Submitted:
22 April 2023
Posted:
23 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
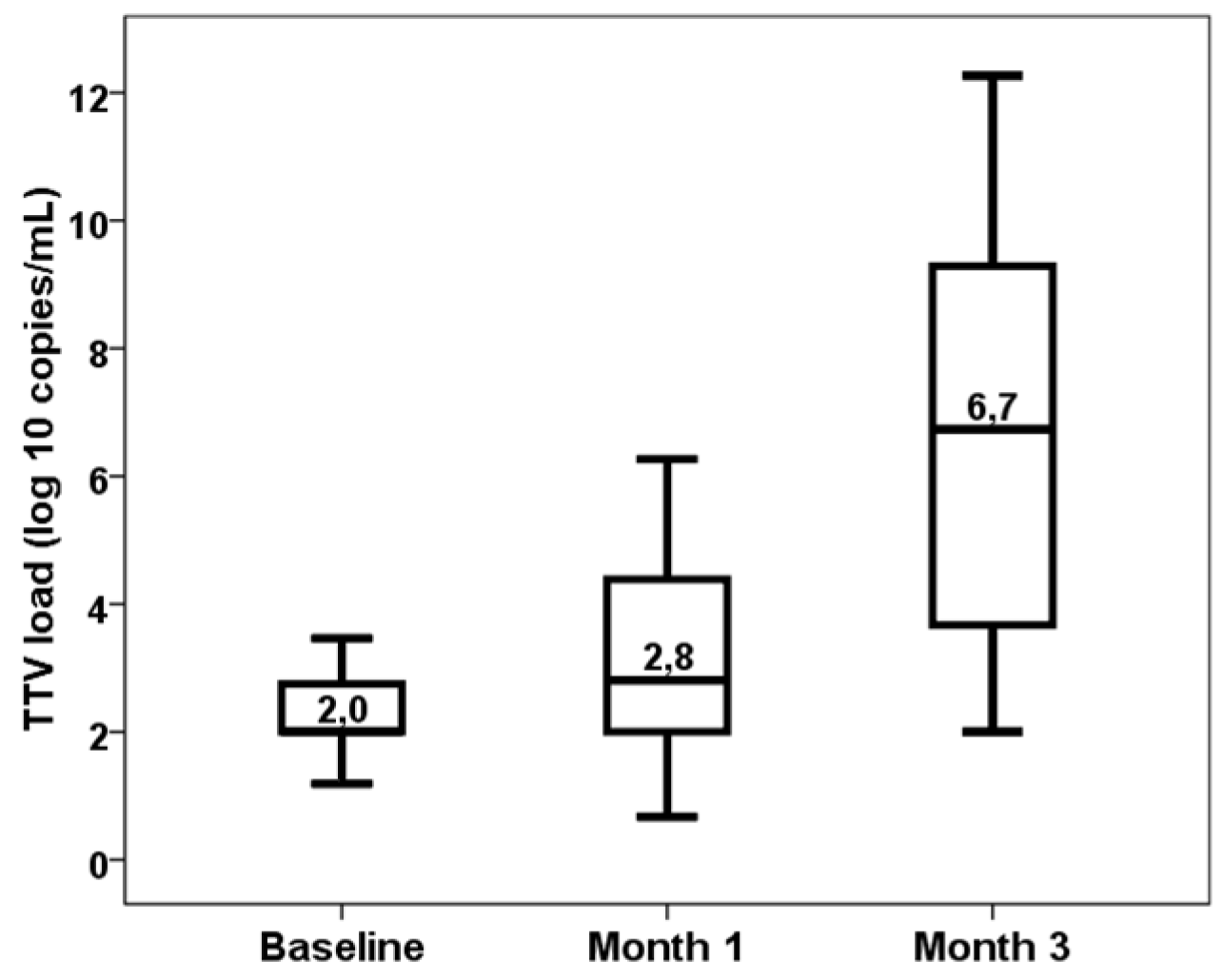
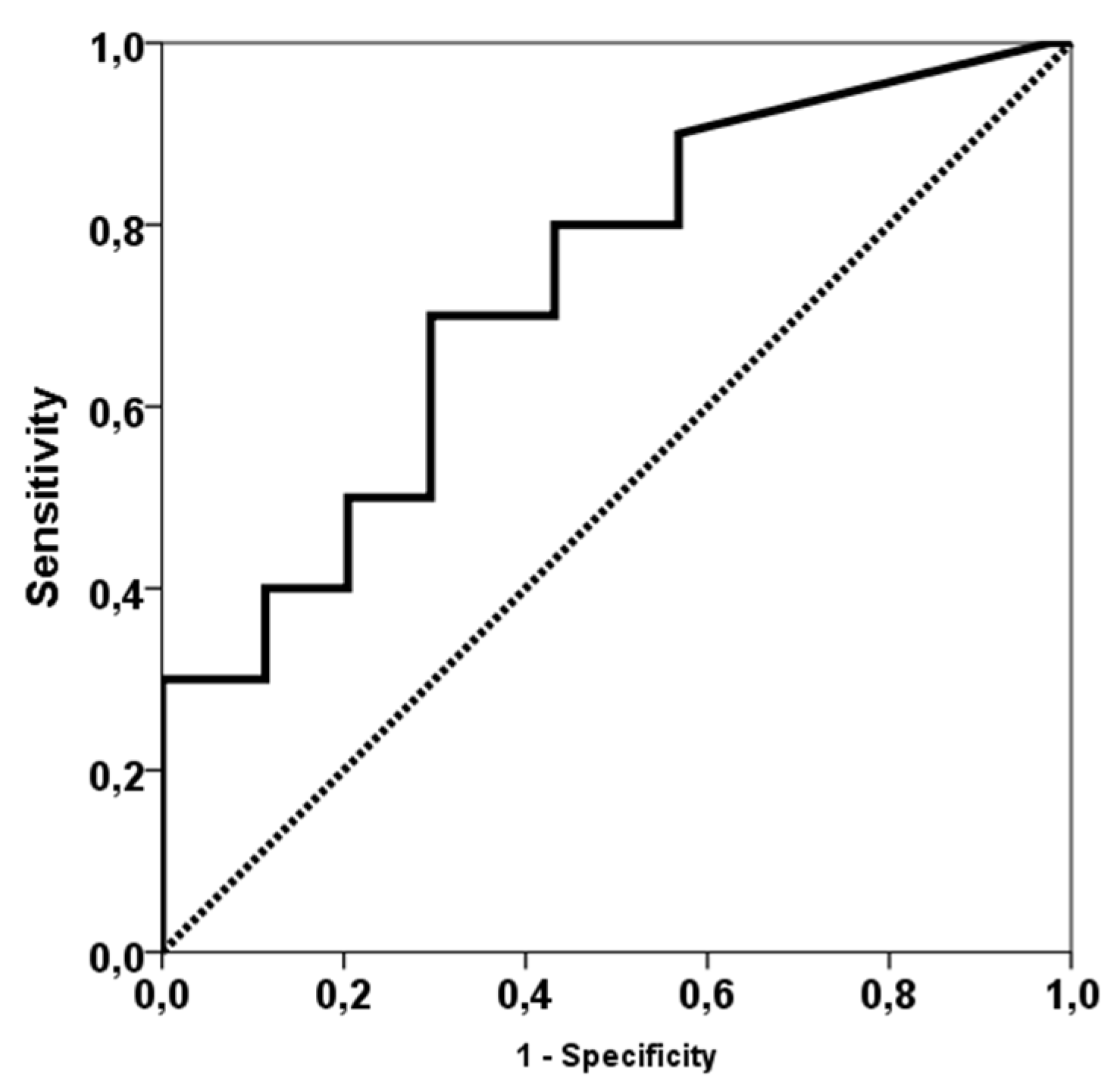
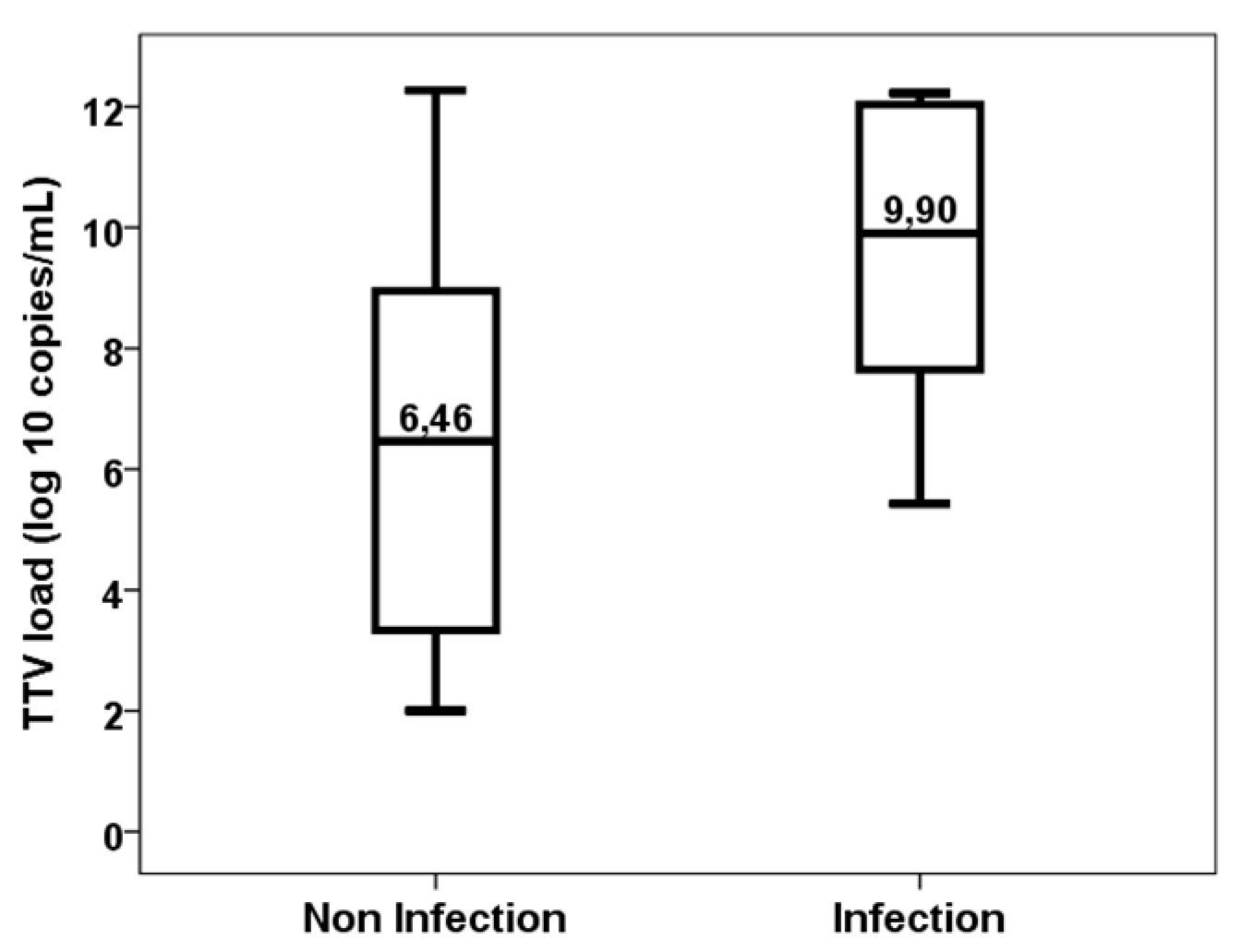
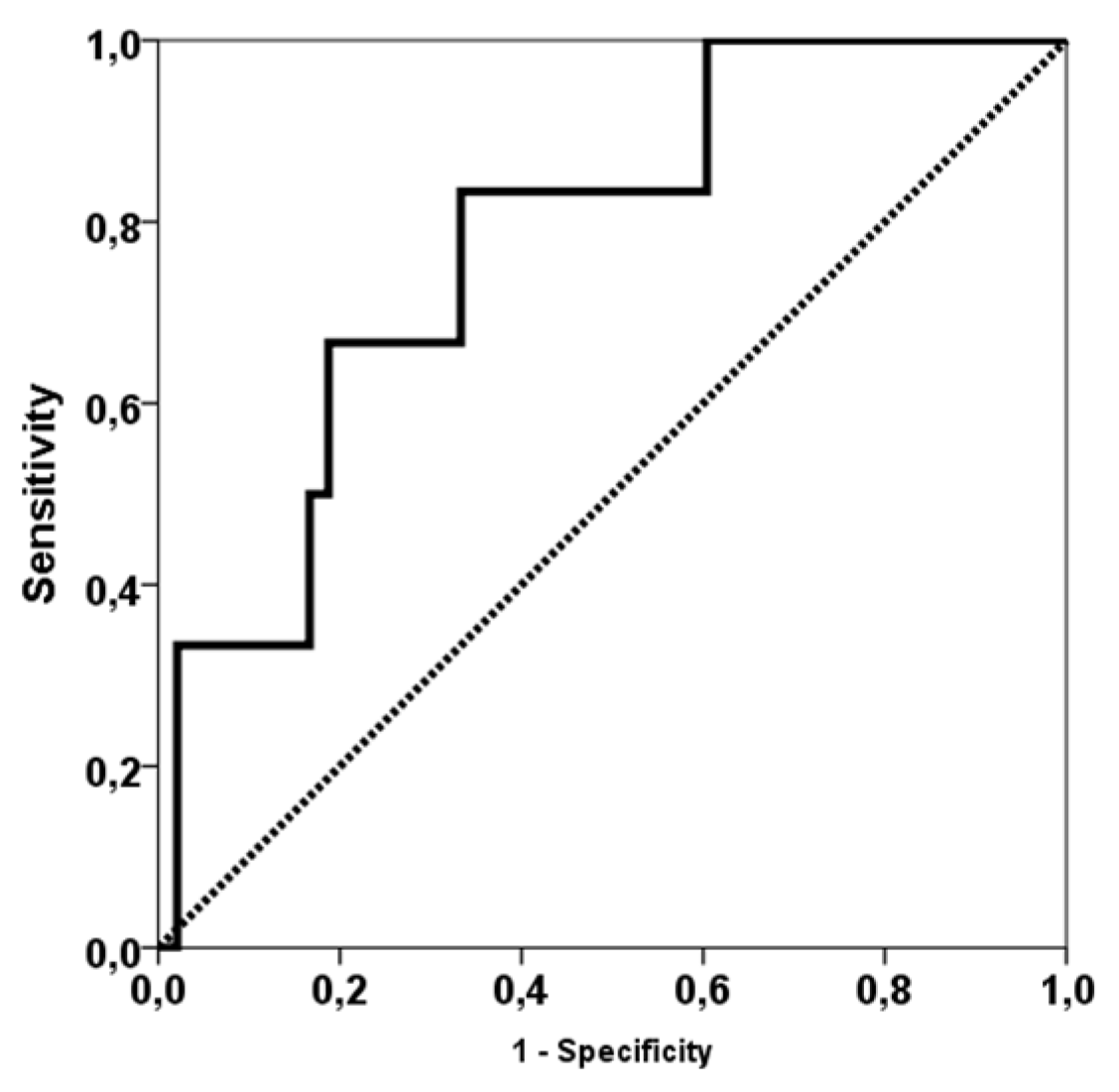
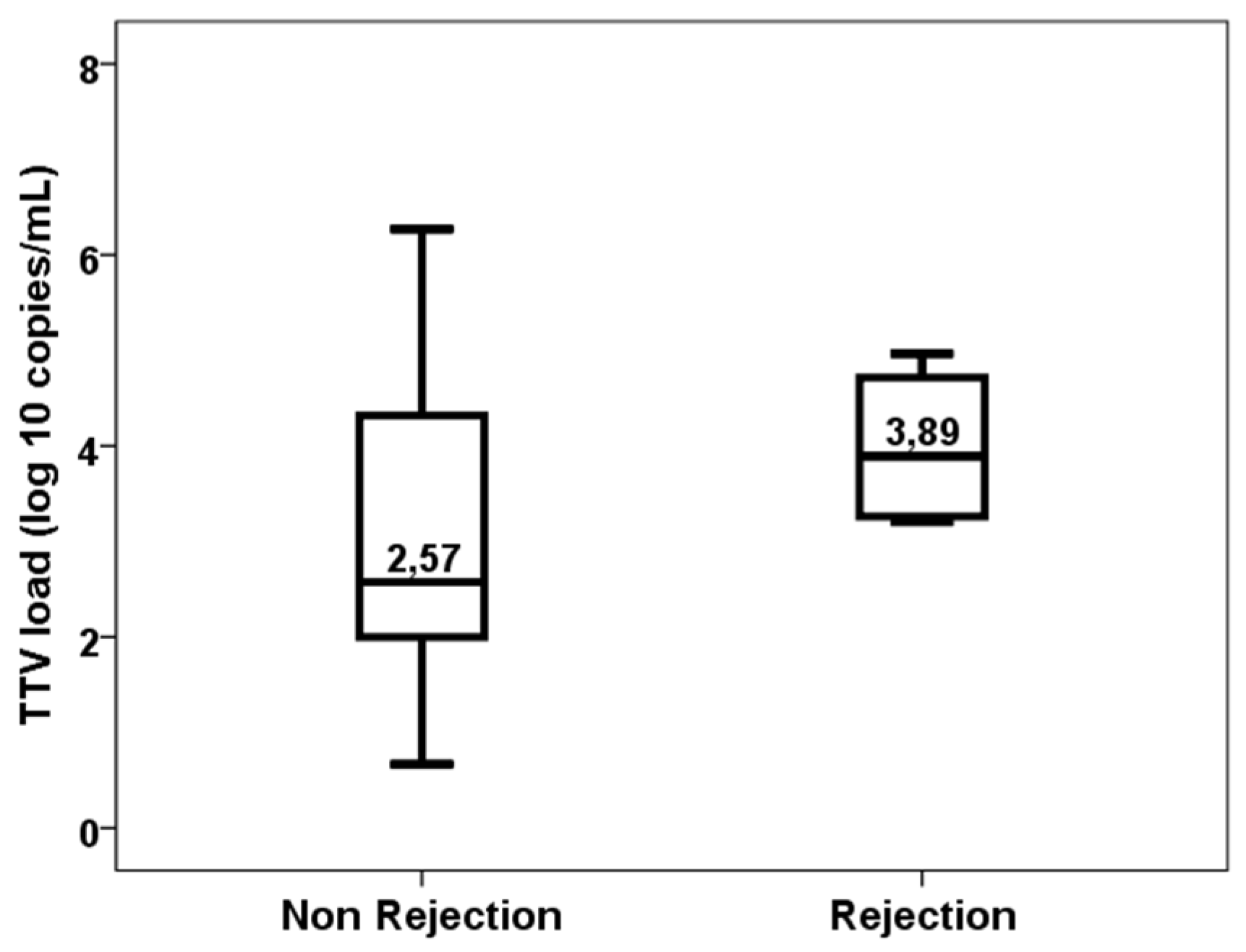
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levitsky, J. Operational tolerance: Past lessons and future prospects. Liver Transpl 2011; 17: 222-32. [CrossRef]
- Dugast E, Soulillou JP, Foucher Y, Papuchon E, Guerif, P., Paul, C.; et al. Failure of Calcineurin Inhibitor (Tacrolimus) Weaning Randomized Trial in Long-Term Stable Kidney Transplant Recipients. Am J Transplant 2016; 16: 3255-3261. [CrossRef]
- Briggs JD. Causes of death after renal transplantation. Nephrol Dial Transplant 2001; 16: 1545-1549. [CrossRef]
