1. Introduction
With overfishing and declining fish stocks, alternative sources of dietary protein have become ever more important and of increasing interest in recent years, especially in aquaculture [
1].
Tenebrio molitor (TM) (Coleoptera: Tenebrionidae), commonly known as yellow mealworm, is increasingly being used as a feed ingredient and substitute for fish meal in aquaculture [
2]. On a proximal composition basis, TM is rich in crude proteins and lipids and low in ash and crude fibers [
3], with an abundance of essential amino acids (EAAs) [
4] and distinct fatty acids profile [
5].
Several authors conducted analyses on the impact of incorporating mealworms into livestock feeds to assess their potential and their effects on livestock growth, animal health, and meat quality. Numerous studies have indicated the efficacy of substituting fishmeal with mealworm meals in aquaculture production, particularly at inclusion rates of up to 25% [
6]. Mealworm-based feeds have received positive evaluations for various freshwater species such as Nile tilapia (
Oreochromis niloticus) [
7,
8,
9], tench (
Tinca tinca) [
10], rainbow trout (
Oncorhynchus mykiss) [
11,
12,
13,
14,
15,
16,
17,
18,
19], yellow catfish (
Pelteobagrus fulvidraco) [
20], black bullhead catfish (
Ameiurus melas) [
21], European perch (
Perca fluviatilis) [
22,
23,
24], African catfish (
Clarias gariepinus) [
25] and several marine water species such as gilthead sea bream (
Sparus aurata) [
1,
11,
12,
13,
26,
27,
28,
29], European sea bass (
Dicentrarchus labrax) [
11,
13,
26], blackspot sea bream (
Pagellus bogaraveo) [
30], olive flounder (
Paralichthys olivaceus) [
31], red sea bream (
Pagrus major) [
32], Pacific white shrimp (
Litopenaeus vannamei) [
33], pearl gentian grouper (
Epinephelus lanceolatus♂
x Epinephelus fuscoguttatus ♀) [
34] and black rockfish (
Sebastes schlegeli) [
35]. Despite the positive evaluations, it should be noted that high levels of inclusion, particularly in fish juveniles, could potentially result in performance decrease [
3,
35].
In recent years the medicinal / aromatic plants (MAPs) market is resilient in growth and development in many Mediterranean countries [
36], while India, Brazil, China, Indonesia, and USA [
37], are the top essential oils (EOs) producing countries [
38]. The Mediterranean has been characterized as a biodiversity hotspot, with exceptional numbers of endemic plants. For instance, more than 7000 native plant taxa occur in the Greek region, with 22% being endemic [
39]. The MAPs market focuses mainly on industrial applications [
37]. The expanding demand for EOs and extracts of MAPs can be attributed to their diverse and extensive applications in various sectors such as cosmetic and fragrance industries, food flavoring industries and industrial solvents [
40].
EOs yield performances fluctuate among MAPs due to different harvest strategies, harvest intensity and hydrological annual profiles [
40]. Nonetheless, the post-distillation residual plant biomass results in a substantial number of wasted by-products due to its significant volume [
41]. These by-products contain phenolic compounds [
42] and if used as feed additives can confer antioxidant and inhibitory effects upon fish and pathogens growth, respectively [
11,
43,
44,
45].
However, adjusting the composition of a diet can potentially result in nutrient deficiencies, particularly when dealing with formulated diets that rely on feedstuff processing techniques, which greatly influence the bioavailability of nutrients [
46]. Such fish feed modifications can lead to diet-induced oxidative stress [
26,
47] thus affecting numerous cellular mechanisms. Heat shock proteins (HSPs), which as molecular chaperones assist in the conformational folding and translocation of proteins [
48,
49] are implicated in the immediate response to exposure to a multitude of stressors [
50]. External stimuli including dietary-related factors [
47,
51] may also modulate the activation of Mitogen-activated protein kinases (MAPKs), which transduce signals for the regulation of several cellular processes such as development and apoptosis [
52,
53]. The latter is crucial under the stress regime for the homeostatic removal of damaged or superfluous cells [
54].
This paper aims to evaluate the impact of the use of T. molitor larvae as a partial fish meal substitute and at the same time examine if the enrichment of the substrate of the larvae with post-distillation MAP residues can benefit and differentiate the produced insect meal from the conventional meals, by examining different stress and inflammatory biomarkers in the blood of gilthead seabream fed these meals. In specific, peripheral white blood cells activation, heat shock proteins, mitogen activated protein kinases and the apoptotic machinery were assessed.
2. Materials and Methods
2.1. Rearing of Tenebrio molitor and production of insect meals
Το produce the insect meals examined in this trial,
T. molitor larvae were reared in two different substrates. The first meal (“Conventional”) was created from larvae reared on a conventional substrate, while the second meal (“Enriched”) was created from insects reared on a substrate partially enriched (20%) with plant material from processed residues of distillation of medicinal aromatic plants: Greek oregano
(Origanum vulgare subsp.
hirtum), thymus (
Thymus vulgaris), sage (
Salvia officinalis), rosemary (
Rosmarinus officinalis) and their essential oils, linseed (
Linum usitatissimum), sea fennel (
Crithmum maritimum), and olive (
Olea europaea subsp
. europaea). All the above were added in analogous percentages corresponding to the 20 % of the insect diet (approx. 2.9% each). Insects were reared for a period of four months in total, starting from newly-hatched larvae until the stage of late-instar larvae, i.e. prior to pupation, as suggested by Rumbos et al. [
55], which was the instar that was used in the feeding trials. After collection, the insect larvae were air-dried for 48 hours at temperature 38°C, using a VK981RH oven (Vencon-N.Varsos A.E., Athens, Greece). The dried material was kept frozen (-20°C) until being used for the preparation of the fish diets. In the Conventional and Enriched rearing substrates, the protein content of the insect meal was 56.25% and 55.16%, respectively. The total phenolics (g GAE/g DW) content of the insect meal in the Conventional and Enriched insect rearing substrates was 314.10% and 257.26%, respectively.
2.2. Fish Diets
Three different diets were prepared as described in Table 2: a) A diet for the control group that was prepared with fishmeal as the main protein source (hereinafter Control Diet); b) A diet with partial substitution of fishmeal with 10% of the “Conventional” insect meal (hereinafter TM-10); c) A diet with partial substitution of fishmeal with the “Enriched” insect meal (hereinafter MAP-TM-10). The diet for the control group was prepared by mixing a commercial feed (Praxis Fish Feeds, Piraeus, Greece) appropriate for gilthead sea bream (Sparus aurata) with fish meal and wheat (extruded). The diets for the other two groups were prepared by combining the same commercial feed with either the “Conventional” (TM-10) or the “Enriched” (MAP-TM-10) dried insect meals.
For the preparation of each diet, the feed ingredients were initially ground in a hammer mill, then mixed, pelleted (5 mm) using a pellet mill, air-dried (25°C), and stored. The three diets were formulated to be isonitrogenous and isocaloric and to meet the nutrient requirements of the gilthead seabream. The AA content of each feed was analyzed in a near infrared reflectance spectrometer (DA 7250 NIR Perten analyzer, Perkin Elmer), crude protein content of the insect meals was analyzed following the guidelines outlined in the Association of Official Chemists (AOAC) methods [
56]. The total phenolic content of the diets was analyzed with the Folin–Ciocalteu method as described by Vasilopoulos et al [
57].
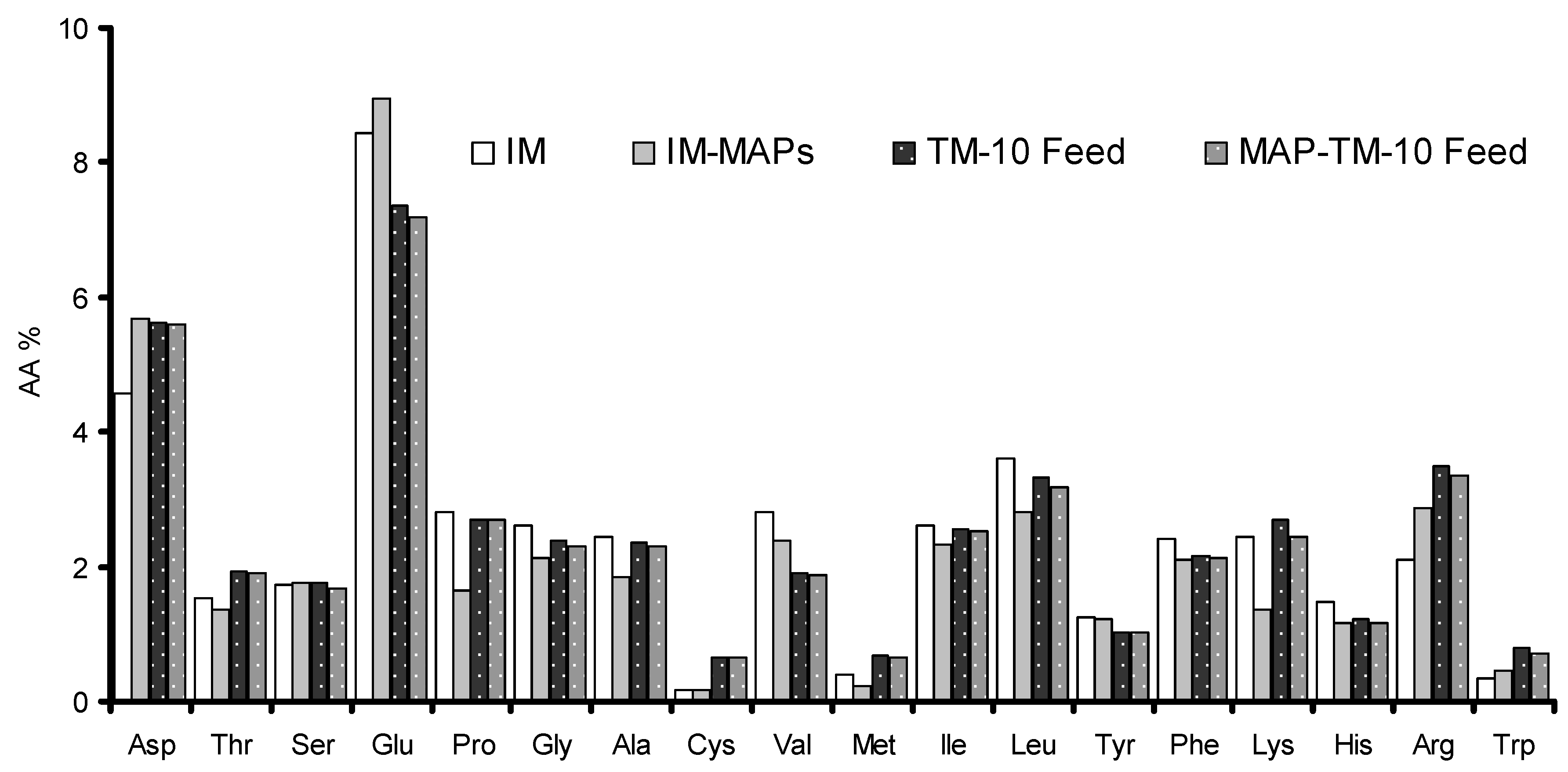
Figure 1.
