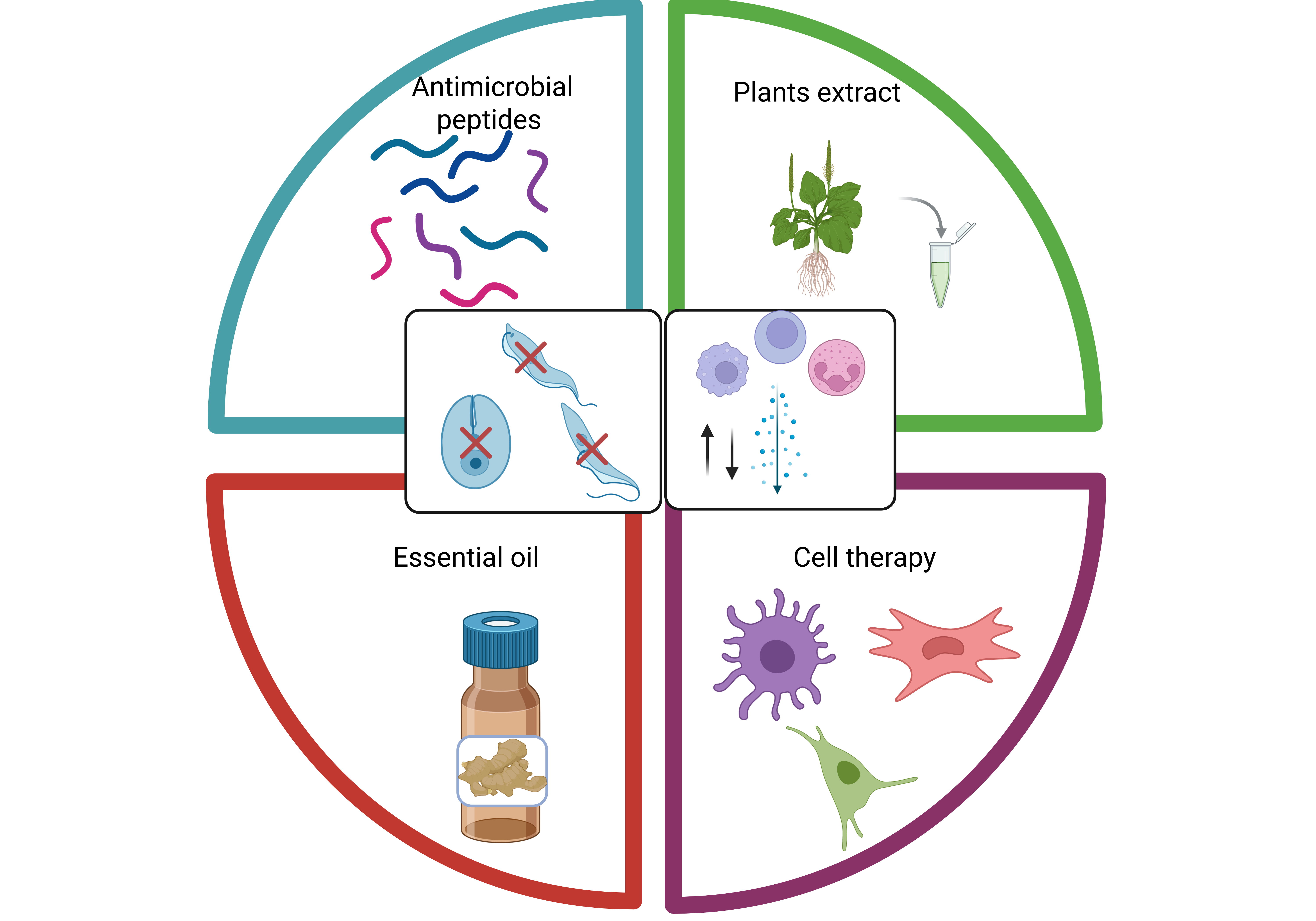
Chagas disease, caused by Trypanosoma cruzi, remains a major public health problem in Latin America and an emerging global health concern due to population mobility. Alt-hough benznidazole and nifurtimox remain the only approved antiparasitic drugs, their limited efficacy in chronic infection, prolonged treatment regimens, frequent adverse ef-fects, and variable activity across parasite strains highlight the need for new therapeutic strategies. In addition, the pathogenesis of chronic Chagas disease is driven not only by parasite persistence but also by immune-mediated tissue damage, particularly in chronic Chagas cardiomyopathy. In this review, we examine emerging therapeutic approaches that extend beyond conventional trypanocidal chemotherapy, with emphasis on plant-derived extracts, essential oils, antimicrobial peptides, and cell-based immuno-modulatory strategies. Plant compounds and essential oils have shown antiparasitic ac-tivity through mechanisms including oxidative stress induction, membrane disruption, interference with sterol biosynthesis, and mitochondrial dysfunction, while some extracts also modulate host immune responses. Antimicrobial peptides display dual potential by directly damaging parasite membranes and organelles or by reshaping infec-tion-associated inflammatory responses. In parallel, cell-based therapies such as mesen-chymal stromal cells, tolerogenic dendritic cells, and bone marrow-derived cells have demonstrated promising cardioprotective and immunoregulatory effects in experimental chronic Chagas disease. Collectively, these approaches support a multitarget therapeutic framework in which parasite-directed and host-directed interventions may complement each other. Further mechanistic studies, standardization, and translational validation will be essential to advance these candidates toward clinically useful therapies for Chagas disease.