Submitted:
09 January 2024
Posted:
17 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
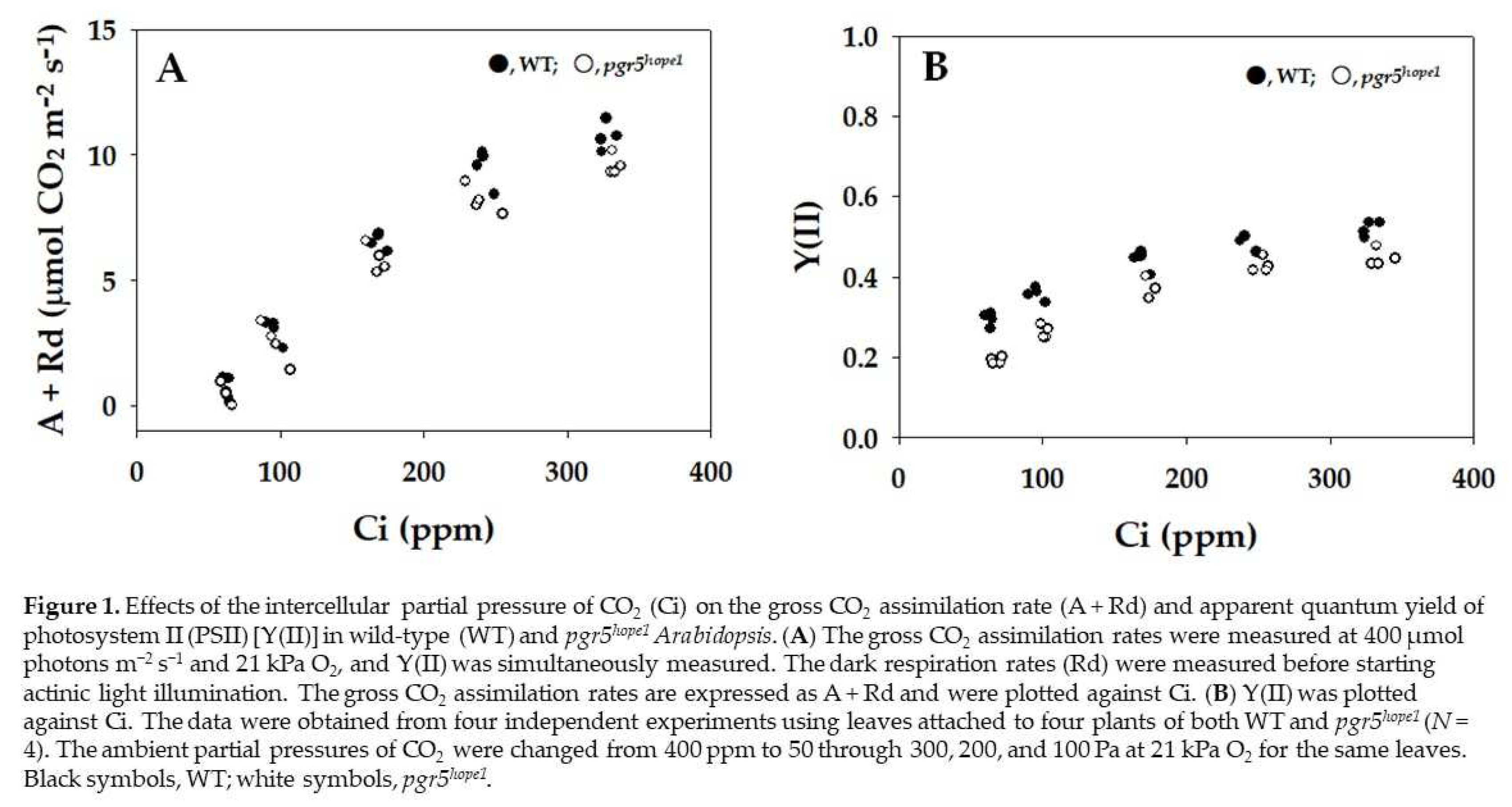
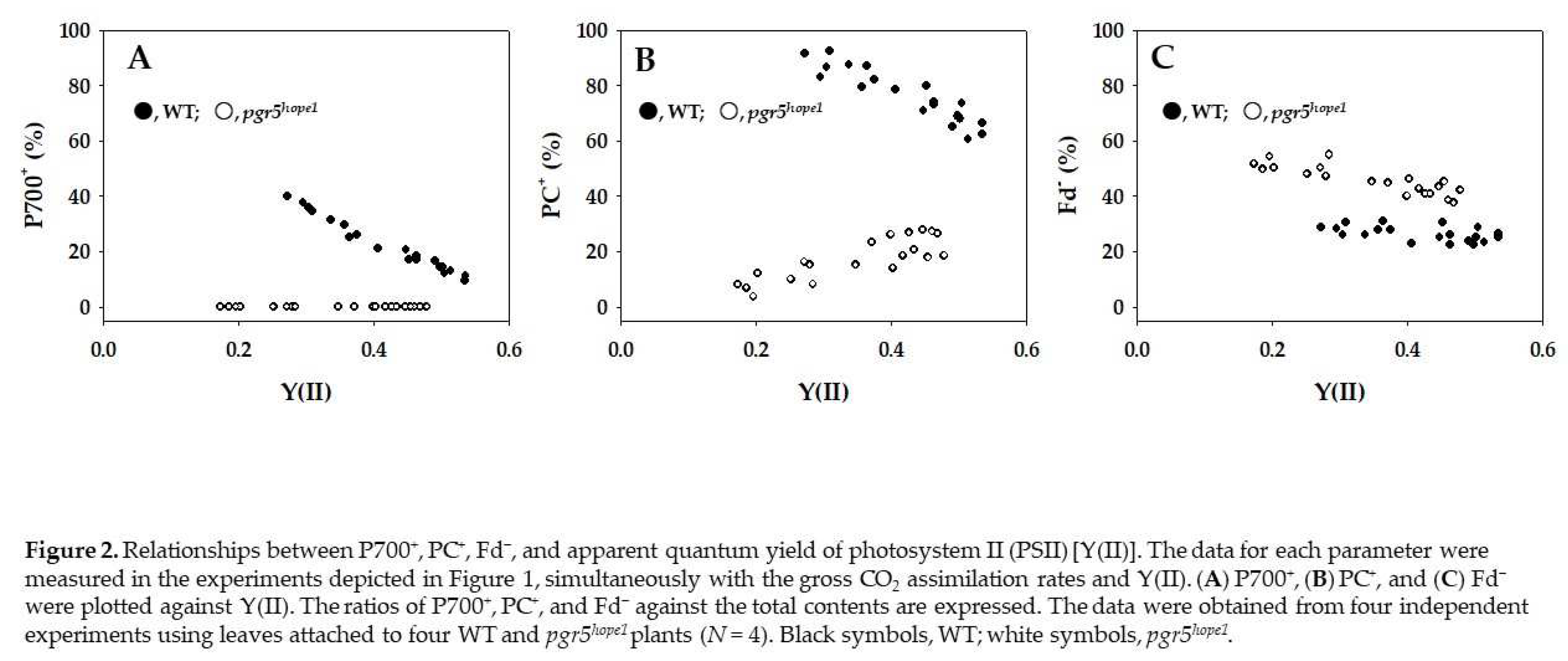
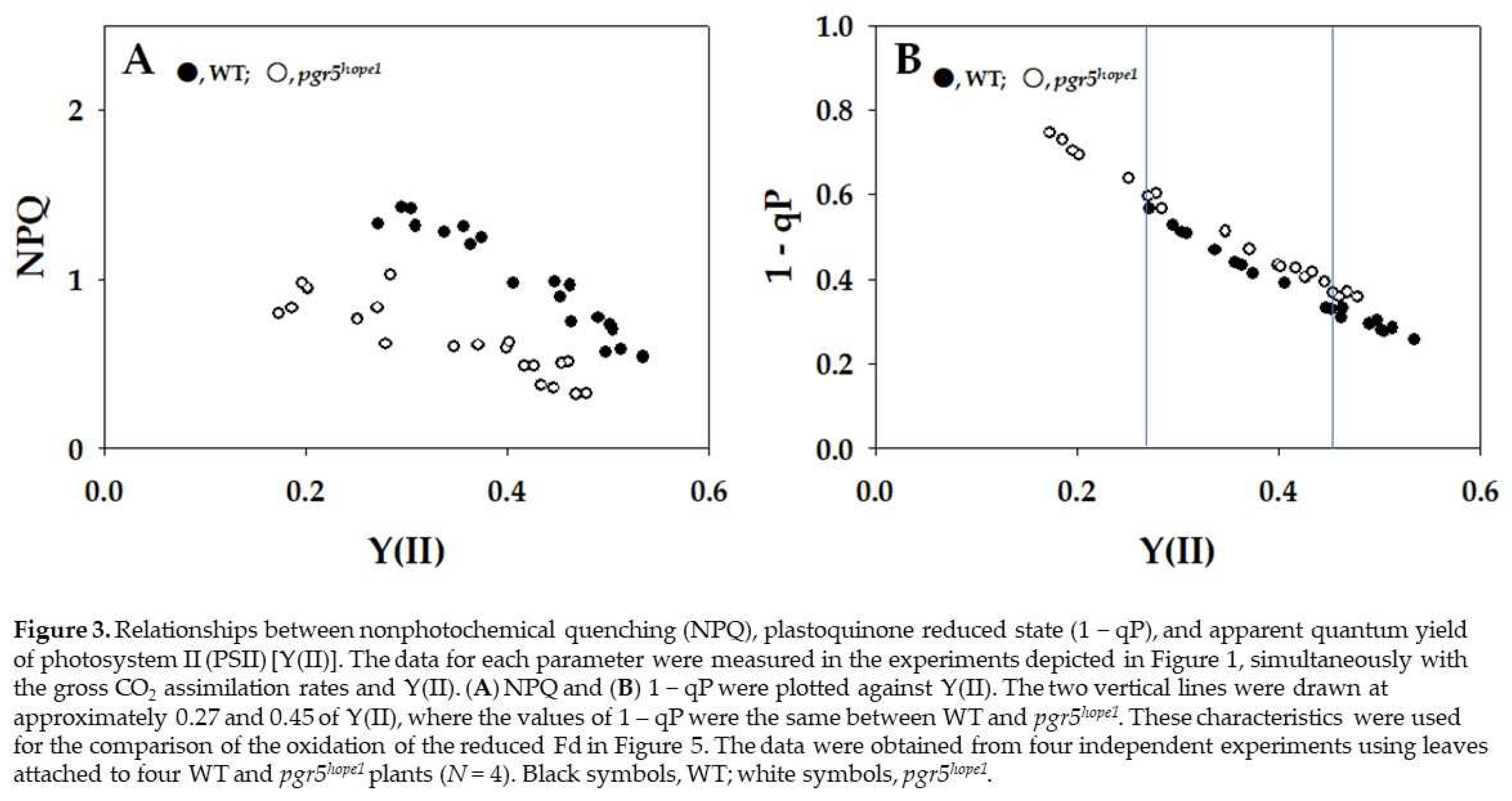
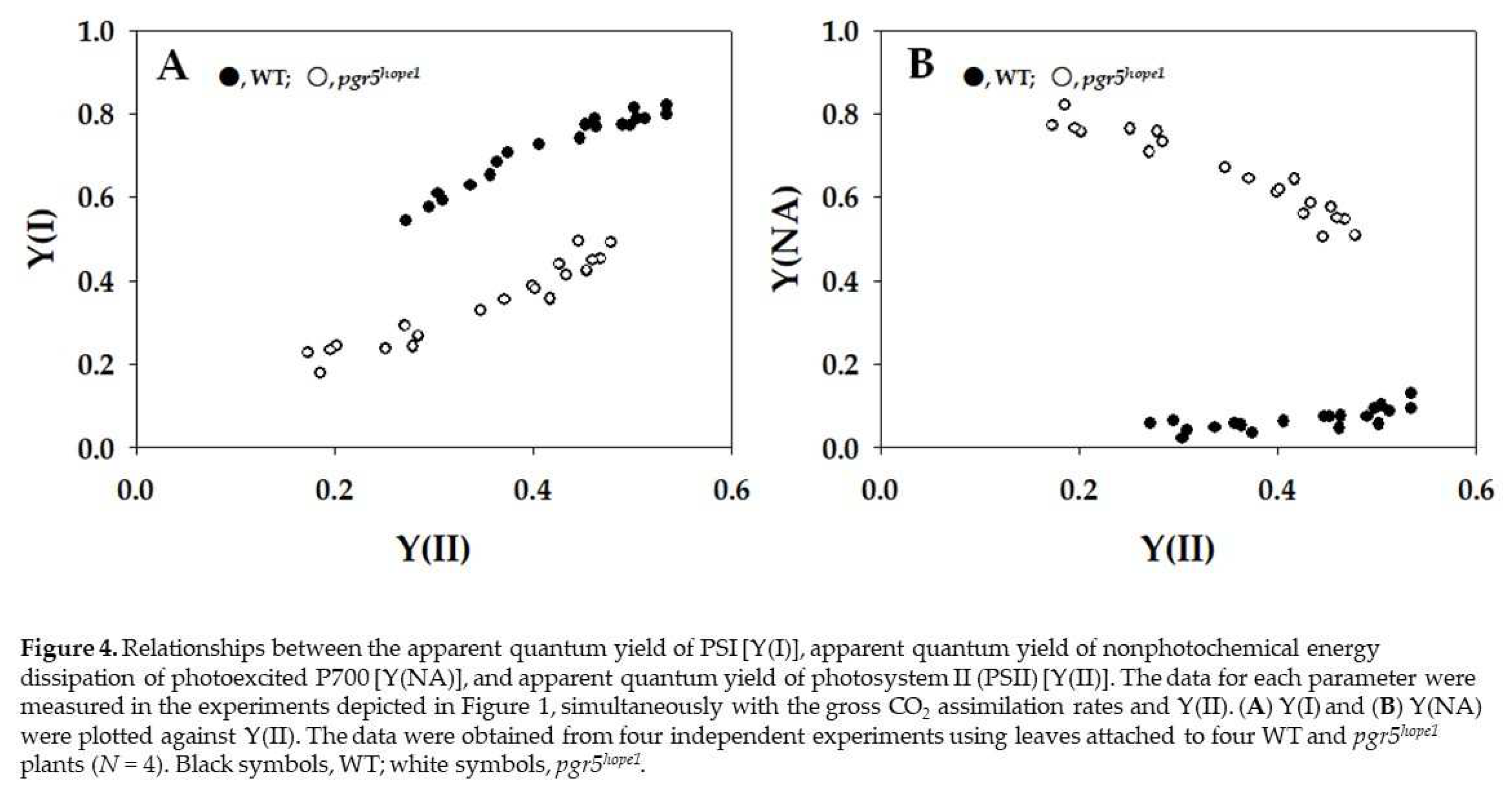
2. Results
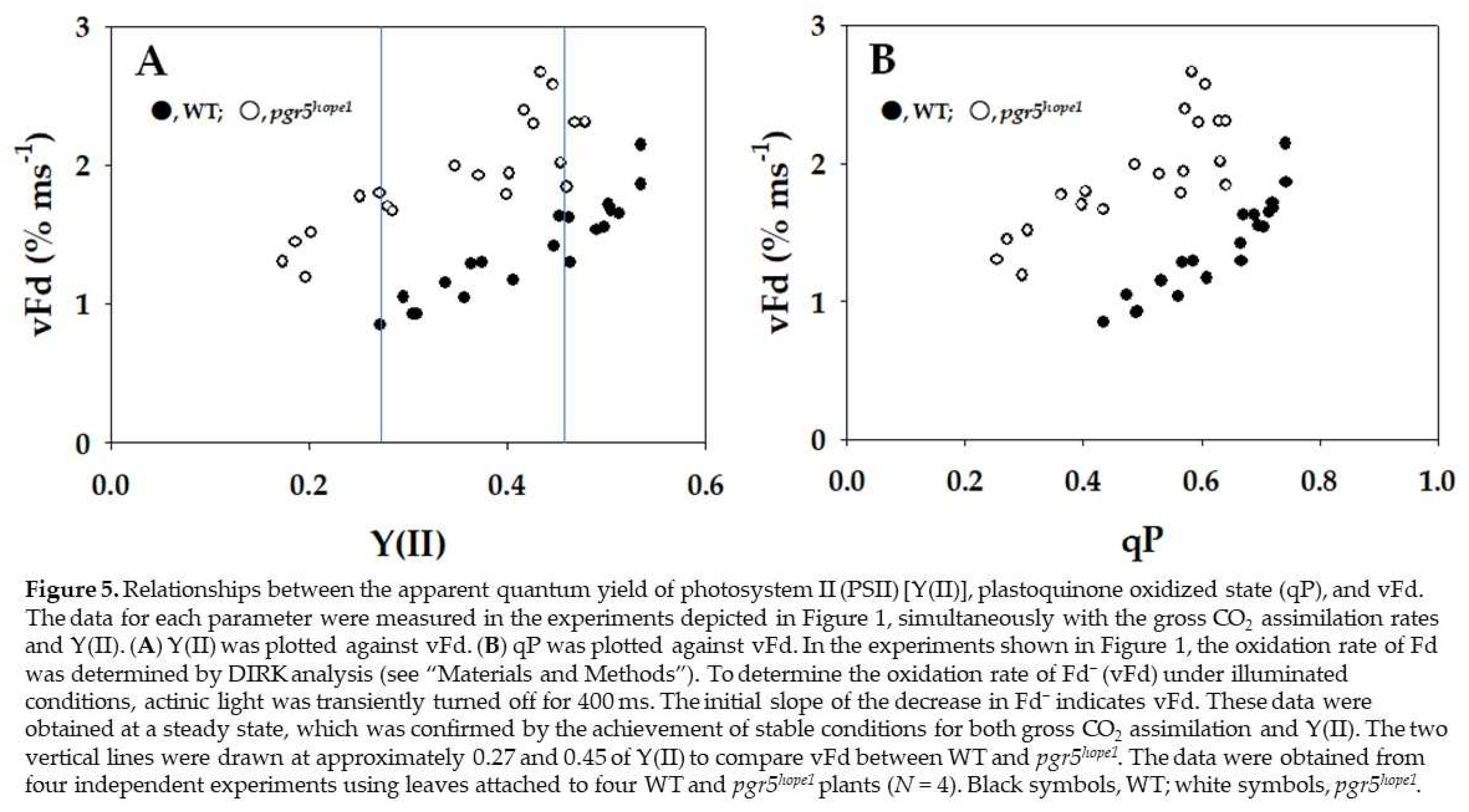
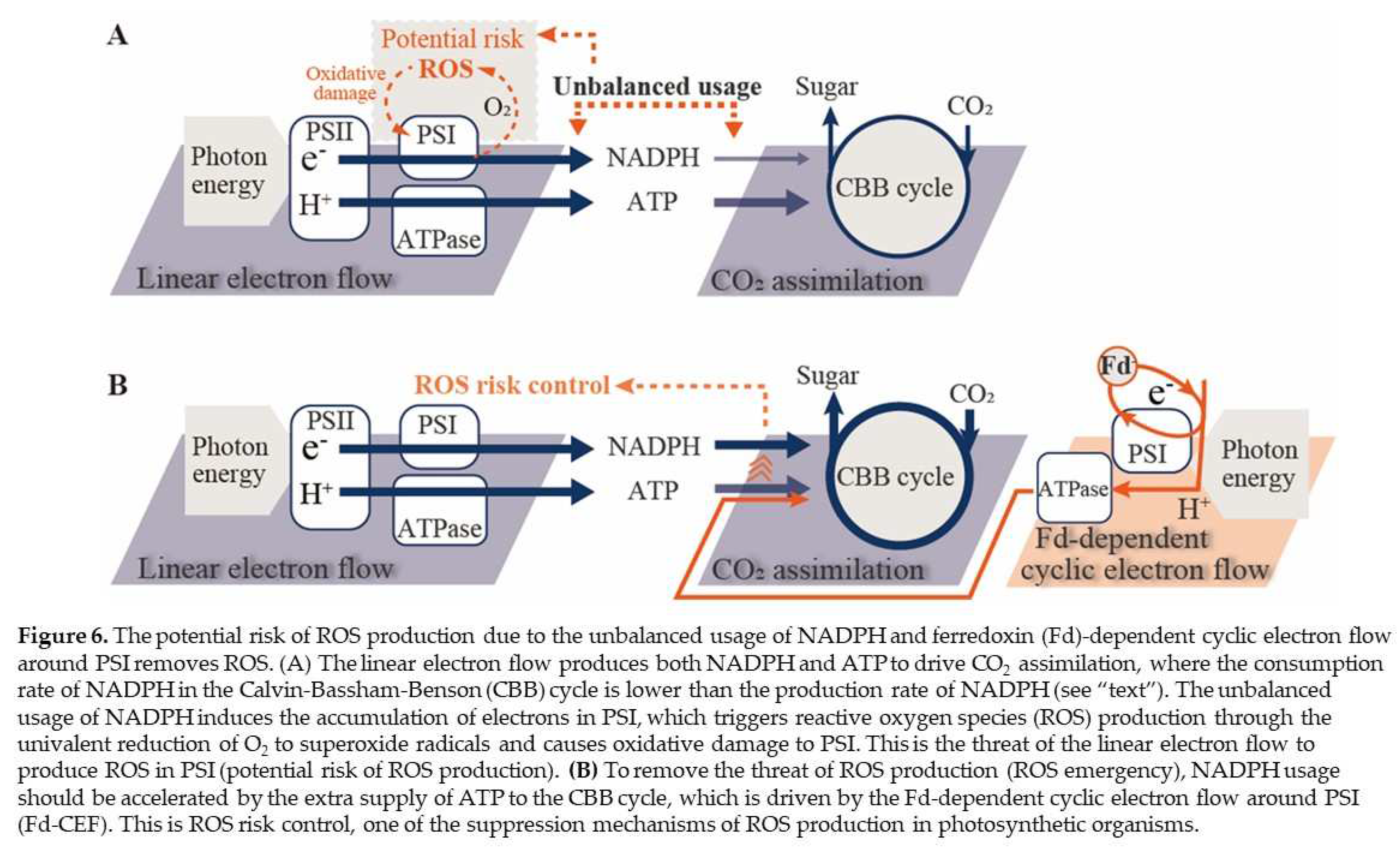
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Determination of Chlorophyll and Nitrogen
4.3. Simultaneous Measurements of Chlorophyll Fluorescence, P700, PC, and Fd Signals with Gas Exchange
4.4. Simultaneous Measurements of Electrochromic shift (ECS)-Signals with Gas Exchange
4.5. Statistical Analytics
Supplemental Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allen, J. F. Cyclic, pseudocyclic and noncyclic photophosphorylation: new links in the chain. Trend. Plant Sci. 2003, 8, (1), 15-9. [CrossRef]
- Miyake, C.; Miyata, M.; Shinzaki, Y.; Tomizawa, K. CO2 response of cyclic electron flow around PSI (CEF-PSI) in tobacco leaves--relative electron fluxes through PSI and PSII determine the magnitude of non-photochemical quenching (NPQ) of Chl fluorescence. Plant Cell Physiol. 2005, 46, (4), 629-37. [CrossRef]
- Nawrocki, W. J.; Bailleul, B.; Picot, D.; Cardol, P.; Rappaport, F.; Wollman, F. A.; Joliot, P. The mechanism of cyclic electron flow. Biochim. Biophys. Acta. Bioerg. 2019, 1860, (5), 433-438. [CrossRef] [PubMed]
- Furutani, R.; Wada, S.; Ifuku, K.; Maekawa, S.; Miyake, C. Higher reduced state of Fe/S-signals, with the suppressed oxidation of P700, causes PSI Inactivation in Arabidopsis thaliana. Antioxidants (Basel, Switzerland) 2022, 12, (1). [CrossRef]
