Submitted:
03 June 2024
Posted:
04 June 2024
You are already at the latest version
Abstract

Keywords:
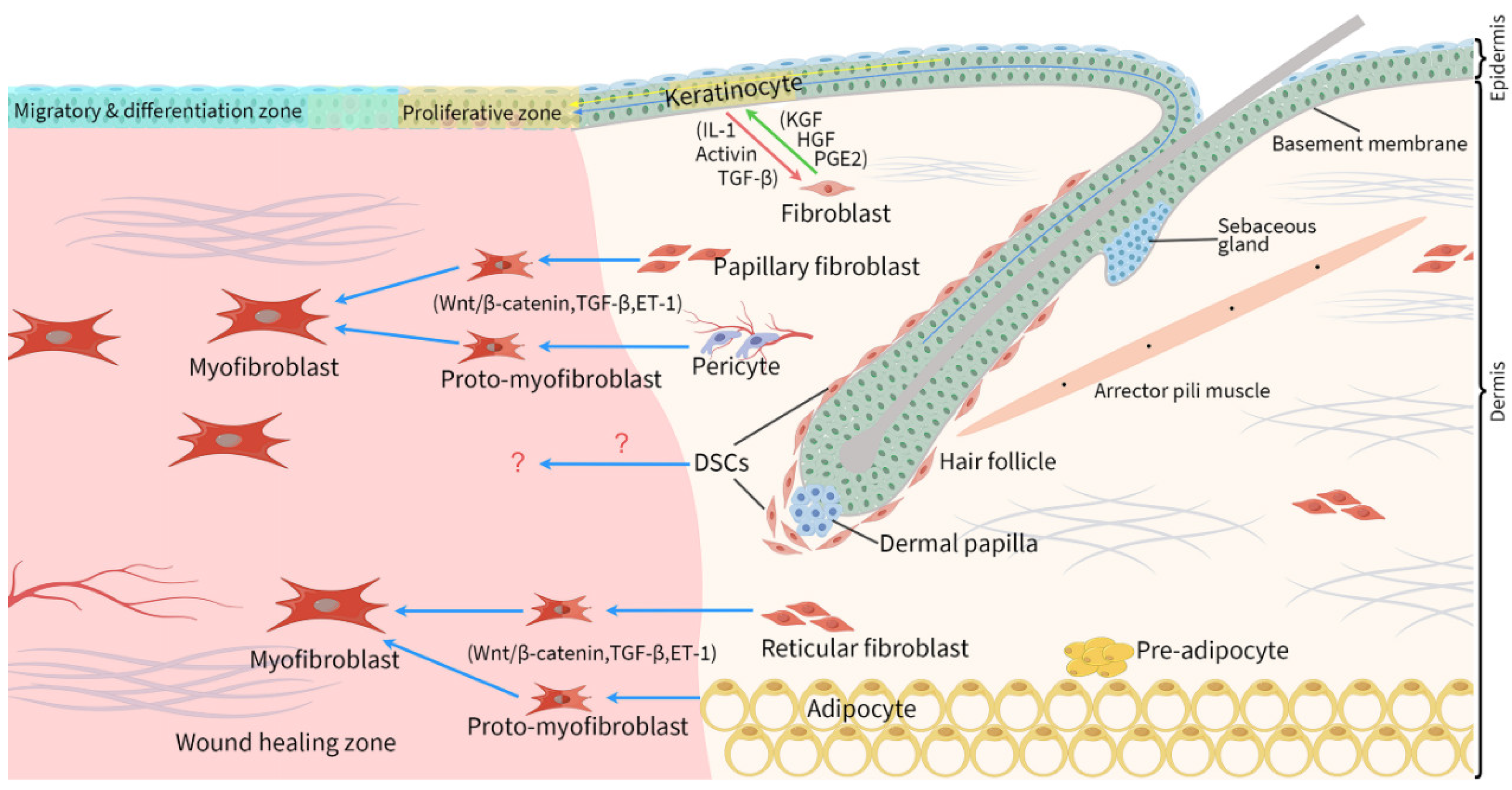
 This review brings to light the significant yet often overlooked role of dermal sheath cells (DSCs) in wound healing. It emphasizes their multifunctional roles in inflammation modulation, proliferation aid, and ECM remodeling, and illuminates DSCs’ paracrine effects and their involvement in fibrosis, offering a new perspective on skin repair processes.
