1. Introduction
Due to their behavior and instinct in response to a fright, horses are very susceptible to trauma, and the skin healing process in this species is hampered by complications, with secondary intention healing being quite common, associated with tissue loss, skin tension, contamination, and time of injury occurrence [
1].
Healing by secondary intention begins with hemorrhage, and formation of clot and crust characteristic of the inflammatory phase, and continues with cleaning of the wound, resulting from fibrinous exudation that forms a crust due to dryness, with polymorphonuclear cells being responsible for removing microorganisms and foreign bodies, with the formation of granulation tissue composed of fibroblasts and neovascularization, characteristic of the proliferation phase. The epithelialization tissue migrates over the granulation tissue from several points, where there is deposition of collagen in the scar tissue. The treatment of these injuries aims to reduce the painful process at the site, prevent hemorrhage, and allow complete tissue repair [
2,
3,
4].
In the treatment of wounds, bacterial cellulose (BC) or biocellulose and its variations have been used, being shown to have a beneficial action in skin healing, due to bio-compatibility, elasticity, malleability, transparency, and the ability to keep the wound environment moist and absorb inflammatory exudates [
5,
6,
7]. BC has been intensely studied and evaluated as a tissue repair material and different variations of BC have been successfully applied to skin healing, demonstrating its quality [
5,
6]. The use of BC gel in equine lumbar wounds increased local perfusion and tissue metabolism, especially in the first 14 days of the healing process [
7].
Due to their absorbent characteristics, alginate hydrogels are normally used for the release of drugs and proteins, for the formation of films and microspheres, and for wound debridement [
5].
Biological dressings, also called skin substitutes, can be classified as temporary or permanent. Permanent dressings are able to stimulate and facilitate tissue repair, remaining integrated into the regenerated tissue. Temporary dressings, after performing their role in stimulating tissue repair, do not remain integrated into the receiving bed, and need to be removed [
8]. The use of these in healing dates back to antiquity, with the use of frog skin being described in the Ebers papyrus, in 1500 BC. [
9].
Oliveira [
10] used frog skin in a non-occlusive dressing on rabbits, and observed acceleration of the healing process and antimicrobial activity. Frog skin was used in 16 human burn victims, proving efficient as a biological dressing, with no statistical differences in relation to dressings with Vaseline gauze in histology and immunohistochemistry tests, but was superior when compared to other membranes, as it is practical for transportation, storage, and stocking, in addition to having antimicrobial activities [
11].
The present work aimed to evaluate the healing of experimentally produced wounds in the lumbar region of horses, using bacterial cellulose hydrogel with alginate, or frog skin, to observe the macroscopic and histopathological aspects of healing, and test the hypothesis that bacterial cellulose hydrogel and frog skin favor the healing process in this species.
2. Materials and Methods
2.1. Animals
Five healthy horses were selected, four castrated males and one female, four with no defined breed and one Purebred Arabian, with a mean weight of 381.2 Kg ± 57.07, owned by the Large Animal Veterinary Hospital of UnB, without skin diseases, which had previously passed through clinical examinations, and blood collection for hematology and serum biochemistry: urea, creatinine, gamma glutamyl transferase (GGT), aspartate aminotransferase (AST), and albumin. The horses were dewormed with oral paste based on ivermectin and praziquantel (Padock Plus NF®, CEVA, Paulína/SP, Brazil) and received a prophylactic dose of antitetanus serum (Venco-sat®, Vencofarma, Londrina/PR, Brazil) administered intramuscularly. After the surgical procedures, the animals were kept in stalls and maintained throughout the experiment in good hygiene conditions, fed with commercial feed (Nutrina Equinos Premium®, Nutrina Rações and Minerais, Brasília/DF, Brazil), coast cross hay, and water ad libitum. Throughout the experimental period, the animals were evaluated daily by measuring heart rate (HR) and respiratory rate (RR), the color of visible mucous membranes, capillary refill time (CRT), rectal temperature (TºC), intestinal motility, attitude, and appetite.
2.2. Bacterial Cellulose Hydrogel with Alginate
The cellulose-based hydrogel (1%) with alginate was supplied by the company Seven Indústria de Produtos Biotecnológicas LTDA. It is a product in the development phase to assist in the healing of deep wounds and cavities in human medicine. Biocellulose is obtained by the biosynthesis of the bacterium Gluconacetobacter xylinum, found in nature, being non-toxic and hypoallergenic.
2.3. Frog Skin (Rana catesbeiana)
The frog skin (
Rana catesbeiana) was supplied by the Ranário Laranjeiras – Ranajax, located in Anápolis (GO) specializing in the cultivation of this anuran to obtain quality products and by-products following the “Code of Responsible Conduct for Frog Cultivation”, a document prepared by the Secretariat of Aquaculture and Fisheries of the Presidency of the Republic (SEAP/PR) in 2002. The skin dehydration process was carried out using a dehydrator, where the professional sanitized the metal plates with neutral soap, after drying, the plates received a layer of frog oil to hydrate the skins and were exposed to a temperature of 28ºC for 45 minutes, time for them to release from the plate, reaching the ideal point. Immediately after cooling, they were placed in surgical grade packaging for sterilization by gamma rays, before being sent for sterilization in Co-60 gamma rays (gamma energy of approximately 1.25 MeV) (Gammacell GC220 MDS Nordion irradiator, Canada), with a dose rate of approximately 1.44kGy/h), with a dosage of 25kGy, at the Gamma Radiation Laboratory (GAMALAB), and sent to INPE-USP in São Paulo/SP (
Figure 1).
