1.0. Background
The treatment of prostate cancer (PCa) has undergone an epochal positive evolution in recent years. In particular, this evolution has led to the main concept of anticipation and intensification of therapy in different phases of PCa progression. The point of failure of this process, which is accentuated due to the numerous therapeutic options that these patients have at their disposal, is the identification of valid prognostic indicators of therapy response and the construction of a tailored precision therapy on each specific patient.
The etiology of prostate cancer (PCa) is driven by a multifaceted interplay of genetic and environmental factors. At the molecular level, one key aspect of this process involves genes responsible for DNA damage repair (DDR), which play a central role in maintaining genomic integrity [
1]. These DDR genes facilitate the detection and repair of DNA damage, such as single-strand breaks (SSBs) and double-strand breaks (DSBs), that can accumulate during cell division. Their function is critical in preventing the accumulation of mutations that could lead to tumorigenesis, as well as inducing apoptosis in cells harboring potentially harmful mutations [
2,
3,
4,
5,
6].
Mutations in DDR genes, particularly those in pathogenic variants (PVs), compromise the efficiency of DNA repair mechanisms, thereby heightening the risk of genomic instability. When DDR systems are impaired, the cell becomes more susceptible to the accumulation of DNA damage, which can result in the uncontrolled growth of cancerous cells. In the context of DDR, the Poly-ADP-ribose polymerase (PARP) system is primarily involved in detecting and repairing SSBs, while the homologous recombination repair (HRR) pathway plays a dominant role in the repair of DSBs [
2,
3,
4,
5,
6].
BRCA1 and BRCA2, the most well-known genes within the HRR pathway, are essential for maintaining genomic stability, and their pathogenic variants (PVs) are strongly associated with an elevated risk of a spectrum of cancers, including breast, ovarian, prostate, pancreatic, and colon cancers. Such insights may offer opportunities for more precise therapeutic strategies, including the potential for targeted therapies that exploit DDR deficiencies in cancer cells [
1,
4].
BRCA mutations, in several neoplasms have been associated with:
- -
Short metastatic-free survival
- -
Short cancer specific survival
- -
Prediction of response to PARP inhibitors
HRR mutations in tumor cells induce a dependency for single strand break reparation through the PARP system, providing the rationale to develop PARP inhibitors. PARP inhibitors block PARP system causing accumulation of DNA damages in DDR defective tumor cells [
6,
7].
The importance of the BRCA1 / 2 test in patients with prostate cancer is justified by the following reasons:
It has been shown that the PVs of the BRCA genes, whether of a germinal or somatic nature, represent a predictive biomarker of greater sensitivity to treatment with inhibitors of the enzyme PARP, in patients with hormone-resistant metastatic prostate cancer.
For patients with PCa and positive for BRCA germline PVs, appropriate surveillance programs should be indicated to manage the risk of developing second cancers associated with BRCA PVs.
In general, the detection of PVs of the BCA genes in PCa patients can help to define the patient’s prognosis and the choice of the therapeutic procedure.
To date, the analysis of PV of HRR genes is recommended in patients with metastatic prostate cancer and linked to a precision therapy with PARP inhibitors. Therapy with PARP inhibitors is recommended for metastatic castration resistant PCa (mCRPC) patients as second line in monotherapy or first line in association with androgen receptor pathway inhibitors (ARPI) [
2,
6].
2.0. DDR Analysis: Prognostic Role in Different Stages of PCA
2.1. Incidence
The incidence of PVs in various DDR genes among PCa patients has been reported in multiple studies and registries. Among men with metastatic PCa (mPCa), the frequency of germline PVs in DDR genes ranges from 11% to 33%, a significantly higher rate compared to non-metastatic PCa (nmPCa), where the incidence is between 5% and 10% [
8,
9,
10,
11]. Notably, BRCA2 mutations are the most common, occurring in 5.3% of cases, followed by mutations in CHEK2 (1.9%), ATM (1.6%), BRCA1 (0.9%), and RAD51 (0.4%). In metastatic castration-resistant prostate cancer (mCRPC), the incidence of somatic DDR mutations is 24%, with BRCA2 mutations accounting for 13%, ATM mutations at 7.3%, MSH2 mutations at 2.0%, and BRCA1 mutations at 0.3% [
8,
9].
The International Stand Up to Cancer (SU2C) Registry, which analyzed mCRPC cases, reported a similar distribution of somatic DDR mutations, with an overall incidence of 23%. Among these, BRCA2 mutations were the most frequent at 13%, followed by ATM mutations at 7.3%, MSH2 mutations at 2%, and BRCA1/RAD51 mutations at 0.3% [
11].
In the Profound study [
8], which examined 2,792 biopsies from mCRPC patients, DDR pathogenic variants were identified in 28% of cases, with no significant difference in frequency between the primary tumors (27%) and metastatic sites (32%). These findings raise important questions regarding the clinical implications of HRR gene mutations in the management of prostate cancer. A similar pattern was observed in a study of 316 Chinese patients (including both mPCa and high-risk localized PCa cases), where 9.8% of patients had pathogenic mutations in DDR genes (95% confidence interval [CI]: 6.5-13%). Among these, 6.3% carried BRCA2 mutations, 0.63% BRCA1, 0.63% ATM, and 2.5% had mutations in 15 other DDR-related genes. This suggests that DDR gene mutations are relatively common and occur independently of ethnicity [
10].
2.2. Role in Non-Metastatic PC
Given the potential impact of DDR gene mutations on the oncological outcomes of treatments for localized PCa, including surgery, radiotherapy, and active surveillance, it is important to consider how somatic DDR mutations might influence treatment efficacy. There is an urgent need for data on the implications of both germline and somatic DDR defects in early-stage PCa to better tailor treatment strategies from a precision medicine perspective.
