Submitted:
21 January 2025
Posted:
27 January 2025
You are already at the latest version
Abstract
Keywords:
Highlights
- Stem CH4 fluxes show large variability within a small spatial range
- Stem CH4 fluxes respond quickly to changing environmental conditions
- Stem CH4 fluxes show time-consistent spatial patterns which are strongly associated with large scale topographic elevation
1. Introduction
- General elevation (i.e., orthometric height of the tree base) explains, at least in part, the spatial variability of stem CH4 emissions.
- The effect of small scale topography (hummocks, hollows) plays a minor role compared to large scale topography (general elevation).
- Stem CH4 emissions increase with an increase in biological activity (i.e., sap flux in trees and soil temperature)
- The water level is the prime factor influencing the temporal variability of stem CH4 flux.
2. Materials and Methods
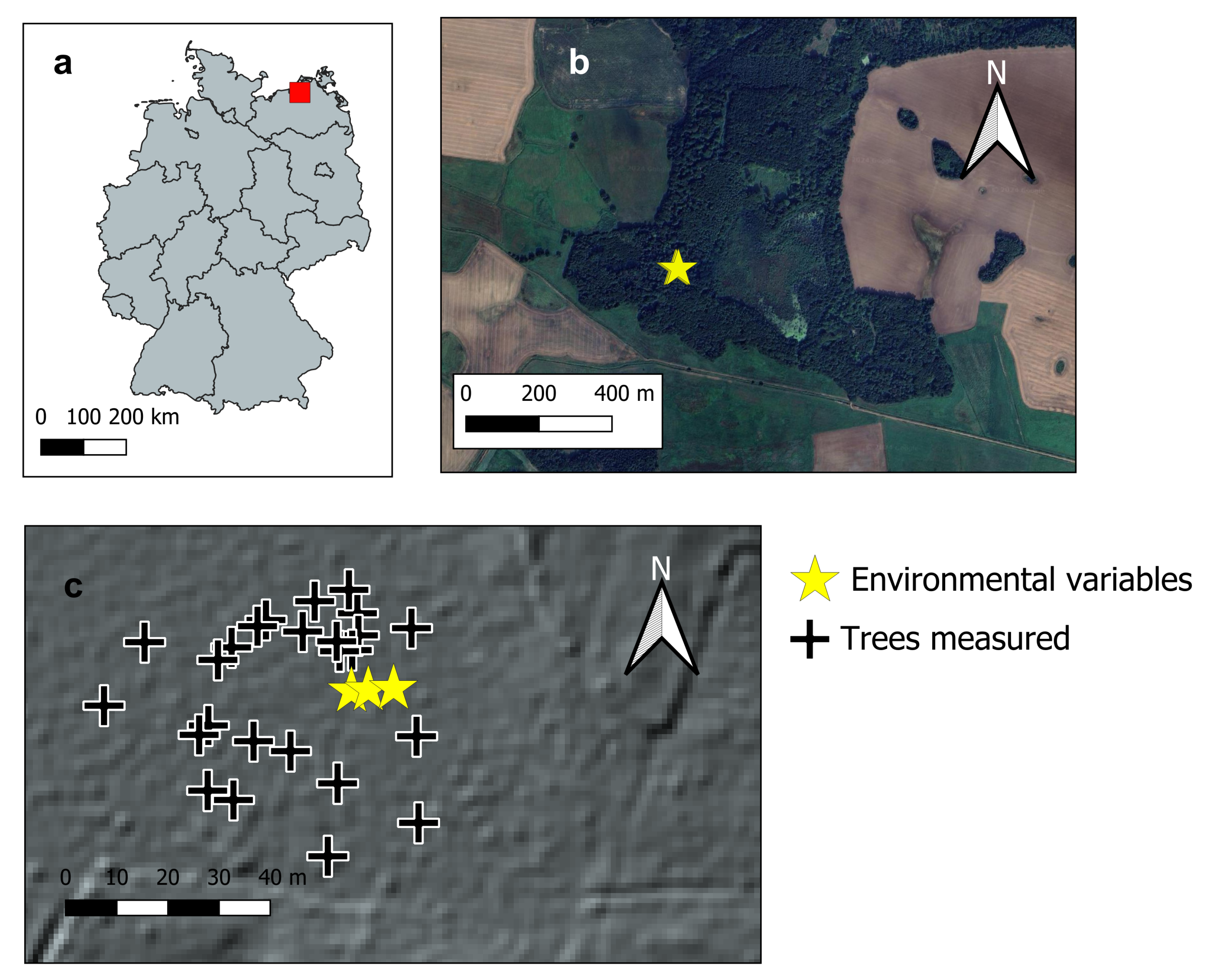
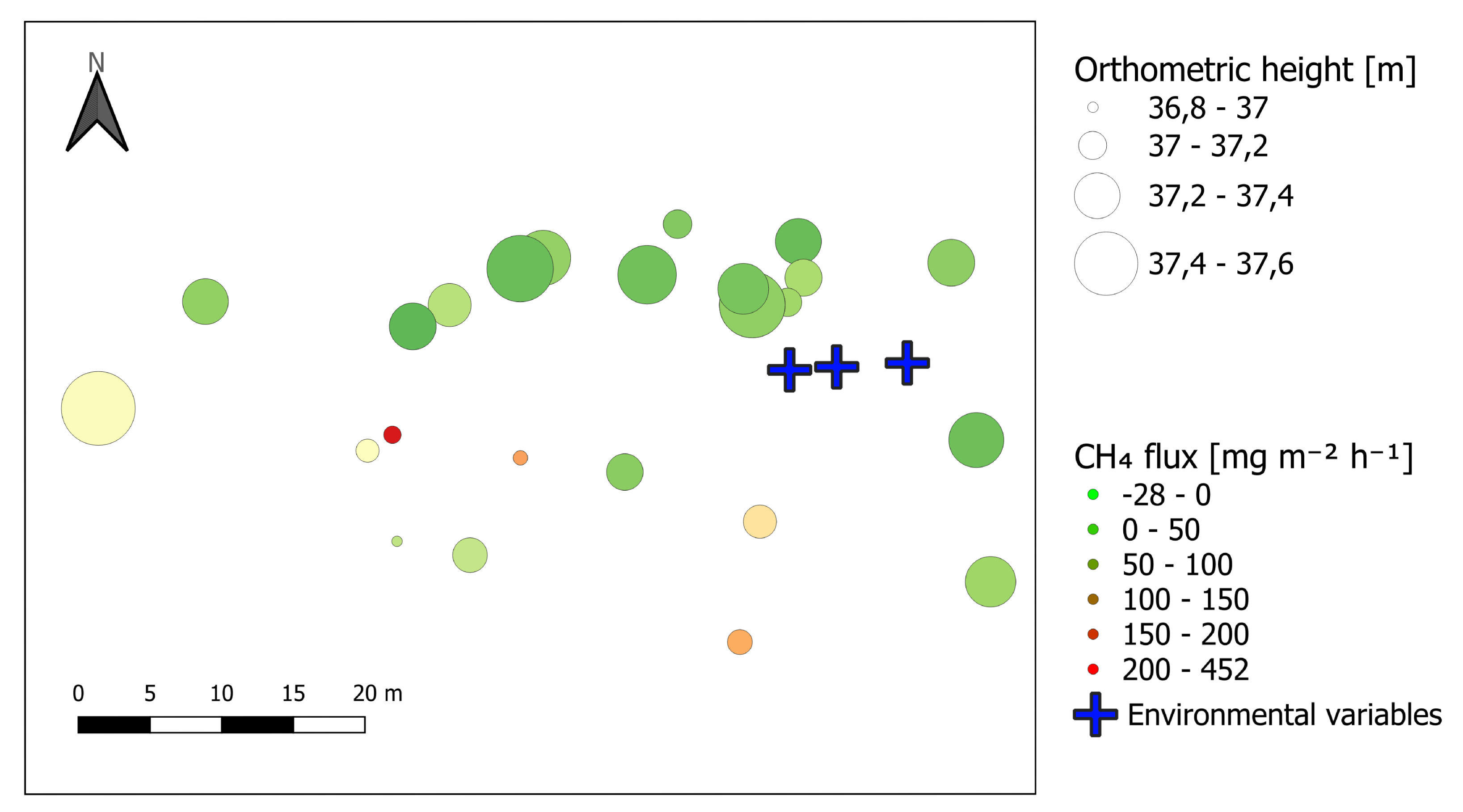
2.1. Study Setup
2.2. Flux Measurements