2.4. Production of the Wounds
The horses underwent a 12-hour food fast and a 6-hour water fast prior to surgery. Tranquilization was performed using 10% xylazine hydrochloride (Equisedan
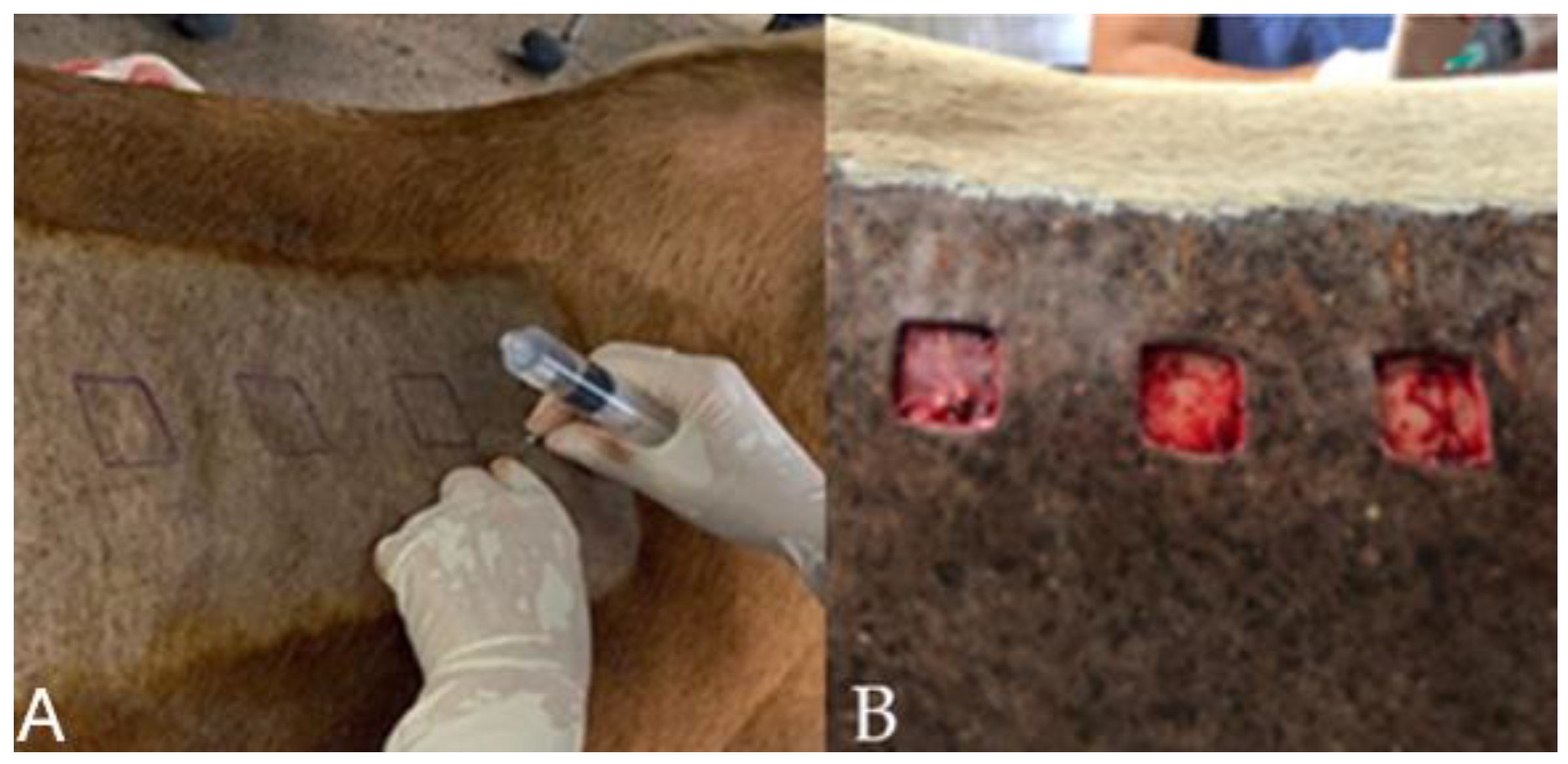
®, J.A. Saúde Animal, Patrocínio Paulista/SP, Brazil), and the dosage of 1mg.kg-1 was used intravenously. Trichotomy was performed on both sides of the lumbar region, cranial to the thigh tuberosity and ventral to the transverse processes of the L1-L6 lumbar vertebrae, and antisepsis was performed with degerming povidone-iodine and 70% alcohol. Soon after, the mold was designed with the aid of a violet-based surgical pen, and local infiltrative anesthesia was performed with 2% lidocaine without vasoconstrictor (Lidocaine hydrochloride 2%®, Hipolabor Farmacêutica, Belo Horizonte/MG, Brazil) (
Figure 2A). On both sides of the lumbar region, cranial to the sacrum and ventral to the transverse process of the lumbar vertebrae (L1-L6), a skin incision was made, with the aid of a scalpel using a No. 24 blade, with the removal of three square-shaped skin flaps, 3 cm on each side, one cranial, one medial, and one caudal, spaced 7 cm apart each (
Figure 2B). All wounds were made using a sterilizable material mold, 3 cm quadrangular, to remove the skin fragment. The depth of the incisions included skin and subcutaneous tissue, leaving the muscle fascia evident.
2.5. Treatments
The animals were randomly chosen for macro and microscopic evaluations, in three horses the right side was used for clinical evaluation of the wound and the left side for microscopic evaluation, and in the other two animals, the reverse procedure was carried out.
Dressing began as soon as the wound hemostasis occurred, and continued daily until healing. All wounds were cleaned with lactated ringer solution (Lactated Ringer Serum - Equiplex - Brazil).