Amino Acid (AA%, Dry Weight basis) concentrations in TM meal produced in conventional (IM, dotted white bars) or in enriched substrate (IM-MAPs, grey bars) and in Fish feeds of the TM-10 (dotted black bars) and the MAP-TM-10 group (dotted grey bars). Aspartic acid - Asp Threonine - Thr Serine - Ser Glutamic acid – Glu, Proline – Pro, Glycine – Gly, Alanine – Ala, Cysteine – Cys, Valine – Val, Methionine – Met, Isoleucine – Ile, Leucine – Leu, Tyrosine – Tyr, Phenylalanine – Phe, Lysine – Lys, Histidine – His, Arginine - Arg , Tryptophan – Trp.
Figure 1.
Amino Acid (AA%, Dry Weight basis) concentrations in TM meal produced in conventional (IM, dotted white bars) or in enriched substrate (IM-MAPs, grey bars) and in Fish feeds of the TM-10 (dotted black bars) and the MAP-TM-10 group (dotted grey bars). Aspartic acid - Asp Threonine - Thr Serine - Ser Glutamic acid – Glu, Proline – Pro, Glycine – Gly, Alanine – Ala, Cysteine – Cys, Valine – Val, Methionine – Met, Isoleucine – Ile, Leucine – Leu, Tyrosine – Tyr, Phenylalanine – Phe, Lysine – Lys, Histidine – His, Arginine - Arg , Tryptophan – Trp.
Table 1.
Composition of the experimental diets.
Table 1.
Composition of the experimental diets.
| Ingredients (g/kg) |
Control |
TM-10 |
MAP-TM-10 |
| Fish feed |
900 |
900 |
900 |
| Wheat (extruded) |
60 |
0 |
0 |
| Fish meal (71% CP) |
40 |
0 |
0 |
| “Conventional” insect meal (35% CP) |
0 |
100 |
0 |
| “Enriched” insect meal (35% CP) |
0 |
0 |
100 |
| |
|
|
|
| Calculated analysis (as fed) |
|
|
|
| Digestible energy (Mj/kg) |
15.79 |
15.80 |
15.80 |
| Total phenolic content (TPC) of Fish feeds (mg GAE/L extract phenol) |
12.42 |
44.13 |
56.08 |
| Crude protein (g/kg) |
439.9 |
440.0 |
440.0 |
| Nitrogen Free Extract (g/kg) |
163.8 |
126.0 |
126.0 |
2.3. Dietary experiments, rearing facilities, and experimental conditions
Growth trial (three independent replicates) was performed in an aquaculture facility in the northwest region of Greece (Skaloma S.A). A total of 4500 (207.19 ± 1.47g average initial body weight) gilthead sea bream (Sparus aurata) of both sexes were individually weighed and were randomly allocated in to 9 floating cages (500 individuals per cage). A total of 3 triplicate experimental groups were formed: The control group A, which was fed the Control diet; The experimental group B which was fed with TM-10; The experimental group C fed with MAP-TM-10. Each diet was administered twice a day (09:00 h and 16:00 h) at 1.5 feeding rate (FR), 7 days per week, for 4 consecutive weeks. All fish were considered clinically healthy based on an external examination for any signs of infestation, skeletal deformities, soft tissue deformities, ocular injuries, or wounds. Prior to the experiment, anti-cohabitation measures took place to ensure equal growth and feed intake potential for all experimental fish. Throughout the duration of the trial, all experimental groups were subjected to similar natural environmental conditions (photoperiod, water temperature, water depth and cohabitation potential). The exact amount of feed distributed to each sea cage (feed intake) was recorded daily. Feeds were administered over the whole water surface to be simultaneously accessible to all the fish. During the trial, all groups were inspected daily to check mortality.
2.4. Sampling procedures
Fish were randomly sampled before feeding from all groups, by confinement and netting in the tanks. Fish were fasted for 18 h before sampling to minimize handling stress. The fish were anaesthetized prior to blood sampling using prescribed buffered Benzocaine at a concentration of 400 ppm. Following the anaesthetization procedure, the fish were individually weighed to the nearest 0.1 g (Kern, PES 2200-2M), their fork length (L) was recorded and thereafter underwent venipuncture.
2.5. Manual haematological analysis
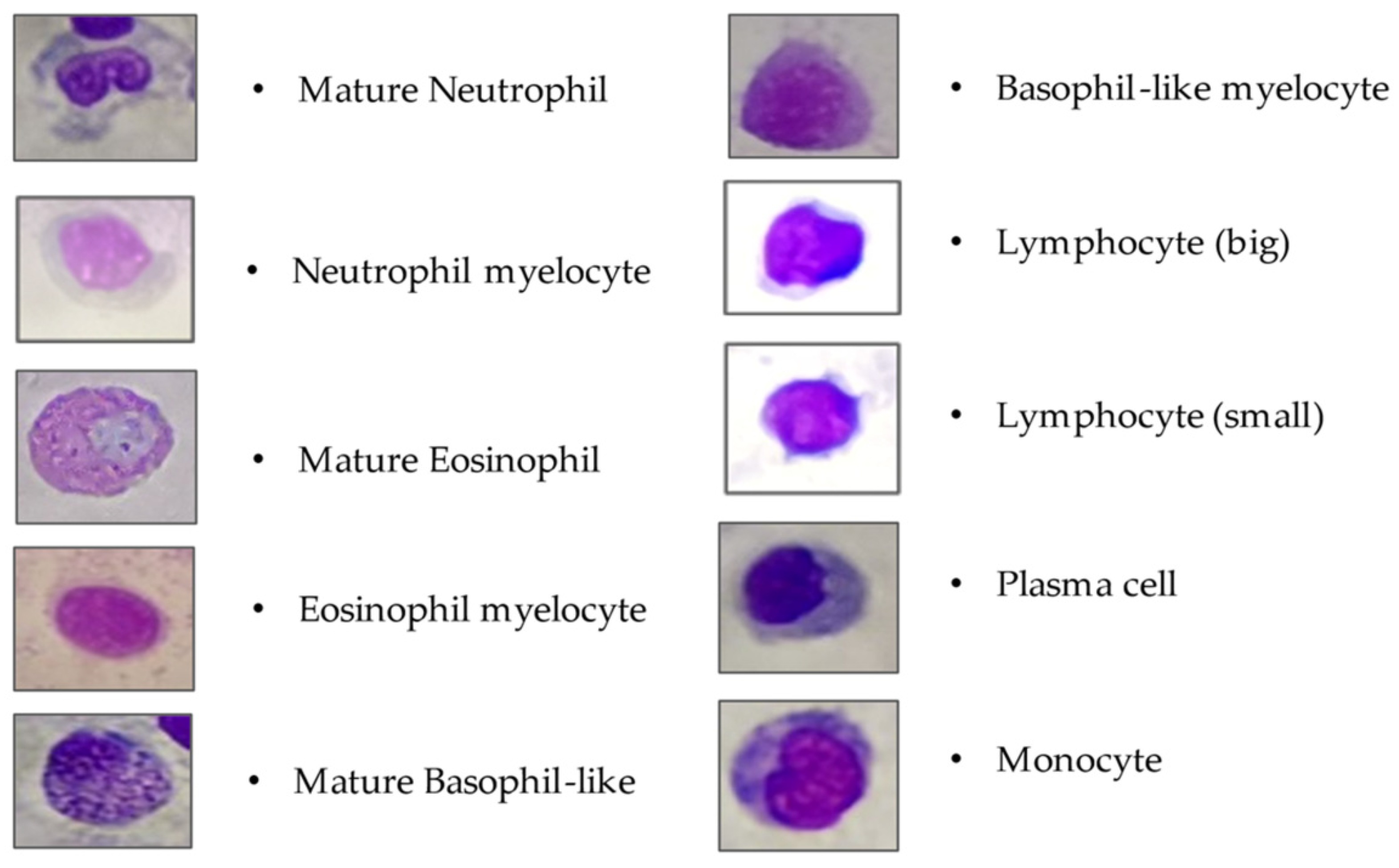
At the end of the 4-week experiment, 5 gilthead sea bream where randomly selected from each of the 9 floating cages (45 fish – 15 per group). Blood samples were obtained from the caudal vein of every fish using a 20 G×1½ syringe without containing any anticoagulant agent to assess the differential leukocyte count (DLC) [
58]. Blood smears were taken immediately given the rapid blood clotting of gilthead sea bream. A blood drop (without anticoagulants) was applied on a degreased glass slide and was smeared out, dried at room temperature, fixed with methanol, and stained (Hemacolor® Rapid Staining Set (Merck KGaG, Darmstadt, Germany). Each smear was divided into 6 randomly selected areas of 1mm
2, and the absolute percentage of each white blood cell was counted (lymphocytes, neutrophils, eosinophils, monocytes, and their blast forms as a percentage were calculated). Smears were studied using an Olympus CX23 light microscope. The cellular size, nuclear morphology, and cytoplasmic staining patterns were the criteria for the cells’ identification and determination [
59,
60,
61]. The DLC was recorded as a percentage of a particular cell type [
58]. Mature and blastic cells were involved in the leukogram (
Figure 2), but thrombocytes and thrombocyte-like cells were excluded in the white blood cell parameters, since they represent a distinct blood cell type [
62]. Conclusively, a total of 270 (45 fish-smears x 6 randomly selected divisions /smear) blood assays were conducted.
2.6. Determination of HSPs, MAPKs and apoptosis
The preparation of whole blood samples and the immunoblot analysis were conducted following well-established protocols. In this study, for SDS-PAGE, equivalent amounts of proteins from whole blood were separated using acrylamide and bisacrylamide gels with concentrations of either 10 and 0.275% (w/v). Subsequently, the proteins were transferred electrophoretically onto nitrocellulose membranes. The antibodies used included polyclonal rabbit anti-heat shock protein 70 kDa, polyclonal rabbit anti-heat shock protein 90 kDa, polyclonal rabbit anti-phospho-p38 MAPK (Thr180-Tyr182) (9211, Cell Signaling), monoclonal rabbit anti-phospho p44/42 MAPK (4376, Cell Signaling), polyclonal rabbit anti-bcl2 (7973, Abcam), polyclonal rabbit anti-bax (B-9) (7480, Santa Cruz Biotechnology) and actin (anti-β actin 3700, Cell Signaling). To ensure quality transfer and protein loading, Ponceau stain was utilized for western blot. Bands were detected using enhanced chemiluminescence, and quantification was performed using laser scanning densitometry with GelPro Analyzer Software (GraphPad).
2.7. Statistical analysis and formulae
Three independent replicates were used in all analyses with exception of differential blood count where six independent replicates were used. Data are presented as the mean ± standard error of means (SEM). Mean values were compared among treatments using One-Way analysis of variance (ANOVA). Post-hoc comparisons were analyzed using Tukey HSD, Bonferroni & Holms test and Scheffe multiple comparison test. The differences were determined significant at p < 0.05. All statistical analyses were carried out using SPSS version 21.0 (SPSS Inc., Chicago, IL, USA). All formulae used are presented below:
Weight gain (WG, %) = [(final body weight − initial body weight) / initial body weight] × 100.
