Submitted:
14 June 2023
Posted:
15 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Mosquito strain and rearing conditions
2.2. Dieldrin selection at the L1 stage
2.3. Effect of dieldrin concentration on sexing
2.4. Effect of dieldrin exposure time on sex sorting
2.5. Measurement of dieldrin toxicity effect on resistant males
2.6. Quantification of dieldrin residues in resistant male adults
2.6.1. Quantification at a small production scale
2.6.2. Quantification of dieldrin on a mass production context
2.7. Dieldrin extraction and quantification
2.8. Statistical analysis
3. Results
3.1. Stability of Tikok line and efficiency of sex sorting at L1 stage
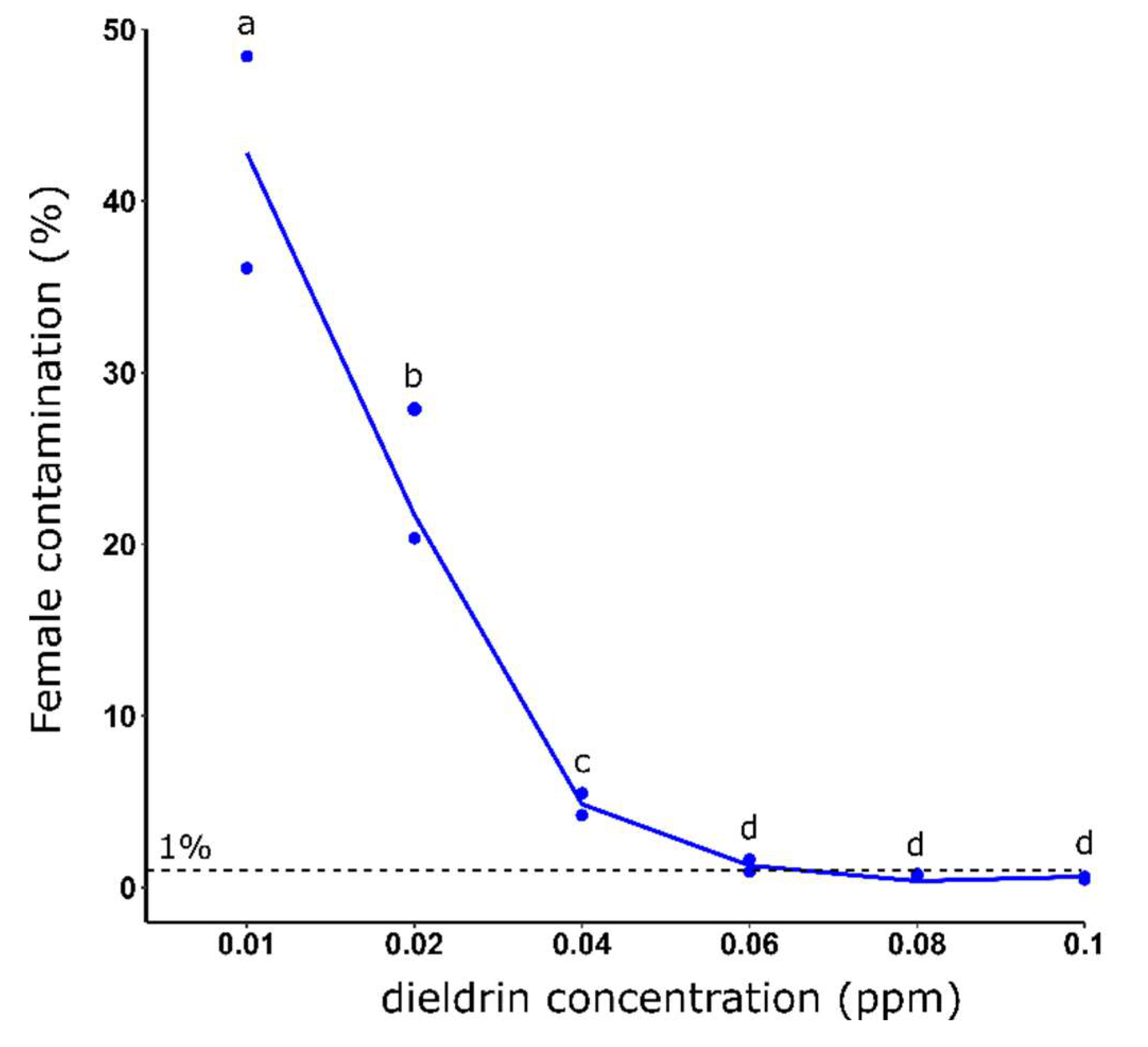
3.2. Effect of dieldrin concentration on the female elimination efficiency
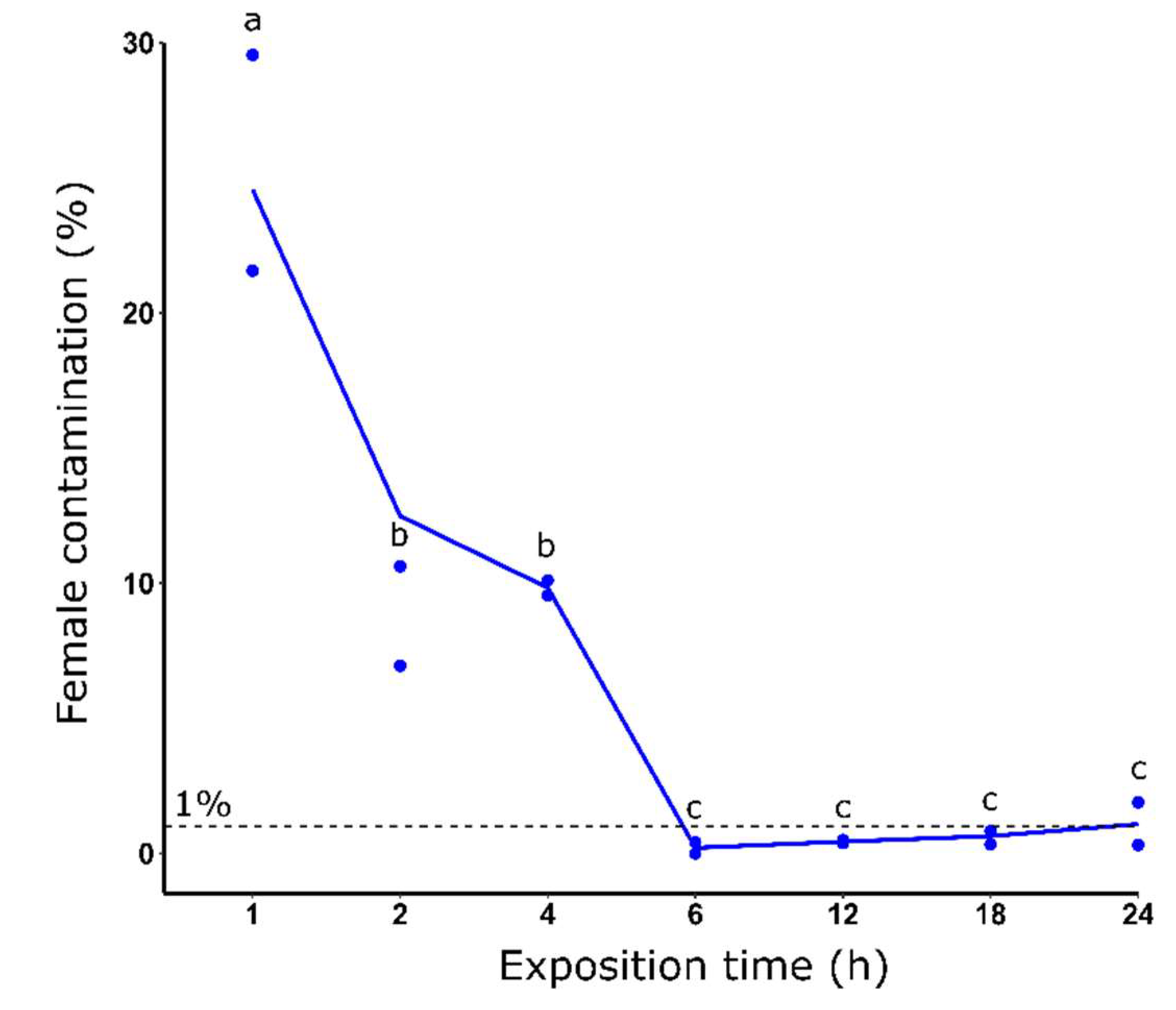
3.3. Effect of dieldrin exposure time on sex sorting
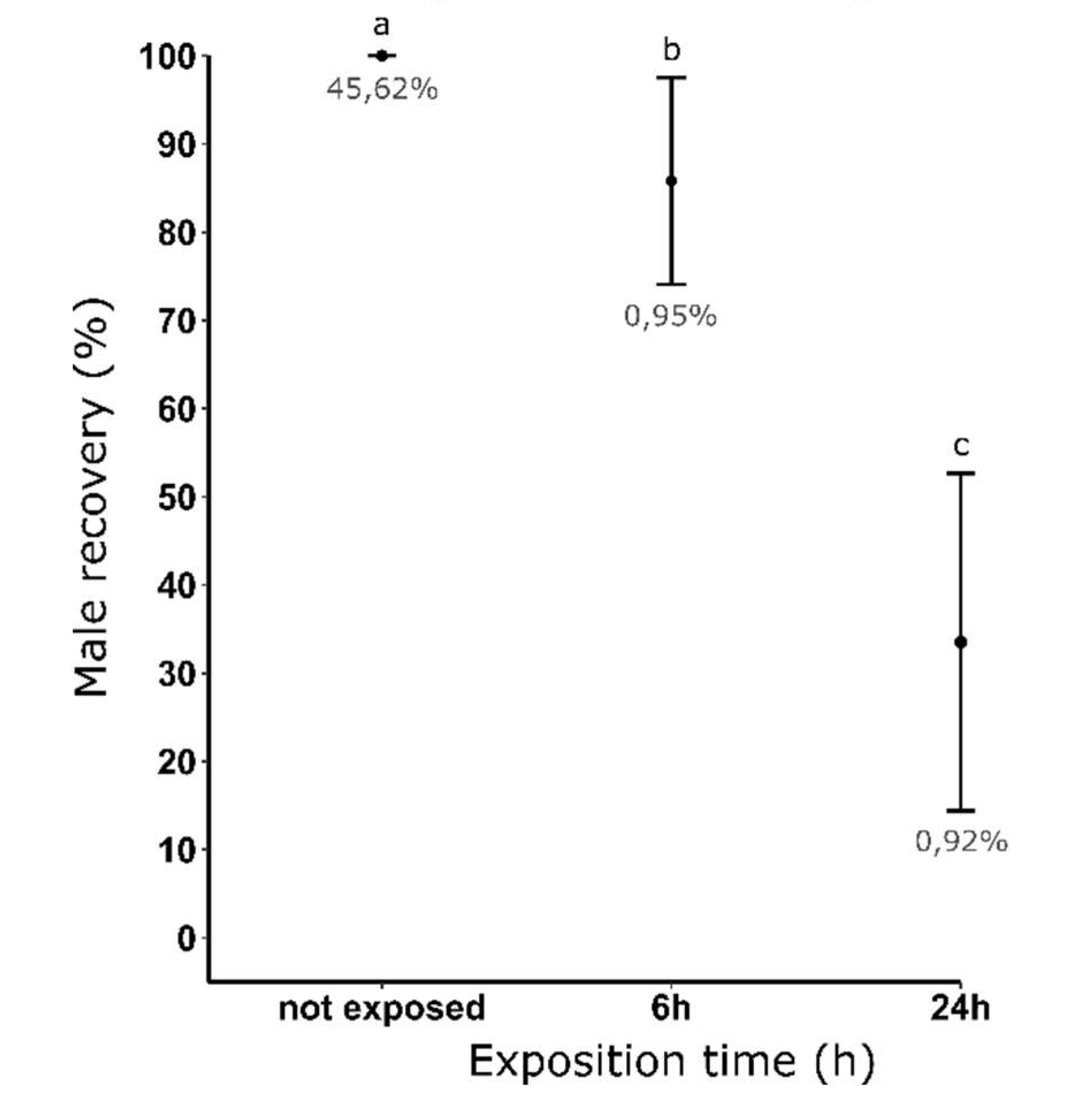
3.3. Measurement of dieldrin toxicity effect on resistant males
3.4. Quantification of dieldrin residues in resistant male adults
3.4.1. Quantification at a small production scale
3.4.2. Quantification of dieldrin in males obtained in a mass production context
4. Discussion
Author Contributions
Conflicts of Interest
References
- Organization, W.H. Global Vector Control Response 2017-2030. Glob. vector Control response 2017-2030. 2017.