- Hashimoto, M.; Endo, T.; Peltier, G.; Tasaka, M.; Shikanai, T. A nucleus-encoded factor, CRR2, is essential for the expression of chloroplast ndhB in Arabidopsis. Plant J. 2003, 36, (4), 541-9. [CrossRef] [PubMed]
- Kono, M.; Noguchi, K.; Terashima, I. Roles of the cyclic electron flow around PSI (CEF-PSI) and O₂-dependent alternative pathways in regulation of the photosynthetic electron flow in short-term fluctuating light in Arabidopsis thaliana. Plant Cell Physiol. 2014, 55, (5), 990-1004. [CrossRef]
- Munekage, Y.; Hojo, M.; Meurer, J.; Endo, T.; Tasaka, M.; Shikanai, T. PGR5 is involved in cyclic electron flow around photosystem I and is essential for photoprotection in Arabidopsis. Cell 2002, 110, (3), 361-71. [CrossRef]
- Shikanai, T.; Endo, T.; Hashimoto, T.; Yamada, Y.; Asada, K.; Yokota, A. Directed disruption of the tobacco ndhB gene impairs cyclic electron flow around photosystem I. Proc. Natl. Acad. Sci. USA 1998, 95, (16), 9705-9. [CrossRef] [PubMed]
- Yamamoto, H.; Shikanai, T. PGR5-dependent cyclic electron flow protects photosystem I under fluctuating light at donor and acceptor sides. Plant Physiol. 2019, 179, (2), 588-600. [CrossRef]
- Yamamoto, H.; Shikanai, T. Does the arabidopsis proton gradient regulation5 mutant leak protons from the thylakoid membrane? Plant Physiol. 2020, 184, (1), 421-427. [CrossRef]
- Yamori, W.; Shikanai, T. Physiological functions of cyclic electron transport around photosystem I in sustaining photosynthesis and plant growth. Annual Recv. Plant Biol. 2016, 67, 81-106. [CrossRef]
- Miyake, C.; Shinzaki, Y.; Miyata, M.; Tomizawa, K. Enhancement of cyclic electron flow around PSI at high light and its contribution to the induction of non-photochemical quenching of chl fluorescence in intact leaves of tobacco plants. Plant Cell Physiol. 2004, 45, (10), 1426-33. [CrossRef] [PubMed]