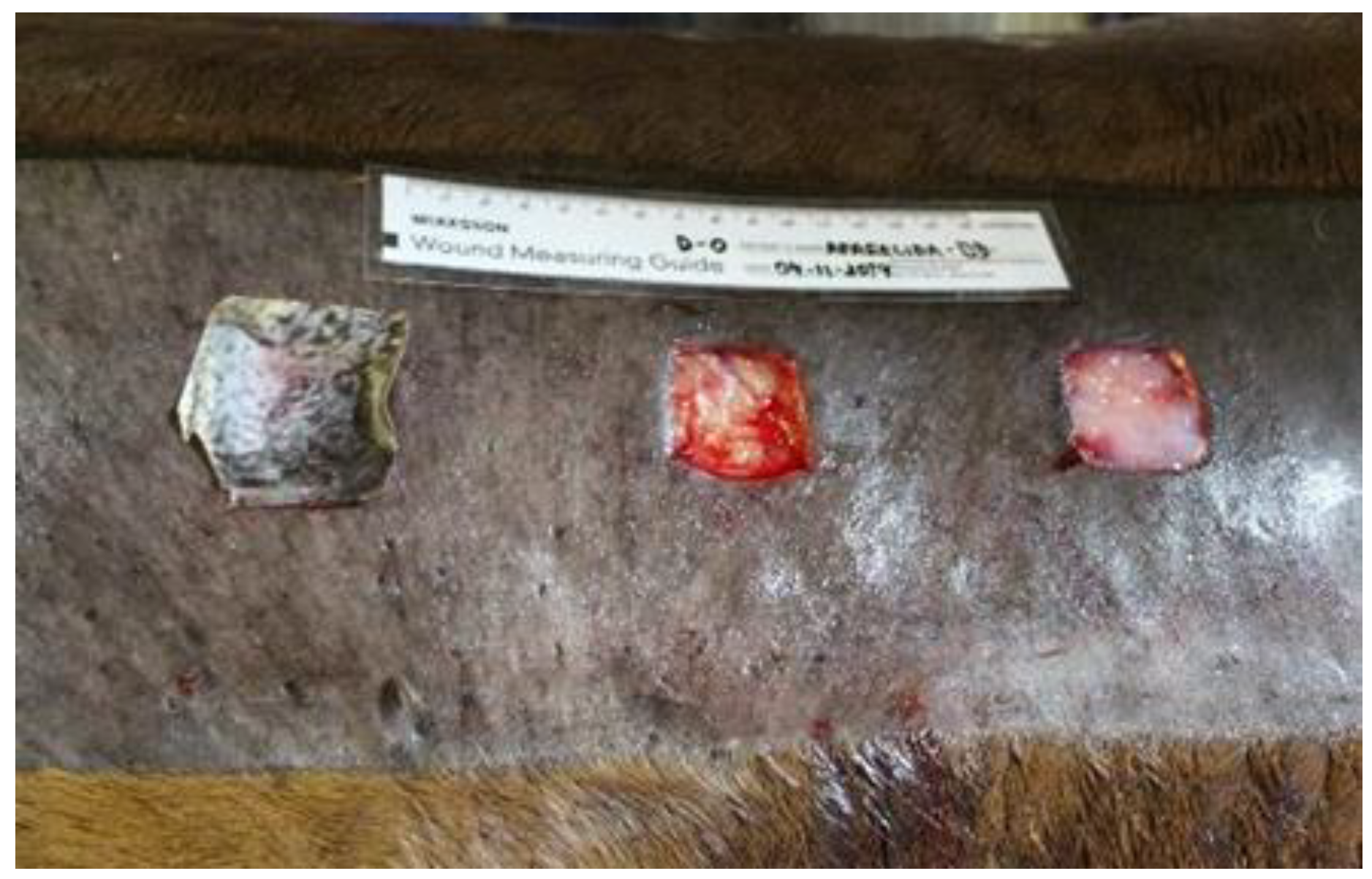
The wounds in the control group (CG) were treated only with Ringer's lactate solution and bandaged with gauze. The wounds selected for the bacterial cellulose hydrogel ointment with alginate (BCAG) received a layer of ointment, enough to cover their entire length and the other wound bed received frog skin (FSG) (
Figure 3). Before positioning the frog skin over the wound, it was cut with sterile scissors to the size of the recipient bed and rehydrated for 10 minutes in a sterile container with lactated ringer. The dressing followed the same pattern during the 28 days of experiment for the CG and BCAG, whereas in the FSG, the dressing was only performed when the skin moved or left the wound area.
On the sides chosen for clinical evaluation, after making the wounds, a photographic evaluation was carried out using a ruler, identifying the day of evaluation, animal number (01, 02, 03, 04 and 05), side of the back, and treated group. Treatments were carried out on all wounds, as follows: treatments for cranial wounds, medial wounds, and caudal wounds were randomly chosen. In animals 01 and 04, the cranial wounds were from the BCAG, the medial wounds from the CG, and the caudal wounds from the FSG. In animals 02 and 05, the cranial wounds were from the FSG, the medial wounds from the BCAG, and the caudal wounds from the CG. Finally, in animal 03, the cranial wounds were from the CG, the medial wounds from the FSG, and the caudal wounds from the BCAG.
The dressing was performed with gauze, fixed with microporous tape and finished with a bandage. Treatments were performed daily for 28 days. All five horses received phenylbutazone in the dosage of 4.4 mg.kg-1 intravenously (Equipalazone®, CEVA, Paulínia/SP, Brazil) immediately post-surgery and for three days, to control pain and edema.
2.6. Clinical and photographic evaluation of wounds.
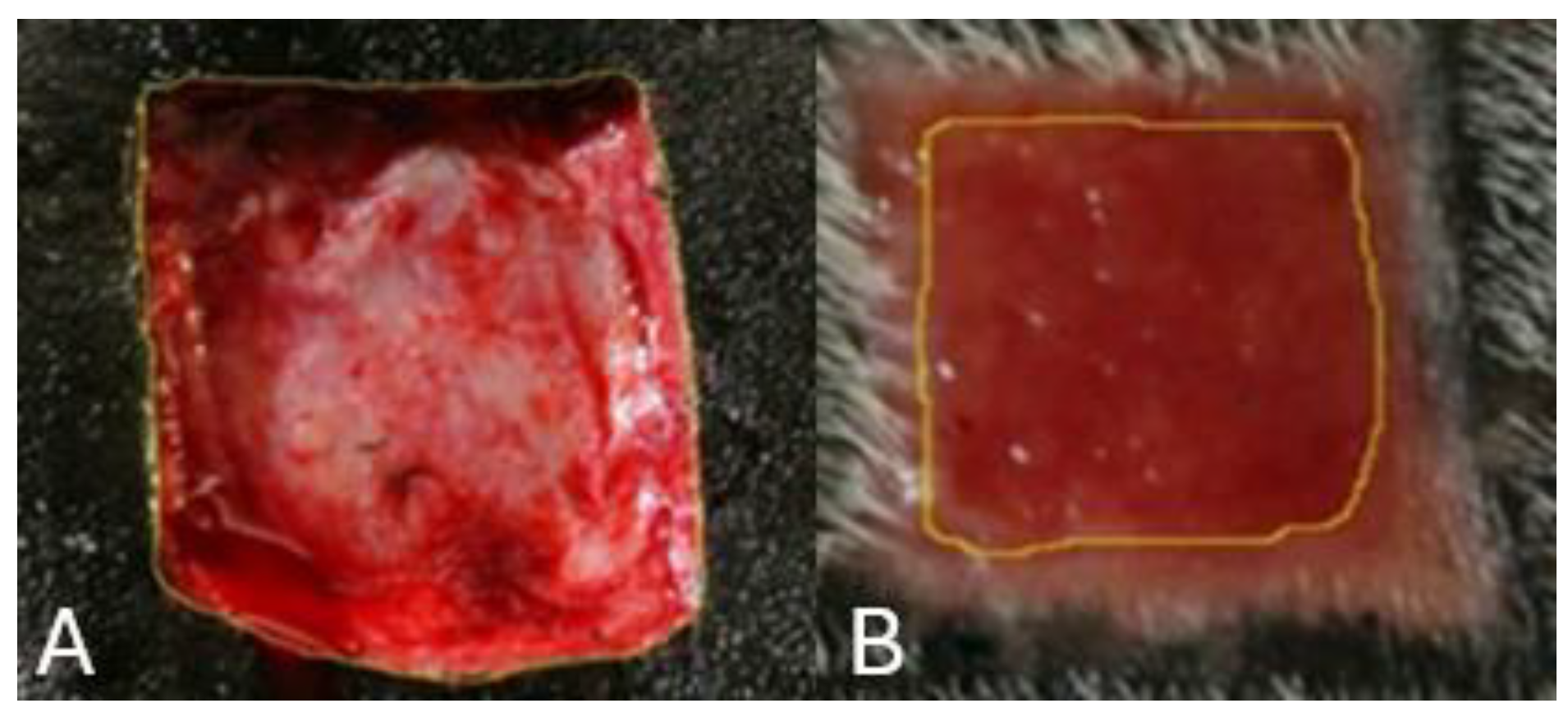
Clinical and photographic evaluations were carried out on the day of the surgery (D0) and on the third (D3), seventh (D7), fourteenth (D14), twenty-first (D21), and twenty-eighth (D28) days. In the clinical evaluation, the following parameters were observed: edema, hyperemia, local hemorrhage, crust, clot formation, presence and type of exudate, granulation tissue, and epithelialization. Through photographic monitoring, the calculation of the wound area and the contraction rate were objectively evaluated. The wound area was measured using Image J Software (Wayne Rasband National Institutes of Health, USA) (
Figure 4). The degree of wound contraction was calculated using the equation proposed by Ramsey and co-workers (1995) [
12]: contraction rate (%) = 100 x (W0 – WA)/W0, where W0 represents the original wound area immediately after its production and WA represents the wound area at the evaluation time (0, 3, 7, 14, 21, or 28 days), expressed as a percentage.
