Submitted:
19 October 2023
Posted:
23 October 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Research Objects. Outbred White Mice Weighing 18-20 g Were Used as a Biological Model in the Experiments. The Formed Strain of Esherichia coli «PL-6» No. 1154115 was Used as Microorganisms
2.2. Obtaining an Immunizing Agent - a Protein-Quinoid Complex (conjugated) Antigen Used as an Inducer of Immune (Therapeutic) Antiradiation Serum
2.3. Obtaining hyperimmune anti-radiating polyclonal antibodies. As an inducer of the synthesis of polyclonal antibodies with bifunctional properties, the conjugated complex protein-quinoid radiotoxin (BCRA) obtained by the above technology was used.
2.4. Obtaining metabolic products of Bifidobacterium bifidum and Bacillus subtilis and a highly dispersed fraction of bentonite.
2.4.1. Setting up a Bentonite Flow Reaction (RBF) for the Indication of Radiotoxins
2.5. Construction of a Composite Medicinal Preparation Based on Polyclonal Antibodies (PCA) of the Apiphyto-Preparation «Vita-Forci» (ACE), Bifidobacterium Bifidummetabolic Products (PM B.b) and Bacillus Subtilis (PM Bs) and a Highly Dispersed Fraction of Bentonite (VDFB).
2.6. Simulation of Radiation Damage
3. Results of Studies
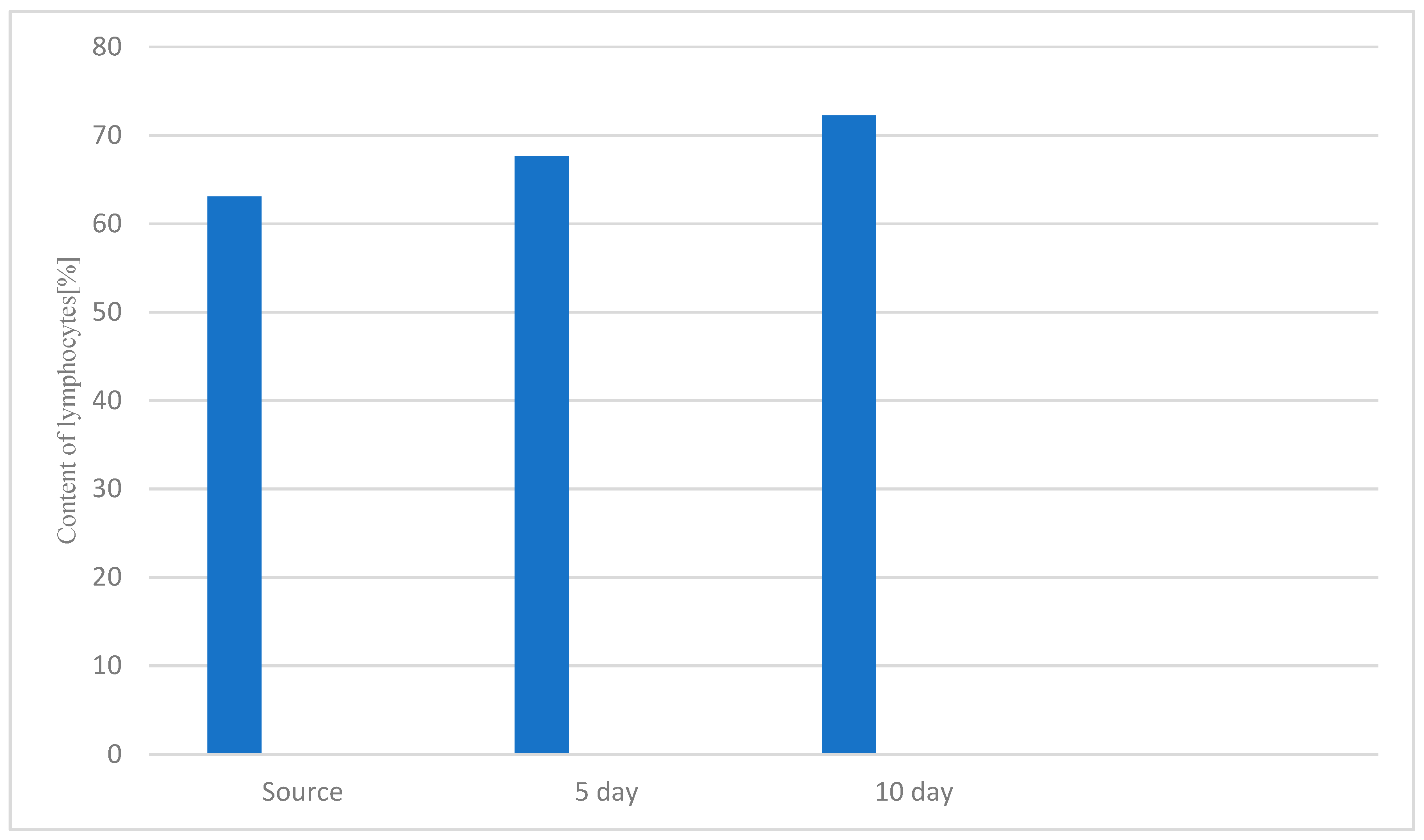
3.1. Study of the Effect of the Drug «Polyapisogen» on the System of Immunohemopoiesis in Health and Disease
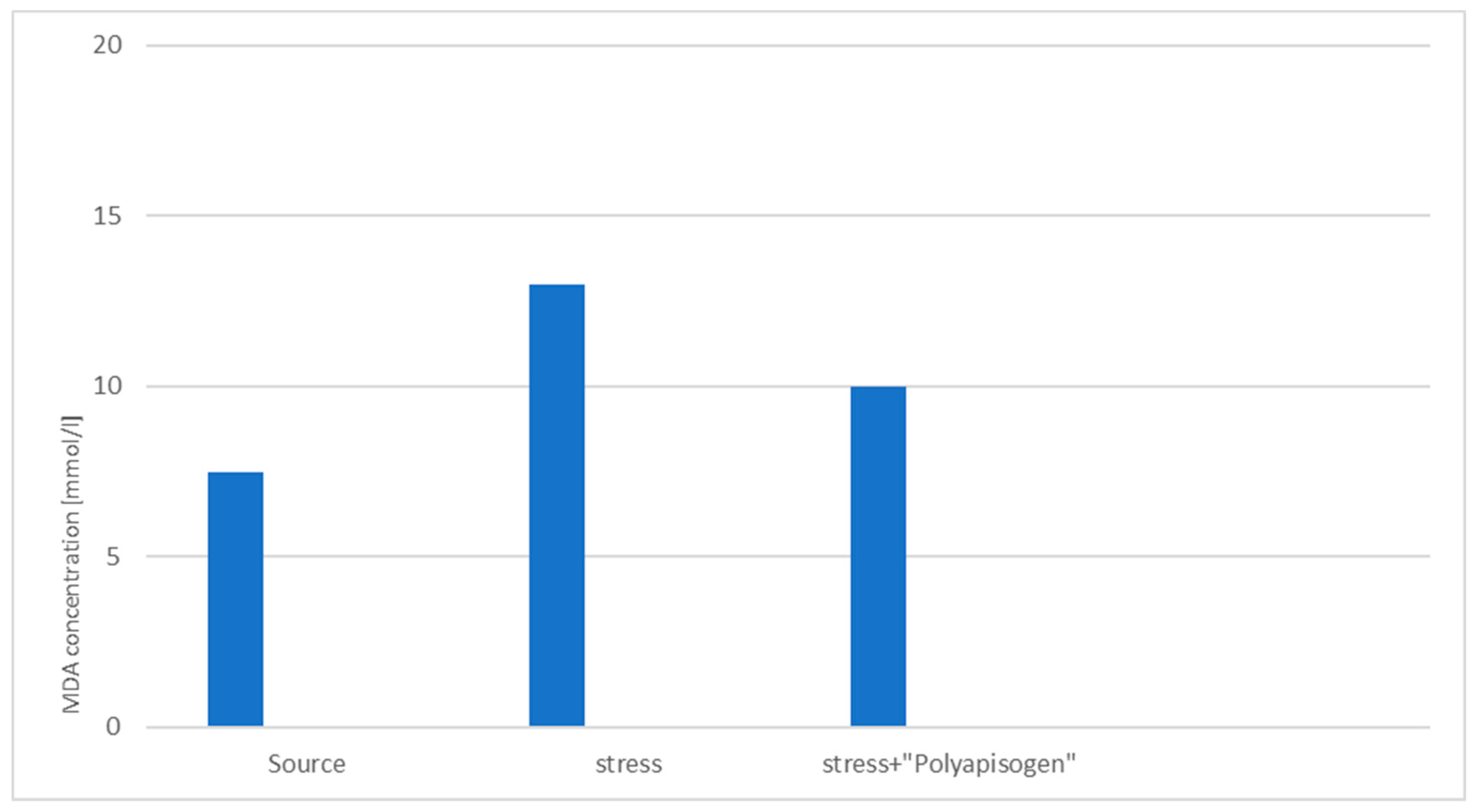
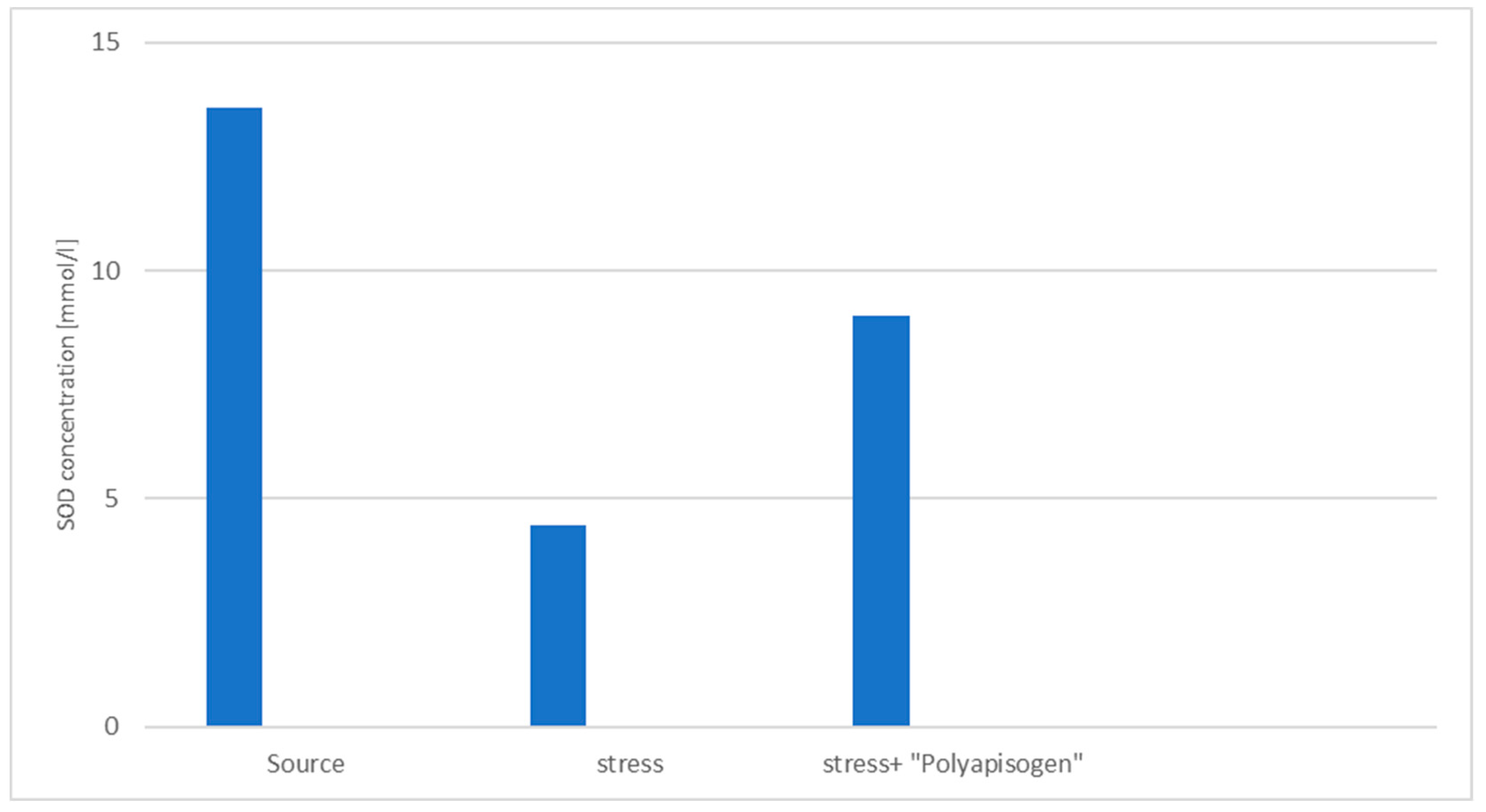
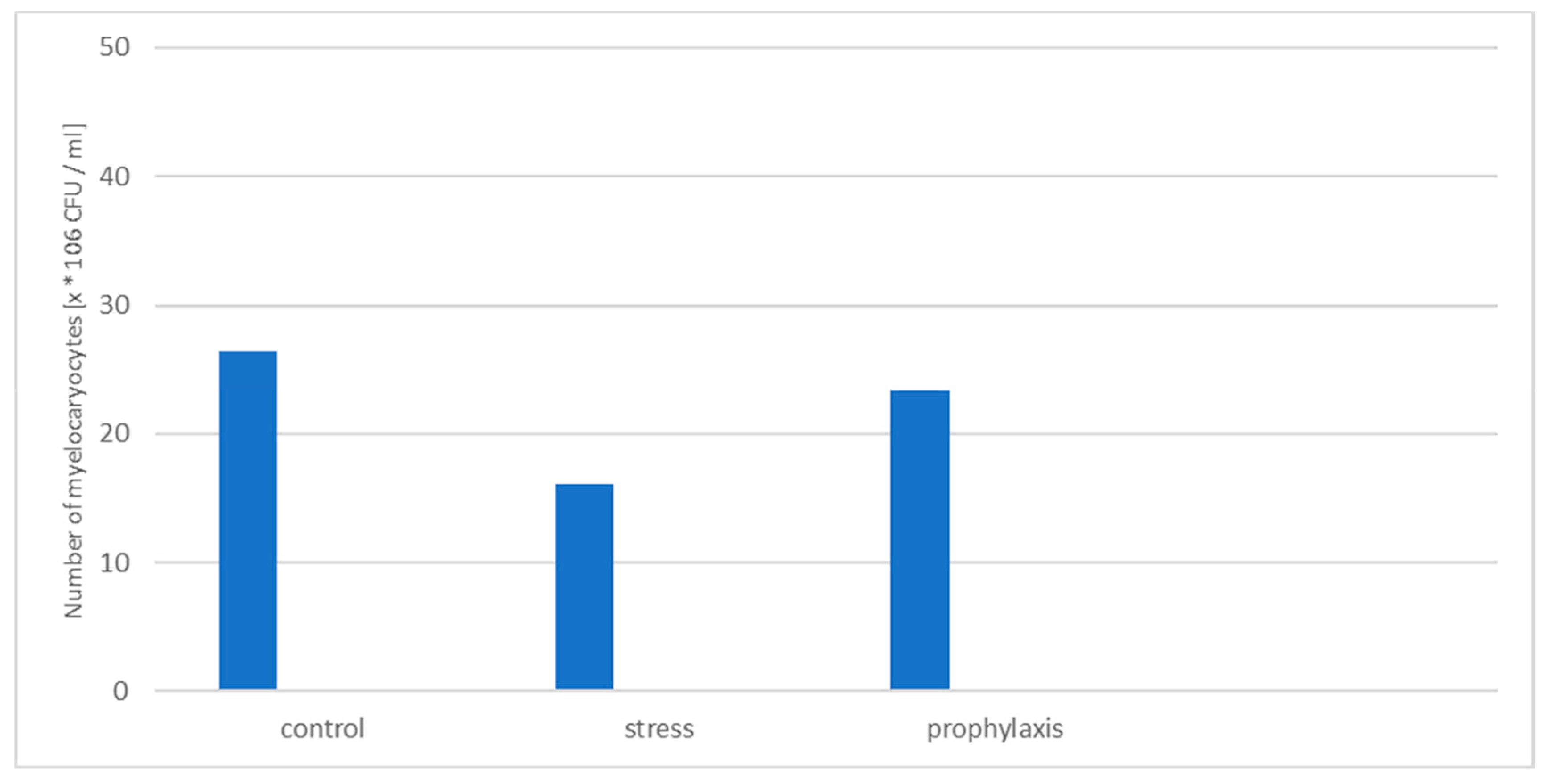
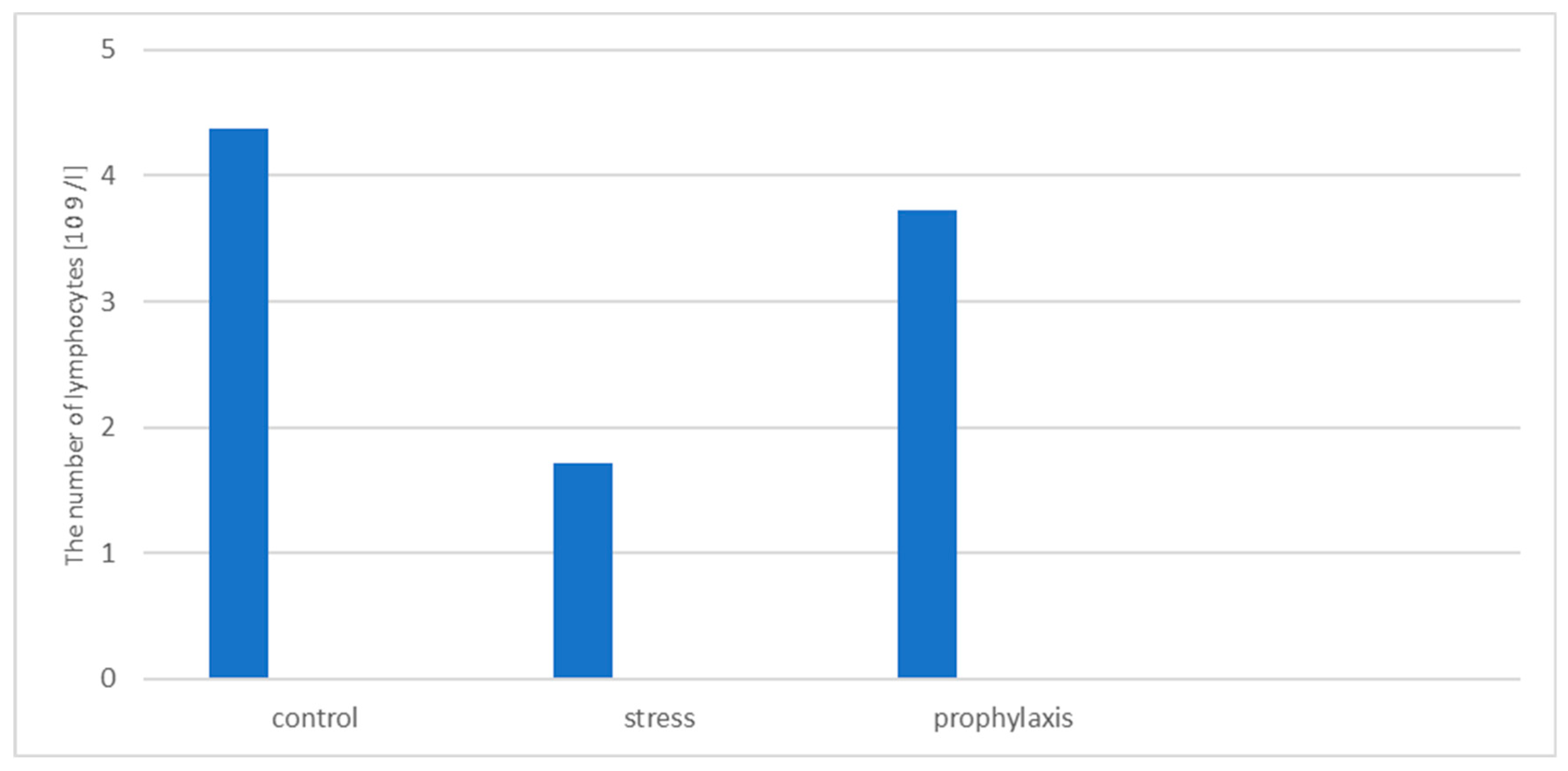
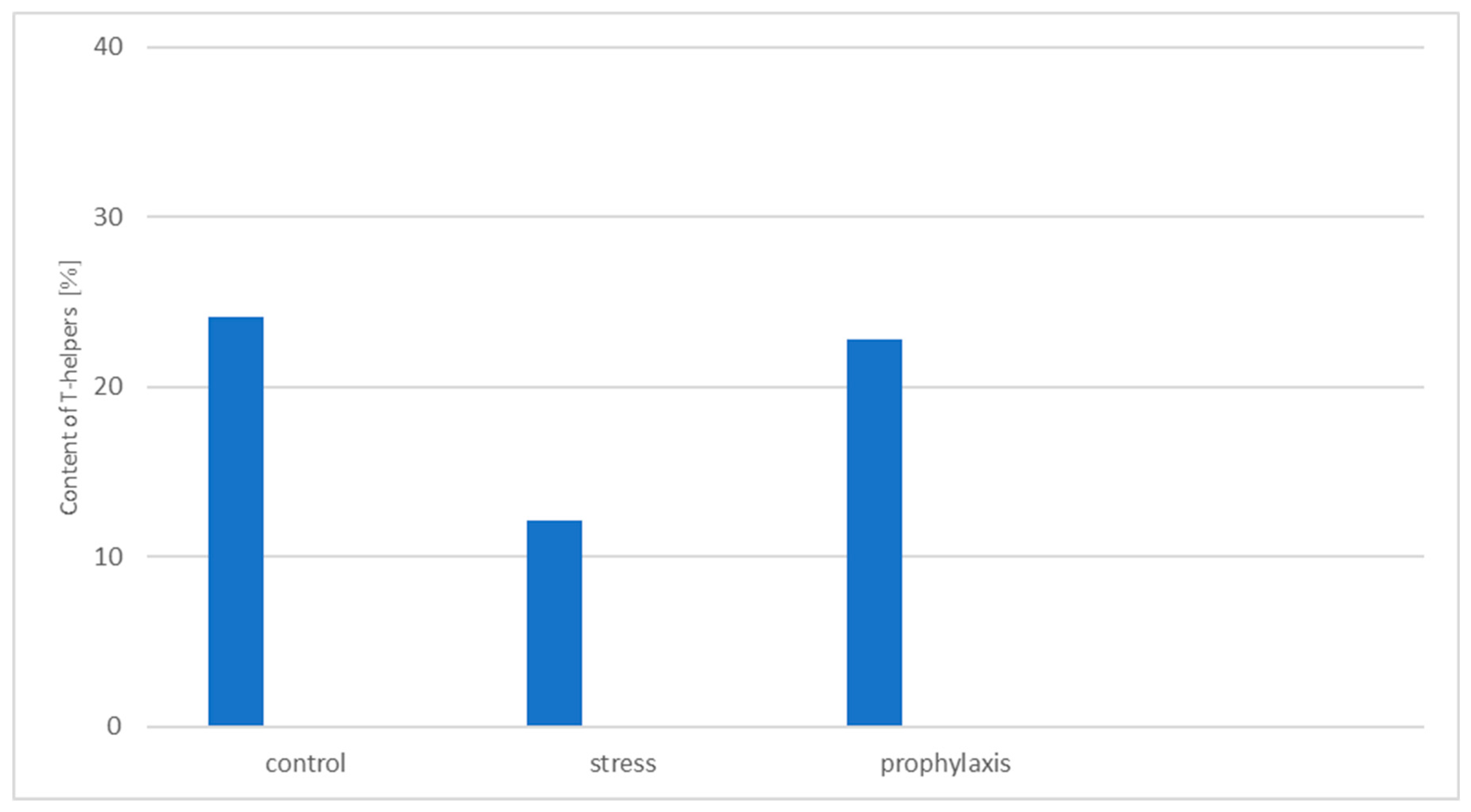
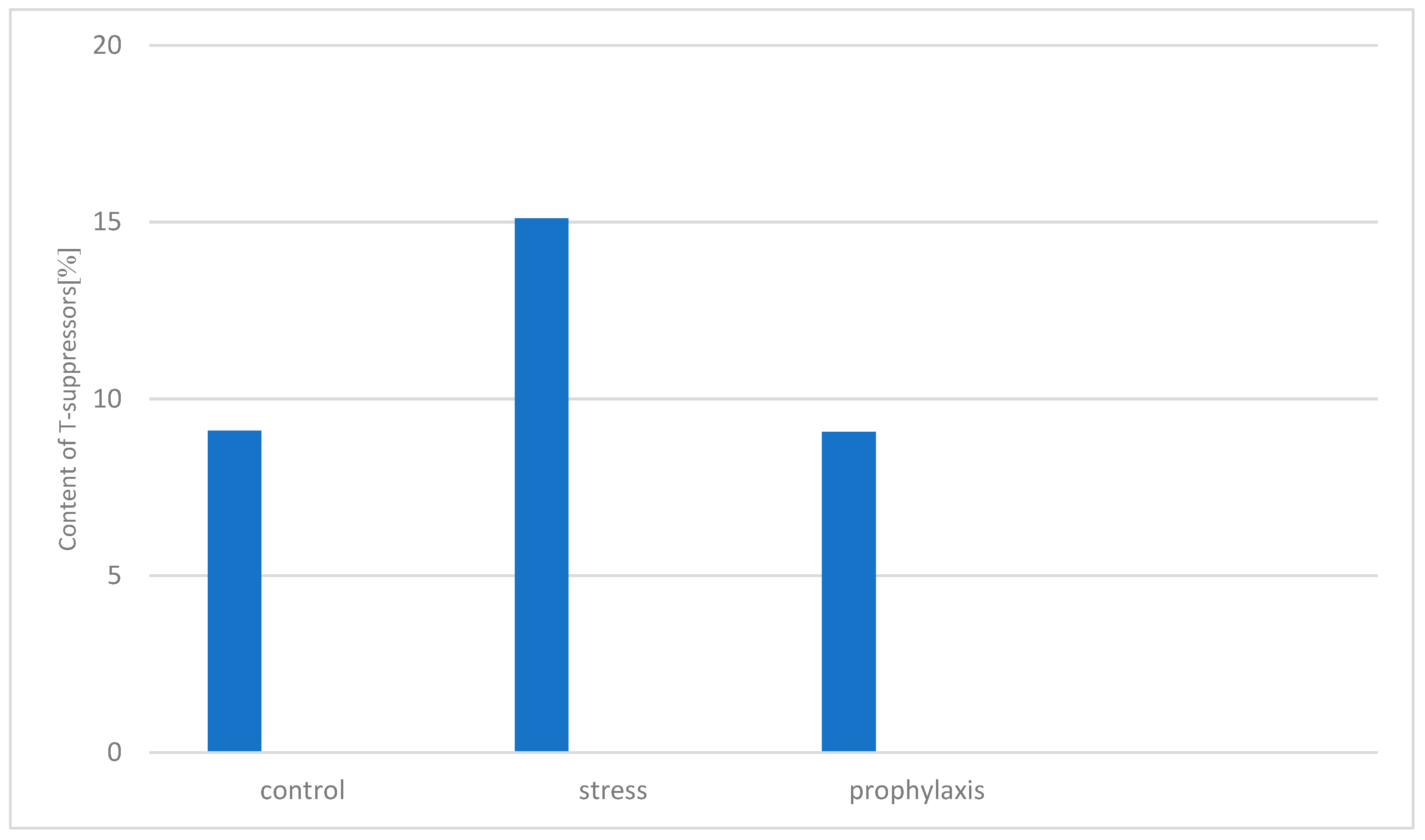
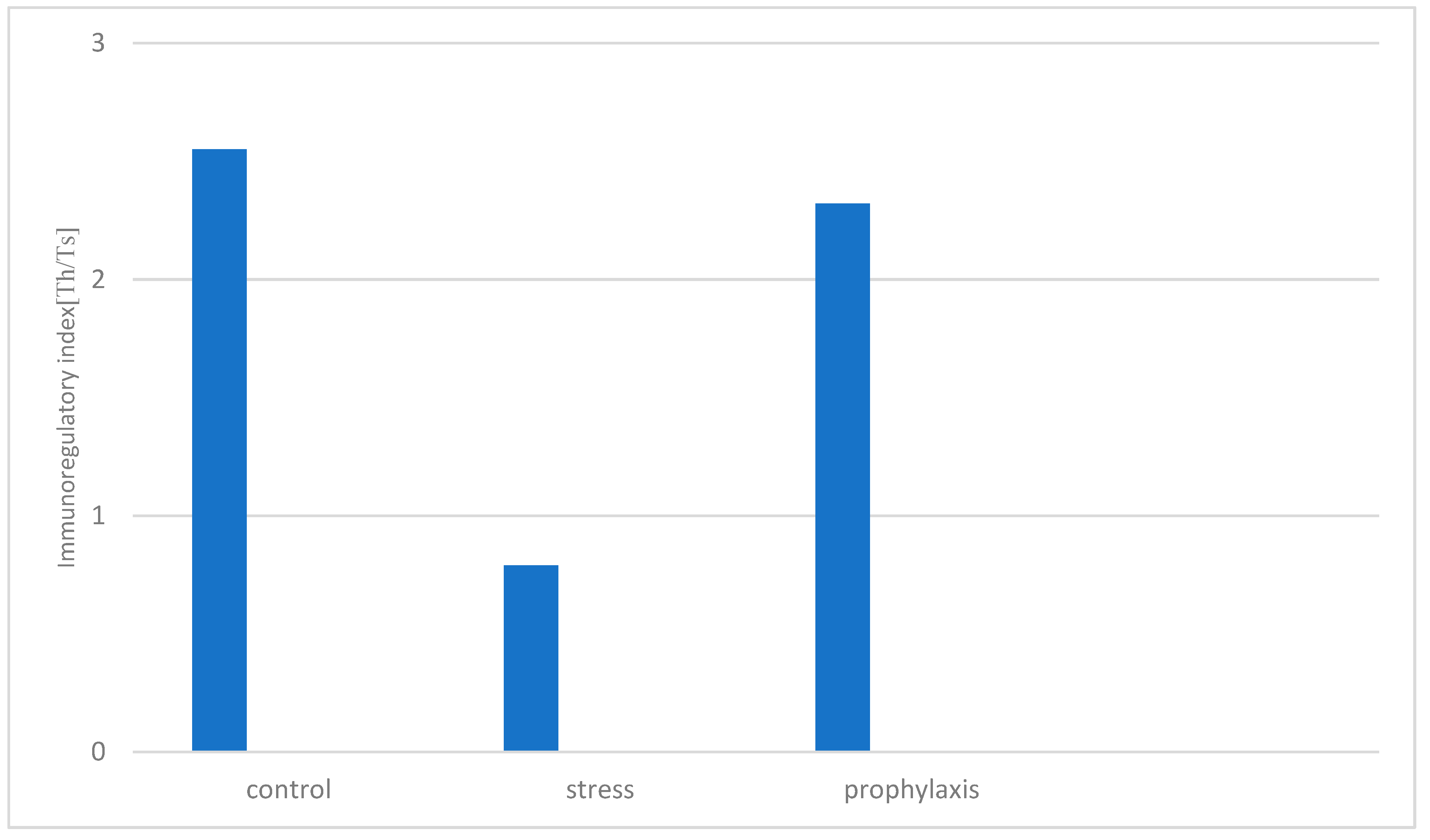
3.2. Study of the Stress-Protective Effect of the Drug «Polyapisogen»
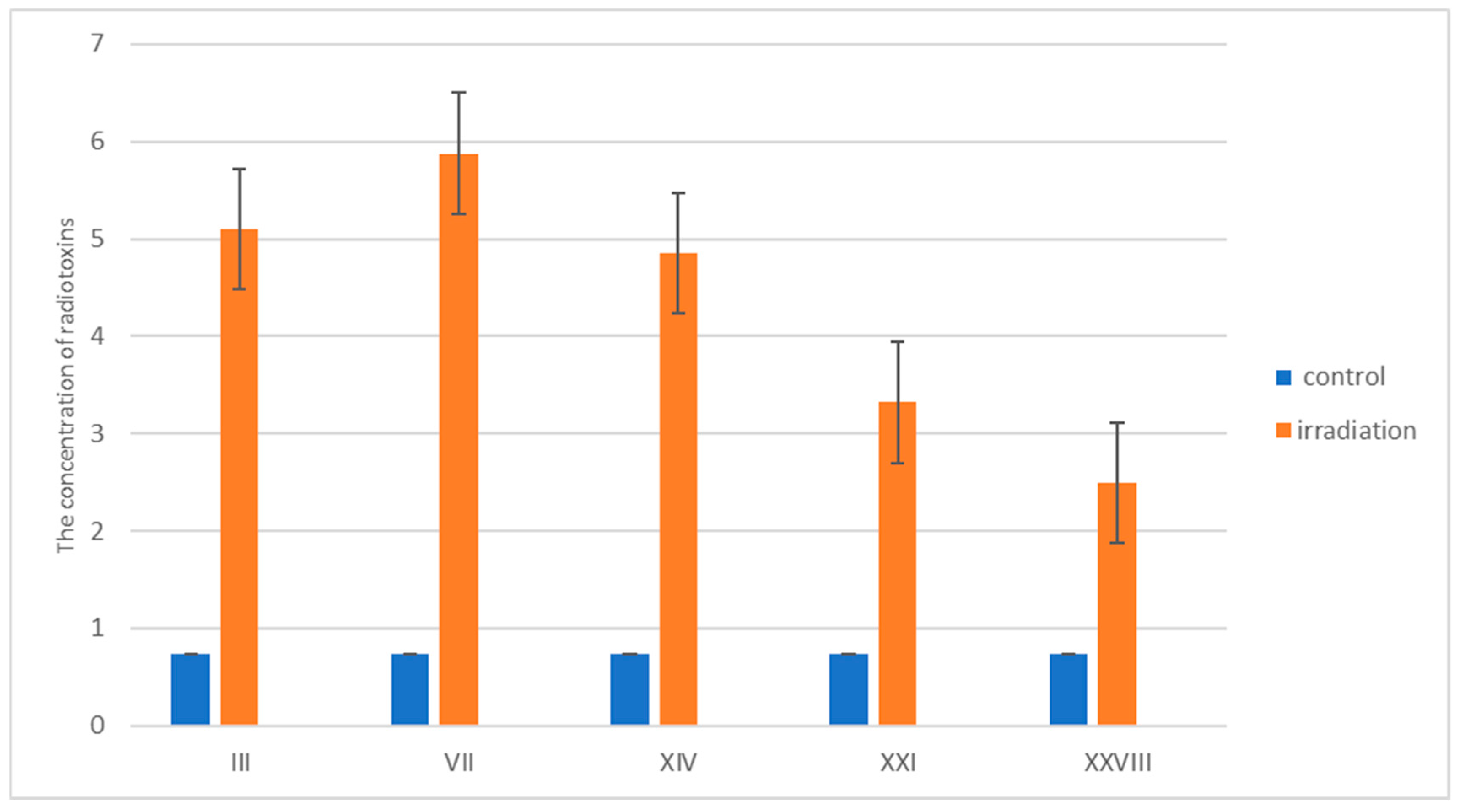
3.3. Study of the Immunotherapeutic Activity of the Drug in Radiation Pathology
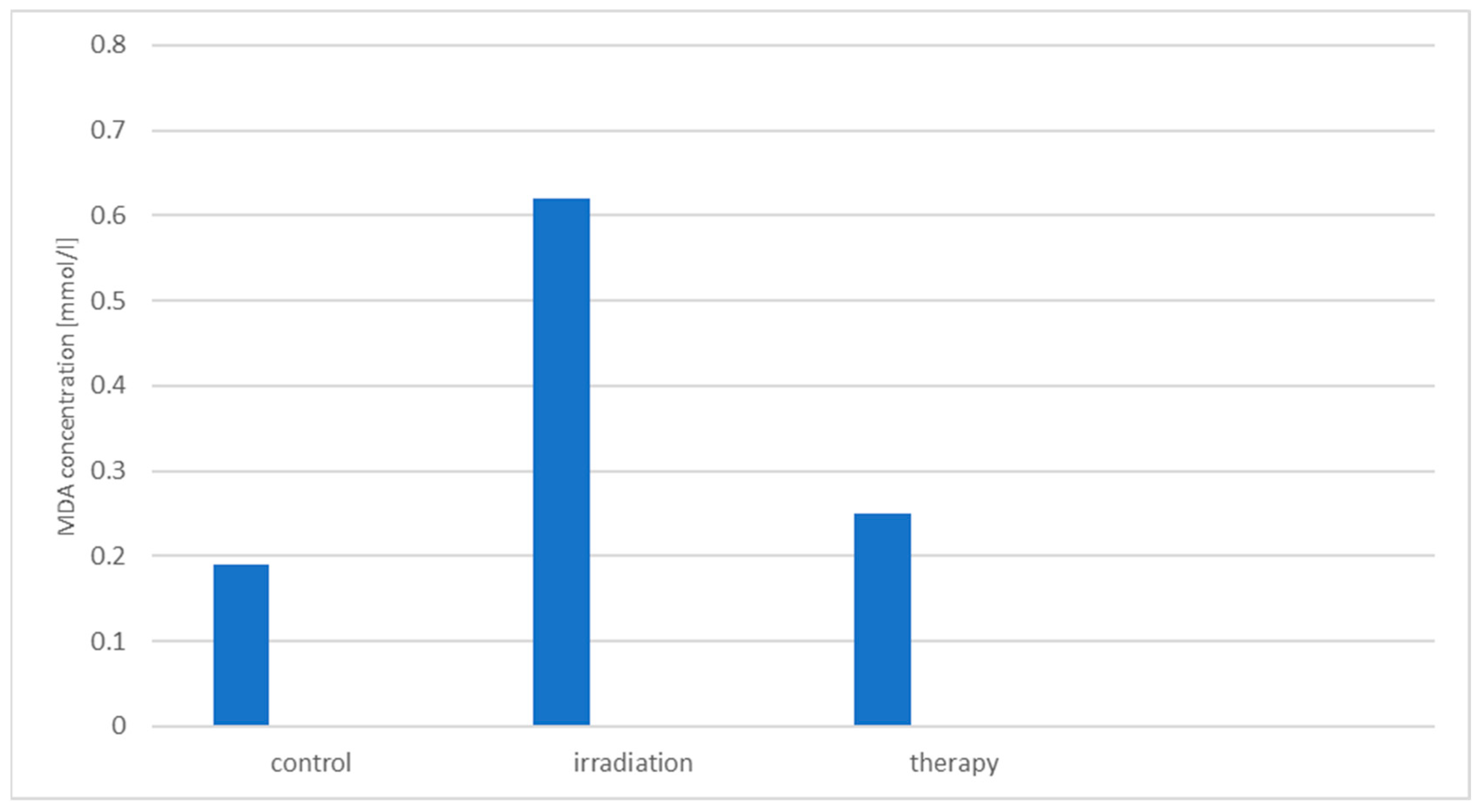
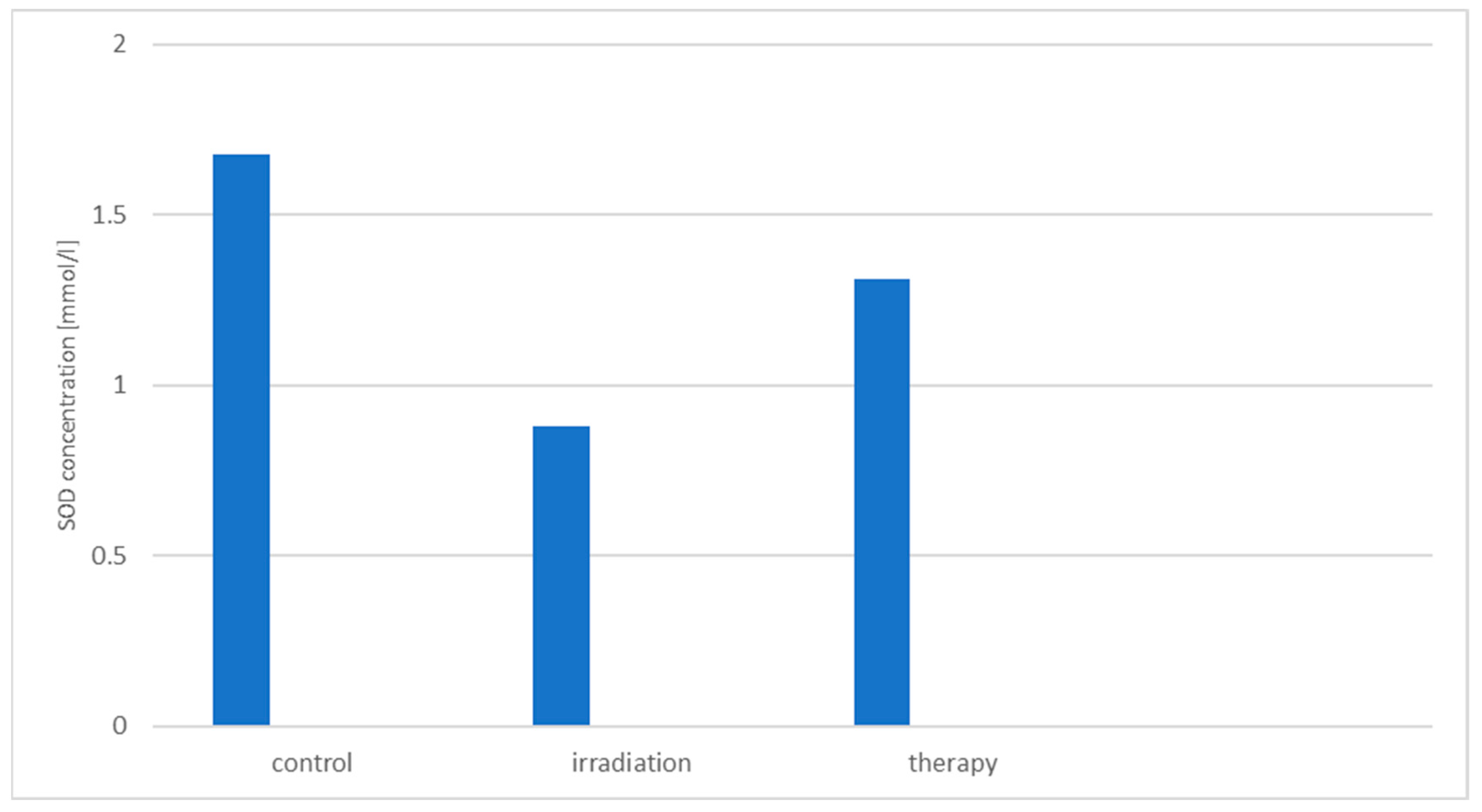
3.4. Study of the Hemoprotective Effect of the Drug «Polyapisogen»
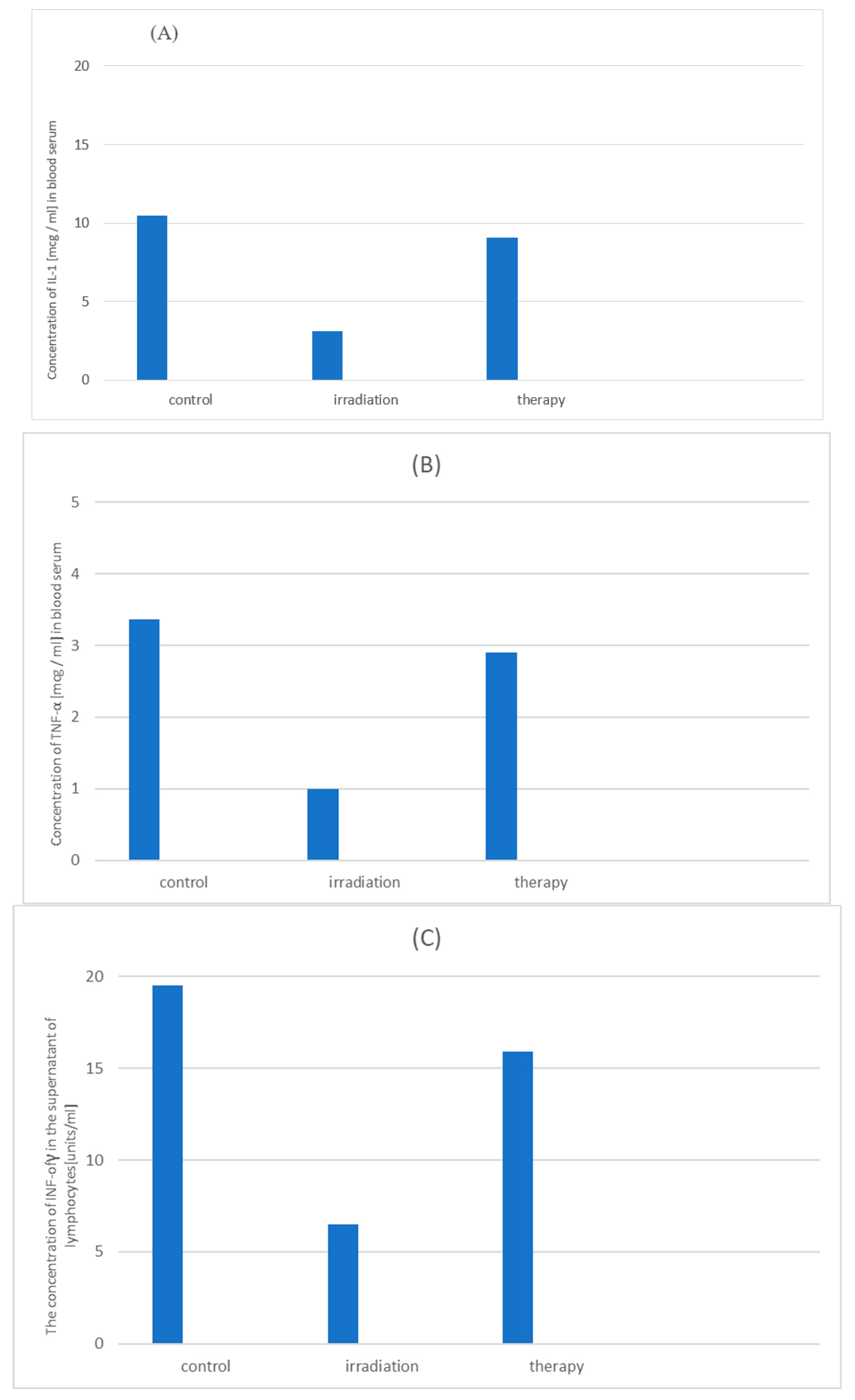
3.5. The Influence of the Drug «Polyapisogen» on Post-Radiation Cytokinesis
Discussion
References
- Hussain, S.M.; Adnan, M.; Rasul, A.; Shah, M.A.; Hussain, G.; Asrar, M.; Riaz, A.; Sarfraz, I.; Hussain, A.; Khorsandi, K.; et al. Radioprotective Role of Natural Polyphenols: From Sources to Mechanisms. Anti-Cancer Agents Med. Chem. 2021, 22, 30–39. [Google Scholar] [CrossRef]