- Bourtzis, K.; Dobson, S.L.; Xi, Z.; Rasgon, J.L.; Calvitti, M.; Moreira, L.A.; Bossin, H.C.; Moretti, R.; Baton, L.A.; Hughes, G.L.; et al. Harnessing Mosquito-Wolbachia Symbiosis for Vector and Disease Control. Acta Trop. 2014, 132, S150–S163. [Google Scholar] [CrossRef] [PubMed]
- Lees, R.S.; Gilles, J.R.; Hendrichs, J.; Vreysen, M.J.; Bourtzis, K. Back to the Future: The Sterile Insect Technique against Mosquito Disease Vectors. Curr. Opin. Insect Sci. 2015, 10, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Papathanos, P.A.; Bourtzis, K.; Tripet, F.; Bossin, H.; Virginio, J.F.; Capurro, M.L.; Pedrosa, M.C.; Guindo, A.; Sylla, L.; Coulibaly, M.B.; et al. A Perspective on the Need and Current Status of Efficient Sex Separation Methods for Mosquito Genetic Control. Parasites and Vectors 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Lutrat, C.; Giesbrecht, D.; Marois, E.; Whyard, S.; Baldet, T.; Bouyer, J. Sex Sorting for Pest Control: It’s Raining Men! Trends Parasitol. 2019, 35, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, F.; Galizi, R.; Menichelli, M.; Papathanos, P.A.; Dritsou, V.; Marois, E.; Crisanti, A.; Windbichler, N. Site-Specific Genetic Engineering of the Anopheles Gambiae y Chromosome. Proc. Natl. Acad. Sci. U. S. A. 2014, 111, 7600–7605. [Google Scholar] [CrossRef] [PubMed]
- Catteruccia, F.; Benton, J.P.; Crisanti, A. An Anopheles Transgenic Sexing Strain for Vector Control. Nat. Biotechnol. 2005, 23, 1414–1417. [Google Scholar] [CrossRef] [PubMed]
- Lutrat, C.; Olmo, R.P.; Baldet, T.; Bouyer, J.; Marois, E. Transgenic Expression of Nix Converts Genetic Females into Males and Allows Automated Sex Sorting in Aedes Albopictus. Commun. Biol. 2022, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- McDonald, P.T.; Asman, S.M. ; others A Genetic-Sexing Strain Based on Malathion Resistance for Culex Tarsalis. Mosq. News 1982, 42, 531–536. [Google Scholar]
- Yamada, H.; Jandric, Z.; Chhem-Kieth, S.; Vreysen, M.J.B.; Rathor, M.N.; Gilles, J.R.L.; Cannavan, A. Anopheles Arabiensis Egg Treatment with Dieldrin for Sex Separation Leaves Residues in Male Adult Mosquitoes That Can Bioaccumulate in Goldfish (Carassius Auratus Auratus). Environ. Toxicol. Chem. 2013, 32, 2786–2791. [Google Scholar] [CrossRef] [PubMed]
- Lebon, C.; Benlali, A.; Atyame, C.; Mavingui, P.; Tortosa, P. Construction of a Genetic Sexing Strain for Aedes Albopictus: A Promising Tool for the Development of Sterilizing Insect Control Strategies Targeting the Tiger Mosquito. Parasites and Vectors 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Vreysen, M.J.B.; Bourtzis, K.; Tschirk, W.; Chadee, D.D.; Gilles, J.R.L. The Anopheles Arabiensis Genetic Sexing Strain ANO IPCL1 and Its Application Potential for the Sterile Insect Technique in Integrated Vector Management Programmes. Acta Trop. 2015, 142, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Esnault, J.; Scussel, S.; Gaudillat, B.; Lejarre, Q.; Toussaint, M.; Duployer, M.; Gárdos, M.; Bán, P.; Mavingui, P.; Tortosa, P.; et al. Development of an Automated Mosquito Pupae Counter. J. Med. Entomol. 2023, 1–5. [Google Scholar] [CrossRef]
- Le Goff, G.; Damiens, D.; Ruttee, A.H.; Payet, L.; Lebon, C.; Dehecq, J.S.; Gouagna, L.C. Field Evaluation of Seasonal Trends in Relative Population Sizes and Dispersal Pattern of Aedes Albopictus Males in Support of the Design of a Sterile Male Release Strategy. Parasites and Vectors 2019, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO) Aldrin and Dieldrin in Drinking-Water. 2003, 14.