This review brings to light the significant yet often overlooked role of dermal sheath cells (DSCs) in wound healing. It emphasizes their multifunctional roles in inflammation modulation, proliferation aid, and ECM remodeling, and illuminates DSCs’ paracrine effects and their involvement in fibrosis, offering a new perspective on skin repair processes.Introduction
Background
Dermal Sheath Cells: Characteristics and Heterogeneity
Modulatory Potential of Dermal Sheath Cells in Inflammatory Responses
Dermal Sheath Cells Are Activated in the Proliferation Phase
Dermal Sheath Cells may Contribute to Angiogenesis and Vascularization
Extracellular Matrix Remodeling and Dermal Sheath Cells
Paracrine Effects of Dermal Sheath Cells
Potential Role of Dermal Sheath Cells in Fibrosis
Dermal Sheath Cells Work Differently in Healing Hairy and Non-Hairy Sites
Conclusions and Future Challenges
Author Contributions
Funding
Conflicts of Interest
References
- Driskell, R.R.; Lichtenberger, B.M.; Hoste, E.; Kretzschmar, K.; Simons, B.D.; Charalambous, M.; Ferron, S.R.; Herault, Y.; Pavlovic, G.; Ferguson-Smith, A.C.; et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 2013, 504, 277–281. [Google Scholar] [CrossRef]
- Tabib, T.; Huang, M.; Morse, N.; Papazoglou, A.; Behera, R.; Jia, M.; Bulik, M.; Monier, D.E.; Benos, P.V.; Chen, W.; et al. Myofibroblast transcriptome indicates SFRP2hi fibroblast progenitors in systemic sclerosis skin. Nat. Commun. 2021, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ascensión, A.M.; Fuertes-Álvarez, S.; Ibañez-Solé, O.; Izeta, A.; Araúzo-Bravo, M.J. Human Dermal Fibroblast Subpopulations Are Conserved across Single-Cell RNA Sequencing Studies. J. Investig. Dermatol. 2020, 141, 1735–1744. [Google Scholar] [CrossRef]
- Williams R, Thornton MJ. Isolation of different dermal fibroblast populations from the skin and the hair follicle. Molecular Dermatology: Methods and Protocols 2020;13-22.
- Jahoda CA, Reynolds AJ. Hair follicle dermal sheath cells: unsung participants in wound healing. The Lancet 2001;358:1445-1448.
- Ahlers, J.M.D.; Falckenhayn, C.; Holzscheck, N.; Solé-Boldo, L.; Schütz, S.; Wenck, H.; Winnefeld, M.; Lyko, F.; Grönniger, E.; Siracusa, A. Single-Cell RNA Profiling of Human Skin Reveals Age-Related Loss of Dermal Sheath Cells and Their Contribution to a Juvenile Phenotype. Front. Genet. 2022, 12, 797747. [Google Scholar] [CrossRef]
- Plikus MV, Wang X, Sinha S, Forte E, Thompson SM, Herzog EL, Driskell RR, Rosenthal N, Biernaskie J, Horsley V. Fibroblasts: Origins, definitions, and functions in health and disease. Cell 2021;184:3852-3872.
- Tao Y, Yang Q, Wang L, Zhang J, Zhu X, Sun Q, Han Y, Luo Q, Wang Y, Guo X, Wu J, Li B, Yang X, He L, Ma G. beta-catenin activation in hair follicle dermal stem cells induces ectopic hair outgrowth and skin fibrosis. J Mol Cell Biol 2019;11:26-38.
- Martino, P.A.; Heitman, N.; Rendl, M. The dermal sheath: An emerging component of the hair follicle stem cell niche. Exp. Dermatol. 2020, 30, 512–521. [Google Scholar] [CrossRef]
- Sharma, R.K.; John, J.R. Role of stem cells in the management of chronic wounds. Indian J. Plast. Surg. 2012, 45, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Barker, N.; Bartfeld, S.; Clevers, H. Tissue-Resident Adult Stem Cell Populations of Rapidly Self-Renewing Organs. Cell Stem Cell 2010, 7, 656–670. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Park E, Lee SM, Jung I-K, Lim Y, Kim J-H. Effects of genistein on early-stage cutaneous wound healing. Biochemical and biophysical research communications 2011;410:514-519.
- Nurden AT, Nurden P, Sanchez M, Andia I, Anitua E. Platelets and wound healing. Front Biosci 2008;13:3532-48.
- A Lenselink, E. Role of fibronectin in normal wound healing. Int. Wound J. 2013, 12, 313–316. [Google Scholar] [CrossRef]
- Nandi, S.; Sommerville, L.; Nellenbach, K.; Mihalko, E.; Erb, M.; Freytes, D.O.; Hoffman, M.; Monroe, D.; Brown, A.C. Platelet-like particles improve fibrin network properties in a hemophilic model of provisional matrix structural defects. J. Colloid Interface Sci. 2020, 577, 406–418. [Google Scholar] [CrossRef]
- Ross R, Odland G. Human wound repair: II. Inflammatory cells, epithelial-mesenchymal interrelations, and fibrogenesis. The Journal of cell biology 1968;39:152-168.