Specific growth rate (SGR, %/day) = [(ln final body weight − ln initial body weight) / days of experiment] × 100.
Feed conversion ratio (FCR) = feed intake / wet weight gain.
Protein efficiency ratio (PER) = Weight gain / Protein intake
Survival (%) = (final number of fish / initial number of fish) × 100.
Differential Leukocyte Count (%) = % of a given white cell type =100% x Absolute number of a given white cell type (cells/μL) / Total WBC (cells/μL).
2.8. Experimental - ethics aspects
The insects used in this study originated from a stock colony maintained at the Entomology Lab of the Institute of Plant Breeding and Genetic Resources (IPGRB) of the Hellenic Agricultural Organization Demeter in Thermi, Greece. The insects were kept under standard conditions: temperature 26°C; 60% relative humidity; 8:16 h (L:D) photoperiod.
The experimental protocol for this trial was reviewed and approved by the Research Ethics Committee of the University of Ioannina (Greece) (protocol No 56652, 26/11/2021). The trial involving gilthead sea bream was conducted by accredited scientists at an aquaculture facility in the northwest region of Greece (Skaloma S.A). Accredited scientists were responsible for the supervision of animal care, procedures, samplings, anesthesia, and euthanasia of experimental fish. The experimental procedures followed the guidelines outlined in the current European Directive (2010/63/EU) for the protection of animals used in scientific research. The use of dried T. molitor as food for humans and animals is considered safe under EU Regulation 882/2021, and any by-products obtained from vertebrate experimental animals were handled in accordance with Regulation (EC) No 1069/2009.
3. Results
3.1. Growth performance and relevant growth indices
There was no significant difference in the mean weight gain among the different groups (
p > 0.05). Specific growth rate (SGR), feed conversion ratio (FCR), and survival rate also displayed no significant differences (
p > 0.05) among the treatments (
Table 2).
3.2. Differential Leukocyte Count (DLC)
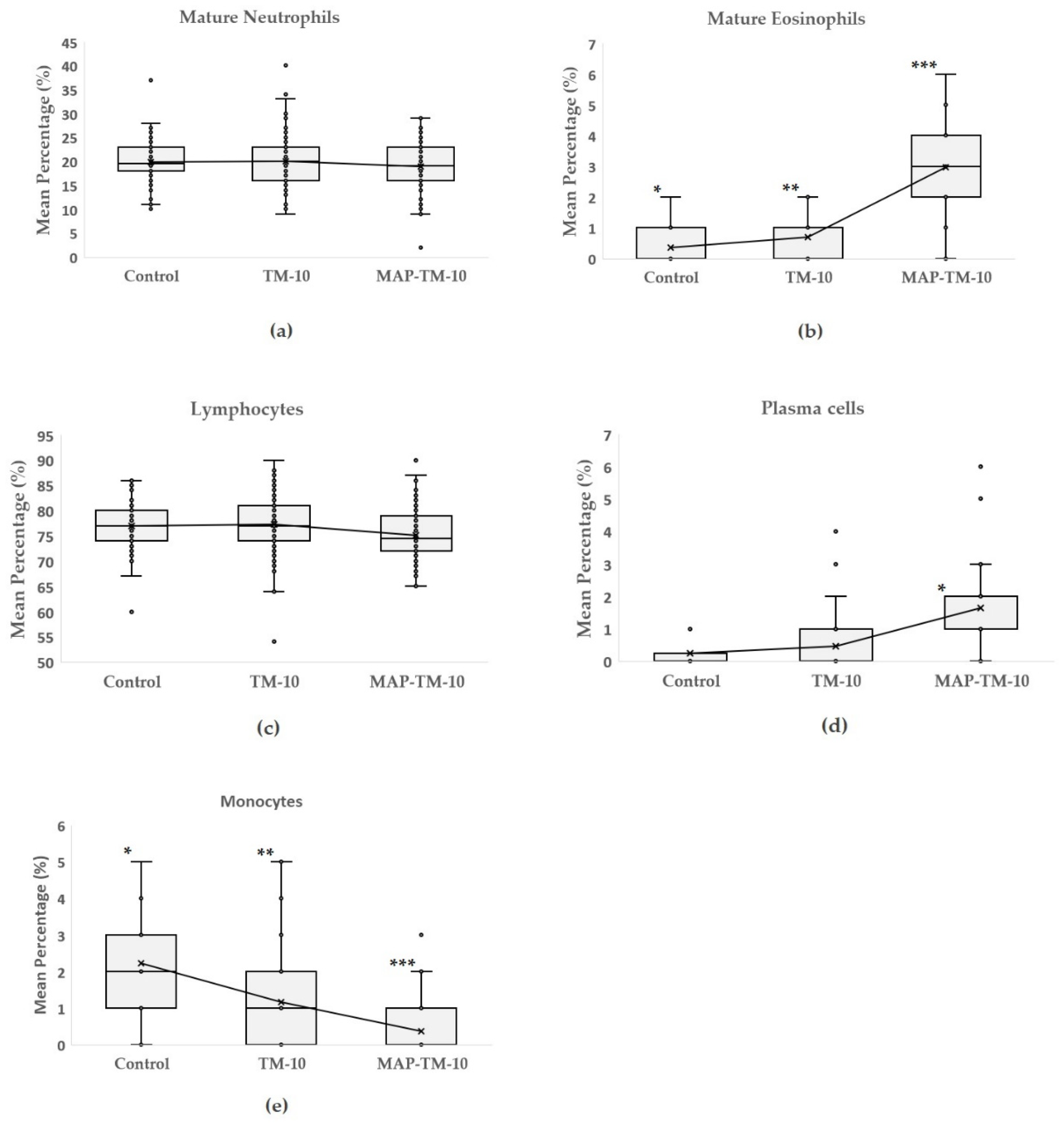
The results of
Figure 2 show the proportions of mature neutrophils, mature eosinophils, lymphocytes, plasma cells, and monocytes of the three groups. Among the three groups, no significant differences (
p > 0.05) were observed between the mean percentage values of mature neutrophils and lymphocytes. However, a significant statistical difference was observed between the groups in mature eosinophils, with the MAP-TM-10 group showing the highest eosinophilic induction (
p < 0.01), followed by the TM-10 (
p < 0.05). A similar pattern was observed in plasma cells, with the MAP-TM-10 group exhibiting the highest mean percentage values and a significant statistical difference from the TM-10 and Control group (
p < 0.01). Monocytes demonstrated significant statistical differences among the three groups (
p < 0.01). The lowest mean percentage value was observed in the MAP-TM-10 group, while the second highest was observed in the TM-10 group.
Figure 3.
Box-and-whisker plots of mean percentage of mature neutrophils (a), mature eosinophils (b), lymphocytes (c), plasma cells (d) and monocytes (e) of the three experimental groups. Significant differences among groups are denoted with asterisks (* p < 0.05; ** p < 0.01;*** p < 0.001).
Figure 3.
Box-and-whisker plots of mean percentage of mature neutrophils (a), mature eosinophils (b), lymphocytes (c), plasma cells (d) and monocytes (e) of the three experimental groups. Significant differences among groups are denoted with asterisks (* p < 0.05; ** p < 0.01;*** p < 0.001).
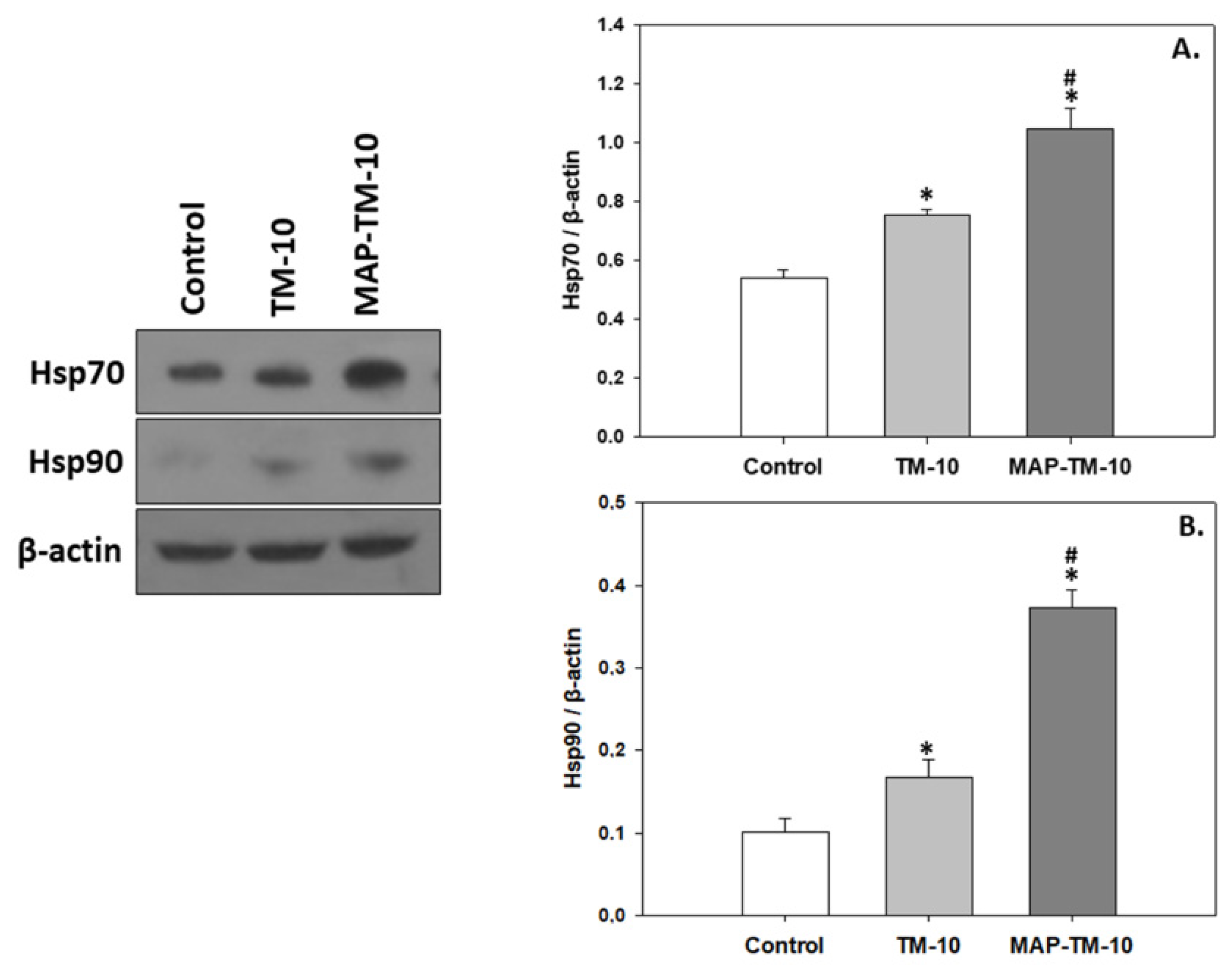
3.3. Heat Shock Induction
Both HSPs members examined in the present study exhibit an increasing pattern in both insect-based feeding regimes compared to Control group. Specifically, the TM-10 group depicted statistically significant increased levels (
p < 0.05) of both HSP70 and HSP90 compared to the Control. Moreover, the MAP-TM-10 group exhibited significantly (
p < 0.05) increased levels of HSP70 (
p < 0.05) and HSP90 (
p < 0.001) in the fish whole blood compared to the Control (
Figure 4A,B).
