Submitted:
12 May 2023
Posted:
15 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant materials and Growing Conditions
2.2. Experimental Design and Treatments
2.2. Growth and Morphology Measurements
2.3. Statistical Analysis
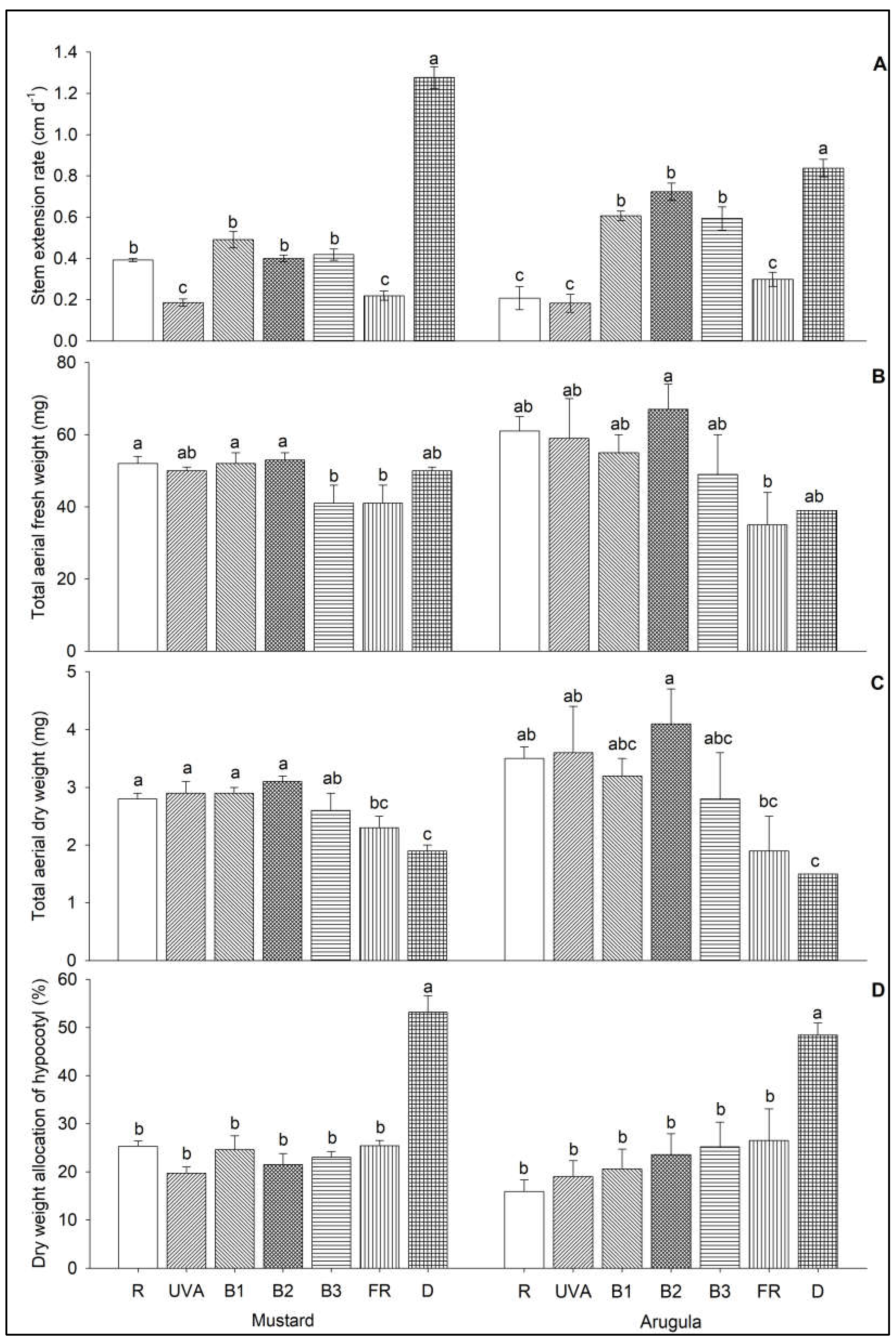
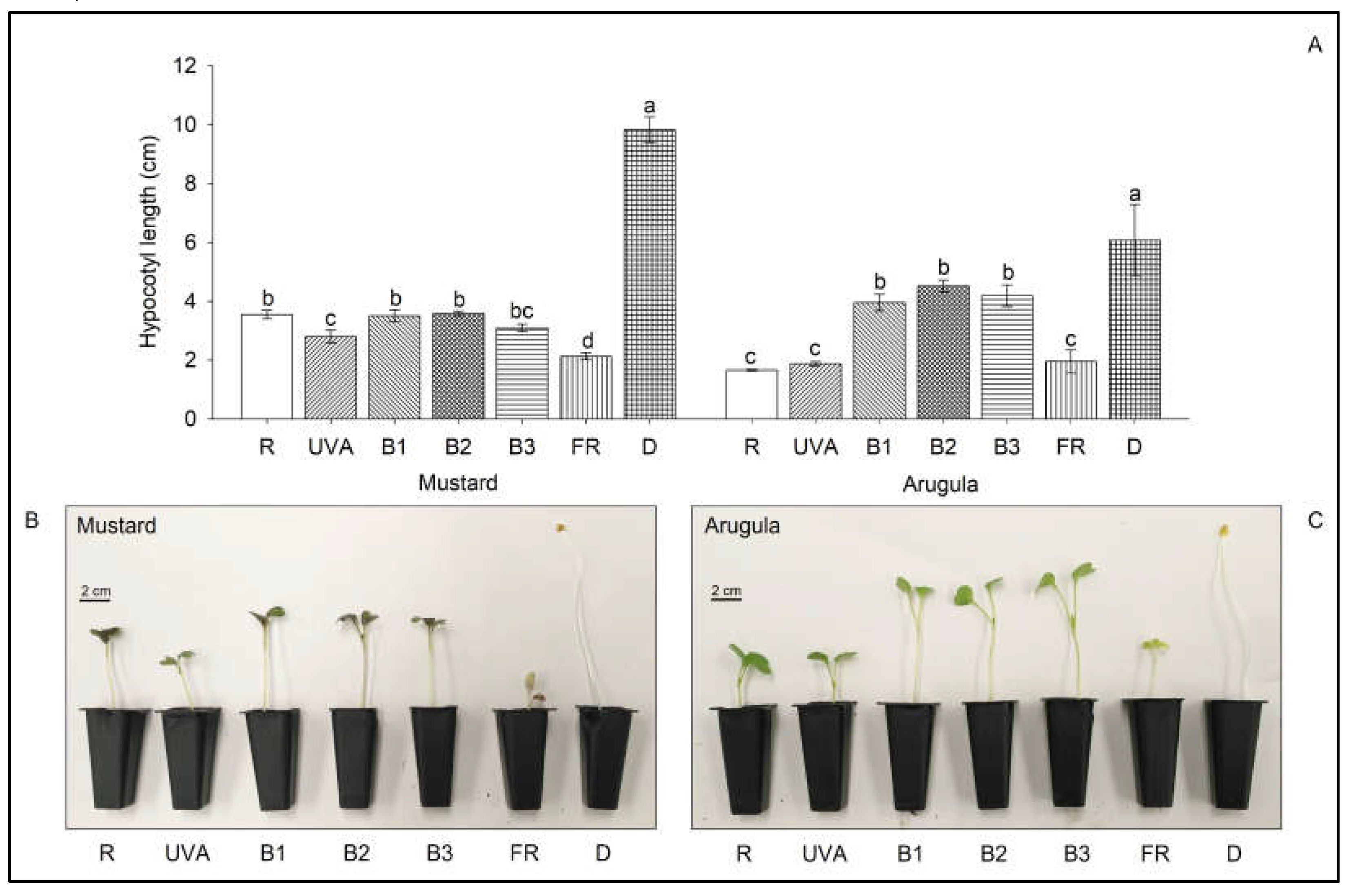
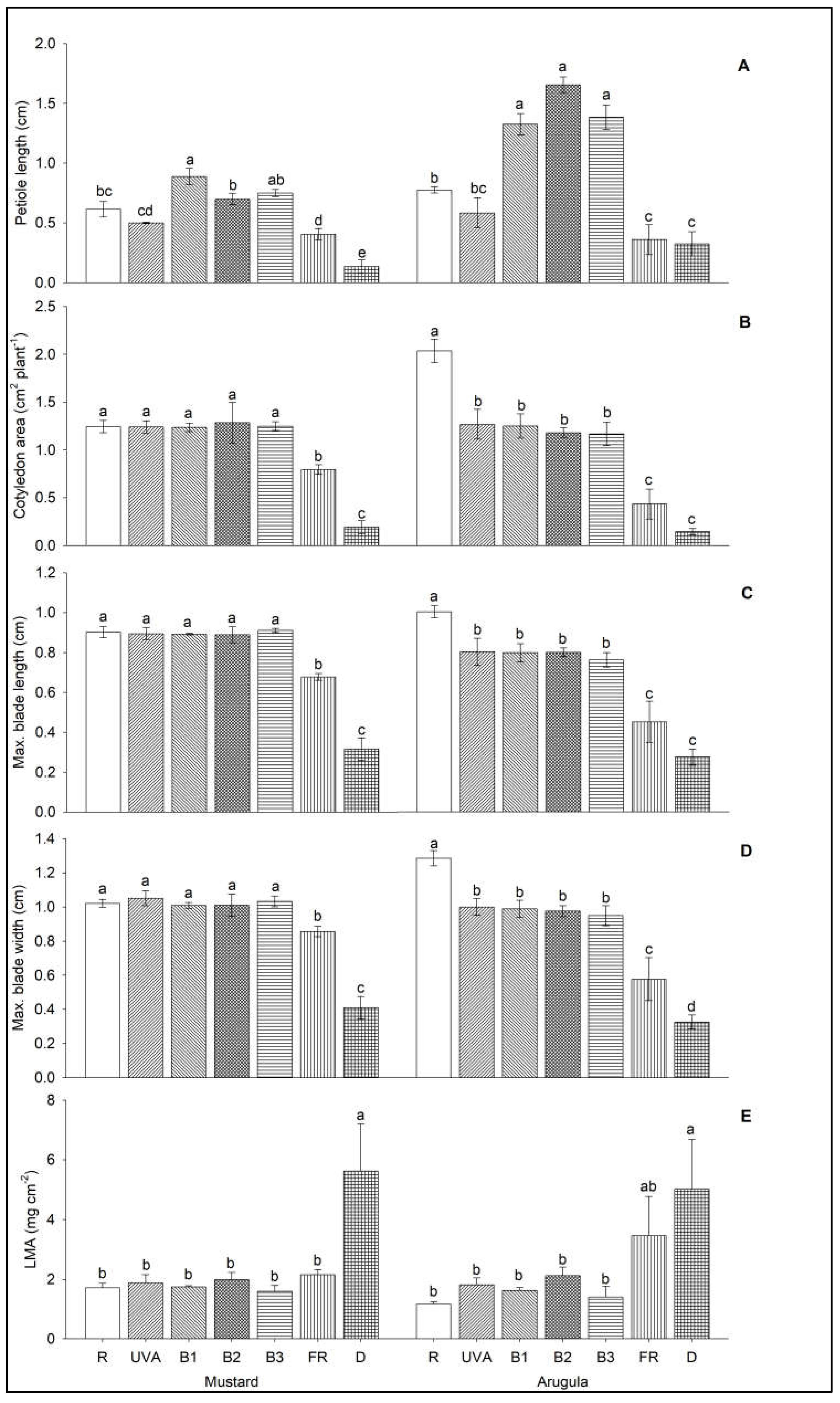
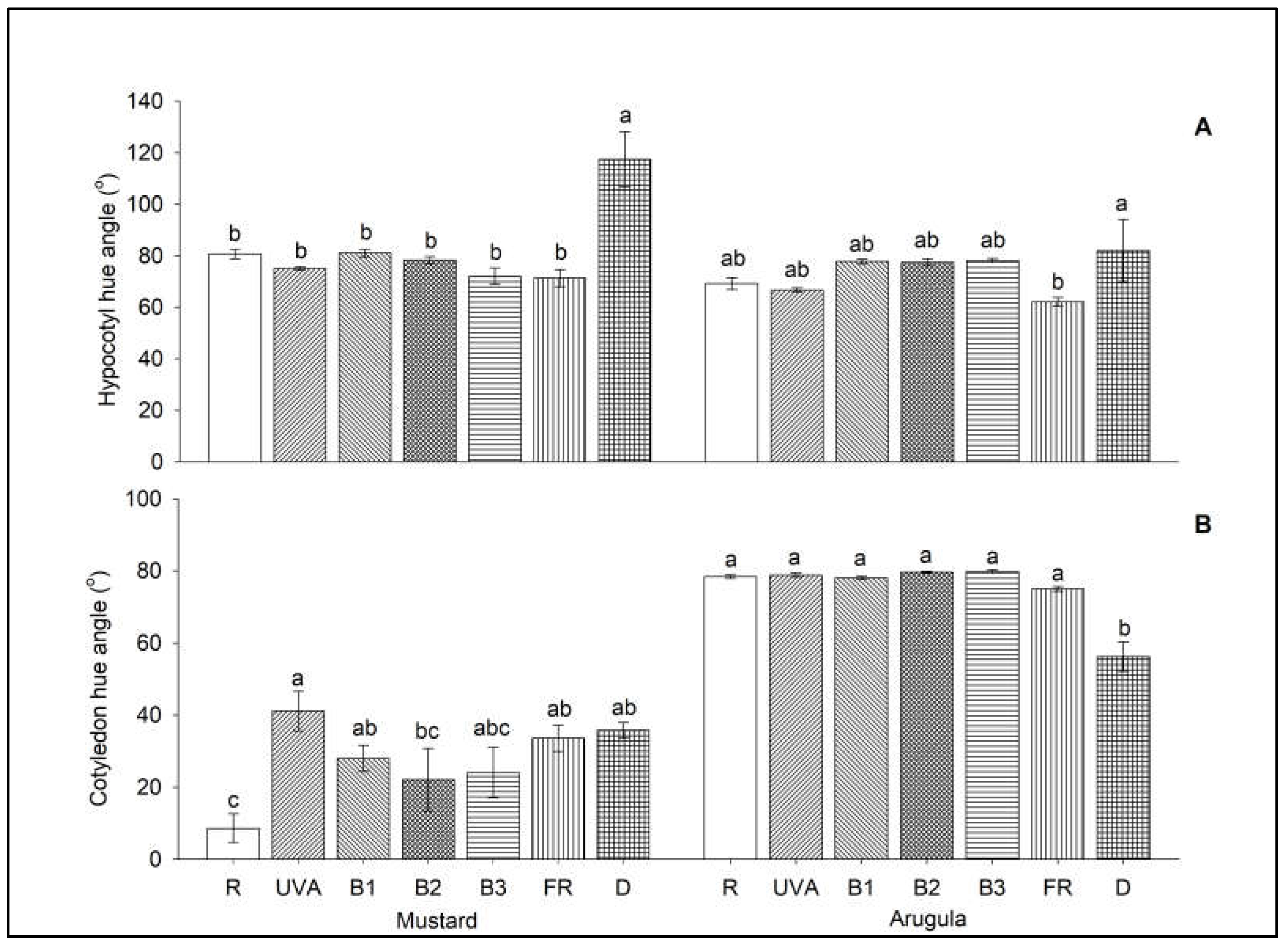
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability
Acknowledgements
Conflicts of Interest
References
- Smith, H., Whitelam G.C. Phytochrome, a family of photoreceptors with multiple physiological roles. Plant Cell Environ. 1990,13, 695–707.
- Smith, H., Whitelam, G.C. The shade avoidance syndrome: multiple responses mediated by multiple phytochromes. Plant Cell Environ. 1997, 20, 840–844.
- Sager, J.C., Smith, W.O., Edwards, J.L., Cyr, K.L. Photosynthetic efficiency and phytochrome photoequilibria determination using spectral data. Trans. ASAE. 1988, 31, 1882–1889.
- Stutte, G.W. Light-emitting diodes for manipulating the phytochrome apparatus. HortScience 2009, 44, 231–234.
- Kusuma, P., Bugbee, B. Improving the predictive value of phytochrome photoequilibrium: Consideration of spectral distortion within a leaf. Front. Plant Sci. 2021, 12, 596943.
- Park, Y., Runkle, E.S. Far-red radiation and photosynthetic photon flux density independently regulate seedling growth but interactively regulate flowering, Env. Expt. Bot. 2018, 155, 206–216.
- Casal, J., Smith, H. The function, action and adaptive significance of phytochrome in light-grown plants. Plant Cell Environ. 1989, 12, 855–862.
- Smith, H. Light quality, photoperception, and plant strategy. Annual Rev. Plant Phys. 1982, 33, 481–518.
- Smith, H. Phytochromes and light signal perception by plants - An emerging synthesis. Nature 2000, 407, 585–591.