- Barbul, A.; Shawe, T.; Rotter, S.; Efron, J.; Wasserkrug, H.; Badawy, S. WOUND-HEALING IN NUDE-MICE - A STUDY ON THE REGULATORY ROLE OF LYMPHOCYTES IN FIBROPLASIA. 1989, 105, 764–769.
- Barbul, A.; Breslin, R.J.; Woodyard, J.P.; Wasserkrug, H.L.; Efron, G. The Effect of In Vivo T Helper and T Suppressor Lymphocyte Depletion on Wound Healing. Ann. Surg. 1989, 209, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Artuc M, Hermes B, Stckelings U, Grützkau A, Henz B. Mast cells and their mediators in cutaneous wound healing–active participants or innocent bystanders? Experimental dermatology 1999;8:1-16.
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: a critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.E.; Minasian, R.A.; Caterson, E. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv. Wound Care 2016, 5, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Y.; Kirsner, R.S. Angiogenesis in wound repair: Angiogenic growth factors and the extracellular matrix. Microsc. Res. Tech. 2002, 60, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Raja KS, Garcia MS, Isseroff RR. Wound re-epithelialization: modulating keratinocyte migration in wound healing. Front Biosci-Landmrk 2007;12:2849-2868.
- Tang, F.; Li, J.; Xie, W.; Mo, Y.; Ouyang, L.; Zhao, F.; Fu, X.; Chen, X. Bioactive glass promotes the barrier functional behaviors of keratinocytes and improves the Re-epithelialization in wound healing in diabetic rats. Bioact. Mater. 2021, 6, 3496–3506. [Google Scholar] [CrossRef] [PubMed]
- Dekker, A.D.; Davis, F.M.; Kunkel, S.L.; Gallagher, K.A. Targeting epigenetic mechanisms in diabetic wound healing. Transl. Res. 2018, 204, 39–50. [Google Scholar] [CrossRef]
- Darby, I.A.; Laverdet, B.; Bonté, F.; Desmouliere, A. Fibroblasts and myofibroblasts in wound healing. Clin. Cosmet. Investig. Dermatol. 2014, 7, 301–311. [Google Scholar] [CrossRef]
- Rippa, A.L.; Kalabusheva, E.P.; Vorotelyak, E.A. Regeneration of Dermis: Scarring and Cells Involved. Cells 2019, 8, 607. [Google Scholar] [CrossRef]
- Schmidt, B.A.; Horsley, V. Intradermal adipocytes mediate fibroblast recruitment during skin wound healing. Development 2013, 140, 1517–1527. [Google Scholar] [CrossRef]
- Rousselle, P.; Braye, F.; Dayan, G. Re-epithelialization of adult skin wounds: Cellular mechanisms and therapeutic strategies. Adv. Drug Deliv. Rev. 2018, 146, 344–365. [Google Scholar] [CrossRef]
- Werner, S.; Krieg, T.; Smola, H. Keratinocyte–Fibroblast Interactions in Wound Healing. J. Investig. Dermatol. 2007, 127, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Russo, B.; Brembilla, N.C.; Chizzolini, C. Interplay Between Keratinocytes and Fibroblasts: A Systematic Review Providing a New Angle for Understanding Skin Fibrotic Disorders. Front. Immunol. 2020, 11, 648. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira RC, Wilson SE. Fibrocytes, Wound Healing, and Corneal Fibrosis. Invest Ophthalmol Vis Sci 2020;61:28.
- Hsu, P.W.; Salgado, C.J.; Kent, K.; Finnegan, M.; Pello, M.; Simons, R.; Atabek, U.; Kann, B. Evaluation of porcine dermal collagen (Permacol) used in abdominal wall reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2009, 62, 1484–1489. [Google Scholar] [CrossRef] [PubMed]
- Midwood, K.S.; Williams, L.V.; Schwarzbauer, J.E. Tissue repair and the dynamics of the extracellular matrix. Int. J. Biochem. Cell Biol. 2004, 36, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Hade, M.D.; Suire, C.N.; Mossell, J.; Suo, Z. Extracellular vesicles: Emerging frontiers in wound healing. Med. Res. Rev. 2022, 42, 2102–2125. [Google Scholar] [CrossRef]
- Xue, M.; Jackson, C.J. Extracellular Matrix Reorganization During Wound Healing and Its Impact on Abnormal Scarring. Adv. Wound Care 2015, 4, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Heitman, N.; Sennett, R.; Mok, K.-W.; Saxena, N.; Srivastava, D.; Martino, P.; Grisanti, L.; Wang, Z.; Ma’ayan, A.; Rompolas, P.; et al. Dermal sheath contraction powers stem cell niche relocation during hair cycle regression. Science 2019, 367, 161. [Google Scholar] [CrossRef] [PubMed]
- Agabalyan NA, Hagner A, Rahmani W, Biernaskie J. SOX2 in the skin. In: Sox2. Elsevier: 2016; 281-300.
