Submitted:
19 January 2023
Posted:
25 January 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
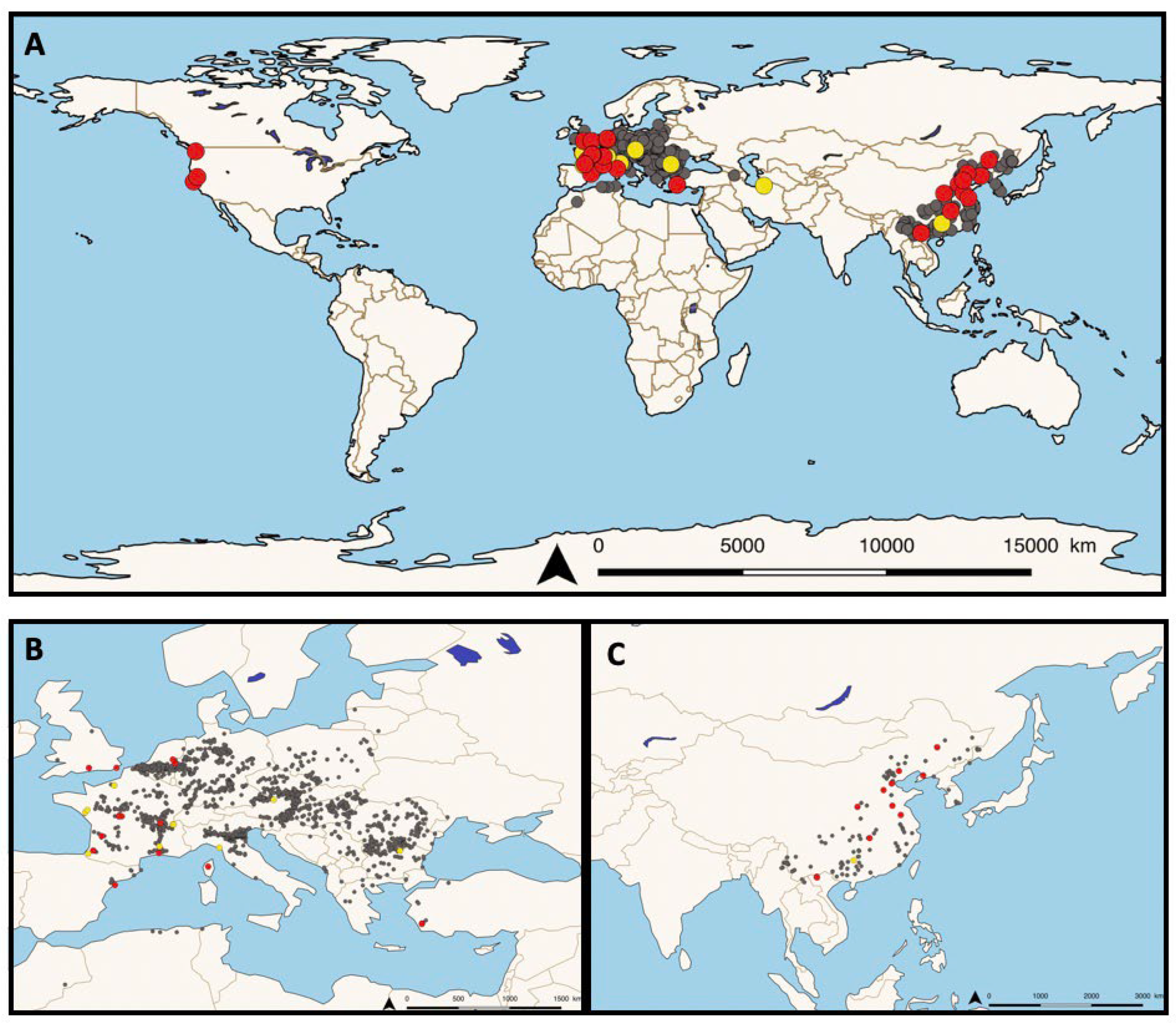
2. Where in The World Has Sphareothecum destruens already Be Localised?
3. Observed Mortalities Associated with The Emergence of S. destruens
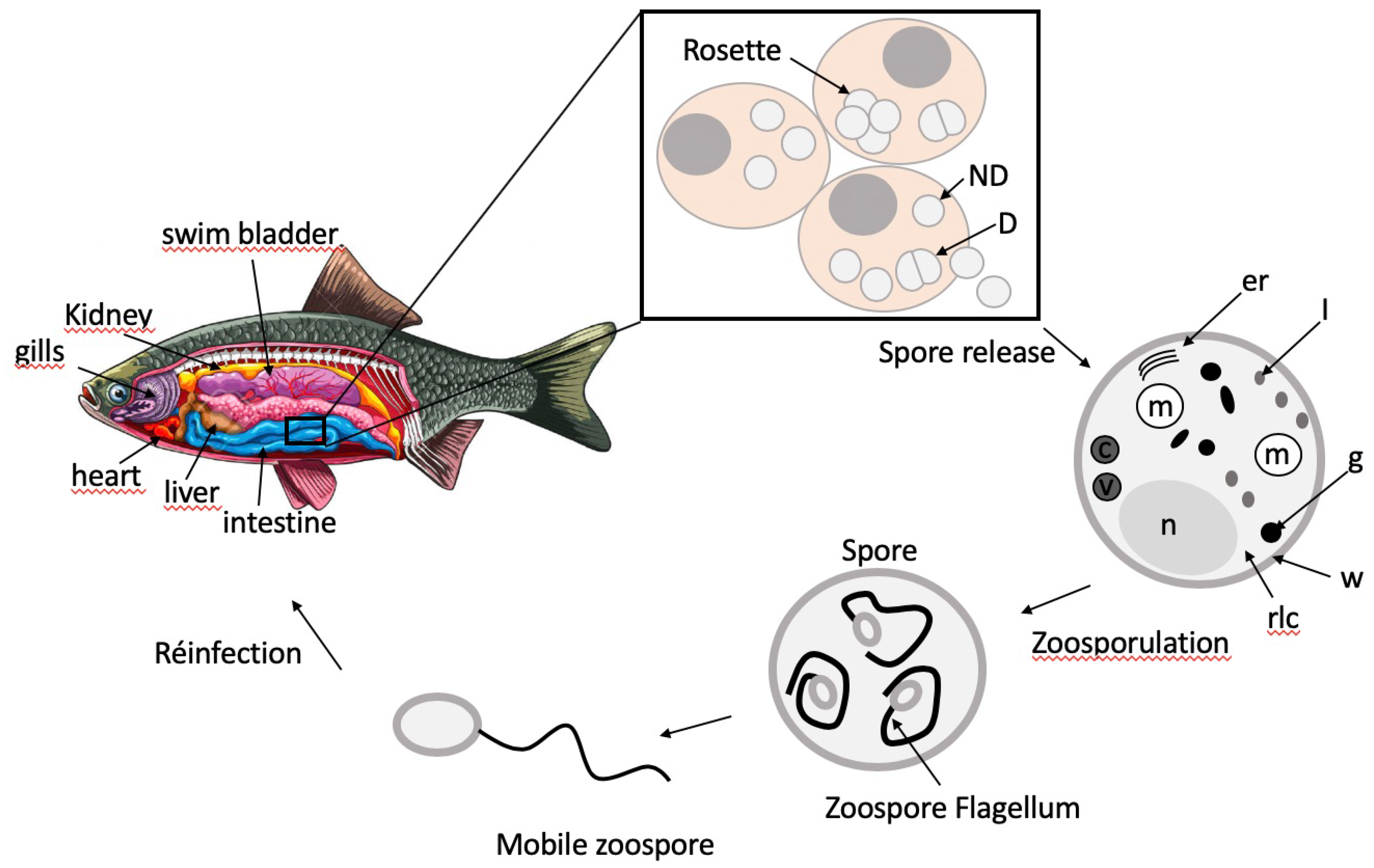
4. Biological Characteristics of S. destruens and Associated Disease Pathology
5. S. destruens Ultrastructure
6. Comparisons with Other Closely Related or Fungal Parasites
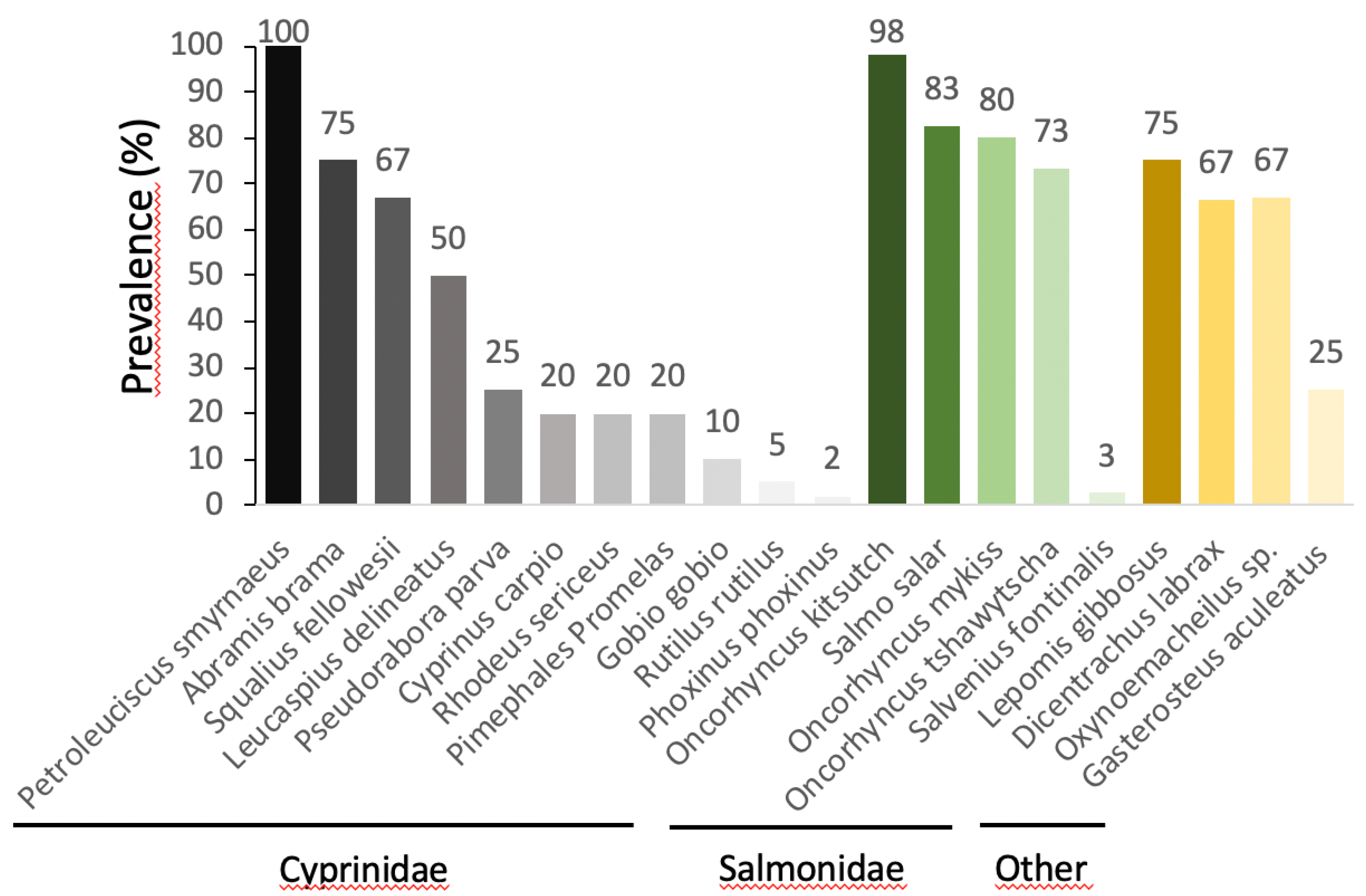
7. Are All Species and Ontogenetic Stages Equally Susceptible to S. destruens?
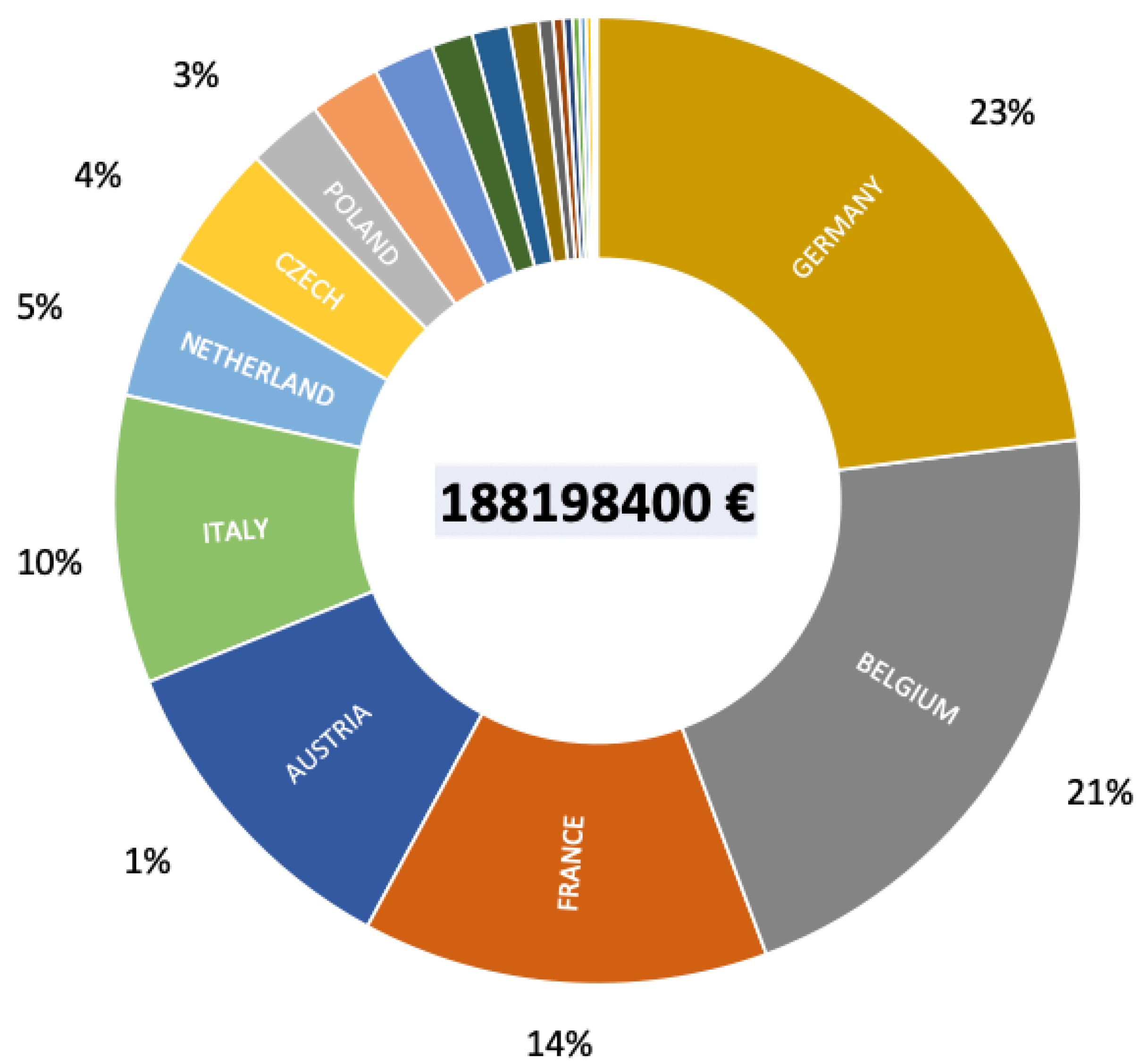
8. What is The Potential Economic Impact of the Emergence of S. destruens?
9. What to Do when The Emergence of S. destruens is Identified in a Water System?
10. Recommendations
Acknowledgments
Conflicts of Interest
References
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef]