2.3. Environmental Variables
3. Results
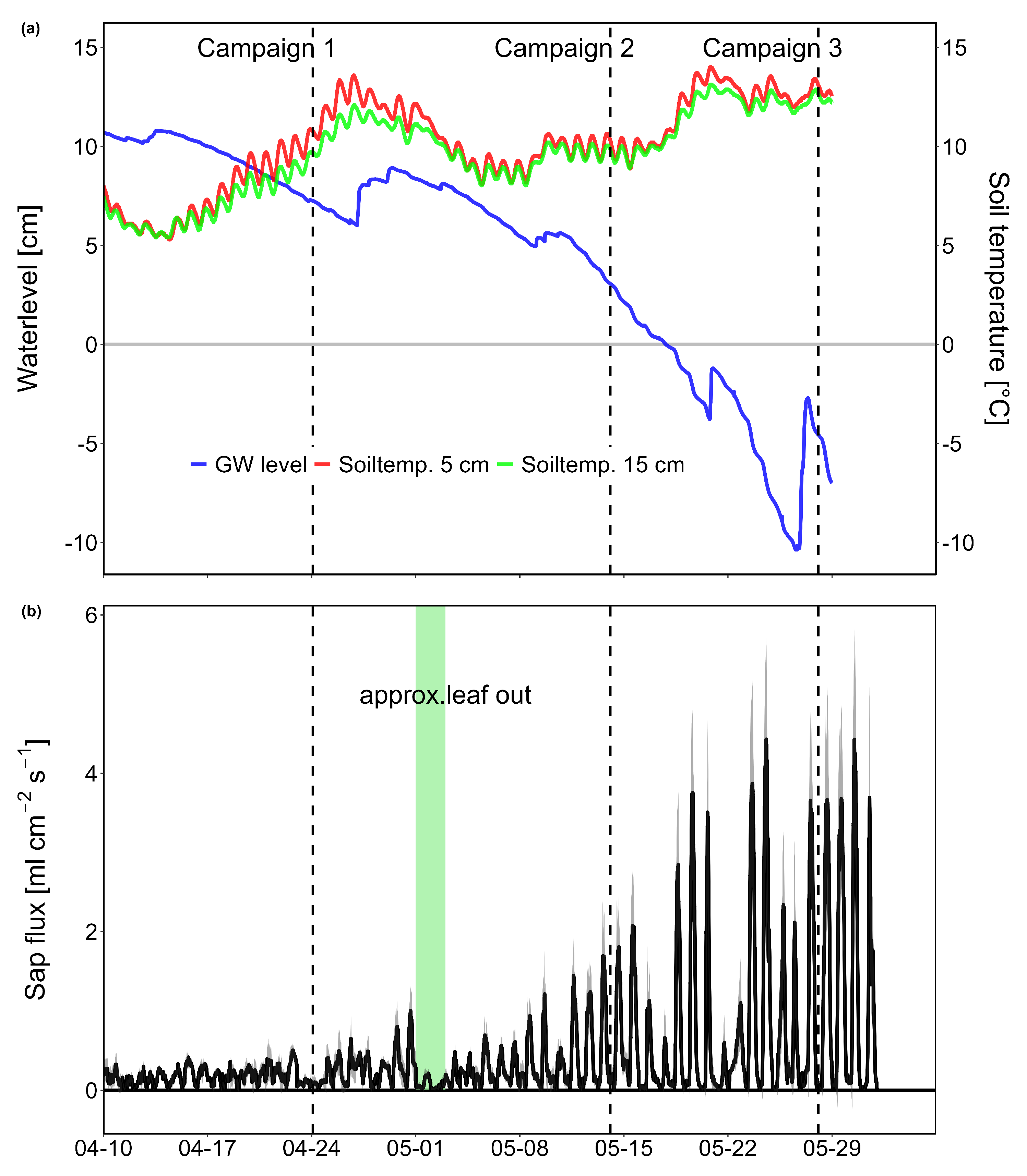
3.1. Environmental Parameters
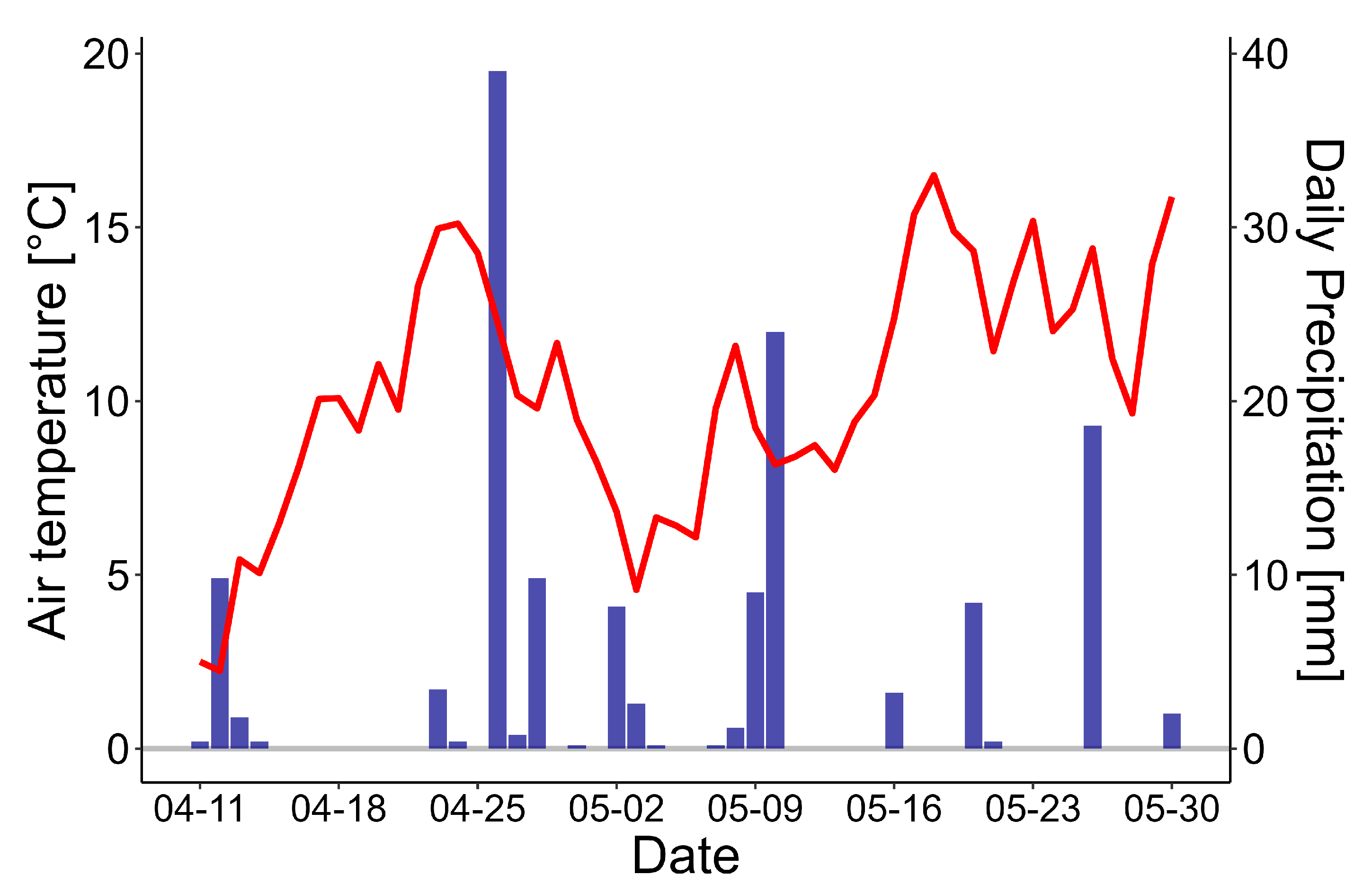
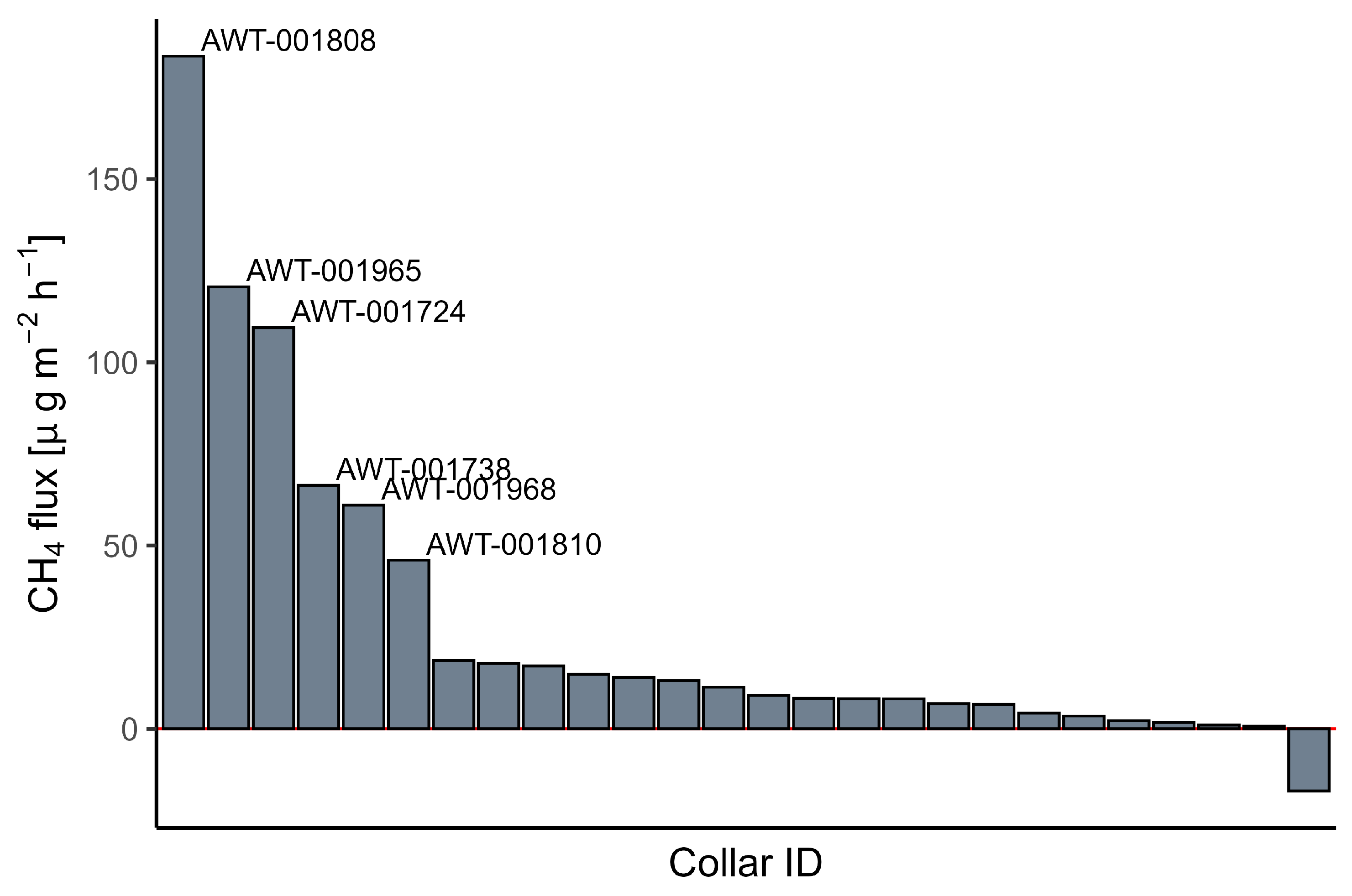
3.2. Tree Stem CH4 and CO2 Exchange
3.3. Topography
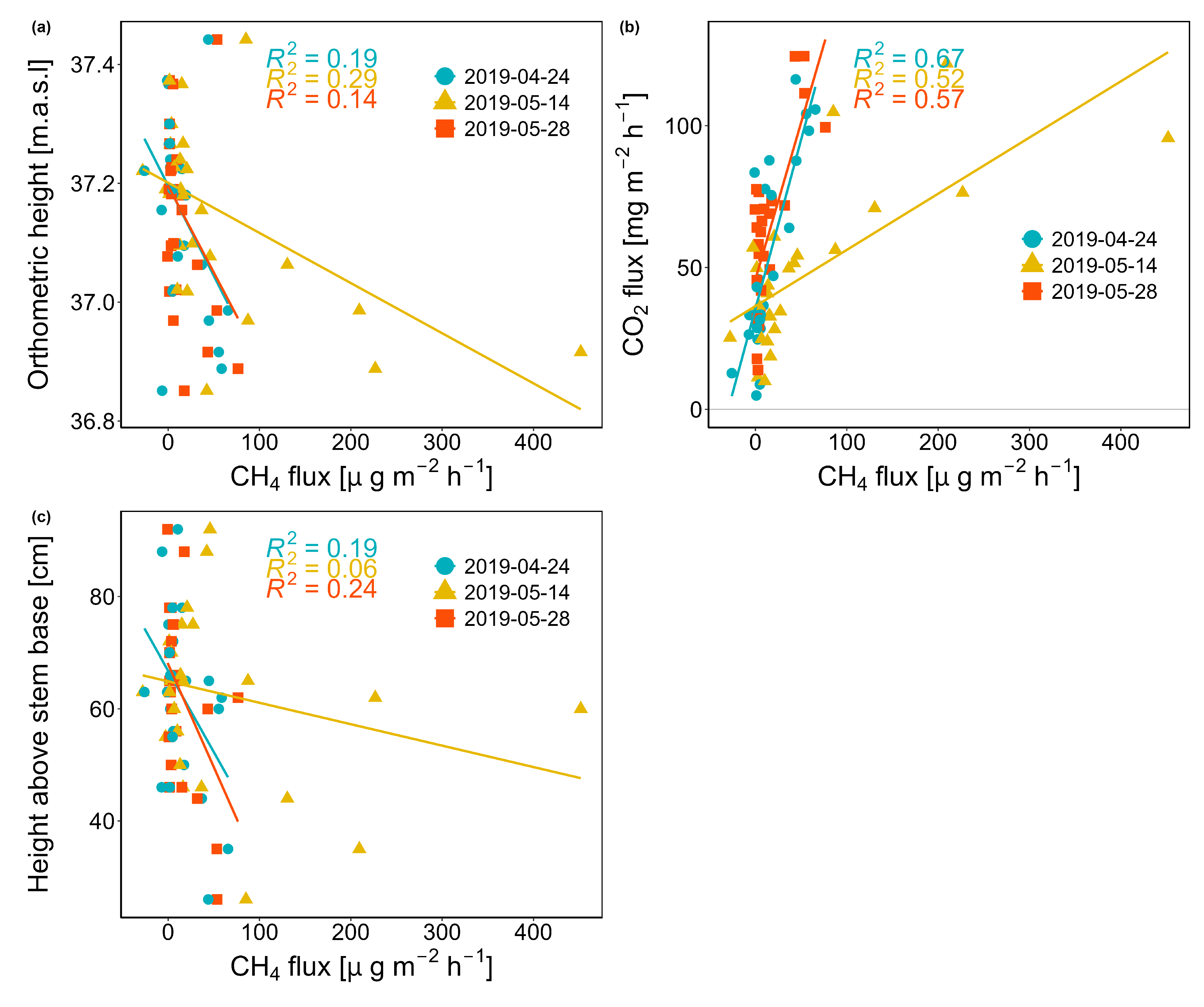
3.4. Controlling Factors of Stem CH4 and CO2 Fluxes
4. Discussion
4.1. Spatial Variation of CH4 Fluxes
4.2. Temporal Variation of CH4 and CO2 Fluxes During Spring
4.3. Variability of CH4 Emissions in Space and Time - What is More Important?
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rusch, H.; Rennenberg, H. Black alder (Alnus Glutinosa (L.) Gaertn.) trees mediate methane and nitrous oxide emission from the soil to the atmosphere. Plant and Soil. [CrossRef]
- Jeffrey, L.C.; Maher, D.T.; Tait, D.R.; Euler, S.; Johnston, S.G. Tree stem methane emissions from subtropical lowland forest ( Melaleuca quinquenervia ) regulated by local and seasonal hydrology. 0123456789. Publisher: Springer International Publishing ISBN: 0123456789. [CrossRef]
