1. Introduction
The geogenic presence of arsenic in groundwater represents a global health threat and currently affects all five continents. Its permanent consumption causes the appearance of various types of cancer, such as cancer of the skin, lungs, kidneys, liver, and potentially the prostate, as well as non-cancerous disorders including skin lesions, cardiovascular diseases, effects on the nervous system, and diabetes.[
1,
2] It is estimated that about 220 million people worldwide are potentially exposed to high concentrations of arsenic in groundwater with the prospect of further increases as new contaminated areas are constantly being discovered [
3,
4] In Bolivia, so far, some studies have documented the presence of geogenic arsenic in the Bolivian Altiplano [
5,
6], in quite high concentrations in the southern part of Lake Poopó, especially in the town of Quillacas with approximately 250 μg/l [
7] and the lower valley of Cochabamba.[
8] Recent research shows the distribution of arsenic in different regions of Bolivia, including some wells in the lowlands, which have As concentrations higher than the maximum permissible value of 10 μg/l established by the Bolivian regulation NB 512 for drinking water [
9] .
There are several technologies available to achieve the demanding arsenic removal required for human consumption of water, however, the economic variable is a determining factor [
10].
According to Hao et al. [
11], among many currently available techniques for arsenic removal from water, the adsorption process is considered one of the most promising techniques due to its low cost, high efficiency, and ease of operation. Iron-based adsorbents have been developed extensively and showed good removal efficiency of arsenic species from water. Removal of arsenic from water by adsorption on nanomaterials is being studied as a promising alternative method to conventional methods (reverse osmosis, nanofiltration, ion exchange, etc.) because of its large specific surface area, catalytic potential, low energy consumption, high efficiency, and reversibility [
12].
A literature search was performed in the Scopus database, focusing on articles related to the remediation of groundwater contaminated with arsenic by adsorption with zerovalent iron nanoparticles (ZVI) and iron sesquioxide (magnetite). 51 records were found. It can be concluded that the application of nanotechnology in the treatment of arsenic-contaminated water, particularly with ZVI nanoparticles and magnetite, is a topic of growing importance in the last decade.
Casentini et al. [
13] analyzed the use of Fe hydroxide nanoparticles encapsulated in an exopolysaccharide gel (FeEPS) for arsenic removal in aqueous solutions. The pH of the hydrogel was found to affect its adsorption capacity, being less favorable than magnetite nanoparticles. The higher removal efficiency was observed in more concentrated FeEPS solutions due to a higher amount of iron nanoparticles present. Immobilization of FeEPS on carbonaceous shells was shown to be effective in arsenic adsorption.
Shahmohammadi et al. [
14] investigated the adsorption of arsenic in well water using a filter with an iron matrix (ZVI). The effectiveness of all layers of the filter was examined and variables such as pH, flow rate, arsenic concentration, turbidity, and chloride concentration were considered to optimize the process. The filter was applied for 10 hours. It was found that the turbidity and flow rate had the highest correlation with adsorption efficiency. Optimal adsorption conditions included: initial arsenic concentration 500 μg/L, flow rate 2 L/h, pH 7, turbidity 0 NTU, and absence of calcium hypochlorite. The filter had a removal efficiency of 98.7%. In conclusion, the proposed filter demonstrated high efficiency in the adsorption of arsenic in water.
According to Sista et al. [
15], iron nanopowder (INP) is a prominent option due to its low cost, high reactivity, and commercial availability. It uses various mechanisms to remove arsenic, such as oxidation, reduction, adsorption, precipitation, and co-precipitation. The properties of the (INP) and the aqueous system, such as pH and the presence of other contaminants, influence its effectiveness. (INP) obtained by electrolytic and reduction routes are preferred. This review highlights the potential of (INP) at micrometric and nanometric levels in arsenic removal in water remediation. The different synthesis routes, removal mechanisms, research evolution, and commercial occurrence of (INP) are also discussed.
The objective of our research is the design, construction, and performance evaluation of a simple and functional filter applied to arsenic removal from groundwaters contaminated with arsenic of geogenic origin. The filter contains zero-valent iron (ZVI) and iron oxide nanoparticles obtained from recycling metal chips by top-down and bottom-up technology, combined with other easily accessible artisanal filtering materials such as charcoal, ground brick, and river sand, in the form of layers for arsenic adsorption. The filter is designed to be used as a household device for water purification.
2. Materials and Methods
Sampling followed the standard protocol [
4,
16]. The pH, electrical conductivity, temperature, and redox potential were determined in situ using a Hanna Instruments model HI 9828 meter. Alkalinity was measured in situ using a Hach model 16900 titrator. Samples were microfiltered (0.45 μm) in polyethylene bottles, acidified with 1% HNO3, for analysis of the majority of cations and trace metals in well water, For anions the sample was not acidified. All samples were refrigerated at 4°C before analysis. Cl-, NO3-, NO2-, SO42-, F- and PO43- ions were determined by ion chromatography (Dionex ICS-1100). Ca, Mg, Na, and K were quantified using a Perkin Elmer Analyst 200 flame atomic absorption spectrophotometer at the Instituto de Investigaciones Químicas, Universidad Mayor de San Andrés. Heavy metals and arsenic were determined by inductively coupled plasma mass spectrometry, ICP-MS, (Agilent 7500) at the laboratory of the Instituto Geológico y Minero de España, IGME, Spain. For adsorption tests and filter performance, arsenic concentrations were measured using a PG Instruments Model AA500 atomic absorption spectrometer with a hydride generator in the laboratories of the Universidad Privada Boliviana in Cochabamba.