- Buell JF, Gross TG, Woodle ES. Malignancy after transplantation. Transplantation 2005; 80(2 Suppl): S254-S264. [CrossRef]
- Woodward RS, Flore MC, Machnicki, G., Brennan DC. The long-term outcomes and costs of diabetes mellitus among renal transplant recipients: Tacrolimus versus cyclosporine. Value Health 2011; 14: 443-449. [CrossRef]
- Mafune A, Tanno Y, Yamamoto H, Kobayashi A, Saigawa H, Yokoo T; et al. A case of BK virus nephropathy and cytomegalovirus infection concurrent with plasma cell-rich acute rejection. Clin Transplant 2012; 26 Suppl 24: 49-53. [CrossRef]
- Bouamar R, Shuker N, Hesselink DA, Weimar W, Ekberg H, Kaplan B; et al. Tacrolimus predose concentrations do not predict the risk of acute rejection after renal transplantation: A pooled analysis from three randomized controlled clinical trials. Am J Transplant 2013; 13: 1253–1261. [CrossRef]
- Percy C, Hassoun Z, Mourad M; et al. Impact of Acute Infection Requiring Hospitalization on Tacrolimus Blood Levels in Kidney Transplant Recipients. Transplant Proc 2017; 49: 2065–2069. [CrossRef]
- Ravaioli M, Neri F, Lazzarotto T, Bertuzzo VR, Di Gioia, P., Stacchini, G.; et al. Immunosuppression Modifications Based on an Immune Response Assay: Results of a Randomized, Controlled Trial. Transplantation 2015; 99: 1625-1632. [CrossRef]
- Jaksch P, Görzer I, Puchhammer-Stöckl, E., Bond, G. Integrated Immunologic Monitoring in Solid Organ Transplantation: The Road Toward Torque Teno Virus-guided Immunosuppression. Transplantation 2022; 106: 1940-1951. [CrossRef]