- Shin, W.; Rosin, N.L.; Sparks, H.; Sinha, S.; Rahmani, W.; Sharma, N.; Workentine, M.; Abbasi, S.; Labit, E.; Stratton, J.A.; et al. Dysfunction of Hair Follicle Mesenchymal Progenitors Contributes to Age-Associated Hair Loss. Dev. Cell 2020, 53, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yang, K.; Wickett, R.R.; Andl, T.; Zhang, Y. Dermal sheath cells contribute to postnatal hair follicle growth and cycling. J. Dermatol. Sci. 2016, 82, 129–131. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Inamatsu, M.; Yoshizato, K. The upper dermal sheath has a potential to regenerate the hair in the rat follicular epidermis. Differentiation 1996, 60, 287–297. [Google Scholar] [CrossRef]
- Rahmani, W.; Abbasi, S.; Hagner, A.; Raharjo, E.; Kumar, R.; Hotta, A.; Magness, S.; Metzger, D.; Biernaskie, J. Hair Follicle Dermal Stem Cells Regenerate the Dermal Sheath, Repopulate the Dermal Papilla, and Modulate Hair Type. Dev. Cell 2014, 31, 543–558. [Google Scholar] [CrossRef] [PubMed]
- Hernaez-Estrada, B.; Gonzalez-Pujana, A.; Cuevas, A.; Izeta, A.; Spiller, K.L.; Igartua, M.; Santos-Vizcaino, E.; Hernandez, R.M. Human Hair Follicle-Derived Mesenchymal Stromal Cells from the Lower Dermal Sheath as a Competitive Alternative for Immunomodulation. Biomedicines 2022, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Soma, T.; Kishimoto, J. Characterization of human dermal sheath cells reveals CD36-expressing perivascular cells associated with capillary blood vessel formation in hair follicles. Biochem. Biophys. Res. Commun. 2019, 516, 945–950. [Google Scholar] [CrossRef]
- Abbasi, S.; Biernaskie, J. Injury modifies the fate of hair follicle dermal stem cell progeny in a hair cycle-dependent manner. Exp. Dermatol. 2019, 28, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Kua, J.E.H.; Lim, W.K.; Lee, S.T.; Chua, A.W.C. In vitro characterization of human hair follicle dermal sheath mesenchymal stromal cells and their potential in enhancing diabetic wound healing. Cytotherapy 2015, 17, 1036–1051. [Google Scholar] [CrossRef] [PubMed]
- Gharzi, A.; Reynolds, A.J.; Jahoda, C.A.B. Plasticity of hair follicle dermal cells in wound healing and induction. Exp. Dermatol. 2003, 12, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Liu, G.; Halim, A.; Ju, Y.; Luo, Q.; Song, G. Mesenchymal Stem Cell Migration and Tissue Repair. Cells 2019, 8, 784. [Google Scholar] [CrossRef] [PubMed]
- Diller, R.B.; Tabor, A.J. The Role of the Extracellular Matrix (ECM) in Wound Healing: A Review. Biomimetics 2022, 7, 87. [Google Scholar] [CrossRef] [PubMed]
- Ather S, Harding K, Tate S. Wound management and dressings. In: Advanced textiles for wound care. Elsevier: 2019; 1-22.
- Fielding, C.A.; McLoughlin, R.M.; McLeod, L.; Colmont, C.S.; Najdovska, M.; Grail, D.; Ernst, M.; Jones, S.A.; Topley, N.; Jenkins, B.J. IL-6 Regulates Neutrophil Trafficking during Acute Inflammation via STAT3. J. Immunol. 2008, 181, 2189–2195. [Google Scholar] [CrossRef]
- Lin, Z.-Q.; Kondo, T.; Ishida, Y.; Takayasu, T.; Mukaida, N. Essential involvement of IL-6 in the skin wound-healing process as evidenced by delayed wound healing in IL-6-deficient mice. J. Leukoc. Biol. 2003, 73, 713–721. [Google Scholar] [CrossRef]
- Bernhard, S.; Hug, S.; Stratmann, A.E.P.; Erber, M.; Vidoni, L.; Knapp, C.L.; Thomaß, B.D.; Fauler, M.; Nilsson, B.; Ekdahl, K.N.; et al. Interleukin 8 Elicits Rapid Physiological Changes in Neutrophils That Are Altered by Inflammatory Conditions. J. Innate Immun. 2021, 13, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Zhang, Q.; Cao, Q.; Kong, R.; Xiang, X.; Liu, H.; Feng, M.; Wang, F.; Cheng, J.; Li, Z.; et al. Liver tumour immune microenvironment subtypes and neutrophil heterogeneity. Nature 2022, 612, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, W.; Sinha, S.; Biernaskie, J. Immune modulation of hair follicle regeneration. npj Regen. Med. 2020, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Abbasi S, Sinha S, Labit E, Rosin NL, Yoon G, Rahmani W, Jaffer A, Sharma N, Hagner A, Shah P, Arora R, Yoon J, Islam A, Uchida A, Chang CK, Stratton JA, Scott RW, Rossi FMV, Underhill TM, Biernaskie J. Distinct Regulatory Programs Control the Latent Regenerative Potential of Dermal Fibroblasts during Wound Healing. Cell Stem Cell 2021;28:581-583.