The iron nanopowder was obtained with top-down technology in a high-energy ball mill, selection, and magnetic separation as reported by Torres [
17]. The collection was optionally performed by: a) the wet method in a liquid solution of iron particles plus isopropyl alcohol with a magnetic field and b) the dry method: in a high-energy ball mill with a filter.
To obtain the iron oxide nanopowder the bottom-up technology was used with the aid of electrochemical synthesis, from a solution of iron sulfate in distilled water. DC electric current was applied between the cathode (stainless steel plate) and the anode (conductive bar of compacted iron microparticles of 45 μm), leading to the deposition and accumulation of iron oxide nanoparticles on the cathode plates and the bottom of the tank. [
18]
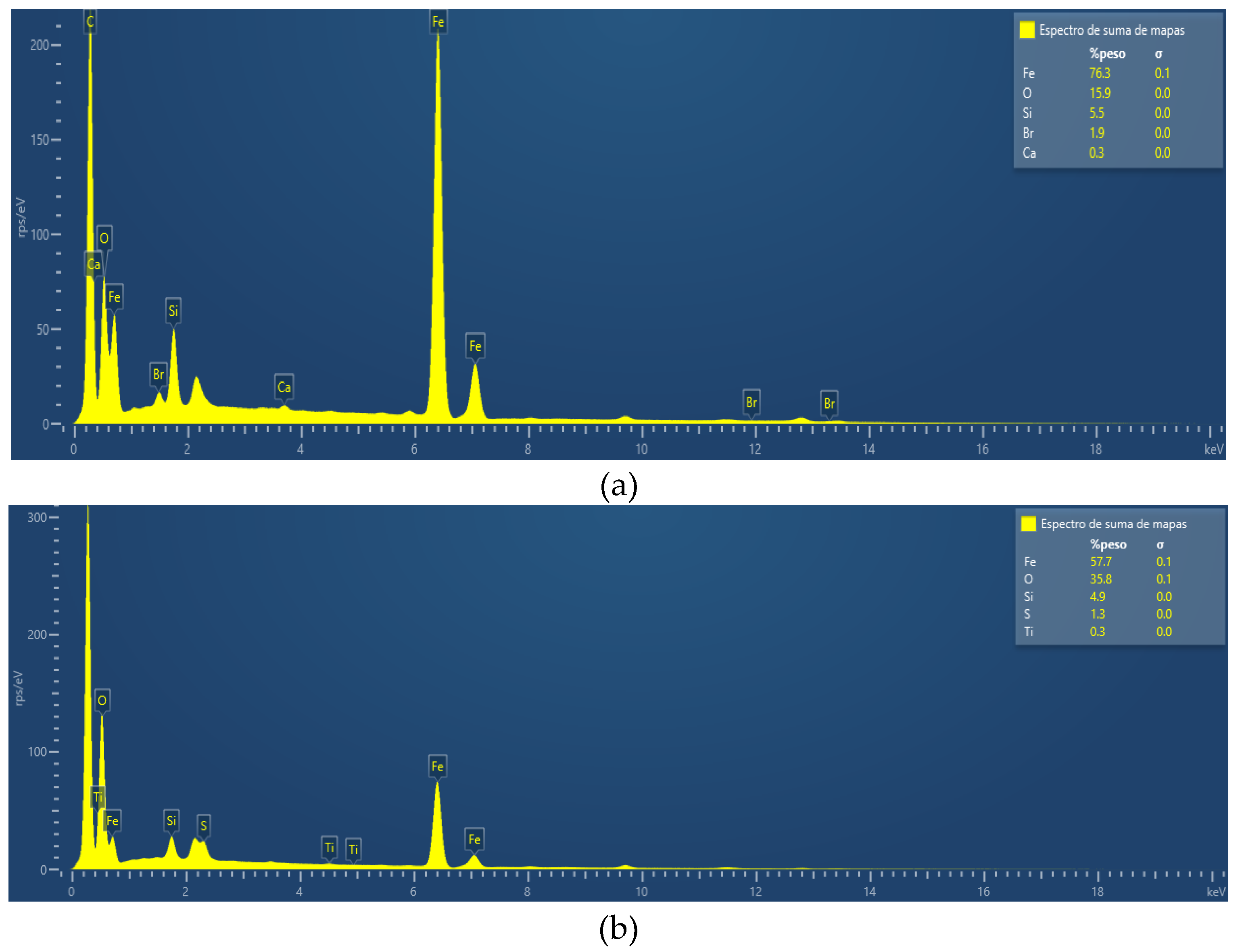
The chemical composition of both nanoparticles was quantified by scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDX).
The size distribution of the nanoparticles was performed on a TESCAN VEGA3 LMU scanning electron microscope. Both analyses were performed at the physics laboratory of the University of San Simon in Cochabamba Bolivia.
