Submitted:
03 January 2024
Posted:
05 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
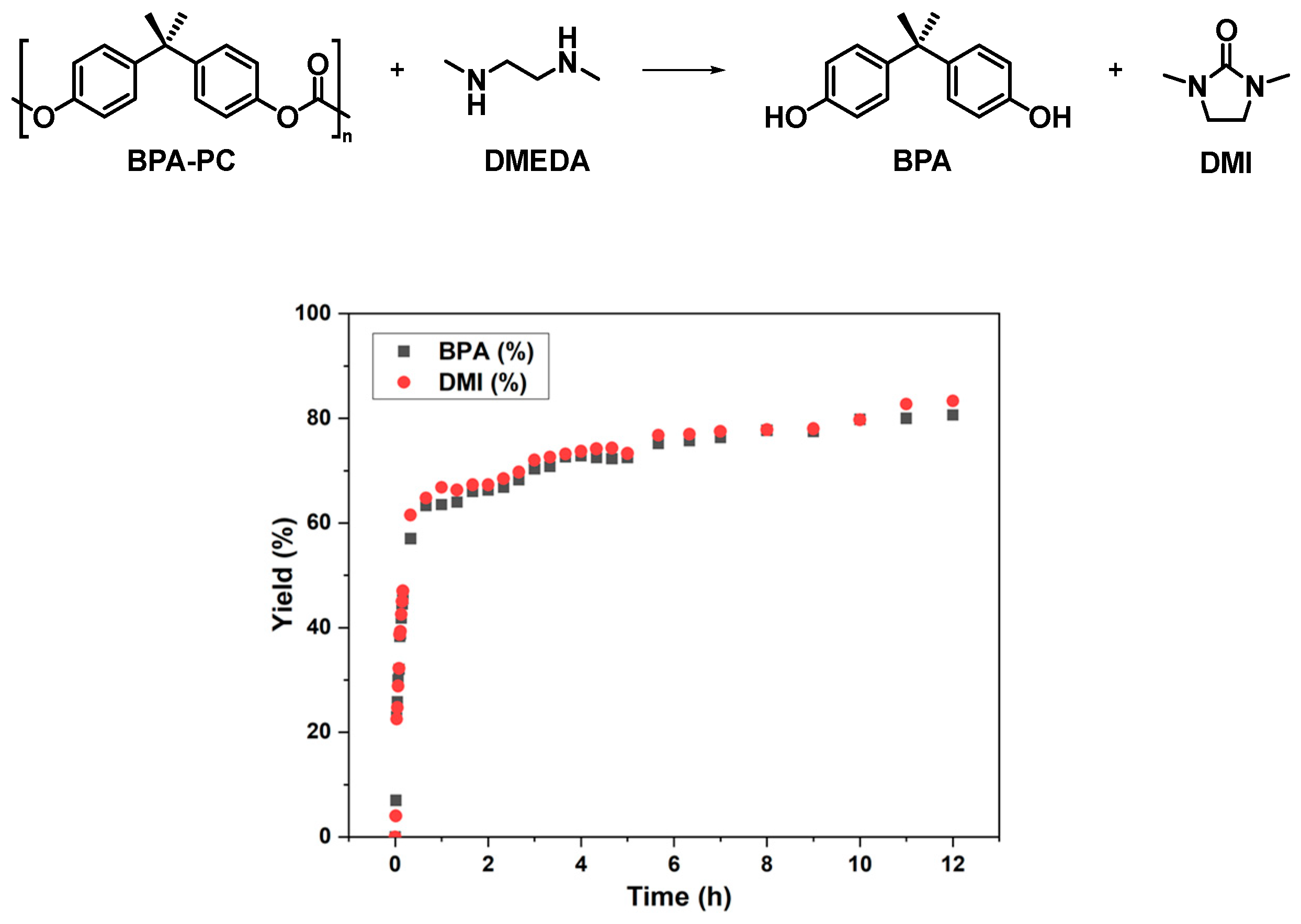
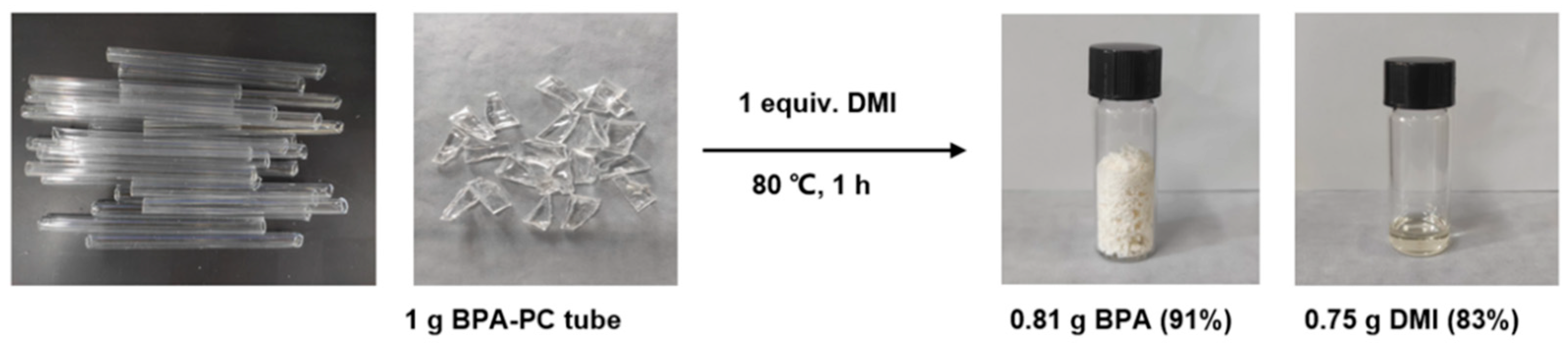
2.1. Depolymerization of BPA-PC under Solvent-Free Conditions
2.2. Effect of Solvent Type on the Reaction
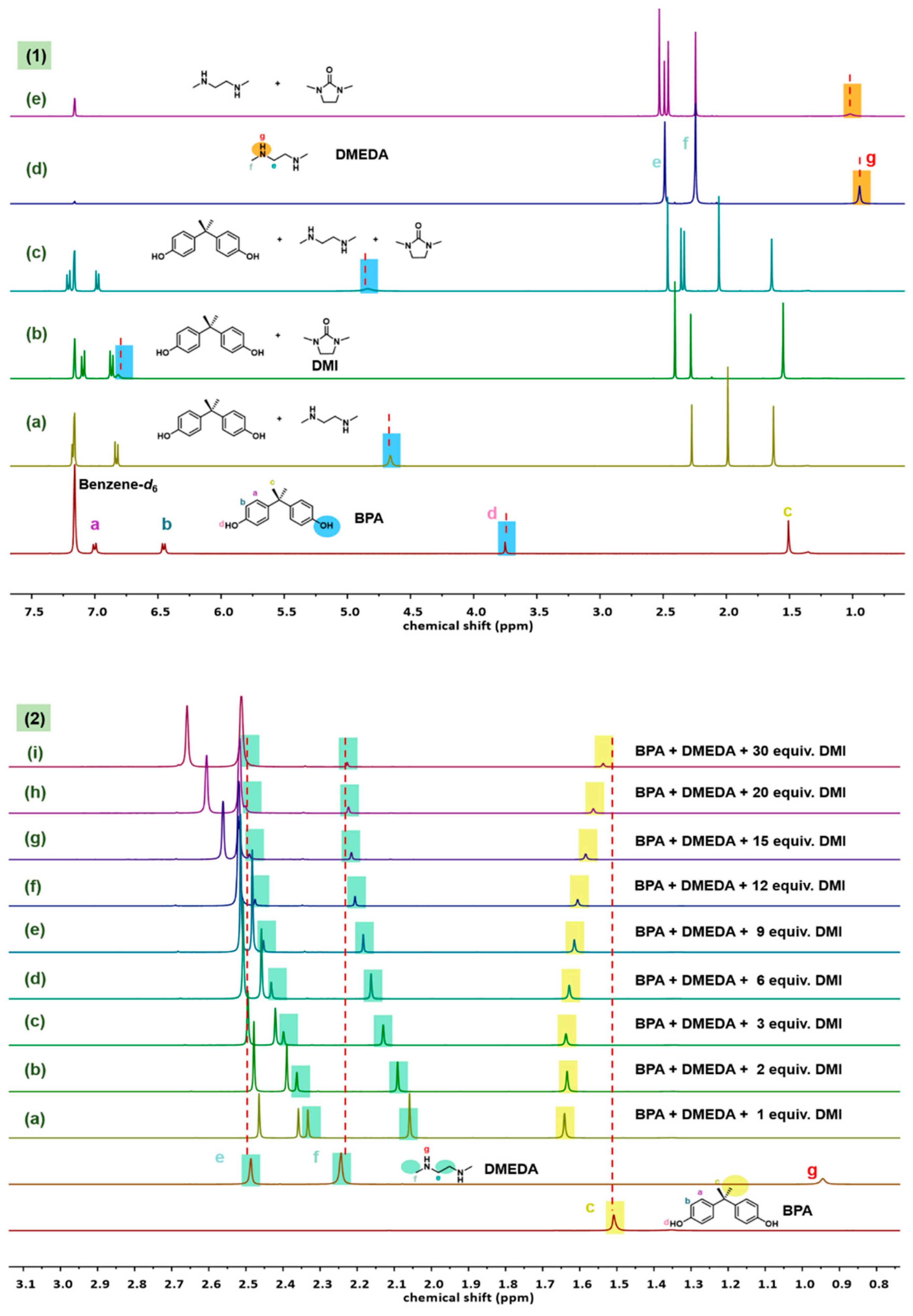
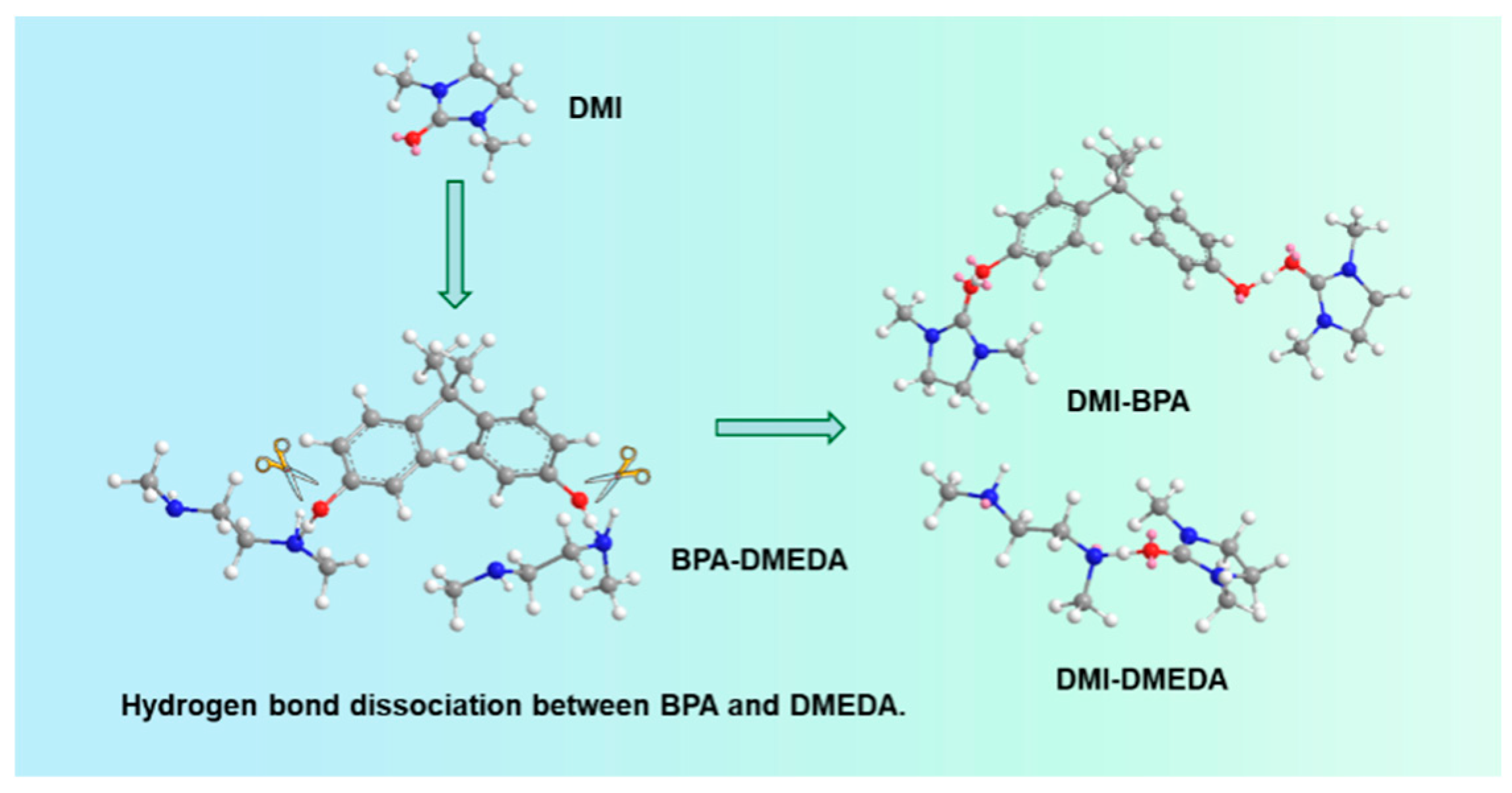
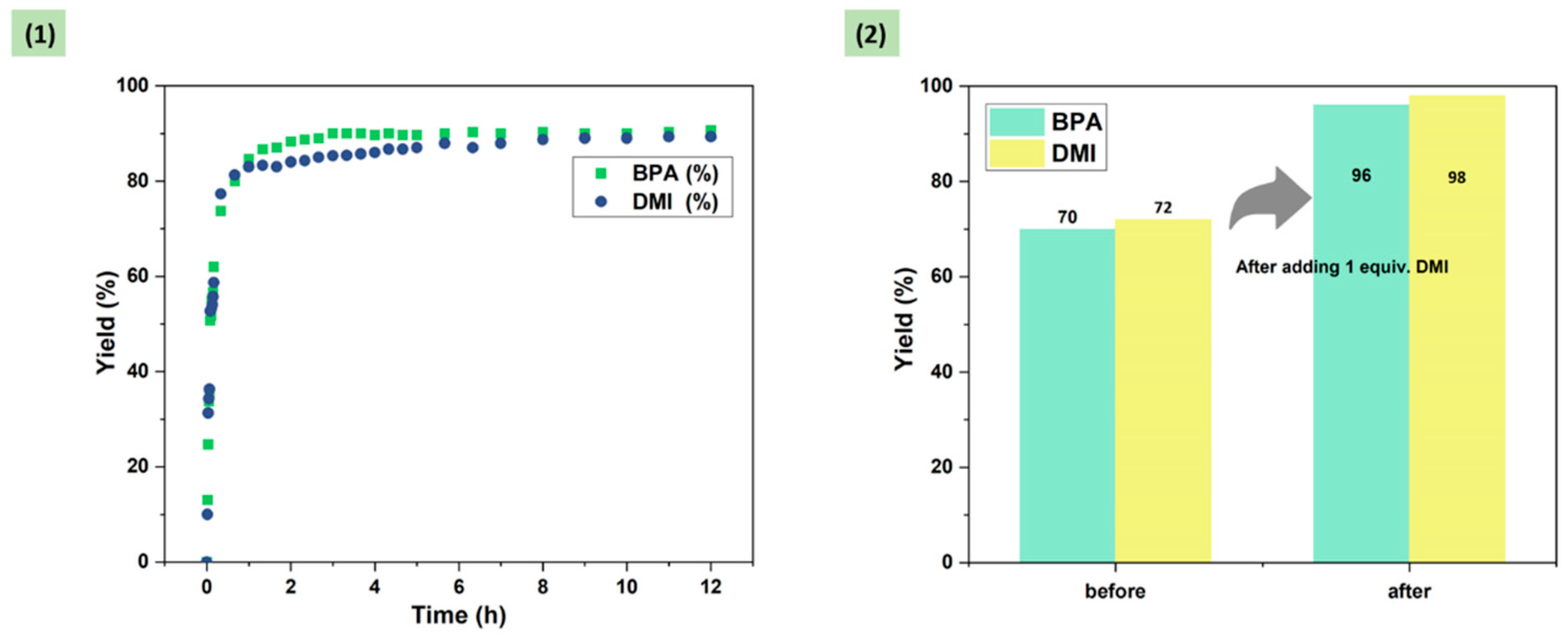
2.3. Probe into the Role of DMI
2.4. Optimization of DMI Equivalent
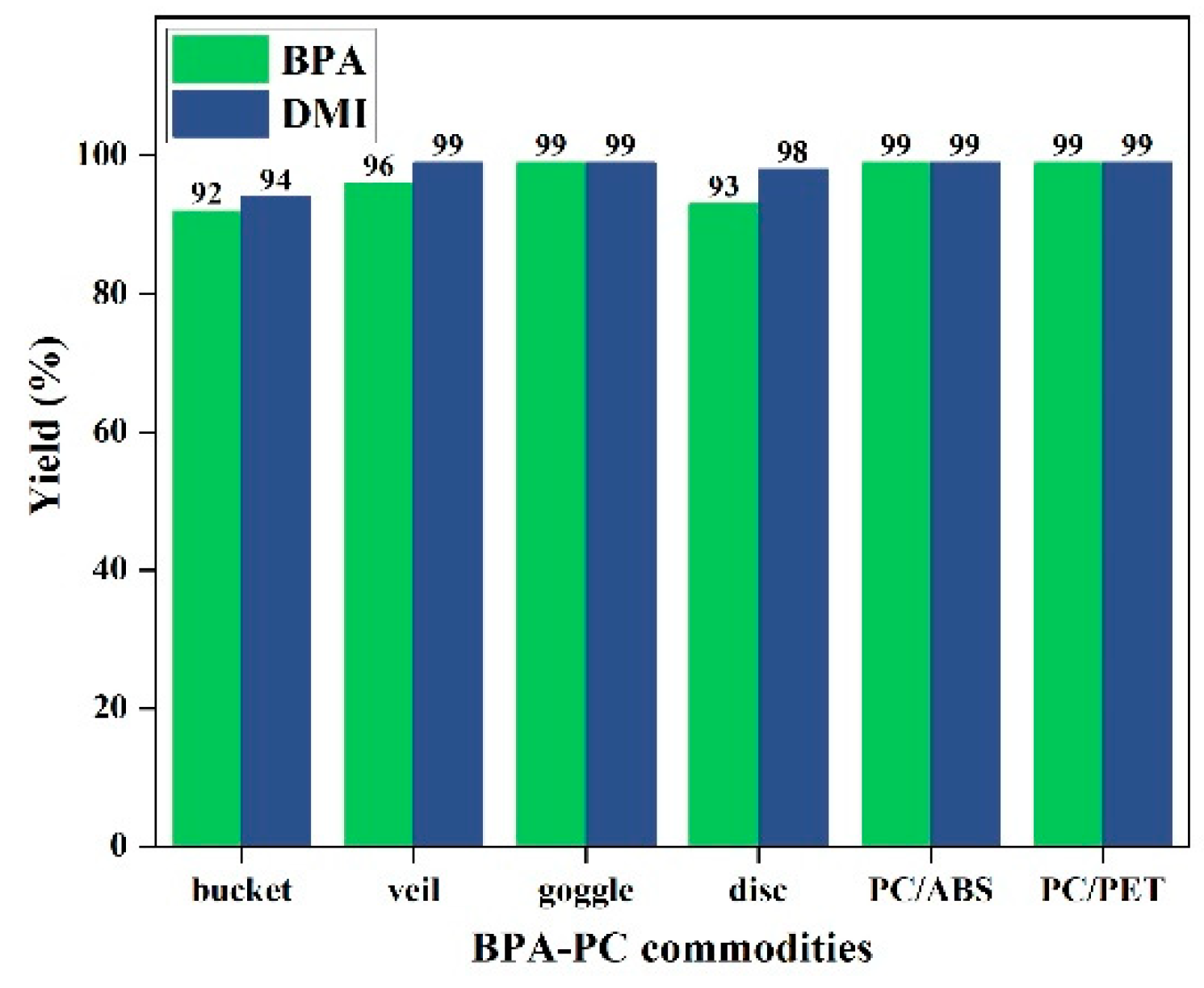
2.5. Degradation of Common BPA-PC Waste Plastics
3. Materials and Methods
3.1. Materials and Reagents
3.2. Depolymerization of BPA-PC
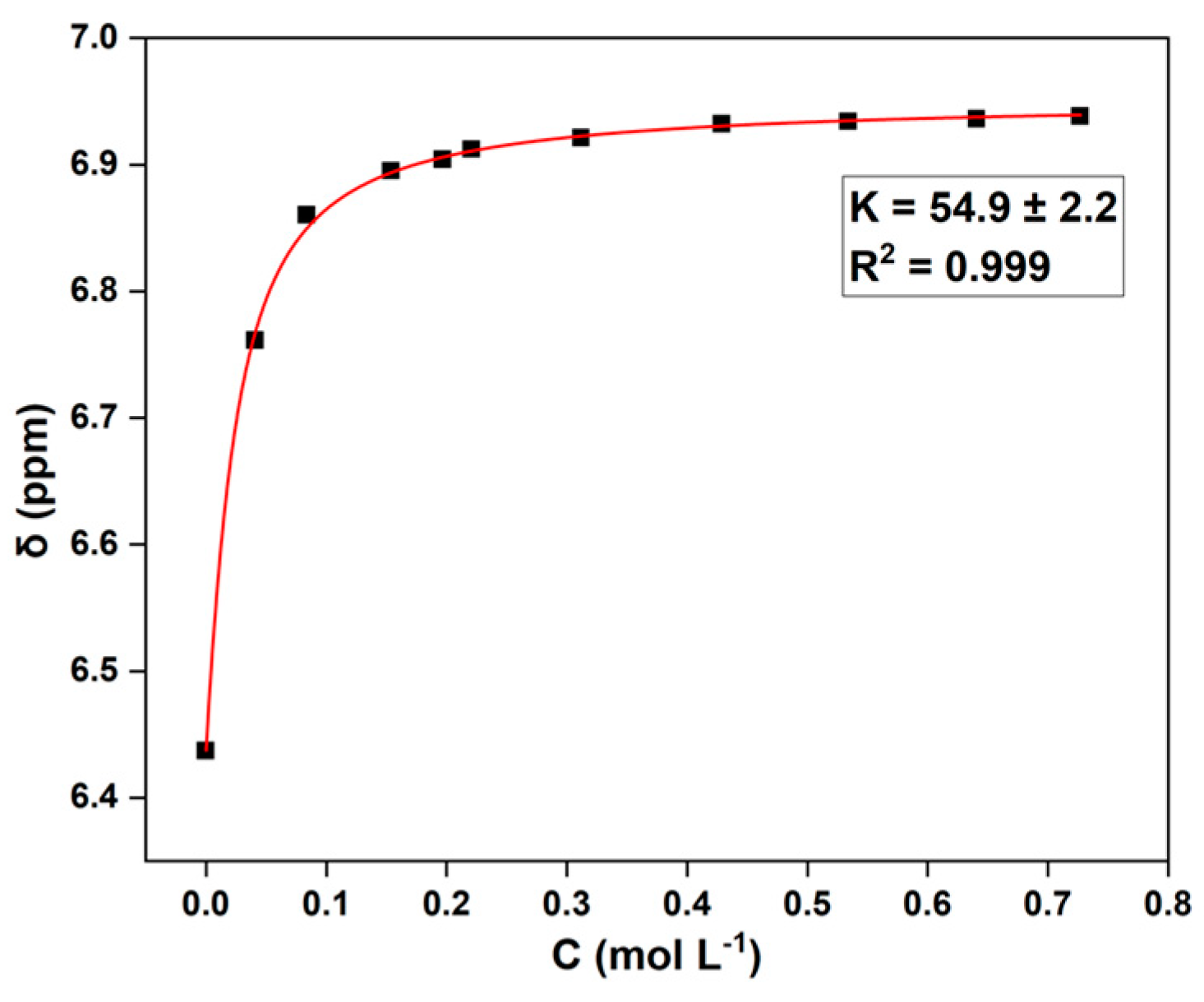
3.3. The Determination of the Binding Constant
3.4. Product Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Potter, H. V. Plastics and some recent applications. Nature 1939, 143, 785-787. [CrossRef]