- Yamamoto, H.; Takahashi, S.; Badger, M. R.; Shikanai, T. Artificial remodelling of alternative electron flow by flavodiiron proteins in Arabidopsis. Nature plants 2016, 2, 16012. [CrossRef]
- Wada, S.; Yamamoto, H.; Suzuki, Y.; Yamori, W.; Shikanai, T.; Makino, A. Flavodiiron protein substitutes for cyclic electron flow without competing CO2 assimilation in rice. Plant Physiol. 2018, 176, (2), 1509-1518. [CrossRef] [PubMed]
- Furutani, R.; Ohnishi, M.; Mori, Y.; Wada, S.; Miyake, C. The difficulty of estimating the electron transport rate at photosystem I. J. Plant Res. 2022, 135, (4), 565-577. [CrossRef]
- Klughammer, C.; Schreiber, U. An improved method, using saturating light pulses, for the determination of photosystem I quantum yield via P700-absorbance changes at 830 nm. Planta 1994, 192, (2), 261-268. [CrossRef]
- Miyake, C.; Schreiber, U.; Asada, K. Ferredoxin-Dependent and Antimycin A-Sensitive Reduction of Cytochrome b-559 by Far-Red Light in Maize Thylakoids; Participation of a Menadiol-Reducible Cytochrome b-559 in Cyclic Electron Flow. Plant Cell Physiol. 1995, 36, (4), 743-748. [CrossRef]
- Peltier, G.; Aro, E. M.; Shikanai, T. NDH-1 and NDH-2 Plastoquinone Reductases in Oxygenic Photosynthesis. Ann. Rev. Plant Biol. 2016, 67, 55-80. [CrossRef]
- Fisher, N.; Kramer, D. M. Non-photochemical reduction of thylakoid photosynthetic redox carriers in vitro: relevance to cyclic electron flow around photosystem I? Biochim. Biophys. Acta Bioenerg. 2014, 1837, (12), 1944-1954. [CrossRef]
- Ohnishi, M.; Maekawa, S.; Wada, S.; Ifuku, K.; Miyake, C. Evaluating the oxidation rate of reduced ferredoxin in Arabidopsis thaliana independent of photosynthetic linear electron flow: Plausible activity of ferredoxin-dependent cyclic electron flow around photosystem I. Int. J. Mol. Sci. 2023, 24, (15). [CrossRef]