- Berger, L.; Speare, R.; Daszak, P.; Green, D.E.; Cunningham, A.A.; Goggin, C.; Slocombe, R.; Ragan, M.A. Hyatt, A.D.; et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl. Acad. Sci. 1998, 95, 9031–9036. [Google Scholar] [CrossRef]
- Longcore, J.E .; Pessier, A.P.; Nichols, D.K. Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia 1999, 91, 219–227. [Google Scholar] [CrossRef]
- Crawford, A.J.; Lips, K.R.; Bermingham, E. Epidemic disease decimates amphibian abundance, species diversity, and evolutionary history in the highlands of central Panama. Proc. Natl Acad. Sci. 2010, 107, 13777–13782. [Google Scholar] [CrossRef]
- Colon-Gaud, C.; Whiles, M.R.; Kilham, S.S.; Lips, K.R.; Pringle, C.M.; Connelly, S.; Peterson, S.D. Assessing ecological responses to catastrophic amphibian declines: Patterns of macroinvertebrate production and food web structure in upland Panamanian streams. Limnol. Oceanogr. 2009, 54, 331–343. [Google Scholar] [CrossRef]
- Stuart, S.N.; Chanson, J.S.; Cox, N.A.; Young, B.E.; Rodrigues, A.S.L.; Fischman, D.L.; Waller, R.W. Status and trends of amphibian declines and extinctions worldwide. Science 2004, 306, 1783–1786. [Google Scholar] [CrossRef]