- Jeffrey, L.C.; Moras, C.A.; Tait, D.R.; Johnston, S.G.; Call, M.; Sippo, J.Z.; Jeffrey, N.C.; Laicher-Edwards, D.; Maher, D.T. Large Methane Emissions From Tree Stems Complicate the Wetland Methane Budget. 128, e2023JG007679. [CrossRef]
- Petaja, G.; Ancāns, R.; Bārdule, A.; Spalva, G.; Meļņiks, R.N.; Purviņa, D.; Lazdiņš, A. Carbon Dioxide, Methane and Nitrous Oxide Fluxes from Tree Stems in Silver Birch and Black Alder Stands with Drained and Naturally Wet Peat Soils. 14, 521. [CrossRef]
- Pitz, S.L.; Megonigal, J.P.; Chang, C.H.; Szlavecz, K. Methane fluxes from tree stems and soils along a habitat gradient. 137, 307–320. Publisher: Springer International Publishing. [CrossRef]
- Warner, D.L.; Villarreal, S.; McWilliams, K.; Inamdar, S.; Vargas, R. Carbon dioxide and methane fluxes from tree stems, coarse woody debris, and soils in an upland temperate forest. Publisher: Springer US. [CrossRef]
- Barba, J.; Bradford, M.A.; Brewer, P.E.; Bruhn, D.; Covey, K.; van Haren, J.; Megonigal, J.P.; Mikkelsen, T.N.; Pangala, S.R.; Pihlatie, M.; et al. Methane emissions from tree stems: A new frontier in the global carbon cycle. 222, 18–28. ISBN: 0000000220187. [CrossRef]
- Köhn, D.; Günther, A.; Schwabe, I.; Jurasinski, G. Short-lived peaks of stem methane emissions from mature black alder (Alnus glutinosa ( L .) Gaertn .) – Irrelevant for ecosystem methane budgets? pp. 1–12. [CrossRef]
- Pangala, S.R.; Hornibrook, E.R.C.; Gowing, D.J.; Gauci, V. The contribution of trees to ecosystem methane emissions in a temperate forested wetland. 21, 2642–2654. ISBN: 1908655151. [CrossRef]
- Pangala, S.R.; Enrich-Prast, A.; Basso, L.S.; Peixoto, R.B.; Bastviken, D.; Hornibrook, E.R.C.; Gatti, L.V.; Ribeiro, H.; Calazans, L.S.B.; Sakuragui, C.M.; et al. Large emissions from floodplain trees close the Amazon methane budget. pp. 1–7. Publisher: Nature Publishing Group. [CrossRef]
- Terazawa, K.; Yamada, K.; Ohno, Y.; Sakata, T.; Ishizuka, S. Spatial and temporal variability in methane emissions from tree stems of Fraxinus mandshurica in a cool-temperate floodplain forest. 123, 349–362. [CrossRef]