The filter was constructed with a 2” (50.8 mm) diameter high-density polyethylene (HDPE) SCH 40 pipe, one meter long, made of recycled material, with threads at the ends to install couplings for reduction to ½” (12.7 mm) as a commercial measure. The tube was filled as shown in
Figure 1 and
Table 1.
To determine the adsorption capacity, different doses (0.01-10 g) of both nanoparticles were placed in contact with synthetic solutions of As(V), As(III), and groundwater. The solutions were stirred for varying periods (20 min to 1 d). The initial and final As concentrations were measured to determine the removal efficiency and adsorption capacity.
Filtration was performed by gravity in continuous downflow mode, and arsenic removal performance was monitored as a function of the volume treated and the number of volumes of nanoparticle beds in the filter. Arsenic concentrations were measured at the inlet and outlet at different times, collecting samples for subsequent analysis.
3. Results
3.1. Untreated Water Quality
Table 2 presents the results of the physical and chemical parameters of the well located in District 4 of Cochabamba Bolivia (Cbba. Bo.). The water is slightly alkaline and the oxidation-reduction potential, Eh, showed a relatively oxidizing system, +198 mV. Its salinity is low to moderate (252 μS/cm). The majority of cations and anions indicate that the water is of the Na-Ca-Mg-HCO3 type, as reported by Escalera & Ormachea (19) for a set of wells located in the western peri-urban zone of (Cbba. Bo.). The quality satisfies the drinking water standard NB 512, except for arsenic and manganese contents. The arsenic content is 113 μg-As/l, much higher than the limit of 10 μg-As/l established by Bolivian standard NB 512. The contents of competitive anions; i.e. sulfates, nitrates, nitrites, and chlorides are relatively low except for phosphates. Arsenic is