- Akita, S. Treatment of Radiation Injury. Adv Wound Care (New Rochelle) 2014, 3, 1–11. [Google Scholar] [CrossRef]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.-Q. Heavy Metals and Pesticides Toxicity in Agricultural Soil and Plants: Ecological Risks and Human Health Implications. Toxics 2021, 9, 42. [Google Scholar] [CrossRef]
- Azad, M.A.K.; Sarker, M.; Wan, D. Immunomodulatory Effects of Probiotics on Cytokine Profiles. Biomed Res Int. 2018, 2018, 8063647. [Google Scholar] [CrossRef]
- Basak, S.; Gokhale, J. Immunity boosting nutraceuticals: Current trends and challenges. J. Food Biochem. 2021, 46, e13902. [Google Scholar] [CrossRef]
- Blake, S.J.; James, J.; Ryan, F.J.; Caparros-Martin, J.; Eden, G.L.; Tee, Y.C.; Salamon, J.R.; Benson, S.C.; Tumes, D.J.; Sribnaia, A.; et al. The immunotoxicity, but not anti-tumor efficacy, of anti-CD40 and anti-CD137 immunotherapies is dependent on the gut microbiota. Cell Rep. Med. 2021, 2, 100464. [Google Scholar] [CrossRef] [PubMed]