- Kadota, K.; Furutani, R.; Makino, A.; Suzuki, Y.; Wada, S.; Miyake, C. Oxidation of P700 induces alternative electron flow in photosystem I in wheat leaves. Plants (Basel, Switzerland) 2019, 8, (6). [CrossRef]
- Shimakawa, G.; Miyake, C. Photosynthetic linear electron flow drives CO2 assimilation in maize leaves. Int. J. Mol. Sci. 2021, 22, (9). [CrossRef]
- Wada, S.; Amako, K.; Miyake, C. Identification of a novel mutation exacerbated the PSI photoinhibition in pgr5/pgrl1 mutants; Caution for overestimation of the phenotypes in Arabidopsis pgr5-1 mutant. Cells 2021, 10, (11). [CrossRef]
- Shimakawa, G.; Miyake, C. Changing frequency of fluctuating light reveals the molecular mechanism for P700 oxidation in plant leaves. Plant Direct 2018, 2, (7), e00073. [CrossRef]
- Miyake, C. Molecular Mechanism of Oxidation of P700 and suppression of ROS production in photosystem I in response to electron-sink limitations in C3 plants. Antioxidants (Basel, Switzerland) 2020, 9, (3). [CrossRef]
- Rantala, S.; Lempiäinen, T.; Gerotto, C.; Tiwari, A.; Aro, E. M.; Tikkanen, M. PGR5 and NDH-1 systems do not function as protective electron acceptors but mitigate the consequences of PSI inhibition. Biochim. Biophys. Acta Bioenerg. 2020, 1861, (3), 148154. [CrossRef]
- Heber, U.; Walker, D. Concerning a dual function of coupled cyclic electron transport in leaves. Plant Physiol. 1992, 100, (4), 1621-6. [CrossRef] [PubMed]
- Miyake, C.; Amako, K.; Shiraishi, N.; Sugimoto, T. Acclimation of tobacco leaves to high light intensity drives the plastoquinone oxidation system-Relationship among the fraction of open PSII centers, non-photochemical quenching of Chl fluorescence and the maximum quantum yield of PSII in the dark. Plant Cell Physiol. 2009, 50, (4), 730-43. [CrossRef] [PubMed]