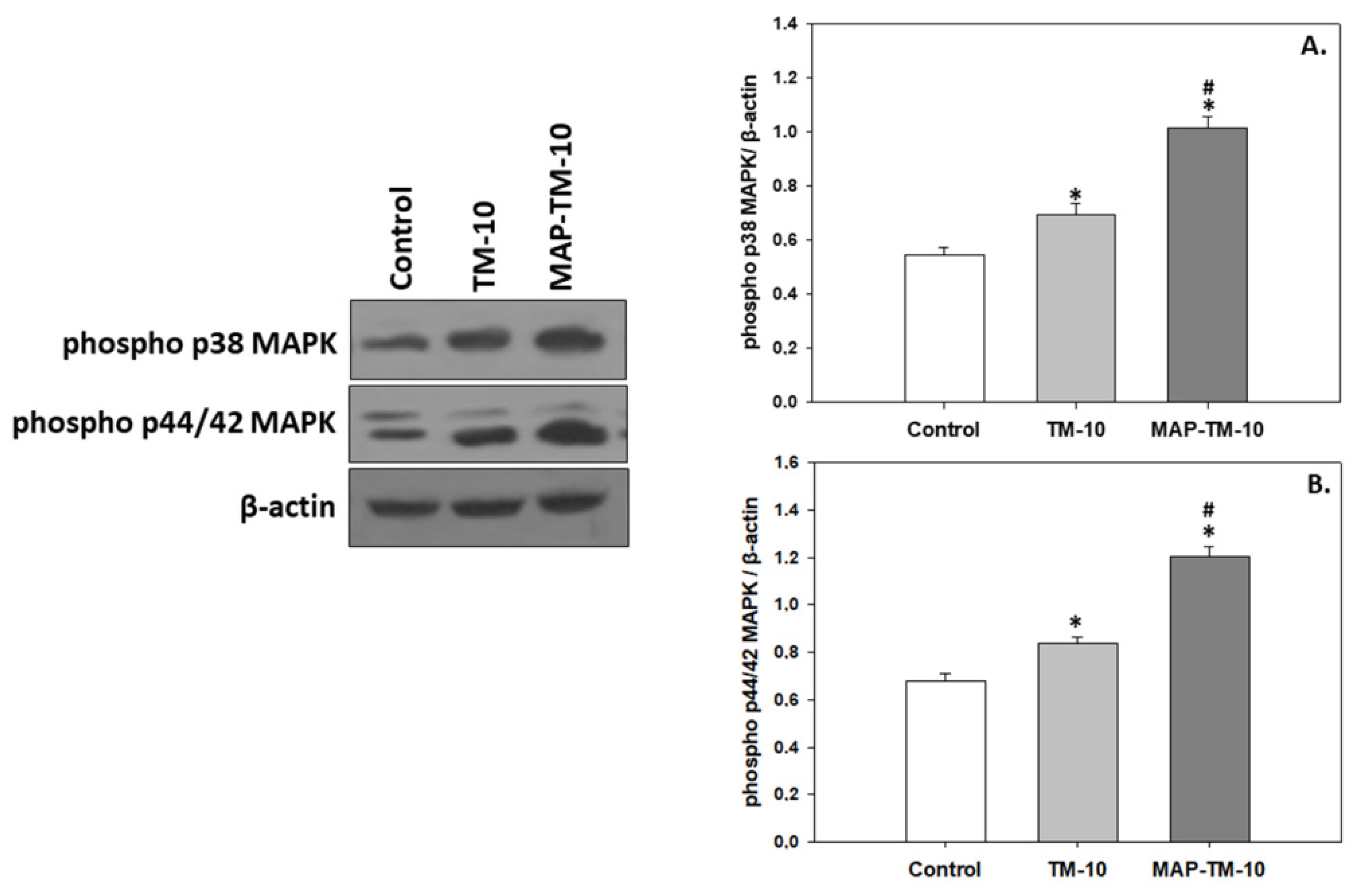
3.4. MAPK signaling
Compared to the HSP induction, the MAPK phosphorylation, exhibited a similar pattern. Specifically, the TM-10 feeding regime depicts significant increased (
p < 0.05) phosphorylated levels of p38 MAPK and p44/42 MAPK compared to the levels exhibited in the Control. Additionally, the MAP-TM-10 feeding regime depicted significantly increased (
p < 0.05) phosphorylated levels of both p38 MAPK and p44/42 MAPK in the fish whole blood compared to the Control (
Figure 5A,B).
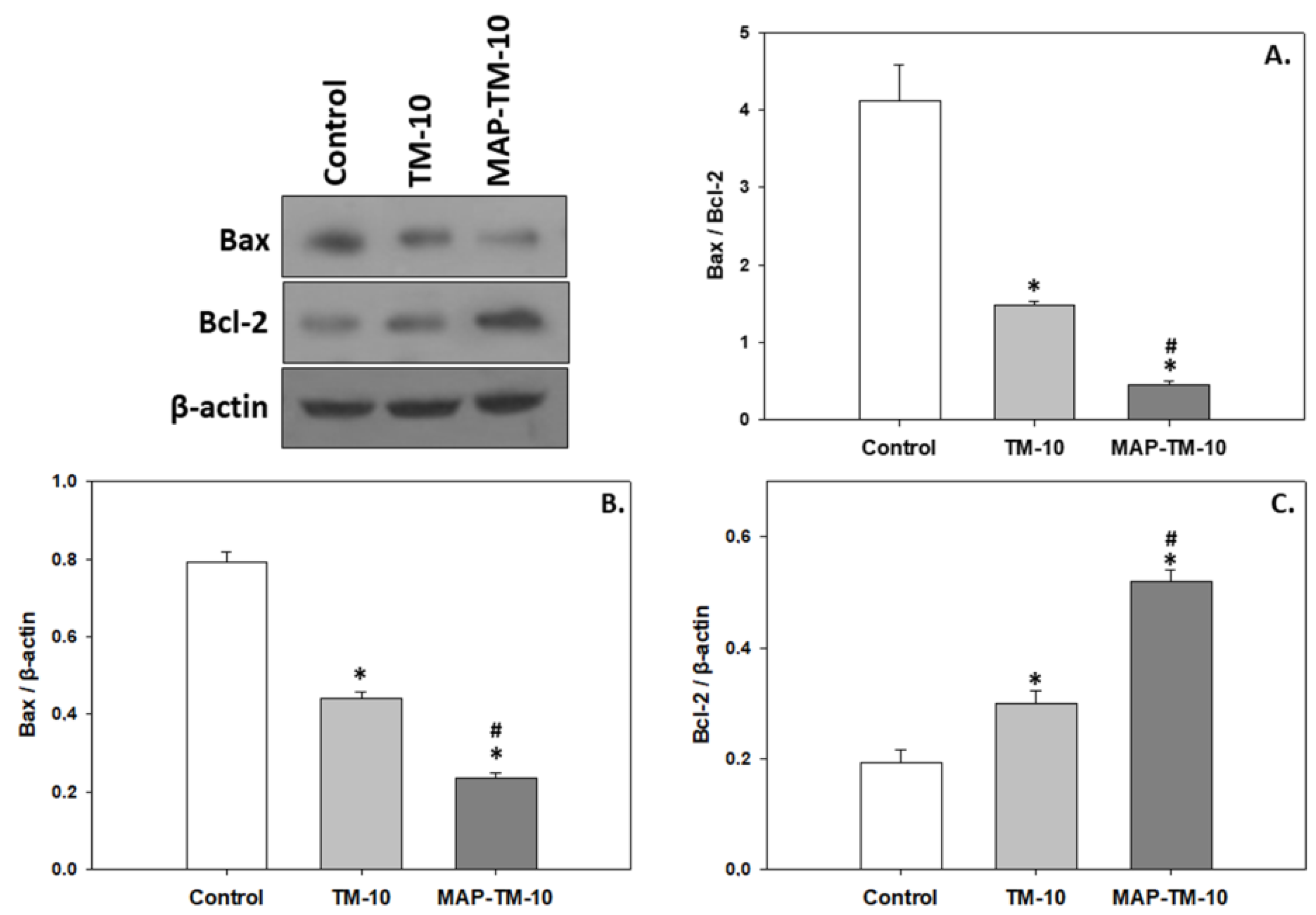
3.5. Apoptosis
Bax and Bcl-2 proteins, which promotes and prevents apoptosis respectively indicate dietary induce effect. In contrast to the pattern observed in both HSPs and MAPKs, Bax levels exhibited statistically significant decreased levels (
p < 0.05) in whole blood of fish fed the TM-10 regime compared to the Control. The MAP-TM-10 regime depicted even more decreased levels (
p < 0.05) of Bax compared to the TM-10 (
Figure 6B). However, Bcl-2 exhibited an opposite pattern with TM-10 resulting in increased levels compared to Control (
p < 0.05), and MAP-TM-10, even more increased levels (
p < 0.05) compared to Control and also the TM-10 feeding regime (
Figure 6C). As expected from the above, the Bax/Bcl-2 ratio, which is potent apoptotic indicator, depicted statistically significant decreased levels (
p < 0.05) in the whole blood of fish fed with both TM-10 and MAP-TM-10 regimes. Moreover, the MAP-TM-10 regime exhibited a statistically significant decrease (
p < 0.05) compared to the TM-10 feeding regime (
Figure 6A).
4. Discussion
Initially, publications regarding the use of TM larvae in gilthead seabream primarily focused on growth performance, feed convertibility, and fillet composition among other indices [
29]. However, recent studies began to investigate the molecular, biochemical and physio-pathological pathways triggered in fish by supplementation of fish meal with insect meal [
13,
26].