present in water in the form of arsenites, As(III). This species is more difficult to adsorb than arsenate, As(V). This section may be divided by subheadings. It should provide a concise and precise description of the experimental results, their interpretation, as well as the experimental conclusions that can be drawn.
3.2. Characterization of Nanoparticles
Iron oxide nanoparticles (Magnetite). The percentage elemental composition is: Fe (57.7%), O (35.8%), Si (4.9%), S (1.3%), and Ti (0.3%). The size distribution is from 60 to 200 nm.
Iron nanoparticles (ZVI). The chemical composition is Fe (76.3%), O (15.9%), Si (5.5%), Br (1.9%), and Ca (0.3%). The nanoparticles have a regular morphology, with sizes ranging from approximately 60 nm to 200 nm
Figure 2 (a), (b),
Table 3 and
Figure 3.
3.3. Adsorption Capacity
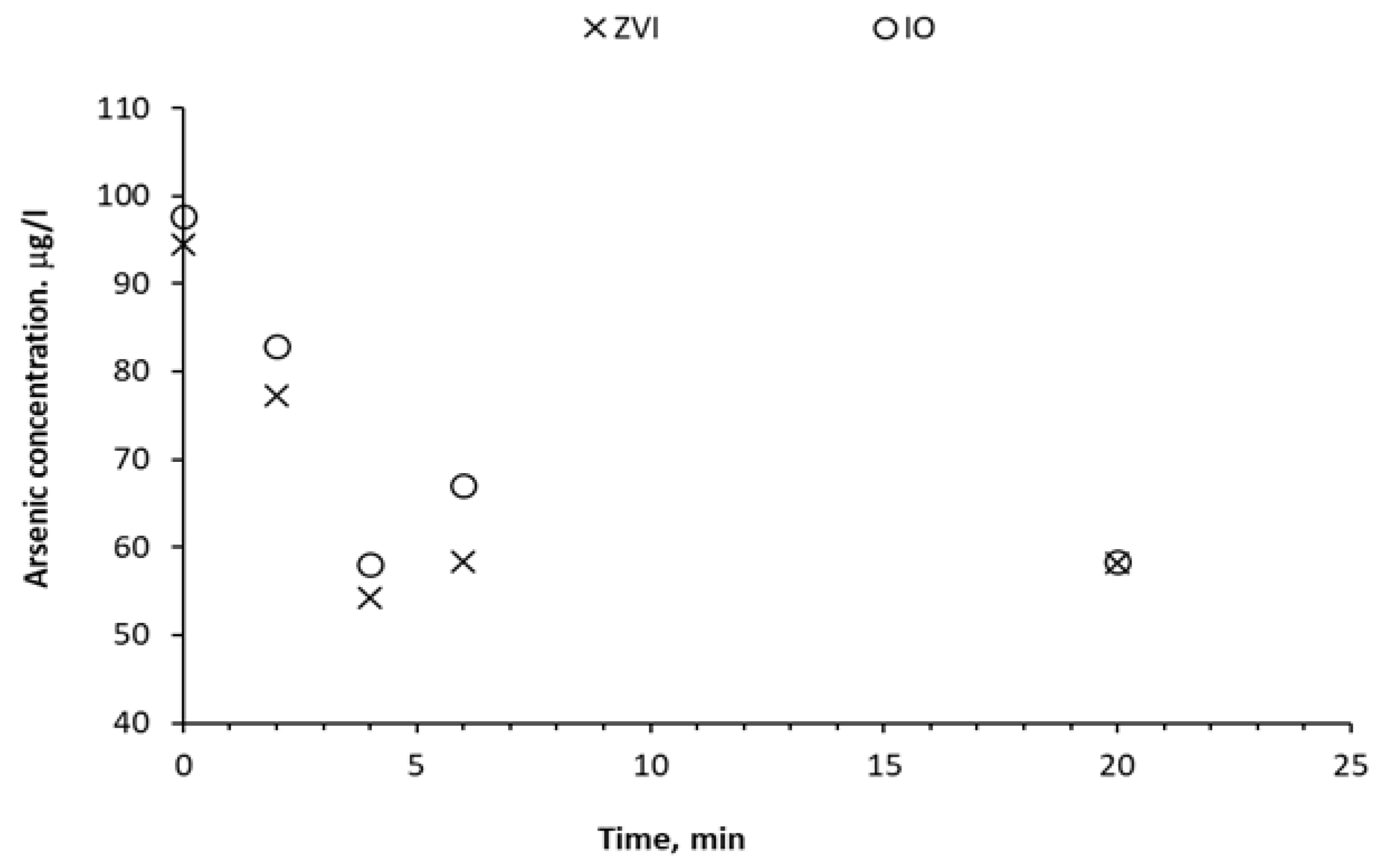
Table 4 and
Figure 4 present the results of the treatment of groundwater characterized in
Table 1. The percentage removal of total As (mostly As(III)) and the adsorption capacity of zero-valent iron and magnetite nanoparticles are presented. Starting from an As(III) concentration of 83 ppb, removal efficiencies of up to 43% could be achieved with ZVI and 41% with magnetite. After 20 min, a saturation of the active sites of the nanoparticles is reached, which allows the establishment of the equilibrium condition to calculate the adsorption capacity, reaching values of approximately 4 mg-As(III)/g for both nanoparticles.
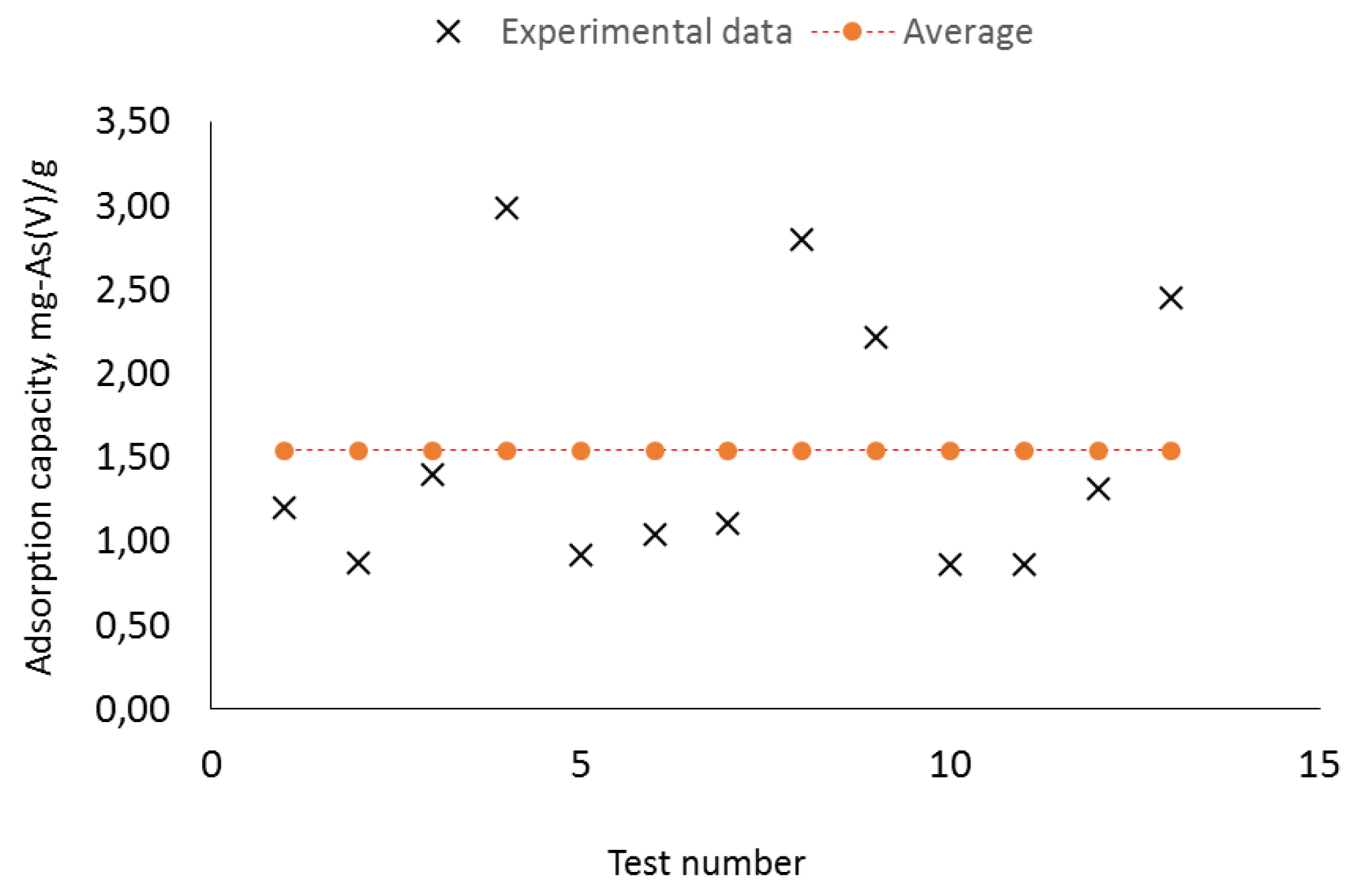
Figure 5 presents the results of the adsorption tests in synthetic waters with As(V) with ZVI iron nanoparticles. Solutions of 500 ppb were subjected in each of the 13 runs and residual concentrations were determined after 1 or 2 h, times at which equilibrium is achieved. Adsorption capacities ranged from 0.86 to 2.99 mg-As(V)/g with an average of 1.54 mg-As(V)/g.
