1. Introduction
An excess of reactive oxygen species and/or nitrogen species beyond cellular antioxidant capacity can result in oxidative damage to the lipids, proteins, and DNA, leading to cell membrane damage, loss of enzyme activities, and mutagenesis [
1]. Free radical molecules adversely affect health status and cause many major diseases [
1], including digestive diseases [
2]. Oxidative stress is essential in the pathogenesis and progression of inflammatory bowel disease and its relationship with the mucosal immune response [
3]. Consequently, oxidative stress generates inflammation in the intestinal wall and flattening of the gross folds of the intestinal lining, leading to a loss of surface area and a syndrome marked by a reduced ability to digest and absorb nutrients.
In rabbits, the causes of this syndrome are often multifactorial. An inappropriate diet or environmental conditions put them at a greater risk of developing it [
4]. Excessive production of reactive oxygen species that cannot be counteracted by the action of antioxidant enzymes in the body causes many diseases; the imbalance between the oxidant species and the antioxidant defense system can trigger specific factors responsible for oxidative damage in the cell [
5]. Therefore, antioxidant substances have been used to prevent or reduce damage to the gastrointestinal tract caused by oxidative stress. The resulting benefits of longevity, immunity, and health should interest those involved in the pet and fur trades because they keep rabbits for a long time. Furthermore, supplementing some antioxidants could improve productive performance, healthiness, and meat quality, which should interest rabbit meat producers [
6].
Synthetic antioxidants are commonly used in the food industry to prevent the deterioration of meat products. However, their use is increasingly limited because they are considered highly unstable. Furthermore, relevant legislation is becoming very strict as they are suspected to have carcinogenetic and mutagenic properties [
7]. Recently, there has been increasing interest in natural antioxidants extracted from plants, which can be used as alternatives to synthetic antioxidants; consumers prefer these natural and organic additives and ingredients because they are perceived as healthier and more sustainable [
8]. Phenolic compounds are secondary metabolites in plants with significant antioxidant and chelating properties; they have been widely studied in the production of livestock and rabbits [
6,
9,
10,
11]. Therefore, it could be hypothesized that plant-based dietary supplementation should be an alternative strategy to provide positive results without side effects.
Clitoria ternatea (CT) is an ornamental perennial climber from the Fabaceae family, commonly known as the butterfly pea. It is native to tropical equatorial Asia including Thailand. In human medicine, CT is thought to have been used in Ayurvedic medicine, one of the world’s oldest medical systems [
12]. A range of properties—including antioxidant, anti-inflammatory, analgesic, antipyretic, and antimicrobial properties—have also been reported in CT studies [
13,
14,
15,
16,
17], probably due to the presence of flavanol and anthocyanin glycosides, triterpenoids, tannins, rutin, and phytosterols [
12,
17].
In rabbits, CT has been used as a protein source rather than for its antioxidant abilities and offered directly to rabbits rather than as an extract [
18,
19]. Rabbit production in Thailand is intensive (50–200 reproductive uses), but it is a primary job for most farmers. However, health problems—mainly digestive and respiratory disorders—have been observed, with negative consequences [
20]. From this point of view, using tropical herb extracts to provide health benefits might be an effective strategy. Therefore, this study evaluated the possible effects of CT crude extract (CTE) supplementation on rabbits’ immunity and health status. Phagocytic activity, serum biochemistry, and intestinal histology were investigated. Furthermore, considering the importance of rabbits in human nutrition, digestibility and meat quality were also evaluated.
2. Materials and Methods
2.1. Plant preparation and extraction
CT (purple pedal breed) was planted at Chiang Mai Animal Husbandry and Research Center, Chiang Mai, Thailand. Plants were harvested at 90 days (during the flowering period). The extraction method was described by Suwannamanee [
21]. Briefly, whole plants (including leaves and flowers, but not roots) were chopped, mixed, milled using rubber-edged mills with water for 30 minutes, and filtrated. Subsequently, the solid part was separated from the liquid part by heating at 60°C for 40 minutes. The solid part was dried by incubating at 60°C for 72 hours, then powdered and kept in a desiccator at 4°C until use.