2.7. Biopsy and Histopathological Evaluation
Skin biopsies were performed with a 6 mm diameter punch, covering the boundaries of the wound and intact tissue, on the day of surgery (D0) and on days (D3), (D7), (D14), (D21), and (D28). The fragments were collected around the wound in a clockwise direction, ensuring that they were not taken from sites of previous biopsies. After removal, the fragments were fixed in 10% buffered formalin, pH 7.0, and sent to the Veterinary Pathology laboratory at the University of Brasília. The samples were routinely processed, and embedded in paraffin, and the 4 µm thick histological sections were stained with hematoxylin and eosin. In the semiquantitative microscopic analysis, the inflammation was evaluated; neutrophils, eosinophils, and monocytes, and the healing process; fibroblasts and neovascularization, and the presence of ulcers and hemorrhage. Pathological alterations were classified as (-) absent, (+) mild, (++) moderate, and (+++) marked.
2.8. Statistical Analysis
Data on wound area and contraction rate were subjected to analysis of variance in split plots with repeated measurements over time, followed by the Tukey test for comparisons of means. The data were tested for normality and homogeneity of variances, necessary prerequisites for analysis of variance. Statistics were considered significant when p<0.05. The median scores attributed in the histopathological evaluation were analyzed using the Kruskal-Wallis test (p<0.05). All analyses were performed using SAS software (v9.4, Cary, North Carolina), adopting a significance level of 5%.
4. Discussion
The shape and size of the mold for creating the wounds allowed the studied treatments to be evaluated for a sufficient period of time, as already used in other studies [
13,
14]. The shape of the wound does not affect healing time in horses [
15].
The use of non-steroidal anti-inflammatory drugs during the first three days aimed to reduce pain and edema, however some animals showed a mild degree of discomfort during the dressing in these first few days. The use of these drugs was also described in studies evaluating wound healing in horses [
14,
16,
17], with discomfort observed in the animals at the time of application of the dressing, shortly after the wounds were made [
18].
The anesthetic protocol established was sedation and local infiltrative anesthesia, which provided comfort to the animals throughout the period when the wounds were being made, and safety for the surgeon, who in turn could perform the entire procedure with peace of mind. The same anesthetic protocol was observed in other experimental studies [
14,
19].
Humans and horses present similarities in the natural healing of wounds. In addition to the development of distinct types of chronic wounds in both species, horses are considered a physiologically relevant model, both for the study of the mechanisms involved in wound repair, as well as for the evaluation of new therapies with possible applicability in the clinical routine of humans [
20].
The skin of humans and horses also presents similarities in terms of architecture, both comprising two main layers that protect the internal organs from mechanical damage and pathogen invasion. The epidermis, with 5 layers of stratified epithelial cells in the case of humans and stratified squamous cells in the case of horses, and the dermis which, in both species, is thick, but with sparse hair follicles in humans, while in horses, the hair follicles are densely distributed [
21,
22].
The determination of one side for macroscopic evaluation and the other for collecting biopsies, was performed to prevent the collections from influencing the measurement of the area and the degree of contraction of the wounds, as also carried previously [
16,
19].
Clinical evaluation of the wounds allowed us to observe an initial inflammatory phase in all animals in all groups, with hemorrhage, edema, hyperemia, and clots until the third experimental day, which were no longer being noticed on the seventh day, as reported by Gomez and co-workers (2004) [
18] during the first seven days of evaluation.
Seromucous exudate was observed from D3 to D7 in CG, and between D3 and D14 in BCAG and FSG, and in the latter group it had a characteristic odor of frog skin and a yellowish color. The exudate is beneficial, as the ability to provide a moist environment in the dressing has been identified as a favorable property for healing, as it contains nutrients and growth factors that stimulate the development of endothelial cells and dermal fibroblasts, favoring granulation and epithelialization [
16], which is one of the reasons why bacterial cellulose and alginate have been studied [
5,
6,
23]. However, this finding disagrees with that reported by Oliveira (2002) [
10] in which the porosity present in frog skin facilitated the passage of oxygen and, in turn, there was no accumulation of liquid and exudate in rabbits, nor accumulation of exudative secretions in humans [
11]. In the present study, regression of the exudate was observed during filling by granulation tissue, but it remained present until the wound was closed. In CG, the exudate was observed only until it was filled with granulation tissue, after which the wounds remained dry.
In general, all animals presented granulation tissue on D7, but not in the entire injured area, as observed in other studies [
14,
24]. FSG presented more advanced development in the initial formation between D14 and D21, with exuberant granulation tissue in 3 animals during this period, but on D28, all groups showed evident but incomplete wound contraction.
Epithelialization began on D14 in most animals, being more evident in BCAG on D21 and in CG on D28. In other studies, good epithelial growth was observed in wounds treated with frog skin in humans, without delay in healing [
11] and faster epithelialization in wounds treated with bacterial cellulose hydrogel in rats [
23], probably due to the humid environment that accelerated the epithelialization process, by supporting the migration of epithelial cells to the moist area of the wound. Also relevant, in BCAG, alginate hydrogels are normally used to release drugs and proteins, for the formation of films and microspheres, and in wound debridement, due to their absorbent characteristic [
25,
26], which can favor the earlier process of epithelialization.