In this context, the IMPACT study explored the role of PSA screening in men with BRCA mutations compared to controls, with a follow-up period of 3 years [
12]. The study found that the differences between BRCA mutation carriers and non-carriers were consistently more pronounced for BRCA2 mutations than for BRCA1 mutations. The incidence of PCa detected at biopsy was 5.2% in BRCA2 carriers compared to 3.0% in non-carriers. The distribution of high- or intermediate-risk PCa cases also differed significantly: 77% of BRCA2 carriers were classified as high or intermediate risk, compared to 40% of non-carriers. Additionally, the proportion of patients with ISUP grade 1 (low-risk) disease changed from 73% in non-carriers to 37% in BRCA2 carriers. These findings suggest that BRCA mutation carriers may have a more aggressive form of disease, which should be considered when selecting treatment options.
In line with this, Carter et al. investigated whether germline mutations were associated with grade reclassification (GR) in patients undergoing active surveillance (AS) for PCa [
13,
14]. Analyzing 1,211 PCa patients enrolled in AS, the study found that the incidence of tumor staging upgrades at 2, 5, and 10 years was higher in BRCA2 carriers than in non-carriers: 27%, 50%, and 78% for BRCA2 carriers versus 10%, 22%, and 40% for non-carriers. However, the retrospective nature of this study, as well as the lack of image-guided biopsies and multiparametric magnetic resonance imaging (mMRI), limits the ability to draw definitive conclusions. Further prospective studies are needed to better evaluate the role of active surveillance in patients with BRCA germline mutations.
The prognostic significance of HRR gene mutations in localized disease has also been explored in the context of active treatments such as surgery and radiotherapy. Castro et al. examined the impact of BRCA mutations on metastatic relapse and cause-specific survival (CSS) following radical treatments (surgery and radiotherapy) for localized PCa [
15]. At 3, 5, and 10 years after treatment, 97%, 94%, and 84% of non-carriers were free from metastasis, compared to 90%, 72%, and 50% of BRCA mutation carriers (p<0.001). The CSS rates were significantly higher in non-carriers at 3, 5, and 10 years (99%, 97%, and 85%, respectively) compared to carriers (96%, 76%, and 61%, respectively; p<0.001). Multivariate analysis confirmed that BRCA mutations were an independent prognostic factor for both metastasis-free survival (MFS) (hazard ratio [HR]: 2.36; 95% confidence interval [CI], 1.38-4.03; p=0.002) and CSS (HR: 2.17; 95% CI, 1.16-4.07; p=0.016).
A similar study by Martinez Chanza et al. in a retrospective cohort of 380 patients with localized and metastatic hormone-sensitive PCa also found that BRCA mutations were associated with a higher risk of relapse in localized disease [
16]. Furthermore, Castro et al. conducted a retrospective analysis of 2,019 PCa patients, which revealed that BRCA1/2 mutations were more frequently associated with high-grade disease (Gleason score ≥8, P = .00003), advanced tumor stage (T3/T4, P = .003), nodal involvement (P = .00005), and metastases at diagnosis (P = .005) compared to non-carriers . These findings further underscore the potential importance of BRCA mutations in the prognosis and management of localized PCa [
17,
18,
19,
20].
2.3. Role in Metastatic PC
Several studies have explored the prognostic significance of DDR mutations in mCRPC patients treated with standard therapies. In a retrospective study of 319 mCRPC patients, Annala et al. found that carriers of DDR mutations had significantly shorter progression-free survival (PFS) compared to non-carriers (3.3 vs. 6.2 months, p=0.01) when treated with first-line androgen receptor pathway inhibitors (ARPI) [
21]. In contrast, Antonarakis et al., in a study of 172 mCRPC patients receiving first-line ARPI, observed a trend toward longer PFS in ATM and BRCA1/2 mutation carriers compared to non-carriers (15 vs. 10.8 months, p=0.090) [
22].
The Prorepair-B trial was the first prospective study to evaluate the prognostic impact of BRCA1/2 and other DDR gene mutations on cause-specific survival (CSS) in mCRPC patients. The study, which analyzed all DDR mutations collectively, did not find significant differences in CSS between carriers and non-carriers (DDR mutation carriers 23.3 months vs. non-carriers 33.2 months; p=0.264; HR: 1.32; 95% CI: 0.81-2.17). However, when looking specifically at BRCA2 mutations, the study found that BRCA2 mutation carriers had significantly worse CSS compared to non-carriers (BRCA2 carriers 17.4 months vs. non-carriers 33.2 months; p=0.027; HR: 2.10; 95% CI: 1.07-4.10). Further subgroup analysis revealed that BRCA2 mutation carriers had a shorter CSS when treated with a docetaxel-ARSI sequence (10.7 months) compared to those treated with an ARPI-docetaxel sequence (24.0 months) [
8].
These findings suggest that BRCA2 mutations have a negative impact on the outcomes of mCRPC, and that the choice of first-line therapy may influence the prognosis for BRCA2 mutation carriers.
3.0. How to Detect
DDR alterations can arise early in the development of aggressive tumors and may be detectable in prostate biopsies or prostatectomies. However, other alterations may emerge later during disease progression, in which case metastatic tumor biopsies might be the preferred method for detection. However, biopsies of metastatic lesions often could be dangerous or not feasible.
3.1. Somatic Samples: Recent vs Archived
In the Profound study, a total of 4,858 tissue samples were centrally tested and analyzed for HRR PVs. Next-generation sequencing (NGS) results were successfully obtained for 58% of the samples, representing 69% of the patients. Of the total samples collected, 83% were from primary tumors (96% of which were archival and 4% newly obtained), and 17% were from metastatic tumors (67% archival and 33% newly obtained). NGS results were more commonly obtained from newly acquired samples (63.9%) compared to archived samples (56.9%), and from metastatic samples (63.9%) compared to primary tumor samples (56.2%) [
8].
