Submitted:
04 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
There shall be neither glory nor grandeur in our creation and formation until the human being is formed.Popol Vuh1
1. Introduction
2. Animal Migration
- (i)
- seasonal migration, such as that of ruminants in the African savannah, following the local rainy season, or of geese and cranes (Weßling 2020) that escape the dark polar winter periodically, driven by external forcing,
- (ii)
- ontogenetic migration, such as that of salmons (McCleave et al. 1984), whales or turtles which spend their lives on certain feeding grounds but return to their place of birth for the breeding period, driven by internal forcing of individual ontogenetic development, and finally,
- (iii)
- density migration, such as that of swarming locusts (Dirsh 1974) that leave their habitat when it becomes overcrowded, driven by climate fluctuations and population dynamics, without return to the starting site.
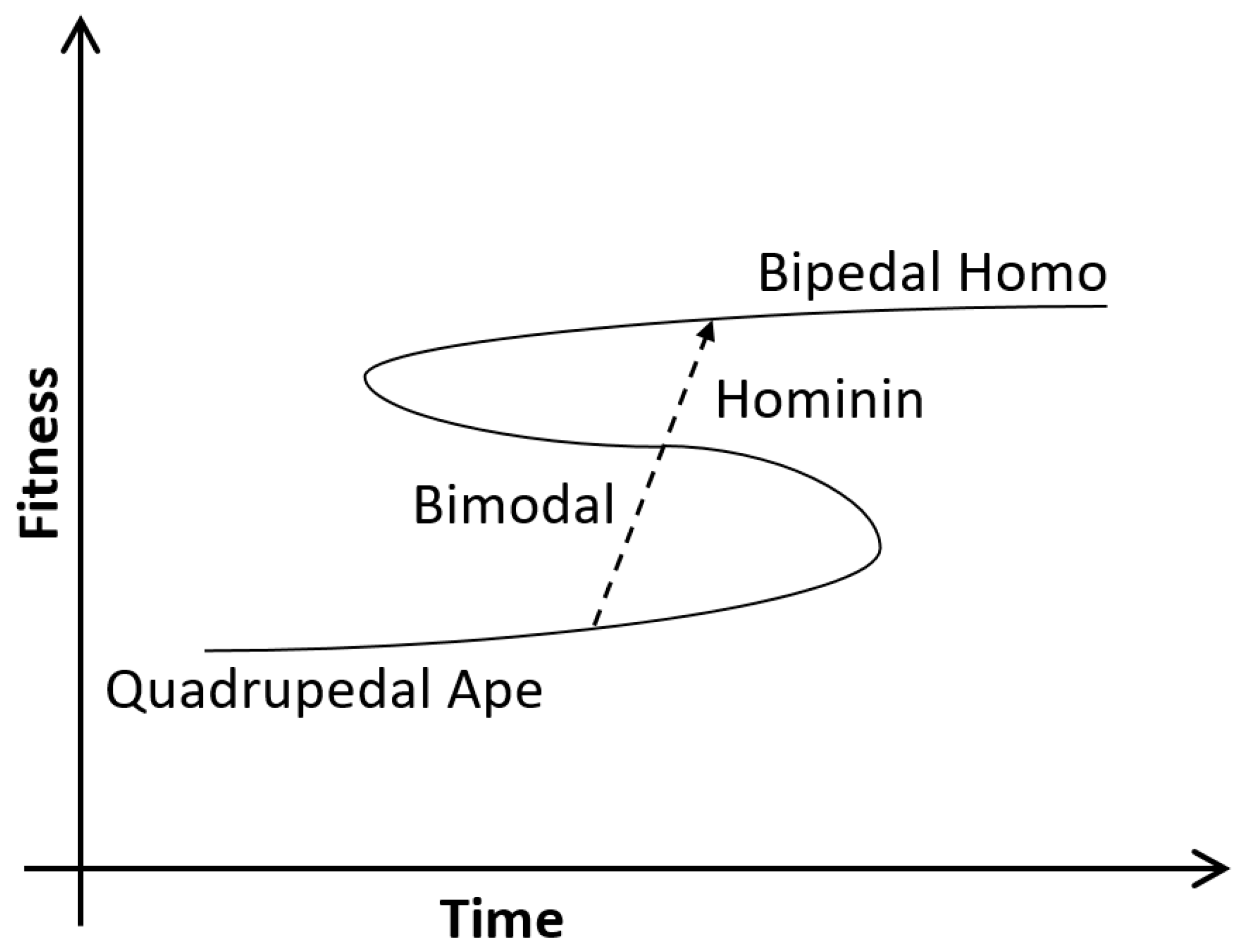
3. Hominin Migration
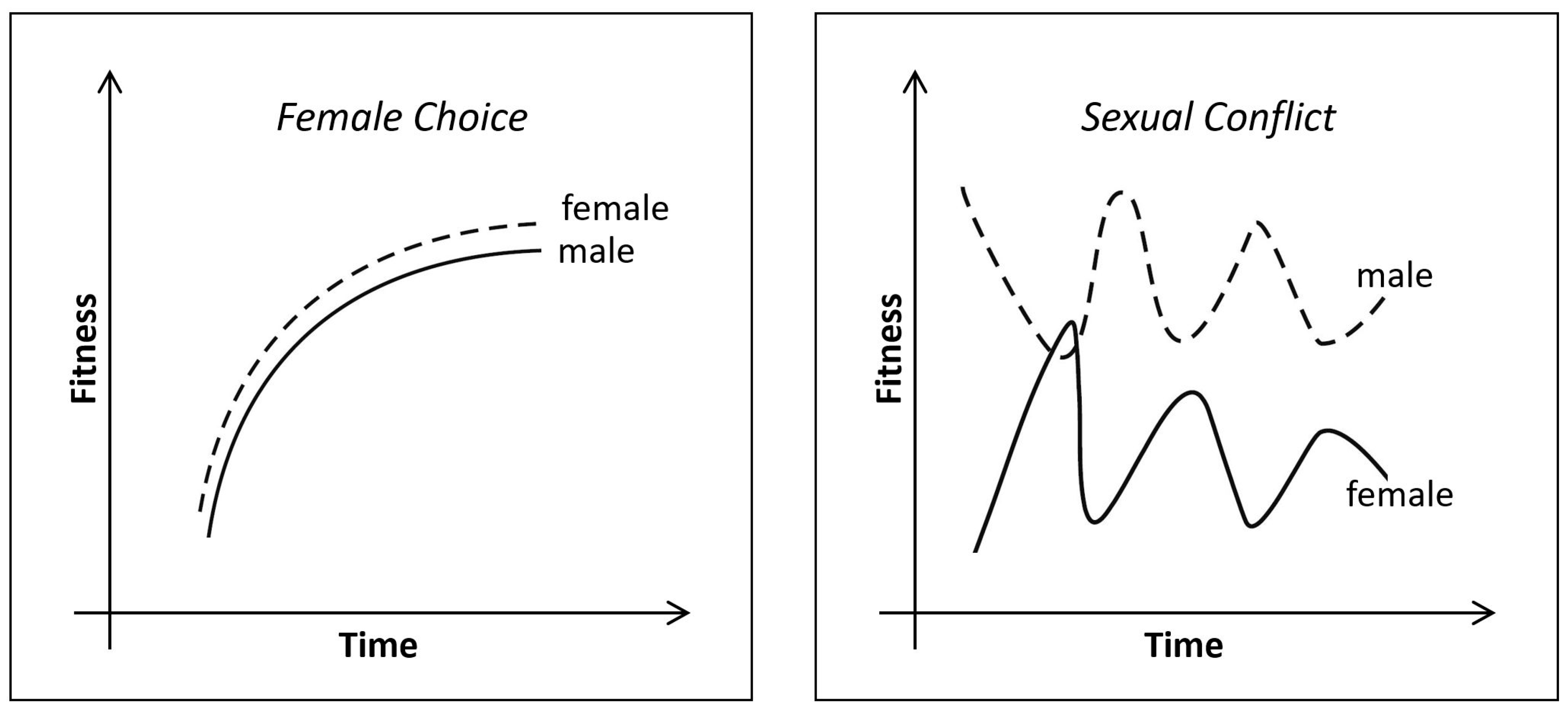
4. Emerging Childhood
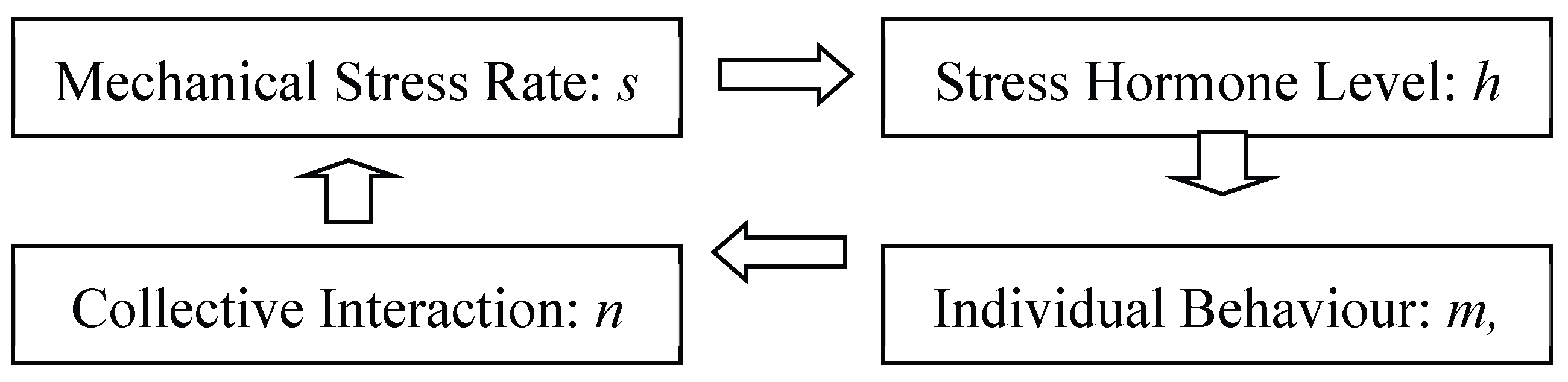
5. Migration Feedback
6. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A: On Geological Conditions 8-2 Myr BP
Appendix B: On Climatic Conditions 8-2 Myr BP
Appendix C: On Fossil Hominin Evidence 8-2 Myr BP
| Myr BP | Event / Fossil | Reference |
| 10 | Beginning Danakil rift opening | Rime et al. (2024) |
| 9-8 | Oreopithecus | Facchini (2006) |
| 8-1 | Gigantopithecus | Facchini (2006) |
| 7.175 | Graecopithecus freybergi in Pyrgos | Böhme et al. (2019) |