- Bou Zerdan, M.; Moussa, S.; Atoui, A.; Assi, H.I. Mechanisms of Immunotoxicity: Stressors and Evaluators. Int J Mol Sci. 2021, 22, 8242. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C. Radioprotective Potential of Plants and Herbs against the Effects of Ionizing Radiation. J Clin Biochem Nutr. 2007, 40, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Cabassi, E. The Immune System and Exposure to Xenobiotics in Animals. Veter- Res. Commun. 2007, 31, 115–120. [Google Scholar] [CrossRef]
- Carpena, N.; Richards, K.; Gonzalez, T.D.J.B.; Bravo-Blas, A.; Housden, N.G.; Gerasimidis, K.; Milling, S.W.F.; Douce, G.; Malik, D.J.; Walker, D. Targeted Delivery of Narrow-Spectrum Protein Antibiotics to the Lower Gastrointestinal Tract in a Murine Model of Escherichia coli Colonization. Front. Microbiol. 2021, 12. [Google Scholar] [CrossRef]
- Chiang, C.S.; Liu, W.C.; Jung, S.M.; Chen, F.H.; Wu, C.R.; McBride, W.H.; Lee, C.C.; Hong, J.H. Compartmental responses after thoracic irradiation of mice: strain differences. Int J Radiat Oncol Biol Phys. 2005, 62, 862–871. [Google Scholar] [CrossRef]
