Submitted:
18 April 2023
Posted:
19 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
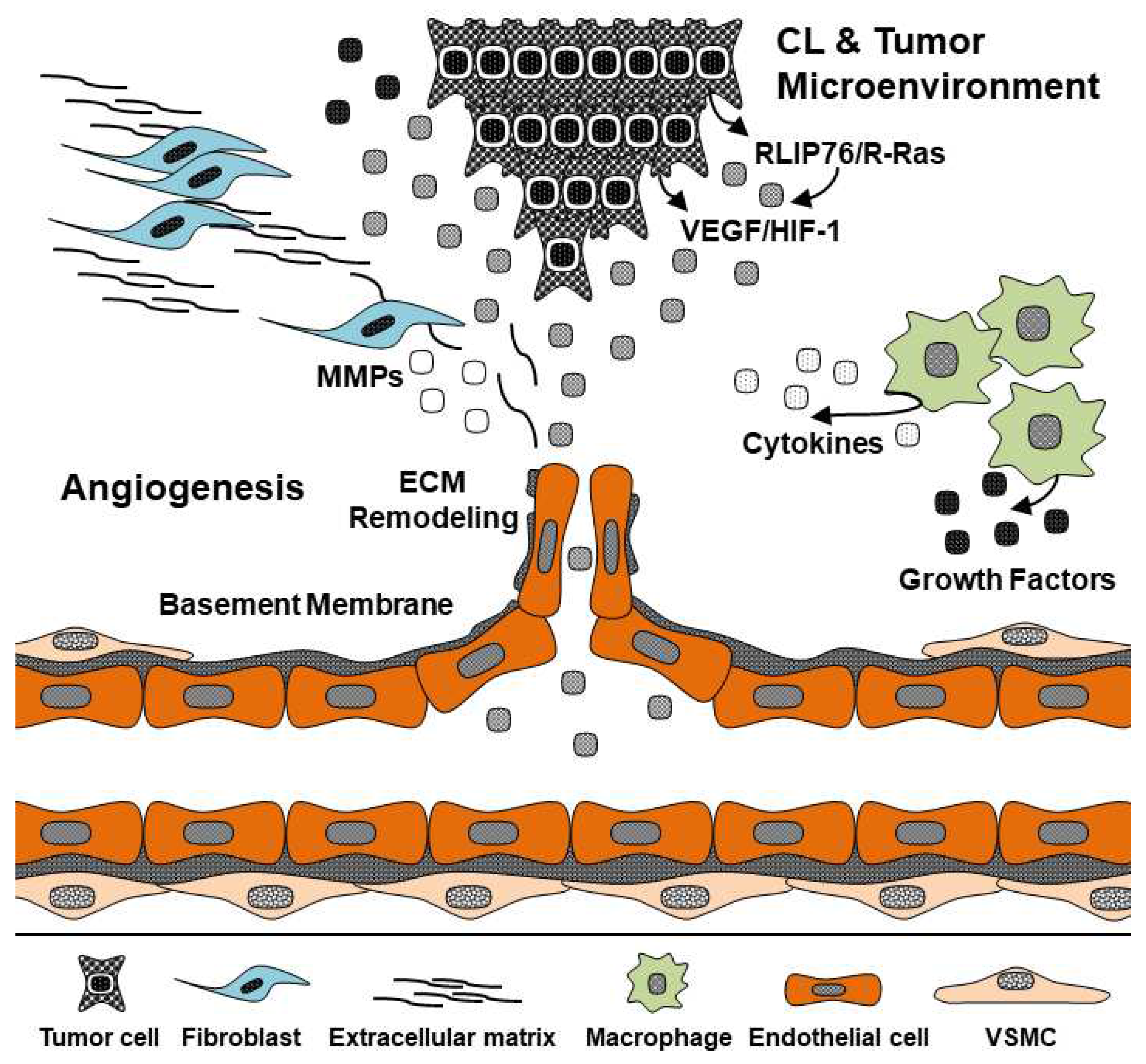
2. RLIP76 in the Tumor Microenvironment
3. RLIP76 and VEGF are Required in Tumor Angiogenesis
4. Microenvironment in Ovarian Corpus Luteum
5. Angiogenesis in the Ovarian Corpus Luteum
6. The Function of RLIP76 and VEGF in the Ovarian Corpus Luteum
7. Conclusion
8. Future Directions
Author Contributions
Funding
Ethics approval and consent to participate
Consent for publication
Availability of data and materials
Acknowledgments
Conflicts of Interest
List of Abbreviations
References
- Telfer:, E.E.; Anderson, R.A. The existence and potential of germline stem cells in the adult mammalian ovary. Climacteric 2019, 22, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Schams, D.; Berisha, B. Regulation of corpus luteum function in cattle - An overview. Reprod. Domest. Anim. 2004, 394, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Devoto, L.; Fuentes, A.; Kohen, P.; Cespedes, P.; Palomino, A.; Pommer, R.; Munoz, A.; Strauss, J.F., III. The human corpus luteum: life cycle and function in natural cycles. Fertil. Steril. 2009, 92, 1067–1079. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, J.; Qi, J.; Zhang, J.; Zhu, Q.; Qin, C. RLIP76 decreases apoptosis through Akt/mTOR signaling pathway in gastric cancer. Oncol. Rep. 2016, 36, 2216–2224. [Google Scholar] [CrossRef]
- Sawada, J.; Urakami, T.; Li, F.; Urakami, A.; Zhu, W.; Fukuda, M.; Li, D.Y.; Ruoslahti, E.; Komatsu, M. Small GTPase R-Ras regulates integrity and functionality of tumor blood vessels. Cancer cell 2012, 22, 235–249. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, S. Change of Ras and its guanosine triphosphatases during development and regression in bovine corpus luteum. Theriogenology 2020, 144, 16–26. [Google Scholar] [CrossRef]
- Lee, S.; Barbe, M.F.; Scalia, R.; Goldfinger, L.E. Three-Dimensional Reconstruction of Neovasculature in Solid Tumors and Basement Membrane Matrix Using Ex Vivo X-ray Microcomputed Tomography. Microcirculation 2014, 21, 159–170. [Google Scholar] [CrossRef]
- Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002, 29, 15–18. [Google Scholar] [CrossRef]
- Gupta, K.; Zhang, J. Angiogenesis: a curse or cure? Postgrad. Med. J. 2005, 81, 236–242. [Google Scholar] [CrossRef]
- Pandya, N.M.; Dhalla, N.S.; Santani, D.D. Angiogenesis—a new target for future therapy. Vasc. Pharmacol. 2006, 44, 265–274. [Google Scholar] [CrossRef]
- Lee, S.H.; Wurtzel, J.G.T.; Singhal, S.S.; Awasthi, S.; Goldfinger, L.E. ; RALBP1/RLIP76 depletion in mice suppresses tumor growth by inhibiting tumor neovascularization. Cancer Res. 2012, 72, 5165–5173. [Google Scholar] [CrossRef] [PubMed]