| >7 | Hominins switched to upright walking | Pennisi (2012) |
| 7-6 | Sahelanthropus tschadensis | Roberts (2011) |
| 6.2-5.6 | Orrorin tugenensis | Roberts (2011) |
| 6.1-5.7 | Bipedal Orrorin tugenensis in Kenya | Richmond & Jungers (2008) |
| 6 | Hominin loss of body hair | Sutou (2014) |
| 6-5 | Main Ethiopian Rift opening | Smith and Reynolds (2025) |
| 5.96-5.33 | Desiccated Mediterranean | Garcia-Castellanos & Villaseñor (2011) |
| 5.8-5.2 | Ardipithecus kadabba | Roberts (2011) |
| 5 | Gorilla separated from apes | Prado-Martinez et al. (2013) |
| 5 | Oceanic Red Sea | Horowitz (2001) |
| 4.5-4.3 | Ardipithecus ramidus | Roberts (2011) |
| 4.4 | Ardipithecus from Awash, Ethiopia | Gibbons (2009) |
| 4.2-3.9 | Australopithecus anamensis | Roberts (2011) |
| 4.2 | Earliest bipedal hominins outside Afar | Smith and Reynolds (2025) |
| 4 | Australopithecus populated Rift Valley | Schrenk et al. (2002) |
| 3.7-3 | Australopithecus afarensis | Roberts (2011) |
| 3.66 | Laetoli bipedal footprints | Deino (2010) |
| 3.6 | Bipedalism of Australopithecus | Haile-Selassie et al. (2010) |
| 3.6-3 | Australopithecus bahrelghazali | Roberts (2011) |
| 3.5-3.3 | Kenyanthropus platyops | Roberts (2011) |
| 3.4 | Human neuronal gene mutation | Geschwind & Konopka (2012) |
| > 3.39 | Stone-tool scraps in Dikika, Ethiopia | McPherron et al. (2010) |
| 3.3-2.1 | Australopithecus africanus | Roberts (2011) |
| 3.2 | Skeleton of »Lucy« from Afar, Ethiopia | Johanson & Edey (1990) |
| 3.032-2.581 | Paranthropus and tool use in Kenya | Plummer et al. (2023) |
| 3 | Homo separated from apes | Prado-Martinez et al. (2013) |
