Submitted:
29 December 2022
Posted:
10 January 2023
You are already at the latest version
Abstract
Keywords:
2. Materials and Methods
2.1. Genetic Material
2.2. Field Study Area
2.3. Postharvest Study Area and Sampling
2.3.1. Parameters Recorded2.3.2. Dry matter and moisture content
2.3.2. Extraction and quantification of non-structural sugars
2.3.3. Total sugar estimation
2.3.4. Reducing and non-reducing sugars estimation
2.3.5. Amylose and Amylopectin
3. Results
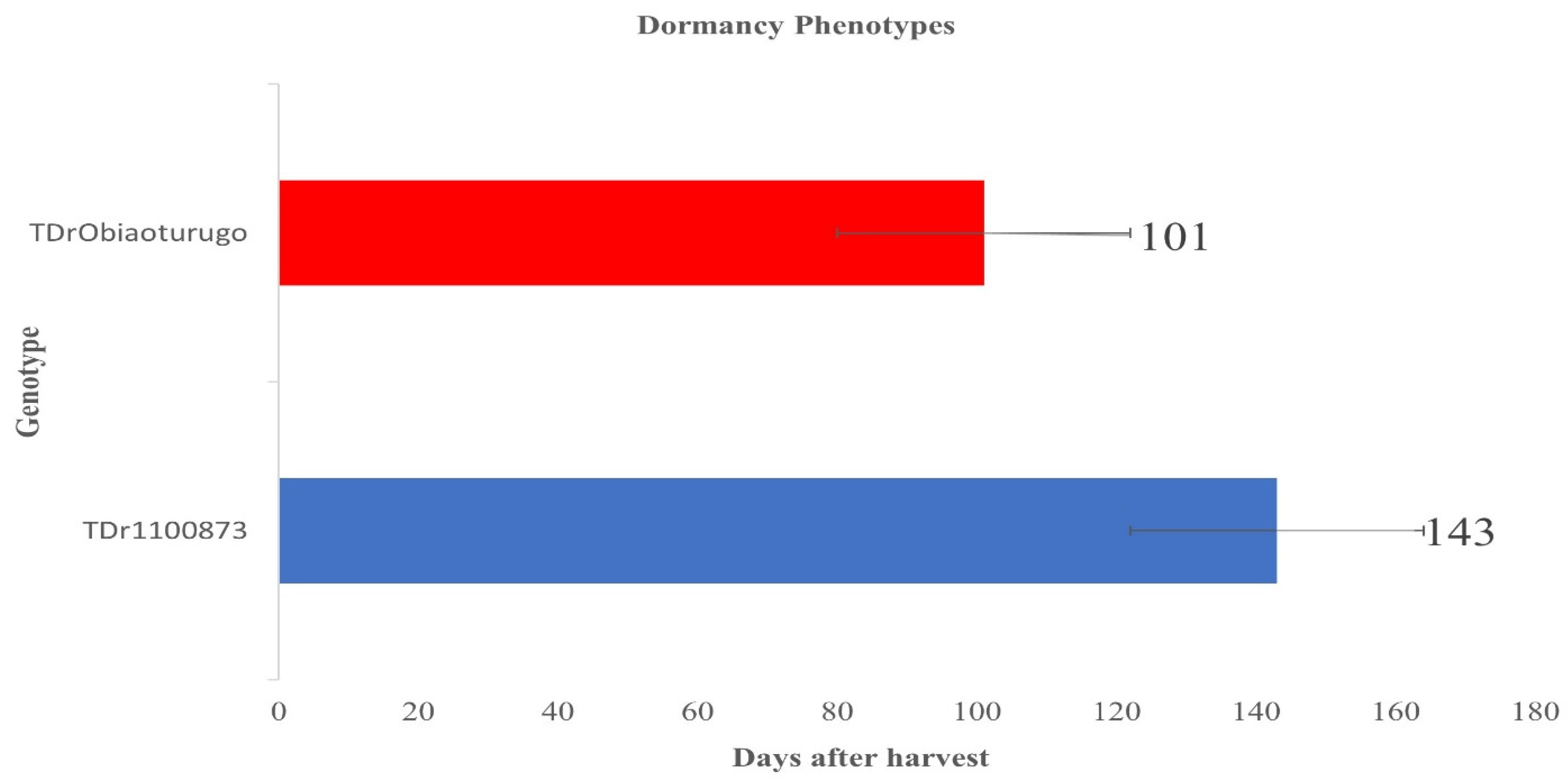
3.1. Tuber dormancy duration
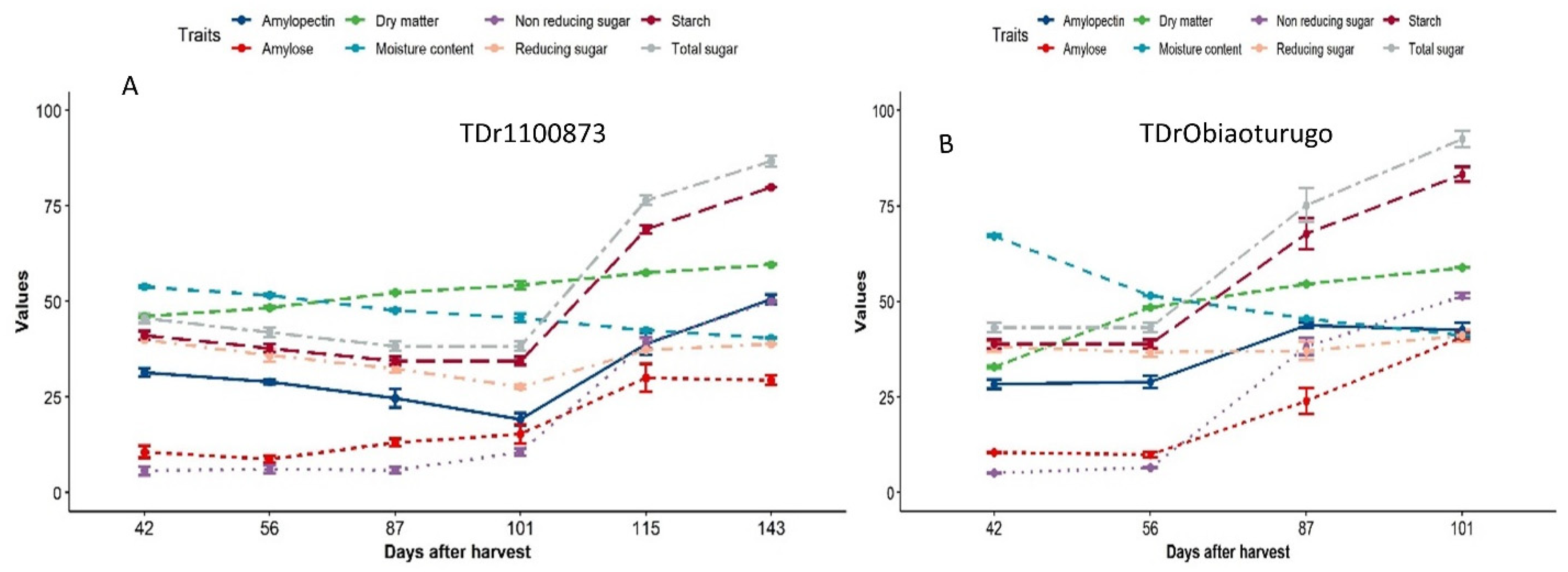
3.2. An overview changes in the metabolic status of non-structural sugar, dry matter and moisture content of white yam tubers during dormancy
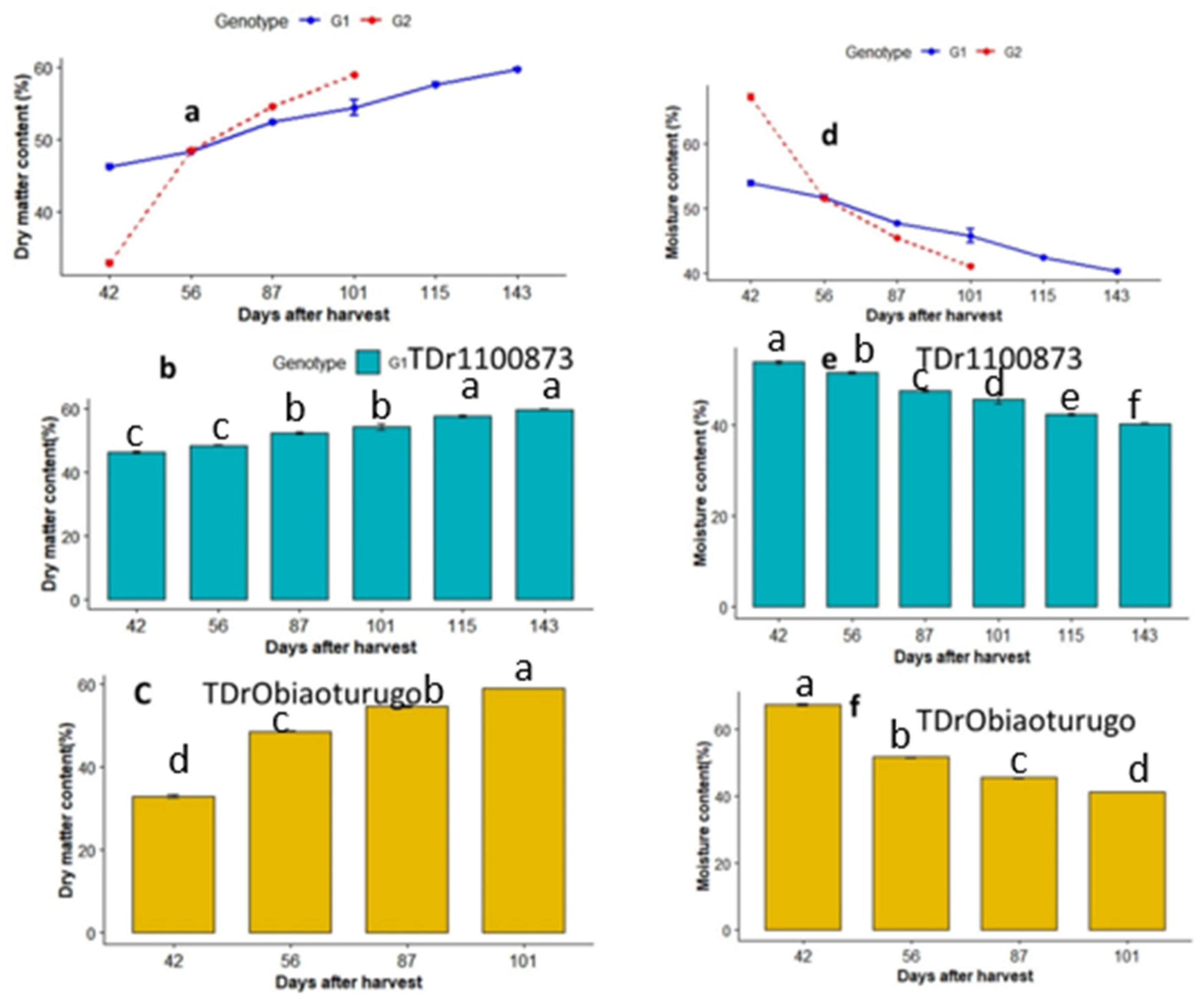
3.3. Dry matter and moisture content
3.4. Metabolic status of non-structural sugar in white yam tubers from dormancy to sprouting