- Maggi F, Focosi D, Statzu M; et al. Early post-transplant torquetenovirus viremia predicts cytomegalovirus reactivations in solid organ transplant recipients. Sci Rep 2018; 8: 15490. [CrossRef]
- Herrmann A, Sandmann L, Adams O; et al. Role of BK polyomavirus (BKV) and Torque teno virus (TTV) in liver transplant recipients with renal impairment. J Med Microbiol 2018; 67: 1496-1508. [CrossRef]
- Strassl R, Schiemann M, Doberer K, Gorzer I, Puchhammer-Stockl, E., Eskandary, F.; et al. Quantification of torque Teno virus viremia as a prospective biomarker for infectious disease in kidney allograft recipients. J Infect Dis 2018; 218: 1191–1199. [CrossRef]
- Fernandez-Ruiz M, Albert E, Gimenez E; et al. Monitoring of alphatorquevirus DNA levels for the prediction of immunosuppression related complications after kidney transplantation. Am J Transpl 2019; 19: 1139-1149. [CrossRef]
- Fernandez-Ruiz M, Albert E, Gimenez E; et al. Early kinetics of Torque Teno virus DNA load and BK polyomavirus viremia after kidney transplantation. Transpl Infect Dis 2020; 22: e13240. [CrossRef]
- Solis M, Velay A, Gantner P; et al. Torquetenovirus viremia for early prediction of graft rejection after kidney transplantation. J Infect 2019; 79: 56-60. [CrossRef]
- Doberer K, Schiemann M, Strassl, R.; et al. Torque teno virus for risk stratification of graft rejection and infection in kidney transplant recipients—A prospective observational trial. Am J Transpl 2020; 20: 2081-2090. [CrossRef]
- Görzer I, Haupenthal F, Maggi F, Gelas F, Kulifaj D, Brossault, L.; et al. Validation of plasma Torque Teno viral load applying a CE-certified PCR for risk stratification of rejection and infection post kidney transplantation. J Clin Virol 2023; 158: 105348. [CrossRef]
- Schiemann M, Puchhammer-Stockl E, Eskandary F, Kohlbeck P, Rasoul-Rockenschaub, S., Heilos, A.; et al. Torque Teno virus load-inverse association with antibody-mediated rejection after kidney transplantation. Transplantation 2017;101: 360–7. [CrossRef]
- Strassl R, Doberer K, Rasoul-Rockenschaub, S.; et al. Torque teno virus for risk stratification of acute biopsy-proven alloreactivity in kidney transplant recipients. J Infect Dis 2019; 219: 1934-1939. [CrossRef]
- van Rijn AL, Wunderink HF, Sidorov IA; et al. Torque teno virus loads after kidney transplantation predict allograft rejection but not viral infection. J Clin Virol 2021; 140: 104871. [CrossRef]
- Doberer K, Haupenthal F, Nackenhorst M, Bauernfeind F, Dermuth F, Eigenschink.
