Submitted:
27 June 2023
Posted:
30 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Umbilical cord lining tissue samples
2.2. Isolation of mesenchymal stem cells
2.3. Transplantation replicates the number of umbilical cord lining mesenchymal stem cells
2.3.1. Evaluation of the ability of cells to create colony
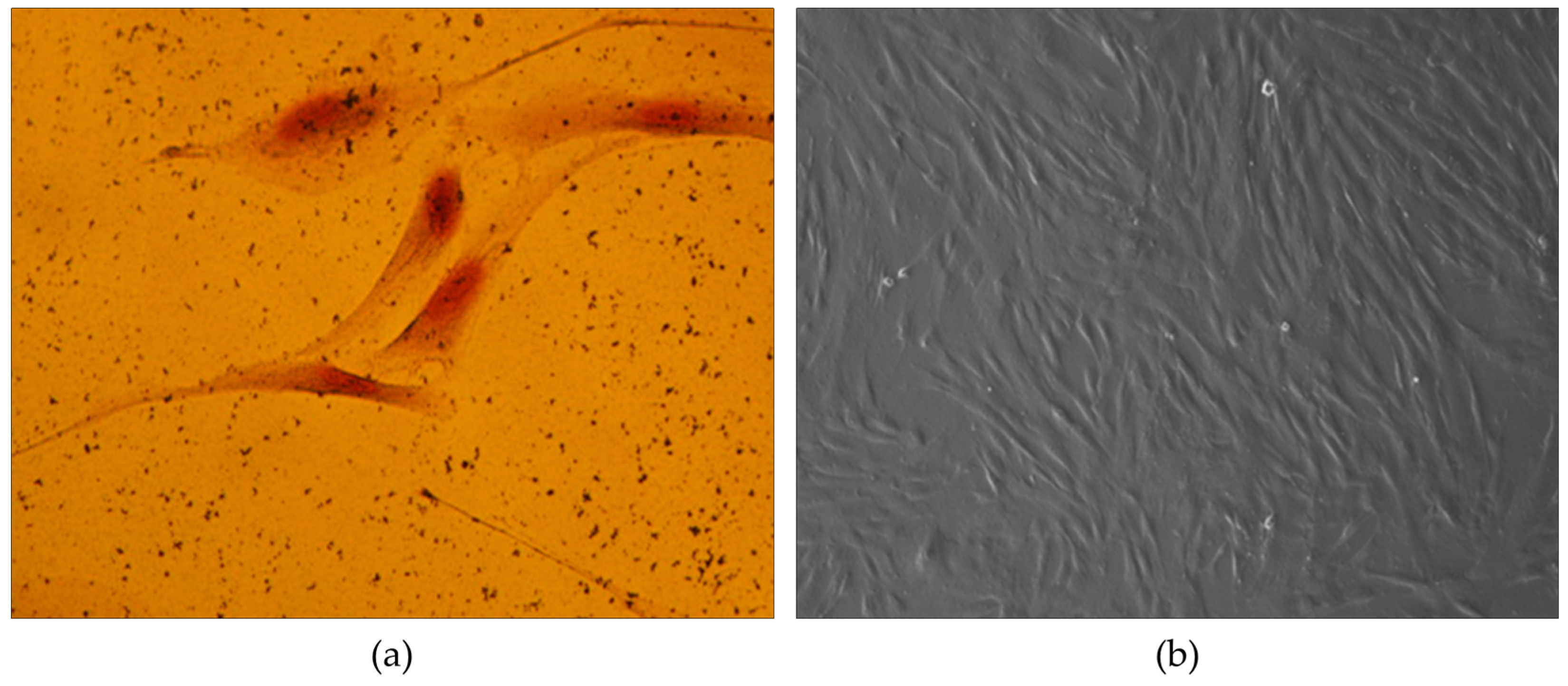
2.3.2. Number and characteristics of cell morphology
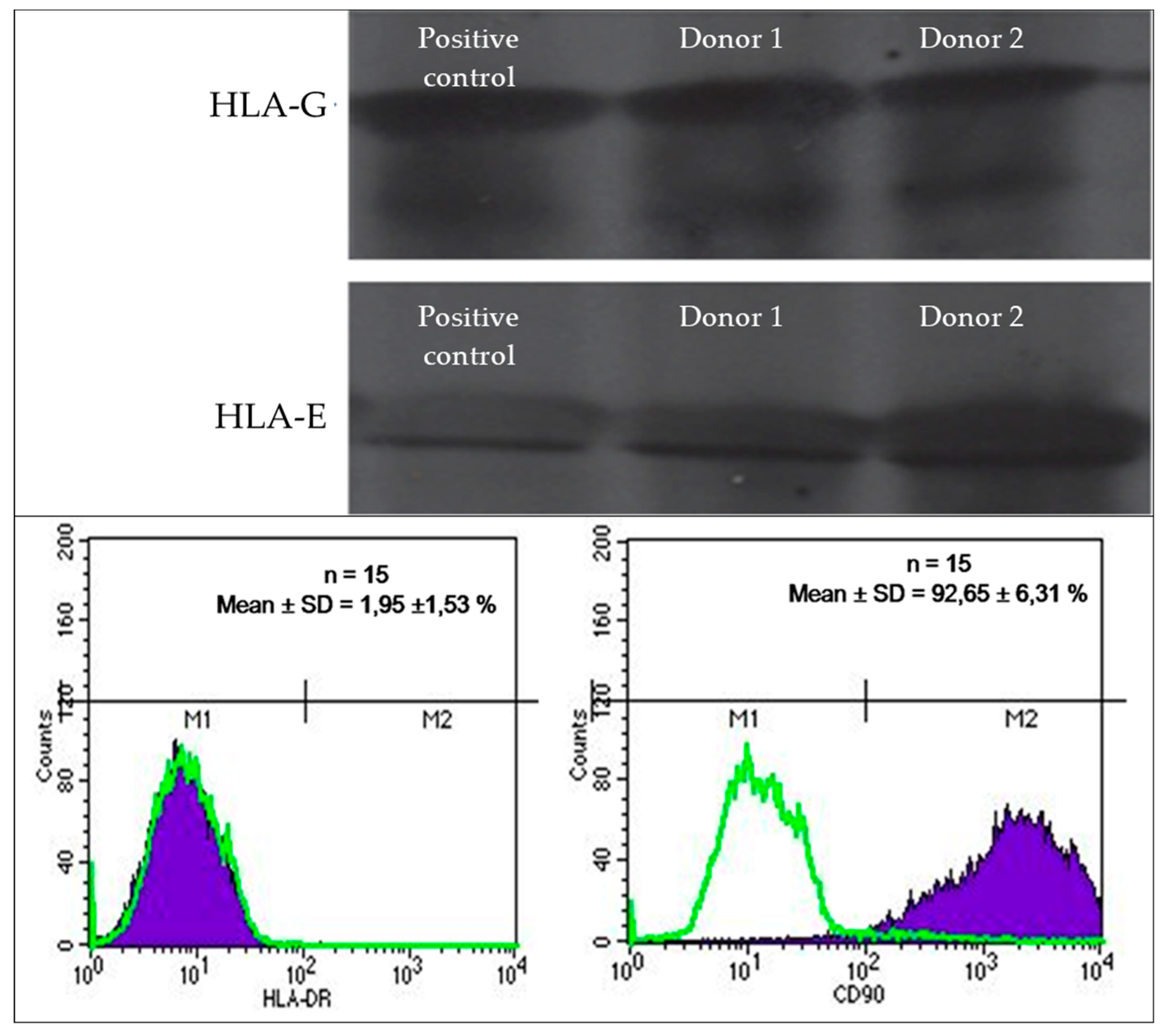
2.4. Investigating the expression of HLA antigens of mesenchymal stem cells
2.4.1. Analysis of the expression of HLA-G and HLA-E
2.4.2. Analysis of the expression of HLA-DR
2.5. Determination of immunogenicity of stem cells
2.5.1. Antigen sample preparation
2.5.2. Rabbit serum
2.5.3. ELISA immunological analysis method
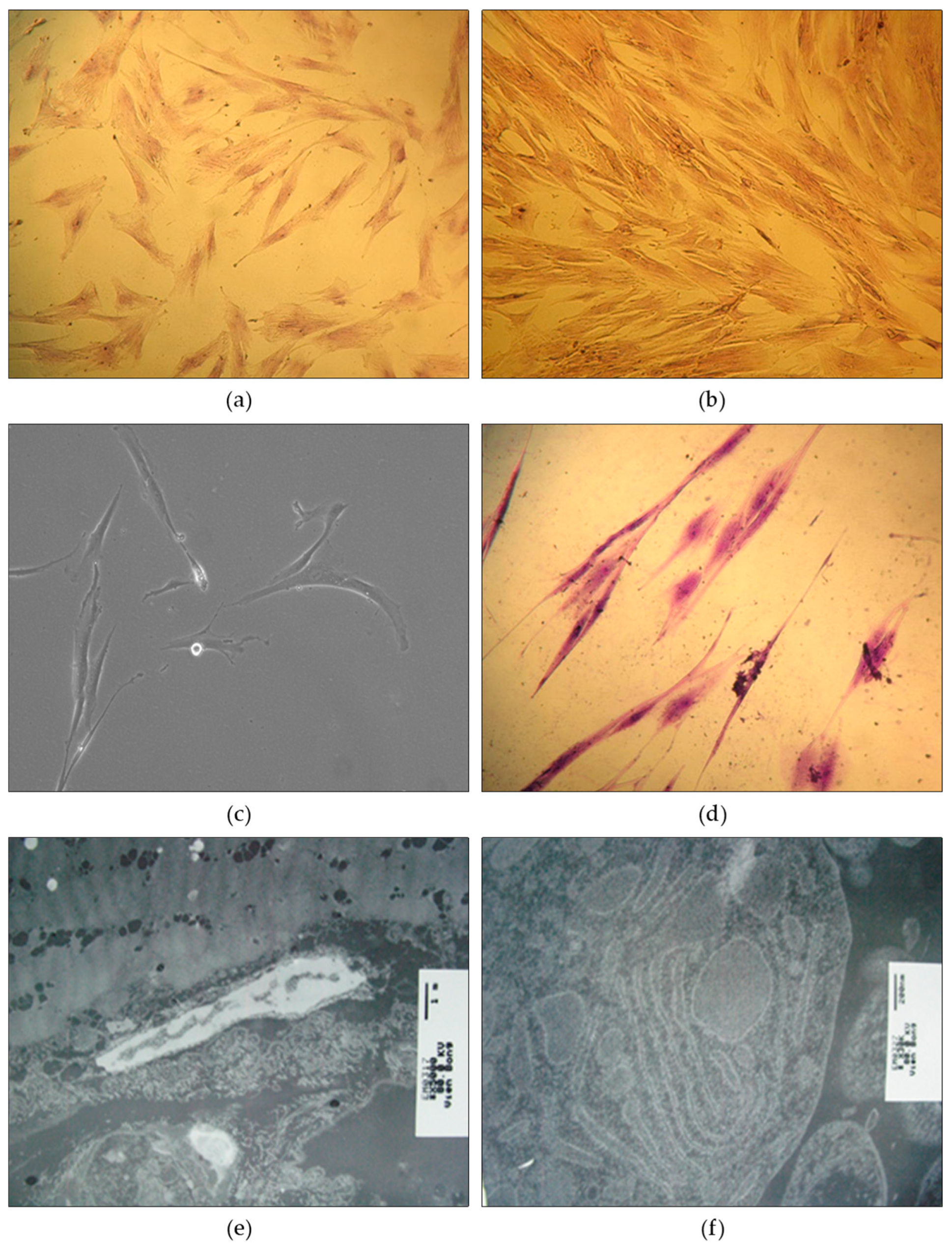
2.6. Differentiation of tissue stem cells into fibroblast cells
2.6.1. Inducing differentiation to generate fibroblasts and mesodermal sheets
2.6.2. ELISA test for collagen type I in the supernatant of differentiated cells
2.7. Burn induction in rabbit models
2.8. Data analysis
3. Results
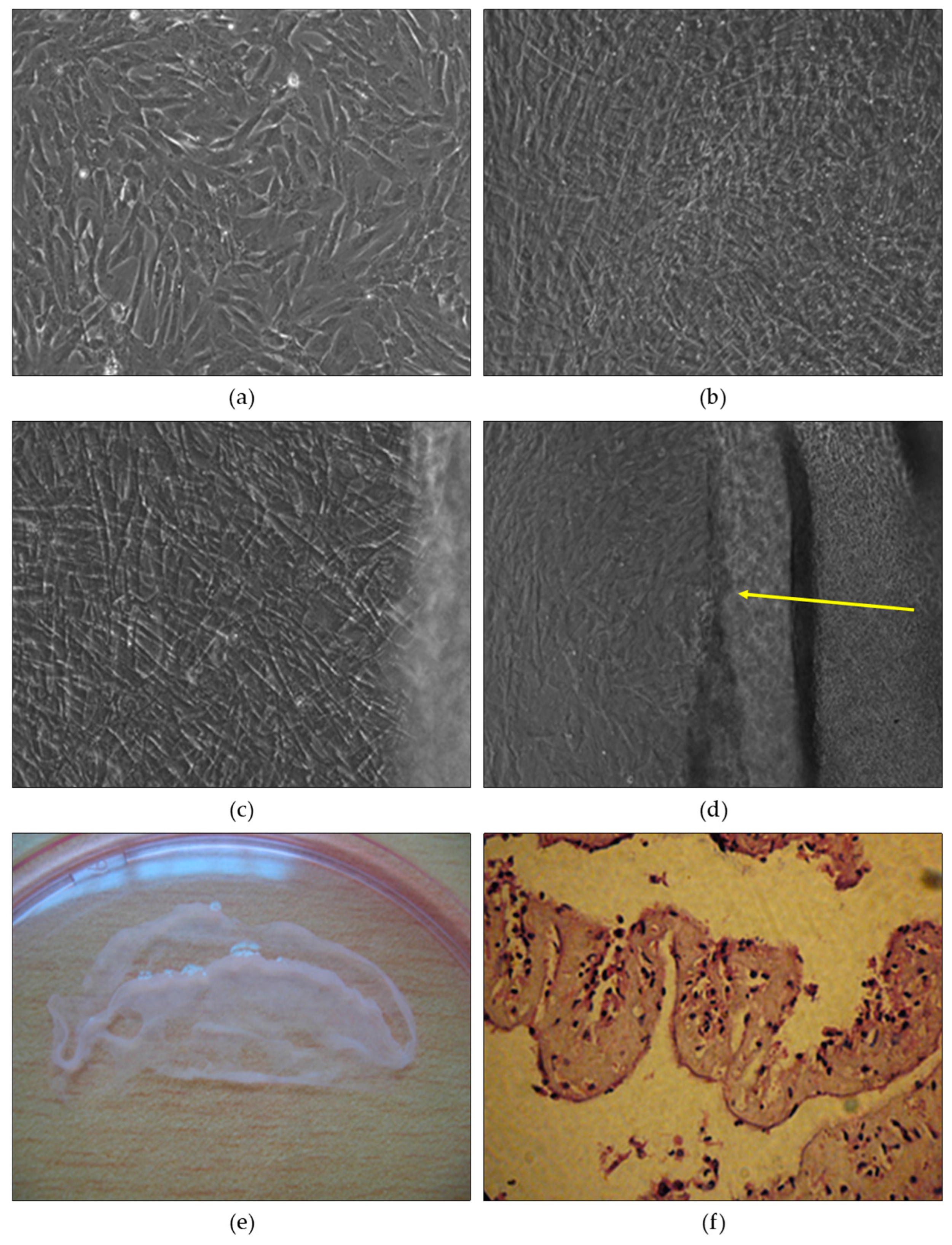