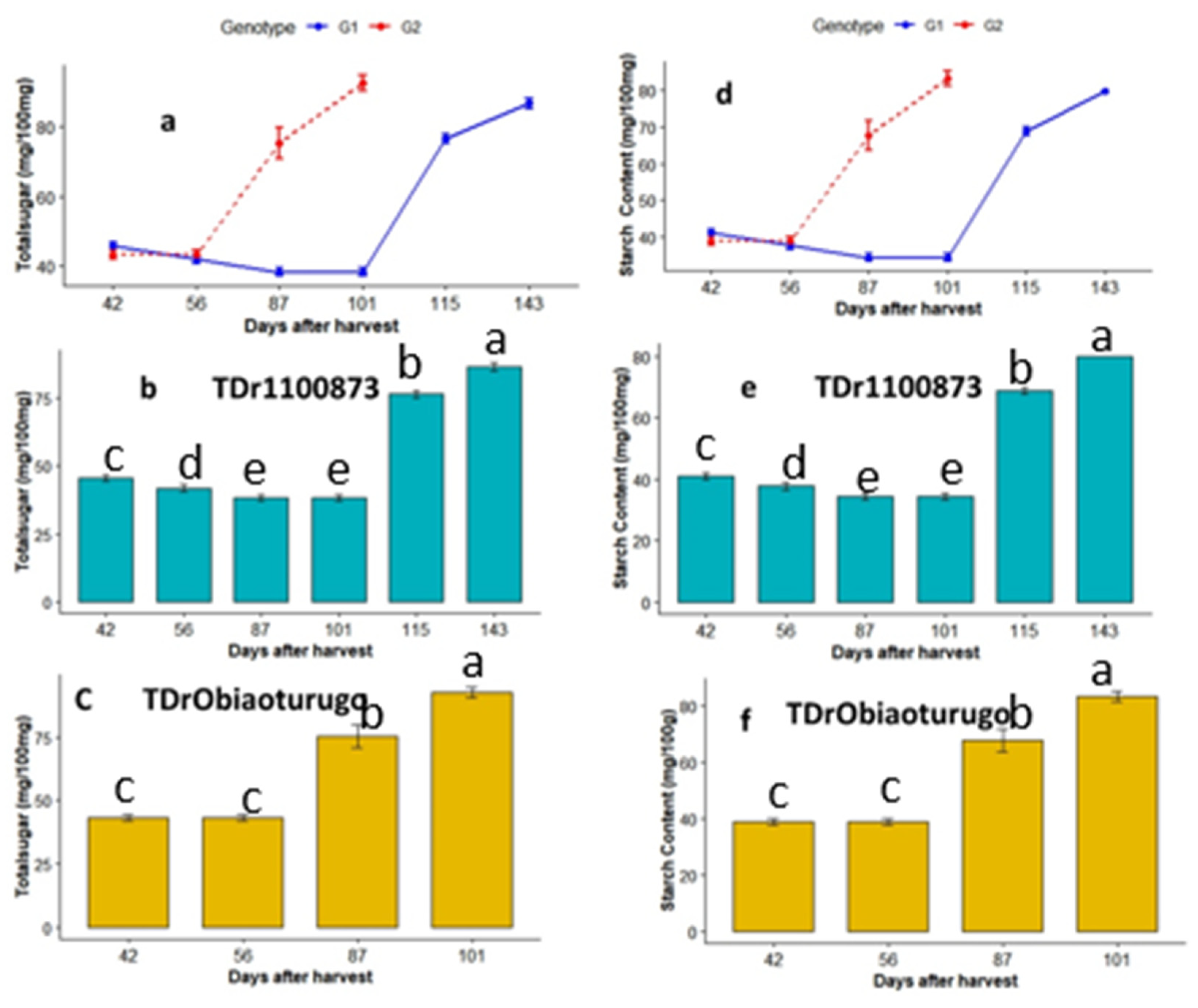
3.4.1. Total Sugar and starch
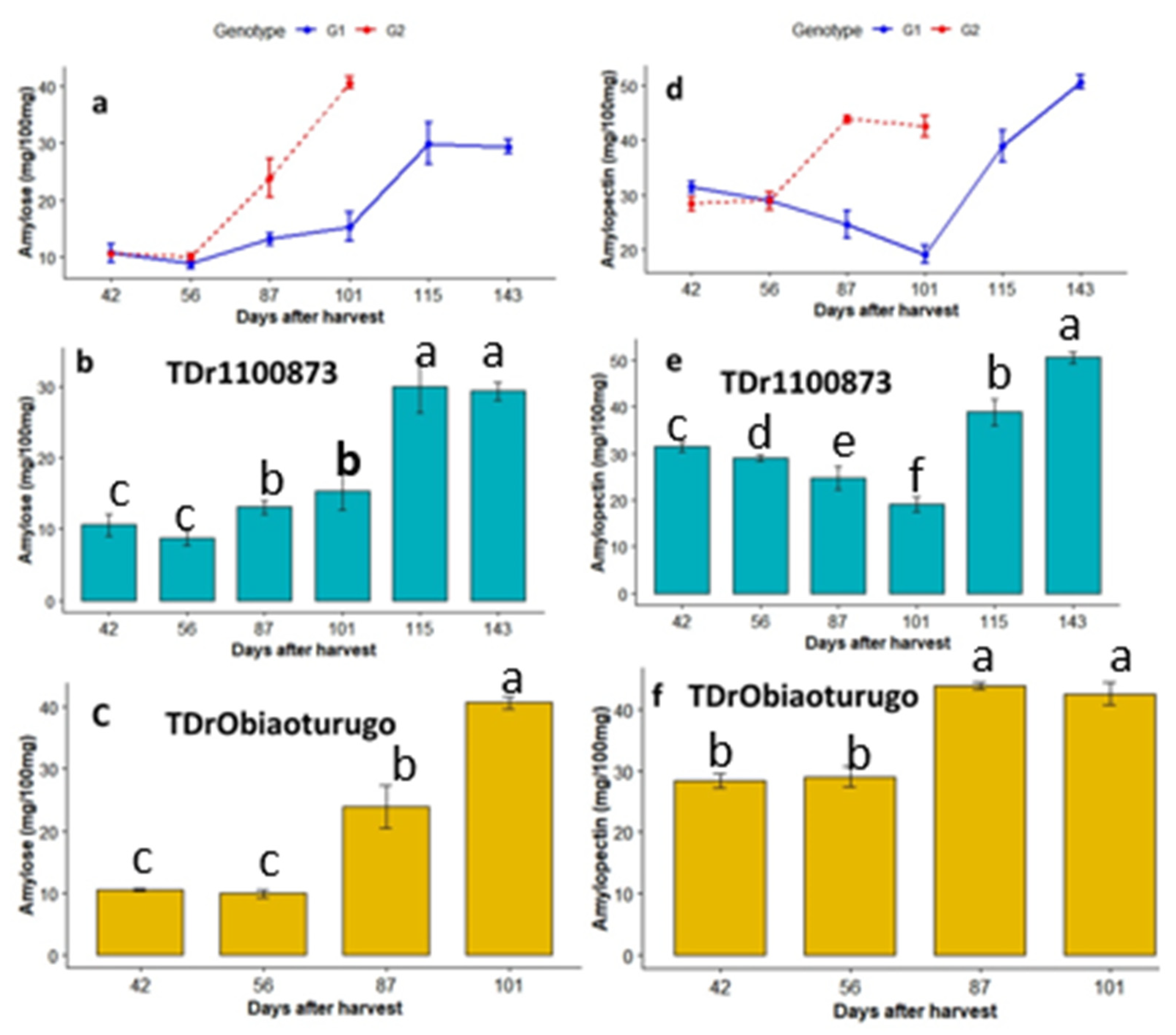
3.4.2. Amylose and Amylopectin
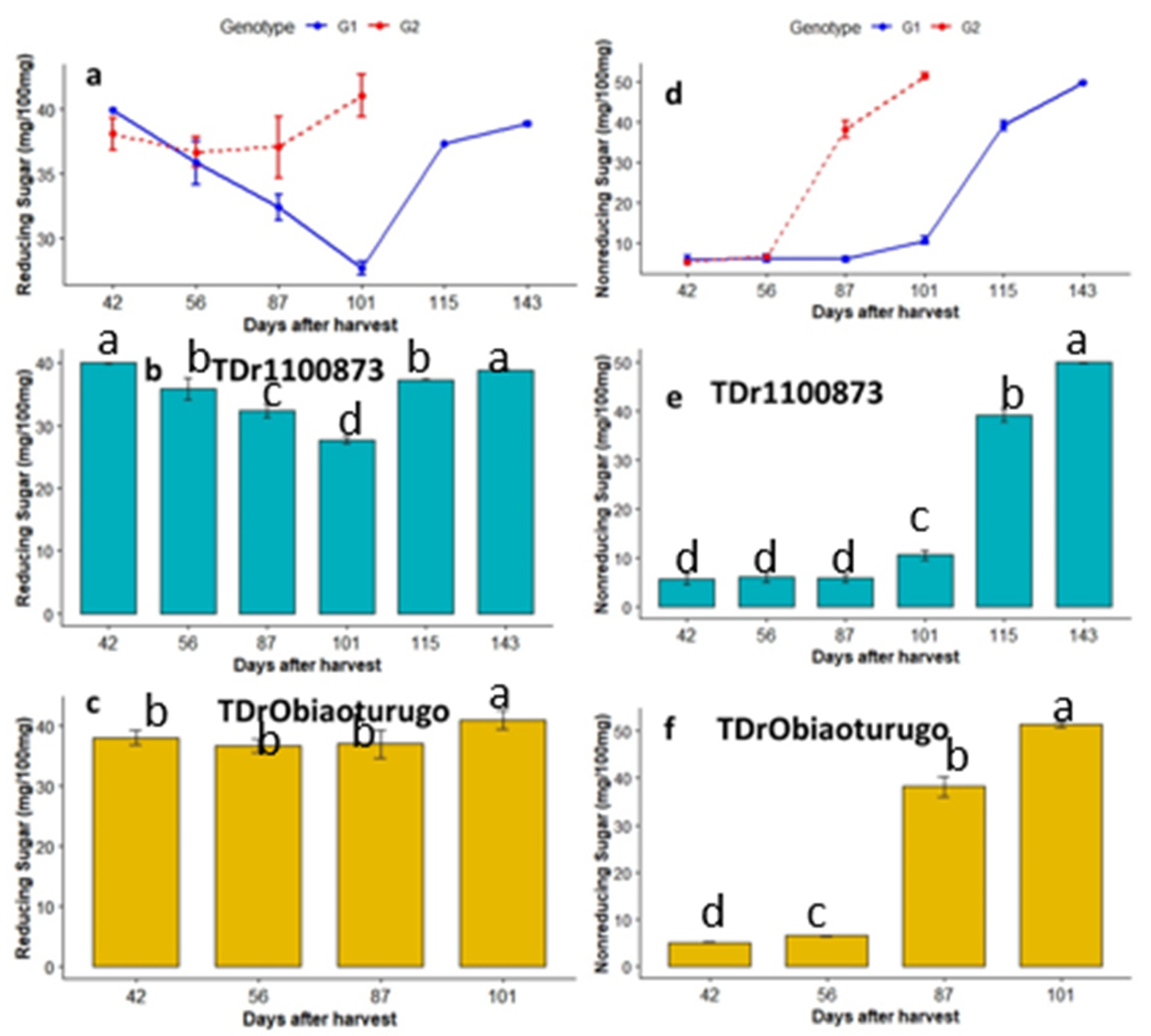
3.4.3. Reducing and nonreducing sugars
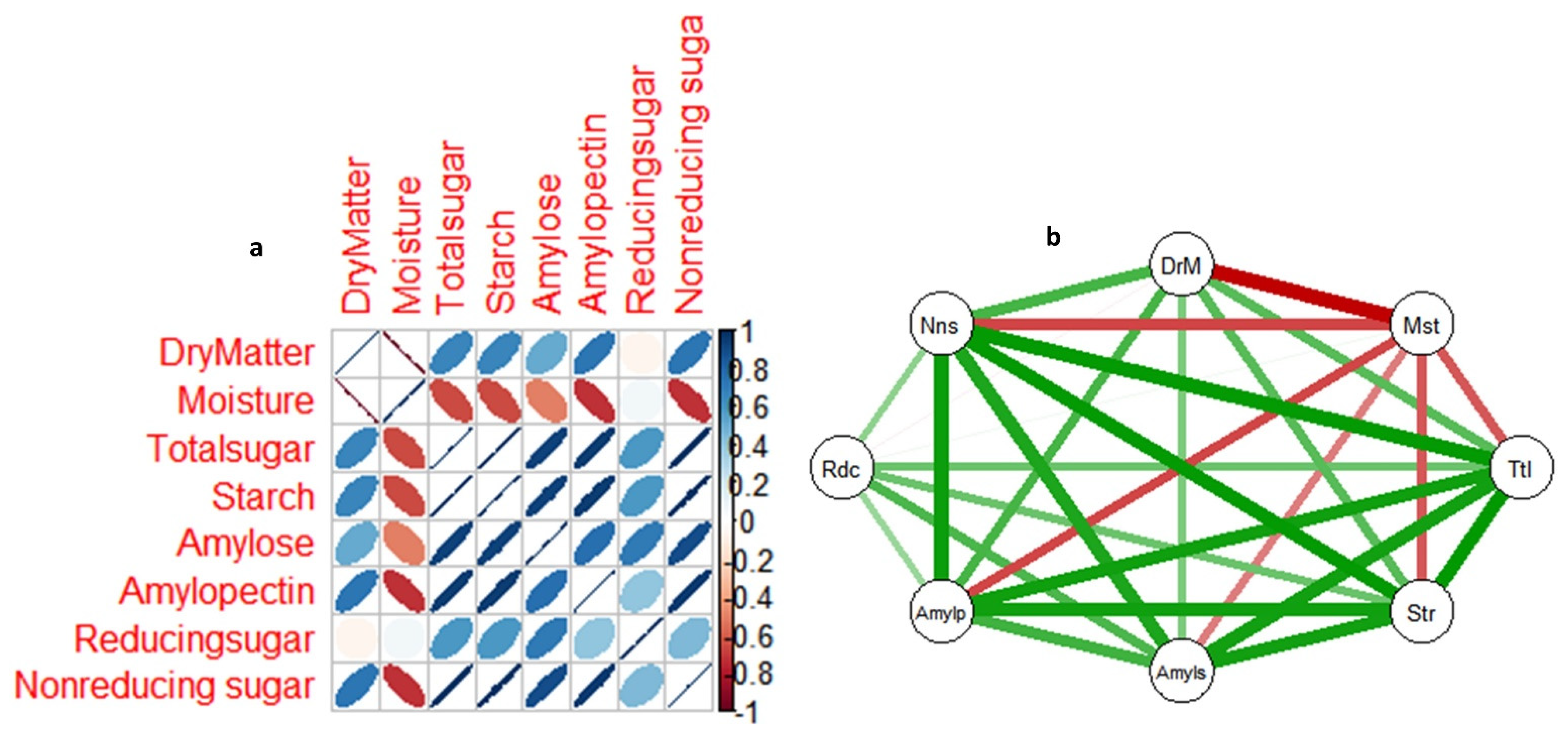
3.5. Correlations among non-structural sugars, dry matter and moisture content in yam tuber from dormancy to sprouting
4. Discussions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Obidiegwu, J.E.; Lyons, J.B.; Chilaka, C.A. The Dioscorea Genus (Yam)—An appraisal of nutritional and therapeutic potentials. Foods 2020, 9, 1304. [Google Scholar] [CrossRef]
- Obidiegwu, J.E.; Akpabio, E.M. The geography of yam cultivation in southern Nigeria: Exploring its social meanings and cultural functions. Journal of Ethnic Foods 2017, 4, 28–35. [Google Scholar] [CrossRef]
