4. Nanomaterials Used in Skincare Products
The inclusion of both hydrophilic and hydrophobic compounds, enhanced stability, controlled release, targeted transport to target cells, and the capacity to elicit a biological reaction at concentrations below therapeutic levels are just a few benefits of using nanomaterials in cosmetics [
29]. Nowadays, it's well understood that using nanoscale materials, which serve as both an active component and/or a carrier, can enhance the performance of cosmetics in a variety of ways. As a result, it may be simple to identify certain technologies as industry standards [
20,
30].
Cubosomes, nanodots, liposomes, dendrimers, and nano-emulsions are only a few examples of nano-sized substances that are now frequently used in cosmetics as a result of the nanotechnology integration into the manufacture and formulation of cosmetics. These nanoparticle-based cosmetics also referred to as nano-cosmeceuticals, have increased the potential uses of cosmetics to treat aging-related skin conditions like distributed hyperpigmentation, dehydration, and wrinkled skin [
3,
31]. In addition, numerous different nanosized varieties can be found in sunscreen, anti-aging products, and curling irons [
31].
Nano-carriers have been utilized for passive drug administration into and through the skin because they have several benefits compared to typical passive distribution techniques, including increased drug loading, site-targeted distribution, the regulated release of the active ingredient, improved stability, soothed skin irritation, protection from degradation, larger surface area, higher solubility, and enhanced active pharmaceutical ingredient penetration into the skin [
30,
32].
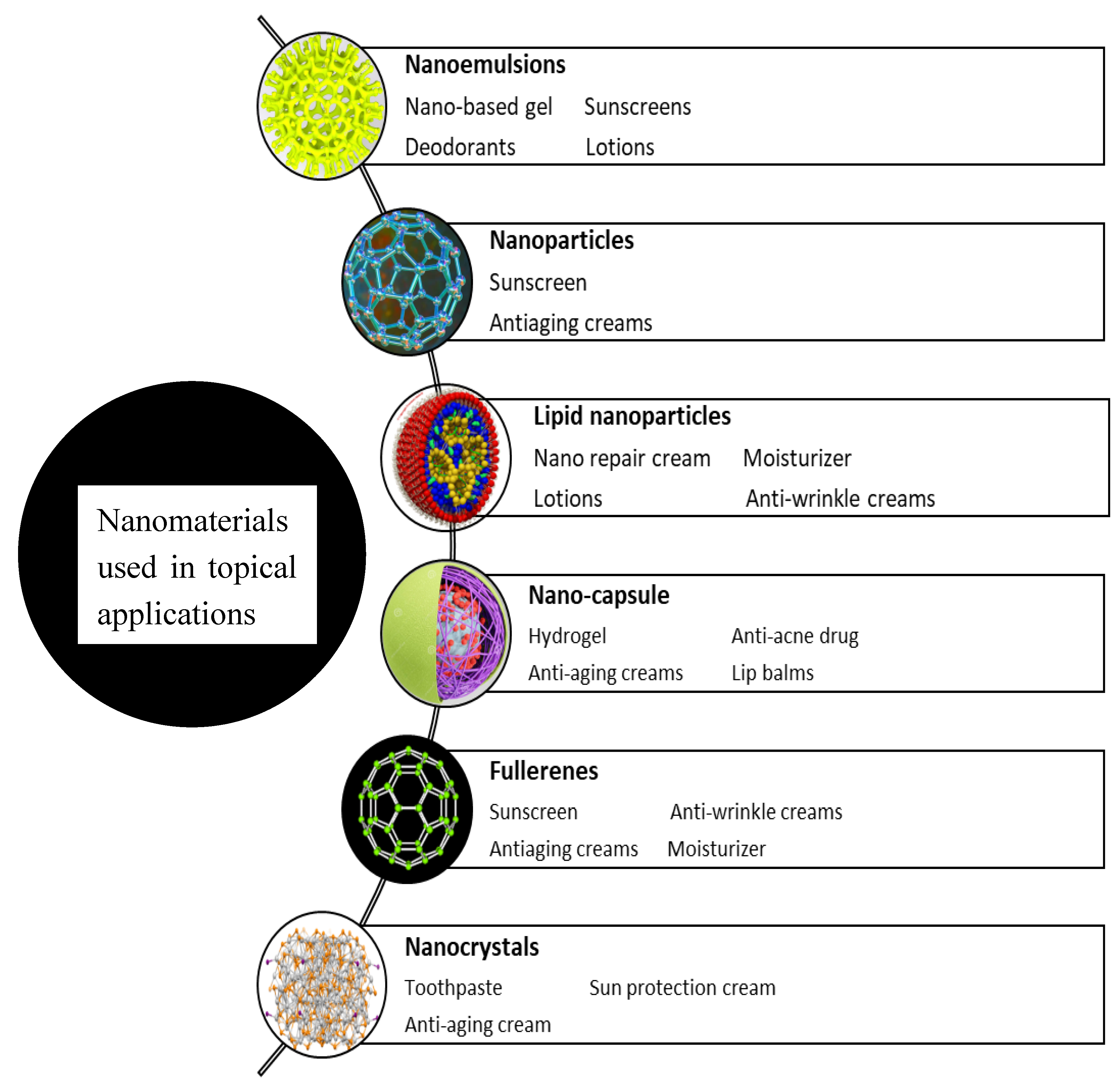
Figure 2.
Nanomaterials used in topical applications.
Figure 2.
Nanomaterials used in topical applications.
When developing drug delivery systems in the early 1970s, liposomes were the first type of nano-carriers used [
32]. Liposomes are composed of a lipid bilayer, which is why Dior invented cream capture in 1986.
Hydrophobic and hydrophilic compounds can be delivered using liposomes, which are non-hazardous, biocompatible, and biodegradable vesicles that can easily encapsulate active ingsredients. By enhancing their skin absorption, liposomes can raise the concentration of active substances [
6]. To raise the percentage of bioactive components (such as vitamins A, E, and CoQ10) in the epidermis without causing any immediate or long-term harm, liposomes are applied topically or for transdermal distribution [
33]. Another one of the reasons liposomes are so used extensively in the cosmetics industry is that they are simple to make [
15].
