Submitted:
09 January 2025
Posted:
10 January 2025
You are already at the latest version
Abstract
Keywords:
 |
1. Introduction
2. Experimental Procedures
2.1. Sorption Materials
2.1.1. Preparation of Hydrotalcite
2.1.2. Preparation of Natural Zeolite Clinoptilolite
2.1.3. Preparation of Synthetic Zeolite
2.1.4. Preparation of Sludge
2.2. Preparation of Synthetic and Real Wastewater
2.3. Adsorption Test
2.4. Analytical Methods
3. Results and Discussion
3.1. Characterization of Sorption Materials
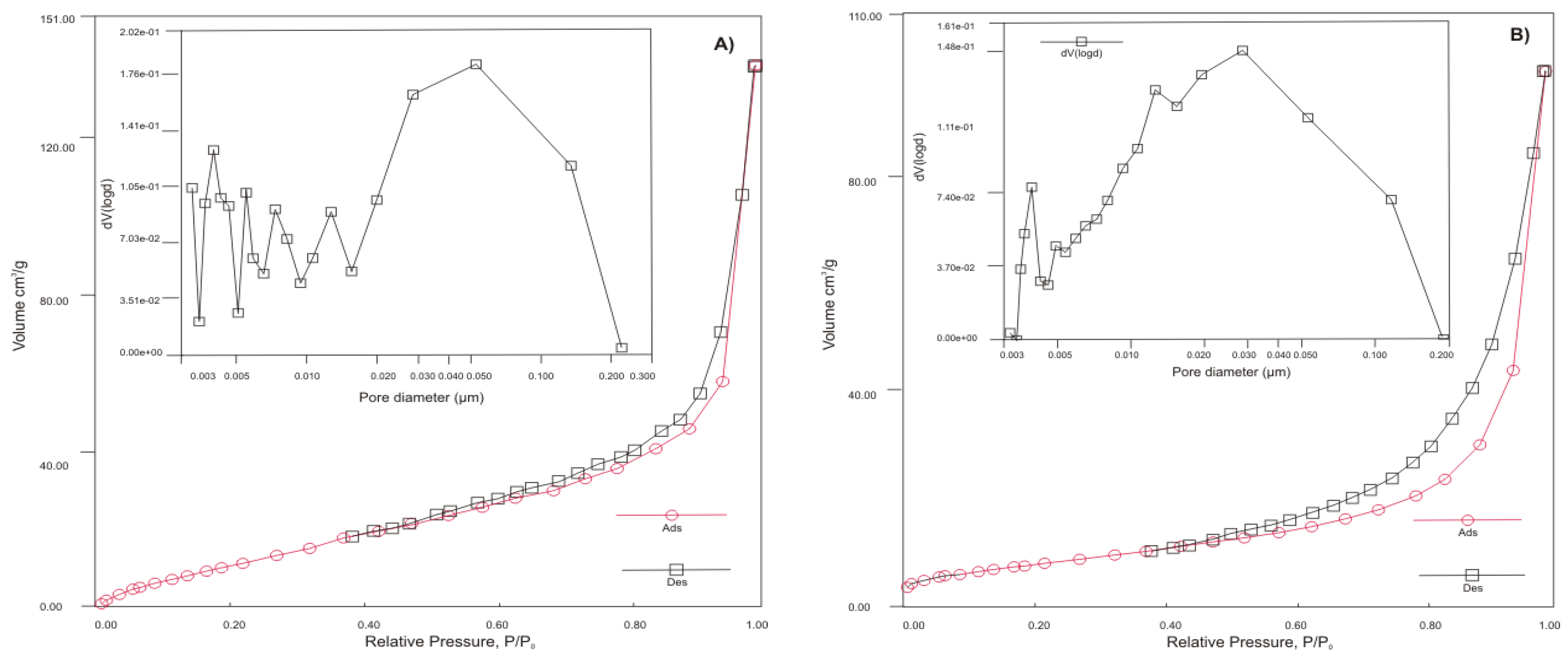
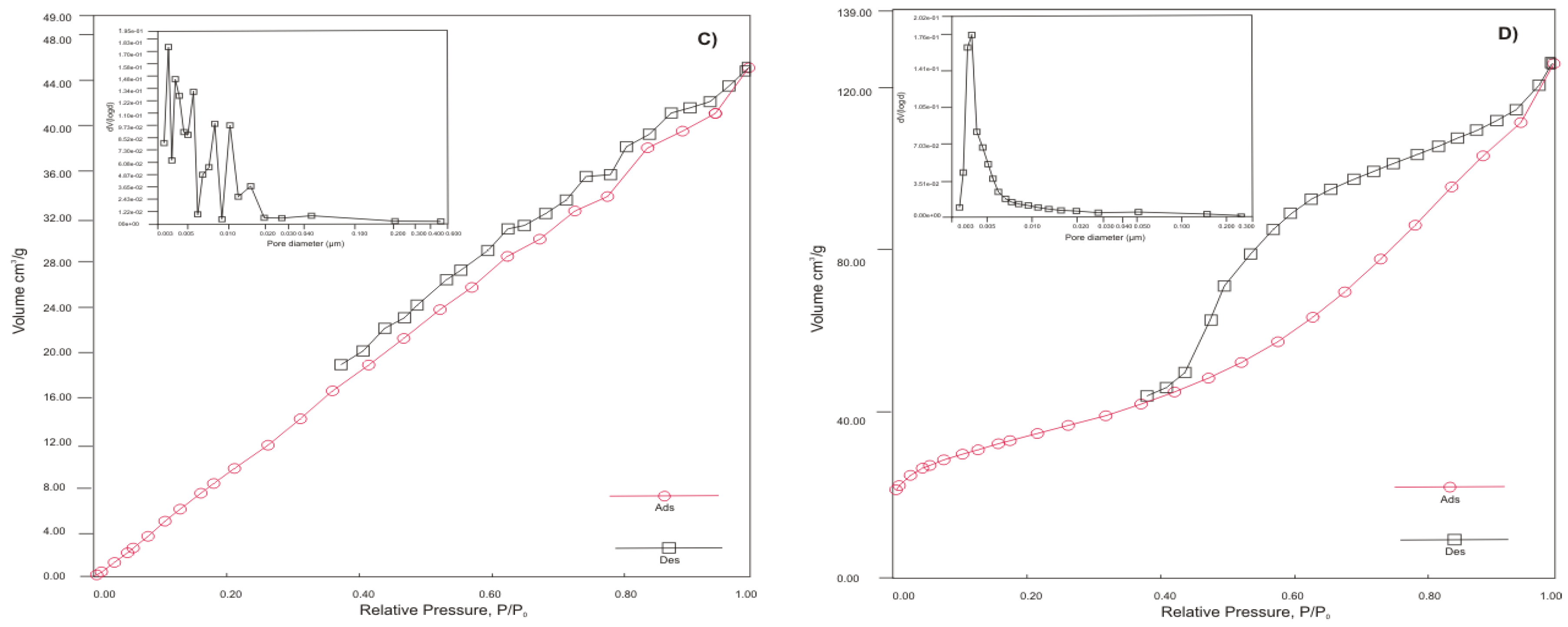
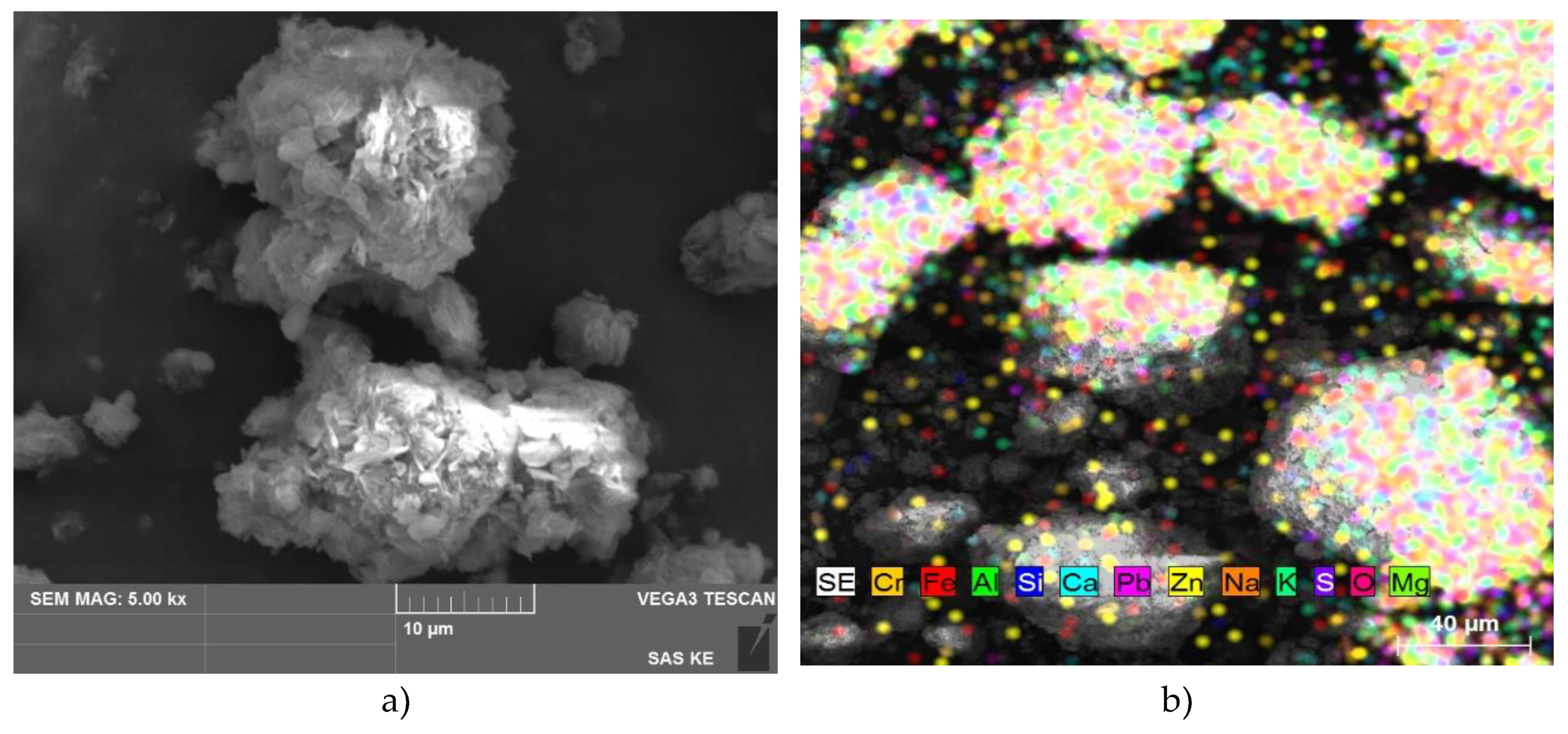
3.1.1. Chemical Composition, Surface Area, Pore Size Distribution and Morphology
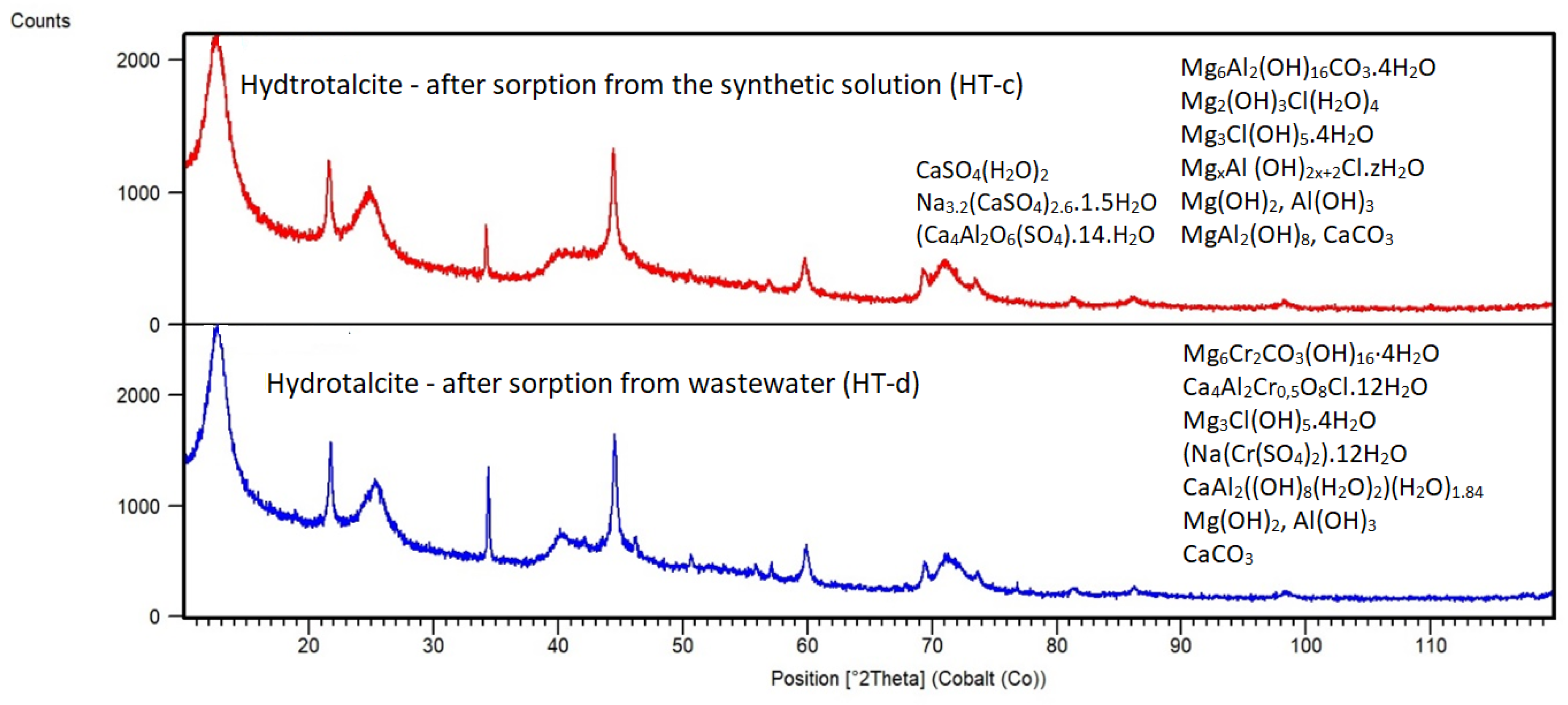
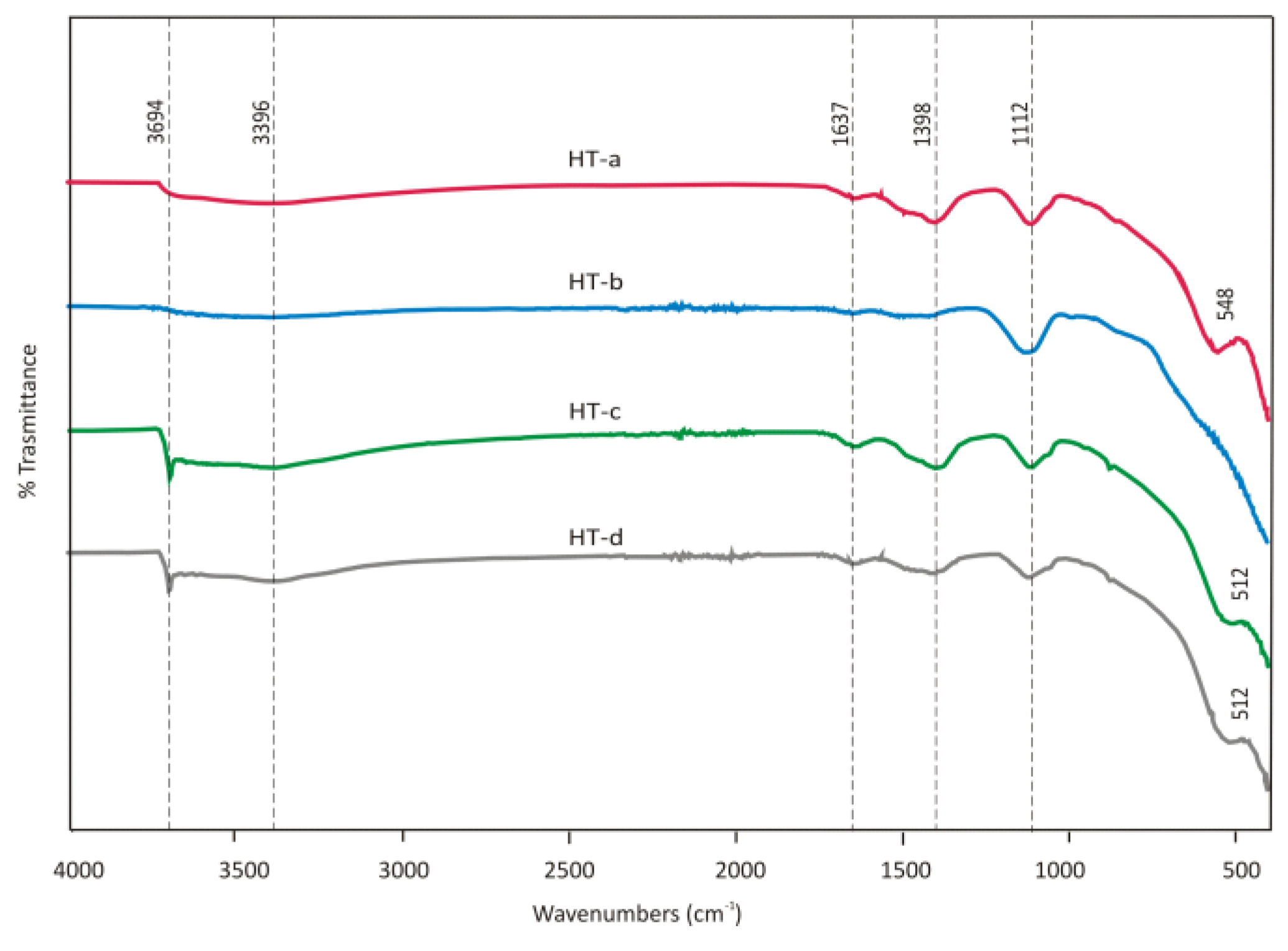