- Nwogha J. S.; J.E. Obidiegwu; R. N Okereke; R. Bhattacharjee; Oselebe, H.O. Preliminary verification of the adoption status of some yam (Dioscorea rotundata and Dioscorea alata) varieties in Nigeria using microsatellites markers. African Journal of Biotechnology 2022, 21, 198–207. [CrossRef]
- Price, E.J.; Wilkin, P.; Sarasan, V.; Fraser, P.D. Metabolite profiling of Dioscorea (yam) species reveals underutilised biodiversity and renewable sources for high-value compounds. Scientific reports 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Mignouna, H.D.; Abang, M.M.; Asiedu, R. Harnessing modern biotechnology for tropical tuber crop improvement: Yam (Dioscorea spp.) molecular breeding. African Journal of Biotechnology 2003, 2, 478–485. [Google Scholar]
- Hartmann, H.; Trumbore, S. Understanding the roles of nonstructural carbohy- drates in forest trees–from what we can measure to what we want to know. New Phytol. 2016, 211, 386–403. [Google Scholar] [CrossRef] [PubMed]
- Ile, E.; Craufurd, P.; Battey, N.; Asiedu, R. Phases of dormancy in yam tubers (Dioscorea rotundata). Annals of Botany 2006, 97, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Passam, H. Dormancy of yams in relation to storage. Yams. Ignames. 1982, 285–293. [Google Scholar]
- Yolou; Elie Idossou1 ADECHOKAN Hibath Audrey, A. ; Didier, M.Z.J. Evaluation of yam (Dioscorea cayenensis–Dioscorea rotundata) seed germination grown in Centre Benin. International Journal 2015, 3, 277–284. [Google Scholar]
- Hamadina, E.I. The control of yam tuber dormancy: a framework for manipulation. 2011.
- Zhang, Y.; He, J. Sugar-induced plant growth is dependent on brassinosteroids. Plant signaling & behavior 2015, 10, e1082700. [Google Scholar]
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709. [Google Scholar] [CrossRef] [PubMed]