- Cox, M.A.; Kahan, S.M.; Zajac, A.J. Anti-viral CD8 T cells and the cytokines that they love. Virology 2013, 435, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Curtsinger, J.M.; Mescher, M.F. Inflammatory cytokines as a third signal for T cell activation. Curr Opin Immunol. 2010, 22, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Dainiak, N. Medical management of acute radiation syndrome and associated infections in a high-casualty incident. J. Radiat. Res. 2018, 59, ii54–ii64. [Google Scholar] [CrossRef]
- Delia, P.; Sansotta, G.; Donato, V.; Frosina, P.; Messina, G.; De Renzis, C.; Famularo, G. Use of probiotics for prevention of radiation-induced diarrhea. World J Gastroenterol. 2007, 13, 912–915. [Google Scholar] [CrossRef]
- Fessas, P.; Possamai, L.A.; Clark, J.; Daniels, E.; Gudd, C.; Mullish, B.H.; Alexander, J.L.; Pinato, D.J. Immunotoxicity from checkpoint inhibitor therapy: clinical features and underlying mechanisms. Immunology 2019, 159, 167–177. [Google Scholar] [CrossRef]
- Fischer, N.; Seo, E.-J.; Efferth, T. Prevention from radiation damage by natural products. Phytomedicine 2017, 47, 192–200. [Google Scholar] [CrossRef]