Nanoemulsions are transparent liquid dispersions that contain a surfactant, an oil phase, and 20 to 200 nm in terms of size [
34]. The creation of nanoemulsions as a means of administering medications orally, parenterally, intranasally, topically, and for intratracheally. Topical nanoemulsions with gamma-aminobutyric acid, an inhibitory neurotransmitter with properties that calm the muscles, are being researched for the treatment of wrinkles [
35]. Bicontinuous nanoemulsions, water-in-oil nanoemulsions, oil-in-water nanoemulsions, and many other varieties of nanoemulsions can be produced depending on the composition. Sinerga's Nanocream
® is an emulsifier mixture that may be used to create oil-in-water nanoemulsions. Wet wipes, sprayable emulsions, and hyper-fluid emulsions are all intended applications for this substance [
19,
36].
Nanoemulsion (O/W nanoemulsion) offers better cosmetic characteristics that make this nanosystem capable of producing micellar nanoparticles of lesser sizes (10 - 200 nm), which promotes uniform distribution on the skin, high stability, boosts active component delivery capabilities, pleasant aesthetic character, bigger surface area, and skin feel [
18,
37]. The reason is that they are stable for the skin's absorption of lipophilic substances, and they are an excellent carrier for the treatment of acne vulgaris [
38].
Microdispersions called nanoemulsions are nanosized droplets of one insoluble liquid in another that are differentiated by their great thermodynamic stability and small dispersed phase size (100 nm) [
39]. Active substances can more easily reach the skin and hair because of nanoemulsions' pleasant, light, and non-greasy nature. They are an effective method of delivering certain critical hydrophobic chemicals (antioxidants, fat-soluble vitamins, lipids, etc.) when used in therapeutic concentrations (antioxidants, fat-soluble vitamins, lipids, etc.) [
40]. Nanoemulsions are utilized to increase the skin's barrier function and decrease transdermal water loss [
29,
40].
Many cosmeceuticals, including deodorants, sunscreens, shampoos, lotions, nail polishes, conditioners, and hair serums, use nanoemulsions as a delivery system [
41]. In the development of cosmetics, nanoemulsions are utilized to promote quick penetration, active transport of active ingredients, and skin hydration [
42].
For instance, a new Nano-based gel has been developed by TRI-K Industries and its patent partner Kemira, to improve the effectiveness of different skin care products. One novel nanoemulsion carrier system is called Kemira Nanogel to is simple to make while simultaneously offering extra advantages as a result of its nanotechnology characteristics [
27].
Lipid nanoparticles, which can range in size from 50 to 1000 nm, are physiological lipid-based submicron colloidal carriers distributed in aqueous surfactant solutions or water. Solid Lipid Nanoparticles (SLNs) are frequently employed in the cosmeceuticals industry due to their intrinsic qualities, such as constrained features, a tiny structure that permits direct proximity to the skin, enhanced skin permeability, minimal toxicity, and other qualities. The benefits of SLNs and nanostructured lipid carriers (NLCs) include improved shelf life of the finished product, improved bioavailability and site-specific action, desired degree of occlusion and consequent skin hydration, and active ingredient stabilization [
6]. The SLN/NLC mark abides by a set of standards.
The following is outlined in the "VW Beetle 1938 Principle": 1. 1. Physical adherence of the skin 2. Occlusion due to physical film formation 3. Physical barriers that prevent drug entry 4. Chemical interactions between skin lipids and particle lipids. It prevents the materials from deteriorating [
6]. In turn, this causes chemicals to be released slowly. It is employed in the perfume business and as an insect repellant. Since it promotes skin moisture and permeates the corneal layer. To lessen the redness of an allergic reaction, they are also utilized by Nano repair creams and lotions [
43]. When compared to oral therapy, solid lipid nanoparticles with cyproterone acetate loaded on them improved transdermal delivery and penetration, which reduced systemic adverse effects and produced therapeutic medication levels in the target area. Acne lesions and sebum production can both be reduced with the help of the oral drug cyproterone acetate. NLCs, OCT, and SLNs were used to encase the BMDBM to block UVA rays. With 2.5% BMDBM and 1% OCT, the best outcomes are attained [
6,
44].
For dermal application of cosmetic items, well-tolerated carrier systems include SLNs and NLCs. There are a wide variety of cosmetic ingredients; Coenzyme Q10, retinol, tocopherol (vitamin E), and ascorbyl palmitate (vitamin A) are just a few. they provide controlled release profiles showing minimal toxicity and cytotoxicity for an extended period [
33,
45]. Additionally, Acne, skin mycosis, psoriasis, atopic eczema, and inflammations have all been treated with lipid nanoparticles in research studies [
46].
When applied to the skin's surface, ZnO and TiO2 nanoparticle mixtures are transparent. When compared to formulations with bigger particles, this transparency offers cosmetic acceptability. The goal of the current investigation was to evaluate the possible systemic exposure and epidermal penetration of an innovative sunscreen formulation containing ZnO nanoparticles. After applying ZnO nanoparticulate sunscreen formulation topically for 24 hours, we were able to pinpoint the ZnO particle distribution throughout the epidermis and measure the cumulative zinc content that penetrated by use of in vitro epidermal membranes. Recent studies on pig skin subjected to sunscreen formulations containing ZnO in minute form (averaging 80 nm for the main particles) and TiO2 (needle-like particles ranging in size from 30 to 60 nm) indicated that none of the particle types could pierce the pig's stratum corneum.
Additionally, the availability of the sunscreen ingredient octyl methoxycinnamate (OMC) in porcine skin was improved by encapsulating it in solid lipid nanoparticles (250 instead of being a direct result of nano-encapsulation itself, it was found in the latter investigation. This effect was produced via simple formulation thermodynamics, which improved partitioning into the stratum corneum. Solid lipid nanoparticles have been linked to higher degrees of penetration as a result of their immediate effect on skin moisture, which results in improved stratum corneum water retention, which causes the skin's surface to form a film and effectively occlude the application location. When compared to rates previously reported for chemical sunscreens, human skin only absorbed a small amount of nanoparticle-coated ZnO. A mineral oil vehicle allowed for the in vitro absorption of oxybenzone at levels of between 5 and 7 percent across human epidermal membranes, compared to 0.03-0.14% for octocrylene, 0.07 and 0.17 percent for ethylhexyl methoxycinnamate, and 0.09 and 0.20 percent for ethyl hexyl dimethyl [
6].