- Mollberg, N.M.; Steinert, G.; Aigner, M.; Hamm, A.; Lin, F.J.; Elbers, H.; Reissfelder, C.; Weitz, J.; Buchler, M.W.; Koch, M. Overexpression of RalBP1 in colorectal cancer is an independent predictor of poor survival and early tumor relapse. Cancer Biol. Ther. 2012, 13, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.S.; Awasthi, Y.C.; Awasthi, S. Regression of melanoma in a murine model by RLIP76 depletion. Cancer Res. 2006, 66, 2354–2360. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.S.; Singhal, J.; Nair, M.P.; Lacko, A.G.; Awasthi, Y.C.; Awasthi, S. Doxorubicin transport by RALBP1 and ABCG2 in lung and breast cancer. Int. J. Oncol. 2007, 30, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.S.; Singhal, J.; Yadav, S.; Dwivedi, S.; Boor, P.J.; Awasthi, Y.C.; Awasthi, S. Regression of lung and colon cancer xenografts by depleting or inhibiting RLIP76 (Ral-binding protein 1). Cancer Res. 2007, 67, 4382–4389. [Google Scholar] [CrossRef]
- Sharma, R.; Gupta, S.; Singh, S.V.; Medh, R.D.; Ahmad, H.; LaBelle, E.F.; Awasthi, Y.C. Purification and characterization of dinitrophenylglutathione ATPase of human erythrocytes and its expression in other tissues. Biochem. Biophys. Res. Commun. 1990, 171, 155–161. [Google Scholar] [CrossRef]
- Awasthi, S.; Singhal, S.S.; Pikula, S.; Piper, J.T.; Srivastava, S.K.; Torman, R.T.; Bandorowicz-Pikula, J.; Lin, J.T.; Singh, S.V.; Zimniak, P.; et al. ATP-dependent human erythrocyte glutathione-conjugate transporter. II. Functional reconstitution of transport activity. Biochemistry 1998, 37, 5239–5248. [Google Scholar] [CrossRef]
- Awasthi, S.; Singhal, S.S.; Srivastava, S.K.; Torman, R.T.; Zimniak, P.; Bandorowicz-Pikula, J.; Singh, S.V.; Piper, J.T.; Awasthi, Y.C.; Pikula, S. ATP-Dependent human erythrocyte glutathione-conjugate transporter. I. Purification, photoaffinity labeling, and kinetic characteristics of ATPase activity. Biochemistry. 1998, 37, 5231–5238. [Google Scholar] [CrossRef]
- Awasthi, S.; Cheng, J.; Singhal, S.S.; Saini, M.K.; Pandya, U.; Pikula, S.; Bandorowicz-Pikula, J.; Singh, S.V.; Zimniak, P.; Awasthi, Y.C. Novel function of human RLIP76: ATP-dependent transport of glutathione conjugates and doxorubicin. Biochemistry 2000, 39, 9327–9334. [Google Scholar] [CrossRef]
- Lim, K.H.; O’Hayer, K.; Adam, S.J.; Kendall, S.D.; Campbell, P.M.; Der, C.J.; Counter, C.M. Divergent roles for RalA and RalB in malignant growth of human pancreatic carcinoma cells. Curr. Biol. 2006, 16, 2385–2394. [Google Scholar] [CrossRef]
- Jullien-Flores, V.; Dorseuil, O.; Romero, F.; Letourneur, F.; Saragosti, S.; Berger, R.; Tavitian, A.; Gacon, G.; Camonis, J.H. Bridging ral GTPase to Rho pathways RLIP76, a ral effector with CDC42/Rac GTPase-activating protein activity. J. Biol. Chem. 1995, 270, 22473–22477. [Google Scholar] [CrossRef]