For As (V) in synthetic waters, a significant variability of the initial adsorption capacities is noted which is related to the variability of their size 20-200 nm and their specific surface area (436 - 131 m
2/g). The adsorption capacities of nanoparticles are higher than other larger adsorbent materials such as ferric oxide-coated sands with 0.64 mg-As/g [
19]. In contrast, smaller magnetic nanocomposites (11-15 nm) have much higher capacities (74.9 mg-As/g), due to their huge specific surface area (thousands of m
2/g)[
20].
For As(III) in well water, despite the presence of phosphates (9 mg/l), the adsorption capacity was 4 mg-As/g for ZVI and magnetite, a result comparable with that reported by Kanel et al. for well water in Bangladesh (maximum capacities of 3-5 mg/g of ZVI). The authors used a size between 10 - 100 nm with 60% smaller than 50 nm. and indicated phosphates reduce adsorption capacities by up to 66% [
21]
3.4. Nanofilter Performance
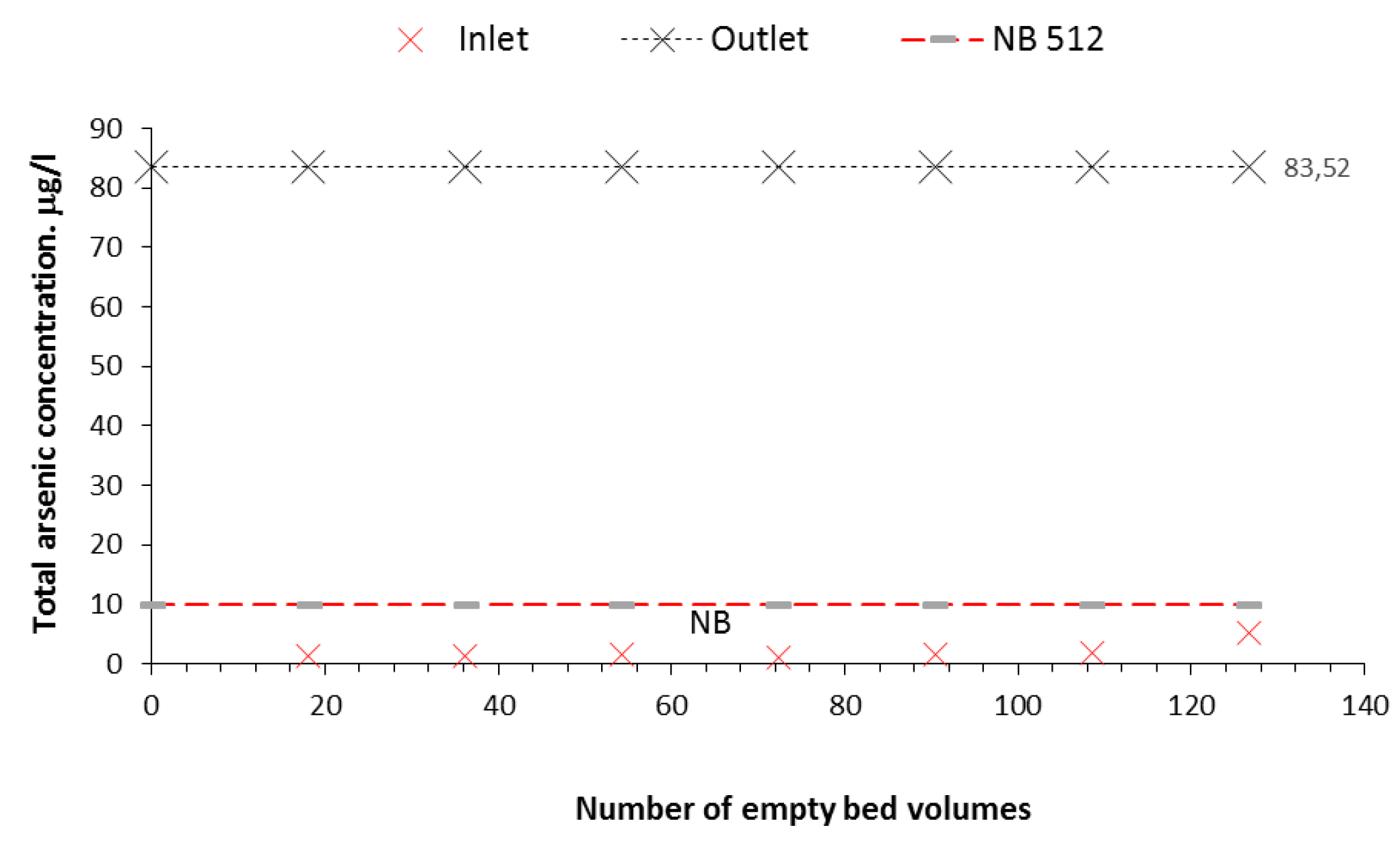
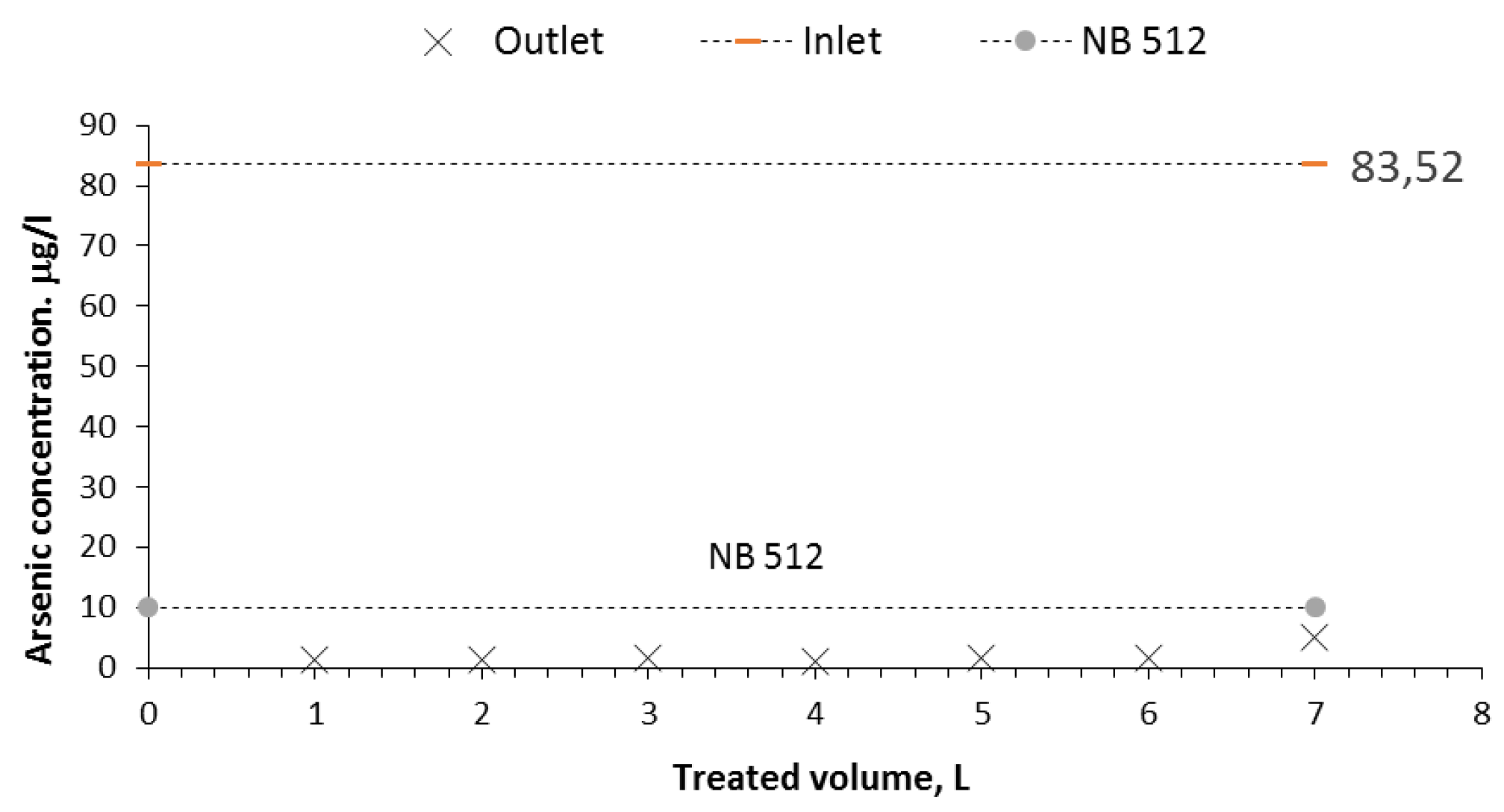
Figure 6 and
Figure 7 illustrate the good performance of the Nanofilter in treating groundwater. The concentrations of the treated water are presented as a function of the volume treated and the number of bed volumes, achieving reductions to values below the maximum permissible limit of 10 ppb established by Bolivian standard 512.
4. Discussion
The original material for obtaining both nanoparticles is iron shavings from machining process. This study takes advantage of this low-cost waste due to its widely known potential to remediate As(V) and As(III) contaminated waters. Top-down and bottom-up technologies are also simple and widely used for the fabrication of micro- and nanoparticles. In contrast, the synthesis of uniform-sized and smaller nanoparticles is more complex and diverse. These aspects make the fabrication cost quite lower in the case of this study.
