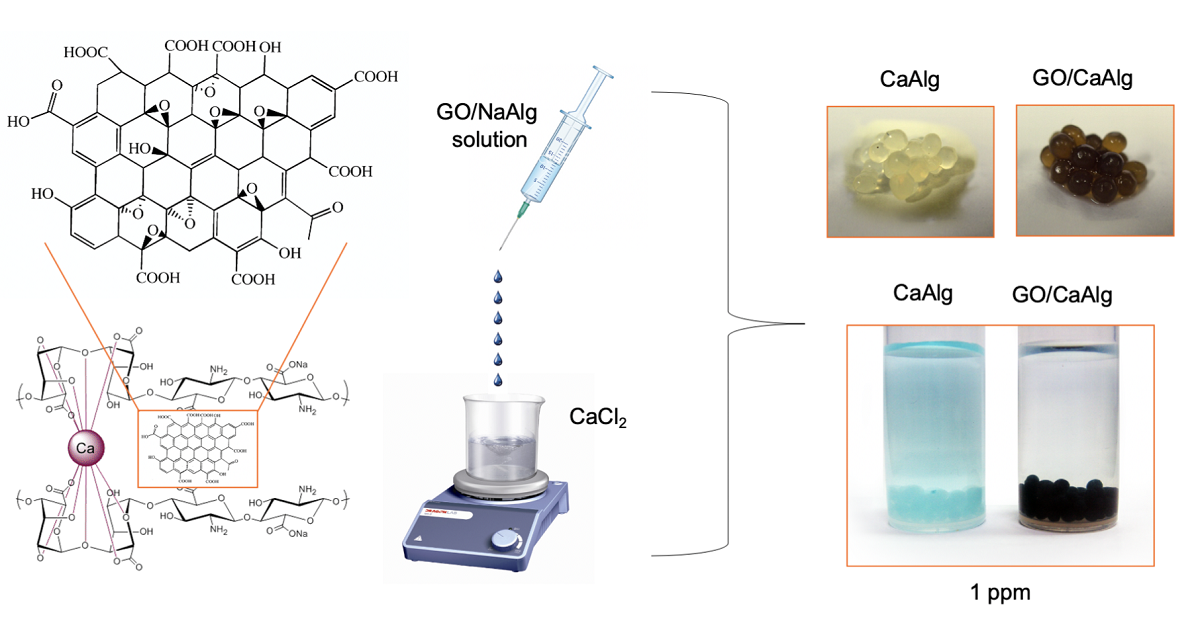
The remarkable adsorption capacity of graphene derived materials has prompted their examination in composite materials suitable for deployment in treatment of contaminated waters. In this study, crosslinked calcium alginate – graphene oxide beads were prepared and activated by exposure to pH 4 by using 0.1M HCl. The activated beads were investigated as novel adsorbents for the removal of organic pollutants (Methylene Blue dye and the pharmaceuticals Famotidine and Diclofenac) with a range of physicochemical properties. Effects of initial pollutant concentration, temperature, pH and adsorbent dose were investigated and kinetic models were examined for fit to the data. Maximum adsorption capacities qmax obtained were 1334, 35.50 and 36.35 mg g-1 for the uptake of Methylene blue, Famotidine and Diclofenac respectively. The equilibrium adsorption had an alignment with Langmuir isotherms while the kinetics were most accurately modelled using a pseudo- first –order and second order models according to the regression analysis. Thermodynamic parameters such as ΔG◦, ΔH◦ and ΔS◦ were calculated and the adsorption process was determined to be exothermic and spontaneous.