- Ciereszko, I. Regulatory roles of sugars in plant growth and development. Acta Societatis Botanicorum Poloniae 2018, 87. [Google Scholar] [CrossRef]
- Wingler, A. Transitioning to the next phase: the role of sugar signaling throughout the plant life cycle. Plant physiology 2018, 176, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Damaris, R.N.; Lin, Z.; Yang, P.; He, D. The rice alpha-amylase, conserved regulator of seed maturation and germination. International journal of molecular sciences 2019, 20, 450. [Google Scholar] [CrossRef] [PubMed]
- Zhaowei, L.; Qian, Z.; Fangmin, C. Sugar starvation enhances leaf senescence and genes involved in sugar signaling pathways regulate early leaf senescence in mutant rice. Rice Science 2020, 27, 201–214. [Google Scholar] [CrossRef]
- Yaliang, W.; Yikai, Z.; Qinghua, S.; Huizhe, C.; Jing, X.; Guohui, H.; Yanhua, C.; Xiaodan, W.; Junke, W.; Zihao, Y. Decrement of sugar consumption in rice young panicle under high temperature aggravates spikelet number reduction. Rice Science 2020, 27, 44–55. [Google Scholar] [CrossRef]
- Rodriguez, M.; Parola, R.; Andreola, S.; Pereyra, C.; Martínez-Noël, G. TOR and SnRK1 signaling pathways in plant response to abiotic stresses: do they always act according to the “yin-yang” model? Plant Science 2019, 288, 110220. [Google Scholar] [CrossRef]
- Pokhilko, A.; Flis, A.; Sulpice, R.; Stitt, M.; Ebenhöh, O. Adjustment of carbon fluxes to light conditions regulates the daily turnover of starch in plants: a computational model. Molecular BioSystems 2014, 10, 613–627. [Google Scholar] [CrossRef]
- Gibson, S.I. Control of plant development and gene expression by sugar signaling. Current Opinion in Plant Biology 2005, 8, 93–102. [Google Scholar] [CrossRef]
- Dennis, R. The role of primary carbohydrate metabolism in wheat grain dormancy and germination. 2019.