3.1. Cell Growth and Confluence Analysis in Mesenchymal Stem Cell Culture
3.2. HLA Expression Profile of Umbilical Cord Lining Mesenchymal Stem Cells
3.3. Analysis of Cell Differentiation and Cellular Immunogenicity
3.4. Analysis of Collagen Production and Sheet Material Formation
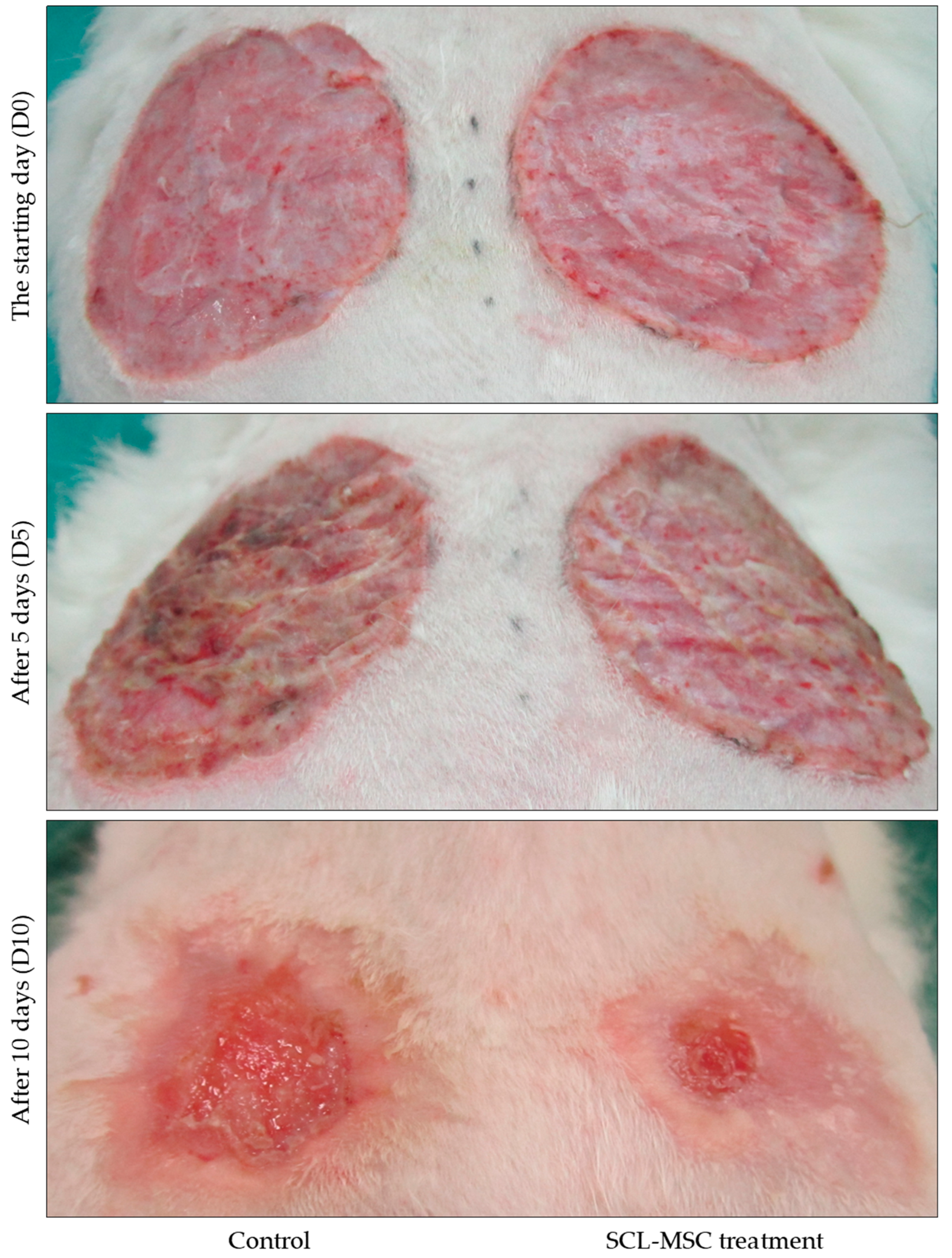
3.5. Alterations in Rabbit Wounds Following Mesenchymal Stem Cell Transplantation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, M.; Xu, X.; Lei, X.; Tan, J.; Xie, H. Mesenchymal stem cell-based therapy for burn wound healing. Burns & Trauma 2021, 9, tkab002. [Google Scholar] [CrossRef]
- Fu, X.; Li, H. Mesenchymal stem cells and skin wound repair and regeneration: possibilities and questions. Cell and Tissue Research 2009, 335, 317–321. [Google Scholar] [CrossRef]
- Nagamura-Inoue, T.; He, H. Umbilical cord-derived mesenchymal stem cells: Their advantages and potential clinical utility. World journal of stem cells 2014, 6, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Harminder, S.D.; Augusto, A.-B. Autologous limbal transplantation in patients with unilateral corneal stem cell deficiency. British Journal of Ophthalmology 2000, 84, 273. [Google Scholar] [CrossRef]