3.1.2. Mineralogical Composition of Sorption Materials
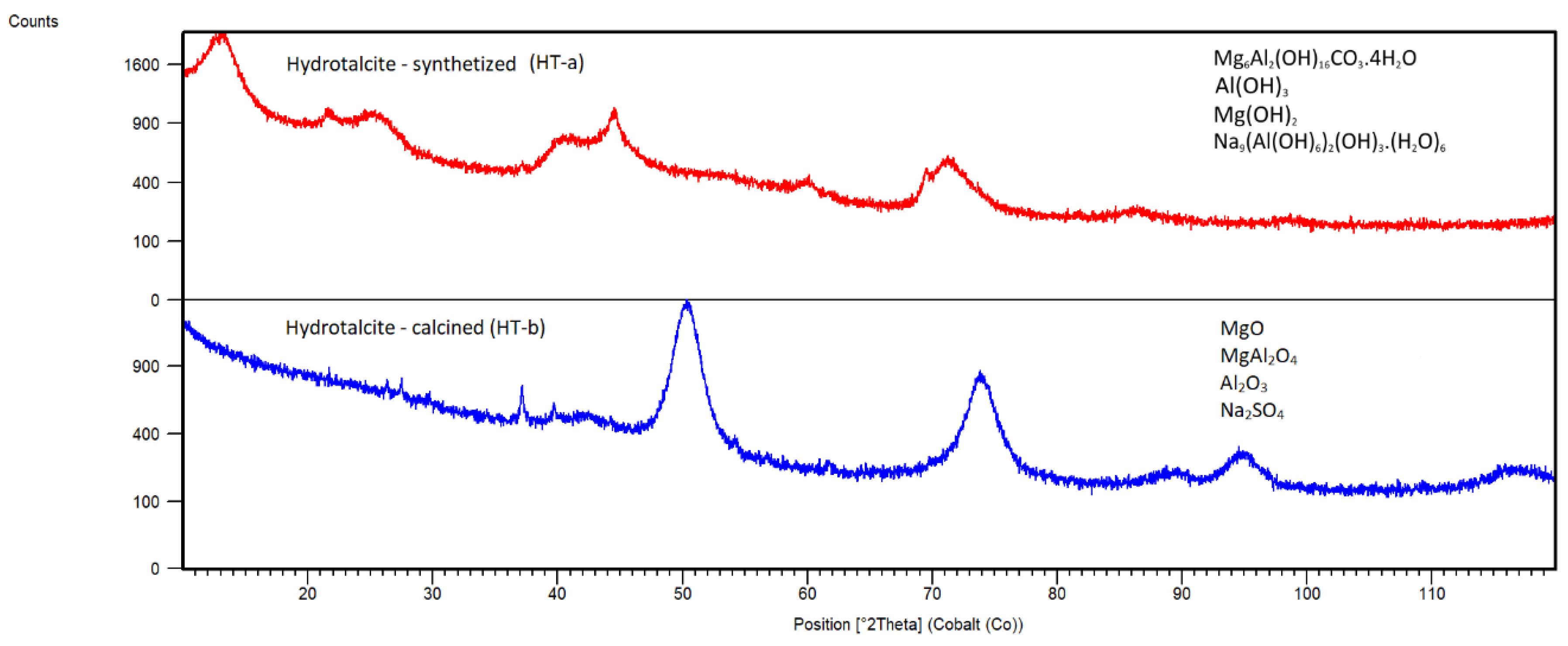
Hydrotalcite
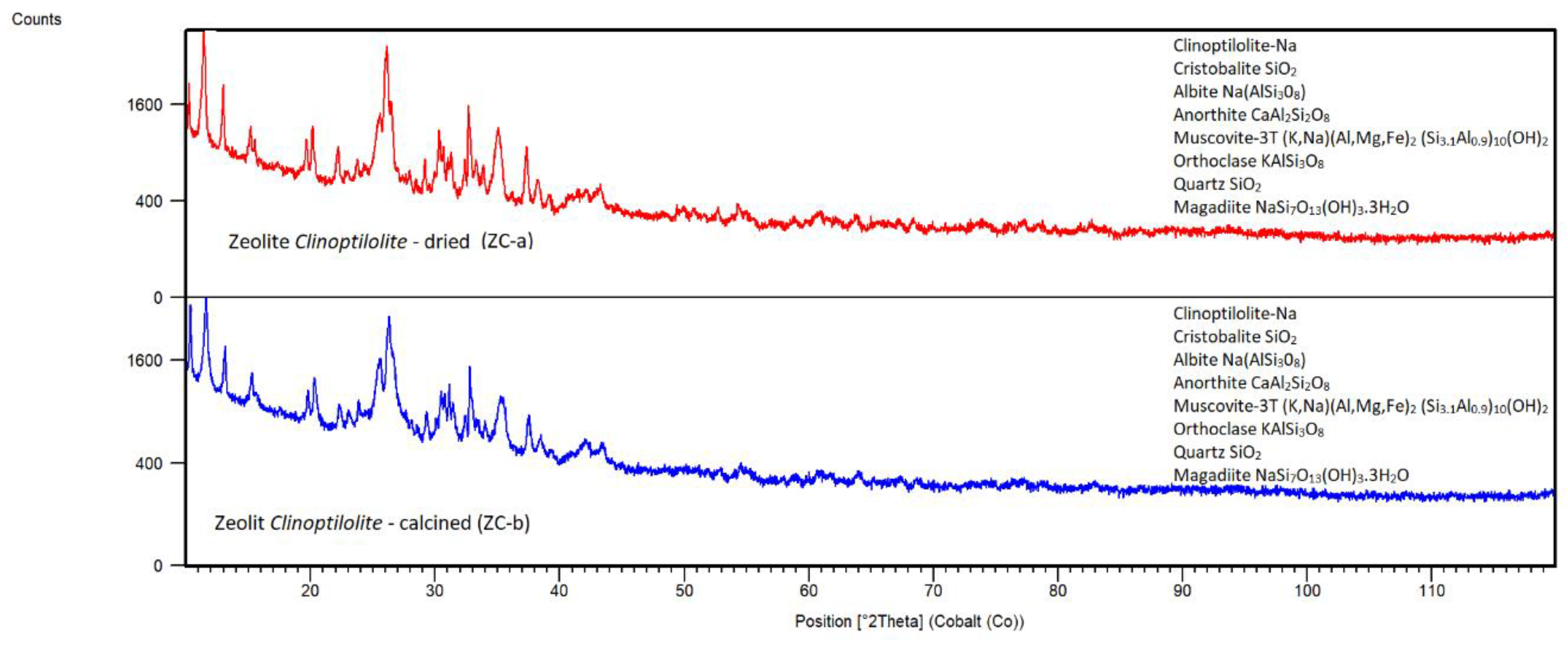
Natural Zeolite Clinoptilolite
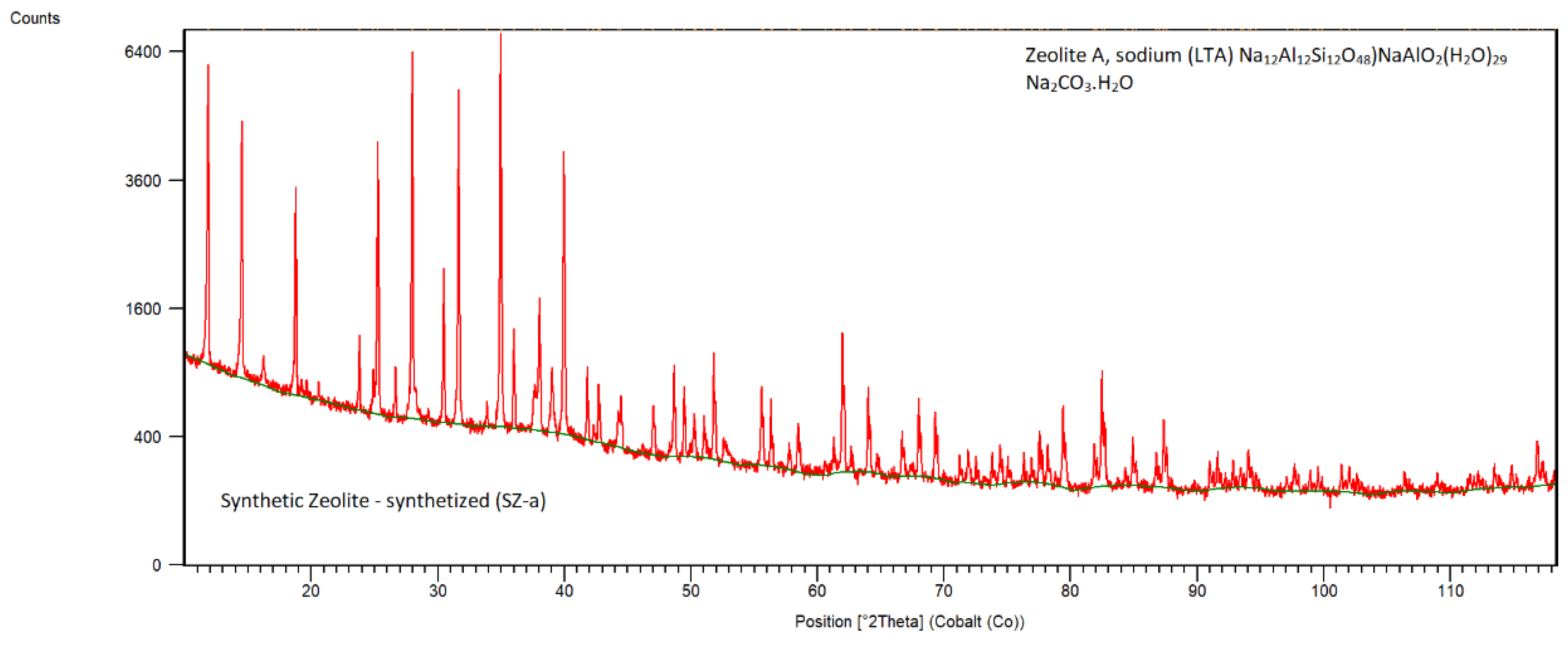
Synthetic Zeolite
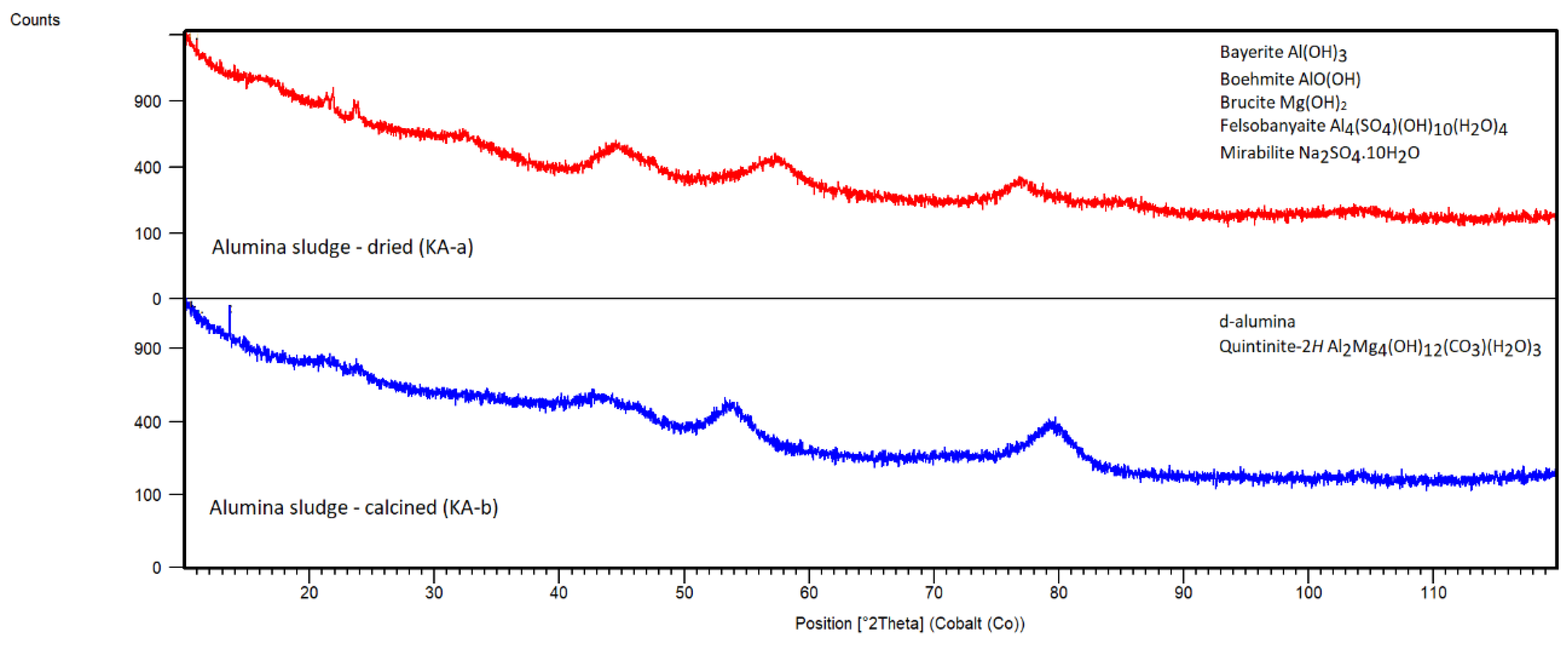
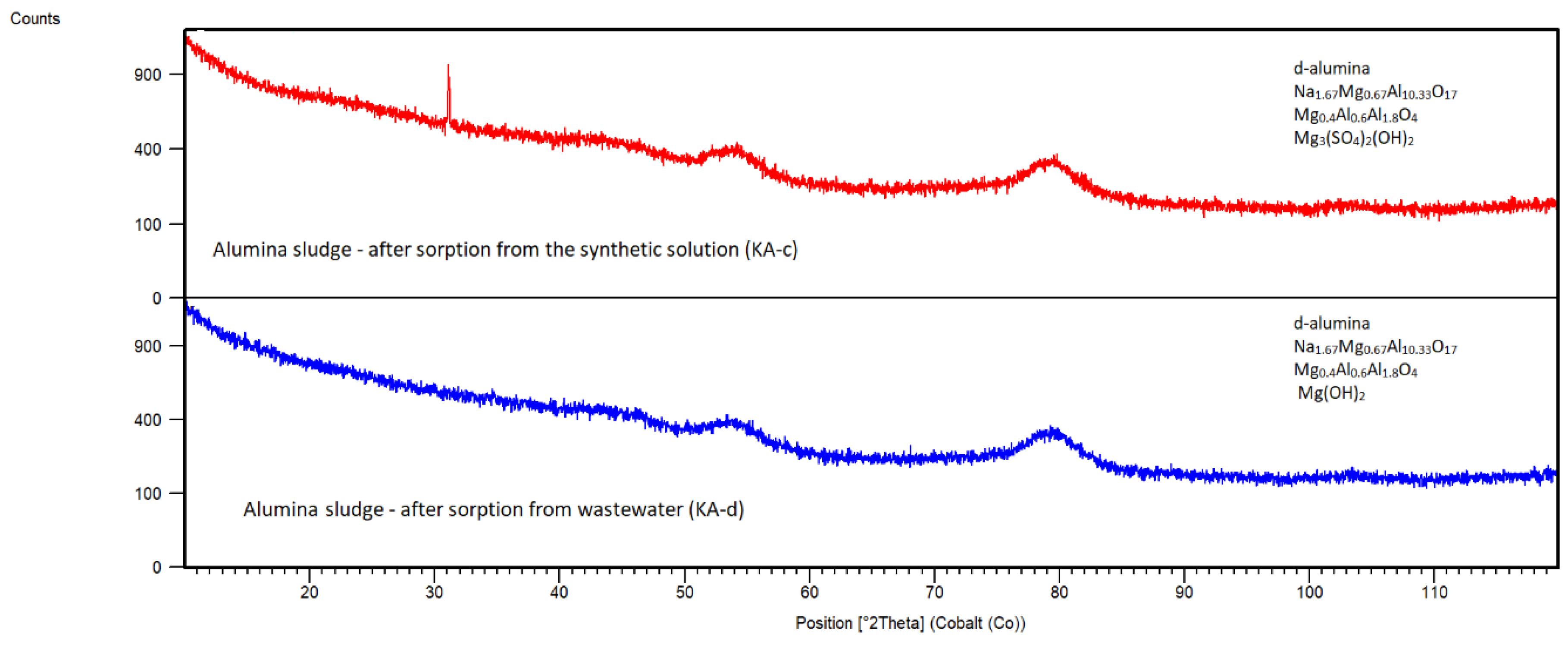
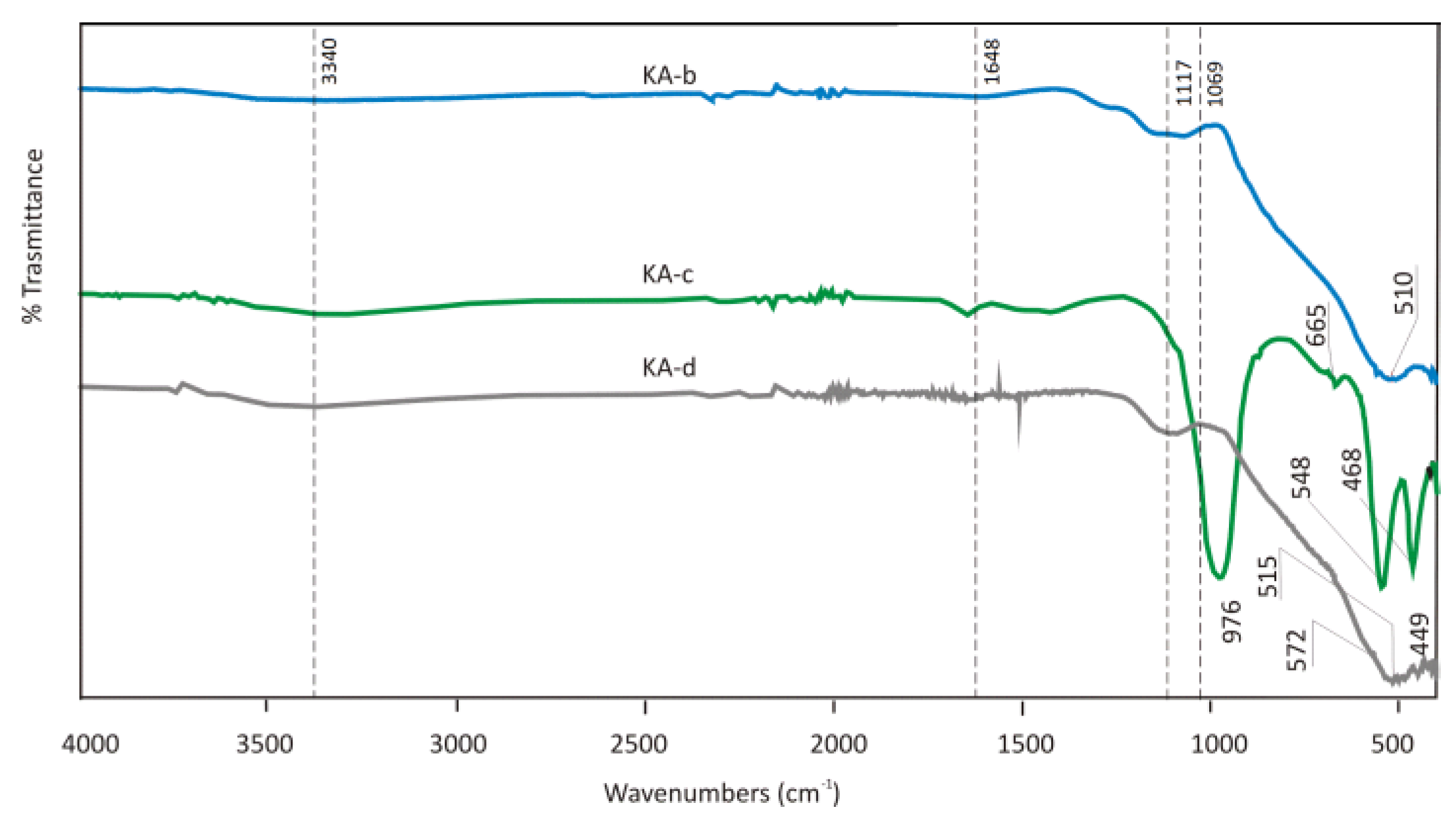
Alumina Sludge
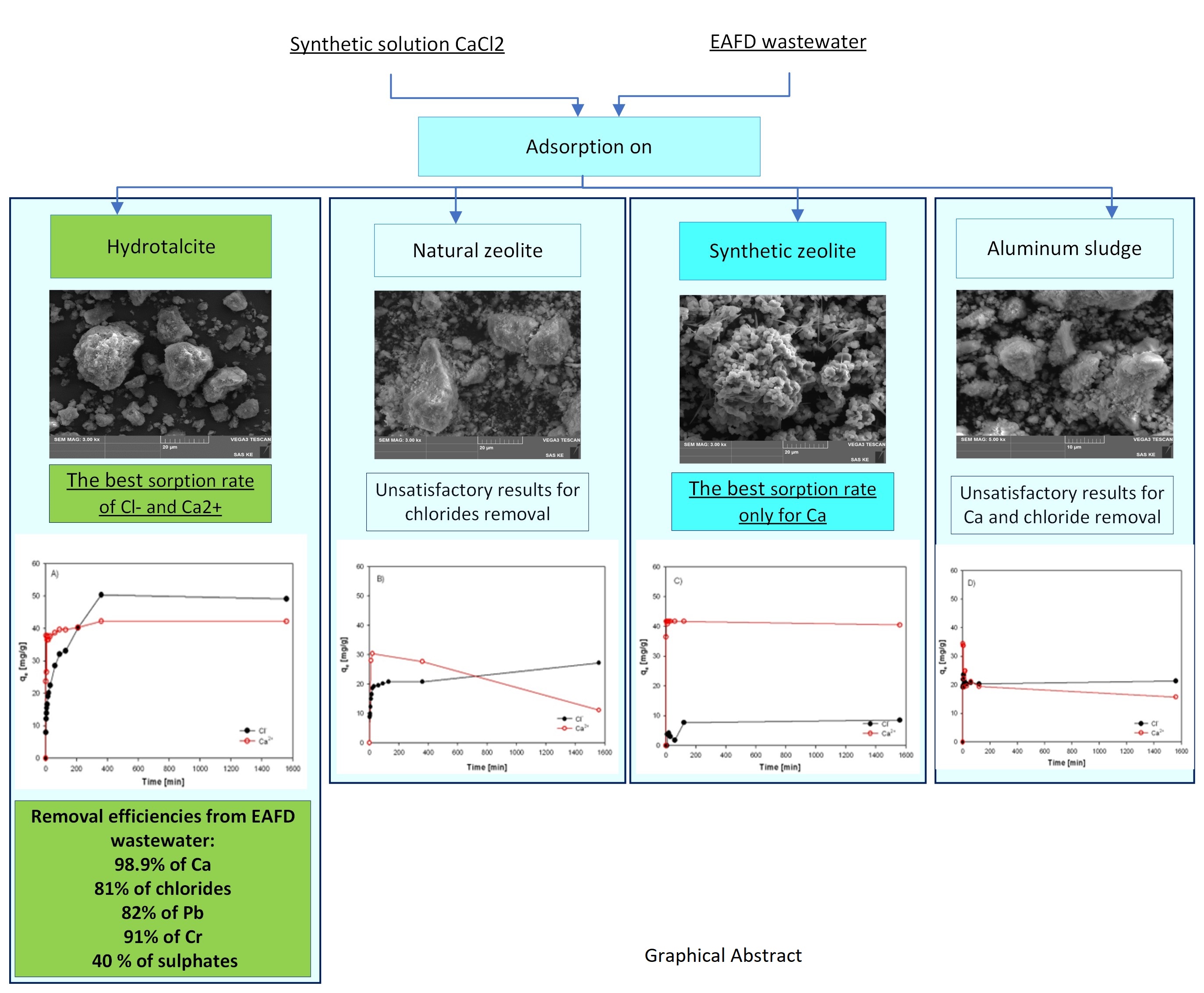