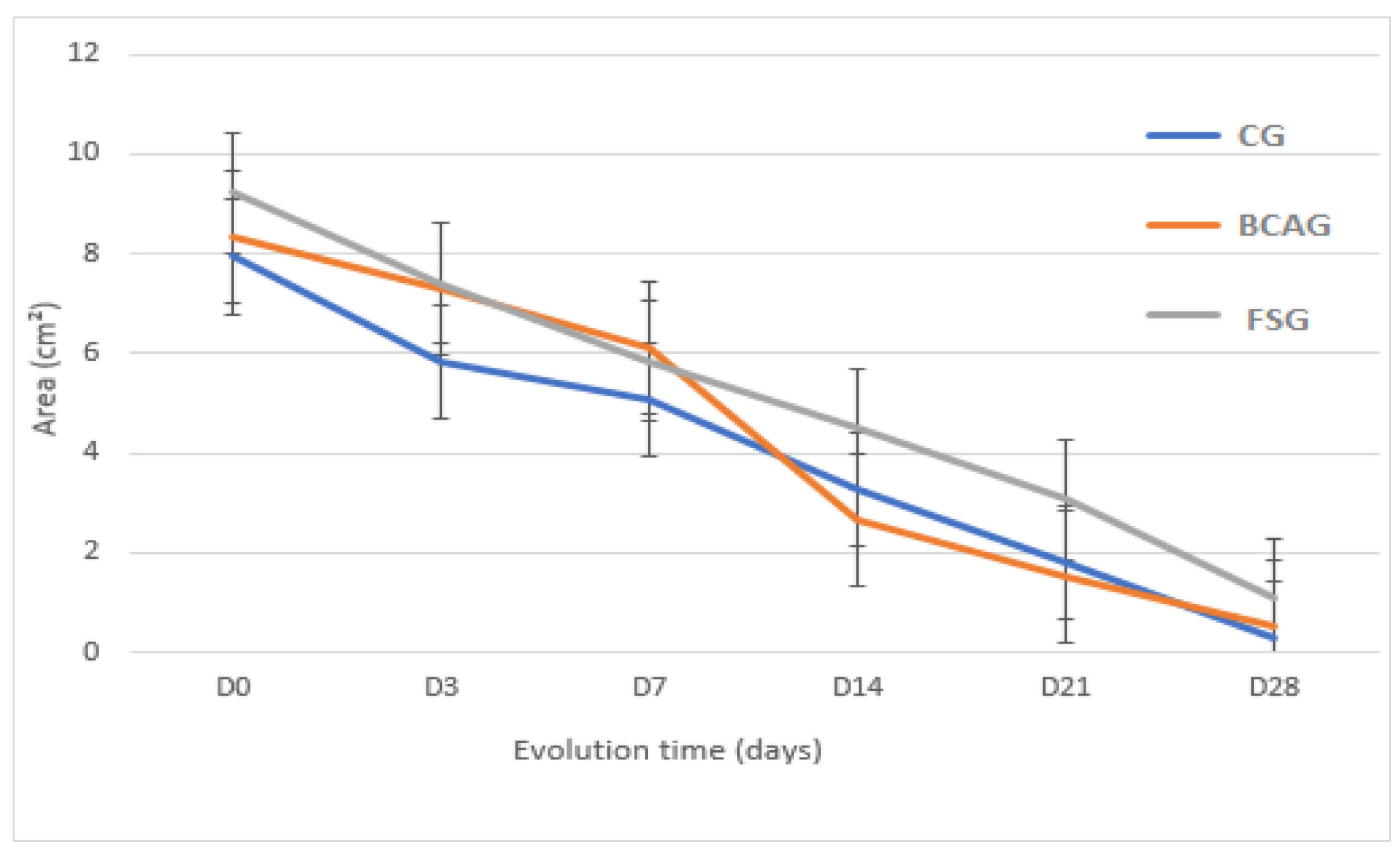
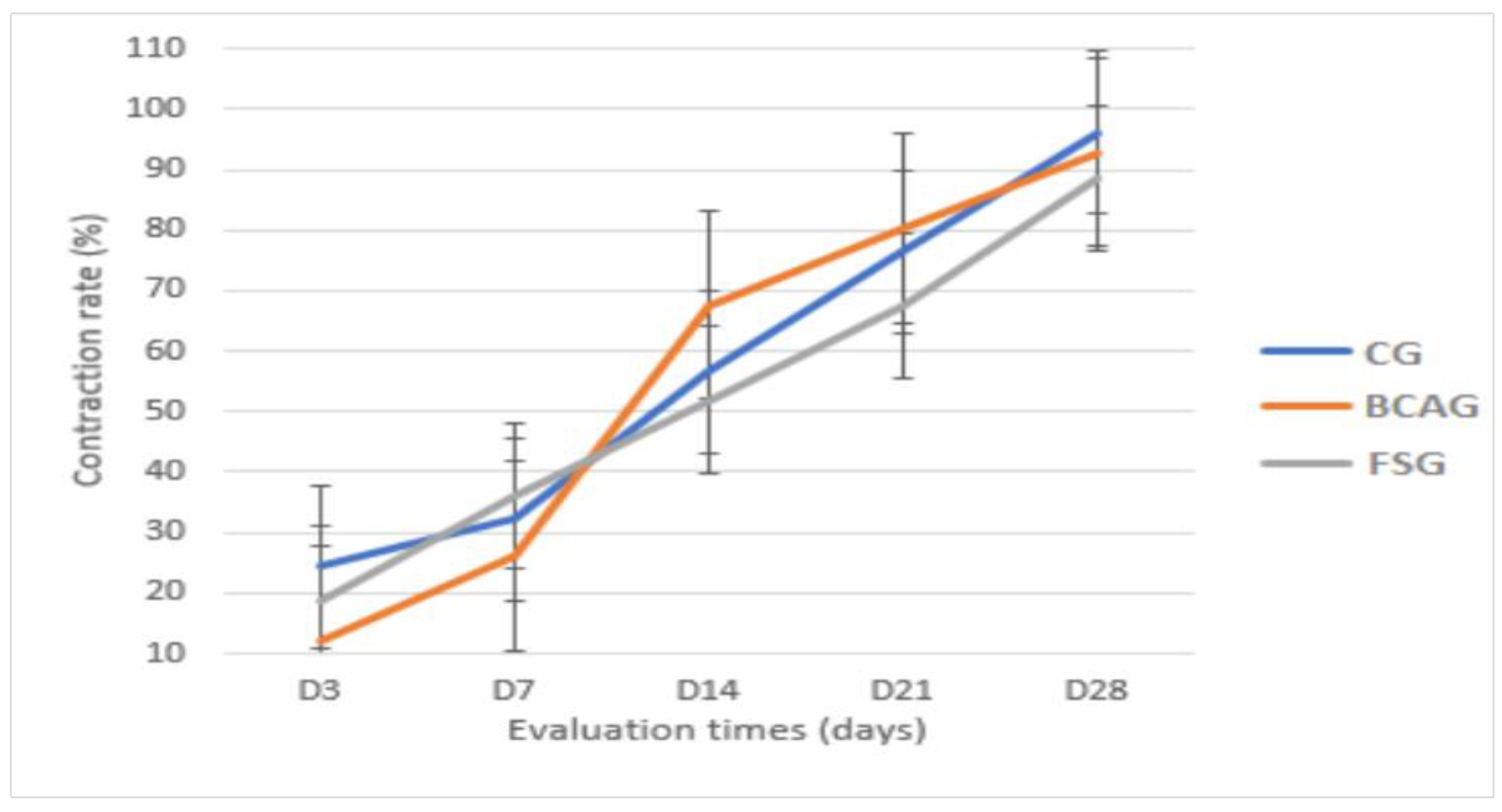
Therefore, it is believed that frog skin may favor the more accelerated formation of granulation tissue in the initial phase of healing, and bacterial cellulose with alginate may favor earlier epithelialization, which was observed in this study, as, although the average wound areas did not present statistical difference, BCAG presented a smaller wound area between D14 and D21 and a higher contraction rate. These data corroborate Pandey et al. [
23] who also did not find significant differences, but reported a reduction in the area and a higher rate of contraction of wounds treated with bacterial cellulose hydrogel, in relation to the control group, from the 7th day of evaluation, in rats. The faster healing of the hydrogel-treated group may be attributed to the moist environment, which accelerated the epithelialization process by supporting the migration of epithelial cells to the wet area of the wound [
27].
In all groups analyzed in the histopathological evaluations for the parameters of the presence of neutrophils, eosinophils, monocytes, fibroplasia, neovascularization, hemorrhage, and ulcer, there were small variations, but these remained within the normal ranges for the species, and statistical differences were not observed.