- Tobin, D.J.; Magerl, M.; Gunin, A.; Handijski, B.; Paus, R. Plasticity and Cytokinetic Dynamics of the Hair Follicle Mesenchyme: Implications for Hair Growth Control. J. Investig. Dermatol. 2003, 120, 895–904. [Google Scholar] [CrossRef]
- Chi, W.Y.; Enshell-Seijffers, D.; Morgan, B.A. De Novo Production of Dermal Papilla Cells during the Anagen Phase of the Hair Cycle. J. Investig. Dermatol. 2010, 130, 2664–2666. [Google Scholar] [CrossRef]
- Feutz, A.-C.; Barrandon, Y.; Monard, D. Control of thrombin signaling through PI3K is a mechanism underlying plasticity between hair follicle dermal sheath and papilla cells. J. Cell Sci. 2008, 121, 1435–1443. [Google Scholar] [CrossRef]
- Higgins, C.; Roger, M.; Hill, R.; Ali-Khan, A.; Garlick, J.; Christiano, A.; Jahoda, C. Multifaceted role of hair follicle dermal cells in bioengineered skins. Br. J. Dermatol. 2017, 176, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Gharzi, A. Dermal and epidermal cell functions in the growth and regeneration of hair follicles and other skin appendages. Durham University: 1998.
- Rognoni, E.; Watt, F.M. Skin Cell Heterogeneity in Development, Wound Healing, and Cancer. Trends Cell Biol. 2018, 28, 709–722. [Google Scholar] [CrossRef]
- Kaushal, G.S.; Rognoni, E.; Lichtenberger, B.M.; Driskell, R.R.; Kretzschmar, K.; Hoste, E.; Watt, F.M. Fate of Prominin-1 Expressing Dermal Papilla Cells during Homeostasis, Wound Healing and Wnt Activation. J. Investig. Dermatol. 2015, 135, 2926–2934. [Google Scholar] [CrossRef]
- Martino, P.A.; Heitman, N.; Rendl, M. The dermal sheath: An emerging component of the hair follicle stem cell niche. Exp. Dermatol. 2020, 30, 512–521. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kumar, A.; Zhang, F.; Lee, C.; Li, Y.; Tang, Z.; Arjunan, P. VEGF-independent angiogenic pathways induced by PDGF-C. Oncotarget 2010, 1, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Crawford, Y.; Kasman, I.; Yu, L.; Zhong, C.; Wu, X.; Modrusan, Z.; Kaminker, J.; Ferrara, N. PDGF-C Mediates the Angiogenic and Tumorigenic Properties of Fibroblasts Associated with Tumors Refractory to Anti-VEGF Treatment. Cancer Cell 2009, 15, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Moriya, J.; Ferrara, N. Inhibition of protein kinase C enhances angiogenesis induced by platelet-derived growth factor C in hyperglycemic endothelial cells. Cardiovasc. Diabetol. 2015, 14, 19–10. [Google Scholar] [CrossRef]
- Folestad, E.; Kunath, A.; Wågsäter, D. PDGF-C and PDGF-D signaling in vascular diseases and animal models. Mol. Asp. Med. 2018, 62, 1–11. [Google Scholar] [CrossRef]
- Lee, C.; Zhang, F.; Tang, Z.; Liu, Y.; Li, X. PDGF-C: a new performer in the neurovascular interplay. Trends Mol. Med. 2013, 19, 474–486. [Google Scholar] [CrossRef]
- Cao, Z.; Liu, Y.; Wang, Y.; Leng, P. Research progress on the role of PDGF/PDGFR in type 2 diabetes. Biomed. Pharmacother. 2023, 164, 114983. [Google Scholar] [CrossRef]
- Zhang ZB, Ruan CC, Lin JR, Xu L, Chen XH, Du YN, Fu MX, Kong LR, Zhu DL, Gao PJ. Perivascular Adipose Tissue-Derived PDGF-D Contributes to Aortic Aneurysm Formation During Obesity. Diabetes 2018;67:1549-1560.