The authors noted that DNA yield had a greater impact on the success of NGS results (AUC = 0.6292) than other variables, such as sample age or tumor content percentage. Although NGS success rates decreased as sample age increased, approximately 50% of samples older than 10 years still produced viable results. Another factor that affected eligibility was the total tissue volume. In the Profound study, samples with a tissue volume greater than 0.6 mm³ had a higher rate of generating NGS results (58.9%; 95% CI, 57.4–60.3) compared to samples with volumes of 0.2 to ≤ 0.6 mm³ (32.1%; 95% CI, 24.4–40.6) [
8]. Overall, the Profound study demonstrated that tissue testing for HRR alterations is feasible, with high-quality tumor tissue being crucial for generating NGS results [
8].
The analysis of DDR mutations in PCa should prioritize somatic testing before germline testing. When DDR gene mutations arise during disease progression, metastatic tumor biopsies are the ideal method for identifying these molecular alterations. However, obtaining biopsies from metastatic lesions can be challenging or even unfeasible due to their location and the invasive nature of the procedure. Additionally, a single biopsy may not capture the full tumor heterogeneity across different metastases. Furthermore, processing bone biopsy samples, which typically requires decalcification, can lead to reduced DNA yield and quality. In the Profound study, bone samples showed a lower proportion of successful NGS results compared to other biopsy sites (42.6%) [
1,
2,
3].
3.2. Germline Analysis in PC
Germline analysis is closely linked to genetic counseling for families of prostate cancer patients with somatic pathogenic variants (PVs). Pre-test counseling should gather detailed family history (spanning at least three generations) and discuss available genetic testing options. Post-test counseling should focus on interpreting the results, explaining the potential risks for various cancers, and outlining the need for any intensive screening strategies. Germline testing can be performed using various approaches, such as small-focused panels (5-6 genes), cancer-specific panels (10-15 genes), or large comprehensive panels (≥80 genes), with tests increasing in complexity depending on the number of genes assessed. If a test result is negative or identifies a "variant of uncertain significance" (VUS), this should not be considered a pathogenic variant. VUS results are often eventually classified as non-pathogenic after further investigation [
1].
Currently, BRCA testing for germline pathogenic variants is typically performed using validated methods, including next-generation sequencing (NGS), and results are generally confirmed through techniques such as Multiplex Ligation Probe Amplification (MLPA) or Multiplex Amplicon Quantification (MAQ) [
3].
Although several methods exist for classifying constitutional BRCA variants, it is recommended to follow the criteria established by the Evidence-based Network for the Interpretation of Germline Mutant Alleles (ENIGMA) [
1,
3]. ENIGMA criteria are used to assess the clinical significance of sequence variants in genes related to breast, ovarian, and prostate cancer. According to ENIGMA, test results are classified into five categories based on the probability of pathogenicity (PP):
Benign variants (PP <0.001)
Likely benign or of limited clinical significance (PP: 0.001–0.049)
Uncertain significance (PP: 0.05–0.949)
Likely pathogenic (PP: 0.95–0.99)
Pathogenic (PP >0.99)
3.3. Circulating DNA (cDNA)
The analysis of free circulating DNA (cDNA) presents a promising approach, especially in cases where obtaining tissue samples is difficult or not possible [
23]. However, current data do not yet completely support the reliable use of cDNA testing in clinical practice. The first study to investigate cDNA in this context was the GHALAND study, a Phase 2 trial evaluating treatment outcomes with Niraparib in mCRPC patients with DDR mutations. The treatment efficacy was assessed based on the presence of circulating tumor cells (CTC) starting from the eighth week of treatment. The best results were seen in the BRCA cohort, which had a 24% CTC response, compared to the non-BRCA cohort [
24]. However, the analysis of HRR mutations through a liquid biopsy and the determination of circulating DNA represents the potentially most interesting method capable of simplifying the difficulties of somatic determination [
25].
Despite this promising initial result, commercial tests for cDNA analysis still show significant inconsistencies, with discrepancies as high as 40% between different tests.
4.0. Personal Experience - Experimental Design
Study protocol: We developed an experimental multidisciplinary prospective study in patients with new diagnosis of high-risk prostate adenocarcinoma at biopsy. The protocol has been approved by our internal Committee Sapienza University (000019_22_ATENEO2022_DDG N.n.4994/2022)
Primary objective: to evaluate and compare the tissue expression of pathogenetic variants (PV) of different genes involved in homologous recombination (HRDR) in patients with the first diagnosis of PCa (de novo) in relation to a metastatic or non-metastatic stage, tumor aggressiveness and early risk of progression.
Population: de novo histological diagnosis of prostatic adenocarcinoma at biopsy.
Inclusion criteria: patients with newly diagnosed histologically confirmed prostate adenocarcinoma at biopsy (de novo), intermediate or high risk (ISUP score 3-5, presence of Gleason Grades 4-5); stage metastatic or non-metastatic.
Exclusion criteria: current or previous androgen deprivation therapies, chemotherapy, radiotherapy or other therapies capable of influencing the growth and progression of PCa.
4.1. Methods
4.1.1. Urologic Evaluation
All cases are submitted to local staging with multiparametric magnetic resonance (mMR) and targeted biopsy. Systemic staging to classify between metastatic and non-metastatic cases is obtained through a PET CT scan (PSMA or choline). Family analysis up to the third generation for prostate, ovarian, breast, pancreatic, colon cancers is reported and longitudinal assessment for early (within 12 months) biochemical (total PSA) and/or radiological progression is analyzed.
Table 1 and
Table 2 describes patients characteristics and results from genetic analysis.