2.2. Animals, housing, diet, and recording
The experiment was carried out in the experimental facility of the Department of Large Animal and Wildlife Clinical Sciences, Faculty of Veterinary Medicine, Kasetsart University, Nakhon Pathom, Thailand. Sixteen crossbred rabbits (New Zealand white x Thai native breed; 484 ± 11.3g) were randomly housed in individual wire cages inside the facility and maintained at 23 ± 2°C (12h light: 12h dark) for the experimental period of 55 days. The rabbits were separated into two same-sex groups of eight individuals. CTE in water (0.5 g/kg body weight) was administered orally to the treated rabbits once a day at 0900 h for the entire experimental period; water (placebo) was administered to the control rabbits. A commercial diet (Lee Feed Mill, Publ. Co., Ltd., Phetchaburi, Thailand) and clean water were provided
ad libitum. The diets and CTE were analyzed in duplicate (
Table 1) for dry matter (DM), crude protein (CP), ether extract (EE), crude fiber, and ash by ignition to 550°C, according to guidelines from the Association of Official Analytical Chemists [
22]. The neutral detergent fiber, acid detergent fiber, and acid detergent lignin were determined according to the procedures of Van Soest et al. [
23]. The level of starch was determined using Ewer’s polarimetric method [
24]. The rabbits’ live weight and feeding intake were checked every two weeks. Mortality and morbidity were controlled daily by the same observer, according to Gidenne et al. [
25].
2.3. Apparent digestibility and digestive tract histology
According to the procedure of Kovitvadhi et al. [
11], feces were collected daily at 46 days for four days (
n = 8 per treatment), then weighed and stored at –20°C for chemical analysis in duplicate, recording DM, ash, EE, and CP, according to the A.O.A.C. [
22]. The apparent digestibility of DM and nutrients was calculated using the European standardized method [
26]. A 10mm length of the mid-jejunum and ileum (
n = 8 per group) was obtained after slaughter at 90 days and preserved in 10% buffered neutral formaldehyde solution (pH 7.2–7.4). Tissue samples were processed, embedded in paraffin, sectioned with 8µm thickness using a rotary microtome (Shandon AS325, Southeast Pathology Instrument Services, SC, USA), and stained with the hemolysin and eosin method. The height of the villi and the depth of the crypt were measured under a BX53 light microscope (Olympus, Tokyo, Japan) using an image analysis program (CellSens standard, Olympus Ver 01 07 imaging software, Tokyo, Japan).
2.4. Phagocytosis assay and serum biochemistry
At 75 and 90 days, blood samples were taken from the lateral saphenous vein (eight rabbits per group) and collected in a sterile tube containing heparin, whereas alveolar macrophage was taken by flushing of 10 mL of Roswell Park Memorial Institute 1640 Medium (RPMI; Sigma-Aldrich, MO, USA) into the lungs of slaughtered rabbits. Both samples were kept at 4ºC and immediately transported to the laboratory for phagocytosis assay. The polymorphonuclear leukocyte fraction was isolated from whole blood using PolymorphprepTM (Axis-shield, Oslo, Norway). This fraction and the alveolar macrophage were harvested and resuspended with RPMI medium with 10% fetal bovine serum (Sigma-Aldrich, MO, USA) to reach the concentrations of 75 x 104 and 20 x 104 cells/mL, respectively. These mixtures were incubated overnight in a 96-well plate at 37°C with 5% carbon dioxide. Subsequently, the phagocytosis assay was performed following the manufacturer’s instructions (CytoSelect TM, Cell Biolabs Inc., CA, USA), and the absorbance was read by a spectrophotometer (Infinite® F50, Tecan, Männedorf, Switzerland) at 450 nm. The results were expressed as optical density from ingested bacterial cells. Serum biochemistry was analyzed using standard protocols (Kasetsart University Veterinary Teaching Hospital, Kasetsart University, Nakorn Pathom, Thailand), where blood samples were collected from eight rabbits per group during slaughter.