- Lim, K.H.; Baines, A.T.; Fiordalisi, J.J.; Shipitsin, M.; Feig, L.A.; Cox, A.D.; Der, C.J.; Counter, C.M. Activation of RalA is critical for Ras-induced tumorigenesis of human cells. Cancer Cell 2005, 7, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Goldfinger, L.E.; Ptak, C.; Jeffery, E.D.; Shabanowitz, J.; Hunt, D.F.; Ginsberg, M.H. RLIP76 (RalBP1) is an R-Ras effector that mediates adhesion-dependent Rac activation and cell migration. J. Cell Biol. 2006, 174, 877–888. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, J.Y.; Zhang, X.P.; Lv, Z.W.; Fu, D.; Lu, Y.C.; Hu, G.H.; Luo, C.; Chen, J.X. RLIP76 is overexpressed in human glioblastomas and is required for proliferation, tumorigenesis and suppression of apoptosis. Carcinogenesis 2012, 34, 916–926. [Google Scholar] [CrossRef]
- Wang, W.; Liu, J.; Qi, J.; Zhang, J.; Zhu, Q.; Qin, C. RLIP76 increases apoptosis through Akt/mTOR signaling pathway in gastric cancer. Oncol. Rep. 2016, 36, 2216–2224. [Google Scholar] [CrossRef] [PubMed]
- Kashatus, D.F.; Lim, K.H.; Brady, D.C.; Pershing, N.L.; Cox, A.D.; Counter, C.M. RALA and RALBP1 regulate mitochondrial fission at mitosis. Nat. Cell Biol. 2011, 13, 1108–1115. [Google Scholar] [CrossRef]
- Singhal, S.S.; Wickramarachchi, D.; Yadav, S.; Singhal, J.; Leake, K.; Vatsyayan, R.; Chaudhary, P.; Lelsani, P.; Suzuki, S.; Yang, S.; et al. Glutathione-conjugate transport by RLIP76 is required for clathrin-dependent endocytosis and chemical carcinogenesis. Mol. Cancer Ther. 2011, 10, 16–28. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Urano, T.; Goi, T.; Feig, L.A. An Eps homology (EH) domain protein that binds to the Ral-GTPase target, RalBP1. J. Biol. Chem. 1997, 272, 31230–31234. [Google Scholar] [CrossRef]
- Margutti, P.; Matarrese, P.; Conti, F.; Colasanti, T.; Delunardo, F.; Capozzi, A.; Garofalo, T.; Profumo, E.; Rigano, R.; Siracusano, A.; et al. Autoantibodies to the C-terminal subunit of RLIP76 induce oxidative stress and endothelial cell apoptosis in immune-mediated vascular diseases and atherosclerosis. Blood 2008, 111, 4559–4570. [Google Scholar] [CrossRef]
- Matarrese, P.; Colasanti, T.; Ascione, B.; Margutti, P.; Franconi, F.; Alessandri, C.; Conti, F.; Riccieri, V.; Rosano, G.; Ortona, E.; et al. Gender disparity in susceptibility to oxidative stress and apoptosis induced by autoantibodies specific to RLIP76 in vascular cells. Antioxid. Redox Signal. 2011, 15, 2825–2836. [Google Scholar] [CrossRef]