- Paterson, S.; Vogwill, T.; Buckling, A.; Benmayor, R.; Spiers, A.J.; Thomson, N.R.; Quail, M.; Smith, F.; Walker, D.; et al. Antagonistic coevolution accelerates molecular evolution. Nature 2010, 464, 275–278. [Google Scholar] [CrossRef]
- Murray, A.G.; Peeler, E.D. A framework for understanding the potential for emerging diseases in aquaculture. Preventing Vet. Med. 2005, 67, 223–235. [Google Scholar] [CrossRef]
- FAO The state of world fisheries and aquaculture. Food and agriculture organization of the United Nations, Rome. FAO Fisheries Department. 2002, ISSN 1020-5489.
- Luis, A. I.S.; Campos, E.V.R.; de Oliveira, J.L.; Fraceto, L.F. Trends in aquaculture sciences: from now to use of nanotechnology for disease control. Rev. Aquac. 2019, 11, 119–132. [Google Scholar] [CrossRef]
- Arkush, K.D.; Mendoza, L.; Adkison, M.A.; Hedrick, R.P. Observations on the Life Stages of Sphaerotecum destruens n.g., n. sp., a Mesomycetozoean Fish Pathogen Formally Referred to as the Rosette Agent. J. Eukaryot. Microbiol. 2003, 50, 430–438. [Google Scholar] [CrossRef]