- Galbiati, V.; Buoso, E.; Bianca, R.D.d.V.; Di Paola, R.; Morroni, F.; Nocentini, G.; Racchi, M.; Viviani, B.; Corsini, E. Immune and Nervous Systems Interaction in Endocrine Disruptors Toxicity: The Case of Atrazine. Front. Toxicol. 2021, 3, 649024. [Google Scholar] [CrossRef] [PubMed]
- Higashi, T.; Takei, H.; Sando, T. Cytosoe catalase: comparison with peroxisomal catalase. Cell. Struct. Funet. 1983, 8, 480. [Google Scholar]
- Hong, J.H.; Jung, S.M.; Tsao, T.C.; Wu, C.J.; Lee, C.Y.; Chen, F.H.; Hsu, C.H.; McBride, W.H.; Chiang, C.S. Bronchoalveolar lavage and interstitial cells have different roles in radiation-induced lung injury. Int J Radiat Biol. 2003, 79, 159–167. [Google Scholar] [CrossRef]
- Khalid, W.; Arshad, M.S.; Ranjha, M.M.A.N.; Różańska, M.B.; Irfan, S.; Shafique, B.; Rahim, M.A.; Khalid, M.Z.; Abdi, G.; Kowalczewski, P. Functional constituents of plant-based foods boost immunity against acute and chronic disorders. Open Life Sci. 2022, 17, 1075–1093. [Google Scholar] [CrossRef]
- Khalil, M.L. Biological Activity of Bee Propolis in Health and Disease. Asian Pac J Cancer Prev. 2006, 7, 22–32. [Google Scholar]
- Lefkovits, I.; Waldman, H. Limiting Dulation Analysis of Cell in the Immune System; London-NewYork: Canbridge Univ. Prees, 1979; 548p.