- Graeber, K.; Nakabayashi, K.; Miatton, E.; Leubner-Metzger, G.; Soppe, W.J.J. Molecular mechanisms of seed dormancy. Plant, Cell and Environment 2012, 35, 1769–1786. [Google Scholar] [CrossRef]
- de Paiva Neto; Vespasiano Borges Otoni; Campos, W. Carbon sources and their osmotic potential in plant tissue culture: does it matter? Scientia Horticulturae 2003, 97, 193–202. [CrossRef]
- Sakr S; Wang M; Dédaldéchamp F; Perez-Garcia M-D; Ogé L; Hamama L; R., A. The sugar-signaling hub: overview of regulators and interaction with the hormonal and metabolic network. I. International journal of molecular sciences 2018., 19. [CrossRef]
- Wingler, A.; Henriques, R. Sugars and the speed of life—Metabolic signals that determine plant growth, development and death. Physiologia Plantarum 2022, 174, e13656. [Google Scholar] [CrossRef] [PubMed]
- Martín-Fontecha, E.S.; Tarancón, C.; Cubas, P. To grow or not to grow, a power-saving program induced in dormant buds. Current Opinion in Plant Biology 2018, 41, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Jaleel, C.A.; Gopi, R.; Manivannan, P.; Kishorekumar, A.; Gomathinayagam, M.; Panneersel Vam, R. Changes in biochemical constituents and induction of early sprouting by triadimefon treatment in white yam (Dioscorea rotundata Poir.) tubers during storage. Journal of Zhejiang University Science B 2007, 8, 283–288. [Google Scholar] [CrossRef]
- Tschannen, A.B.; Girardin, O.; Nindjin, C.; Daouda, D.; Farah, Z.; Stamp, P.; Escher, F. Improving the application of gibberellic acid to prolong dormancy of yam tubers (Dioscorea spp). Journal of the Science of Food and Agriculture 2003, 83, 787–796. [Google Scholar] [CrossRef]
- Hariprakash, C.; Nambisan, B. Carbohydrate metabolism during dormancy and sprouting in yam (Dioscorea) tubers: changes in carbohydrate constituents in yam (Dioscorea) tubers during dormancy and sprouting. Journal of Agricultural and Food Chemistry 1996, 44, 3066–3069. [Google Scholar] [CrossRef]
- Landhäusser, M.S.; Pak, S. Chow, L.; Turin Dickman; E., M.; Furze, I.K.; JSandra Schmid, J.W.; Birgit Wild; Gerd Gleixner; Henrik Hartmann; Günter Hoch; et al. Standardized protocols and procedures can precisely and accurately quantify non-structural carbohydrates. Tree Physiology 2018, 38, 1764–1778. [Google Scholar] [CrossRef]
- DUBOIS, M., K. A. GILLES, J. K. HAMILTON, P. A. REBERS, and FRED SMITH. Colorimetric Method for Determination of Sugars and Related Substances. Analytical Chemistry 1956, 28, 3, 350-356. [CrossRef]
- Widdowson, E.M. A method for the determination of small quantities of mixed reducing sugars and its application to the estimation of the products of hydrolysis of starch. Imperial College of Science and Technology, London. 1931.
- Sowbhagya, C.M.a.K.R.B., Mysore. A Simplified Colorimetric Method for Determination of Amylose Content in Rice. Die Starke . Jahrg 1971, 23.
- Huang, C.-C.; Chiang, P.-Y.; Chen, Y.-Y.; Wang, C.-C. Chemical compositions and enzyme activity changes occurring in yam (Dioscorea alata L.) tubers during growth. LWT-Food Science and Technology 2007, 40, 1498–1506. [Google Scholar] [CrossRef]
- Muzac-Tucker, I.; Helen, N.; Ahmad, M. Biochemical composition and storage of Japanican yams (Dioscorea spp.). Journal of the Science of Food and Agriculture 1993, 62, 219–224. [Google Scholar] [CrossRef]
- Paul, M.J.; Lawlor, D.W. GENETIC MODIFICATION OF PRIMARY METABOLISM | Photosynthesis. Encyclopedia of Applied Plant Sciences 2003, 484–493. [Google Scholar] [CrossRef]
- Jabrin, S.; Ravanel, S.; Gambonnet, B.; Douce, R.; Rébeillé, F. One-carbon metabolism in plants. Regulation of tetrahydrofolate synthesis during germination and seedling development. Plant physiology 2003, 131, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Landhäusser, S. Aspen shoots are carbon autonomous during bud break. Trees- Structure And Function 2011, 25. , 531–536. [CrossRef]
- Ohanenye, I.C.; Alamar, M.C.; Thompson, A.J.; Terry, L.A. Fructans redistribution prior to sprouting in stored onion bulbs is a potential marker for dormancy break. Postharvest Biology and Technology 2019, 149, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Smeekens, S.; Hellmann; A., H. Sugar sensing and signaling in plants. Frontiers in Plant Science 2014, 5, 185–205.