Recently, the use of nanoparticles made of titanium dioxide (TiO2) and zinc oxide (ZnO) has increased due to their ability to sustain the UV blocking and absorbing qualities of conventional sunscreens without having the white chalky appearance. Meanwhile, it has been claimed that a few changes to the standard ZnO or TiO2 UV protection system have increased the sun protection factor (SPF)[
33,
47]. Since 1995, L'Oreal has employed nanocapsules containing active substances including retinol, vitamin A, and beta-carotene that can penetrate the skin's deep layers to provide antiaging effects. The dimensions of these tiny biodegradable polymeric shells range from 300 to 600 nm [
48].
Essential oils are composed of lipophilic and extremely volatile secondary plant metabolites with their antibacterial activity, they can be employed in the cosmetics sector, complementary medicine, and agro-alimentary purposes [
49]. The active ingredient is contained in a cavity within the liquid/solid core of a nano-capsule, it has a polymer barrier surrounding it made of either natural or manmade polymers. The production of hydrogels and emulgels depends on nano-capsules. Additionally, hydrogel is employed in the production of several goods for personal care [
15]. The nanocapsule's sensory properties have been established that when the cosmetics industry grows, certain companies change the product's features to improve quality and effectiveness. Sensory analysis is used to accomplish this purpose. Only human reactions can determine how much this product's use is impacted. The study and development of the beauty industry have proven that this sensory analysis is a powerful analytical tool.
The active components in lipoic acid nanocapsules serve to both prevent and treat skin anti-aging. Increase the physiochemical stability of lipoic acid once the substance has been protected against deterioration via nanocapsulation. Lipoic acid-containing cosmetics outperform substitutes in terms of dependability, stickiness, oiliness, and sulfur odor [
50]. Furthermore, as compared to Benzoyl peroxide alone, the Chitosan-alginate Nanoparticles successfully benzoyl peroxide (BP), an anti-acne drug that is often encapsulated, which demonstrated stronger antibacterial efficacy against propionic bacterium acnes while causing less damage to eukaryotic cells [
6].
It is possible to load desired chemicals inside nanocapsules, which are polymeric nanoparticles with a hollow interior. These nanocapsules can then be shielded from the outside environment. To get retinol into the skin's deepest layers, L'Oreal (a French business with a nanotechnology patent in the US) has employed polymer nanocapsules [
26]. In addition, Lancôme developed a product called Primordiale Optimum Lip that delivered 100% botanically pure vitamin E using nanocapsule technology to prevent lip bleeding and feathering brought on by fine lines and wrinkles [
31].
Nanocapsules are employed in the manufacturing of cosmetics to safeguard chemicals, lessen chemical smells, and fix compatibility problems between formulation elements. Polymeric nanocapsule suspensions can either be used as a finished product that is applied directly to the skin or as an ingredient in semisolid formulations [
51]. UV filters, for example, can only penetrate a certain amount of skin with the use of nanocapsules [
52], or administer active chemicals to the skin's deep layers, such as genistein [
26,
53].
Following the nanocapsules' topical use produces a light coating of skin that evaporates when in contact with water. The increased potential of the skin to keep the vital element in reserve in comparison to this thin film for long-term delivery [
25,
54].
Fullerenes are a novel class of materials produced via nanotechnology, such as carbon fullerenes. The term "buckyballs" is also used to describe fullerenes. In terms of antioxidant activity, fullerenes outperform vitamins by at least two orders of magnitude. A brightening effect of fullerene is also present. By eliminating UV-induced free radicals and lowering excessive melanin formation, it enhances the appearance of the skin. The use of fullerenes in cosmeceutical treatments for skin renewal has received attention because of their antioxidant characteristics, which have been used in many cosmetic goods. They are utilized to create creams with a healthy appearance that are then applied to lighten under-eye shadows [
55,
56].
Due to their potent anti-radical oxygen species (ROS) scavenging abilities, fullerenes have been considered for usage in the creation of skin-rejuvenating cosmetic compositions [
57]; But there is still considerable disagreement over their safety [
33]. Because they penetrate the skin so well, 'Fullerene' nanoparticles made of carbon are used in moisturizers and anti-aging products. As a result, nano-scale compounds are frequently used in the cosmetics business. [
5].
The carbon spheres known as fullerenes measure 1 nm in diameter and contain 60 carbon atoms. Despite being hydrophobic, fullerenes can be chemically functionalized by incorporating a hydrophilic component, allowing them to be water-soluble and able to transport biomolecules like DNA and proteins [
58]. The therapeutic uses are made possible by their small size, spherical form, and hollow interior [
38]. Because aging skin is linked to increased oxidative stress and apoptosis, the development of cosmetic treatments for skin rejuvenation spurred a lot of scientists to research the application of fullerenes. In experiments with human keratinocytes, fullerenes showed substantial antioxidant properties and no cytotoxicity [
57,
59].
The cosmetic industry, which explores new cosmetic uses of scientific advancements, is a rapidly expanding sector. Fullerenes were the focus of research into cosmetics last year because of their potent antioxidant abilities and capacity to absorb free radicals. Although several research assessing the biological activities of fullerenes have produced highly encouraging results, their application in cosmetic items is still very low [
57].
The development of sunscreen formulas may make use of fullerenes. Effectively shields the skin from UVA and UVB radiation, as well as shielding it from DNA damage and immunosuppression. In a recent patent, it was stated that a sunscreen formulation comprising carbon nanotubes (structures similar to fullerene made of molecular-scale tubes of graphitic carbon) was developed [
57].
Nanocrystals demonstrated several properties, including increased membrane penetration, greater adhesion, and permeation of the gastrointestinal membrane; these qualities were subsequently utilized to affix nanoparticles for topical administration [
32]. Also, nanotechnology is a major help in toothpaste formulations for preventing the degeneration of tooth enamel [
6,
60].
Rutin is a flavonoid that is present in nanocrystals, which are made up of atoms in a monocrystalline or polycrystalline structure and have a minimum of one dimension smaller than 100 nanometers. A nanocrystal version of the antioxidant rutin called Juvedical was introduced in 2000 by the cosmetics company Juvena. According to a study, the original rutin molecule is 500 times more bioactive in this form than it is in its water-soluble form, rutin glycoside. Rutin glycoside and rutin nanocrystals were compared using the sun protection factor [
61]. It was particularly interesting since the skin contained a significantly higher concentration of the active ingredients synthesized as nanocrystals than the glycoside or powder, despite the nanocrystal solution being less soluble. A similar study discovered that Gel-based rutin nanocrystals had the strongest antioxidant (neutralizing) activity when compared to the powder or the form that is readily accessible for purchase [
31,
62].