- Ruban, A. V.; Murchie, E. H. Assessing the photoprotective effectiveness of non-photochemical chlorophyll fluorescence quenching: a new approach. Biochim. Biophys. Acata Bioenerg. 2012, 1817, (7), 977-82. [CrossRef]
- Cramer, W. A.; Zhang, H.; Yan, J.; Kurisu, G.; Smith, J. L. Transmembrane traffic in the cytochrome b6f complex. Ann. Rev. Biochem. 2006, 75, 769-90. [CrossRef] [PubMed]
- Cramer, W. A.; Zhang, H. Consequences of the structure of the cytochrome b6f complex for its charge transfer pathways. Biochim. Biophys. Acta Bioenerg. 2006, 1757, (5-6), 339-45. [CrossRef]
- Sarewicz, M.; Pintscher, S.; Pietras, R.; Borek, A.; Bujnowicz, Ł.; Hanke, G.; Cramer, W. A.; Finazzi, G.; Osyczka, A. Catalytic reactions and energy conservation in the cytochrome bc1 and b6f complexes of energy-transducing membranes. Chem. Rev. 2021, 121, (4), 2020-2108. [CrossRef] [PubMed]
- Driever, S. M.; Baker, N. R. The water-water cycle in leaves is not a major alternative electron sink for dissipation of excess excitation energy when CO2 assimilation is restricted. Plant Cell Env. 2011, 34, (5), 837-46. [CrossRef]
- Genty, B.; Harbinson, J.; Briantais, J. M.; Baker, N. R. The relationship between non-photochemical quenching of chlorophyll fluorescence and the rate of photosystem 2 photochemistry in leaves. Photosyn. Res. 1990, 25, (3), 249-57. [CrossRef]
- Ruuska, S. A.; Badger, M. R.; Andrews, T. J.; von Caemmerer, S. Photosynthetic electron sinks in transgenic tobacco with reduced amounts of Rubisco: Little evidence for significant Mehler reaction. J. Exp. Bot. 2000, 51 Spec No, 357-68. [CrossRef]