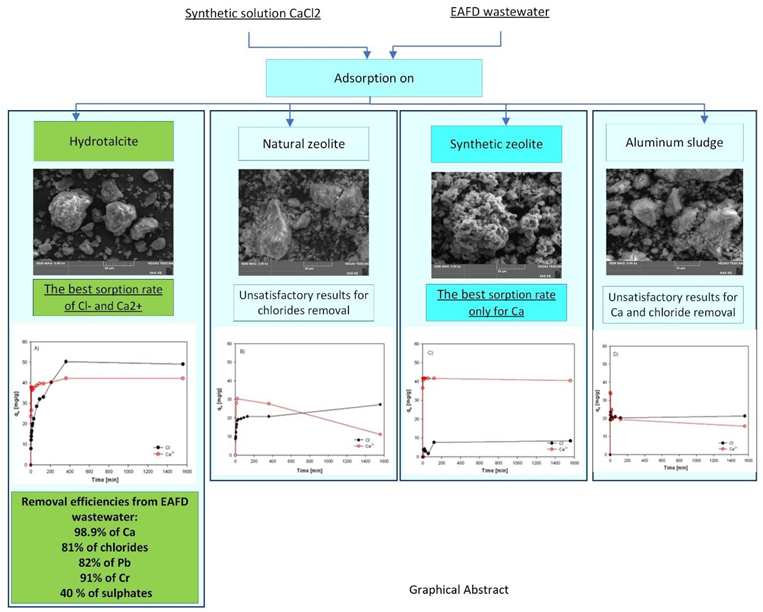
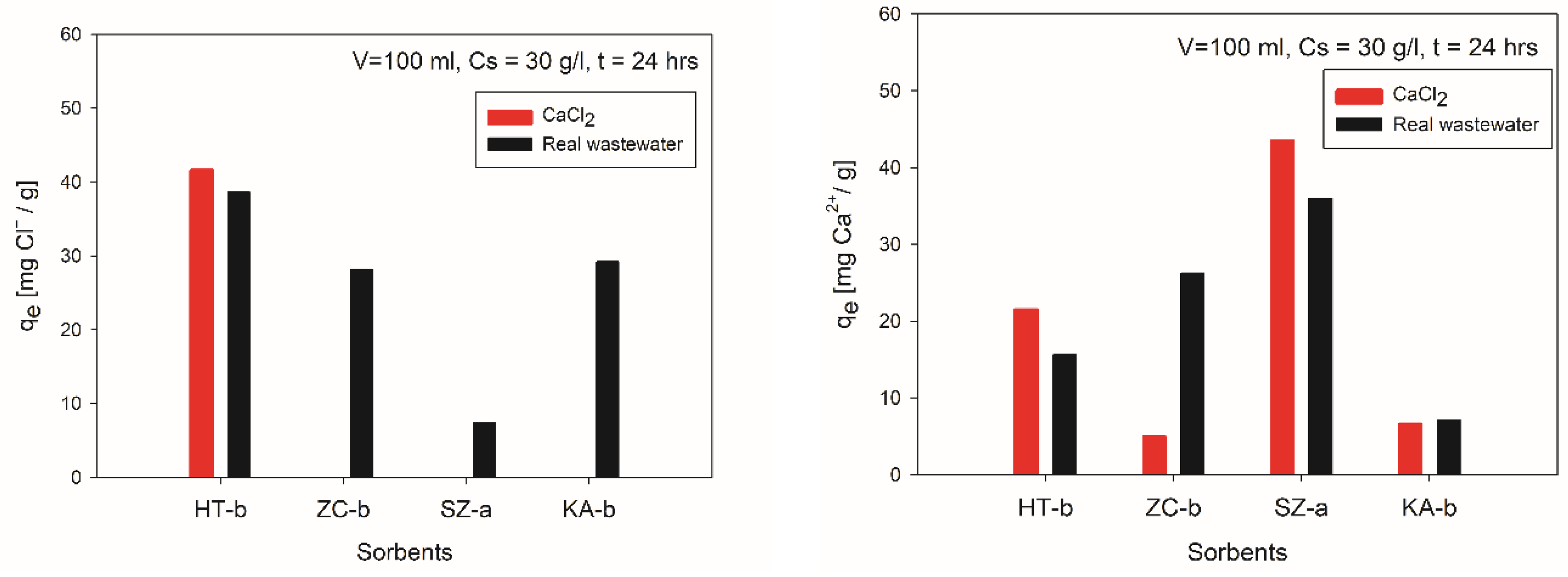
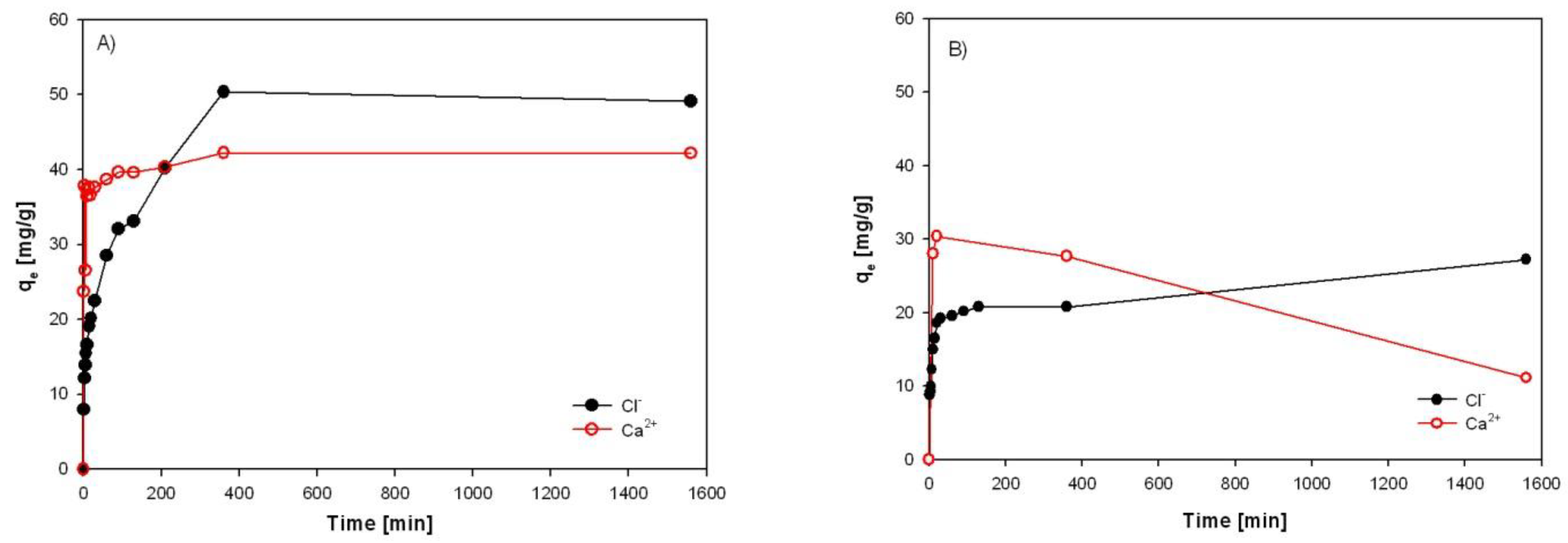
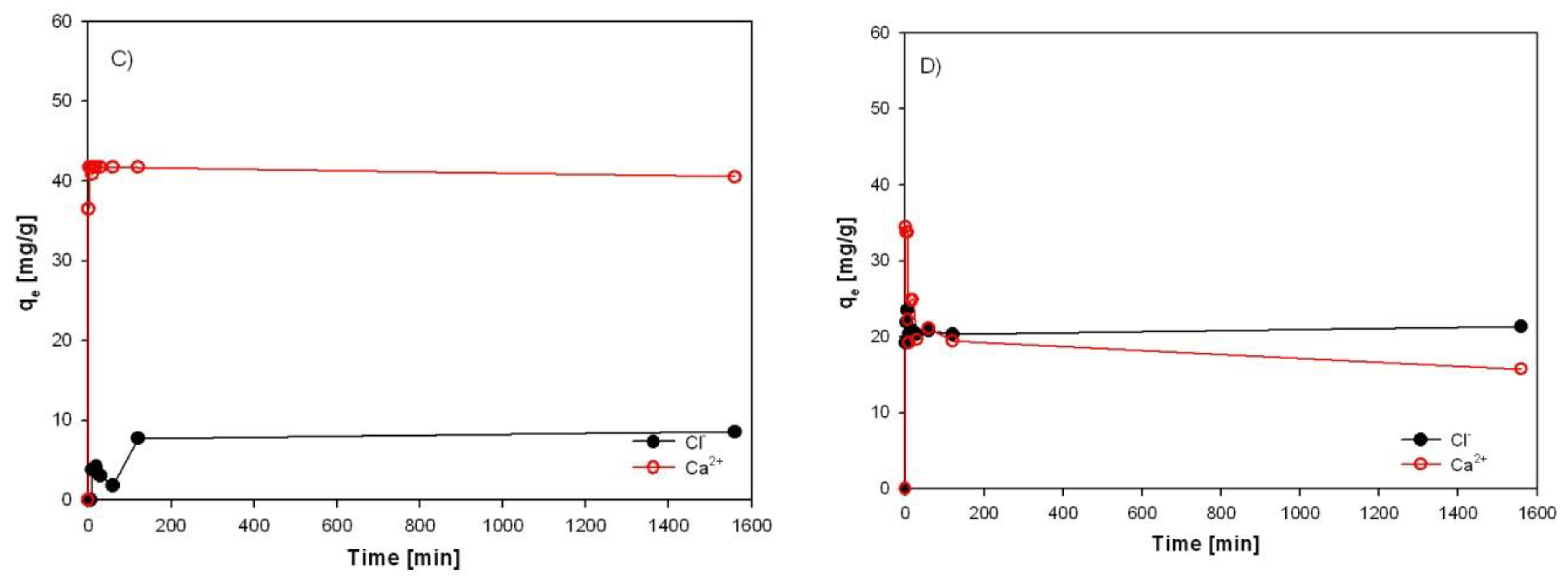
3.2. Results of Chlorides and Calcium Adsorption from Synthetic Solution and EAFD Wastewater
3.3. Evaluation of Sorption Results
3.3.1. Characterization of Adsorbents after Sorption
Hydrotalcite
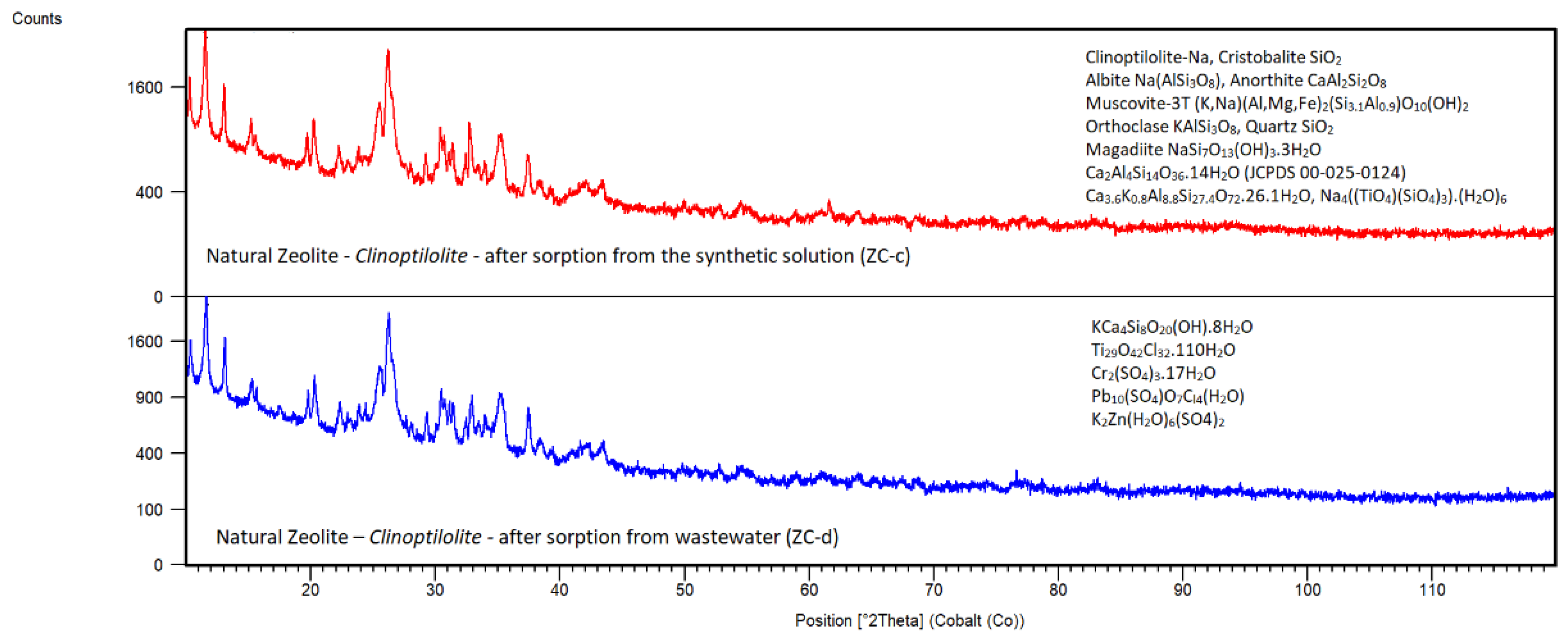