Nanogold and nanosilver both possess antifungal and antibacterial properties. They are regularly used in cosmeceutical products such as face masks, deodorant, and anti-aging lotion. They have been added to facial masks and moisturizing creams [
63,
64].
Cosmetic manufacturers exploit nanosilver's enhanced antibacterial properties for a variety of applications. Several manufacturers currently provide underarm deodorants that purport to provide up to 24-hour antibacterial protection due to the silver in the product. The usage of gold nanoparticles in toothpaste is similar to that of nanosilver since they are thought to be very effective at removing oral germs [
3].
The size of gold nanoparticles, also known as nanogold, ranges from 5 nm to 400 nm. They can be formed into forms in a variety, including nanoshells, nanospheres, nanoclusters, nanorods, nanocubes, nanotriangles, and branching nanotriangles. Shape, size, dielectric qualities, and environmental factors all have a significant impact on the frequency response of gold nanoparticles.
By guiding the drug to the proper therapeutic region, niosomes, which are biodegradable and typically harmless vesicles made of nonionic surfactants, aid in the attainment of site-specific delivery. Although it is similar to liposomes, it is stable and has hydrophilic and hydrophobic ends. Lancôme was hence the first item L'Oreal released in 1987. Amphiphilic or lipophilic medicines are transported by niosomes [
29,
32]
Dendrimers are unimolecular nanostructures that are globular, symmetrical and have several strongly branching segments. The total number of branch series determines dendrimer formation [
19,
65]. Dendrimers have been investigated for use in cosmetics and pharmaceuticals. These monodisperse, unimolecular functional end groups are highly concentrated at the perimeter of symmetrical, unimolecular micellar nanostructures [
33].
A high density of surface functioning is provided by dendrimers, which are symmetrical structures that are clearly defined, consistently branched and have a tree-like shape with branch terminals. Numerous cosmetic products use it because of its size and molecular weight. Lavender and nail polish are comparable. They are beneficial in the fields of artificial tanning, hair, skin, and nail care because of their revolutionary film-forming ability. They can be used to treat acne [
6].
Cubosomes are distinct nanoparticles of the crystal phase that are continuously cubic and liquid with a significantly increased specific surface area than the parent cubic phase. Additionally, it may moisturize the skin and has a high heat stability [
6,
66].
It is created when a certain ratio of water and a microstructure is combined with liquid crystalline particles of a particular surfactant. Due to the affordable monetary value of the basic materials and the possibility of regulating the act of functionalizing release, they are a desirable option for both medicine delivery and cosmetic applications [
3].
Nanospheres are spherical polymeric matrices the size of the particles ranges from 10 to 200 nm. Both biodegradable and non-biodegradable nanospheres are present in various nanoscale carriers, whether crystalline or amorphous. Pure or modified starch, gelatin, and albumin-based nanospheres are examples of biodegradable nanospheres, as opposed to Polylactic acid is an example of a non-biodegradable nanosphere. The skin's outermost layer receives active chemicals when nanospheres are employed in cosmeceutical products for the skin or body [
19].
The drugs or medicine is bound, broken down, adhered to, or encapsulated by nanospheres, which shield it from enzymatic and chemical destruction. To more precisely and effectively deliver active substances to the skin's deeper layers and their therapeutic potential on the affected area, nanospheres are utilized in dermal treatments and cosmetic products [
42].
5. Unique Features of Nanomaterials in Skincare Products
Numerous techniques have already been shown by research on nanotechnology to improve the effectiveness of cosmetics, including 1) enhancing the skin penetration and entrapment effectiveness of the active ingredient, 2) regulating medication release, 3) enhancing physical stability, 4) enhancing moisturizing ability, and 5) enhancing UV protection [
20]. Nanoscale materials frequently have different chemical or physical characteristics from their bigger counterparts, such as elevated biologically harmful action, owing to their small size and peculiarly high surface area-to-volume ratio. This is one of the reasons why nanoscale compounds are becoming more and more popular in the beauty sector [
5].
Table 1.
Nanomaterials and specific properties of nanomaterials used in personal care products.
Table 1.
Nanomaterials and specific properties of nanomaterials used in personal care products.
| Nanomaterials |
Personal Care Products
|
Specific properties of nanomaterials |
References |
| Nanoemulsions |
Nano-based gel
Deodorants
Sunscreens
Shampoos
Lotions
Nail Polishes
Conditioners
Hair Serums
|
Improve the effectiveness of products
As a delivery system
Promote quick penetration
Active transport of active ingredients
Skin hydration
|
[41]
[42]
[6]
|
| Lipid nanoparticles |
Nano repair cream
Lotions
Moisturizer
Anti-wrinkle creams
Sunscreen |
Permits direct proximity to the skin
Enhance skin permeability minimal toxicity
Improve the shelf life of the finished product
Improve bioavailability and site-specific action,
The desired degree of occlusion
Consequent skin hydration active ingredient stabilization
|
[31].
[67]
[43] |
| Nanoparticle |
Sunscreen
Antiaging creams |
Transparency than large particles
Higher degrees of penetration
Immediate effect on skin moisture
|
[6]
[47,33] |
| Nano-capsule |
Hydrogel
Anti-aging creams
Anti-acne drug
Lip balms
UV filters |
Improve quality and effectiveness
Increase the physiochemical stability of ingredients
Stronger antibacterial efficacy
Able to deliver targeted tissue or cells
The active ingredient is shielded from potentially hazardous external influences |
[15]
[50]
[52] |
| Fullerenes |
Sunscreen
Antiaging creams
Anti-wrinkle creams
Eye gels
Moisturizer
|
Antioxidant activity
Penetration in the skin
Able to transport biomolecules
Anti-radical oxygen species (ROS) scavenging abilities
|
[55,56,57]
|
| Nanocrystals |
Toothpaste
Anti-aging cream
Sun protection cream |
Increased membrane penetration
Greater adhesion
A higher concentration of the active ingredients synthesized
Strongest antioxidant (neutralizing) activity |
[60]
[32]
[61] |
The use of nanoparticles in the cosmetic sector aims for long-lasting advantages and improved stability. The extensive surfaces of nanomaterials make it possible to transfer ingredients through the skin more effectively [
28,
51]. The stiffness, hydrophobicity, size, charge, and other physicochemical properties of the nanocarriers have an impact on how well they penetrate the skin. By lowering the dose of medications supplied, the using nanoscaled carriers for medication delivery is anticipated to boost therapeutic specificity and decrease side effects [
38,
68].