- Sejima, T.; Hanawa, H.; Shimakawa, G.; Takagi, D.; Suzuki, Y.; Fukayama, H.; Makino, A.; Miyake, C. Post-illumination transient O2 -uptake is driven by photorespiration in tobacco leaves. Physiol. Plant. 2016, 156, (2), 227-238. [CrossRef]
- Smith, K.; Strand, D. D.; Kramer, D. M.; Walker, B. J. The role of photorespiration in preventing feedback regulation via ATP synthase in Nicotiana tabacum. Plant Cell Env. 2023. [CrossRef]
- von Caemmerer, S.; Farquhar, G. D. Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 1981, 153, (4), 376-87. [CrossRef]
- Sejima, T.; Takagi, D.; Fukayama, H.; Makino, A.; Miyake, C. Repetitive short-pulse light mainly inactivates photosystem I in sunflower leaves. Plant Cell Physiol. 2014, 55, (6), 1184-93. [CrossRef]
- Ozaki, H.; Mizokami, Y.; Sugiura, D.; Sohtome, T.; Miyake, C.; Sakai, H.; Noguchi, K. Tight relationship between two photosystems is robust in rice leaves under various nitrogen conditions. J. Plant Res. 2023, 136, (2), 201-210. [CrossRef]
- Wada, S.; Takagi, D.; Miyake, C.; Makino, A.; Suzuki, Y. Responses of the photosynthetic electron transport reactions stimulate the oxidation of the reaction center chlorophyll of photosystem I, P700, under drought and high temperatures in rice. Int. J. Mol. Sci. 2019, 20, (9). [CrossRef]
- Wada, S.; Suzuki, Y.; Miyake, C. Photorespiration enhances acidification of the thylakoid lumen, reduces the plastoquinone pool, and contributes to the oxidation of P700 at a lower partial pressure of CO2 in wheat leaves. Plants (Basel, Switzerland) 2020, 9. [CrossRef]
- Kasting, J. F. Atmospheric science. How was early earth kept warm? Science (New York, N.Y.) 2013, 339, (6115), 44-5. [CrossRef]