4.1.2. Pathologic Preparation
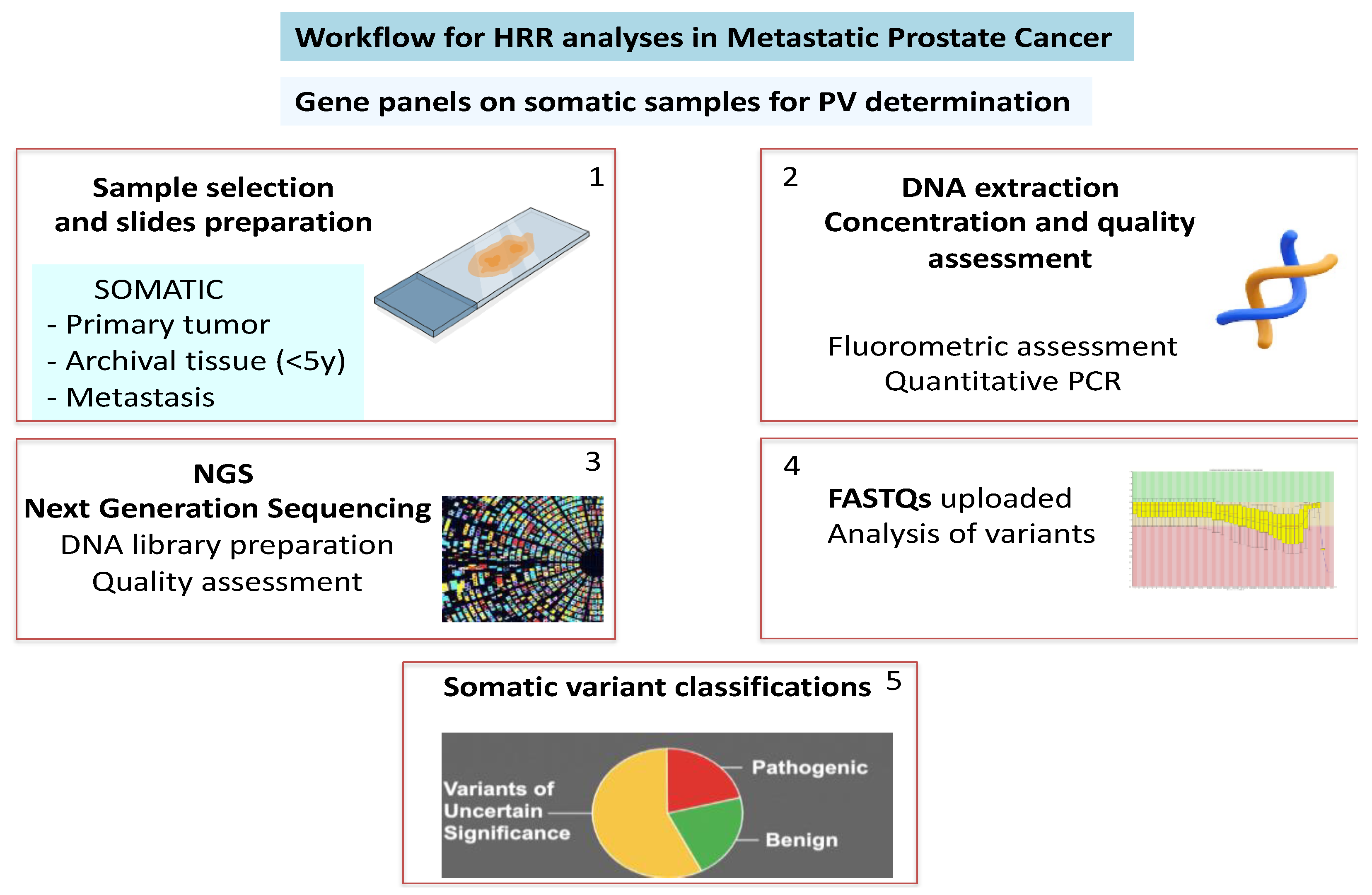
In each case, selection of the most representative prostate adenocarcinoma tissue with Grades 4-5 at biopsy and description of cellularity in each slide is performed. At now formalin fixed-paraffin-embedded (FFPE) tumor samples from 22 PCa patients have been collected. The diagnosis of cancer samples is evaluated by genitourinary pathologist on hematoxiline and eosine (H&E)-stained slides. From each patient we had chosen from 1 to 3 slides. Selected samples are cut into 5-10 × 5 µm sections with the last H&E stained 4 µm sections to confirm and assess tumor cellularity.
4.1.3. Genetic Analysis
DNA was extracted from 43 Formalin-fixed paraffin-embedded (FFPE) slices obtained from the prostate adenocarcinoma biopsies of 22 patients, using the MagCore® Super Automated Nucleic Acid Extractor (Diatech Lab Line, Jesi, Italy) by the MagCore® Genomic DNA FFPE One-Step Kit according to the manufacturer's instructions. DNA concentration was assessed by fluorometric method using Qubit 3.0 and the Qubit™ 1X dsDNA High Sensitivity (HS) (ThermoFisher). The DNA quality was tested by a quantitative PCR using Archer PreSeq DNA QC Assay on the Archer VariantPlex instrument.
Next Generation Sequencing (NGS): the DNA libraries were prepared by the Archer™ VARIANTPlex™ Pan Solid Tumor Panel according to the manufacturer's instructions. The quality and concentration of the prepared libraries were evaluated using Qubit 3.0 and the Qubit™ 1X dsDNA High Sensitivity (HS) and TapeStation with D1000 ScreenTape. Libraries that met the quality threshold are pooled and run on NextSeq 550dx. The resulting FASTQs were uploaded to the Archer Server for analysis and variant reporting.
Somatic variants were classified according to the standards and guidelines of the American Society of Clinical Oncology, the joint recommendations of the Clinical Genome Resource (ClinGen), the Cancer Genomics Consortium (CGC) and the Variant Interpretation for Cancer Consortium (VICC) using ArcherDX analysis software (
Figure 1). Only variants assigned to tier I (oncogenic) tier II (likely oncogenic) and tier III (variant of uncertain significance) were included in this study.