- Awasthi, Y.C.; Chaudhary, P.; Vatsyayan, R.; Sharma, A.; Awasthi, S.; Sharma, R. Physiological and pharmacological significance of glutathione-conjugate transport. J. Toxicol. Env. Health-Pt-b-Crit. Rev. 2009, 12, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.Z.; Sharma, R.; Yang, Y.; Singhal, S.S.; Sharma, A.; Saini, M.K.; Singh, S.V.; Zimniak, P.; Awasthi, S.; Awasthi, Y.C. Accelerated metabolism and exclusion of 4-hydroxynonenal through induction of RLIP76 and hGST5. 8 is an early adaptive response of cells to heat and oxidative stress. J. Biol. Chem. 2001, 276, 41213–41223. [Google Scholar] [CrossRef]
- Awasthi, Y.C.; Yang, Y.; Tiwari, N.K.; Patrick, B.; Sharma, A.; Li, J.; Awasthi, S. Regulation of 4-hydroxynonenal-mediated signaling by glutathione S-transferases. Free Radic. Biol. Med. 2004, 37, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Heitzer, T.; Schlinzig, T.; Krohn, K.; Meinertz, T.; Münzel, T. Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation 2001, 104, 2673–2678. [Google Scholar] [CrossRef] [PubMed]
- Jiang, E.; Yan, T.; Xu, Z.; Shang, Z. Tumor microenvironment and cell fusion. BioMed Res. Int. 2019, 5013592. [Google Scholar] [CrossRef]
- Wu, T.; Dai, Y. Tumor microenvironment and therapeutic response. Cancer Lett. 2017, 387, 61–68. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, H.; He, J.; Wang, Q. Functions of Group 2 Innate Lymphoid Cells in Tumor Microenvironment. Front. Immunol. 2019, 10, 1615. [Google Scholar] [CrossRef]
- Lee, S.H.; Goldfinger, L.E. RLIP76 regulates HIF-1 activity, expression and secretion in tumor cells, and secretome transactivation of endothelial cells. Faseb, J. 2014, 28, 4158–4168. [Google Scholar] [CrossRef]
- Cantor, S.B.; Urano, T.; Feig, L.A. Identification and characterization of Ral-binding protein 1, a potential downstream target of Ral GTPases. Mol. Cell. Biol. 1995, 15, 4578–4584. [Google Scholar] [CrossRef]
- Marshall, C.J. Ras effectors. Curr. Opin. Cell Biol. 1996, 8, 197–204. [Google Scholar] [CrossRef]
- Matsubara, K.; Hinoi, T.; Koyama, S.; Kikuchi, A. The post-translational modifications of Ral and Rac1 are important for the action of Ral-binding protein 1, a putative effector protein of Ral. FEBS Lett. 1997, 410, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.L.; Khosravi-Far, R.; Rossman, K.L.; Clark, G.J.; Der, C.J. Increasing complexity of Ras signaling. Oncogene 1998, 1998. 17, 1395–1413. [Google Scholar] [CrossRef]
- Oertli, B.; Han, J.; Marte, B.M.; Sethi, T.; Downward, J.; Ginsberg, M.; Hughes, P.E. The effector loop and prenylation site of R-Ras are involved in the regulation of integrin function. Oncogene 2000, 19, 4961–4969. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, S.; Boissel, L.; Iouzalen, N.; Moreau, J. RLIP mediates downstream signalling from RalB to the actin cytoskeleton during Xenopus early development. Mech. Dev. 2004, 121, 1481–1494. [Google Scholar] [CrossRef] [PubMed]