- M; et al. Torque Teno Virus Load Is Associated With Subclinical Alloreactivity in Kidney Transplant Recipients: A Prospective Observational Trial. Transplantation 2021; 105: 2112-2118. [CrossRef]
- van Rijn AL, Roos R, Dekker FW, Rotmans JI, Feltkamp, M. Torque teno virus load as marker of rejection and infection in solid organ transplantation—A systematic review and meta-analysis. Rev Med Virol 2023; 33: e2393. [CrossRef]
- Görzer I, Haloschan M, Jaksch P, Klepetko W, Puchhammer-Stöckl, E. Plasma DNA levels of Torque teno virus and immunosuppression after lung transplantation. J Heart Lung Transplant 2014; 33: 320-323. [CrossRef]
- Focosi D, Macera L, Boggi U, Nelli LC, Maggi, F. Short-term kinetics of torque Teno virus viraemia after induction immunosuppression confirm T lymphocytes as the main replication-competent cells. J Gen Virol 2015; 96: 115–117. [CrossRef]
- Görzer I, Jaksch P, Kundi M, Seitz T, Klepetko W, Puchhammer-Stöckl, E. Pre-transplant plasma Torque Teno virus load and increase dynamics after lung transplantation. PLoS One 2015; 10: e0122975. [CrossRef]
- Metz DK, Holford N, Kausman JY, Walker A, Cranswick N, Staatz CE, Barraclough KA, Ierino, F. Optimizing Mycophenolic Acid Exposure in Kidney Transplant Recipients: Time for Target Concentration Intervention. Transplantation 2019; 103: 2012-2030. [CrossRef]
- Sharma A, Cherukuri A, Mehta RB, Sood, P., Hariharan, S. High Calcineurin Inhibitor Intrapatient Variability Is Associated With Renal Allograft Inflammation, Chronicity, and Graft Loss. Transplant Direct 2019; 5: e424. [CrossRef]
- Rayar M, Tron C, Jézéquel C, Beaurepaire JM, Petitcollin A, Houssel-Debry P; et al. High Intrapatient Variability of Tacrolimus Exposure in the Early Period After Liver Transplantation Is Associated with Poorer Outcomes. Transplantation 2018; 102: e108-e114. [CrossRef]
- Song T, Yin S, Jiang Y, Huang Z, Liu J, Wang Z; et al. Increasing Time in Therapeutic Range of Tacrolimus in the First Year Predicts Better Outcomes in Living-Donor Kidney Transplantation. Front Immunol 2019; 10: 2912. [CrossRef]
- Jouve T, Fonrose X, Noble J, Janbon B, Fiard G, Malvezzi P; et al. The TOMATO Study (Tacrolimus Metabolization in Kidney Transplantation): Impact of the Concentration-Dose Ratio on Death-censored Graft Survival. Transplantation 2020; 104: 1263-1271. [CrossRef]
- Pawinski T, Luszczynska P, Durlik M, Majchrzak J, Baczkowska T, Chrzanowska M; et al. Development and validation of limited sampling strategies for the estimation of mycophenolic acid area under the curve in adult kidney and liver transplant recipients receiving concomitant enteric-coated mycophenolate sodium and tacrolimus. Ther Drug Monit 2013; 35: 760-769. [CrossRef]
- Maggi F, Pifferi M, Fornai C, Andreoli E, Tempestini E, Vatteroni, M.; et al. TT virus in the nasal secretions of children with acute respiratory diseases: Relations to viremia and disease severity. J Virol 2003; 77: 2418-2425. [CrossRef]
- Maggi F, Fornai C, Vatteroni ML, Siciliano G, Menichetti F, Tascini, C.; et al. Low prevalence of TT virus in the cerebrospinal fluid of viremic patients with central nervous system disorders. J Med Virol 2001; 65: 418–422. [CrossRef]
- Pistello M, Morrica A, Maggi F, Vatteroni ML, Freer, G., Fornai, C.; et al. TT virus levels in the plasma of infected individuals with different hepatic and extrahepatic pathology. J Med Virol 2001; 63: 189-195.
- Forqué L, Fernández-Ruiz M, Albert E, Giménez E, Monzó C, Chaves, J.; et al. Dynamics of Human Anelloviruses in Plasma and Clinical Outcomes Following Kidney Transplantation. Transplantation 2023; 107: 511-520. [CrossRef]
- Handala L, Descamps V, Morel V, Castelain S, François C, Duverlie, G.; et al. No correlation between Torque Teno virus viral load and BK virus replication after kidney transplantation. J Clin Virol 2019; 116: 4-6. [CrossRef]