- Llewellyn, D., Zheng, Y., Dixon. M.Survey of how hanging baskets influence the light environment at lower crop level in ornamental greenhouses in Ontario. HortTechnology 2013, 23, 823–829.
- Kusuma, P., Bugbee, B. Far-red fraction: An improved metric for characterizing phytochrome effects on morphology. J. Am. Soc. Hort. Sci. 2020, 146, 3–13.
- Appelgren, M. Effects of light quality on stem elongation of Pelargonium in vitro. Sci. Hort. 1991, 45, 345–351.
- Brown, C.S., Schuerger, A.C., Sager, J.C. Growth and photomorphogenesis of pepper plants under red light-emitting diodes with supplemental blue or far-red lighting. J. Am. Soc. Hort. Sci. 1995, 120, 808–813.
- Cosgrove, D.J. Photomodulation of growth. In Photomorphogenesis in Plants; Kendrick, R.E., Kronenberg, G.H.M. Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1994; pp. 631–658.
- Hoenecke, M.E., Bula, R.J., Tibbitts, T.W. Importance of 'blue' photon levels for lettuce seedlings grown under red-light-emitting diodes. HortScience 1992, 27, 427–430.
- Kong, Y., Wang, S., Chen, J., Chen, Q., Yao, Y. Effect of supplemental lighting with red and blue light on the characters of container-growing seedlings of muskmelon. Acta Hort. 2012, 944, 141–146.
- Wheeler, R.M., Mackowiak, C.L., Sager, J.C. Soybean stem growth under high-pressure sodium with supplemental blue lighting. Agron. J. 1991, 83, 903–906.
- Bergstrand, K.J., Asp, H., Schüssler, H.K. Development and acclimatisation of horticultural plants subjected to narrowband lighting. Eur. J. Hort. Sci. 2014, 79, 45–51.
- Kook, H.S., Park, S.H., Jang, Y.J., Lee, G.W., Kim, J.S., Kim, H.M., Oh, B.T., Chae, J.C., Lee, K.J. Blue LED (light-emitting diodes)-mediated growth promotion and control of Botrytis disease in lettuce, Acta Agriculturae Scandinavica 2013, 63, 271–277.
- Lee, N.Y., Lee, M.J., Kim, Y.K., Park, J.C., Park, H.K., Choi, J.S., Hyun, J.N., Kim, K.J., Park, K.H., Ko, J.K., Kim, J.G. Effect of light emitting diode radiation on antioxidant activity of barley leaf. J. Korean Soc. Appl. Biol. Chem. 2010, 53, 685–690.
- Wollaeger, H., Runkle, E. Growth of impatiens, petunia, salvia, and tomato seedlings under blue, green, and red light-emitting diodes. HortScience 2014a, 49, 734–740.
- Wollaeger, H., Runkle, E. Producing commercial-quality ornamental seedlings under sole-source LED lighting. Acta Hort. 2014b. 1037, 269–276.
- Wollaeger, H., Runkle, E. Growth and acclimation of impatiens, salvia, petunia, and tomato seedlings to blue and red light. HortScience 2015, 50, 522–529.
- Hata, N., Hayashi, Y., Ono, E., Satake, H., Kobayashi, A., Muranaka, T., Okazawa, A. Differences in plant growth and leaf sesamin content of the lignan-rich sesame variety ‘Gomazou’under continuous light of different wavelengths. Plant Biotechnol. 2013, 30, 1–8.
- Hernández, R., Kubota, C. Physiological responses of cucumber seedlings under different blue and red photon flux ratios using LEDs. Environ. Exp. Bot. 2016, 121, 66–74.
- Hirai, T., Amaki, W., Watanabe, H. Action of blue or red monochromatic light on stem internodal growth depends on plant species. Acta Hort. 2006, 711, 345–350.
- Kim, E.Y., Park, S.A., Park, B.J., Lee, Y., Oh, M.M. Growth and antioxidant phenolic compounds in cherry tomato seedlings grown under monochromatic light-emitting diodes. Hort. Environ. Biotechnol. 2014, 55, 506–513.
- Schwend, T., Prucker, D., Mempel, H. Red light promotes compact growth of sunflowers. Eur. J. Hort. Sci. 2015, 80, 56–61.
- Kong, Y., Stasiak, M., Dixon, M.A., Zheng, Y. Blue light associated with low phytochrome activity can promote elongation growth as shade-avoidance response: a comparison with red light in four bedding plant species. Environ. Exp. Bot. 2018, 155, 345–359.