- Schindler, T.; Mander, U.; Machacova, K.; Espenberg, M.; Krasnov, D. Short-term flooding increases CH4 and N2O emissions from trees in a riparian forest soil-stem continuum. 10, 1–10. ISBN: 4159802060. [CrossRef]
- Terazawa, K.; Tokida, T.; Sakata, T.; Yamada, K.; Ishizuka, S. Seasonal and weather-related controls on methane emissions from the stems of mature trees in a cool-temperate forested wetland. 156, 211–230. [CrossRef]
- Sakabe, A.; Takahashi, K.; Azuma, W.; Itoh, M.; Tateishi, M.; Kosugi, Y. Controlling Factors of Seasonal Variation of Stem Methane Emissions From Alnus japonica in a Riparian Wetland of a Temperate Forest. 126, e2021JG006326. [CrossRef]
- Machacova, K.; Bäck, J.; Vanhatalo, A.; Halmeenmäki, E.; Kolari, P.; Mammarella, I.; Pumpanen, J.; Acosta, M.; Urban, O.; Pihlatie, M. Pinus sylvestris as a missing source of nitrous oxide and methane in boreal forest. 6, 2–10. Publisher: Nature Publishing Group ISBN: 2045–2322 (Electronic)\r2045-2322 (Linking). [CrossRef]