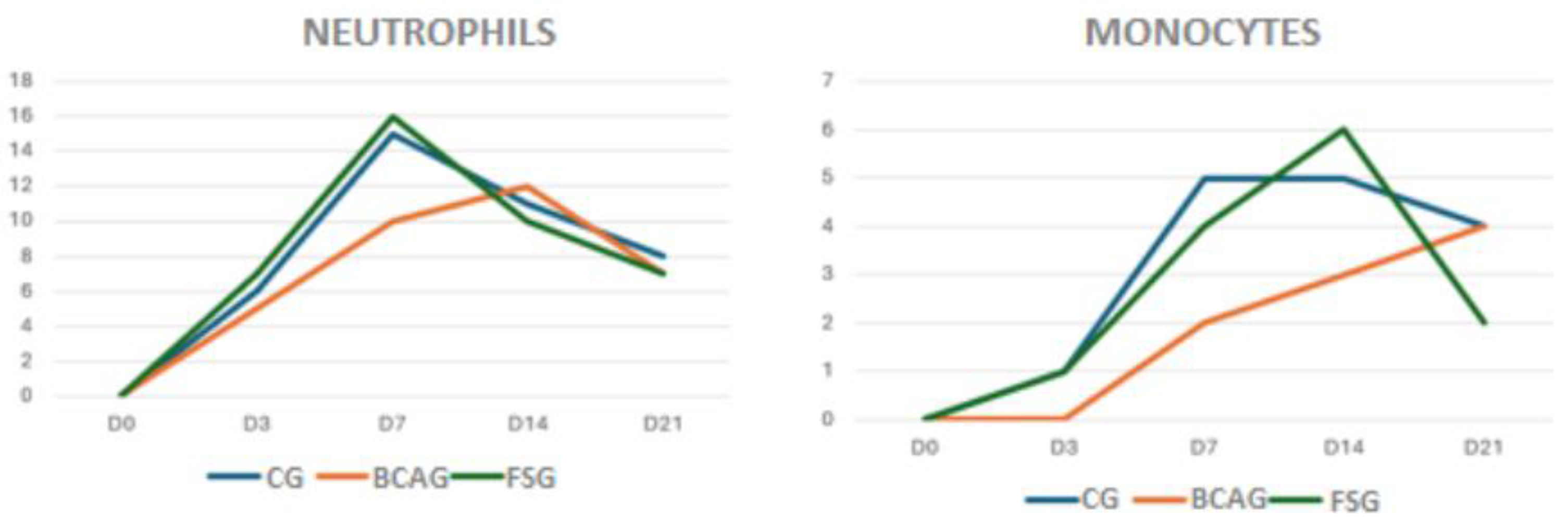
When evaluating neutrophils, higher values were observed on D7 in FSG, suggesting a greater inflammatory reaction in this group. Polymorphonuclear cells are the first cells to arrive at the wound site, shortly after the injury, reaching a maximum level within 48 hours, and are responsible for removing necrotic tissue and bacteria, in addition to releasing chemotactic factors that recruit macrophages. The presence of polymorphonuclear cells indicates a process of secretory activity, with removal of cellular debris from necrotic tissues and activation of the microbicidal function. Polymorphonuclear cells can also secrete anti-inflammatory cytokines, such as IL-10 [
28].
Regarding the assessment of monocytes, on D7 and D14 the CG and FSG groups presented higher values compared to BCAG, suggesting that the hydrogel promoted anti-inflammatory action in the wounds. The number of mononuclear cells tends to increase during the healing period, with polymorphonuclear cells being replaced by the mononuclear infiltrate [
29].