- Lu, J.-F.; Hu, Z.-Q.; Yang, M.-X.; Liu, W.-Y.; Pan, G.-F.; Ding, J.-B.; Liu, J.-Z.; Tang, L.; Hu, B.; Li, H.-C. Downregulation of PDGF-D Inhibits Proliferation and Invasion in Breast Cancer MDA-MB-231 Cells. Clin. Breast Cancer 2021, 22, e173–e183. [Google Scholar] [CrossRef]
- Ma S, Tang T, Wu X, Mansour AG, Lu T, Zhang J, Wang L-S, Caligiuri MA, Yu J. PDGF-D− PDGFRβ signaling enhances IL-15–mediated human natural killer cell survival. PNAS 2022;119:e2114134119.
- Heitman, N.; Sennett, R.; Mok, K.-W.; Saxena, N.; Srivastava, D.; Martino, P.; Grisanti, L.; Wang, Z.; Ma’ayan, A.; Rompolas, P.; et al. Dermal sheath contraction powers stem cell niche relocation during hair cycle regression. Science 2019, 367, 161. [Google Scholar] [CrossRef]
- Williams, R.; Westgate, G.E.; Pawlus, A.D.; Sikkink, S.K.; Thornton, M.J. Age-Related Changes in Female Scalp Dermal Sheath and Dermal Fibroblasts: How the Hair Follicle Environment Impacts Hair Aging. J. Investig. Dermatol. 2020, 141, 1041–1051. [Google Scholar] [CrossRef]
- Keskin ES, Keskin ER, Öztürk MB, Çakan D. The effect of MMP-1 on wound healing and scar formation. Aesthetic Plast Surg 2021;45:2973-2979.
- Fonseca-Camarillo, G.; Furuzawa-Carballeda, J.; Martínez-Benitez, B.; Barreto-Zuñiga, R.; Yamamoto-Furusho, J.K. Increased expression of extracellular matrix metalloproteinase inducer (EMMPRIN) and MMP10, MMP23 in inflammatory bowel disease: Cross-sectional study. Scand. J. Immunol. 2020, 93, e12962. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Hu, Q.; Yang, Y.; Yuan, H.; Cai, Y.; Liu, Z.; Xu, Z.; Xiong, Y.; Zhou, J.; Ye, Q.; et al. Effect of Matrix Metalloproteinase 23 Accelerating Wound Healing Induced by Hydroxybutyl Chitosan. ACS Appl. Bio Mater. 2023, 6, 1460–1470. [Google Scholar] [CrossRef] [PubMed]
- Niiyama, S.; Ishimatsu-Tsuji, Y.; Nakazawa, Y.; Yoshida, Y.; Soma, T.; Ideta, R.; Mukai, H.; Kishimoto, J. Gene Expression Profiling of the Intact Dermal Sheath Cup of Human Hair Follicles. Acta Dermato-Venereologica 2018, 98, 694–698. [Google Scholar] [CrossRef] [PubMed]
- Matziari, M.; Dive, V.; Yiotakis, A. Matrix metalloproteinase 11 (MMP-11; stromelysin-3) and synthetic inhibitors. Med. Res. Rev. 2006, 27, 528–552. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wei, Y.; Fan, X.; Zhang, P.; Wang, P.; Cheng, S.; Zhang, J. MicroRNA-125b as a tumor suppressor by targeting MMP11 in breast cancer. Thorac. Cancer 2020, 11, 1613–1620. [Google Scholar] [CrossRef] [PubMed]
- de Almeida, L.G.; Thode, H.; Eslambolchi, Y.; Chopra, S.; Young, D.; Gill, S.; Devel, L.; Dufour, A. Matrix Metalloproteinases: From Molecular Mechanisms to Physiology, Pathophysiology, and Pharmacology. Materials 2022, 74, 712–768. [Google Scholar] [CrossRef] [PubMed]
- Couture, C.; Zaniolo, K.; Carrier, P.; Lake, J.; Patenaude, J.; Germain, L.; Guérin, S.L. The tissue-engineered human cornea as a model to study expression of matrix metalloproteinases during corneal wound healing. Biomaterials 2016, 78, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Waresijiang, N.; Sun, J.; Abuduaini, R.; Jiang, T.; Zhou, W.; Yuan, H. The downregulation of miR-125a-5p functions as a tumor suppressor by directly targeting MMP-11 in osteosarcoma. Mol. Med. Rep. 2016, 13, 4859–4864. [Google Scholar] [CrossRef] [PubMed]
- Brew, K.; Nagase, H. The tissue inhibitors of metalloproteinases (TIMPs): An ancient family with structural and functional diversity. Biochim. Biophys. Acta BBA Mol. Cell Res. 2010, 1803, 55–71. [Google Scholar] [CrossRef]
- Vandenbroucke RE, Libert C. Is there new hope for therapeutic matrix metalloproteinase inhibition? Nat Rev Drug Discov 2014;13:904-927.