- Thoft, R.A.; Wiley, L.A.; Sundarraj, N. The multipotential cells of the limbus. Eye 1989, 3, 109–113. [Google Scholar] [CrossRef]
- Tseng, S.C.G. Concept and application of limbal stem cells. Eye 1989, 3, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Ang, L.P.K.; Tan, D.T.H. Ocular surface stem cells and disease: current concepts and clinical applications. Ann Acad Med Singap 2004, 33, 576–580. [Google Scholar] [PubMed]
- Puangsricharern, V.; Tseng, S.C.G. Cytologlogic Evidence of Corneal Diseases with Limbal Stem Cell Deficiency. Ophthalmology 1995, 102, 1476–1485. [Google Scholar] [CrossRef]
- Liang, L.; Sheha, H.; Li, J.; Tseng, S.C.G. Limbal stem cell transplantation: new progresses and challenges. Eye 2009, 23, 1946–1953. [Google Scholar] [CrossRef]
- Yiu, S.C.; Thomas, P.B.; Nguyen, P. Ocular surface reconstruction: recent advances and future outlook. Current Opinion in Ophthalmology 2007, 18, 509–514. [Google Scholar] [CrossRef]
- Ang, L.P.K.; Tanioka, H.; Kawasaki, S.; Ang, L.P.S.; Yamasaki, K.; Do, T.P.; Thein, Z.M.; Koizumi, N.; Nakamura, T.; Yokoi, N.; et al. Cultivated Human Conjunctival Epithelial Transplantation for Total Limbal Stem Cell Deficiency. Investigative Ophthalmology & Visual Science 2010, 51, 758–764. [Google Scholar] [CrossRef]
- Tan, D.T.H.; Ang, L.P.K.; Beuerman, R.W. Reconstruction of the Ocular Surface by Transplantation of a Serum-Free Derived Cultivated Conjunctival Epithelial Equivalent. Transplantation 2004, 77. [Google Scholar] [CrossRef] [PubMed]
- Ang, L.P.K.; Nakamura, T.; Inatomi, T.; Sotozono, C.; Koizumi, N.; Yokoi, N.; Kinoshita, S. Autologous Serum–Derived Cultivated Oral Epithelial Transplants for Severe Ocular Surface Disease. Archives of Ophthalmology 2006, 124, 1543–1551. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Yamato, M.; Hayashida, Y.; Watanabe, K.; Yamamoto, K.; Adachi, E.; Nagai, S.; Kikuchi, A.; Maeda, N.; Watanabe, H.; et al. Corneal Reconstruction with Tissue-Engineered Cell Sheets Composed of Autologous Oral Mucosal Epithelium. New England Journal of Medicine 2004, 351, 1187–1196. [Google Scholar] [CrossRef]