4.2. Findings
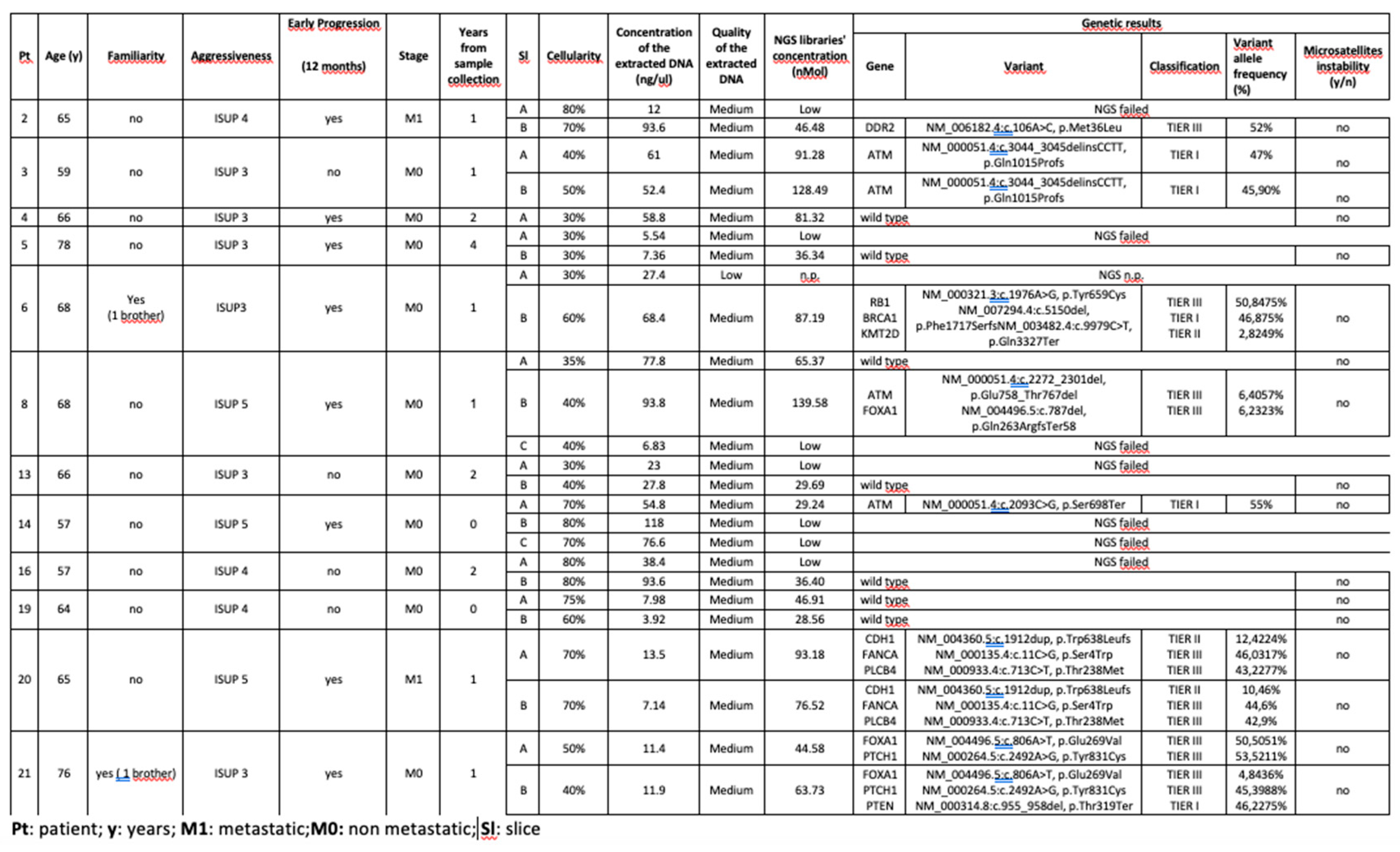
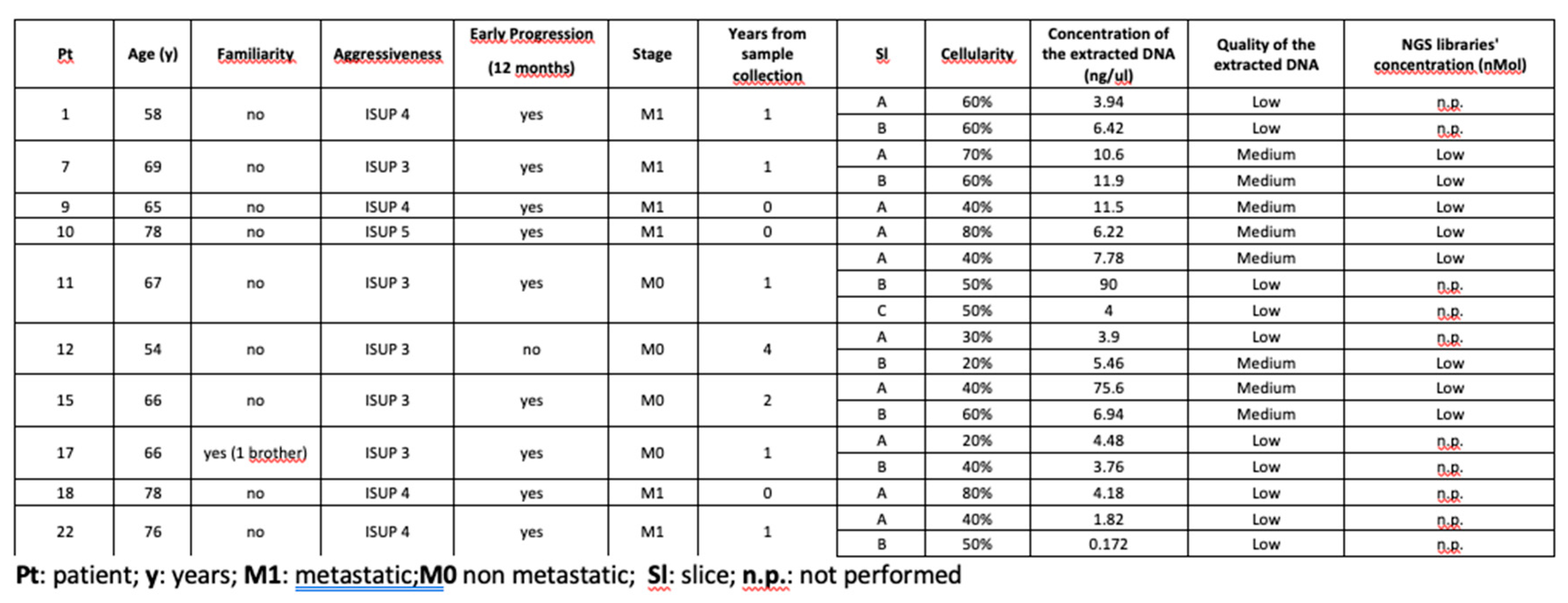
Among the DNA samples from the 43 FFPE slices, the concentration and quality of 25 were sufficient to proceed with the preparation of the NGS libraries (
Table 1). Thereafter, the sequencing of 7 samples failed due to low libraries’ concentration (
Table 2).