- Ohnishi, M.; Furutani, R.; Sohtome, T.; Suzuki, T.; Wada, S.; Tanaka, S.; Ifuku, K.; Ueno, D.; Miyake, C. Photosynthetic parameters show specific responses to essential mineral deficiencies. Antioxidants (Basel, Switzerland) 2021, 10, (7). [CrossRef]
- Baker, N. R. Chlorophyll fluorescence: a probe of photosynthesis in vivo. Ann. Rev. Plant Biol. 2008, 59, 89-113. [CrossRef]
- Bilger, W.; Björkman, O. Relationships among violaxanthin deepoxidation, thylakoid membrane conformation, and nonphotochemical chlorophyll fluorescence quenching in leaves of cotton (Gossypium hirsutum L.). Planta 1994, 193, (2), 238-246. [CrossRef]
- Oxborough, K.; Baker, N. R. An evaluation of the potential triggers of photoinactivation of photosystem II in the context of a Stern-Volmer model for downregulation and the reversible radical pair equilibrium model. Phil. Trans. Royal Soc. Lond. Series B, Biol. Sci. USA 2000, 355, (1402), 1489-98. [CrossRef]
- Klughammer, C.; Schreiber, U. Deconvolution of ferredoxin, plastocyanin, and P700 transmittance changes in intact leaves with a new type of kinetic LED array spectrophotometer. Photosyn. Res. 2016, 128, (2), 195-214. [CrossRef]
- Böhme, H. Quantitative determination of ferredoxin, ferredoxin-NADP+ reductase and plastocyanin in spinach chloroplasts. Eur. J. Biochem. 1978, 83, (1), 137-41. [CrossRef]
- Kozuleva, M.; Goss, T.; Twachtmann, M.; Rudi, K.; Trapka, J.; Selinski, J.; Ivanov, B.; Garapati, P.; Steinhoff, H. J.; Hase, T.; Scheibe, R.; Klare, J. P.; Hanke, G. T. Ferredoxin:NADP(H) oxidoreductase abundance and location influences redox poise and stress tolerance. Plant Physiol. 2016, 172, (3), 1480-1493. [CrossRef] [PubMed]