2.5. Meat quality and lipid peroxidation
At slaughter, the carcasses were prepared following the indications of Kovitvadhi et al. [
11]. After 24 h of chilling, eight carcasses per group were halved, and then two longissimus dorsi (LD) muscles were excised. The left part of the LD muscle was used to assess pH after 24 h of chilling (pH24). An ultra basic portable pH meter (Denver InstrumentTM, NY, USA) was used, with a homogenized mixture of 10 g of LD and 100 mL of distilled deionized water to establish color (Color checker NR-1, Nippon Denshoku Industries, Tokyo, Japan). The approximate chemical composition (moisture, CP, EE and ash) was then analyzed according to the procedure of Kovitvadhi et al. [
11]. The right part of the LD was separated into four pieces to perform the thiobarbituric acid reactive substances (TBARS) assay after preservation at 4°C and covering with polyvinyl chloride wrap for 1, 3, 5, or 7 days. Fresh meat (5 g,
n = 8 per group, in duplicate) was homogenized with 25 ml of 7.5% trichloroacetic acid using a Polytron tissue homogenizer (Type PT 10-35, Kinematica GmbH, Luzern, Switzerland). Lipid peroxidation was determined using a modified TBARS method according to the protocol of Witte et al. [
27]. The results were expressed as µg malonyldialdehyde per kg of fresh meat, using a standard curve that covered a concentration range of 0.5–10 µM 1,1,3,3-tetramethoxypropane (Sigma-Aldrich, Steinheim, Germany). Absorbance was measured at 532 nm using a UV-1601 spectrophotometer (Shimadzu Corporation, Kyoto, Japan).
2.6. Statistical analysis
All statistical analyses were performed using the R statistical program with the package Rcmdr. Statistically significant differences between the groups in each parameter were analyzed using the Student t-test, while Welch’s t-test was applied when groups had unequal variances. Statistical significance was established at p < 0.05.
3. Results and Discussion
3.1. Apparent digestibility and digestive tract histology
No statistically significant differences were observed in terms of performance. However, the digestibility of organic matter and EE increased significantly (
p < 0.05) in rabbits fed CTE compared to the control group (
Table 2). Differences in the digestibility of DM and CP showed a slight trend toward significance (
p = 0.09 and 0.07, respectively). An improvement in digestibility of EE was observed in rabbits and broilers after supplementation with thyme (
Thymus vulgaris) [
10], essential oils of thyme and star anise as the main active component [
28] and the essential oil blend of oregano, cinnamon, and pepper [
29]. Digestibility may be improved by active compounds and/or functions that these plants share with CT. However, a study on the mechanisms of each chemical compound should be performed. Gut histology (villi height and crypt depth) is another possible indicator of digestive function, but there was no statistically significant difference in this study (
Table 2). However, villi height and crypt depth of the jejunum and ilium of CTE-fed rabbits tended to increase compared to control rabbits, possibly indicating improved digestion and absorption in the intestinal tract. Intestinal crypts are invaginations of the epithelium around the villi. They are lined by enzyme-secreting epithelial cells, and the base of the crypts is constantly dividing to maintain the villi structure [
30]. No morbidity or mortality was observed during the experimental period, reflecting good environmental conditions.
3.2. Phagocytic function and serum biochemistry
No statistically significant differences were observed regarding phagocytic activities (
Table 2). Phagocytosis is a nonspecific defense mechanism with five stages. Both neutrophils and macrophages have similar mechanisms during the destruction stage. Respiratory burst involves the production of potent oxidants, which is a chain reaction to eradicate ingested pathogens working with several enzymes. In a normal situation, glutathione, redox-active metals, and antioxidants such as vitamins C and E have enough potential to detoxify the end products after the destructive mechanism [
31]. However, unsuitable conditions can cause increased formation of oxidative molecules beyond the protective level, damaging cellular biomolecules [
6]. Therefore, supplementing exogenous antioxidants should optimize the eradicated mechanism, improving phagocytic function. Unfortunately, phagocytic activity was not improved in either circulating leukocytes or alveolar macrophages after CTE supplementation in this study. The reason may be that the experiment was conducted in normal conditions, as the formation of reactive oxygen species did not exceed the cellular antioxidant capacity. Therefore, clear effects on phagocytic function after supplementation should be observed in experiments with poor conditions, pathogen infection, or stimulated stress, each promoting increased oxidative stress.
In this study, blood chemistry was used to establish the physiological and biochemical status of the rabbits. Alanine aminotransferase (AST) and aspartate aminotransferase (ALT) were present in lower concentrations in treated rabbits than in the control group, but these differences were not significant (50.3 vs. 60.2 U/L,
p = 0.52, and 35.7 vs. 53.2 U/L,
p = 0.20, respectively;
Table 2). The hepatoprotective property of AST and ALT reduction, induced by acetaminophen with CTE supplementation at 200 mg/kg [
32] and other phytoadditions [
33], was reported in mice. An unclear result in the present study might be due to the study design, which did not include induction of hepatotoxicity [
33]. A decrease in blood nitrogen and creatinine was observed in rabbits with CTE supplementation compared to the control group (12.0 vs. 14.8%,
p = 0.05, and 0.86 vs. 1.08%,
p = 0.12, respectively;
Table 2), which was similar to a study on CTE addition to alloxan-induced diabetic rats [
34]. These results suggest that CTE may contain hepatoprotective or renal properties, although further study is required to confirm these benefits. Other parameters of serum biochemistry were unaffected by the treatment.