Therefore, the genetic NGS data were obtained for 17 samples belonging to 12 different patients. DNA variants were filtered by the following criteria: (i) read depth >300X; (ii) MAF (Minor Allele Frequency) in gnomAD global population database <5%, (iii) number of times a variant was observed on the system in different runs <5; (iv) sample’s allele frequency >2%. In total, 14 different DNA variants were prioritized in 10 bioptic samples from 9 different patients. The Variant Allele Frequency (VAF) was comprised between 3% and 53%. Since blood DNA samples from the patients were not available, we cannot exclude that the variants with a high VAF were constitutional (
Table 1).
Five samples from non-metastatic patients (cases 3,6,8,14,21) and one sample from a metastatic case at first diagnosis (case 20), all with early progression (no later than 12 months from treatment) (except case 3 without early progression) were found to carry at least one tier I/II variant.
According to the standards and guidelines of the American Society of Clinical Oncology, the variant NM_000314.8:c.955_958del (p.Thr319Ter) in the PTEN gene was classified as Tier I. Indeed, the p.Thr319Ter has been validated for an FDA-approved therapy for the treatment of patients with PTEN-mutant ER+/HER2- metastatic breast cancer, but the clinical benefit of this combination in patients with PTEN Thr319Ter mutant prostate cancer is unknown. This variant was only found in one of the two FFPE samples from patient 21, a patient with familiarity (1 brother with prostate cancer), aged 76 years, non-metastatic PCa, ISUP 3 with early biochemical progression in a hormone-sensitive tumor.
Three out of the 14 different prioritized variants were found in the
ATM gene, all non-metastatic cases with early progression. A
BRCA1 oncogenic variant was detected in only one PCa case (case 6), non-metastatic, early progression and familiarity for prostate cancer. The same patient also carried one tier II variant in
KMT2D, as well as a variant of uncertain significance in
RB1. Very recently, in mouse, the Kmt2c deficiency has been found as a metastatic driver [
26]. A truncating tier II variant in the
CDH1 gene was found in the PCa biopsy from patient 20 (metastatic and early progression); the role of
CDH1 mutations in not-gastric cancers has yet to be defined.
BRCA2 variants were not identified. Of note, the PCa biopsy of two patients (cases 8,21, both non-metastatic and early progression) carried variants in
FOXA1, a gene that plays an important role in regulating steroid receptor functions and whose expression has been reported to be related to several human cancers, as both an oncogene and a tumor suppressor gene depending on the specific cancer types gene [
27]. In particular,
FOXA1 is thought to play an important role in prostate cancer progression and may be a factor leading to aggressive prostate cancer [
28]
Five samples passed the QC metric for MSI (microsatellite site instability) and no instability was detected. No copy number variants (CNV) were identified in any sample.
4.2.1. Critical Analysis
To date, the analysis of HRR PVs in PCa patients remains complex, time-consuming and at high cost, so that it is often limited to single gene such as BRCA. BRCA PVs have been shown to have the highest predictive value in patients with metastatic PCa, regarding sensitivity for PARP inhibitors therapy. However, the predictive value of a more extensive analysis of a large pool of PVs in the HRR family, could offer prognostic indications and response to therapies not only limited to the use of PARP inhibitors. This expansion of analysis could be particularly useful in an earlier phase of disease, in non-metastatic PCa. The high complexity and costs of this type of analysis can limit its use in clinical practice and a selection in high-risk patients or based on family histories is certainly necessary. It is possible that genes other than BRCA and other PVs may be predominant and prognostically more useful in this earlier disease setting.
5.0. Conclusions
Prostate cancer is one of the most common cancers affecting men, with a substantial impact on both survival and quality of life, as well as significant social and psychological consequences. Despite extensive research in recent years, awareness of the genetic risks associated with PCa remains low. There is a critical unmet need to develop effective health promotion strategies and screening programs targeted at younger men with genetic mutations linked to a higher risk of developing PCa.
Currently, there is no international consensus on the optimal management approach for men at genetic risk for prostate cancer. However, some key findings have emerged:
The incidence of pathogenic variants (PVs) in HRR genes among men with metastatic PCa ranges from 11% to 33%, which is notably higher than in non-metastatic prostate cancer (nmPC). Within the metastatic setting, BRCA2 mutations are more prevalent compared to other HRR gene mutations.
Identifying somatic or germline HRR PVs, particularly BRCA2 mutations, plays a crucial role in personalizing treatment with PARP inhibitors in metastatic castration-resistant prostate cancer (mCRPC). This approach has shown significant improvements in radiographic progression-free survival (rPFS) and overall survival (OS). As a result, this strategy has been recommended by international guidelines and has received approval from both the FDA and EMA.