- Lefkovits, I.; Waldman, H. Limiting Dulation Analysis of Cell in the Immune System; London-NewYork: Canbridge Univ. Prees, 1979; 548 p.
- Lierova, A.; Jelicova, M.; Nemcova, M.; Proksova, M.; Pejchal, J.; Zarybnicka, L.; Sinkorova, Z. Cytokines and radiation-induced pulmonary injuries. J. Radiat. Res. 2018, 59, 709–753. [Google Scholar] [CrossRef]
- López, M.; Martín, M. Medical management of the acute radiation syndrome. Rep. Pr. Oncol. Radiother. 2011, 16, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Mutinelli, F. Antioxidant Activity in Bee Products: A Review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Mazziotta, C.; Tognon, M.; Martini, F.; Torreggiani, E.; Rotondo, J.C. Probiotics Mechanism of Action on Immune Cells and Beneficial Effects on Human Health. Cells 2023, 12, 184. [Google Scholar] [CrossRef] [PubMed]
- Mazziotta, C.; Tognon, M.; Martini, F.; Torreggiani, E.; Rotondo, J.C. Probiotics Mechanism of Action on Immune Cells and Beneficial Effects on Human Health. Cells 2023, 12, 184. [Google Scholar] [CrossRef] [PubMed]
- Miglani, R.; Parveen, N.; Kumar, A.; Ansari, M.A.; Khanna, S.; Rawat, G.; Panda, A.K.; Bisht, S.S.; Upadhyay, J.; Ansari, M.N. Degradation of Xenobiotic Pollutants: An Environmentally Sustainable Approach. Metabolites 2022, 12, 818. [Google Scholar] [CrossRef]
- Miguel, J.A.; Tulipani, S.; Romandini, S.; Bertoli, E.; Battino, M. Contribution of honey in nutrition and human health:a review. Mediterr J Nutr Metab. 2010, 3, 15–23. [Google Scholar]
- Münstedt, K.; Männle, H. Using Bee Products for the Prevention and Treatment of Oral Mucositis Induced by Cancer Treatment. Molecules 2019, 24, 3023. [Google Scholar] [CrossRef]
- Nizamov, R.N.; Nasybullina, J.R.; Vagin, K.N.; Gainullin, R.R.; Vasilevsky, N.M.; Plotnikova, E.M. (20210 Conceptual foundations of the design of immunotherapeutic agents in multifactorial ecopathology. Kazan.
- Odum E.P. (1954) Fundamentals of Ecology. – Philadelphia – London. – 670p.
- Olaitan, P.B.; E Adeleke, O.; O Ola, I. Honey: a reservoir for microorganisms and an inhibitory agent for microbes. Afr. Heal. Sci. 2007, 7, 159–165. [Google Scholar]
- Patent RU no. 2169572 A61K 35/28 Method for treating radiation damage to the body and a method for obtaining a drug for the treatment of radiation damage to the body / V. M. Avilov, A. Z. Ravilov, V. A. Kirshin, R. N. Nizamov et al. - publ. 27.06.2001.
- Patent RU no. 2324361, A23K 1/100 Natural biologically active feed additive «Vita-Force» / A.V. Ivanov, R.N. Nizamov, Konyukhov G. V. et al. – Publ. 2008.
- Patent RU no. 2524612 C2 A61K 39/295 Method of treatment of radiation, chemical and/or biological damage to the body and a method for obtaining globulins for the treatment of radiation, chemical and /or biological damage to the body / A.V. Ivanov, R.N. Nizamov, G. V. Konyukhov and others – publ. 20.07.2014.
- Paulino, N.; Abreu, S.R.; Uto, Y.; Koyama, D.; Nagasawa, H.; Hori, H.; Dirsch, M.; Vollmar, A.; Scremin, A.; Bretz, W. Anti-inflammatory Effects of a Bioavailable Compound, Artepillin C, in Brazilian Propolis. Eur J Pharmacol. 2008, 587, 296–301. [Google Scholar] [CrossRef]
- Peterson, R.A. Regulatory T-Cells: Diverse Phenotypes Integral to Immune Homeostasis and Suppression. Toxicol. Pathol. 2012, 40, 186–204. [Google Scholar] [CrossRef]
- Rasheed, N. Inclination toward herbal remedies as immunity boosters and anti-stress agents during COVID-19 pandemic: A short report. Int J Health Sci (Qassim) 2021, 15, 1–3. [Google Scholar]
- Reeves, G. Overview of Use of G-CSF and GM-CSF in the Treatment of Acute Radiation Injury. Heal. Phys. 2014, 106, 699–703. [Google Scholar] [CrossRef]