3.3. Meat quality and lipid peroxidation
The treated group was found to have heavier livers (6.78 vs. 5.41% of cold carcass weight, SEM = 0.33,
p = 0.03), while carcass traits and meat chemical composition did not differ between groups (
Table 2). Increased liver weight in treated rabbits may be due to enhanced liver function and/or fat storage. However, there were no clinical signs or gross lesions due to hepatic lipidosis or liver enlargement in the treated rabbits. In addition, serum AST and ALT, which indicate liver damage, did not increase. For these reasons, it can be concluded that CTE supplementation did not adversely affect liver function. CTE supplementation decreased pH
24, yellowness (b*), hue angle (H*), and chromosome (C*) while increasing the lightness (L*) of rabbit meat (
Table 2). Meat color influences consumers’ purchasing decisions, and surface discoloration can lead to rejection by consumers, directly influencing economic losses. In most studies, antioxidant supplementation does not influence meat color [
6]. However, higher meat brightness was reported after the dietary addition of vitamin E in rabbits [
35] and of CTE in broilers [
21] and rabbits in this study. The reason may be the protection of the cellular membrane by antioxidants, reducing the loss of liquids into the extracellular space and thus increasing light refraction and maintaining a bright color. Furthermore, this mechanism might lead to higher moisture in the meat of treated rabbits, increasing the meat’s brightness (
Table 2). Lo Fiego et al. [
35] found a negative correlation between pH
24 and L* in rabbit meat, as was also found in the present study. The lower pH
24 in CTE-treated rabbits should be beneficial because proteolytic bacteria developed rapidly when the pH value was around 6 [
36]. Lower yellowness in the CTE-treated group may lead to a smaller hue angle and lower saturation. The small amount of fat in the meat of treated rabbits may be related to a lower yellowness value compared to the control group. Rabbit meat is considered healthy from a commercial point of view [
36]; therefore, brighter and less yellow meat should promote attention from consumers who aim to eat healthily.
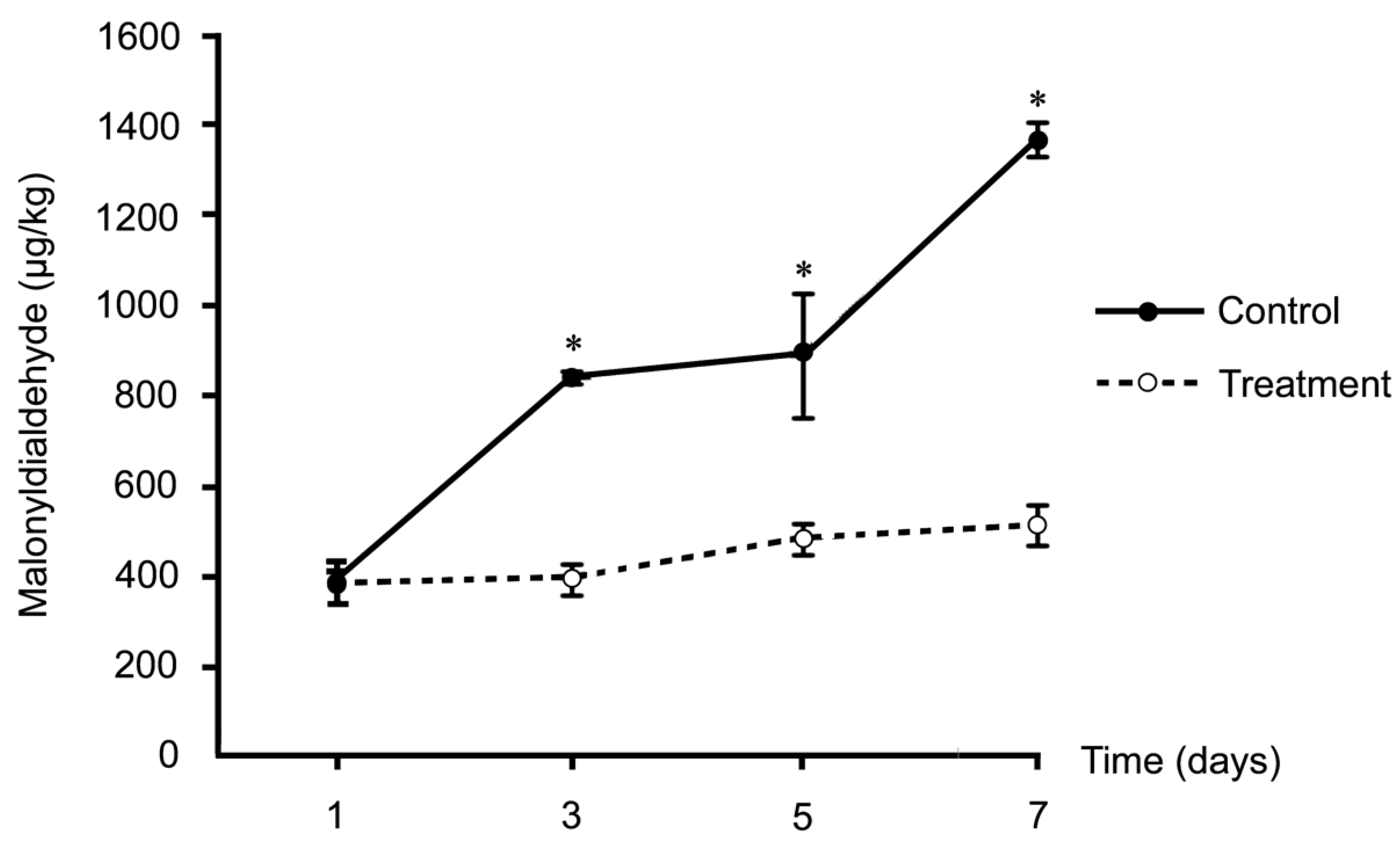