| 3 | Australopithecus eating grass, Chad | Lee-Thorp et al. (2012) |
| 2.7-2.5 | East African humid period | Trauth et al. (2005) |
| 2.7-2.3 | Paranthropus aethiopicus | Roberts (2011) |
| 2.6 | First stone tools from Afar, Ethiopia | Semaw et al. (2003) |
| 2.6 | Evolution of early Homo | Antón et al. (2014) |
| 2.6 | Homo regularly ate meat | Roach et al. (2013) |
| 2.5 | Homo separated from Paranthropus | Schrenk et al. (2002) |
| 2.5 | Stone tools used for hunting weapons | Deacon (1997) |
| 2.5 ± 0.1 | Quaternary ice-age oscillations began | Hewitt (2000), Horowitz (2001) |
| 2.5 | Formation of the Sahara desert | Kröpelin (2006) |
| 2.5-2.3 | Australopithecus garhi | Roberts (2011) |
| 2.45-2 | Ngorongoro Crater activity | Nigel (2001) |
| 2.4 | Human neuronal gene mutation | Geschwind & Konopka (2012) |
| 2.4-1.6 | Homo habilis | Roberts (2011) |
| 2.3-1.4 | Paranthropus boisei | Roberts (2011) |
| 2-1.2 | Paranthropus robustus | Roberts (2011) |
| 2 | Endurance running ofHomo | Bramble & Liebermann (2004) |
| 2 | “Genetic bottleneck” of Homo erectus | Hawks et al. (2000) |
| 1.95-1.78 | Australopithecus sediba | Roberts (2011) |
| 1.9-1.5 | Homo ergaster | Roberts (2011) |
| 1.85 | dispersal of Homo to Eurasia | Antón et al. (2014) |
| 1.8 | Homo georgicus | Roberts (2011) |
| 1.8-0.03 | Homo erectus | Roberts (2011) |
| 1.75 | Human spoken language | Morgan et al. (2015) |
| 1.6 | Homo ergaster in Koobi Fora, Kenya | Facchini (2006) |
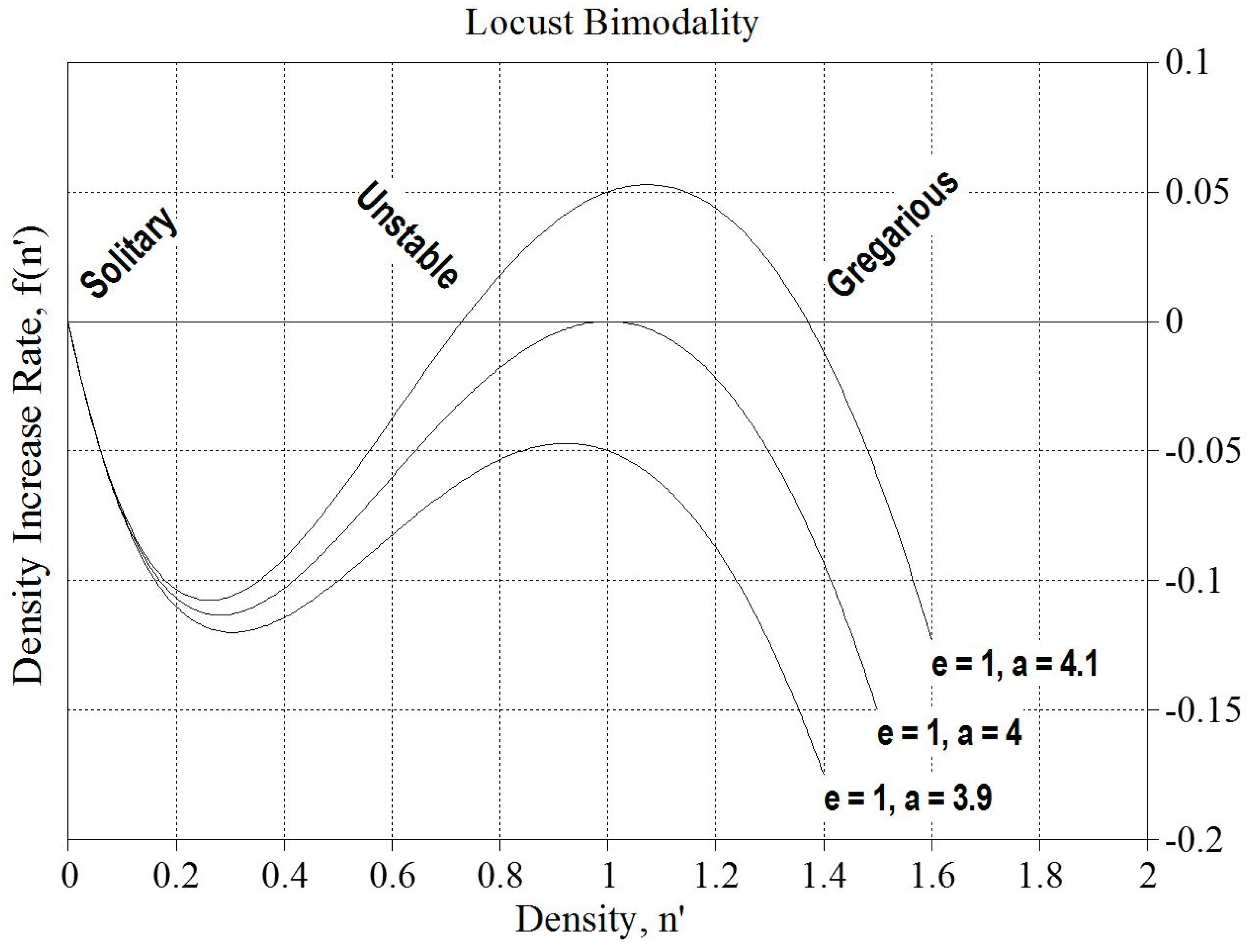
Appendix D: Mathematical Feedback Model for Migrating Locusts