- Kong, Y., Kamath, D., Zheng, Y. Blue versus red light can promote elongation growth independent of photoperiod: a study in four Brassica microgreens species. HortScience 2019a, 54, 1955–1961.
- Kong, Y., Schiestel, K., Zheng, Y. Pure blue light effects on growth and morphology are slightly changed by adding low-level UVA or far-red light: A comparison with red light in four microgreen species. Environ. Exp. Bot. 2019b, 157, 58–68.
- Kong, Y., Schiestel, K., Zheng, Y. Maximum elongation growth promoted as a shade-avoidance response by blue light is related to deactivated phytochrome: a comparison with red light in four microgreen species. Can. J. Plant Sci. 2020, 100, 314–326.
- Kong, Y., Zheng, Y. Growth and morphology responses to narrowband blue light and its co-action with low-level UVB or green light: A comparison with red light in four microgreen species. Environ. Exp. Bot. 2020a,178, 104189.
- Kong, Y., Zheng., Y. Phytochrome contributes to blue-light-mediated stem elongation and flower initiation in mature Arabidopsis thaliana plants. Can. J. Plant Sci. 2021, 102, 449–458.
- Kong, Y., Zheng, Y. Phototropin is partly involved in blue-light-mediated stem elongation, flower initiation, and leaf expansion: A comparison of phenotypic responses between wild Arabidopsis and its phototropin mutants. Environ. Exp. Bot. 2020b, 171, 104189.
- Kong, Y., Zheng, Y. Low-activity cryptochrome 1 plays a role in promoting stem elongation and flower initiation of mature Arabidopsis under blue light associated with low phytochrome activity. Can. J. Plant Sci. 2022, 102, 755–759.
- Lee, J.S., Ae, C., Lee, Y., Kim, H., Song, J.Y. Shorter wavelength blue light promotes growth of green perilla (Perilla frutescens). Int. J. Agric. Biol. Eng. 2014, 16, 1177–1182.
- Demotes-Mainard, S., Péron, T., Corot, A., Bertheloot, J., Le, G.J., Pelleschi-Travier, S., Crespel, L., Morel, P., Huché-Thélier, L., Boumaza, R., Vian, A., Guérin, V., Leduc, N., Sakr, S. Plant responses to red and far-red lights, applications in horticulture. Environ. Exp. Bot. 2016. 121, 4–21.
- Maloof, J., Borevitz, J., Dabi, T. Lutes, J., Nehring, R., Redfern, J., Trainer, G., Wilson, J., Asami, T., Berry, C., Weigel, D., Chory, J. Natural variation in light sensitivity of Arabidopsis. Nat. Genet. 2021, 29, 441–446.
- Razzak, A., Ranade, S.S., Strand, Å., García-Gil, M.R. Differential response of Scots pine seedlings to variable intensity and ratio of red and far-red light. Plant Cell Environ. 2017, 40, 1332–1340.
- Johnson, R.E., Kong, Y., Zheng, Y.J.E., Botany, E. Elongation growth mediated by blue light varies with light intensities and plant species: A comparison with red light in arugula and mustard seedlings. Environ. Exp. Bot. 2020, 169, 1–9.
- Alabadí, D., Blázquez, M.A. Molecular interactions between light and hormone signaling to control plant growth. Plant Mol. Biol. 2009, 69, 409–417.
- Chaiwanon, J., Wang, W., Zhu, J.Y., Oh, E., Wang, Z.Y. Information integration and communication in plant growth regulation. Cell 2016, 164, 1257–1268.
- Chen, M., Chory, J., Fankhauser, C. Light signal transduction in higher plants. Annu. Rev. Genet. 2004, 38, 87–117.
- McNellis, T.W., and Deng, X.-W. Light control of seedling morphogenetic pattern. Plant Cell 1995, 7, 1749–1761.
- Mah, J.J., D. Llewellyn, Y. Zheng. Protocol for converting spectrometer radiometric data to photon flux units [Microsoft Excel Spreadsheet]. Guelph, University of Guelph. 2019. Available from: https://cesrf.uoguelph.ca/technotes/ [right click on “TechNote 001-2019” and use “save link as” to save file, last accessed: 19 April, 2023].