The level of lipid peroxidation did not differ between the two groups on the first storage day, but this chemical reaction was clearly delayed in the treated group after 3, 5, and 7 storage days, compared to the control group (
p < 0.05;
Figure 1). High levels of polyunsaturated fatty acids are characteristic of rabbit meat, reducing meat quality during storage in terms of nutritive value and safety from lipid peroxidation. However, supplementation with antioxidant-containing substances can prevent or slow this reduction. In an in vitro study, In an in vitro study, CTE had antioxidant activities in a 2,2-diphenyl-1-picrylhydrazyl radical (DPPH) scavenging assay, and it was suggested that anthocyanin is the main active chemical compound responsible for these activities [
15,
16,
32]. Increased DPPH in broiler thigh and breast meat was reported following dietary CTE-supplementation of 0.25 or 0.5% in the diet [
21], the same level as in the present study. These beneficial results in preventing lipid peroxidation in meat should confirm the bioavailability of CTE following oral supplementation. However, further studies should be performed on isolating the responsible compounds and their mode of action.
Figure 1.
Muscular lipid peroxidation-malonyldialdehyde (MDA) formation in control (n=8) and Clitoria ternatea extract (CTE) treated (n=8) rabbits during 7 days of storage.
Figure 1.
Muscular lipid peroxidation-malonyldialdehyde (MDA) formation in control (n=8) and Clitoria ternatea extract (CTE) treated (n=8) rabbits during 7 days of storage.
4. Conclusion
this study indicated that supplementation of CT crude extract in rabbit diets had no effect on phagocytic function but did improve digestibility. Modification of the meat color and delayed lipid peroxidation were also observed. Therefore, supplementation at 0.5 g/kg body weight could be suggested in the production of rabbits for meat consumption, prolonging shelf life by extending lipid peroxidation.
Author Contributions
Conceptualization, A.K., I.G., I.Z., and T.R.; methodology, A.K., I.G., I.Z. and T.R.; formal analysis, A.K.; writing—original draft preparation, A.K. and T.R.; writing—review and editing, T.R.; visualization, A.K.; supervision, I.G., I.Z. and T.R.; project administration, A.K. and T.R.; funding acquisition, T.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research and the APC was funded by Faculty of Veterinary Medicine, Kasetsart University.