- Gore EJ, Gomes-Neto AW, Wang L, Bakker SJL, Niesters HGM, de Joode AAE; et al. Torquetenovirus Serum Load and Long-Term Outcomes in Renal Transplant Recipients. J Clin Med 2020; 9: 440. [CrossRef]
- Kulifaj D, Durgueil-Lariviere B, Meynier F, Munteanu E, Pichon N, Dubé M; et al. Development of a standardized real time PCR for Torque teno viruses (TTV) viral load detection and quantification: A new tool for immune monitoring. J Clin Virol 2018; 105: 118-127. [CrossRef]
- Haupenthal F, Rahn J, Maggi F, Gelas F, Bourgeois P, Hugo C, et al.; TTVguideTX consortium partners. A multicentre, patient- and assessor-blinded, non-inferiority, randomised and controlled phase II trial to compare standard and torque teno virus-guided immunosuppression in kidney transplant recipients in the first year after transplantation: TTVguideIT. Trials 2023; 24: 213. [CrossRef]

| Number of patients | 54 |
| Recipient age (years) | 57.8 ± 12.0 |
| Recipient gender (male) | 74.1% |
| Diabetic nephropathy | 27.8% |
| Time in renal replacement therapy (months) | 49 ± 100 |
| Retransplant | 14.9% |
| Preemptive transplantation | 20.4% |
| Donor age (years) | 54.0 ± 15.7 |
| HLA mismatches | 4.5 ± 1.1 |
| Cold ischemia time (hours) | 19.5 ± 6.6 |
| Induction | 74.1% |
| Thymoglobulin | 25.9% |
| Delayed graft function | 27.8% |
| First year acute rejection | 12 (22.2%) |
| First month eGFR (mL/min/1.73 m2) | 51 ± 21 |
| First month albuminuria (mg/g) | 159 ± 321 |
| Third month eGFR (mL/min/1.73 m2) | 53 ± 21 |
| Third month albuminuria (mg/g) | 105 ± 196 |
| Month 1 Prednisone dose (mg) | 14.1 ± 2.4 |
| TTR 8-12 at month 1 (%) | 45 ± 24% |
| TTR > 12 at month 1 (%) | 49 ± 27% |
| Mean tacrolimus level at month 1 (ng/mL) | 12.7 ± 2.3 |
| Any tacrolimus level < 5 at month 1 | 11.1% |
| Any tacrolimus level < 6 at month 1 | 14.8% |
| Coefficient of variability at month 1 (%) | 29.1 ± 12.2 |
| Tacrolimus trough level/Dose at month 1 | 1.8 ± 0.9 |
| Month 3 Prednisone dose (mg) | 7.4 ± 1.6 |
| TTR 8-12 at month 3 (%) | 59 ± 21% |
| TTR > 12 at month 3 (%) | 30 ± 18% |
| Mean tacrolimus level at month 3 (ng/mL) | 11.6 ± 1.4 |
| Any tacrolimus level < 5 at month 3 | 14.8% |
| Any tacrolimus level < 6 at month 3 | 27.8% |
| CV at month 3 (%) | 31.1 ± 10.0 |
| C/D at month 3 | 2.3 ± 1.3 |
| Fast tacrolimus metabolizers | 14.8% |
| AUC-MPA at month 3 (µg x h/mL) | 40.2 ± 14.6 |
| rho | p | |
|---|---|---|
| Recipient age (years) | -0.066 | 0.637 |
| Time in renal replacement therapy (months) | 0.052 | 0.708 |
| Donor age (years) | 0.123 | 0.376 |
| HLA mismatches | -0.168 | 0.228 |
| Cold ischemia time (hours) | 0.088 | 0.526 |
| First month eGFR (mL/min/1.73 m2) | -0.207 | 0.134 |
| First month albuminuria (mg/g) | -0.158 | 0.283 |
| Month 1 Prednisone dose (mg) | 0.008 | 0.955 |
| TTR 8-12 at month 1 (%) | -0.060 | 0.667 |
| TTR > 12 at month 1 (%) | -0.012 | 0.931 |
| Mean tacrolimus level at month 1 (ng/mL) | -0.070 | 0.617 |
| CV at month 1 (%) | -0.016 | 0.909 |
| C/D at month 1 | -0.142 | 0.306 |
| Mycophenolic acid trough level at month 1 | 0.223 | 0.116 |
| rho | p | |
|---|---|---|
| Recipient age (years) | -0.066 | 0.637 |
| Time in renal replacement therapy (months) | 0.052 | 0.708 |
| Donor age (years) | 0.328 | 0.015 |
| HLA mismatches | -0.168 | 0.228 |
| Cold ischemia time (hours) | -0.014 | 0.919 |
| Third month eGFR (mL/min/1.73 m2) | -0.113 | 0.426 |
| Third month albuminuria (mg/g) | 0.000 | 0.999 |
| Month 3 Prednisone dose (mg) | 0.215 | 0.125 |
| TTR 8-12 at month 3 (%) | -0.211 | 0.125 |
| TTR > 12 at month 3 (%) | 0.062 | 0.656 |
| Mean tacrolimus level at month 3 (ng/mL) | -0.070 | 0.615 |
| CV at month 3 (%) | 0.105 | 0.451 |
| C/D at month 3 | -0.026 | 0.855 |
| AUC-MPA at month 3 | 0.060 | 0.680 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