- Pangala, S.R.; Moore, S.; Hornibrook, E.R.C.; Gauci, V. Trees are major conduits for methane egress from tropical forested wetlands. 197, 524–531. ISBN: 1469–8137. [CrossRef]
- Sjögersten, S.; Siegenthaler, A.; Lopez, O.R.; Aplin, P.; Turner, B.; Gauci, V. Methane emissions from tree stems in neotropical peatlands. 225, 769–781. [CrossRef]
- Terazawa, K.; Ishizuka, S.; Sakata, T.; Yamada, K.; Takahashi, M. Methane emissions from stems of Fraxinus mandshurica var. japonica trees in a floodplain forest. 39, 2689–2692. ISBN: 0038–0717. [CrossRef]
- Wang, Z.P.; Gu, Q.; Deng, F.D.; Huang, J.H.; Megonigal, J.P.; Yu, Q.; Lü, X.T.; Li, L.H.; Chang, S.; Zhang, Y.H.; et al. Methane emissions from the trunks of living trees on upland soils. 211, 429–439. [CrossRef]
- Covey, K.R.; Wood, S.A.; Warren, R.J.; Lee, X.; Bradford, M.A. Elevated methane concentrations in trees of an upland forest. 39, 1–6. ISBN: 0094–8276. [CrossRef]
- Chanton, J.P.; Whiting, G.J.; Happell, J.D.; Gerard, G. Contrasting rates and diurnal patterns of methane emission from emergent aquatic macrophytes. 46, 111–128. [CrossRef]
- Garnet, K.N.; Megonigal, J.P.; Litchfield, C.; Taylor, G.E. Physiological control of leaf methane emission from wetland plants. 81, 141–155. [CrossRef]
- Krähenmann, S.; A., W.; S, B.; F, I.; A, M. Monthly, daily and hourly grids of 12 commonly used meteorological variables for Germany estimated by the Project TRY Advancement. Technical report, DWD - German Weather Service, 2016. [CrossRef]
- Jurasinski, G.; Ahmad, S.; Anadon-Rosell, A.; Berendt, J.; Beyer, F.; Bill, R.; Blume-Werry, G.; Couwenberg, J.; Günther, A.; Joosten, H.; et al. From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach. 4, 1–27. [CrossRef]
- Livingston, G.; Hutchinson, G. Enclosure-based measurement of trace gas exchange: Applications and sources of error. In Biogenic Trace Gases: Measuring Emissions from Soil and Water; Matson, P.; Harris, R., Eds.; Blackwell Science Ltd., Oxford; pp. 14–53.