- Combe, M.; Gozlan, R.E. The Rise of the Rosette Agent in Europe: an Epidemiological Enigma. Transbound. Emerg. Dis. 2018, 65, 1474–1481. [Google Scholar] [CrossRef]
- Kerk, D.; Gee, A.; Standish, M.; Wainwright, P.O.; Drum, A.S.; Elston, R.A.; Sogin, M.L. The rosette agent of Chinook salmon (Oncorhynchus tshawytscha) is closely-related to Choanoflagellates, As Determined by the phylogenetic analyses of Its small ribosomal subunit RNA. Mar. Biol. 1995, 122, 187–192. [Google Scholar] [CrossRef]
- Ragan, M.; Goggin, C.L.; Cawthorn, R.J.; Cerenius, L.; Jamieson, A.; Plourde, S.M.; Rand, K.; Söderhäll, R.; Gutell, R. A novel clade of protistan parasites near the animal-fungal divergence. Proc. Natl. Acad. Sci. 1996, 93, 11907–12. [Google Scholar] [CrossRef] [PubMed]
- Cavalier-Smith, T. Neomonada and the origin of animal and fungi. In: Coombs, G. H., Vickerman, K., Sleigh, M. A. & Warren, A. (ed.), Evolutionary Relationships Among Protozoa. Chapman & Hall, London. 1998, p. 375–407.