- European Commission Regulation (EC) No 396/2005, Maximum Residue Levels of Pesticides in/on Food and Feed of Plant and Animal. Off. J. Eur. Union 2005, 1–16.
- INERIS Dieldrine. Données Tech. sur les Subst. Chim. en Fr. 2007, 1–13.
| Larvae stage exposed | Generations | Number of male pupae | Number of female pupae | Total of pupae | Female contamination (%) |
|---|---|---|---|---|---|
| L3 | F27 | 592 | 7 | 599 | 1.17 |
| F28 | 306 | 4 | 310 | 1.29 | |
| F29 | 457 | 4 | 461 | 0.87 | |
| F30 | 469 | 3 | 472 | 0.64 | |
| F31 | 2372 | 15 | 2387 | 0.63 | |
| F32 | 2041 | 13 | 2054 | 0.63 | |
| F33 | 3331 | 31 | 3362 | 0.92 | |
| F34 | 2470 | 16 | 2486 | 0.64 | |
| L1 | F35 | 2463 | 10 | 2473 | 0.40 |
| F36 | 841 | 7 | 848 | 0.83 | |
| F37 | 1153 | 8 | 1161 | 0.69 | |
| F38 | 638 | 5 | 643 | 0.78 | |
| F39 | 3239 | 26 | 3265 | 0.80 | |
| F40 | 3001 | 30 | 3031 | 0.99 | |
| F41 | 4326 | 33 | 4359 | 0.76 | |
| F42 | 4822 | 37 | 4859 | 0.76 |