- Pavelka, M.; Acosta, M.; Kiese, R.; Altimir, N.; Brümmer, C.; Crill, P.; Darenova, E.; Fuß, R.; Gielen, B.; Graf, A.; et al. Standardisation of chamber technique for CO2, N2O and CH4 fluxes measurements from terrestrial ecosystems. 32, 569–587. [CrossRef]
- Jurasinski, G.; Koebsch, F.; Guenther, A.; Beetz, S. flux: Flux Rate Calculation from Dynamic Closed Chamber Measurements, 2022. R package version 0.3-0.1.
- Siegel, A.F. Robust regression using repeated medians. 69, 242–244. [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, 2021.
- Böhner, J.; Selige, T. Spatial prediction of soil attributes using terrain analysis and climate regionalisation. 115, 13–28.
- Conrad, O.; Bechtel, B.; Bock, M.; Dietrich, H.; Fischer, E.; Gerlitz, L.; Wehberg, J.; Wichmann, V.; Böhner, J. System for Automated Geoscientific Analyses ( SAGA ) v . 2 . 1 . 4. Geoscientific Model Development, pp. 1991–2007. [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer-Verlag New York, 2016.
- Lüdecke, D.; Ben-Shachar, M.S.; Patil, I.; Waggoner, P.; Makowski, D. performance: An R Package for Assessment, Comparison and Testing of Statistical Models. Journal of Open Source Software 2021, 6, 3139. [Google Scholar] [CrossRef]
- Gauci, V.; Gowing, D.J.; Hornibrook, E.R.; Davis, J.M.; Dise, N.B. Woody stem methane emission in mature wetland alder trees. 44, 2157–2160. [CrossRef]
- Machacova, K.; Warlo, H.; Svobodová, K.; Agyei, T.; Uchytilová, T.; Horáček, P.; Lang, F. Methane emission from stems of European beech ( Fagus sylvatica ) offsets as much as half of methane oxidation in soil. 238, 584–597. [CrossRef]
- Dinsmore, K.J.; Skiba, U.M.; Billett, M.F.; Rees, R.M. Effect of water table on greenhouse gas emissions from peatland mesocosms. Plant and Soil 2009, 318, 229–242. [Google Scholar] [CrossRef]
- Aben, R.C.; Barros, N.; Van Donk, E.; Frenken, T.; Hilt, S.; Kazanjian, G.; Lamers, L.P.; Peeters, E.T.; Roelofs, J.G.; De Senerpont Domis, L.N.; et al. Cross continental increase in methane ebullition under climate change. 8, 1–8. Publisher: Springer US ISBN: 4146701701535. [CrossRef]
- Christensen, T.; Panikov, N.; Mastepanov, M.; Joabsson, a.; Stewart, a.; {\textbackslash}{\textbackslash}"Oquist, M.; Sommerkorn, M.; Reynaud, S.; Svensson, B. Biotic controls on CO 2 and CH 4 exchange in wetlands–a closed environment study. 64, 337–354. [CrossRef]
- Koebsch, F.; Jurasinski, G.; Koch, M.; Hofmann, J.; Glatzel, S. Controls for multi-scale temporal variation in ecosystem methane exchange during the growing season of a permanently inundated fen. 204, 94–105. Publisher: Elsevier B.V. ISBN: 0168–1923. [CrossRef]
- Yvon-Durocher, G.; Allen, A.P.; Bastviken, D.; Conrad, R.; Gudasz, C.; St-Pierre, A.; Thanh-Duc, N.; del Giorgio, P.A. Methane fluxes show consistent temperature dependence across microbial to ecosystem scales. 507, 488–491. Publisher: Nature Publishing Group ISBN: 0028–0836; 1476–4687. [CrossRef]
- Zeikus, J.G.; Ward, J.C. Methane Formation in Living Trees - A Microbial Origin - Zeikus.pdf. 184, 1181–1183.
- Keppler, F.; Hamilton, J.T.G.; Braß, M.; Röckmann, T. Methane emissions from terrestrial plants under aerobic conditions. 439, 187–191. ISBN: 00280836. [CrossRef]
- Teskey, R.O.; McGuire, M.A. CO2 transported in xylem sap affects CO2 efflux from Liquidambar styraciflua and Platanus occidentalis stems, and contributes to observed wound respiration phenomena. 19, 357–362. ISBN: 0931–1890. [CrossRef]
- Takahashi, K.; Sakabe, A.; Azuma, W.A.; Itoh, M.; Imai, T.; Matsumura, Y.; Tateishi, M.; Kosugi, Y. Insights into the mechanism of diurnal variations in methane emission from the stem surfaces of Alnus japonica. 235, 1757–1766. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