Several points remain undefined and relevant unmet need must be addressed:
- -
to better define clinical and pathological characteristics of newly diagnosed prostate cancer associated to DDR genes defects
- -
to offer a platform for approaching personalized medicine based on genetic assessment of PVs in DDR genes also in a non-metastatic stage
- -
to define whether the expression of PVs of DDR genes is also relevant in non -metastatic prostate cancer at high risk
- -
to define whether BRCA2 remains the main PV expressed also in non-metastatic prostate cancer cases or other PVs for different DDR genes are similarly expressed and useful
- -
to simplify the detection of PVs of DDR genes, exploring not only the somatic but also other approaches
Author Contributions
All authors significantly contributed to the research and the manuscript. Irene Bottillo, Paola Grammatico, Francesca Bacigalupo, Francesco Pastacaldi, Maria Pia Ciccone: organization and execution of each genetic analysis; interpretation of data Fabio Magliocca, Elisabetta Merenda: organization and execution of each pathologic analysis and preparation Valerio Santarelli,Alessandro Gentilucci, Beatrice Sciarra: collecting data, writing manuscript, figures and tables Laura De Marchis, Daniele Santini: contributing to clinical trial definition and progression, methodology,literature research Alessandro Sciarra, Stefano Salciccia, Giulio Bevilacqua: organizing the review; data collection, Writing the manuscript, data interpretation, study design, supervision
Funding
This research received no external funding. Sapienza University covered the costs relating to the materials for genetic analysis
Institutional Review Board Statement
The experimental protocol was approved by our internal committee Sapienza University (000019_22_ATENEO2022_DDG N.n.4994/2022)
Informed Consent Statement
all patients gave their informed consensus for the procedure.
Data Availability Statement
experimental data are available, and all presented in
Table 1 and
Table 2
Acknowledgments
no acknowledgments
Conflicts of Interest
The authors declare no conflicts of interest. None of the contributing authors have any conflict of interest, including specific financial interests or relationships and affiliations relevant to the subject matter or materials discussed in the manuscript.
References
- Castro E, Romero-Laorden N, Del Pozo A, Lozano R, Medina A, Puente J, Piulats JM, Lorente D, Saez MI, Morales-Barrera R, Gonzalez-Billalabeitia E, Cendón Y,García-Carbonero I, Borrega P, Mendez Vidal MJ, Montesa A, Nombela P, Fernández-Parra E, Gonzalez Del Alba A, Villa-Guzmán JC, Ibáñez K, Rodriguez-Vida A,Magraner-Pardo L, Perez-Valderrama B, Vallespín E, Gallardo E, Vazquez S,Pritchard CC, Lapunzina P, Olmos D. PROREPAIR-B: A Prospective Cohort Study of the Impact of Germline DNA Repair Mutations on the Outcomes of Patients With Metastatic Castration-Resistant Prostate Cancer. J Clin Oncol. 2019 Feb 20;37(6):490-503.
- Sciarra A, Frisenda M, Bevilacqua G, Gentilucci A, Cattarino S, Mariotti G,Del Giudice F, Di Pierro GB, Viscuso P, Casale P, Chung BI, Autorino R,Crivellaro S, Salciccia S. How the Analysis of the Pathogenetic Variants of DDR Genes Will Change the Management of Prostate Cancer Patients. Int J Mol Sci.2022 Dec 30;24(1):674-82.
- Sciarra A, Fiori C, Del Giudice F, DI Pierro G, Bevilacqua G, Gentilucci A,Cattarino S, Mariotti G, Salciccia S. DDR genes analysis and PARP-inhibitors therapy as tailored management in metastatic prostate cancer: achieved answers, open questions and future perspectives. Minerva Urol Nephrol. 2022 Dec;74(6):649-652.
- Giglia-Mari G, Zotter A, Vermeulen W. DNA damage response. Cold Spring Harb Perspect Biol. 2011;3(1):a000745.
- Caldecott KW. Mammalian single-strand break repair: mechanisms and links with chromatin. DNA Repair (Amst). 2007;6(4):443-453.
- Rouleau, M., Patel, A., Hendzel, M. PARP inhibition: PARP1 and beyond. Nat Rev Cancer 2010; 10: 293–301.
- Castro E, Goh C, Olmos D. Germline BRCA mutations are associated with higher risk of nodal involvement, distant metastasis, and poor survival outcomes in prostate cancer. J Clin Oncol. 2013;31(14):1748-1757.
- Hussain M, Corcoran C, Sibilla C, Fizazi K, Saad F, Shore N, Sandhu S, Mateo J, Olmos D, Mehra N, Kolinsky MP, Roubaud G, Özgüroǧlu M, Matsubara N, Gedye C, Choi YD, Padua C, Kohlmann A, Huisden R, Elvin JA, Kang J, Adelman CA, Allen A, Poehlein C, de Bono J. Tumor Genomic Testing for >4,000 Men with Metastatic Castration-resistant Prostate Cancer in the Phase III Trial PROfound (Olaparib). Clin Cancer Res. 2022 Apr 14;28(8):1518-1530.
- Wei Y, Wu J, Gu W, Qin X, Dai B, Lin G, Gan H, Freedland SJ, Zhu Y, Ye D. Germline DNA Repair Gene Mutation Landscape in Chinese Prostate Cancer Patients. Eur Urol. 2019 Sep;76(3):280-283.
- Pritchard CC, Mateo J, Walsh MF, De Sarkar N, Abida W, Beltran H, Garofalo A, Gulati R, Carreira S, Eeles R, Elemento O, Rubin MA, Robinson D, Lonigro R, Hussain M, Chinnaiyan A, Vinson J, Filipenko J, Garraway L, Taplin ME, AlDubayan S, Han GC, Beightol M, Morrissey C, Nghiem B, Cheng HH, Montgomery B, Walsh T, Casadei S, Berger M, Zhang L, Zehir A, Vijai J, Scher HI, Sawyers C, Schultz N, Kantoff PW, Solit D, Robson M, Van Allen EM, Offit K, de Bono J, Nelson PS. Inherited DNA-Repair Gene Mutations in Men with Metastatic Prostate Cancer. N Engl J Med. 2016 Aug 4;375(5):443-53.