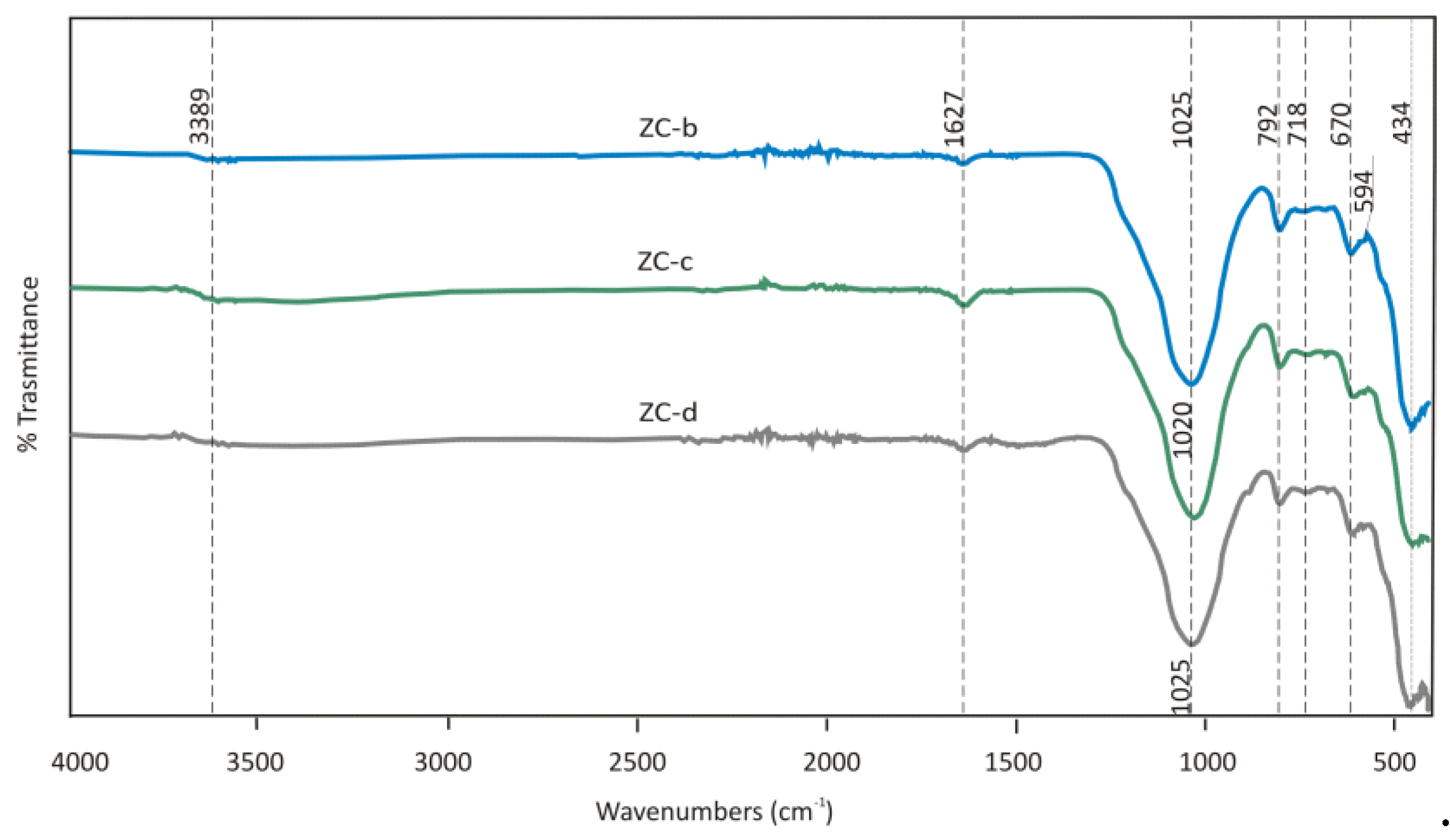
Natural Zeolite – Clinoptilolite
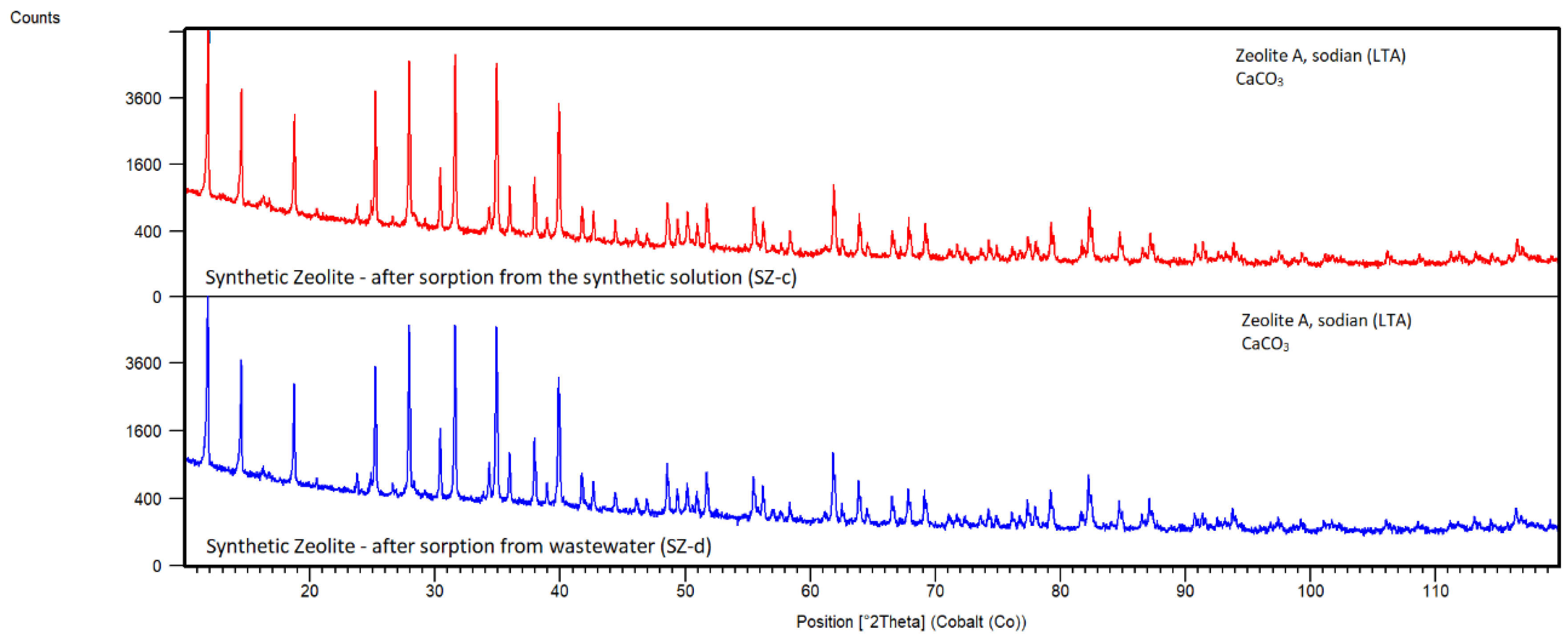
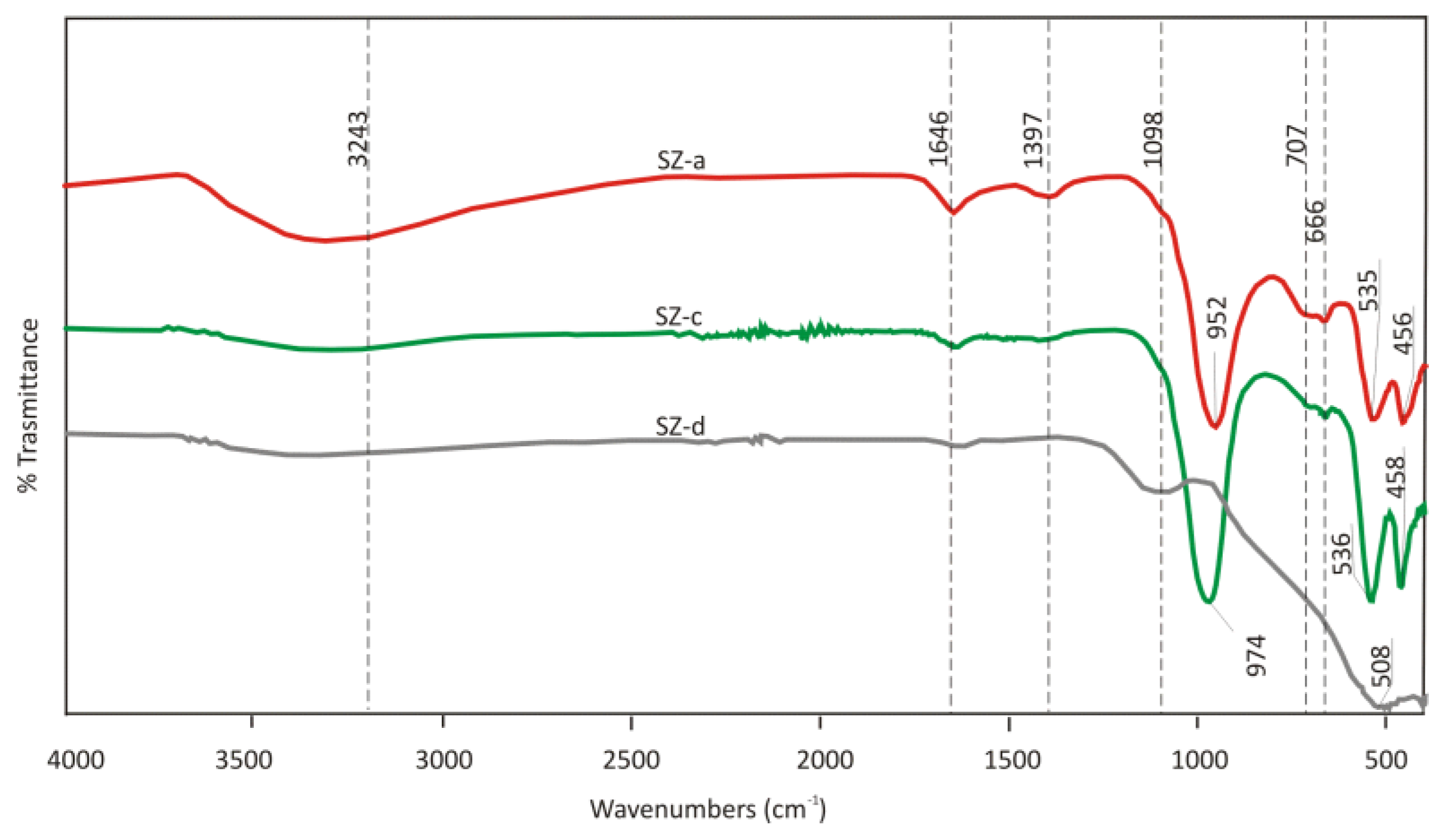
Synthetic Zeolite
Alumina Sludge
3.4. Investigation of Sorption Rate of Individual Sorbents from EAFD Wastewater
3.5. The sorption of Other Species from EAFD Wastewater by Hydrotalcite Sorbent
4. Conclusions
- It was approved the possibility of simultaneous removal of chloride anions and calcium cations by sorption from EAFD wastewater (generated during EAFD washing) by laboratory prepared and calcined Hydrotalcite sorbent (500°C). The highest sorption efficiencies of 98.9% for Ca2+ and 81% for Cl- ions were achieved by using calcined HT adsorbent after 24 hours for EAFD wastewater. The results implies that the sorption process of calcium is faster and preferential to chlorides. This could be caused by two different mechanisms which occur during the simultaneous sorption of Ca2+ and Cl- ions. Calcium is probably adsorbed into the interlayers where it is physically bonded to the carboxyl groups. The rate of chloride sorption is slower than calcium due to the outer parts of the Hydrotalcite sorbent, which has positively charged layers.