Although the masses of magnetite and zerovalent iron nanoparticles in the prototype were quite small, the As removal efficiency is over 95%. In treated well water, arsenic is present in the water in the form of arsenites, As(III) which is more difficult to adsorb than arsenate, As(V). However, in the nanofilter it was possible to reduce its content to 50% below that established by Bolivian Standard 512, at a low cost since nanoparticles obtained by the recycling process of iron chips by top-down and bottom-up methods were used, a technology developed and tested to obtain iron and iron oxide nanoparticles with size and chemical composition characteristics suitable for the remediation of water contaminated with arsenic.
The novelty of this work lies in the development of a new low-cost filtration system using known materials that are easily accessible, which allows predicting that this remediation method can be widely applied in all the sites identified with this problem.
In a work similar to this study (20), a gravity filter was developed using a polyurethane (PU) foam support loaded with iron oxide nanoparticles. The author comments that it is an innovative filter that is low cost, easy to manufacture, fast, practical, and efficient for arsenic removal, demonstrating between 50 and 70 % effectiveness. However, it should be noted that the iron oxide nanoparticles used were purchased at an average cost of hydrated iron oxide is $586/lb, and nanoscale zero-valent iron of $4,850/lb, respectively is over 50% of the cost that we produced.
From the results obtained in this research, it can be concluded that the filter constructed with iron and iron oxide nanoparticles has a higher efficiency because it was able to reduce the arsenic in contaminated water with 83.52 g/l to 5 g/l, representing a 94% reduction. On the other hand, the cost and availability of the materials used make the nanofilter very accessible in technical and economic terms.
The filter built with iron and iron oxide nanoparticles and other filler elements as easily accessible filtering elements, the object of the present research, was able to reduce the arsenic content in groundwater to 50% below that established by Bolivian Standard NB 512, at a low cost since nanoparticles obtained by the recycling process of iron chips by top-down and bottom-up method were used. Technology was developed and tested to obtain iron and iron oxide nanoparticles with size and chemical composition characteristics suitable for the remediation of water contaminated with arsenic. Its construction at industrial and commercial scale is feasible because its technology is within reach and available in the environment.
Quality of life and socioeconomic aspects
The potential of the nanofilter as a system applied at the point of use lies in the possibility of directly benefiting people who do not have alternative access to safe water sources, in the three ecological floors of Bolivia that have the problem of arsenic-contaminated water especially in rural areas. So it could improve the quality of life with the improvement of water quality.
It is a fact that one of the current and future problems in our country is the scarcity of drinking water sources for human consumption and irrigation. Hence, the optimization of its use is essential. The filter also has the potential to remediate water contaminated with heavy metals in industrial and mining processes for its reuse, once it’s scaling up becomes possible.
5. Conclusions
From the results obtained in the present investigation, it can be concluded that the filter constructed with iron and iron oxide nanoparticles has a higher efficiency than that achieved in other similar studies, managing to reduce the arsenic in well water naturally contaminated with 83.52 g/l to 5 g/l, which represents a 94% reduction. On the other hand, the cost and availability of the materials used make this filter very accessible in technical and economic terms.
The technology developed and tested is suitable for obtaining iron and iron oxide nanoparticles with size and chemical composition characteristics suitable for the remediation of arsenic-contaminated water. This is confirmed by similar work using the adsorption remediation process.
The industrial and commercial scale construction of the nanofilter is feasible because its technology is within reach and available in the environment.
One of the crucial problems identified in Bolivia is the presence of arsenic in groundwater in very high concentrations, particularly in rural areas in the three ecological levels: altiplano, valleys, and the Amazon, with greater incidence in rural areas. This fact endangers the health of the inhabitants of these areas and their biodiversity since this liquid element coming from wells is the only source of supply for human use and irrigation. The potential of the nanofilter as a system applied at the point of use lies in the possibility of directly benefiting the population that does not have alternative access to safe water sources. With the application of this filtration system, the quality of life could be improved by improving water quality.
Funding
This research received no external funding.
Data Availability Statement
The data used in this research are available in this paper and are included in the author’s references so other researchers can use them.
Acknowledgments
The authors appreciate the participation of Jorge Alvarez graduate of the Production Engineering Department at Universidad Privada Boliviana, who carried out the experimental adsorption runs. To Dr. Victor Achá from La Salle University in France for the preliminary results of nanoparticles characterization.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kapaj, S.; Peterson, H.; Liber, K.; Bhattacharya, P. Human health effects from chronic arsenic poisoning--a review. J Environ Sci Health Part A Tox Hazard Subst Environ Eng. 2006, 41, 2399–2428. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.F.; Beck, B.D.; Chen, Y.; Lewis, A.S.; Thomas, D.J. Arsenic exposure and toxicology: A historical perspective. Toxicol Sci. 2011, 123, 305–332. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behaviour and distribution of arsenic in natural waters. Appl Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Polya, D.; Jovanovic, D. Best Practice Guide on the Control of Arsenic in Drinking Water. IWA Publishing; 2017. 308 p.