- Bergmann, M.; Collard, F.; Fabres, J.; Gabrielsen, G. W.; Provencher, J. F.; Rochman, C. M.; Sebille, E.; Tekman, M. B. Plastic pollution in the arctic. Nat. Rev. Earth Environ. 2022, 3, 323-337. [CrossRef]
- Fan, Y. V.; Jiang, P.; Tan, R. R.; Aviso, K.B.; You, F.; Zhao, X.; Lee, C. T.; Klemeš, J. J. Forecasting plastic waste generation and interventions for environmental hazard mitigation. J. Hazard. Mater. 2022, 424, 127330. [CrossRef]
- Zhao, X.; You, F. Life cycle assessment of microplastics reveals their greater environmental hazards than mismanaged polymer waste losses. Environ. Sci. Technol. 2022, 56, 11780-11797. [CrossRef]
- Zhao, X.; Korey, M.; Li, K.; Copenhaver, K.; Tekinalp, H.; Celik, S.; Kalaitzidou, K.; Ruan, R.; Ragauskas, A. J.; Ozcan, S. Plastic waste upcycling toward a circular economy. Chem. Eng. J. 2022, 428, 131928. [CrossRef]
- Garcia, J. M.; Robertson, M. L. The future of plastics recycling. Science 2017, 358, 870-872. [CrossRef]
- Ignatyev, I. A.; Thielemans, W.; Vander Beke, B. Recycling of polymers: a review. ChemSusChem 2014, 7, 1579-1593. [CrossRef]