In comparison to their parent or bigger size equivalents, nanoscale particles are distinct regarding hue, consistency of the structure, optical activity, transparency, chemical reactivity, and solubility. For instance, the size-matching properties of nanoscale materials with the biological architecture of skin cells provide easier contact, improved solubility, and the selective control of cellular activities at their naturally occurring scale [
4]. Cosmetics made with these nanoparticles have enhanced skin penetration, longer-lasting benefits, and superior UV protection [
1,
3].
Sivanskar and Kumar attempted to calculate the proportion of a spherical micrometer-sized particle that was shrunk to nanometer size and had more surface area [
4]. Increased surface area improves dispersibility (sedimentation rate is reduced to nearly zero) and dissolution rate of drug or cosmetic ingredients. Because nanoscale materials have a bigger surface area and more surface imperfections than bulk materials, more contact points can support van der Waals attraction, which improves adhesion. A better cosmetic outcome will be obtained with improved skin adherence. The surface area has a greater impact on particle reactivity. Different bioactivity could be produced by these distinctive chemical characteristics and may aid in the prediction of potential biological responses obtained from nanomaterials used in cosmetics [
69].
Nanomaterials' optical transparency and activity are advantageous for cosmetic items. This is particularly obvious in numerous lotions and sunscreens. Titanium oxide and zinc oxide are the active chemicals in sunscreen. Their bigger size particles leave a conspicuous white tint on the skin when used, but nanosized particles in comparable products do not, improving the aesthetics of the nanoproduct [
70].
Even materials with a high solid density that is less than a nanometer can be suspended in a liquid without settling because of Brownian motion and form well-dispersed systems. As a result, cosmetics containing nanoscale ingredients will remain suspended for a longer duration than those containing microscale ingredients. This type of nanosized skin preparation has a longer shelf life as a result of this trait since unstable characteristics like creaming, cracking, Ostwald ripening or phase inversion take longer to observe or manifest. Therefore, it should not be shocking that there is a strong desire to acquire and utilize available nanomaterials in the manufacture of cosmetics. The reactivity of materials with nanoscale surface areas is increased because there are more active sites for chemical reactions [
31].
The color cosmetics business was one of the most passionate early users of nanotechnology. Nano-sized materials frequently have different chemical or physical properties from their larger equivalents because of their small dimensions and extraordinarily high surface area-to-volume ratio such as increased unfavorable biological activity. This is one of the reasons why nano-scale materials are already becoming extremely prevalent in the personal care industry. Increased effectiveness, transparency, distinctive consistency, preservation of bioactive substances, and improved attentiveness from customers are a few potential advantages of adding nanoparticles to cosmetics. When viewed at a macroscale, larger particles like Zinc oxide and Titanium dioxide are both white and impenetrable, however, when observed at a nanometer scale, they are translucent. This makes it possible for them to be used in moisturizers and foundation. Wrinkles can be covered by the "soft focus" effect created by aluminum oxide and other nanoparticles. These components may be found in expensive face powders, foundations, and concealer sticks [
5].
As cosmetic items, moisturizers were developed to help cure skin dehydration, which showed up as flaking, scaling, and drying of the skin's surface. Humectants, which are often found in moisturizers, help the skin's surface retain moisture, giving it a smooth, fresh appearance. When the skin is hydrated and supple, atopic dermatitis and pruritus can be avoided and controlled. To increase solubility and add sparkle, cosmetic manufacturers started employing nanoscale materials (like liposomes) as envelopes or carriers for cosmetic chemicals. One such component in cosmetics is vitamin E.
These products' particular benefits are supposed to counteract the problems that cause aging, providing firmness and suppleness to aging skin. The triceramide nano-capsules in Lancôme's Hydrazen cream are said to protect skin from daily stress over the long run while regaining the skin's ideal comfort and softness. Nanosized UV filters and antioxidants are utilized in Caudalie's Vinosun Anti-Aging Suncare and L'Oreal's Plentitude Revitalift, an anti-wrinkle cream that also contains nanoparticles, to regenerate aging skin [
31].
The release of silver ions from silver nanoparticles may be the cause of their antibacterial activity, according to published literature, and comparable studies on nanosilver and its derivatives have been carried out extensively to take advantage of its special antibacterial and antifungal capabilities [
71,
72]. However, because of their special capabilities for loading and unloading medications as well as their simplicity in production, gold nanoparticles have potential use in cosmetics. They can be conjugated with chemicals from cosmetic formulations to raise the caliber of the final product because they are readily functionalized, often via thiol linkages. According to studies, toothpaste, and other commercially available personal care products now contain nanogold to generate excellent oral cleansing [
16,
31].
As their names suggest, UV filters and sunscreens shield the skin from the sun's damaging ultraviolet rays. In UV filters, the two main materials used as nanoparticles are zinc oxide (ZnO) and titanium dioxide (TiO). Even though certain organic substitutes (such as those produced by ivy plants) are being investigated, the nanoparticles of these two oxides have been widely used in sunscreens to prevent UV damage to the skin. They reflect UV-A and UV-B radiation while more effectively absorbing UV light than their bigger particle-sized competitors as a unique quality. TiO nanoparticle-based sunscreen formulations have fewer oily, translucent, pungent, and visually attractive characteristics. They also don't leave behind the same white residue on the skin as their counterparts with bigger particle sizes do, which is chalky in appearance. These ivy nanoparticles protect UV rays four times better than metallic oxide nanoparticles, according to studies. Sunforgettable powder by Colorescience, which contains titanium dioxide nanoparticles, is a nice illustration [
31].
Broad-spectrum antibacterial action is present in silver nanoparticles, which is especially effective against bacteria. Silver nanoparticles are believed to be especially efficient for skin protection and as an antiseptic, in organic Korean soap and hand sanitizer. It is asserted that even Evolut is hypoallergenic and increases resistance to airborne diseases [
31].