- Banks, D.; Markland, H.; Smith, P.; Mendez, C.; Rodriguez, J.; Huerta, A.; et al. Distribution, salinity and pH dependence of elements in surface waters of the catchment areas of the Salars of Coipasa and Uyuni, Bolivian Altiplano. J Geochem Explor. 2004, 84, 141–166. [Google Scholar] [CrossRef]
- Ramos Ramos, O.; Choque, L.; Ormachea Muñoz, M.; Bhattacharya, P.; Quino, I.; Aguirre, J.; et al. Sources and behavior of arsenic and trace elements in groundwater and surface water in the Poopó Lake Basin, Bolivian Altiplano. Environ Earth Sci. 2012, 66, 793–807. [Google Scholar] [CrossRef]
- Ormachea Munoz, M.; Wern, H.; Johnsson, F.; Bhattacharya, P.; Sracek, O.; Thunvik, R.; et al. Geogenic arsenic and other trace elements in the shallow hydrogeologic system of Southern Poopo Basin, Bolivian Altiplano. J Hazard Mater. 2013, 262, 924–940. [Google Scholar] [CrossRef] [PubMed]
- Ormachea Muñoz, M.; Wern, H.; Johnsson, F.; Bhattacharya, P.; Sracek, O.; Thunvik, R.; et al. Geogenic arsenic and other trace elements in the shallow hydrogeologic system of Southern Poopo Basin, Bolivian Altiplano. J Hazard Mater. 2013, 262, 924–940. [Google Scholar] [CrossRef] [PubMed]
- NB512-REGLAM-CtrlCalidadAguaCH.pdf [Internet]. [citado 27 de diciembre de 2023]. Disponible en: http://www.anesapa.org/data/files/NB512-REGLAM-CtrlCalidadAguaCH.pdf.
- D’ambrosio. Congreso Hidrogeológico Argentino. Río Cuarto, Argentina, 2005.
- Hao L, Wang N, Wang C, Li G. Arsenic removal from water and river water by the combined adsorption - UF membrane process. Chemosphere 2018, 202, 768–776.
- Torasso, N.; Vergara-Rubio, A.; Rivas-Rojas, P.; Huck-Iriart, C.; Larrañaga, A.; Fernández-Cirelli, A.; et al. Enhancing arsenic adsorption via excellent dispersion of iron oxide nanoparticles inside poly(vinyl alcohol) nanofibers. J Environ Chem Eng. 2021, 9, 104664. [Google Scholar] [CrossRef]
- Casentini, B.; Gallo, M.; Baldi, F. Arsenate and arsenite removal from contaminated water by iron oxides nanoparticles formed inside a bacterial exopolysaccharide. J Environ Chem Eng. 2019, 7, 102908. [Google Scholar] [CrossRef]
- Shahmohammadi, S.; Shahmoradi, B.; Maleki, A.; Yang, J.K.; Lee, S.M. Efficiency of an iron matrix-based filter in adsorption of arsenic from water. DESALINATION WATER Treat. 2019, 163, 198–205. [Google Scholar] [CrossRef]
- Sista, K.S.; Kumar, D.; Sinha, G.R.; Moon, A.P.; Dwarapudi, S. Iron Powders as a Potential Material for Arsenic Removal in Aqueous Systems. ISIJ Int. 2021, 61, 2687–2702. [Google Scholar] [CrossRef]
- Sampling-protocol-water-samples-july-2020.pdf [Internet].
- Torres Espada, J.; Simon, J. Journal of Nanotechnology and Materials Science Obtaining Iron Nanoparticles from Chip through Top down Technology. Int J Green Nanotechnol Mater Sci Eng. 2021, 17. [Google Scholar]
- Torres Espada, J.; Simón, J. articulo OBTENCION DE NANOPARTICULAS DE OXIDO DE HIERRO revista CTe Innovación. 2021, 18, 11–24.
- Escalera Vásquez, R.; Ormachea Muñoz, M. HIDROQUÍMICA DE LA PRESENCIA NATURAL DE ARSÉNICO EN AGUAS SUBTERRÁNEAS DE ÁREAS SUBURBANAS DE COCHABAMBA-BOLIVIA Y EVALUACIÓN DE LA VIABILIDAD TÉCNICA DE PROCESOS DE REMOCIÓN. Investig Desarro. 2017, 1, 27–41. [Google Scholar] [CrossRef]
- Zubair, Y.; Fuchida, S.; Tokoro, C. Insight into the Mechanism of Arsenic(III/V) Uptake on Mesoporous Zerovalent Iron–Magnetite Nanocomposites: Adsorption and Microscopic Studies. ACS Appl Mater Interfaces. 2020, 12, 49755–49767. [Google Scholar] [CrossRef] [PubMed]
- Kanel, S.R.; Manning, B.; Charlet, L.; Choi, H. Removal of Arsenic(III) from Groundwater by Nanoscale Zero-Valent Iron. Environ Sci Technol. 2005, 39, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- Pillai, A.; Zarandi, M.A.F.; Hussein, F.B.; Pillai, K.M.; Abu-Zahra, N.H. Towards developing a low-cost gravity-driven arsenic filtration system using iron oxide nanoparticle-loaded PU foam. Water Qual Res J. 2020, 55, 234–248. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).