- Cabral-Pacheco, G.A.; Garza-Veloz, I.; la Rosa, C.C.-D.; Ramirez-Acuña, J.M.; A Perez-Romero, B.; Guerrero-Rodriguez, J.F.; Martinez-Avila, N.; Martinez-Fierro, M.L. The Roles of Matrix Metalloproteinases and Their Inhibitors in Human Diseases. Int. J. Mol. Sci. 2020, 21, 9739. [Google Scholar] [CrossRef]
- Li, Z.; Guo, S.; Yao, F.; Zhang, Y.; Li, T. Increased ratio of serum matrix metalloproteinase-9 against TIMP-1 predicts poor wound healing in diabetic foot ulcers. J. Diabetes its Complicat. 2013, 27, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Meissburger, B.; Stachorski, L.; Röder, E.; Rudofsky, G.; Wolfrum, C. Tissue inhibitor of matrix metalloproteinase 1 (TIMP1) controls adipogenesis in obesity in mice and in humans. Diabetologia 2011, 54, 1468–1479. [Google Scholar] [CrossRef] [PubMed]
- Alexander, C.M.; Selvarajan, S.; Mudgett, J.; Werb, Z. Stromelysin-1 Regulates Adipogenesis during Mammary Gland Involution. J. Cell Biol. 2001, 152, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Martins, V.L.; Caley, M.; O’toole, E.A. Matrix metalloproteinases and epidermal wound repair. Cell Tissue Res. 2012, 351, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Ravanti, L.; Kähäri, V.M. Matrix metalloproteinases in wound repair (review). Int. J. Mol. Med. 2000, 6. [Google Scholar] [CrossRef]
- Sarrazy, V.; Billet, F.; Micallef, L.; Coulomb, B.; Desmoulière, A. Mechanisms of pathological scarring: Role of myofibroblasts and current developments. Wound Repair Regen. 2011, 19, s10–s15. [Google Scholar] [CrossRef]
- Mirastschijski, U.; Schnabel, R.; Claes, J.; Schneider, W.; Ågren, M.S.; Haaksma, C.; Tomasek, J.J. Matrix metalloproteinase inhibition delays wound healing and blocks the latent transforming growth factor-β1-promoted myofibroblast formation and function. Wound Repair Regen. 2010, 18, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Yoon, M.; Choi, K.-Y. Approaches for Regenerative Healing of Cutaneous Wound with an Emphasis on Strategies Activating the Wnt/β-Catenin Pathway. Adv. Wound Care 2022, 11, 70–86. [Google Scholar] [CrossRef]
- Liu B, Zhou H, Zhang T, Gao X, Tao B, Xing H, Zhuang Z, Dardik A, Kyriakides TR, Goodwin JE. Loss of endothelial glucocorticoid receptor promotes angiogenesis via upregulation of Wnt/beta-catenin pathway. Angiogenesis 2021;24:631-645.
- Guo R, Wang X, Fang Y, Chen X, Chen K, Huang W, Chen J, Hu J, Liang F, Du J, Dordoe C, Tian X, Lin L. rhFGF20 promotes angiogenesis and vascular repair following traumatic brain injury by regulating Wnt/beta-catenin pathway. Biomed Pharmacother 2021;143:112200.
- Popp T, Steinritz D, Breit A, Deppe J, Egea V, Schmidt A, Gudermann T, Weber C, Ries C. Wnt5a/β-catenin signaling drives calcium-induced differentiation of human primary keratinocytes. J Invest Dermatol 2014;134:2183-2191.
- Zhu, X.-J.; Liu, Y.; Dai, Z.-M.; Zhang, X.; Yang, X.; Li, Y.; Qiu, M.; Fu, J.; Hsu, W.; Chen, Y.; et al. BMP-FGF Signaling Axis Mediates Wnt-Induced Epidermal Stratification in Developing Mammalian Skin. PLOS Genet. 2014, 10, e1004687–e1004687. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.; Kim, E.; Seo, S.H.; Kim, G.-U.; Choi, K.-Y. KY19382 Accelerates Cutaneous Wound Healing via Activation of the Wnt/β-Catenin Signaling Pathway. Int. J. Mol. Sci. 2023, 24, 11742. [Google Scholar] [CrossRef]
- Yoon M, Seo SH, Choi S, Han G, Choi K-Y. Euodia daniellii Hemsl. Extract and Its Active Component Hesperidin Accelerate Cutaneous Wound Healing via Activation of Wnt/β-Catenin Signaling Pathway. Molecules 2022;27:7134.