- Vollmer, I.; Jenks, M. J. F.; Roelands, M. C. P.; White, R. J.; Harmelen, T.; Wild, P.; Laan, G. P.; Meirer, F.; Keurentjes, J. T. F.; Weckhuysen, B. M. Beyond mechanical recycling: giving new life to plastic waste. Angew. Chem. Int. Ed. 2020, 59, 15402-15423. [CrossRef]
- Badia, J. D.; Ribes-Greus, A. Mechanical recycling of polylactide, upgrading trends and combination of valorization techniques. Eur. Polym. J. 2016, 84, 22-39. [CrossRef]
- Petrus, R.; Bykowski, D.; Sobota, P. Solvothermal alcoholysis routes for recycling polylactide waste as lactic acid esters. ACS Catal. 2016, 6, 5222-5235. [CrossRef]
- Xu, G.; Wang, Q. Chemically recyclable polymer materials: polymerization and depolymerization cycles. Green Chem. 2022, 24, 2321-2346. [CrossRef]
- McKeown, P.; Kamran, M.; Davidson, M. G.; Jones, M. D.; Román-Ramírez, L. A.; Wood, J. Organocatalysis for versatile polymer degradation. Green Chem. 2020, 22, 3721-3726. [CrossRef]
- Chen, X.; Wang, Y.; Zhang, L. Recent progress in the chemical upcycling of plastic wastes. ChemSusChem 2021, 14, 4137-4151. [CrossRef]
- Chen, H.; Wan, K.; Zhang, Y.; Wang, Y. Waste to wealth: chemical recycling and chemical upcycling of waste plastics for a great future. ChemSusChem 2021, 14, 4123-4136. [CrossRef]
- Zhang, S.; Hu, Q.; Zhang, Y.-X.; Guo, H.; Wu, Y.; Sun, M.; Zhu, X.; Zhang, J.; Gong, S.; Liu, P.; et al. Depolymerization of polyesters by a binuclear catalyst for plastic recycling. Nat. Sustain. 2023, 6, 965-973. [CrossRef]
- Cederholm, L.; Wohlert, J.; Olsén, P.; Hakkarainen, M.; Odelius, K. "Like recycles like”: selective ring-closing depolymerization of poly(L-lactic acid) to L-lactide. Angew Chem. Int. Ed. 2022, 61, e202204531. [CrossRef]
- An, W.; Liu, X.; Li, J.; Zhao, X.; Long, Y.; Xu, S.; Wang, Y.-Z. Water-solvent regulation on complete hydrolysis of thermosetting polyester and complete separation of degradation products. J. Hazard. Mater. 2023, 453, 131423. [CrossRef]