- Yamamoto, H.; Kato, H.; Shinzaki, Y.; Horiguchi, S.; Shikanai, T.; Hase, T.; Endo, T.; Nishioka, M.; Makino, A.; Tomizawa, K.; Miyake, C. Ferredoxin limits cyclic electron flow around PSI (CEF-PSI) in higher plant-Stimulation of CEF-PSI enhances non-photochemical quenching of Chl fluorescence in transplastomic tobacco. Plant Cell Physiol. 2006, 47, (10), 1355-71. [CrossRef]
- Oja, V.; Eichelmann, H.; Peterson, R. B.; Rasulov, B.; Laisk, A. Deciphering the 820 nm signal: redox state of donor side and quantum yield of Photosystem I in leaves. Photosyn. Res. 2003, 78, (1), 1-15. [CrossRef] [PubMed]
- Cherepanov, D. A.; Milanovsky, G. E.; Petrova, A. A.; Tikhonov, A. N.; Semenov, A. Y. Electron Transfer through the Acceptor Side of Photosystem I: Interaction with Exogenous Acceptors and Molecular Oxygen. Biochem. Biokhim. 2017, 82, (11), 1249-1268. [CrossRef] [PubMed]
- Sacksteder, C. A.; Kramer, D. M. Dark-interval relaxation kinetics (DIRK) of absorbance changes as a quantitative probe of steady-state electron transfer. Photosyn. Res. 2000, 66, (1), 145-158. [CrossRef] [PubMed]
- Avenson, T. J.; Cruz, J. A.; Kramer, D. M. Modulation of energy-dependent quenching of excitons in antennae of higher plants. Proc. Natl. Acad. Sci. USA 2004, 101, (15), 5530-5. [CrossRef] [PubMed]
- Cruz, J. A.; Sacksteder, C. A.; Kanazawa, A.; Kramer, D. M. Contribution of electric field (ΔΨ) to steady-state transthylakoid proton motive force (pmf) in vitro and in vivo. control of pmf parsing into ΔΨ and ΔpH by ionic strength. Biochem. 2001, 40, (5), 1226-37. [CrossRef]
- Klughammer, C.; Siebke, K.; Schreiber, U. Continuous ECS-indicated recording of the proton-motive charge flux in leaves. Photosyn. Res. 2013, 117, (1-3), 471-87. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