4.1. Gilthead sea bream growth performance
As stated in previous studies, in the context of growth performance, the replacement of 25% fishmeal (FM) with TM larvae meals has shown encouraging results in growth indicators, although a slight decrease in protein efficiency ratio (PER) and feed conversion ratio (FCR) was noticed compared to the FM-supplemented group [
28]. Higher replacement percentages (50% FM) resulted in some cases in improved gut microbial diversity [
11] and amino acid composition [
2,
12], while in some others in increased operational taxonomic units without corresponding to higher diversity in gut microbiota [
1]. Our results are in accordance with previous literature findings that have examined similar insect meal supplementation rates, in terms of growth performance indices of other farmed fish species [
3,
13,
16,
17,
18,
19,
20,
21,
22] and gilthead seabream. [
63]. More specifically, both TM-10 insect meal supplementation and MAP-TM-10 groups had no adverse effects on WG, SGR and FCR in comparison to the Control group. The results show that there were no significant differences in PER between the control group (A), the TM-10 group (B), and the MAP-TM-10 group (C). This means that all three groups were able to retain protein equally well which was reflected on the PER (Table 3). In fact, the addition of TM-10 or MAP-TM-10 to the diet did not have a significant effect on protein retention and the values were within the range of PER reported by Piccolo et al [
38] in their study on the effects of
T. molitor larvae meal on the growth performance of gilthead seabream.
4.2. White blood cells modultation
Blood analysis in fish has become an essential method for assessing stress and inflammatory biomarkers. Unlike the liver, where these biomarkers are often rapidly cleared, blood allows for easy detection and measurement of such biomarkers, providing valuable insights into the physiological responses of fish to different stressors and environmental challenges [
64]. Blood tests on fish have been conducted for several decades, both in laboratory and field settings, to evaluate endocrine, reproductive, and immune functions, as well as maturation, nutrition, and health status [
65]. The primary stress responses involve the release of catecholamines and corticosteroids [
65]. Subsequently, secondary stress responses occur and have various effects on multiple tissues, including the blood. These secondary responses can include accelerated energy mobilization through glucose, changes in hydromineral balance, increased lactate levels, as well as decreased blood pH, hematocrit, and leukocytes activation [
66,
67,
68].
One interesting finding was the significant increase in eosinophil values among the groups, with higher values observed in the MAP-TM-10 group. At first glance, the results may seem contradictory, as there are several reports on the immunoregulatory capacity of essential oils in humans [
69,
70,
71], though increased eosinophil values have been observed in Nile tilapia after administration of oregano essential oil (although without statistically significant differences compared to the controls) [
73]. On the other hand, someone could hypothesize that the observed eosinophilia is a reactive response to the insect meal substitute. Henry et al. [
27], reported no significant changes on gilthead seabream lymphocytes and eosinophils between the control and the 10% full-fat substitute group [i.e.
Zophobas morio (F.) larvae] after 100 days of administration. It is important to consider the heterogeneity of the seven different MAP residues used in our study, which in combination with TM, may have resulted in this eosinophilic induction.
Another important finding is the significant decrease in monocyte cells (TM-10 and MAP-TM-10 groups) compared to the control, with lowest percentage values observed in the MAP-TM-10 group. Similar monocyte pattern findings were reported by Henry et al. [
27] following insect meal inclusion. Moreover, it is worth noting that MAP residues have been associated with potential antibacterial properties due to the presence of phenolic compounds [
43,
74]. In the present study, the antibacterial activity of MAP residues may have contributed to the significant reduction of monocytes observed in the MAP-TM-10 group, amplifying the effect. Like other vertebrates [
75], the relative ratios of specific leukocyte populations in fish blood can provide valuable information about their response to specific treatments or environmental factors.
Changes in the number of mature neutrophils and leukocytes and the ratio of neutrophilic/heterophilic granulocytes, can indicate stress-induced WBC activation [
68,
76,
77,
78]. However, in the present work, there were no apparent differences among the three experimental groups in the number of mature neutrophils and leukocytes. This suggests that the dietary treatment did not induce any changes in WBC activation in the experimental fish suggesting a well-tolerated immune-homeostatic capacity.
4.3. Apoptosis, Heat Shock Proteins & MAPK signaling
Recent studies have investigated the potential impact of TM on the pathophysiology (physiological dysfunction) of gilthead seabream. For instance, Mente, et al. [
13] reported dietary inclusion of 25% TM larvae meal in farmed fish led the induction of TM-related oxidative stress, which activated cell death mechanisms in the liver.
In this context, Bousdras et al. [
26] exemplified that apoptosis was induced in seabream’s heart following the inclusion of 25% TM. However, tissue specificity was apparent in the abovementioned study considering that apoptosis was suppressed in the muscle [
26], which is in accordance with the present findings in the blood of both experimental diets. Numerous studies have highlighted that the impact of dietary insect meal highly depends on the levels of inclusion [
79,
80,
81]. The substitution of fishmeal with mealworm meal at 75% provoked hepatic apoptosis in largemouth bass (
Micropterus salmoides), compared to lower inclusion levels, which was attributed to the decreased feed utilization due to intestinal damage under that feeding regime [
81]. Considering that growth performance indices displayed no alteration herein among the three diets, it is likely that such a low level of substitution exerts no detrimental effects which may induce the apoptotic cell death. In addition, the enrichment of
T. molitor substrate with MAP seems to strengthen the anti-apoptotic effects. Plant compounds have been shown both
in vivo and
in vitro to up-regulate Bcl-2 and suppress pro-apoptotic cytochrome c release from mitochondria in rat [
82,
83]. Furthermore, the ameliorative effects of herbal extracts in terms of apoptosis have also been observed in breast tissue of broiler chicken following dietary supplementation [
84].
MAPs are known to have antioxidant properties, and they have been shown to enhance the total phenolic and flavonoid content as well as the antioxidant potential of
T. molitor [
41]. Likewise, in the present study, the MAP-TM-10 group had a significantly higher level of total phenolics than the control group, this may have contributed to the anti-apoptotic effects observed in the MAP-TM-10 group.
Contrary to the apoptosis suppression, both insect-based diets led to the induction of HSPs and the activation of MAPKs compared to the control. In agreement with the present findings, several fish species, including gilthead seabream, exhibited up-regulation of HSPs in response to dietary inclusion of insect meal [
26,
85,
86]. However, the induced HSPs expression in the abovementioned studies was observed under considerable higher levels of inclusion compared to the 10% fishmeal substitution of the present study. Therefore, it is likely that the apparent HSPs increase may lean more towards the recruitment of molecular chaperones for the protein metabolism rather than the involvement in nutrient stress response. Insect meals have been previously demonstrated to modify the amino acid catabolism in fish species such as rainbow trout, tench and gilthead seabream [
10,
87]. Specifically, the elevated free amino acids and peptides from insect-based diets may favor the protein synthesis [
86], which requires HSPs for the proper folding of the newly synthesized polypeptides [
73]. Furthermore, the higher HSPs levels in the gilthead seabream fed the MAP-enriched insect meal may also imply involvement in protein metabolism considering that previous studies have demonstrated that supplementation of plant organic compounds contribute to the increase of free amino acids [
88,
89]. Interestingly, Lee et al. [
90] indicated that garlic extract supplementation in sterlet sturgeon (
Acipenser ruthenus) was accompanied by elevated circulating insulin, which may enhance the blood transfer of free amino acid to muscle. In addition, the regulation of HSPs induction in fish has been proposed to partially involve the activation of MAPKs [
91], which may explain the identical expression pattern shared among the two protein families herein. The tendency for a similar modification pattern of HSPs and MAPKs was previously reported by Stenberg et al. [
92] in Atlantic salmon following dietary substitution of fish meal with black soldier fly larvae meal. However, further research is required especially for the insect meal-induced alterations in the activation of MAPKs. Seo et al. [
93] suggested that ethanol extracts of
T. molitor larvae suppresses adipogenic differentiation via p44/42 MAPK inactivation and p38 MAPK activation in mice.
5. Conclusions
The results of this study suggest that the addition of TM-10 or MAP-TM-10 insect meal to the diet of gilthead sea bream does not have a significant effect on SGR, PER or FCR. However, there were some differences observed in the white blood cell profiles of the fish in the different groups. This suggests that the MAP-TM-10 diet may have a different effect on the immune system of the fish than the other two diets. Plant extracts have been found to promote various activities in fish and shrimp aquaculture, including anti-stress effects, growth promotion, appetite stimulation, enhancement of tonicity, immune stimulation, maturation of culture species, and antibacterial properties. MAPs (medicinal-aromatic plants) were not directly added to the diets in this experiment, but rather served as a substrate for
T. molitor larvae. As a result, the only way MAPs might have impacted the fish was through their consumption of insect meal produced in MAP-enriched substrate. Given the tissue-specific nature of fish biochemical processes [
94,
95,
96], as well as the growing need for sustainable aquaculture practices [
97], more research is needed on the molecular and metabolic pathways in insects reared in MAPs-enriched substrate. Additionally, future research should also look into whether MAP-enriched insect meal has an effect on the fatty acid profiles of the fish flesh. This would help to elucidate the mechanism implicated in the modulation of physiological parameters and the potential effect on fish flesh quality of fish fed with MAP-TM-10 feed.
Author Contributions
Conceptualization, E.A., I.G.,C.Z., E.B., S.A. and C.N.; software, E.A., N.P., K.F., E.B., C.Z. and I.G.; validation, E.A., N.P., M.K., K.F, E.G.; formal analysis, E.A., M.K., N.P., P.K., K.F., K. B., C.Z.; investigation, E.A., M.K., N.P., E.G., P.K., K.F., A.C., K.B., C.Z., E.B., G.S., and C.N.; resources, E.A., C.N., E.B., I.S., I.G , A.C., and K.B.; data curation, E.A., M.K., N.P., E.G., and E.B.; writing—original draft preparation, E.A., M.K., N.P., K.F., E.G., E.B., and C.N.; writing—review and editing, E.A., M.K., N.P., E.G., K.F., E.B., I.G., S.A., G.S., C.A. and C.N; supervision, E.A., E.B., I.G. and C.N.; project administration, E.B.; funding acquisition, E.A., E.B. I.S. C.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research has been co-financed by Greece and the European Union (European Regional Development Fund) in context “Research–Create–Innovate” within the Operational Program (Competitiveness, Entrepreneurship and Innovation (ΕΠAΝΕΚ) of the NSRF 2014–2020, Project Code: Τ2ΕΔΚ-02356. Acronym: InsectFeedAroma.