- Höcker, H.; Keul, H. Ring-opening polymerization and ring-closing depolymerization. Adv. Mater. 1994, 6, 21-36. [CrossRef]
- Endo, T.; Nagai, D. A novel construction of ring-opening polymerization and chemical recycling system. Macromol. Symp. 2005, 226, 79-86. [CrossRef]
- Olsén, P.; Odelius, K.; Albertsson, A.-C. Thermodynamic presynthetic considerations for ring-opening polymerization. Biomacromolecules 2016, 17, 699-709. [CrossRef]
- Olsén, P.; Undin, J.; Odelius, K.; Keul, H.; Albertsson, A.-C. Switching from controlled ring-opening polymerization (cROP) to controlled ring-closing depolymerization (cRCDP) by adjusting the reaction parameters that determine the ceiling temperature. Biomacromolecules 2016, 17, 3995-4002. [CrossRef]
- Xiong, W.; Chang, W.; Shi, D.; Yang, L.; Tian, Z.; Wang, H.; Zhang, Z.; Zhou, X.; Chen, E.-Q; Lu, H. Geminal dimethyl substitution enables controlled polymerization of penicillamine-derived β-thiolactones and reversed depolymerization. Chem 2020, 6, 1831-1843. [CrossRef]
- Yang, R.; Xu, G.; Dong, B.; Hou, H.; Wang, Q. A “polymer to polymer” chemical recycling of PLA plastics by the “DE–RE polymerization” strategy. Macromolecules 2022, 55, 1726-1735. [CrossRef]
- Zhou, X.; Liu, Q.; Xu, G.; Yang, R.; Sun, H.; Wang, Q. Chemical upcycling of poly(lactide) plastic waste to lactate ester, lactide and new poly(lactide) under Mg-catalysis condition. Chin. Chem. Lett. 2023, 34, 108158. [CrossRef]
- Yang, R.; Xu, G.; Dong, B.; Guo, X.; Wang, Q. Selective, sequential, and “one-pot” depolymerization strategies for chemical recycling of commercial plastics and mixed plastics. ACS Sustainable Chem. Eng. 2022, 10, 9860-9871. [CrossRef]
- Zhou, X.; Chai, M.; Xu, G.; Yang, R.; Sun, H.; Wang, Q. Catalyst-free amino-alcoholysis depolymerization strategy: a facile and powerful tool for chemical recycling of poly(bisphenol A carbonate). Green Chem. 2023, 5, 952-959. [CrossRef]