A unique set of multifunctional features, including a high surface-to-volume ratio, stability, catalytic performance, electromagnetic properties, and biological activities, are provided by effective nanoparticle deployment. The properties of nanoparticles described above, in particular, allow for boosting the comprehensive functionality of the product of interest [
30]. To capitalize on the benefits of activities involving form and size, cosmetics are frequently made with nanoparticles of various morphologies and chemical contents [
73,
74], which can improve product performance. Due to their capacity to deflect and refract UV rays, many different forms of inorganic oxide-based nanoparticles, including TiO2, ZrO2, ZnO, and CeO2, are utilized as physical UV filters in sunscreen preparations as opposed to UV absorbers. Due to the UV radiation attenuation capabilities of these nanostructured materials, their resistance to UV light is increased [
19,
75].
Recent findings, Haddada et al. examined the safety of gold nanoparticles as a cosmetic additive by evaluating their antioxidant and dermoprotective effects [
76]. Because they are inert, non-cytotoxic, biocompatible, and extremely stable, gold nanoparticles are remarkable for these qualities. Gold nanoparticles have grown in popularity because of their effective antibacterial and antifungal capabilities. Due to their effective antibacterial and antifungal qualities, gold nanoparticles have become more popular. These nanoparticles are found in a variety of cosmeceutical treatments, such as face packs, deodorants, creams, lotions, and anti-aging creams. The fact that lipid nanoparticles are made from disposable and physiological lipids makes them a "nanoscale" carrier technique with low toxicity/cytotoxicity and notable tolerance [
46].
Lipid nanoparticles can also increase the chemical stability of materials that are sensitive to light, degradation, and hydrolysis. Through inclusion into the lipid nanoparticle drug delivery system, many cosmetic ingredients, including Coenzyme Q10, vitamin E, ascorbyl palmitate, and vitamin A, have increased their chemical stability [
77]. Intrinsic viscosity is a crucial dendrimer characteristic that makes them valuable in cosmetic formulations. L'Oreal has combined film-forming polymers with terminal hydroxyl functionalized polyester dendrimers in cosmetic formulations for skin applications. The potency of many L'Oreal cosmetics products has grown due to the introduction of dendrimers [
19,
78].
TiO
2 and ZnO nanoparticles meet consumer needs better than micron-sized particles because they produce a translucent effect by reflecting only a small amount of the light particles that are incident [
79,
80]. ZnO and TiO
2 nanoparticles as small as 20 nm are also widely used in sunscreens as UV filters. They have superior dispersion and produce a more attractive cosmetic outcome [
51]. The primary benefit of nanoparticles is that they release the active component in the outermost skin layers, guaranteeing that substantial concentrations of this chemical enter the epidermis [
20,
81].
For cellular distribution, inorganic nanoparticles are well suited due to their vast availability, rich functionality, strong biocompatibility, possibility for targeted administration, and regulated drug administration [
58]. These are effective for the transdermal distribution of chemicals as well as for the dermatological treatment of disorders like vitiligo and skin cancer. They are also used in the cosmetics sector for therapies for both acne and aging, as well as for hydration and skin care products [
38].
Due to their hydrophilic surface, which encourages longer circulation, and their low production cost, silica nanoparticles have caught the attention of the cosmetic industry [
82]. The effectiveness, smoothness, and shelf life of cosmetic products are all improved by the use of nano-silica. It functions as an anti-caking agent and enhances absorbency [
51].
Nanometer-sized solid lipid nanoparticles (SLNs) have a solid lipid matrix that maintains its form at body temperature [
83], which preserves its structure at body temperature. SLNs feature an oily or lipoid core and a single layer of shells. Since SLNs are tiny, they come into close contact with the stratum corneum, allowing for greater ingredient penetration into the skin [
46]. They can aid in boosting the water content of the skin while also acting as potential UV blockers. Even though nanostructured lipid carriers (NLCs) can carry more active substances, SLNs can increase chemical stability. And they are easily scalable, and their production methods are well-established. Also, they improve the ability to penetrate the skin, to be biocompatible, and to be stable as a carrier delivery agent [
22]. It is possible to produce SLNs and NLCs both in the lab and on a large scale, and they are highly tolerated carrier systems for dermal applications [
46].
As a result of their tiny size and low-toxicity physiological and disposable lipid composition, they remain on the skin's surface and facilitate the dispersion of different compounds within the body, SLNs are widely used in cosmetics. SLNs also have occlusive properties that contribute to increased skin hydration [
8,
46].
The more recent nanoscale materials NLCs and SLNs distribute and stabilize more effectively than liposomes. Because of their high bioavailability and controlled occlusion when used in moisturizers, the skin is better hydrated. Nanogold is also said to have healing and anti-oxidant properties and is used in one moisturizer available in the UK. A moisturizer branded Hydra Flash Bronzer Daily Face from the cosmetics company Lancôme claims to provide customers with a natural, glowing complexion because it contains nanocapsules of vitamin E [
31].
The capacity to prevent encapsulated chemicals from degrading, controlled release, and improved penetration through the stratum corneum are only a few benefits of using these nanomaterials in cosmetic products (coenzyme Q10 and retinol) [
83,
84]. Because of their occlusive properties, they also increase skin hydration [
29].
For dermal application, NLC and SLN both have many benefits. They are colloidal carriers that offer a variety of chemical-regulated release characteristics. Due to its diminutive size, which ensures direct contact with the stratum corneum and could boost the amount of medication absorbed by the skin. Skin hydration rises as a result of the occlusive features of lipid nanoparticles. Furthermore, materials that are susceptible to oxidation, UV, and hydrolysis can have their chemical stability increased by the addition of lipid nanoparticles [
46].
Labile Active Pharmaceutical Ingredients can be protected from hydrolysis and oxidation by the solid matrix of SLNs. This is important for anti-aging and minor acne topical agents. Comparing SLN to an aqueous dispersion, tocopherol, retinol, and coenzyme Q10's chemical stability is increased by 57% [
85] and tocopherol by 57%. [
21].
SLNs have several advantageous properties, including: a) the existence of biodegradable, low-toxic, physiological lipids b) Obstructive characteristics that enhance skin moisture levels, and c) greater uptake of active substances due to small size-like properties. Because of these properties, they have become popular in cosmetics [
46,
86].