- Schams, D.; Berisha, B. Regulation of Corpus luteum function in cattle – an overview. Reprod. Domest. Anim. 2004, 39, 241–251. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.D.; Rodgers, R.J.; D’Occhio, M.J. Cellular composition of the cyclic corpus luteum of the cow. J. Reprod. Fertil. 1989, 85, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Stocco, C.; Telleria, C.; Gibori, G. ; The molecular control of corpus luteum formation, function, and regression. Endocr. Rev. 2007, 28, 117–149. [Google Scholar] [CrossRef] [PubMed]
- Xavier, P.; Leão, R.; Oliveira e Silva, P.; Marques Júnior, A. Histological characteristics of the corpus luteum of Nelore cows in the first, second and third trimester of pregnancy. Arq. Bras. Med. Vet. Zoo. 2012, 64, 505–509. [Google Scholar] [CrossRef]
- Stocco, C.; Telleria, C.; Gibori, G. The molecular control of corpus luteum formation, function, and regression. Endocr. Rev. 2007, 28, 117–149. [Google Scholar] [CrossRef]
- Pate, J.L. Regulation of prostaglandin synthesis by progesterone in the bovine corpus luteum. Prostaglandins 1988, 36, 303–315. [Google Scholar] [CrossRef]
- Lee, S.H.; Jung, B.D.; Lee, S. Effect of prostaglandin F2 alpha on E-cadherin, N-cadherin and cell adhesion in ovarian luteal theca cells. Korean, J. Clin. Lab. Sci. 2019, 51, 360–369. [Google Scholar] [CrossRef]
- Lee, S.H.; Acosta, T.J.; Yoshioka, S.; Okuda, K. Prostaglandin F2 alpha regulates the nitric oxide generating system in bovine luteal endothelial cells. J. Reprod. Dev. 2009, 55, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Acosta, T.J.; Nakagawa, Y.; Okuda, K. Role of nitric oxide in the regulation of superoxide dismutase and prostaglandin F2 alpha production in bovine luteal endothelial cells. J. Reprod. Dev. 2010, 56, 454–459. [Google Scholar] [CrossRef]
- Okuda, K.; Sakumoto, R. Multiple roles of TNF super family members in corpus luteum function. Reprod. Biol. Endocrinol. 2003, 1, 95. [Google Scholar] [CrossRef] [PubMed]
- Korzekwa, A.; Lukasik, K.; Skarzynski, D. Leukotrienes are auto-/paracrine factors in the bovine corpus luteum: an in vitro study. Reprod. Domest. Anim. 2010, 45, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Bishop, C.V. Progesterone inhibition of oxytocin signaling in endometrium. Front. Neurosci. 2013, 7, 138. [Google Scholar] [CrossRef]
- Li, X.H.; Matsuura, T.; Xue, M.; Chen, Q.Y.; Liu, R.H.; Lu, J.S.; Shi, W.; Fan, K.; Zhou, Z.; Miao, Z.; Yang, J.; Wei, S.; Wei, F.; Chen, T.; Zhuo, M. Oxytocin in the anterior cingulate cortex attenuates neuropathic pain and emotional anxiety by inhibiting presynaptic long-term potentiation. Cell Rep. 2021, 36, 109411. [Google Scholar] [CrossRef]