Institutional Review Board Statement
The experimental protocol for this trial was reviewed and approved by the Ethics and Research Ethics Committee of the University of Ioannina of Greece (protocol number 56652, 26/11/2021).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
Acknowledgments
We would like to thank Dr. K. Grigoriadou of the Institute of Plant Breeding and Genetic Resources, Hellenic Agricultural Organization ELGO-DIMITRA, for her invaluable contribution and inspiration in the field of Mediterranean medicinal aromatic plant extracts in animal feeds.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Fabrikov, D.; Vargas-García, M.D.C.; Barroso, F.G.; Sánchez-Muros, M.J.; Cacua Ortíz, S.M.; Morales, A.E.; Cardenete, G.; Tomás-Almenar, C.; Melenchón, F. Effect on Intermediary Metabolism and Digestive Parameters of the High Substitution of Fishmeal with Insect Meal in Sparus aurata Feed. Insects 2021, 12, 965. [Google Scholar] [CrossRef]
- Shafique, L.; Abdel-Latif, H.M.R.; Hassan, F.; Alagawany, M.; Naiel, M.A.E.; Dawood, M.A.O.; Yilmaz, S.; Liu, Q. The Feasibility of Using Yellow Mealworms (Tenebrio molitor): Towards a Sustainable Aquafeed Industry. Animals 2021, 11, 811. [Google Scholar] [CrossRef]
- Gasco, L.; Henry, M.; Piccolo, G.; Marono, S.; Gai, F.; Renna, M.; Lussiana, C.; Antonopoulou, E.; Mola, P.; Chatzifotis, S. Tenebrio molitor Meal in Diets for European Sea Bass ( Dicentrarchus labrax L.) Juveniles: Growth Performance, Whole Body Composition and in Vivo Apparent Digestibility. Animal Feed Science and Technology 2016, 220, 34–45. [Google Scholar] [CrossRef]
- Ghosh, S.; Lee, S.-M.; Jung, C.; Meyer-Rochow, V.B. Nutritional Composition of Five Commercial Edible Insects in South Korea. Journal of Asia-Pacific Entomology 2017, 20, 686–694. [Google Scholar] [CrossRef]
- Gkinali, A.-A.; Matsakidou, A.; Vasileiou, E.; Paraskevopoulou, A. Potentiality of Tenebrio molitor Larva-Based Ingredients for the Food Industry: A Review. Trends in Food Science & Technology 2022, 119, 495–507. [Google Scholar] [CrossRef]
- Gasco, L.; Biasato, I.; Dabbou, S.; Schiavone, A.; Gai, F. Animals Fed Insect-Based Diets: State-of-the-Art on Digestibility, Performance and Product Quality. Animals 2019, 9, 170. [Google Scholar] [CrossRef]
- Tippayadara, N.; Dawood, M.A.O.; Krutmuang, P.; Hoseinifar, S.H.; Doan, H.V.; Paolucci, M. Replacement of Fish Meal by Black Soldier Fly (HermetiaiIllucens) Larvae Meal: Effects on Growth, Haematology, and Skin Mucus Immunity of Nile Tilapia, Oreochromis Niloticus. Animals 2021, 11, 193. [Google Scholar] [CrossRef]
- Sánchez-Muros, M.; De Haro, C.; Sanz, A.; Trenzado, C.E.; Villareces, S.; Barroso, F.G. Nutritional Evaluation of Tenebrio molitor Meal as Fishmeal Substitute for Tilapia ( Oreochromis niloticus ) Diet. Aquacult Nutr 2016, 22, 943–955. [Google Scholar] [CrossRef]
- Tubin, J.S.B.; Paiano, D.; Hashimoto, G.S.D.O.; Furtado, W.E.; Martins, M.L.; Durigon, E.; Emerenciano, M.G.C. Tenebrio Molitor Meal in Diets for Nile Tilapia Juveniles Reared in Biofloc System. Aquaculture 2020, 519, 734763. [Google Scholar] [CrossRef]
- Fabrikov, D.; Sánchez-Muros, M.J.; Barroso, F.G.; Tomás-Almenar, C.; Melenchón, F.; Hidalgo, M.C.; Morales, A.E.; Rodriguez-Rodriguez, M.; Montes-Lopez, J. Comparative Study of Growth Performance and Amino Acid Catabolism in Oncorhynchus mykiss, Tinca tinca and Sparus aurata and the Catabolic Changes in Response to Insect Meal Inclusion in the Diet. Aquaculture 2020, 529, 735731. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Nikouli, E.; Piccolo, G.; Gasco, L.; Gai, F.; Chatzifotis, S.; Mente, E.; Kormas, K.A. Reshaping Gut Bacterial Communities after Dietary Tenebrio molitor Larvae Meal Supplementation in Three Fish Species. Aquaculture 2019, 503, 628–635. [Google Scholar] [CrossRef]
- Iaconisi, V.; Secci, G.; Sabatino, G.; Piccolo, G.; Gasco, L.; Papini, A.M.; Parisi, G. Effect of Mealworm (Tenebrio Molitor L.) Larvae Meal on Amino Acid Composition of Gilthead Sea Bream (Sparus aurata L.) and Rainbow Trout (Oncorhynchus mykiss W.) Fillets. Aquaculture 2019, 513, 734403. [Google Scholar] [CrossRef]
- Mente, E.; Bousdras, T.; Feidantsis, K.; Panteli, N.; Mastoraki, M.; Kormas, K.A.; Chatzifotis, S.; Piccolo, G.; Gasco, L.; Gai, F.; et al. Tenebrio molitor Larvae Meal Inclusion Affects Hepatic Proteome and Apoptosis and/or Autophagy of Three Farmed Fish Species. Sci Rep 2022, 12, 121. [Google Scholar] [CrossRef] [PubMed]
- Terova, G.; Gini, E.; Gasco, L.; Moroni, F.; Antonini, M.; Rimoldi, S. Effects of Full Replacement of Dietary Fishmeal with Insect Meal from Tenebrio molitor on Rainbow Trout Gut and Skin Microbiota. J Anim Sci Biotechnol 2021, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Rema, P.; Saravanan, S.; Armenjon, B.; Motte, C.; Dias, J. Graded Incorporation of Defatted Yellow Mealworm (Tenebrio molitor) in Rainbow Trout (Oncorhynchus mykiss) Diet Improves Growth Performance and Nutrient Retention. Animals 2019, 9, 187. [Google Scholar] [CrossRef]
- Jeong, S.-M.; Khosravi, S.; Mauliasari, I.R.; Lee, S.-M. Dietary Inclusion of Mealworm (Tenebrio molitor) Meal as an Alternative Protein Source in Practical Diets for Rainbow Trout (Oncorhynchus mykiss) Fry. Fish Aquatic Sci 2020, 23, 12. [Google Scholar] [CrossRef]
- Melenchón, F.; Larrán, A.M.; De Mercado, E.; Hidalgo, M.C.; Cardenete, G.; Barroso, F.G.; Fabrikov, D.; Lourenço, H.M.; Pessoa, M.F.; Tomás-Almenar, C. Potential Use of Black Soldier Fly ( Hermetia illucens ) and Mealworm (Tenebrio molitor) Insectmeals in Diets for Rainbow Trout ( Oncorhynchus mykiss ). Aquacult. Nutr. 2021, 27, 491–505. [Google Scholar] [CrossRef]
- Chemello, G.; Renna, M.; Caimi, C.; Guerreiro, I.; Oliva-Teles, A.; Enes, P.; Biasato, I.; Schiavone, A.; Gai, F.; Gasco, L. Partially Defatted Tenebrio molitor Larva Meal in Diets for Grow-Out Rainbow Trout, Oncorhynchus mykiss (Walbaum): Effects on Growth Performance, Diet Digestibility and Metabolic Responses. Animals 2020, 10, 229. [Google Scholar] [CrossRef]
- Belforti, M.; Gai, F.; Lussiana, C.; Renna, M.; Malfatto, V.; Rotolo, L.; De Marco, M.; Dabbou, S.; Schiavone, A.; Zoccarato, I.; et al. Tenebrio molitor Meal in Rainbow Trout ( Oncorhynchus mykiss ) Diets: Effects on Animal Performance, Nutrient Digestibility and Chemical Composition of Fillets. Italian Journal of Animal Science 2015, 14, 4170. [Google Scholar] [CrossRef]
- Su, J.; Gong, Y.; Cao, S.; Lu, F.; Han, D.; Liu, H.; Jin, J.; Yang, Y.; Zhu, X.; Xie, S. Effects of Dietary Tenebrio Molitor Meal on the Growth Performance, Immune Response and Disease Resistance of Yellow Catfish (Pelteobagrus fulvidraco). Fish Shellfish Immunol 2017, 69, 59–66. [Google Scholar] [CrossRef]
- Roncarati, A.; Gasco, L.; Parisi, G.; Terova, G. Growth Performance of Common Catfish ( Ameiurus Melas Raf.) Fingerlings Fed Mealworm ( Tenebrio molitor ) Diet. Journal of Insects as Food and Feed 2015, 1, 233–240. [Google Scholar] [CrossRef]
- Tran, H.Q.; Kiljunen, M.; Van Doan, H.; Stejskal, V. European Perch (Perca fluviatilis) Fed Dietary Insect Meal (Tenebrio Molitor): From a Stable Isotope Perspective. Aquaculture 2021, 545, 737265. [Google Scholar] [CrossRef]
- Tran, H.Q.; Van Doan, H.; Stejskal, V. Does Dietary Tenebrio molitor Affect Swimming Capacity, Energy Use, and Physiological Responses of European Perch Perca fluviatilis? Aquaculture 2021, 539, 736610. [Google Scholar] [CrossRef]
- Tran, H.Q.; Prokešová, M.; Zare, M.; Matoušek, J.; Ferrocino, I.; Gasco, L.; Stejskal, V. Production Performance, Nutrient Digestibility, Serum Biochemistry, Fillet Composition, Intestinal Microbiota and Environmental Impacts of European Perch (Perca fluviatilis) Fed Defatted Mealworm (Tenebrio molitor). Aquaculture 2022, 547, 737499. [Google Scholar] [CrossRef]
- Ng, W.-K.; Liew, F.-L.; Ang, L.-P.; Wong, K.-W. Potential of Mealworm ( Tenebrio Molitor ) as an Alternative Protein Source in Practical Diets for African Catfish, Clarias Gariepinus: Mealworm as a Protein Source for Catfish. Aquaculture Research 2001, 32, 273–280. [Google Scholar] [CrossRef]
- Bousdras, T.; Feidantsis, K.; Panteli, N.; Chatzifotis, S.; Piccolo, G.; Gasco, L.; Gai, F.; Antonopoulou, E. Dietary Tenebrio molitor Larvae Meal Inclusion Exerts Tissue-Specific Effects on Cellular, Metabolic, and Antioxidant Status in European Sea Bass (Dicentrarchus labrax) and Gilthead Seabream (Sparus aurata). Aquaculture Nutrition 2022, 2022, 1–20. [Google Scholar] [CrossRef]
- Henry, M.A.; Golomazou, E.; Asimaki, A.; Psofakis, P.; Fountoulaki, E.; Mente, E.; Rumbos, C.I.; Athanassiou, C.G.; Karapanagiotidis, I.T. Partial Dietary Fishmeal Replacement with Full-Fat or Defatted Superworm (Zophobas morio) Larvae Meals Modulates the Innate Immune System of Gilthead Seabream, Sparus aurata. Aquaculture Reports 2022, 27, 101347. [Google Scholar] [CrossRef]
- Piccolo, G.; Iaconisi, V.; Marono, S.; Gasco, L.; Loponte, R.; Nizza, S.; Bovera, F.; Parisi, G. Effect of Tenebrio molitor Larvae Meal on Growth Performance, in Vivo Nutrients Digestibility, Somatic and Marketable Indexes of Gilthead Sea Bream (Sparus aurata). Animal Feed Science and Technology 2017, 226, 12–20. [Google Scholar] [CrossRef]