- O’Neal, M., Landis, D., Isaacs, R. An inexpensive, accurate method for measuring leaf area and defoliation through digital image analysis. J. Economic Entomology 2003, 95, 1190–1194.
- Karcher, D.E., Richardson, M.D. Quantifying turfgrass color using digital image analysis. Crop Sci. 2003, 43, 943–951.
- Jones-Baumgardt, C., Ying, Q., Zheng, Y., Bozzo, G. The growth and morphology of microgreens is associated with modified ascorbate and anthocyanin profiles in response to the intensity of sole-source light-emitting diodes. Can. J. Plant Sci. 2020, 101, 212–228.
- Ying, Q., Jones-Baumgardt, C., Zheng, Y., Bozzo, G. The proportion of blue light from light-emitting diodes alters microgreen phytochemical profiles in a species-specific manner. HortScience 2020, 56, 1–8.
- Kutschera, U., Briggs, W.R. Seedling development in buckwheat and the discovery of the photomorphogenic shade-avoidance response. Plant Biol. 2013, 15, 931–940.
- Gommers, C.M., Visser, E.J., St, K.R., Voesenek, L.A., Pierik, R. Shade tolerance: when growing tall is not an option. Trends Plant Sci. 2013, 18, 65–71.
- Sheerin, D.J., Hiltbrunner, A. Molecular mechanisms and ecological function of far-red light signalling. Plant Cell Environ. 2017, 40, 2509–2529.
- Yang, C., Xie, F., Jiang, Y., Li, Z., Huang, X., Li, L. Phytochrome A negatively regulates the shade avoidance response by increasing auxin/indole acidic acid protein stability. Dev. Cell. 2018, 44, 29–41.
- Fankhauser, C., Chory, J. Light control of plant development. Annu Rev. Cell Dev. Biol. 1997, 13, 203–229.
- Seluzicki, A., Burko, Y., Chory, J. Dancing in the dark: darkness as a signal in plants. Plant Cell Environ. 2017, 40, 2487–2501.