- Shirasuna, K.; Shimizu, T.; Hayashi, K.-G.; Nagai, K.; Matsui, M.; Miyamoto, A. Positive Association, in Local Release, of Luteal Oxytocin with Endothelin 1 and Prostaglandin F2alpha during Spontaneous Luteolysis in the Cow: A Possible Intermediatory Role for Luteolytic Cascade Within the Corpus Luteum. Biol. Reprod. 2007, 76, 965–970. [Google Scholar] [CrossRef]
- Lee, S.H.; Billhaq, D.H. A potential function of RLIP76 in the ovarian corpus luteum. J. Ovarian Res. 2019, 12, 34. [Google Scholar]
- Kim, M.; Lee, S.H.; Lee, S.; Kim, G.Y. Expression of Fas and TNFR1 in the luteal cell types isolated from the ovarian corpus luteum. Biomed. Sci. Lett. 2019, 25, 107–112. [Google Scholar] [CrossRef]
- Abulafia, O.; Shere, D.M. Angiogenesis of the ovary. Am. J. Obstet. Gynecol. 2000, 182, 240–246. [Google Scholar] [CrossRef]
- Kim, M.; Lee, S.H.; Lee, S. Expression of H-Ras, RLIP76 mRNA and protein, and angiogenic receptors in corpus luteum tissues during estrous cycles. Korean, J. Clin. Lab. Sci. 2018, 50, 457–461. [Google Scholar] [CrossRef]
- Reynolds, L.P.; Killilea, S.D.; Redmer, D.A. Angiogenesis in the female reproductive system. Faseb, J. 1992, 6, 886–892. [Google Scholar] [CrossRef]
- Schams, D.; Berisha, B. Regulation of corpus luteum function in cattle-an overview. Reprod. Domest. Anim. 2004, 394, 241–251. [Google Scholar] [CrossRef]
- de Tarso, S.G.S.; Gastal, G.D.A.; Bashir, S.T.; Gastal, M.O.; Apgar, G.A.; Gastal, E.L. Follicle vascularity coordinates corpus luteum blood flow and progesterone production. Reprod. Fertil. Dev. 2017, 29, 448–457. [Google Scholar] [CrossRef]
- Xu, M.Q.; Jiang, H.; Zhang, L.Q.; Sun, X.L.; Luo, D.; Fu, Y.; Gao, Y.; Yuan, B.; Zhang, J.B. MiR-29b affects the secretion of PROG and promotes the proliferation of bovine corpus luteum cells. PLoS ONE 2018, 13, e0195562. [Google Scholar] [CrossRef]
- Kfir, S.; Basavaraja, R.; Wigoda, N.; Ben-Dor, S.; Orr, I.; Meidan, R. Genomic profiling of bovine corpus luteum maturation. PLoS ONE 2018, 13, e0194456. [Google Scholar] [CrossRef]
- Ikeda, M.; Ishida, O.; Hinoi, T.; Kishida, S.; Kikuchi, A. Identification and characterization of a novel protein interacting with Ral-binding protein 1, a putative effector protein of Ral. J. Biol. Chem. 1998, 273, 814–821. [Google Scholar] [CrossRef]
- Koshiba, S.; Kigawa, T.; Iwahara, J.; Kikuchi, A.; Yokoyama, S. Solution structure of the Eps15 homology domain of a human POB1 (partner of RalBP1). FEBS Lett. 1999, 442, 138–142. [Google Scholar] [CrossRef]
- Jullien-Flores, V.; Mahé, Y.; Mirey, G.; Leprince, C.; Meunier-Bisceuil, B.; Sorkin, A.; Camonis, J.H. RLIP76, an effector of the GTPase Ral, interacts with the AP2 complex: involvement of the Ral pathway in receptor endocytosis. J. Cell Sci. 2000, 113, 2837–2844. [Google Scholar] [CrossRef]