Institutional Review Board Statement
The present study was conducted under animal care approved by the Institutional Animal Care and Use Committee in accordance to the guidelines of animal care and use under the Ethics Board of the Office of National Research Council of Thailand (NRCT) for the conduction of the scientific research. The approved number was ACKU61-VET-001..
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Acknowledgments
This study was financially supported by the Department of Large Animal and Wildlife Clinical Sciences, Faculty of Veterinary Medicine, Kasetsart University. Dr. Supachai Sritiwong is thanked for providing the butterfly pea extracts for the whole experiment.
Conflicts of Interest
The authors declare that there is no conflict of interest with any financial organizations with respect to the material discussed in the manuscript.
References
- Devasagayam TPA, Tilak JC, Boloor KK, Sane KS, Ghaskadbi SS, Lele RD. Free radicals and antioxidants in human health: current status and future prospects. J Assoc Phys India 2004;52:794-804.
- Liu Z, Ren Z, Zhang J, Chuang CC, Kan-daswamy E, Zhou T, et al. Review article. Role of ROS and nutritional antioxidants in human diseases. Front Physiol 2018;9:477. [CrossRef]
- Tian T, Wang Z, Zhang J. Phatomechanisms of oxidative stress in inflammatory bowel disease and potential antioxidant therapies. Oxid Med Cell Longev 2017;4535194. [CrossRef]
- Oglesbee BL, Lord B. Gastrointestinal diseases of rabbits. In: Ferrets, rabbits, and rodents: Clinical medicine and Surgery (4th ed.). 2020;174-187p. [CrossRef]
- Skowron M, Zalejska-Fiolka J, Blaszczyk U, Chwalinska E, Owczarek A, Brikner E. Antioxidant enzyme activities in rabbits under oxidative stress induced by hight fat diet. J Vet Res 2018;62:199-205. [CrossRef]
- Abdel-Khalek, AM. Supplemental antioxidants in rabbit nutrition: A review. Livest Sci 2013;158:95-105. [CrossRef]
- Krishnaiah D, Sarbatly R, Nithyanandam R. A review of the antioxidant potential of medicinal plant species. Food Bioprod Process 2011;89:217-33. [CrossRef]
- Brewer, MS. Natural antioxidants: sources, compounds, mechanisms of action, and potential applications. Compr Rev Food Sci Food Saf 2011;10:221-47. [CrossRef]
- Christaki E, Bonos E, Giannenas I, Florou-Paneri P. Aromatic plants as a source of bioactive compounds. Agriculture 2012;2:228-43. [CrossRef]
- Gerencsér ZS, Szendrő ZS, Matics ZS, Radnai I, Kovacs M, Nagy I, et al. Effect of Dietary supplementation of Spirulina (Arthrospira platensis) and Thyme (Thymus vulgaris) on apparent digestibility and productive performance of growing rabbits. World Rabbit Sci 2014;22: 1-9. [CrossRef]
- Kovitvadhi A, Gasco L, Ferrocino I, Rotolo L, Dabbou S, Malfatto V, et al. Effect of purple loosestrife (Lythrum salicaria) diet supplementation in rabbit nutrition on performance, digestibility, health and meat quality. Animal 2016;10:10-8. [CrossRef]
- Mukherjee PK, Kumar V, Kumar NS, Heinrich M. The ayurvedic medicine Clitoria ternatea- from traditional use to scientific assessment. J Ethnopharmacol 2008;20:291-301. [CrossRef]
- Devi BP, Boominathan R, Mandal SC. Anti-inflammatory, analgesic and antipyretic properties of Clitoria ternatea root. Fitoterapia 2003;74:345-9. [CrossRef]
- Ponnusamy S, Gnanaraj WE, Antonisamy JM, Selvakumar V, Nelson J. The effect of leaves extracts of Clitoria ternatea Linn against the fish pathogens. Asian Pac J Trop Med 2010;3:723-6. [CrossRef]
- Mukhopadhyay R, Bhattacharya S, Biswas M. In vitro free radical scavenging activity of Clitorea ternatea leaf extracts. J Adv Pharm Educ Res 2012;2:206-9.
- Rabeta MS, An Nabil Z. Total phenolic compounds and scavenging activity in Clitoria ternatea and Vitex negundo linn. Int Food Res J 2013;20:495-500.