- Von Arnim, A., Deng, X.W. Light control of seedling development. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 215–243.
| Spectrum treatments | Temperature (°C) | Relative Humidity (%) | ||||
|---|---|---|---|---|---|---|
| Rep 1 | Rep 2 | Rep 3 | Rep 1 | Rep 2 | Rep 3 | |
| R | 21.2 ± 0.22 | 21.1 ± 0.19 | 21.0 ± 0.20 | 74.6 ± 0.73 | 73.5 ± 0.73 | 72.9 ± 0.89 |
| UVA | 21.1 ± 0.24 | 21.0 ± 0.25 | 20.4 ± 0.28 | 69.6 ± 0.95 | 70.0 ± 1.02 | 76.0 ± 1.10 |
| B1 | 21.0 ± 0.29 | 21.0 ± 0.20 | 20.7 ± 0.35 | 72.2 ± 1.05 | 72.0 ± 0.69 | 77.5 ± 1.58 |
| B2 | 21.2 ± 0.20 | 21.0 ± 0.23 | 19.8 ± 0.32 | 73.1 ± 0.69 | 71.4 ± 0.92 | 79.1 ± 1.78 |
| B3 | 21.2 ± 0.23 | 20.9 ± 0.33 | 21.0 ± 0.21 | 71.5 ± 0.54 | 75.6 ±1.62 | 71.9 ± 0.96 |
| FR | 21.1 ± 0.29 | 20.9 ± 0.32 | 20.9 ± 0.18 | 70.8 ± 1.28 | 73.5 ± 1.10 | 75.3 ± 0.94 |
| D | 20.3 ± 0.40 | 20.1 ± 0.32 | 20.9 ± 0.21 | 76.3 ± 1.86 | 76.2 ± 1.84 | 71.1 ± 0.97 |
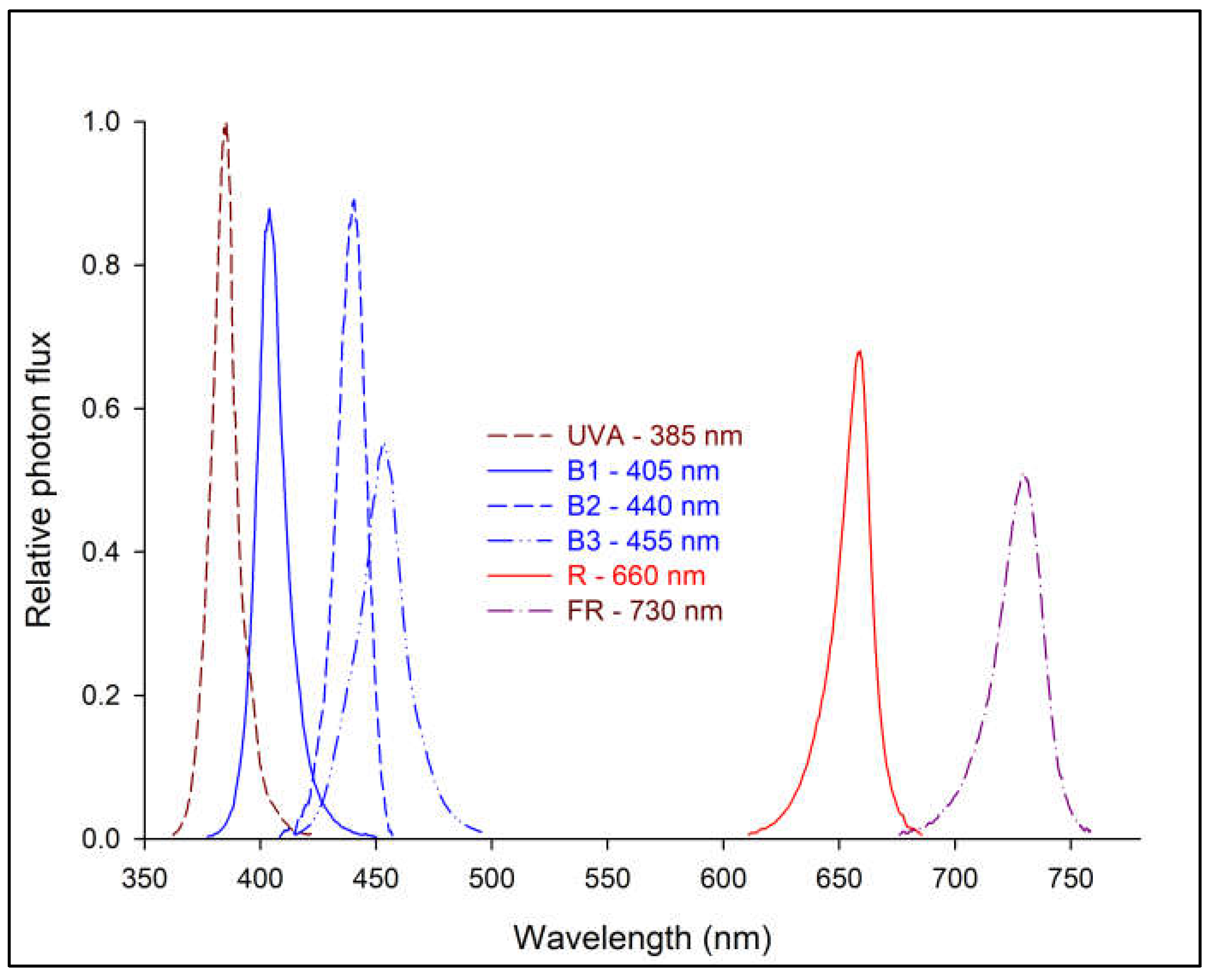
| Spectrum treatment | λpeak (nm) |
FWHM (nm) |
PFD (μmol m–2 s–1) |
PPSz | LED lighting source |
|---|---|---|---|---|---|
| UVA | 385 | 11.5 | 52.0 ± 0.4y | 0.77 | Yunustech Inc., Mississauga, Canada |
| B1 | 405 | 13.6 | 49.9 ± 0.1 | 0.58 | RX30, Heliospectra AB, Gothenburg, Sweden |
| B2 | 440 | 14.6 | 50.7 ± 1.0 | 0.49 | LX601C, Heliospectra AB, Gothenburg, Sweden |
| B3 | 455 | 21.2 | 50.0 ± 0.6 | 0.48 | Pro650, LumiGrow, Emeryville, USA |
| R | 660 | 15.6 | 50.0 ± 0.4 | 0.89 | LX601C, Heliospectra AB, Gothenburg, Sweden |
| FR | 730 | 21.9 | 49.5 ± 0.5 | 0.16 | LX601C, Heliospectra AB, Gothenburg, Sweden |
| D | / | / | / | / | / |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