- Mendoza, L.; Ajello, L.; Taylor, J.W. The taxonomic status of Lacazia loboi and Rhinosporidium seeberi has been finally resolved with the use of molecular tools. Rev. Iberoam. Micol. 2001, 18, 95–98. [Google Scholar] [PubMed]
- Mendoza, L.; Taylor, J.W.; Ajello, L. The Class Mesomycetozoea: A heterogeneous group of microorganisms at the animal-fungal boundary. Annu. Rev. Microbiol. 2002, 56, 315–344. [Google Scholar] [CrossRef]
- Herr, R.A.; Ajello, L.; Taylor, J.W.; Arseculeratne, S.N.; Mendoza, L. Phylogenetic analysis of Rhinosporidium seeberi’s 18S small-subunit ribosomal DNA groups this pathogen among members of the protoctistan Mesomycetozoa clade. J. Clin. Microbiol. 1999, 37, 2750–2754. [Google Scholar] [CrossRef]
- Adl, S.M.; Simpson, A.G.B.; Farmer, M.A.; Andersen, R.A.; Anderson, O.R.; Barta, J. R.; et al. The new higher level classification of eukaryotes with emphasis on the taxonomy of protists. J. Eukaryot. Microbiol. 2005, 52, 399–451. [Google Scholar] [CrossRef]
- Hardouin, E.A.; Andreou, D.; Zhao, Y.; Chevret, P.; Fletcher, D.H. ; Britton, Gozlan, R.E. Reconciling the biogeography of an invader through recent and historic genetic patterns: the case of topmouth gudgeon Pseurorasbora parva. Biol Invasions 2018, 20, 2157–2171. [Google Scholar] [CrossRef]
- Brazier, T.; Cherif, E.; Martin, J-F.; Gilles, A.; Blanchet, S.; Zhao, Y.; Combe, M.; Mccairns R.J.S., Gozlan. R.E. The influence of native populations' genetic history on the reconstruction of invasion routes: the case of a highly invasive aquatic species. Biol. Invasions 2022, 24,, 2399–2420. [Google Scholar] [CrossRef]
- Combe, M.; Cherif, E.; Charrier, A.; Barbey, B.; Chague, M.; Carrel, G.; Chasserieau, C.; Foissy, J.-M.; Gerard, B. Towards unravelling the Rosette agent enigma: Spread and emergence of the co-invasive host-pathogen complex, Pseudorasbora parva-Sphaerothecum destruens. Sci. Total Environ. 2022, 806, 150427. [Google Scholar]
- Harrell, L.W.; Elston, R.A.; Scott, T.M.; Wilkinson, M.T. A significant new systemic disease of net-pen reared chinook salmon (Oncorhynchus tshawytscha) brood stock. Aquaculture 1986, 55, 249–262. [Google Scholar] [CrossRef]
- Elston, R.A.; Harrell, L.; Wilkinson, M.T. Isolation and in vitro characteristics of chinook salmon (Oncorhynchus tshawytscha) rosette agent. Aquaculture 1986, 56, 1–21. [Google Scholar] [CrossRef]
- Hedrick, R.; Friedman, C.; Modin, J. Systemic infection in Atlantic salmon Salmo salar with a Dermocystidium-like species. Dis. Aquat. Organ 1989, 7, 171–177. [Google Scholar] [CrossRef]
- Arkush, K.D.; Frasca, S.; Hedrick, R.P. Pathology associated with the rosette agent, a systemic protist infecting salmonid fishes. J. Aquat. Anim. Health 1998, 10, 1–11. [Google Scholar] [CrossRef]
- Gozlan, R.E.; St-Hilaire, S.; Feist, S.W.; Martin, P.; Kent, M.L. Disease threat to European fish. Nature 2005, 435, 1046. [Google Scholar] [CrossRef] [PubMed]