- Schaue, D.; Kachikwu, E.L.; McBride, W.H. Cytokines in Radiobiological Responses: A Review. Radiat. Res. 2012, 178, 505–523. [Google Scholar] [CrossRef] [PubMed]
- Segner, H.; Bailey, C.; Tafalla, C.; Bo, J. Immunotoxicity of Xenobiotics in Fish: A Role for the Aryl Hydrocarbon Receptor (AhR)? Int J Mol Sci. 2021, 22, 9460. [Google Scholar] [CrossRef]
- Silverman, M.N.; Pearce, B.D.; Biron, C.A.; Miller, A.H. Immune Modulation of the Hypothalamic-Pituitary-Adrenal (HPA) Axis during Viral Infection. Viral Immunol. 2005, 18, 41–78. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Romaine, P.L.; Newman, V.L. Biologics as countermeasures for acute radiation syndrome: where are we now? Expert Opin Biol Ther. 2015, 15, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Toka, F.N.; Suvas, S.; Ronsi, B.T. CD4 + CD25 + T cells regulate vaccine-generated primoy and memory CD8 + T-cell responses against herpes simplex virus type. J. Virol. 2004, 78, 13082–13089. [Google Scholar] [CrossRef] [PubMed]
- Toma, C.L.; Serbescu, A.; Alexe, M.; Cervis, L.; Ionita, D.; Bogdan, M.A. The bronchoalveolar lavage pattern in radiation pneumonitis secondary to radiotherapy for breast cancer. Maedica 2010, 5, 250–257. [Google Scholar]
- Tyrinova, T.V.; Leplina, O.Y.; Mishinov, S.V.; Tikhonova, M.A.; Shevela, E.Y.; Stupak, V.V.; et al. Cytotovic activity ej ex-vivo generated IFN-α – induced monocytic-derived dendritic cells in brain glioma paticuts. Cell. Immunal. 2013, 284, 146–153. [Google Scholar] [CrossRef]
- Van den Brand, A.D.; Bokkers, B.G.H.; Te Biesebeek, J.D.; Mengelers, M.J.B. Combined Exposure to Multiple Mycotoxins: An Example of Using a Tiered Approach in a Mixture Risk Assessment. Toxins (Basel) 2022, 14, 303. [Google Scholar] [CrossRef]
- Veraldi, A.; Costantini, A.S.; Bolejack, V.; Miligi, L.; Vineis, P.; van Loveren, H. Immunotoxic effects of chemicals: A matrix for occupational and environmental epidemiological studies. Am. J. Ind. Med. 2006, 49, 1046–1055. [Google Scholar] [CrossRef]
- Wang, Z.; van der Fels-Klerx, H.J.; Lansink, A.G.J.M.O. Optimization of Sampling for Monitoring Chemicals in the Food Supply Chain Using a Risk-Based Approach: The Case of Aflatoxins and Dioxins in the Dutch Dairy Chain. Risk Anal. 2020, 40, 2539–2560. [Google Scholar] [CrossRef]
- Yadav, H.; Sharma, R.S.; Singh, R. Immunotoxicity of radiofrequency radiation. Environ. Pollut. 2022, 309, 119793. [Google Scholar] [CrossRef]
- Zhang, X.; Song, H.; Tang, X.; Wang, S.; Li, J.; Hao, Y. Research progress on radioprotective effects of bee products. Int J Radiat Biol. 2021, 97, 444–451. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, Y.; Li, Z.; Wu, H.; Zou, B.; Xu, Y. Exploring Natural Products as Radioprotective Agents for Cancer Therapy: Mechanisms, Challenges, and Opportunities. Cancers (Basel) 2023, 15, 3585. [Google Scholar] [CrossRef]
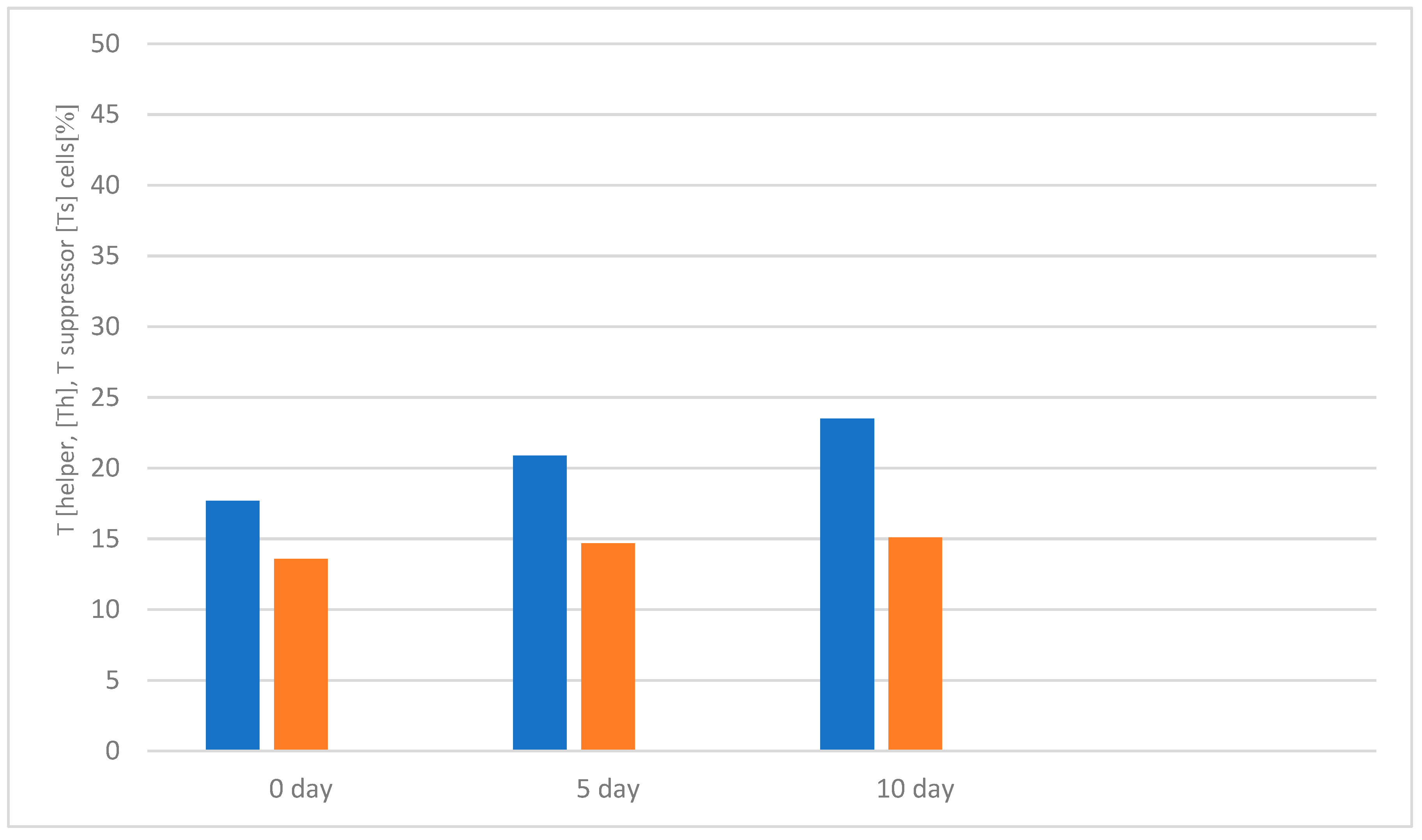
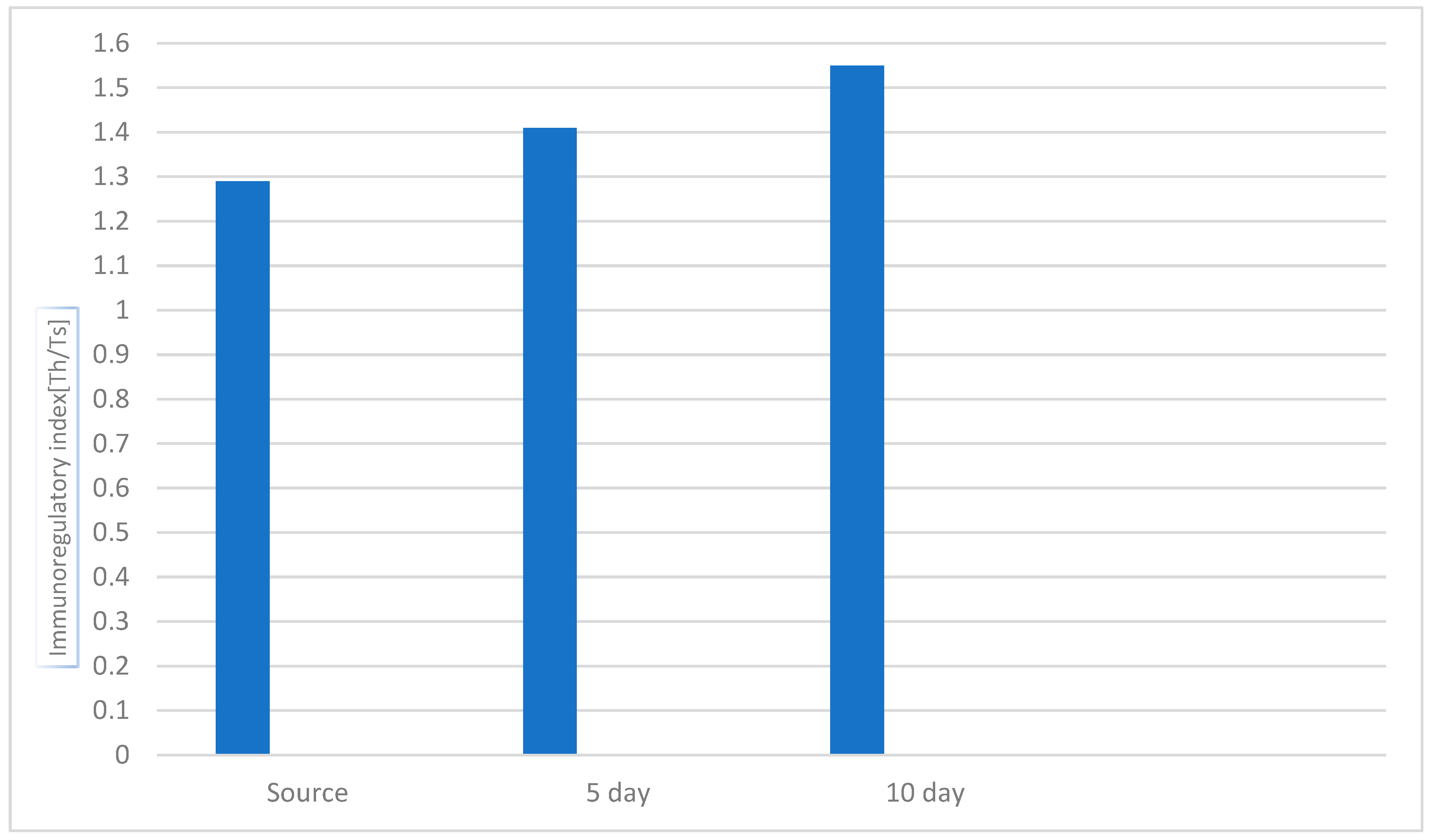
| The nature of destruction | Indicator | Groups of animals | ||
|---|---|---|---|---|
| Intacted | Controlled (stress+Н2О) | Practiced 1 (stress + «Polyapsinogen») | ||
| Striated hemorrhage | % damage to animals | 0 | 81,1 ± 1,5 | 38,3 ±0,9 |
| Average number of destruction per animal, % | 0 | 3,2 ± 0,2 | 1,1 ± 0,1 | |
| Erosion | % damage to animals | 0 | 100 | 51 ± 0,8 |
| Average number of destruction per animal, % | 0 | 1,6 ± 0,1 | 1,2±0,1 | |
| Striped ulcers | % damage to animals | 0 | 100 | 0 |
| Average number of destruction per animal, % | 0 | 1,5 ± 0,3 | 0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