- Doan, N.; Huynh, T.Q.; Tran, S.; Wang, G.; Hamlet, S.; Dau, v.; Dao, D.; Nguyen, N.-T.; Nguyen, H.T.; Doan, J.; et al. Multidisciplinary approach to maximize angiogenesis and wound healing using piezoelectric surgery, concentrated growth factors and photobiomodulation for dental implant placement surgery involving lateral wall sinus lift: two case reports. Vascular Cell 2020. [Google Scholar] [CrossRef]
- Reza, H.M.; Ng, B.-Y.; Phan, T.T.; Tan, D.T.H.; Beuerman, R.W.; Ang, L.P.-K. Characterization of a Novel Umbilical Cord Lining Cell with CD227 Positivity and Unique Pattern of P63 Expression and Function. Stem Cell Reviews and Reports 2011, 7, 624–638. [Google Scholar] [CrossRef]
- Freshney, R.I. Culture of animal cells: a manual of basic technique and specialized applications; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Nguyen, T.B.P.; Dinh, V.H.; Nguyen, N.T.; Nguyen, N.L.; Nguyen, T.H. In Vitro Assessment of The Effect of Low Level Laser Therapy on the Proliferation and Migration of Fibroblasts Derived from Patients with Chronic Wounds. Vietnam Medical Journal 2023, 525, 230–235. [Google Scholar] [CrossRef]
- Secunda, R.; Vennila, R.; Mohanashankar, A.M.; Rajasundari, M.; Jeswanth, S.; Surendran, R. Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: a comparative study. Cytotechnology 2015, 67, 793–807. [Google Scholar] [CrossRef]