Skin thinning, loss of suppleness, dryness, wrinkles, the emergence of spots, and the loss of the skin's barrier function are all signs of aging skin. Accelerated aging has been associated with exposure to chemical contaminants, UV and infrared radiation, abrasion, and stress. Skin regeneration and replenishment, on the other hand, are dependent on the level of skin's collagen quality. Certain skin care products contain proteins made from progenitor cells that are wrapped in nanoparticles called liposomes that also contain anti-aging ingredients. This allows for effective protein delivery [
67]. When creating anti-aging cosmetics, some people incorporate retinoids and botulinum toxins into nanoscale materials to promote skin renewal.
Encapsulation with liposomes (some manufacturers call them "nanosomes") preserves the medication against metabolic deterioration and allows for the controlled release of its bioactive constituents [
87]. These are appropriate for delivering pharmacologically active substances with both hydrophobic and hydrophilic characteristics. Their sizes range between 20 nanometers and many micrometers, which determines how effective they are at targeting, and they may have a unilamellar or multilamellar structure [
42]. Antioxidants like lycopene or carotenoids can be added to liposomes to increase their physical and chemical stability in water [
19]. Cosmetics containing liposomes have a high substantivity, making them difficult to wash away. Liposomes, in particular, can increase the lipid content of the stratum corneum, increasing moisturization and decreasing skin dryness [
20,
81].
Liposomes are widely used in the cosmetic industry for a variety of reasons, including their ease of preparation, safety, structural similarity to the skin, biocompatibility, the capacity to enhance active component penetration through the skin, and the viability of directing the entrapped chemicals to particular skin layers while the skin moisturizing and restorative benefits of their inherent lipids and limiting negative effects caused by systemic absorption. Furthermore, liposomes may establish an active chemical reservoir in the skin and its appendages, providing a sustained release that may be effective in extending the effects of active compounds [
55].
Several scientists hypothesized that making liposomes more elastic would enhance medication absorption through the skin. In addition, leakage-resistant flexible liposomes were developed to address the stiffness and stability issues that are common with traditional liposomes [
32]. The cream Capture, introduced by Dior in 1986, was the first cosmetic product to use liposomes [
3]. The utilization of liposomes is increasing due to their simplicity in manufacture and capacity to boost the skin's ability to absorb bioactive components, despite their instability and also the requirement for antioxidants, cryoprotectants, and other additions [
29].
Liposomes are appropriate for skin care applications because of their ease of manufacture, improved absorption of active compounds via the skin, and protracted delivery of chemicals into cells [
3]. The distribution of both hydrophobic and hydrophilic substances is possible with the help of liposomes, which are non-hazardous, biocompatible, and able to easily encapsulate active ingredients. Both hydrophilic and hydrophobic substance transport can be made more appropriate with the help of liposomes. And increased concentrations of drug products are possible with liposomes, by improving their absorption by the skin [
6].
To improve penetration through the skin barrier, proteins are mixed with nanoliposomes and nanocapsule particles for usage in cosmetic applications [
88]. Because of their water-containing structure, nanoliposome particles can moisturize skin; after liposomes penetrate the skin's stratum corneum layer, and the liposomes' inner water core serves as a source of hydration for the skin. [
48].
Liposomes are relatively small spheres having an outer shell comprised of both hydrophilic and lipophilic materials. They mostly consist of water and active substances that are soluble in water, such as vitamins. They are minuscule (less than 1/10,000 mm) and have a cell-like exterior structure, which allows them to penetrate the skin [
89].
Typically, liposomes have a size range of 20 nanometers to several hundred microns. These are essentially spherical vesicles that have a phospholipid bilayer trapping some of the solvents. They are appropriate for the administration of both hydrophilic and hydrophobic substances due to their capacity to shield the drug from the outside environment. Due to its softening and conditioning qualities, phosphatidylcholine, one of the liposome's essential components, has been found in a variety of skin care formulas and hair care products [
60]. Due to the biocompatibility, biodegradability, and nontoxicity of liposomes, as well as the ease with which they encapsulate the active component, liposomes are employed in several cosmeceuticals [
60,
90].
Nanoemulsions have desirable attributes such as a broad excellent solubilization capacity, high kinetic stability, interfacial area, and low viscosity [
91]. Smaller-sized particles of nanoemulsions promote applicability and stability for carrying active ingredients; they also boost the longevity of the product's shelf life [
40,
92]. These are widely used as a delivery medium for a variety of cosmetic products, including nail polish, lotions, shampoos, conditioners, sunscreens, deodorants, and hair serums [
93].
Because of their interior lipophilic, nanoemulsions are better suited for transporting lipophilic compounds than liposomes. They, like liposomes, help active ingredients penetrate the skin and consequently boost their skin concentration. The high surface area of the little droplet, which enables efficient active transport to the skin, is another benefit [
27].
The following characteristics explain why a nanoemulsion is superior to a macroemulsion broad surface area, enhanced inclusiveness, skin film generation, excellent stability, modified release and drug-delivery capacities, attractive visual character, and skin feel [
94]. Liposomes are less stable than nanoemulsions, and occasionally a lamellar liquid-crystalline phase might develop surrounding the droplets [
37,
95].
Nanoemulsions in cosmeceutical products allow components to penetrate the skin quickly. As a result of the tiny droplet size, the potential benefits of using nanoemulsions in cosmetic products include (i) strong occlusive action of keeping skin hydrated, (ii) rapid skin penetration of active ingredients, (iii) visual aspects and skin with a gleaming sheen, (iv) excessive fluidity without gelation, and (v) enhanced diffusion into small gaps like hair scale spacings and pilosebaceous follicles [
96].
Active chemicals can swiftly infiltrate the skin thanks to the nanoemulsion formulation's enormous surface area of the droplets. Nanoemulsions have even been found to be easily able to penetrate tough skin. Because of this nanoemulsion feature, it is no longer necessary to use a particular penetration enhancer, which is what causes formulation incompatibility [
93].
In recent years, nanoemulsions have become more well-known as prospective delivery systems for cosmetics and as a means of improving the distribution of active ingredients in certain skin layers. Transporting lipophilic compounds is better suited with nanoemulsions than liposomes due to their lipophilic interior. They increase the concentration of active chemicals in the skin by facilitating their absorption into the skin, similar to liposomes [
27,
93].