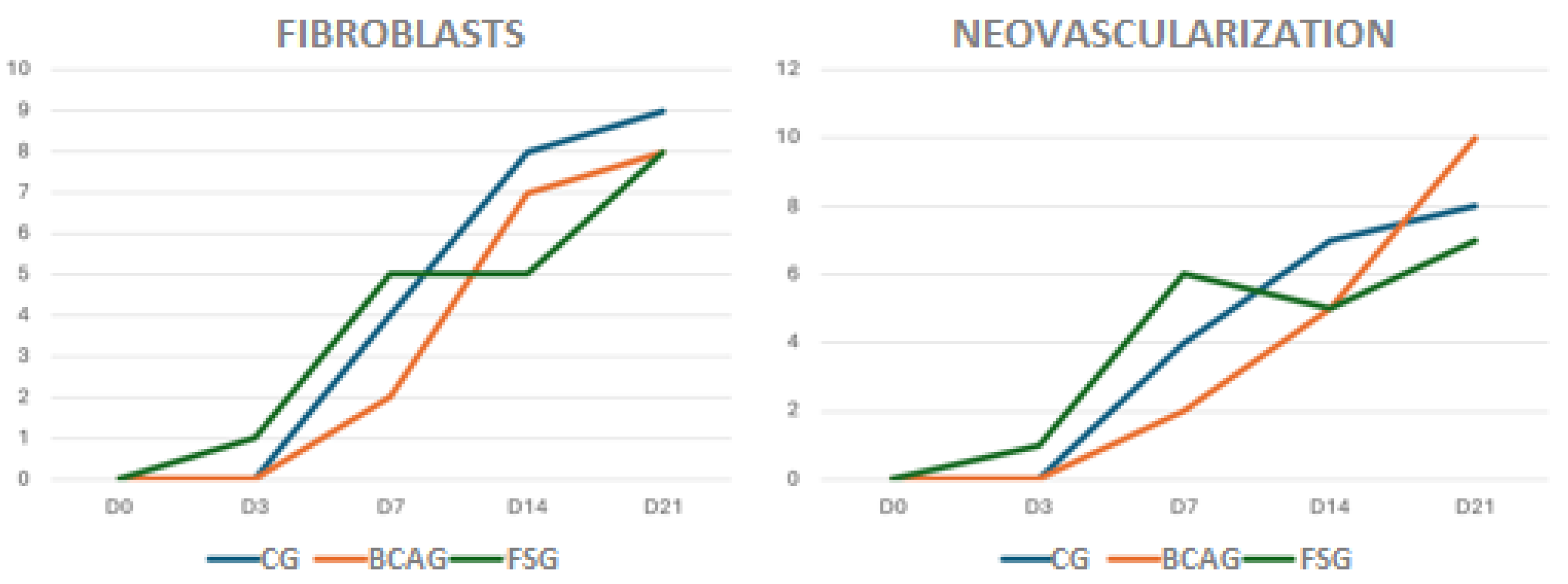
Fibroplasia indicates formation of the granulation tissue, extracellular matrix synthesis, and collagen deposition. Fibroblast synthesis was observed early and discreetly in FSG, however the phase of intense synthesis was observed on D14 and D21 in all groups. In rats, bacterial cellulose hydrogel accelerated the healing process by promoting re-epithelialization and proliferation of fibroblasts, promoting rapid healing in rats, compared to the control group [
23].
Although no significant differences were found between the groups, neovascularization occurred earlier on D7 in the FSG, remaining mild to moderate, a fact also reported in humans [
11]. On D21, greater neovascularization was observed in BCAG, corroborating Pandey and co-workers (2017) [
23] who reported greater neovascularization in wounds treated with bacterial cellulose hydrogel when compared to control wounds, a relevant result, since greater local vascularization can provide an increase in the concentration of cytokines associated with healing at the injury site. The use of bacterial cellulose gel in equine lumbar wounds also increased local perfusion and tissue metabolism, especially in the first 14 days of the healing process [
7].
Antimicrobial action was not the objective of investigation in this study, however viable skins of
Ranna catesbiana secrete several peptides, including relaxin, which have antimicrobial potential [
30]. No benefits were observed in the use of frog skin as a biological dressing on the healing of integumentary wounds produced in dogs [
31], however, the authors did not use any method to alter the antigenicity of the xenograft, such as the gamma radiation used in the current study and by Adorno (2005) [
11]. The use of gamma rays reduces antigenicity to food proteins and allergenicity [
32].
Although there was no statistical difference between the groups in wound healing in this study in horses, in clinical evaluations an early start of filling the area of the wounds treated with frog skin was observed, representing a safe, effective, and low-cost option, and favoring the re-epithelialization of wounds, as observed in humans [
11] and with tilapia skin xenografts, in wounds on equine limbs. [
33].
On D14 and D21, a smaller area and higher contraction rate were observed in BCAG, which can be attributed to more efficient re-epithelialization and fibroblast proliferation [
23]. The hydrogel proved to be easy to apply, maintaining contact across the entire length of the wound, forming a protective film, and promoting tissue healing.
Author Contributions
Conceptualization, Andressa B. Oliveira, José Adorno, Hernane S. Barud and Rita C. Campebell; methodology, Andressa B. Oliveira, Jéssyca L. A. Fagundes, Beatriz N. A. Fortes, Henrique C. Veado, Isabel L. Macedo, Márcio B. Castro, Bruno S. L. Dallago, Hernane S. Barud, José Adorno, Pablo A. V. Salvador, Paulo S. Santos and Rita C. Campebell; validation, Andressa B. Oliveira, Márcio B. Castro, José Adorno, Bruno S. L. Dallago, Hernane S. Barud, and Rita C. Campebell; formal analysis, Andressa B. Oliveira, Márcio B. Castro, Bruno S. L. Dallago, Hernane S. Barud, José Adorno and Rita C. Campebell; investigation, Andressa B. Oliveira, Jéssyca L. A. Fagundes, Beatriz N. A. Fortes, Henrique C. Veado, Isabel L. Macedo, Márcio B. Castro, Bruno S. L. Dallago, Hernane S. Barud, José Adorno, Pablo A. V. Salvador, Paulo S. Santos and Rita C. Campebell; data curation, Andressa B. Oliveira, Márcio B. Castro, Bruno S. L. Dallago and Rita C. Campebell.; writing— Andressa B. Oliveira and Rita C. Campebell; original draft preparation, Andressa B. Oliveira and Rita C. Campebell; writing—review and editing, Andressa B. Oliveira, Márcio B. Castro, Bruno S. L. Dallago, Hernane S. Barud, José Adorno and Rita C. Campebell; visualization, Rita C. Campebell; supervision, Rita C. Campebell; project administration, Rita C. Campebell. All authors have read and agreed to the published version of the manuscript.
Figure 1.
Frog skin packed in surgical grade paper, sterilized by gamma rays.
Figure 1.
Frog skin packed in surgical grade paper, sterilized by gamma rays.
Figure 2.
Performing the skin wounds on horses: (A) Mold marking; (B) Square-shaped wounds measuring 3cm on each side, including skin and subcutaneous tissue.
Figure 2.