- Monaco, M. R.; Prévost, S.; List, B. Catalytic asymmetric synthesis of thiols. J. Am. Chem. Soc. 2014, 136, 16982-16985. [CrossRef]
- Monaco, M. R.; Poladura, B.; Diaz de Los Bernardos, M.; Leutzsch, M.; Goddard, R.; List, B. Activation of carboxylic acids in asymmetric organocatalysis. Angew. Chem. Int Ed. 2014, 53, 7063-7067. [CrossRef]
- Luo, Y.; Ma, H.; Sun, Y.; Che, P.; Nie, X.; Wang, T.; Xu, J. Understanding and measurement for the binding energy of hydrogen bonds of biomass-derived hydroxyl compounds. J. Phys. Chem. A 2018, 122, 843-848. [CrossRef]
- Luo, Y.; Ma, H.; Zhang, S.; Zheng, D.; Che, P.; Liu, X.; Zhang, M.; Gao, J.; Xu, J. Binding energy as driving force for controllable reconstruction of hydrogen bonds with molecular scissors. J. Am. Chem. Soc. 2020, 142, 6085-6092. [CrossRef]
- Scheiner, S. Assessment of the presence and strength of H-Bonds by means of corrected NMR. Molecules 2016, 21, 1426. [CrossRef]
- Hata, S.; Goto, H.; Yamada, E.; Oku, A. Chemical conversion of poly(carbonate) to 1,3-dimethyl-2-imidazolidinone (DMI) and bisphenol A: a practical approach to the chemical recycling of plastic wastes, Polymer 2002, 43, 2109-2116. [CrossRef]
- Yang, J.; Li, Y.; Wang, J.; Sun, X.; Shah, S. M.; Cao, R.; Chen, J. Novel sponge-like molecularly imprinted mesoporous silica material for selective isolation of bisphenol A and its analogues from sediment extracts. Anal. Chim. Acta 2015, 853, 311-319. [CrossRef]
- Mizuno, T.; Nakai, T.; Mihara, M. Efficient solvent-free synthesis of urea derivatives using selenium-catalyzed carbonylation of amines with carbon monoxide and oxygen. Synthesis 2010, 2010, 4251-4255. [CrossRef]
| Entry | Solvent | Yield of BPA (%) | Yield of DMI (%) |
|---|---|---|---|
| 1 | DCM | 77 | 79 |
| 2 | THF | 68 | 66 |
| 3 | EtOAc | 75 | 75 |
| 4 | Tol. | 72 | 71 |
| 5 | Ace. | 70 | 68 |
| 6 | DMI | 87 | 85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