- Mastoraki, M.; Katsika, L.; Enes, P.; Guerreiro, I.; Kotzamanis, Y.P.; Gasco, L.; Chatzifotis, S.; Antonopoulou, E. Insect Meals in Feeds for Juvenile Gilthead Seabream (Sparus aurata): Effects on Growth, Blood Chemistry, Hepatic Metabolic Enzymes, Body Composition and Nutrient Utilization. Aquaculture 2022, 561, 738674. [Google Scholar] [CrossRef]
- Iaconisi, V.; Marono, S.; Parisi, G.; Gasco, L.; Genovese, L.; Maricchiolo, G.; Bovera, F.; Piccolo, G. Dietary Inclusion of Tenebrio molitor Larvae Meal: Effects on Growth Performance and Final Quality Treats of Blackspot Sea Bream ( Pagellus bogaraveo ). Aquaculture 2017, 476, 49–58. [Google Scholar] [CrossRef]
- Jeong, S.-M.; Khosravi, S.; Yoon, K.-Y.; Kim, K.-W.; Lee, B.-J.; Hur, S.-W.; Lee, S.-M. Mealworm, Tenebrio molitor, as a Feed Ingredient for Juvenile Olive Flounder, Paralichthys olivaceus. Aquaculture Reports 2021, 20, 100747. [Google Scholar] [CrossRef]
- Ido, A.; Hashizume, A.; Ohta, T.; Takahashi, T.; Miura, C.; Miura, T. Replacement of Fish Meal by Defatted Yellow Mealworm (Tenebrio molitor) Larvae in Diet Improves Growth Performance and Disease Resistance in Red Seabream (Pargus major). Animals 2019, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Motte, C.; Rios, A.; Lefebvre, T.; Do, H.; Henry, M.; Jintasataporn, O. Replacing Fish Meal with Defatted Insect Meal (Yellow Mealworm Tenebrio molitor) Improves the Growth and Immunity of Pacific White Shrimp (Litopenaeus vannamei). Animals 2019, 9, 258. [Google Scholar] [CrossRef]
- Song, S.-G.; Chi, S.-Y.; Tan, B.-P.; Liang, G.-L.; Lu, B.-Q.; Dong, X.-H.; Yang, Q.-H.; Liu, H.-Y.; Zhang, S. Effects of Fishmeal Replacement by Tenebrio molitor Meal on Growth Performance, Antioxidant Enzyme Activities and Disease Resistance of the Juvenile Pearl Gentian Grouper ( Epinephelus lanceolatus ♂ × Epinephelus fuscoguttatus ♀ ). Aquac Res 2018, 49, 2210–2217. [Google Scholar] [CrossRef]
- Khosravi, S.; Kim, E.; Lee, Y.-S.; Lee, S.-M. Dietary Inclusion of Mealworm (Tenebrio Molitor) Meal as an Alternative Protein Source in Practical Diets for Juvenile Rockfish (Sebastes schlegeli). Entomological Research 2018, 48, 214–221. [Google Scholar] [CrossRef]
- So, O.; O, A.; Ka, J. Medicinal Plants and Sustainable Human Health: A Review. HIJ 2018, 2. [Google Scholar] [CrossRef]
- Kant, R.; Kumar, A. Review on Essential Oil Extraction from Aromatic and Medicinal Plants: Techniques, Performance and Economic Analysis. Sustainable Chemistry and Pharmacy 2022, 30, 100829. [Google Scholar] [CrossRef]
- Aqeel, U.; Aftab, T.; Khan, M.M.A.; Naeem, M. Regulation of Essential Oil in Aromatic Plants under Changing Environment. Journal of Applied Research on Medicinal and Aromatic Plants 2022, 100441. [Google Scholar] [CrossRef]
- Taghouti, I.; Cristobal, R.; Brenko, A.; Stara, K.; Markos, N.; Chapelet, B.; Hamrouni, L.; Buršić, D.; Bonet, J.-A. The Market Evolution of Medicinal and Aromatic Plants: A Global Supply Chain Analysis and an Application of the Delphi Method in the Mediterranean Area. Forests 2022, 13, 808. [Google Scholar] [CrossRef]
- Zuazo, V.H.D.; García-Tejero, I.F.; Gálvez Ruiz, B.; Cermeño Sacristán, P.; Cuadros Tavira, S. Response of Essential-Oil Yield of Aromatic and Medicinal Plants to Different Harvesting Strategies. Com. Sci. 2019, 10, 429–437. [Google Scholar] [CrossRef]
- Andreadis, S.S.; Panteli, N.; Mastoraki, M.; Rizou, E.; Stefanou, V.; Tzentilasvili, S.; Sarrou, E.; Chatzifotis, S.; Krigas, N.; Antonopoulou, E. Towards Functional Insect Feeds: Agri-Food By-Products Enriched with Post-Distillation Residues of Medicinal Aromatic Plants in Tenebrio molitor (Coleoptera: Tenebrionidae) Breeding. Antioxidants 2021, 11, 68. [Google Scholar] [CrossRef]
- de Elguea-Culebras, G.O.; Bravo, E.M.; Sánchez-Vioque, R. Potential Sources and Methodologies for the Recovery of Phenolic Compounds from Distillation Residues of Mediterranean Aromatic Plants. An Approach to the Valuation of by-Products of the Essential Oil Market–A Review. Industrial Crops and Products 2022, 175, 114261. [Google Scholar] [CrossRef]
- Mandalakis, M.; Anastasiou, T.I.; Martou, N.; Keisaris, S.; Greveniotis, V.; Katharios, P.; Lazari, D.; Krigas, N.; Antonopoulou, E. Antibacterial Effects of Essential Oils of Seven Medicinal-Aromatic Plants Against the Fish Pathogen Aeromonas Veronii Bv. Sobria: To Blend or Not to Blend? Molecules 2021, 26, 2731. [Google Scholar] [CrossRef]
- Anastasiou, T.I.; Mandalakis, M.; Krigas, N.; Vézignol, T.; Lazari, D.; Katharios, P.; Dailianis, T.; Antonopoulou, E. Comparative Evaluation of Essential Oils from Medicinal-Aromatic Plants of Greece: Chemical Composition, Antioxidant Capacity and Antimicrobial Activity against Bacterial Fish Pathogens. Molecules 2019, 25, 148. [Google Scholar] [CrossRef]
- Miguel, M.G. Antioxidant Activity of Medicinal and Aromatic Plants. A Review. Flavour Fragr. J. 2010, 25, 291–312. [Google Scholar] [CrossRef]
- Oliva-Teles, A. Nutrition and Health of Aquaculture Fish. Journal of fish diseases 2012, 35, 83–108. [Google Scholar] [CrossRef] [PubMed]
- Feidantsis, K.; Soumalevris, A.; Panteli, N.; Chatzifotis, S.; Antonopoulou, E. Synergistic Effect of Long-Term Feed Deprivation and Temperature on the Cellular Physiology of Meagre (Argyrosomus regius). Journal of Thermal Biology 2022, 105, 103207. [Google Scholar] [CrossRef]
- Craig, E.A. Chaperones: Helpers along the Pathways to Protein Folding. Science 1993, 260, 1902–1903. [Google Scholar] [CrossRef]
- Jäättelä, M. Heat Shock Proteins as Cellular Lifeguards. Ann Med 1999, 31, 261–271. [Google Scholar] [CrossRef]
- Iwama, G.K.; Vijayan, M.M.; Forsyth, R.B.; Ackerman, P.A. Heat Shock Proteins and Physiological Stress in Fish. American Zoologist 1999, 39, 901–909. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Chouri, E.; Feidantsis, K.; Lazou, A.; Chatzifotis, S. Effects of Partial Dietary Supplementation of Fish Meal with Soymeal on the Stress and Apoptosis Response in the Digestive System of Common Dentex (Dentex dentex). Journal of Biological Research-Thessaloniki 2017, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wada, T.; Penninger, J.M. Mitogen-Activated Protein Kinases in Apoptosis Regulation. Oncogene 2004, 23, 2838–2849. [Google Scholar] [CrossRef] [PubMed]
- Panteli, N.; Demertzioglou, M.; Feidantsis, K.; Karapanagiotis, S.; Tsele, N.; Tsakoniti, K.; Gkagkavouzis, K.; Mylonas, C.C.; Kormas, K.A.; Mente, E.; et al. Advances in Understanding the Mitogenic, Metabolic, and Cell Death Signaling in Teleost Development: The Case of Greater Amberjack (Seriola dumerili, Risso 1810). Fish Physiol Biochem 2022, 48, 1665–1684. [Google Scholar] [CrossRef]
- Watson, A.J.; Pritchard, D.M. Lessons from Genetically Engineered Animal Models. VII. Apoptosis in Intestinal Epithelium: Lessons from Transgenic and Knockout Mice. Am J Physiol Gastrointest Liver Physiol 2000, 278, G1-5. [Google Scholar] [CrossRef]
- Rumbos, C.I.; Adamaki-Sotiraki, C.; Gourgouta, M.; Karapanagiotidis, I.T.; Asimaki, A.; Mente, E.; Athanassiou, C.G. Strain Matters: Strain Effect on the Larval Growth and Performance of the Yellow Mealworm, Tenebrio molitor L. Journal of Insects as Food and Feed 2021, 7, 1195–1205. [Google Scholar] [CrossRef]
- Horowitz, W.; Latimer, G.W. Official Methods of Analysis of AOAC International. Gaithersburg, Md. AOAC International 2006, 18. [Google Scholar]
- Vasilopoulos, S.; Giannenas, I.; Savvidou, S.; Bonos, E.; Rumbos, C.I.; Papadopoulos, E.; Fortomaris, P.; Athanassiou, C.G. Growth Performance, Welfare Traits and Meat Characteristics of Broilers Fed Diets Partly Replaced with Whole Tenebrio molitor Larvae. Animal Nutrition 2023, 13, 90–100. [Google Scholar] [CrossRef]
- Witeska, M.; Kondera, E.; Ługowska, K.; Bojarski, B. Hematological Methods in Fish – Not Only for Beginners. Aquaculture 2022, 547, 737498. [Google Scholar] [CrossRef]
- López-Ruiz, A.; Esteban, M.A.; Meseguer, J. Blood Cells of the Gilthead Seabream (Sparus aurata L.): Light and Electron Microscopic Studies: Blood Cells of Gilthead Seabream. Rec. 1992, 234, 161–171. [Google Scholar] [CrossRef]
- Clauss, T.M.; Dove, A.D.M.; Arnold, J.E. Hematologic Disorders of Fish. Veterinary Clinics of North America: Exotic Animal Practice 2008, 11, 445–462. [Google Scholar] [CrossRef]
- Ainsworth, A.J. Fish Granulocytes: Morphology, Distribution, and Function. Annual Review of Fish Diseases 1992, 2, 123–148. [Google Scholar] [CrossRef]
- Khandekar, G.; Kim, S.; Jagadeeswaran, P. Zebrafish Thrombocytes: Functions and Origins. Advances in Hematology 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Mastoraki, M., Katsika, L., Enes, P., Guerreiro, I., Kotzamanis, Y. P., Gasco, L., ... & Antonopoulou, E. (2022). Insect meals in feeds for juvenile gilthead seabream (Sparus aurata): Effects on growth, blood chemistry, hepatic metabolic enzymes, body composition and nutrient utilization. Aquaculture 561, 738674.