- Lastdrager, J.; Hanson, J.; Smeekens, S. Sugar signals and the control of plant growth and development. Journal of experimental botany 2014, 65, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Vilalta, J.; Sala, A.; Asensio, D.; Galiano, L.; Hoch, G.; Palacio, S.; Piper, F.I.; Lloret, F. Dynamics of non-structural carbohydrates in terrestrial plants: a global synthesis. Ecological Monographs 2016, 86, 495–516. [Google Scholar] [CrossRef]
- Doblin, M.S.; Kurek, I.; Jacob-Wilk, D.; Delmer, D.P. Cellulose biosynthesis in plants: from genes to rosettes. Plant and cell physiology 2002, 43, 1407–1420. [Google Scholar] [CrossRef]
- Tsai, A., Y-L .,; Gazzarrini, S. Trehalose-6-phosphate and SnRK1 kinases in plant development and signaling: the emerging picture. Frontiers in Plant Science 2014, 5, 1–11. [CrossRef]
- Durán-Soria, S.; Pott, D.M.; Osorio, S.; Vallarino, J.G. Sugar signaling during fruit ripening. Frontiers in Plant Science 2020, 11, 564917. [Google Scholar] [CrossRef]
- Hartmann, H.; Adams, H.D.; Hammond, W.M.; Hoch, G.; Landhäusser, S.M.; Wiley, E.; Zaehle, S. Identifying differences in carbohydrate dynamics of seedlings and mature trees to improve carbon allocation in models for trees and forests. Environmental and Experimental Botany 2018, 152, 7–18. [Google Scholar] [CrossRef]
- Yadav, U.P.; Ivakov, A.; Feil, R.; Duan, G.Y.; Walther, D.; Giavalisco, P.; Piques, M.; Carillo, P.; Hubberten, H.-M.; Stitt, M. The sucrose–trehalose 6-phosphate (Tre6P) nexus: specificity and mechanisms of sucrose signalling by Tre6P. Journal of experimental botany 2014, 65, 1051–1068. [Google Scholar] [CrossRef]
- Baena-González, E.; Lunn, J.E. SnRK1 and trehalose 6-phosphate–two ancient pathways converge to regulate plant metabolism and growth. Current Opinion in Plant Biology 2020, 55, 52–59. [Google Scholar] [CrossRef]
- Goddijn, O.J.; van Dun, K. Trehalose metabolism in plants. Trends in plant science 1999, 4, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, C.M.; Feil, R.; Ishihara, H.; Watanabe, M.; Kölling, K.; Krause, U.; Höhne, M.; Encke, B.; Plaxton, W.C.; Zeeman, S.C. Trehalose 6–phosphate coordinates organic and amino acid metabolism with carbon availability. The Plant Journal 2016, 85, 410–423. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhu, J.-Y.; Roh, J.; Marchive, C.; Kim, S.-K.; Meyer, C.; Sun, Y.; Wang, W.; Wang, Z.-Y. TOR signaling promotes accumulation of BZR1 to balance growth with carbon availability in Arabidopsis. Current Biology 2016, 26, 1854–1860. [Google Scholar] [CrossRef] [PubMed]
- Bledsoe, S.W.; Henry, C.; Griffiths, C.A.; Paul, M.J.; Feil, R.; Lunn, J.E.; Stitt, M.; Lagrimini, L.M. The role of Tre6P and SnRK1 in maize early kernel development and events leading to stress-induced kernel abortion. BMC Plant Biology 2017, 17, 1–17. [Google Scholar] [CrossRef]
| Genotype | DAPM | DM% | Moisture% | Total Sugar(mg/100mg) | Starch(mg/100mg) | Amylopectin(mg/100mg) | Amylose(mg/100mg) | Reducing Sugar(mg/100mg) | Nonreducing Sugar(mg/100mg) | |
|---|---|---|---|---|---|---|---|---|---|---|
| TDr1100873 | 42days | 46.12c | 53.88a | 45.63c | 41.07c | 31.45c | 10.62c | 39.97a | 5.66d | |
| TDr1100873 | 56days | 48.35c | 51.65b | 41.93d | 37.74d | 28.98d | 8.76c | 35.87b | 6.07d | |
| TDr1100873 | 87days | 52.3b | 47.69c | 38.23e | 34.41e | 24.67e | 13.07b | 32.42c | 5.81d | |
| TDr1100873 | 101days | 54.29b | 45.72d | 38.23e | 34.41e | 19.12f | 15.29b | 27.68d | 10.56c | |
| TDr1100873 | 115days | 57.55a | 42.45e | 76.47b | 68.82b | 38.85b | 27.5a | 37.32b | 39.15b | |
| TDr1100873 | 143days | 59.91a | 40.09f | 86.7a | 79.92a | 50.57a | 29.35a | 38.87a | 49.94a | |
| TDrObia | 42days | 43.24d | 67.17a | 44.4c | 38.85c | 28.37b | 10.48c | 38.1ab | 5.07d | |
| TDrObia | 56days | 48.5c | 51.5b | 43.17c | 38.85c | 28.98b | 9.86c | 36.71b | 6.46c | |
| TDrObia | 87days | 54.47b | 45.46c | 75.23b | 67.71b | 43.78a | 23.93b | 37.14ab | 38.18b | |
| TDrObia | 101days | 59.88a | 40.12d | 92.5a | 83.25a | 42.55a | 40.7a | 41.1a | 51.44a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