- Besides Ca and Cl-, calcined HT also removes Pb, Cr (82-91%), and partially sulphates (40%).
- Adsorption with Hydrotalcite (HT-b) requires a long contact time (6 h), which indicates a diffusion-controlled process and the anion-exchange mechanism.
- Other tested sorption materials of ZC-b, KA-b, SZ-a have shown unsatisfactory results for chloride removal. However, a sample of SZ-a exhibits a very high sorption capacity for calcium and could be used in wastewater purification except in very acid solutions. These results support knowledge and enhance efforts to recycle and reutilize waste materials to produce high-quality raw materials.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Steel Association: Crude Steel Production. Available online: https://worldsteel.org/data/steel-data-viewer/ (accessed on 28 June 2024).
- Sustainable Water Treatment, Delivered Anywhere. Available online: https://www.fluencecorp.com/ (accessed on 28 June 2024).
- Colla, V.; Branca, T. A.; Rosito, F.; Lucca, C.; Vivas, B.P.; Delmiro, V.M. Sustainable reverse osmosis application for wastewater treatment in the steel industry. J. Clean. Prod. 2016, 130, 103–115. [Google Scholar] [CrossRef]
- Water management in the steel industry. Available online: https://worldsteel.org/wp-content/uploads/ Water-management-in-the-steel-industry.pdf (accessed on 28 June 2024).
- Kirkelund, G. M.; Magro, C.; Guedes, P.; Jensen, P. E.; Ribeiro, A. B.; Ottosen, L. M. Electrodialytic removal of heavy metals and chloride from municipal solid waste incineration fly ash and air pollution control residue in suspension - test of a new two compartment experimental cell. Electrochimica Acta 2015, 181, 73–81. [Google Scholar] [CrossRef]
- Ozugbe, E.O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Kameda, T.; Miyano, Y.; Yoshioka, Y.; Uchida, M.; Okuwaki, A. New treatment methods for waste water containing chloride ion using magnesium-aluminium oxide. Chem. Lett. 2000, 29, 1136–1137. [Google Scholar] [CrossRef]
- Wang, J.; Qu, D.; Tie, M.; Ren, H.; Peng, X.; Luan, Z. Effect of coagulation pretreatment on membrane distillation process for desalination of recirculating cooling water. Sep. Purif. Technol. 2008, 64, 108–115. [Google Scholar] [CrossRef]
- Shehata, N.; Egirani, D.; Olabi, A.G.; Inayat, A.; Abdelkareem, M.A.; Chae, K.-J.; Sayed, E.T. Membrane-based water and wastewater treatment technologies: Issues, current trends, challenges, and role in achieving sustainable development goals, and circular economy. Chemosphere 2023, 320, 137993. [Google Scholar] [CrossRef]
- Dron, J.; Dodi, A. Comparison of adsorption equilibrium models for the study of Cl−, NO3− and SO42− removal from aqueous solutions by an anion exchange resin. J. Hazard. Mat. 2011, 190, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Hilal, N.; Kochkodan, V.; Al Abdulgader, H.; Mandale, S.; Al-Jlil S., A. A combined ion exchange-nanofiltration process for water desalination: I. sulphate-chloride ion-exchange in saline solutions. Desalination 2015, 363, 44–50. [Google Scholar] [CrossRef]
- Darracq, G.; Baron, J.; Joyeux, M. Kinetic and isotherm studies on perchlorate sorption by ion-exchange resins in drinking water treatment. J. Water Process Eng., 2014, 3, 123–131. [Google Scholar] [CrossRef]
- Iakovleva, E.; Mäkilä, E.; Salonen, J.; Sitarz, M.; Sillanpää, M. Industrial products and wastes as adsorbents for sulphate and chloride removal from synthetic alkaline solution and mine process water, Chem. Eng. J. 2015, 259, 364–371. [Google Scholar]
- Dahmani, K.; Kherroub, D.E.; Boucherdoud, A.; Bestani, B. Removal of Ca(II) and Mg(II) hardness by ion exchange resins and soda ash for seawater pretreatment to reduce scale formation in evaporators multi-stage flash desalination. Desalin. Water. Treat. 2021, 221, 23–30. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Hailu, Y.; Tilahun, E.; Brhane, A.; Resky, H.; Sahu, O. Ion exchanges process for calcium, magnesium and total hardness from ground water with natural zeolite. Groundwater for Sustainable Development 2019, 8, 457–467. [Google Scholar] [CrossRef]
- Kameda, T.; Yoshioka, T.; Mitsuhashi, T.; Uchida, M.; Okuwaki, A. The simultaneous removal of calcium and chloride ions from calcium chloride solution using magnesium–aluminum oxide. Water Res. 2023, 37, 4045–4050. [Google Scholar] [CrossRef]
- Kameda, T.; Yoshioka, T.; Hoshi, T.; Uchida, M.; Okuwaki, A. The removal of chloride from solutions with various cations using magnesium–aluminum oxide, Sep. Purif. Technol. 2005, 42, 25–29. [Google Scholar] [CrossRef]
- Hamidi, R.; Kazemi, P. Kinetics and mechanism of sorption of chloride ion from sodium carbonate manufacturing wastewater by Mg–Al oxide. Desal. Water Treat. 2014, 54, 332–341. [Google Scholar] [CrossRef]
- Miyata, S. Anion-exchange properties of hydrotalcite-like compounds. Clays Clay Miner. 1983, 4, 4–311. [Google Scholar] [CrossRef]
- Jun-yi, L.; Zheng-song, Q.; Yang, L. Nano-pore structure characterization of shales using gas adsorption and mercury intrusion techniques. Journal of Chemical and Pharmaceutical Research 2014, 6, 850–857. [Google Scholar]
- Shoumkova, A.; Stoyanova, V. Zeolites formation by hydrothermal alkali activation of coal fly ash from thermal power station “Maritsa 3”, Bulgaria. Fuel 2013, 103, 533–541. [Google Scholar] [CrossRef]
- About zeolite. Available online: https://worldsteel.org/data/steel-data-viewer/ (accessed on 1 July 2024).
- Yang, S.; Lach-hab, M.; Vaisman, I.I.; Blaisten-Barojas, E.; Li, X.; Karen, V.L. Framework-Type Determination for Zeolite Structures in the Inorganic Crystal Structure Database. J. Phys. Chem. Ref. Data 2010, 39, 3. [Google Scholar] [CrossRef]
- Frost, R.L.; Martens, W.; Ding, Z.; Kloprogge, J.T.; Johnson, T.E. The role of water in synthesized hydrotalcites of formula MgxZn6-xCr2(OH)16(CO3).4H2O and NixCo6-xCr2(OH)16(CO3).4H2O – an infrared spectroscopic study. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2003, 59, 291–302. [Google Scholar] [CrossRef]
- Morimoto, K.; Anraku, S.; Hoshino, J.; Yoneda, T.; Sato, T. Surface complexation reactions of inorganic anions on hydrotalcite-like compounds. J. Colloid. Interface Sci. 2012, 384, 99–104. [Google Scholar] [CrossRef]
- Polatoglu, I.; Cakicioglu-Ozkan, F. Aqueous interactions of zeolitic material in acidic and basic solutions. Microporous and Mesoporous Mater. 2010, 132, 219–225. [Google Scholar] [CrossRef]
- Ye, N.; Yang, J.; Liang, S.; Hu, Y.; Hu, J.; Xiao, B.; Huang, O. Synthesis and strength optimization of one-part geopolymer based on red mud. Constr. Build. Mater. 2016, 111, 317–325. [Google Scholar] [CrossRef]
- Demortier, A.; Gobeltz, N.; Lelieur, J.P.; Duhayon, C. Infrared evidence for the formation of an intermediate compound during the synthesis of zeolite Na–A from metakaolin. Intern. J. Inorg. Mat. 1999, 1, 129–134. [Google Scholar] [CrossRef]







| Elements | Cl- | SO42- | Ca2+ | Pb2+ | Cr | Na+ | K+ |
|---|---|---|---|---|---|---|---|
| Ci [mg/l] | 1995.2 | 1200 | 1084 | 6.16 | 1.69 | 525 | 694 |
| Sample | Metal content [wt.%] | ||
|---|---|---|---|
| - | Mg | Al | |
| HT-b | - | 24.42 | 3.72 |
| Si | Ca | Al | |
| ZC-b | 13.32 | 3.38 | 2.01 |
| SZ-a | 34.7 | 0.13 | 11.5 |
| KA-b | 0.27 | 0.04 | 30.265 |
| Sample | BET surface area [m2 g-1] | Single point pore volume [cm3 g-1] | Pore diameter Dv (d) [nm] |
|---|---|---|---|
| HT-b | 48.532 | 0.209 | 3 |
| ZC-b | 27.060 | 0.157 | 4 |
| SZ-a | 78.267 | 0.064 | 3 |
| KA-b | 118.890 | 0.200 | 4 |
| Adsorption material | Synthetic | EAFD wastewater | |
|---|---|---|---|
| Initial pH value (pHi) | - | 6.87 | 12.78 |
| Final pH value (pHf) | HT-b | 11.77 | 12.54 |
| ZC-b | 8.53 | 12.24 | |
| SZ-a | 12.34 | 12.87 | |
| KA-b | 4.41 | 7.78 |
| Ion | SO42- | K+ | Na+ | Pb2+ | Cr3+ |
|---|---|---|---|---|---|
| Initial concentration [mg/l] | 1910 | 838 | 560 | 5.52 | 4.52 |
| Final concentration [mg/l] | 1150 | x | x | 0.97 | 0.4 |
| Sorption capacity [mg/g] | 25.33 | - | - | 0.15 | 0.14 |
| Removal efficiency [%] | 39.79 | - | - | 82.43 | 91.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