- Verma PR, Itankar RP, Arora SK. Evaluation of antidiabetic antihyperlipidemic and pancreatic regeneration, potential of aerial parts of Clitoria ternatea. Rev Bras Farmacogn 2013;23:819-29. [CrossRef]
- Lowry JB, Schlink AC, Hoffmann D. Evaluation of three tropical legumes in diets for growing rabbits. Asian Austral J Anim Sci 1992;5:257-9. [CrossRef]
- Elamin KM, Elkhairey MA, Ahmed HB, Musa AM, Bakhiet AO. Effect of different feeds on performance and some blood constituents of local rabbits. Res J Vet Sci 2011;4:37-42. [CrossRef]
- Kovitvadhi, A. , Chandang, P., Tirawattanawanich, C., Kasemsuwan, S., Rukkwamsuk, T. The study on cluster and obstacles of meat rabbit production in Thailand. J Mahanakorn Vet Med 2019;4:115-27.
- Suwannamanee J, 2014. The utilization of extracted butterfly pea concentrate as a supplement for broiler chickens. (unpublished M.S. thesis, Chiang Mai Rahabhat University).
- A.O.A.C. 2000. Official Method of Analysis, 17th edition, Association of Official Analytical Chemists. Gaithersburg, MD, USA.
- Van Soest PJ, Robertson JB, Lewis BA. Methods for dietary fibre, neutral detergent fibre, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 1991;74: 3583-97. [CrossRef]
- European Economic Community. Analytical determination of starch. OJEC 1972;123:7.
- Gidenne T, Combes S, Feugier A, Jehl N, Arveux P, Boisot P, et al. Feed restriction strategy in the growing rabbit. 2. Impact on digestive health, growth and carcass characteristics. Animal 2009;3:509-15. [CrossRef]
- Perez JM, Lebas F, Gidenne T, Maertens L, Xiccato G, Parigi-Bini R, Dalle Zotte A, et al. European reference method for in vivo determination of diet digestibility in rabbits. World Rabbit Sci 1995;3:41-3. [CrossRef]
- Witte VC, Krause GF, Bailey ME. A new extraction method for determining 2- thiobarbituric acid values of pork and beef during storage. J Food Sci 1970;35:582-5. [CrossRef]
- Amad AA, Männer K, Wendler KR, Neumann K, Zentek J. Effects of a phytogenic feed additive on growth performance and ileal nutrient digestibility in broiler chickens. Poult Sci 2011;90:2811-6. [CrossRef]
- Hernández F, Madrid J, García V, Orengo J, Megías MD. Influence of two plant extracts on broilers performance, digestibility, and digestive organ size. Poult Sci 2004;83:169-74. [CrossRef]
- Chwen LT, Foo HL, Than NT, Choe DW. Growth performance, plasma fatty acids, villous height and crypt depth of preweaning piglets fed with medium chain triacylglycerol. Asian Australs J Anim Sci 2013;26: 700-4. [CrossRef]
- Tizard, I. 2013. Veterinary Immunology, 9th edition, Elsevier Saunders, St. Louis, MO, USA.
- Nithianantham K, Ping KY, Latha LY, Jothy SL, Darah I, Chen Y, et al. Evaluation of hepatoprotective effect of methanolic extract of Clitoria ternatea (Linn.) flower against acetaminophen-induced liver damage. Asian Pac J Trop Dis 2013;3:314-9. [CrossRef]
- Kumar CH, Ramesh A, Kumar JNS, Mohammed ishaq B. A review on hepatoprotective activity of medicinal plants. Int J Pharm Sci Res 2011;2:501-515. [CrossRef]
- Daisy P, Santosh K, Rajathi M. Antihyperglycemic and antihyperlipidemic effects of Clitoria ternatea Linn. in alloxan-induced diabetic rats. Afr J Microbiol Res 2009;3: 287-91.
- Lo Fiego DP, Santoro P, Macchioni P, Mazzoni D, Piattoni F, Tassone F, et al. The effect of dietary supplementation of vitamins C and E on the a-tocopherol content of muscles, liver and kidney, on the stability of lipids, and on certain meat quality parameters of the longissimus dorsi of rabbits. Meat Sci 2004;67: 319-327. [CrossRef]
- Dalle Zotte, A. Perception of rabbit meat quality and major factors influencing the rabbit carcass and meat quality. Livest Prod Sci 2002;75:11-32. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).