- Barton, B. A. (2002). Stress in Fishes: A Diversity of Responses with Particular Reference to Changes in Circulating Corticosteroids. Integrative and Comparative Biology, Volume 42, Issue 3, July 2002, Pages 517–525. [CrossRef]
- Cooke, S.J.; Lawrence, M.J.; Raby, G.D.; Teffer, A.K.; Jeffries, K.M.; Danylchuk, A.J.; Clark, T.D. Comment: Practices for Drawing Blood Samples from Teleost Fish. North American Journal of Aquaculture 2019, 81, 424–426. [Google Scholar] [CrossRef]
- Wendelaar Bonga, S.E. The Stress Response in Fish. Physiological reviews 1997, 77, 591–625. [Google Scholar] [CrossRef] [PubMed]
- Chrousos, G.P. Stress and Disorders of the Stress System. Nature reviews endocrinology 2009, 5, 374–381. [Google Scholar] [CrossRef]
- Barton, B.A.; Iwama, G.K. Physiological Changes in Fish from Stress in Aquaculture with Emphasis on the Response and Effects of Corticosteroids. Annual Review of fish diseases 1991, 1, 3–26. [Google Scholar] [CrossRef]
- Anastasiou, C.; Buchbauer, G. Essential Oils as Immunomodulators: Some Examples. Open Chemistry 2017, 15, 352–370. [Google Scholar] [CrossRef]
- Sandner, G.; Heckmann, M.; Weghuber, J. Immunomodulatory Activities of Selected Essential Oils. Biomolecules 2020, 10, 1139. [Google Scholar] [CrossRef]
- Zuo, X.; Gu, Y.; Wang, C.; Zhang, J.; Zhang, J.; Wang, G.; Wang, F. A Systematic Review of the Anti-Inflammatory and Immunomodulatory Properties of 16 Essential Oils of Herbs. Evidence-Based Complementary and Alternative Medicine 2020, 2020, 1–14. [Google Scholar] [CrossRef]
- Lang, M.; Ferron, P.-J.; Bursztyka, J.; Montjarret, A.; Duteil, E.; Bazire, A.; Bedoux, G. Evaluation of Immunomodulatory Activities of Essential Oils by High Content Analysis. Journal of Biotechnology 2019, 303, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Shourbela, R.M.; El-Hawarry, W.N.; Elfadadny, M.R.; Dawood, M.A.O. Oregano Essential Oil Enhanced the Growth Performance, Immunity, and Antioxidative Status of Nile Tilapia (Oreochromis niloticus) Reared under Intensive Systems. Aquaculture 2021, 542, 736868. [Google Scholar] [CrossRef]
- Mssillou, I., Bakour, M., Slighoua, M., Laaroussi, H., Saghrouchni, H., Amrati, F. E. Z., ... & Derwich, E. (2022). Investigation on wound healing effect of Mediterranean medicinal plants and some related phenolic compounds: A review. Journal of Ethnopharmacology 298, 115663. [CrossRef]
- Silva, M.B. da; Fraga, R.E.; Nishiyama, P.B.; Silva, I.S.S. da; Costa, N.L.B.; de Oliveira, L.A.A.; Rocha, M.A.; Juncá, F.A. Leukocyte Profiles in Odontophrynus carvalhoi (Amphibia: Odontophrynidae) Tadpoles Exposed to Organophosphate Chlorpyrifos Pesticides. Water, Air, & Soil Pollution 2020, 231, 372. [Google Scholar] [CrossRef]
- Weyts, F.A.A.; Verburg-van Kemenade, B.M.L.; Flik, G. Characterisation of Glucocorticoid Receptors in Peripheral Blood Leukocytes of Carp, Cyprinus carpio L. General and comparative endocrinology 1998, 111, 1–8. [Google Scholar] [CrossRef]
- Ortuno, J.; Esteban, M.A.; Meseguer, J. Effects of Short-Term Crowding Stress on the Gilthead Seabream (Sparus aurata L.) Innate Immune Response. Fish & shellfish immunology 2001, 11, 187–197. [Google Scholar] [CrossRef]
- Pickering, A.D. Cortisol-Induced Lymphocytopenia in Brown Trout, Salmo trutta L. General and comparative endocrinology 1984, 53, 252–259. [Google Scholar] [CrossRef]
- Hua, K. A Meta-Analysis of the Effects of Replacing Fish Meals with Insect Meals on Growth Performance of Fish. Aquaculture 2021, 530, 735732. [Google Scholar] [CrossRef]
- Reyes, M.; Rodríguez, M.; Montes, J.; Barroso, F.G.; Fabrikov, D.; Morote, E.; Sánchez-Muros, M.J. Nutritional and Growth Effect of Insect Meal Inclusion on Seabass (Dicentrarchuss labrax) Feeds. Fishes 2020, 5, 16. [Google Scholar] [CrossRef]
- Su, J.; Liu, Y.; Xi, L.; Lu, Q.; Liu, H.; Jin, J.; Yang, Y.; Zhu, X.; Han, D.; Xie, S. The Effect of Dietary Tenebrio Molitor Meal Inclusion on Growth Performance and Liver Health of Largemouth Bass (Micropterus salmoides). Journal of Insects as Food and Feed 2022, 8, 1297–1309. [Google Scholar] [CrossRef]
- Ma, H.; Lin, H.; Feng, H.; Putheti, R. Effects of Angelica Polysaccharide on Hepatocytes Apoptosis Induced by Exhaustive Exercise. Afr. J. Microbiol. Res 3(11), 774–777.
- Zhou, B.; Wu, L.-J.; Li, L.-H.; Tashiro, S.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin Protects against Isoproterenol-Induced Rat Cardiac Myocyte Injury through Mitochondrial Pathway after up-Regulation of SIRT1. Journal of pharmacological sciences 2006, 102, 387–395. [Google Scholar] [CrossRef]
- Dokou, S.; Mellidou, I.; Savvidou, S.; Stylianaki, I.; Panteli, N.; Antonopoulou, E.; Wang, J.; Grigoriadou, K.; Tzora, A.; Jin, L. A Phytobiotic Extract, in an Aqueous or in a Cyclodextrin Encapsulated Form, Added in Diet Affects Meat Oxidation, Cellular Responses and Intestinal Morphometry and Microbiota of Broilers. Frontiers in Animal Science 2023, 4, 1050170. [Google Scholar] [CrossRef]
- Cardinaletti, G.; Randazzo, B.; Messina, M.; Zarantoniello, M.; Giorgini, E.; Zimbelli, A.; Bruni, L.; Parisi, G.; Olivotto, I.; Tulli, F. Effects of Graded Dietary Inclusion Level of Full-Fat Hermetia illucens Prepupae Meal in Practical Diets for Rainbow Trout (Oncorhynchus mykiss). Animals 2019, 9, 251. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ji, H.; Zhang, B.; Zhou, J.; Yu, H. Defatted Black Soldier Fly (Hermetia Illucens) Larvae Meal in Diets for Juvenile Jian Carp (Cyprinus carpio Var. Jian): Growth Performance, Antioxidant Enzyme Activities, Digestive Enzyme Activities, Intestine and Hepatopancreas Histological Structure. Aquaculture 2017, 477, 62–70. [Google Scholar] [CrossRef]
- Roques, S.; Deborde, C.; Guimas, L.; Marchand, Y.; Richard, N.; Jacob, D.; Skiba-Cassy, S.; Moing, A.; Fauconneau, B. Integrative Metabolomics for Assessing the Effect of Insect (Hermetia illucens) Protein Extract on Rainbow Trout Metabolism. Metabolites 2020, 10, 83. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Saravana Bhavan, P.; Seenivasan, C.; Muralisankar, T.; Shanthi, R. Effects of Native Medicinal Herbs (Alternanthera sessilis,Eclipta alba and Cissus quadrangularis) on Growth Performance, Digestive Enzymes and Biochemical Constituents of the Monsoon River Prawn M Acrobrachium Malcolmsonii. Aquaculture nutrition 2015, 21, 496–506. [Google Scholar] [CrossRef]
- Xu, Z.; Li, X.Q.; Yang, H.; Poolsawat, L.; Wang, P.; Leng, X.J. Dietary Rutin Promoted the Growth, Serum Antioxidant Response and Flesh Collagen, Free Amino Acids Contents of Grass Carp (Ctenopharyngodon idella). Aquaculture Nutrition 2021, 27, 544–555. [Google Scholar] [CrossRef]
- Lee, D.-H.; Ra, C.-S.; Song, Y.-H.; Sung, K.-I.; Kim, J.-D. Effects of Dietary Garlic Extract on Growth, Feed Utilization and Whole Body Composition of Juvenile Sterlet Sturgeon (Acipenser ruthenus). Asian-Australasian Journal of Animal Sciences 2012, 25, 577. [Google Scholar] [CrossRef]
- Feidantsis, K.; Poertner, H.O.; Markou, T.; Lazou, A.; Michaelidis, B. Involvement of P38 MAPK in the Induction of H Sp70 During Acute Thermal Stress in Red Blood Cells of the Gilthead Sea Bream, Sparus aurata. Journal of Experimental Zoology Part A: Ecological Genetics and Physiology 2012, 317, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Stenberg, O.K.; Holen, E.; Piemontese, L.; Liland, N.S.; Lock, E.-J.; Espe, M.; Belghit, I. Effect of Dietary Replacement of Fish Meal with Insect Meal on in Vitro Bacterial and Viral Induced Gene Response in Atlantic Salmon (Salmo salar) Head Kidney Leukocytes. Fish & shellfish immunology 2019, 91, 223–232. [Google Scholar] [CrossRef]
- Seo, M.; Goo, T.-W.; Chung, M.Y.; Baek, M.; Hwang, J.-S.; Kim, M.-A.; Yun, E.-Y. Tenebrio molitor Larvae Inhibit Adipogenesis through AMPK and MAPKs Signaling in 3T3-L1 Adipocytes and Obesity in High-Fat Diet-Induced Obese Mice. International Journal of Molecular Sciences 2017, 18, 518. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Chatzigiannidou, I.; Feidantsis, K.; Kounna, C.; Chatzifotis, S. Effect of Water Temperature on Cellular Stress Responses in Meagre (Argyrosomus regius). Fish physiology and biochemistry 2020, 46, 1075–1091. [Google Scholar] [CrossRef]
- Feidantsis, K.; Antonopoulou, E.; Lazou, A.; Pörtner, H.O.; Michaelidis, B. Seasonal Variations of Cellular Stress Response of the Gilthead Sea Bream (Sparus aurata). Journal of Comparative Physiology B 2013, 183, 625–639. [Google Scholar] [CrossRef]
- Somayeh, S.M.; Jiun, Z.; Loh, Y.; Mousavi, S.; Loh, J.-Y.; Zahedinezhad, S. A review on insect meals in aquaculture: The immunomodulatory and physiological effects. International Aquatic Research 2020, 12, 100–115. [Google Scholar] [CrossRef]
- Reverter, M.; Tapissier-Bontemps, N.; Sarter, S.; Sasal, P.; Caruso, D. Moving towards More Sustainable Aquaculture Practices: A Meta-Analysis on the Potential of Plant-Enriched Diets to Improve Fish Growth, Immunity and Disease Resistance. Reviews in Aquaculture 2021, 13, 537–555. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).