| Exposition time (h) | Female contamination (%) | Male recovery (%) | Dieldrin (µg/L) | Dieldrin (µg/kg) | Dieldrin / mosquito (µg) |
|---|---|---|---|---|---|
| 0 | 45.57 | 100.00 | |||
| 6 | 0.68 | 90.70 | 2.05 (±0.07) | 18.72 (±1.81) | 8.32 x 10-6 (±3.08 10-7) |
| 24 | 0.86 | 33.88 | 486 (±153.48) | 3841.09 (±1427.40) | 1.94 x 10-3 (±6.14 10-4) |
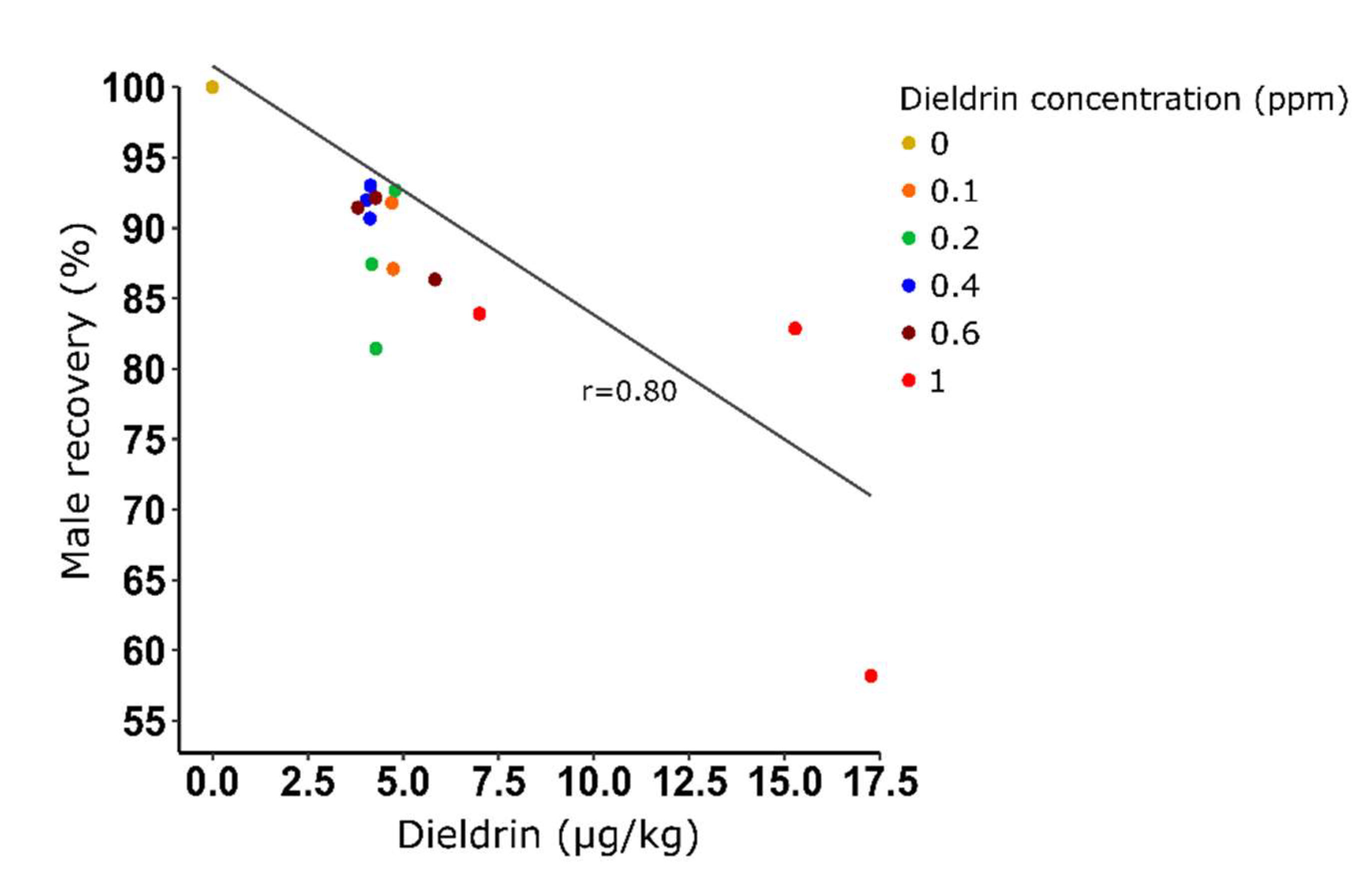
| Dieldrin concentration (ppm) | Female contamination (%) | Male recovery (%) | Dieldrin (µg/L) | Dieldrin (µg/kg) | Dieldrin / mosquito (µg) |
|---|---|---|---|---|---|
| 0 | 51,1 | 100 | |||
| 0,1 | 18,65 (±3,51) | 93,87 (±8,01) | ≤ 2 | ≤ 4,71 (±0,03) | ≤ 2 x 10-6 |
| 0,2 | 6,87 (±2,78) | 87,19 (±5,63) | ≤ 2 | ≤ 4,42 (±0,33) | ≤ 2 x 10-6 |
| 0,4 | 4,10 (±1,25) | 91,89 (±1,17) | ≤ 2 | ≤ 4,10 (±0,06) | ≤ 2 x 10-6 |
| 0,6 | 3,09 (±0,29) | 89,98 (±3,17) | ≤ 2,33 | ≤ 4,64 (±1,06) | ≤ 2,33 x 10-6 |
| 1 | 2,42 (±1,07) | 74,97 (±14,56) | 6,33 | 13,17 (±5,44) | 6,66 x 10-6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