- Ito, M.; Yang, Z.; Andl, T.; Cui, C.; Kim, N.; Millar, S.E.; Cotsarelis, G. Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature 2007, 447, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Wang X, Chen H, Tian R, Zhang Y, Drutskaya MS, Wang C, Ge J, Fan Z, Kong D, Wang X, Cai T, Zhou Y, Wang J, Wang J, Wang S, Qin Z, Jia H, Wu Y, Liu J, Nedospasov SA, Tredget EE, Lin M, Liu J, Jiang Y, Wu Y. Macrophages induce AKT/beta-catenin-dependent Lgr5(+) stem cell activation and hair follicle regeneration through TNF. Nat Commun 2017;8:14091.
- Rinkevich, Y.; Walmsley, G.G.; Hu, M.S.; Maan, Z.N.; Newman, A.M.; Drukker, M.; Januszyk, M.; Krampitz, G.W.; Gurtner, G.C.; Lorenz, H.P.; et al. Identification and isolation of a dermal lineage with intrinsic fibrogenic potential. Science 2015, 348, aaa2151. [Google Scholar] [CrossRef] [PubMed]
- Martino, P.; Sunkara, R.; Heitman, N.; Rangl, M.; Brown, A.; Saxena, N.; Grisanti, L.; Kohan, D.; Yanagisawa, M.; Rendl, M. Progenitor-derived endothelin controls dermal sheath contraction for hair follicle regression. Nature 2023, 25, 222. [Google Scholar] [CrossRef] [PubMed]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Castella, L.F.; Buscemi, L.; Godbout, C.; Meister, J.-J.; Hinz, B. A new lock-step mechanism of matrix remodelling based on subcellular contractile events. J. Cell Sci. 2010, 123, 1751–1760. [Google Scholar] [CrossRef] [PubMed]
- Follonier Castella, L.; Gabbiani, G.; McCulloch, C.A.; Hinz, B. Regulation of myofibroblast activities: Calcium pulls some strings behind the scene. Exp. Cell Res. 2010, 316, 2390–2401. [Google Scholar] [CrossRef]
- Jimenez, F.; Poblet, E.; Izeta, A. Reflections on how wound healing-promoting effects of the hair follicle can be translated into clinical practice. Exp. Dermatol. 2014, 24, 91–94. [Google Scholar] [CrossRef]
- Debono, R. Histological and immunohistochemical studies of excisional wounds in the rat with special reference to the involvement of the hair follicles in the wound healing process. Durham University: 2000.
| DSC | DP cell | DF | DSC, DP cell | DSC, DF | DSC, DF, DP cell |
| Col3a1 | Col9a2 | Col4a5 | Col5a3 | Col1a1 | Col6a3 |
| Col4a1 | Col15a1 | Col7a1 | Col1a2 | Col16a1 | |
| Col4a2 | Col23a1 | Col25a1 | Col5a1 | ||
| Col6a6 | Col26a1 | Col5a2 | |||
| Col8a1 | Col6a1 | ||||
| Col8a2 | Col6a2 | ||||
| Col11a1 | |||||
| Col12a1 | |||||
| Col27a1 |
| MMPs | DSCs[76] | DFs[2,76] | DP cells[76] | Substrates[84,93,94] |
| Gelatinases | ||||
| MMP2 (gelatinase A) | + (human healthy DFs; human SSc DFs) | Gelatin; collagen I, IV, V, VII and X; laminin; aggrecan; fibronectin; tenascin | ||
| Stromelysins | ||||
| MMP11 (stromelysin-3) | + (mouse; human scalp) | + (human SSc DFs) | Serine protease inhibitors; α1-proteinase inhibitor | |
| Membrane-type MMPs | ||||
| MMP14 (MT1-MMP) | + (human SSc DFs) | Collagen I, II and III; gelatin; fibronectin; laminin; vitronectin; aggrecan; tenascin; nidogen; perlecan; fibrillin; α1-proteinase inhibitor; alpha2-macroglobulin; fibrin | ||
| MMP17 (MT4-MMP) | + (mouse) | Fibrin; fibrinogen; tumor necrosis factor | ||
| Other MMPs | ||||
| MMP19 | + (mouse) | + (mouse) | Gelatin; aggrecan; cartilage oligomeric matrix protein; collagen IV; laminin; nidogen; tenascin | |
| MMP23 | + (mouse) | + (mouse; human healthy DFs; human SSc DFs) | Unknown | |
| MMP27 | + (mouse) | Unknown | ||
| TIMPs | MMP inhibition[87] | |||
| TIMP1 | + (mouse) | + (human healthy DFs; human SSc DFs) | weak for MMP14, MMP16, MMP19 and MMP24 | |
| TIMP2 | + (human healthy DFs; human SSc DFs) | All | ||
| TIMP3 | + (human healthy DFs; human SSc DFs) | All | ||
| TIMP4 | Most |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