- Gozlan, R.E.; Whipps, C.M.; Andreou, D.; Arkush, K.D. Identification of a rosette-like agent as Sphaerothecum destruens, a multi-host fish pathogen. Int. J. Parasitol. 2009, 39, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Pollux, B.J.A.; Korosi, A. On the occurrence of the Asiatic cyprinid Pseudorasbora parva in the Netherlands. J. Fish Biol. 2006, 69, 1575–1580. [Google Scholar] [CrossRef]
- Spikmans, F.; van Tongeren, T.; van Alen, T.A.; van der Velde, G.; Op den Camp, H.J.M. High prevalence of the parasite Sphaerothecum destruens in the invasive topmouth gudgeon Pseudorasbora parva in the Netherlands, a potential threat to native freshwater fish. Aquatic Invasions 2013, 8, 355–360. [Google Scholar] [CrossRef]
- Spikmans, F., Lemmers, P., op den Camp, H. J. M., van Haren, E., Kappen, F., Blaakmeer, A., van der Velde G.; van Langevelde F.; Leuven, R.S.E.W.; et al. Impact of the invasive alien topmouth gudgeon (Pseudorasbora parva) and its associated parasite Sphaerothecum destruens on native fish species. Biol. Invasions 2020, 22, 587–601. [CrossRef]
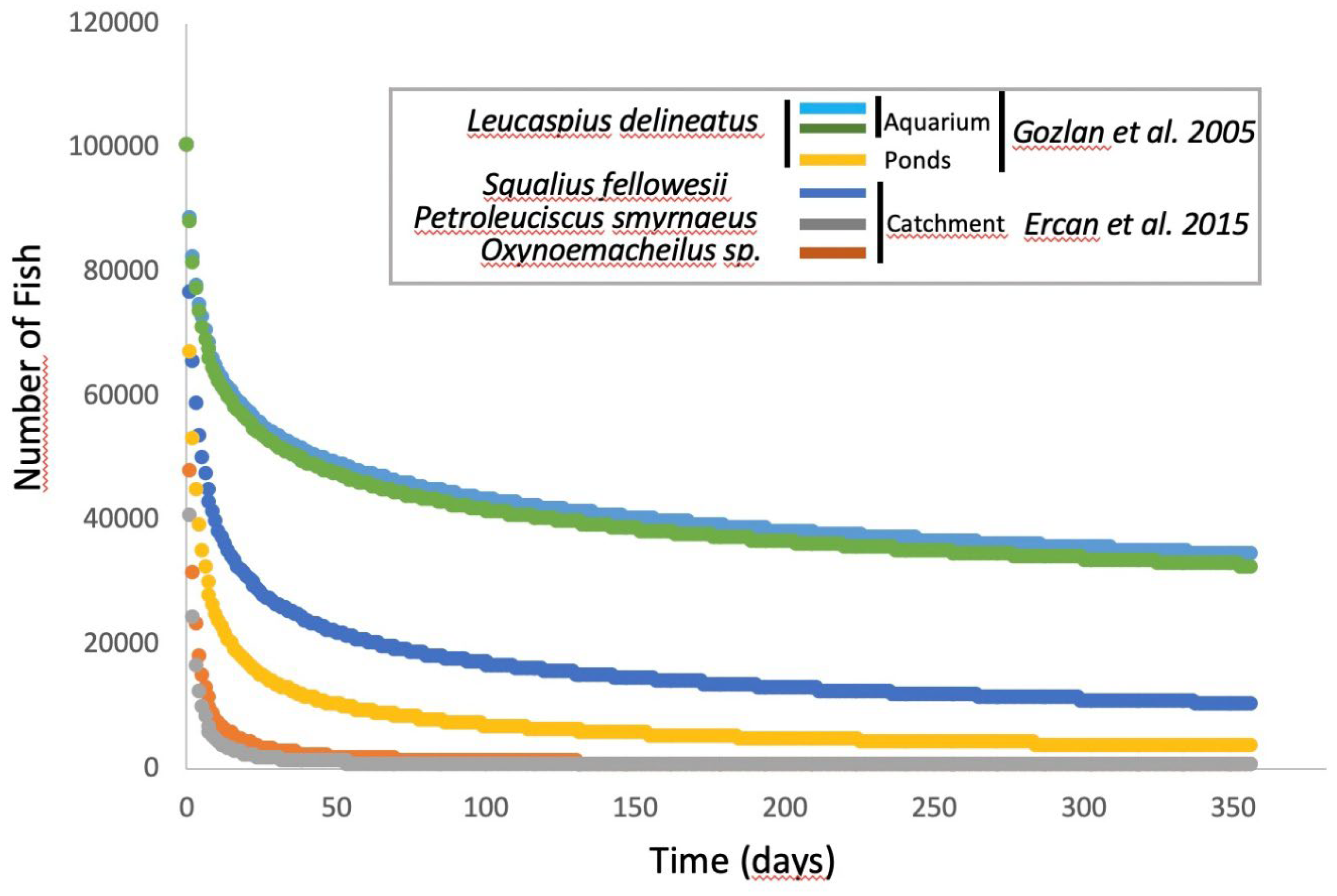
- Ercan, D.; Andreou, D.; Sana, S.; Öntaş, C.; Baba, E.; Top, N.; Karakuş, U.; Tarkan, S.A.; Gozlan, G.E. Evidence of threat to European economy and biodiversity following the introduction of an alien pathogen on the fungal-animal boundary. Emerg. Microbes Infect. 2015, 4, e52. [Google Scholar] [CrossRef]
- Allardi, J.; Chancerel, F. Note ichtyologique. Sur la présence en France de Pseudorasbora parva (Schlegel, 1842). Bull. Fr. Pêche Piscic. 1988, 308, 35–37. [Google Scholar] [CrossRef]
- Poulet, N.; Beaulaton, L.; Dembski, S. Time trends in fish populations in metropolitan France: insights from national monitoring data. J. Fish Biol. 2011, 79, 1436–1452. [Google Scholar] [CrossRef]
- Charrier, A.; Peudpiece, M.; Lesne, M.; Daniel, P. First report of the intracellular fish parasite Sphaerothecum destruens associated with the invasive topmouth gudgeon (Pseudorasbora parva) in France. Knowl. Manag. Aquat. Ecosyst. 2016, 417, 44. [Google Scholar] [CrossRef]
- Boitard, P.M.; Charrier, A.; Labrut, S.; Jamin, M. First detection of sphaerothecum destruens in salmonids in France. Bull. Eur. Assoc. Fish Pathol. 2017, 37, 198–204. [Google Scholar]
- Sana, S.; Hardouin, E.A.; Gozlan, R.E.; Ercan, D.; Tarkan, A.S.; Zhang, T.; Andreou, D. Origin and invasion of the emerging infectious pathogen Sphaerothecum destruens. Emerg. Microbes Infect. 2017, 6, e76. [Google Scholar] [CrossRef]
- Gozlan, R.E. Monitoring fungal infections in fish. Nature 2012, 285, 446. [Google Scholar] [CrossRef]
- Alshorbaji, F.; Gozlan, R.E.; Roche, B.; Britton, J.R.; Andreou, D. The alternate role of direct and environmental transmission in fungal infectious disease in wildlife: threats for biodiversity. Scientific Report 2015, 5, 10368. [Google Scholar] [CrossRef]
- Lelek, A. The Freshwater Fishes of Europe. Volume 9. Threatened Fishes of Europe. AULA-. Verlag, Wiesbaden, 1987, 343 pages.