- Alegre, E.; Rebmann, V.; LeMaoult, J.; Rodriguez, C.; Horn, P.A.; Díaz-Lagares, A.; Echeveste, J.I.; González, A. In vivo identification of an HLA-G complex as ubiquitinated protein circulating in exosomes. European Journal of Immunology 2013, 43, 1933–1939. [Google Scholar] [CrossRef]
- Kwak, J.; Choi, W.; Bae, Y.; Kim, M.; Choi, S.; Oh, W.; Jin, H. HLA-A2 Promotes the Therapeutic Effect of Umbilical Cord Blood-Derived Mesenchymal Stem Cells in Hyperoxic Lung Injury. Bioengineering 2022, 9. [Google Scholar] [CrossRef]
- Busuioc, C.J.; Mogoşanu, G.D.; Popescu, F.C.; Lascăr, I.; Pârvănescu, H.; Mogoantă, L. Phases of the cutaneous angiogenesis process in experimental third-degree skin burns: histological and immunohistochemical study. Romanian journal of morphology and embryology = Revue roumaine de morphologie et embryologie 2013, 54, 163–171. [Google Scholar] [PubMed]
- Li, M.; Luan, F.; Zhao, Y.; Hao, H.; Liu, J.; Dong, L.; Fu, X.; Han, W. Mesenchymal stem cell-conditioned medium accelerates wound healing with fewer scars. International Wound Journal 2017, 14, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Grahovac, T.; Oh, S.J.; Ieraci, M.; Rubin, J.P.; Marra, K.G. Evaluation of a multi-layer adipose-derived stem cell sheet in a full-thickness wound healing model. Acta Biomaterialia 2013, 9, 5243–5250. [Google Scholar] [CrossRef] [PubMed]
| Monitoring Indicators | Zone A | Zone B | P-value |
|---|---|---|---|
| Percent of Wound Healing | |||
| D5 | 77.44±12.24 | 68.76±11.14 | < 0.01 |
| D10 | 93.23±3.74 | 87.36±4.78 | < 0.001 |
| Area of Epithelialization (cm²) | |||
| D5 | 14.18±3.33 | 12.52±2.83 | < 0.05 |
| D10 | 16.95±2.31 | 15.74±1.93 | < 0.05 |
| Number of Inflammatory Cells per Unit Area | |||
| D5 | 18.86±4.46 | 18.01±5.08 | 0.475 |
| D10 | 8.96±1.80 | 12.5±3.01 | < 0.001 |
| Number of Fibroblasts per Unit Area | |||
| D5 | 37.2±8.26 | 36.56±9.35 | 0.76 |
| D10 | 67.1±9.09 | 52.73±10.38 | < 0.001 |
| Number of Neo-vessels per Unit Area | |||
| D5 | 3.50±1.04 | 3.67±1.02 | 0.484 |
| D10 | 7.23±1.13 | 4.90±0.95 | < 0.001 |
| Mitotic Index (MI × 100) | |||
| D5 | 1.53±0.50` | 1.47±0.51 | 0.189 |
| D10 | 2.47±0.56 | 1.86±0.51 | < 0.010 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