References
- Akbari, A.; Perry, A.; Barton, A.R.; et al. (2026): Ancient DNA reveals pervasive directional selection across West Eurasia. Nature 2026. [Google Scholar] [CrossRef]
- Almalki, K.A.; Betts, P.G.; Ailleres, L. Incipient seafloor spreading segments: Insights from the Red Sea. Geophys. Res. Lett. 2016, 43, 2709–2715. [Google Scholar] [CrossRef]
- Antón, S.C.; Potts, R.; Aiello, L.C. Evolution of early Homo: An integrated biological perspective. Science 2014, 345, 1236828. [Google Scholar] [CrossRef] [PubMed]
- ARISE (2022): Mobilize for relief: Emergency response to flood affected communities in Afar. Advancement for Recovery Initiative for Safer Ethiopia (ARISE) Project. https://alnap.hacdn.io/media/documents/2022-ydt2Wj-ADPC-4_CaseStudies2028129.pdf accessed 06 Apr 2026.
- Ashley, G.M. Gornitz, V., Ed.; Human Evolution and Climate Change. In Encyclopedia of Paleoclimatology and Ancient Environments. Encyclopedia of Earth Sciences Series.; Springer; Dordrecht, 2009. [Google Scholar] [CrossRef]
- Augustin, N.; van der Zwan, F.M.; Devey, C.W.; Brandsdottir, B. 13 million years of seafloor spreading throughout the Red Sea Basin. Nature Communications 2021, 12, 2427. [Google Scholar] [CrossRef] [PubMed]
- Barash, D.P.; Lipton, J.E. How Women Got Their Curves and Other Just-So Stories: Evolutionary Enigmas; Columbia University Press; New York, 2009. [Google Scholar]
- Benanav, M. The Afar of Ethiopia. Nomads navigating a troubled twenty-first century. Traditional Cultures Project. 2020. Available online: https://www.afarofethiopia.com/ (accessed on 06 Apr 2026).