- Awasthi, S.; Singhal, S.S.; Awasthi, Y.C.; Martin, B.; Woo, J.H. Cunningham CC and Frankel AE: RLIP76 and Cancer. Clin. Cancer Res. 2008, 14, 4372–4377. [Google Scholar] [CrossRef] [PubMed]
- Arbiser, J.L.; Moses, M.A.; Fernandez, C.A.; Ghiso, N.; Cao, Y.; Klauber, N.; Frank, D.; Brownlee, M.; Flynn, E.; Parangi, S.; Byers, H.R.; Folkman, J. Oncogenic H-ras stimulates tumor angiogenesis by two distinct pathways. Proc. Natl. Acad. Sci. USA. 1997, 94, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Maisonpierre, P.C.; Suri, C.; Jones, P.F.; Bartunkova, S.; Wiegand, S.J.; Radziejewski, C.; Compton, D.; McClain, J.; Aldrich, T.H.; Papadopoulos, N.; Daly, T.J.; Davis, S.; Sato, T.N.; Yancopoulos, G.D. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 1997, 277, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Parmar, M.; Yadav, V.; Reshma, R.; Bharati, J.; Bharti, M.; Paul, A.; Chouhan, V.S.; Taru Sharma, G.; Gingh, G.; Sarkar, M. Expression and localization of angiopoietin family in corpus luteum during different stages of oestrous cycle and modulatory role of angiopoietins on steroidogenesis, angiogenesis and survivability of cultured buffalo luteal cells. Reprod. Domest. Anim. 2016, 51, 855–869. [Google Scholar] [CrossRef]
- Hayashi, K.-G.; Acosta, T.J.; Tetsuka, M.; Berisha, B.; Matsui, M.; Schams, D.; Ohtani, M.; Miyamoto, A. Involvement of Angiopoietin-Tie System in Bovine Follicular Development and Atresia: Messenger RNA Expression in Theca Interna and Effect on Steroid Secretion. Biol. Reprod. 2003, 69, 2078–2084. [Google Scholar] [CrossRef] [PubMed]
- Sugino, N.; Suzuki, T.; Sakata, A.; Miwa, I.; Asada, H.; Taketani, T.; Yamagata, Y.; Tamura, H. Angiogenesis in the Human Corpus Luteum: Changes in Expression of Angiopoietins in the Corpus Luteum throughout the Menstrual Cycle and in Early Pregnancy. J. Clin. Endocrinol. Metab. 2005, 90, 6141–6148. [Google Scholar] [CrossRef]
- Ferrara, N.; Chen, H.; Davis-Smyth, T.; Gerber, H.P.; Nguyen, T.N.; Peers, D.; Chisholm, V.; Hillan, K.J.; Schwall, R.H. Vascular endothelial growth factor is essential for corpus luteum angiogenesis. Nat. Med. 1998, 4, 336–340. [Google Scholar] [CrossRef]
- Takahashi, T.; Ueno, H.; Shiuya, M. VEGF activates protein kinase C-dependent, but Ras-independent Raf-MEK-MAP kinase pathway for DNA synthesis in primary endothelial cells. Oncogene 1999, 18, 2221–2230. [Google Scholar] [CrossRef]
- Gecaj, R.M.; Schanzenbach, C.I.; Kirchner, B.; Pfaffl, M.W.; Riedmaier, I.; Tweedie-Cullen, R.Y.; Berisha, B. The Dynamics of microRNA Transcriptome in Bovine Corpus Luteum during Its Formation, Function, and Regression. Fron. Genet. 2017, 8, 213. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



