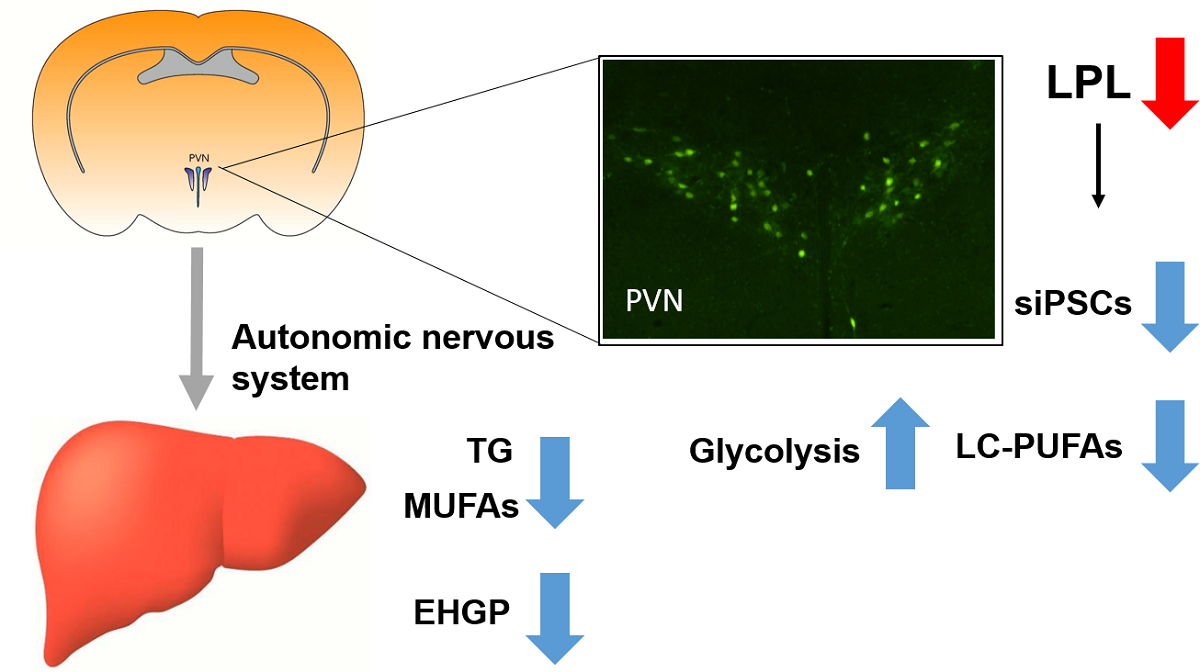
The autonomic regulation of hepatic metabolism offers a novel target for the treatment of non-alcoholic fatty liver disease (NAFLD). However, the molecular characteristics of neurons that regulate the brain-liver axis remain unclear. Since mice lacking neuronal lipoprotein lipase (LPL) develop perturbations in neuronal lipid-sensing and systemic energy balance, we reasoned that LPL might be a component of pre-autonomic neurons involved in the regulation of hepatic metabolism. Here we show that despite obesity, mice with reduced neuronal LPL (NEXCreLPLflox [LPL KD]) show improved glucose tolerance and reduced hepatic lipid accumulation with aging compared to WT controls (LPLflox). To determine the effect of LPL deficiency on neuronal physiology, liver-related neurons were identified in the paraventricular nucleus (PVN) of the hypothalamus using the transsynaptic retrograde tracer PRV-152. Patch-clamp studies revealed reduced inhibitory post-synaptic currents in liver-related neurons of LPL KD mice. Fluorescence Lifetime Imaging Microscopy (FLIM) was used to visualize metabolic changes in LPL-depleted neurons. Quantification of the free vs. bound Nicotinamide Adenine Dinucleotide (NADH) and Flavin Adenine Dinucleotide (FAD) revealed increased glucose utilization and TCA cycle flux in LPL-depleted neurons compared to controls. Global metabolomics from hypothalamic cell lines either deficient in, or over-expressing, LPL recapitulated these findings. Our data suggest that LPL is a novel feature of liver–related preautonomic neurons in the PVN. Moreover, LPL loss is sufficient to cause changes in neuronal substrate utilization and function, which may precede changes in hepatic metabolism.