- Andreou, D.; Arkush, K.D.; Guégan, J-F.; Gozlan, R.E. Introduced pathogens and native freshwater biodiversity: A case study of Sphaerothecum destruens. PLoS One 2012, 7, e36998. [Google Scholar] [CrossRef]
- Olson, R.E.; Dungan, C.F.; Holt, R.A. Water-borne transmission of Dermocystidium salmonis in the laboratory. Dis. Aquat. Org. 1991, 12, 41–48. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Taylor, L.H.; Haydon, D.T. Population biology of multihost pathogens. Science 2001, 292, 1109–1112. [Google Scholar] [CrossRef]
- Andreou, D.; Gozlan, R.E.; Paley, R. Temperature influence on production and longevity of Sphaerothecum destruens zoospores. J. Parasitol. 2009, 95, 1539–1541. [Google Scholar] [CrossRef]
- Andreou, D.; Gozlan, R.E.; Stone, D.; Martin, P.; Bateman, K.; Feist, S.W. Sphaerothecum destruens pathology in cyprinids. Dis. Aquat. Organ. 2011, 95, 145–151. [Google Scholar] [CrossRef]
- Paley, R.K.; Andreou, D.; Bateman, K.S.; Feist, S.W. ; Isolation and culture of Sphaerothecum destruens from Sunbleak (Leucaspius delineatus) in the UK and pathogenicity experiments in Atlantic salmon (Salmo salar). Parasitology 2012, 139, 904–914. [Google Scholar] [CrossRef]
- Lotman, K.; Pekkarinen, M.; Kasesalu, J. Morphological observations on the life cycle of Dermocystidium cyprini Cervinka and Lom, 1974, parasitic in carps (Cyprinus carpio). Acta Protozool. 2000, 39, 125–134. [Google Scholar]
- van der Moer, A.; Manier, J.-F.; Bouix, G. Étude ultrastructural de Dermocystidium macrophagi n. sp. parasite intracellular de Salmo gairdneri Richardson, 1836. Ann. Sci. Nat. Zoo. Biol. Anim. 1986, 8, 143–151. [Google Scholar]
- Nash, G.; Southgate, P.; Richards, R.H.; Sochon, E. A systemic protozoal disease of cultured salmonids. J. Fish Dis. 1989, 12, 157–173. [Google Scholar] [CrossRef]
- McVicar, A.H.; Wootten, R. Disease in farmed juvenile Atlantic salmon caused by Dermocystidium sp. In: Ahne, W. (ed.), Fish Diseases, 3rd COPRAQ session. Springer-Verlag, Berlin. 1980, p. 165–173.
- Arseculeratne, S.N.; Ajello, L. Rhinosporidium seeberi. In: Ajello, L. and Hay, R.J. (ed.), Topley and Wilson's Microbiology and Microbial Infections. Vol. 4. Arnold, London/Sydney/Auckland. 1998, p. 595–643.
- Voyles, J.; Young, S.; Berger, L.; Campbell, C.; Voyles, W.F.; Dinudom, A.; Cook, D.; Webb, R.; Alford, R.A.; et al. Pathogenesis of chytridiomycosis, a cause of catastrophic amphibian declines. Science 2009, 326, 582–585. [Google Scholar] [CrossRef]
- Berger, L.; Hyatt, A.D.; Speare, R.; Longcore, J.E. Life cycle stages of the amphibian chytrid Batrachochytrium dendrobatidis. Dis. Aquat. Org. 2005, 68, 51–63. [Google Scholar] [CrossRef]
- Diagne C.; Leroy B.; Vaissière A-V.; Gozlan R.E.; Roiz D.; Jaric I.; Bradshaw C.J.A.; Courchamp F. High and rising economic costs of biological invasions worldwide. Nature. 2020, 592, 571–576.