- ICGC/TCGA Pan-Cancer Analysis of Whole Genomes Consortium. Pan-cancer analysis of whole genomes. Nature. 2020 Feb;578(7793):82-93. [CrossRef]
- Page EC, Bancroft EK, Brook MN, Assel M, Hassan Al Battat M, Thomas S, Taylor N, Chamberlain A, Pope J, Raghallaigh HN, et al. Interim Results from the IMPACT Study: Evidence for Prostate-specific Antigen Screening in BRCA2 Mutation Carriers. Eur. Urol. 2019, 76, 831–842.
- Chen RC, Rumble RB, Loblaw DA. Active surveillance for the management of localized prostate cancer (Cancer Care Ontario Guideline): American Society of Clinical Oncology Clinical Practice Guideline Endorsement. J Clin Oncol 2016;34:2182–90.
- Carter HB, Helfand B, Mamawala M. Germline Mutations in ATM and BRCA1/2 Are Associated with Grade Reclassification in Men on Active Surveillance for Prostate Cancer. Eur Urol. 2019;75(5):743-749.
- Castro E, Goh C, Leongamornlert D. Effect of BRCA Mutations on Metastatic Relapse and Cause-specific Survival After Radical Treatment for Localised Prostate Cancer. Eur Urol. 2015;68(2):186-193.
- Martinez Chanza N, Bernard B, Barthelemy P, Accarain A, Paesmans M, Desmyter L, T’Kint de Roodenbeke D, Gil T, Sideris S, Roumeguere T, et al. Prevalence and clinical impact of tumor BRCA1 and BRCA2 mutations in patients presenting with localized or metastatic hormone-sensitive prostate cancer. Prostate Cancer Prostatic Dis. 2022, 25, 199–207.
- Lang SH, Swift SL, White H, Misso K, Kleijnen J, Quek RGW. A systematic review of the prevalence of DNA damage response gene mutations in prostate cancer. Int J Oncol. 2019;55(3):597-616.
- Eeles R, Goh C, Castro E. The genetic epidemiology of prostate cancer and its clinical implications. Nat Rev Urol. 2014;11(1):18-31.
- Kim IE Jr, Kim S, Srivastava A. Similar incidence of DNA damage response pathway alterations between clinically localized and metastatic prostate cancer. BMC Urol. 2019;19(1):33.
- Marshall CH, Fu W, Wang H, Baras AS, Lotan TL, Antonarakis ES. Prevalence of DNA repair gene mutations in localized prostate cancer according to clinical and pathologic features: association of Gleason score and tumor stage. Prostate Cancer Prostatic Dis. 2019;22(1):59-65.
- Annala M, Struss WJ, Warner EW, Beja K, Vandekerkhove G, Wong A, Khalaf D, Seppälä IL, So A, Lo G, Aggarwal R, Small EJ, Nykter M, Gleave ME, Chi KN, Wyatt AW. Treatment Outcomes and Tumor Loss of Heterozygosity in Germline DNA Repair-deficient Prostate Cancer. Eur Urol. 2017 Jul;72(1):34-42.
- Antonarakis ES, Lu C, Luber B, Liang C, Wang H, Chen Y, Silberstein JL, Piana D, Lai Z, Chen Y, Isaacs WB, Luo J. Germline DNA-repair Gene Mutations and Outcomes in Men with Metastatic Castration-resistant Prostate Cancer Receiving First-line Abiraterone and Enzalutamide. Eur Urol. 2018 Aug;74(2):218-225.
- Carr TH, Adelman C, Barnicle A, Kozarewa I, Luke S, Lai Z, Hollis S,Dougherty B, Harrington EA, Kang J, Saad F, Sala N, Thiery-Vuillemin A, Clarke NW, Hodgson D, Barrett JC. Homologous Recombination Repair Gene Mutation Characterization by Liquid Biopsy: A Phase II Trial of Olaparib and Abiraterone in Metastatic Castrate-Resistant Prostate Cancer. Cancers (Basel). 2021 Nov 20;13(22):5830-42.
- Smith MR, Scher HI, Sandhu S, Efstathiou E, Lara PN Jr, Yu EY, George DJ, Chi KN, Saad F, Ståhl O, Olmos D, Danila DC, Mason GE, Espina BM, Zhao X, Urtishak KA, Francis P, Lopez-Gitlitz A, Fizazi K; GALAHAD investigators. Niraparib in patients with metastatic castration-resistant prostate cancer and DNA repair gene defects (GALAHAD): a multicentre, open-label, phase 2 trial. Lancet Oncol. 2022 Mar;23(3):362-373.
- Torga G, Pienta KJ. Patient-Paired Sample Congruence Between 2 Commercial Liquid Biopsy Tests. JAMA Oncol. 2018 Jun 1;4(6):868-870.
- Cai H, Zhang B, Ahrenfeldt J, Joseph JV, Riedel M, Gao Z, Thomsen SK,Christensen DS, Bak RO, Hager H, Vendelbo MH, Gao X, Birkbak N, Thomsen MK.CRISPR/Cas9 model of prostate cancer identifies Kmt2c deficiency as a metastatic driver by Odam/Cabs1 gene cluster expression. Nat Commun. 2024 Mar 7;15(1):2088.
- Wang D, Garcia-Bassets I, Benner C, Li W, Su X, Zhou Y, Qiu J, Liu W,Kaikkonen MU, Ohgi KA, Glass CK, Rosenfeld MG, Fu XD. Reprogramming transcription by distinct classes of enhancers functionally defined by eRNA. Nature. 2011 May 15;474(7351):390-4.
- Song B, Park SH, Zhao JC, Fong KW, Li S, Lee Y, Yang YA, Sridhar S, Lu X, Abdulkadir SA, Vessella RL, Morrissey C, Kuzel TM, Catalona W, Yang X, Yu J.Targeting FOXA1-mediated repression of TGF-β signaling suppresses castration-resistant prostate cancer progression. J Clin Invest. 2019 Feb 1;129(2):569-582.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).