Performing the skin wounds on horses: (A) Mold marking; (B) Square-shaped wounds measuring 3cm on each side, including skin and subcutaneous tissue.
Figure 3.
Treatment of wounds, where the frog skin group (FSG), control group (CG) and bacterial cellulose and alginate hydrogel group (BCAG) are observed, respectively, in the lumbar region of the horses.
Figure 3.
Treatment of wounds, where the frog skin group (FSG), control group (CG) and bacterial cellulose and alginate hydrogel group (BCAG) are observed, respectively, in the lumbar region of the horses.
Figure 4.
Measurement of the wound in the lumbar region of a horse, with the aid of Image J software. A) Wound area on D0. B) Wound area on D14.
Figure 4.
Measurement of the wound in the lumbar region of a horse, with the aid of Image J software. A) Wound area on D0. B) Wound area on D14.
Figure 5.
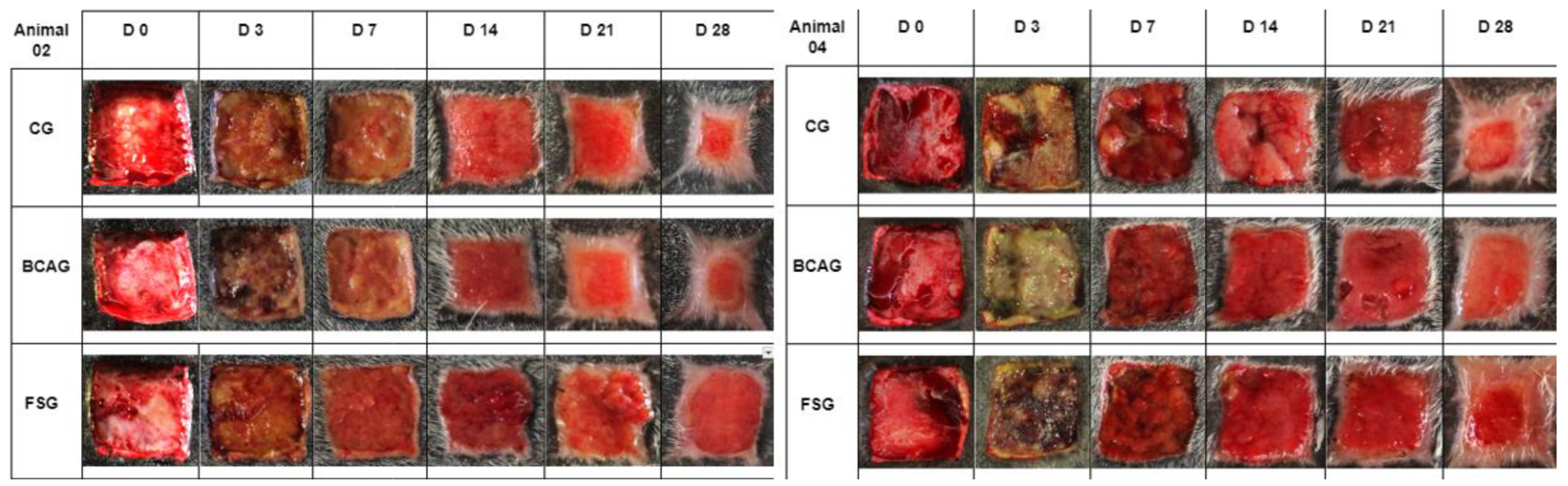
Photographic images of wounds in the lumbar region of horses 02 and 04, from the control group (CG), bacterial cellulose and alginate hydrogel group (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21) and 28 (D28).
Figure 5.
Photographic images of wounds in the lumbar region of horses 02 and 04, from the control group (CG), bacterial cellulose and alginate hydrogel group (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21) and 28 (D28).
Figure 6.
Mean wound areas in cm², in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 6.
Mean wound areas in cm², in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 7.
Mean wound contraction rate in percentages, in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 7.
Mean wound contraction rate in percentages, in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 8.
Relationship of neutrophils and monocytes, on a scale of mild, moderate, or severe, in wounds in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 8.
Relationship of neutrophils and monocytes, on a scale of mild, moderate, or severe, in wounds in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 9.
Relationship of fibroblasts and neovascularization, on a mild, moderate, or severe scale, in wounds in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Figure 9.
Relationship of fibroblasts and neovascularization, on a mild, moderate, or severe scale, in wounds in the lumbar region of horses in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Table 1.
Mean and standard deviation of wound areas (cm2) in the lumbar region of horses, in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Table 1.
Mean and standard deviation of wound areas (cm2) in the lumbar region of horses, in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
| Groups |
D0 |
D3 |
D7 |
D14 |
D21 |
D28 |
| CG |
7.94 ± 2.55 Aa
|
5.84 ± 1.22 ABa
|
5.07 ± 1.06 Ba
|
3.29 ± 1.68 BCa
|
1.79 ± 1.32 CDa
|
0.31 ± 0.30 Da
|
| CBAG |
8.32 ± 1.67 Aa
|
7.29 ± 1.55 Aa
|
6.11 ± 1.16 Aa
|
2.65 ± 0.99 Ba
|
1.54 ± 0.83 Ba
|
0.54 ± 0.31 Ba
|
| FSG |
9.23 ± 1.37 Aa
|
7.4 ± 1.46 ABa
|
5.85 ± 2.28 BCa
|
4.5 ± 2.08 CDa
|
3.08 ±1.60 DEa
|
1.09 ± 0.72 Ea
|
Table 2.
Mean and standard deviation of the contraction rate in percentages, of the lumbar region of horses, in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
Table 2.
Mean and standard deviation of the contraction rate in percentages, of the lumbar region of horses, in the control group (CG), group treated with bacterial cellulose hydrogel with alginate (BCAG), and frog skin group (FSG), on days 0 (D0), 3 (D3), 7 (D7), 14 (D14), 21 (D21), and 28 (D28).
| Groups |
D3 |
D7 |
D14 |
D21 |
D28 |
| CG |
24.40 ± 11.86Aa
|
32.28 ± 17.95ABa
|
56.57 ± 24.48BCa
|
76.51 ± 18.81CDa
|
96.08 ± 3.92Da
|
| CBAG |
12.24. ± 5.57Aa
|
26.07 ± 7.52Aa
|
67.68 ± 10.97Ba
|
80.34 ± 12.33Ba
|
92.87 ± 5.38Ba
|
| FSG |
18.83 ± 14.32Aa
|
36.14 ± 23.85ABa
|
51.95 ± 18.68BCa
|
67.39 ± 14.87CDa
|
88.60 ± 6.53Da
|