- ritton, J.R.; Davies, G.D.; Brazier, M. Towards the successful control of Pseudorasbora parva in the UK. Biological Invasions 2010, 12, 25–31. [Google Scholar]
- Britton, J.R.; Copp, G.H.; Davies, G.D.; Brazier, M. A modular assessment tool for managing introduced fishes according to risks of species and their populations, and impacts of management actions. Biological Invasions 2011, 13, 2847–2860. [Google Scholar] [CrossRef]
- Al-Shorbaji, F.; Roche, B.; Gozlan, R.E.; Britton, J.R.; Andreou, D. The consequences of reservoir host eradication on disease epidemiology in animal communities. Emerging Microbes & Infections 2016, 5, e46. [Google Scholar]
- Al-Shorbaji, F.; Andreou, D.; Roche, B.; Stafford, R.; Britton, J.R.; Gozlan, R.E. The influence of host competition on community resilience to disease. Journal of Animal Ecology. 2017, 86, 1147–1158. [Google Scholar] [CrossRef] [PubMed]
| Fish species | Infected Organs/Tissues | Histopathology of Infected Tissues | RA Dissemination within Tissues | References |
|---|---|---|---|---|
| Chinook salmon (O. tshawytscha) |
Spleen, kidneys | Focal areas of RA spore growth, necrosis | Intracellular localization of spores in macrophages and endothelial cells | [24] |
| Chinook salmon (O. tshawytscha) |
Spleen, kidneys | Edema, focal necrosis | Spherical organisms of 2-7 µm in diameter with a peripheral halo that occurred in cluster “rosette”, organisms accumulate in macrophages, intracellular organisms found within the interstitium parenchyma | [23] |
| Spleen, kidney, liver, gonad, heart, brain, intestinal mucosae | Hepatomegaly, splenomegaly | Organisms observed in peripheral blood and vascular spaces of these organs | ||
| Atlantic salmon (Salmo salar) |
Spleen, kidney, liver, gonads | Widely disseminated nodules, with involvement of hematopoietic tissues | Spores of 2-7 µm in diameter found principally in macrophages but also as cell-free forms | [25] |
| Spleen | Granulomas in splenic and hepatic lesions with macrophages at the periphery of the lesions | |||
| Spleen, kidney, testes | More diffuse granulomatous response | |||
| Winter-run chinook salmon (O. tshawytscha) |
Spleen, kidney, liver, heart, mesentery surrounding the intestinal tract, pyloric cecae |
Nodular form: multifocal granulomas that replaced the normal parenchyma, nodules observed in visceral organs. Granulomas characterized by central cores of eosinophilic necrotic material or closely apposed macrophages | Aggregates of RA found within central zones of granulomas and within macrophages |
[11] |
| Spleen, kidney, liver, heart, gill, brain, ovary, testis, hindgut | Disseminated form: edema, focal necrosis, enlargement and pallor of the spleen kidney, liver | RA spores found in hematopoietic, epithelial and mesenchymal cells, as intracellular or extracellular forms, clusters of 4-5 rosettes | ||
| Kidney | Necrosis of the renal tubular epithelium, loss of tubules, membranous glomerulonephritis, necrotizing interstitial nephritis | Parasite present as single or in aggregates within the cytoplasm of the bilary, renal tubular epithelium, lumina of bile ductules and renal tubules | ||
| Spleen | Necrotizing vasculitis of splenic arterioles | RA spores largely disseminated individually or in aggregates in the pulp spaces, in the cytoplasm of sinusoidal macrophages and reticuloendithelial cells. RA spores found in the lumina and tunicae media of splenic arterioles | ||
| Gills (in early infections) | RA spores found within vessels of the gill | |||
| Swimbladder (in advanced infections) | RA spores found in subserosal aggregates | |||
| Epidermis, urine, seminal and ovarian fluids, intestine mucosa | RA sometimes observed | |||
| Sunbleak (L. delineatus) |
Spleen, kidney, liver, intestine, gonad, eye, adipose tissue surrounding the intestinal tract, skeletal tissue | Nodular form Disseminated form Vacuolar degeneration, necrosis |
RA spores located intracellularly in various types of host cell, including renal tubule and collecting-duct epithelial cells. Presence of spores within giant cells. |
[45] |
| Kidney, testis | Intense inflammation | Numerous stages of RA spore, mostly intracellular in the nodular form, intracellular and extracellular form for disseminated disease, aggregates “rosette” | ||
| Liver | Inflammatory response resulting in an influx of phagocytic cells, lymphocytic infiltration of the hepatic parenchyma. Multifocal granuloma of different size | |||
| Eyes | RA spores within macrophages | |||
| Testis | Multifocal granuloma of different size, necrosis and intense inflammation | |||
| Sunbleak (L. delineatus) |
Kidney, liver, testis, gill | Nodular form: multifocal granulomas in liver and testis Disseminated form: Hepatocellular necrosis of the liver |
Granulomas enclosed different stages of RA spores, 2-4 µm in diameter Intracellular and extracellular RA spores |
[28] |
| Atlantic salmon (Salmo salar) |
Spleen, kidney, liver, heart, choroidal rete, cranial connective tissue | Inflammatory lesions | [46] | |
| Kidney | Granulomatous lesions surrounded by hepatocytes and inflammatory cells | Proliferation of RA spores in haematopoietic tissues, RA spores within macrophages | [46] | |
| Liver | Numerous plaques of pale tissues and extensive inflammation | RA spores of different sizes | [46] | |
| Spleen | Numerous plaques of pale tissues and extensive inflammation | [46] | ||
| Sunbleak (L. delineatus) |
Pancreatitis, severe inflammation of the spleen and renal interstitial haematopoietic tissue | Intense proliferation of RA spores | [46] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