High fluidity, low viscosity, homogeneity, high solubilization capacity, low sedimentation, and thermal stability are characteristics of nanoemulsions [
97]. Inserting chemicals that are physiologically active into them is relatively simple. This method is preferred due to the improvement in the efficacy of the components in nanoemulsions. They have been used in skin care products for aged skin due to their properties [
98].
The enhanced methods for dermal creams and gels to penetrate the skin are being provided by nano-based compositions. It may be possible for dendrimers to successfully distribute dermal formulations via the skin [
28,
65].
The dendrimer structure has assisted in increasing resveratrol's systemic absorption and skin absorption (It is well-known for its antioxidant and anti-aging qualities.)[
99], resulting in the subsequent scaling up and commercialization of this product with a dendrimer structure [
51]. Dendrimers have the potential to improve bioavailability and drug solubility because of the possibility that molecules could be trapped by their hyper-branched structure and molecules that attach to their surface. The main factor driving dendrimers' strong demand in medication delivery systems and product development is due to their qualities [
65].
Carbosiloxane dendrimer, according to Furukawa et al., can give the skin a healthy quantity of glossiness and sebum, as well as sensory perception, water repellency, and adhering characteristics [
100]. Dendrimers' intrinsic viscosity is a crucial aspect that might accentuate them as a significant benefit for cosmetic innovators. And because of the nonlinear relationship between dendrimer molecular weight and inherent viscosity [
65]. Dendrimers cover a significant portion of the cosmeceutical fields [
8].
Although they are formed of artificial, non-ionic surfactants, niosomes are shaped similarly to liposomes [
101]. Lancome introduced the first Niosome product to the market in 1987. Some of the benefits of using niosomes in cosmetics include their ability to improve the incorporated active ingredient's stability and raise the rate of skin absorption [
29].
Niosomes can transport substances that are both hydrophilic and hydrophobic. Niosomes can be used as a novel medication administration system for poorly absorbed medicines [
102]. Because they encapsulate the drug, niosomes can be used as a vehicle for poorly absorbable drugs [
102]. It encapsulates the drug, extending its time in the systemic circulation and increasing entry into the target tissue. Niosomes eliminate the problems of liposomes, such as inconsistency, expensive demand, and vulnerability to oxidation [
42,
103].
Cubosomes have a substantial surface area, a low viscosity, and the ability to subsist with virtually any level of dilution. They are very thermostable and are capable of carrying both hydrophobic and hydrophilic compounds [
3].
Because of its tiny pore size (5—10 nm), ability to hydrolyze hydrophilic, hydrophobic, and amphiphilic components, and biodegradability by simple enzyme activity, the cubic phase of cubosomes is appealing for controlled release. The cubic phase, which is exceptionally bioadhesive, is thought to be a skin penetration booster because of its exceptional tolerance to topical and mucosal accumulation and active ingredient administration [
67].
Depending on whether or not their surfaces have a formal charge, nanocapsules are categorized as ionic or non-ionic. The inclusion of charge can boost the hydrolysis pressure at the surface of nanocapsules, encouraging repulsion among suspended particles and decreasing cluster formation [
26,
104].
The use of nanocapsules has the advantage of the targeted tissue or cells being delivered and releasing active substances under controlled conditions. As opposed to that, applying too many direct application of biologically active substances to the skin may impair the effectiveness of the medicine or cosmetic product. The active ingredient is shielded from potentially hazardous external influences like moisture or oxygen thanks to this nanoformulation, which is still another benefit [
98,
105].
Nanocrystals have higher kinetic saturation solubility due to their nano-dimension size, which, when combined with the large surface area, results in rapid dissolution. For rutin nanocrystals developed for oral administration, these effects have already been described [
62,
106].
Lipid particles provide an adhesion that covers the skin surface and prevents water from evaporating from the lipid nanodispersion that has been applied to it. As a result of decreased corneocyte packing and increased inter-corneocyte gaps, the stratum corneum's moisture may rise, enabling medication penetration into deeper skin layers [
85,
107].
The superficial connections of corneocyte clusters and the furrows between corneocyte islands can be closely contacted by small particles, facilitating sustained drug release over several hours [
108]. While this is also true for liposomes, they differ in that they fall apart, which is thought to compromise percutaneous penetration [
107]. Because of their strong adhesive properties, the application site's SLN and NLC should endure longer. Adhesion increases as particle size decreases. Furthermore, in the lipid matrix of the carrier, the medicine is dissolved or finely disseminated, or it sticks to the surface of the carrier, allowing contact with the outermost skin layers [
21].
Chemical-specific receptors are also attached to nanocarriers, allowing them to reach any desired location in the body [
101]. This increases the effectiveness of the active ingredients while decreasing the possibility of side effects [
109]. Active ingredients like hyaluronic acid, antioxidants, vitamins, and different types of peptides are frequently transferred via nanocarriers in moisturizing lotions, nutrient preparations, and sunscreens [
88,
98].
NanoCarriers are particularly advantageous as cosmetic systems because they can improve cosmetic product delivery perceived or measured performance. Because NCs have numerous benefits over conventional topical formulations, such as improved active chemical stability and water solubility, greater bioavailability targeting specific skin layers, and so on, these carriers may help boost anti-oxidant topical efficiency. In particular, liposomes, niosomes, solid lipid nanoparticles, and so forth [
55].
Natural inertness, extreme stability, biocompatibility, and noncytotoxicity characterize gold nanoparticles. After membrane staining, Nanogold is extremely stable in liquid or dried form and does not fade; it is also offered in conjugated and unconjugated forms [
110]. Their small size, enormous surface area, shape, and crystallinity make them easy to move into the target cell and provide them with a high drug-loading capacity [
111].
UV radiation can be reflected, absorbed, or dispersed by nanoparticles of compounds such as zinc oxide (ZnO) and titanium dioxide (TiO2). According to reports, nanoparticles can reach the tops of human hair follicles or the Horny layer, but they are incapable of passing through intact skin and reaching the critical epidermal layers [
112,
113]. Liposomes and SLNs, for example, have been used as penetration enhancers in various sunscreen formulations [
114,
115].
When compared to microparticles of the same chemical, nanomaterials may have different biological characteristics since a considerable portion of their atoms are on the surface of the particle. These distinct chemical properties may result in distinctive bioactivity [
69]. Therefore, it is crucial to comprehend which chemical characteristics may be crucial for foretelling a biological response to a nanomaterial [
1].