- Böhme, M.; Braun, R.; Breier, F. Wie wir Menschen wurden; Heyne; München, 2019. [Google Scholar]
- Bohm, K.; Kaakinen, A.; Stevens, T.; Lahaye, Y.; O’Brien, H.; Tang, H.; Shang, Y.; Zhang, H.; Lu, H. Neogene global climate change and East Asian dust sources: Combined rutile geochemistry and zircon U-Pb analysis from the northern Chinese Loess Plateau. Global and Planetary Change 2023, 221, 104049. [Google Scholar] [CrossRef]
- Bondevik, S.; Stormo, S.K.; Skjerdal, G. Green mosses date the Storegga tsunami to the chilliest decades of the 8.2 ka cold event. Quaternary Science Reviews 2012, 45, 1–6. [Google Scholar] [CrossRef]
- Bramble, D.M.; Lieberman, D.E. Endurance running and the evolution of Homo. Nature 2004, 432, 345–352. [Google Scholar] [CrossRef]
- Callaway, E. Oldest stone tools raise questions about their creators. The 3.3-million-year-old implements predate the first members of the Homo genus. Nature 2015, 520, 421. [Google Scholar] [CrossRef]
- Callaway, E. (2026): Landmark ancient-genome study shows surprise acceleration of human evolution. Nature 2026. [Google Scholar] [CrossRef]
- Campbell, R.M.; Vinas, G.; Henneberg, M.; Diogo, R. Visual Depictions of Our Evolutionary Past: A Broad Case Study Concerning the Need for Quantitative Methods of Soft Tissue Reconstruction and Art-Science Collaborations. Front. Ecol. Evol. 2021, 9, 639048. [Google Scholar] [CrossRef]
- Campbell, K.E., Jr.; Prothero, D.R.; Romero-Pittman, L.; Hertel, F.; Rivera, N. Amazonian magnetostratigraphy: Dating the first pulse of the Great American Faunal Interchange. Journal of South American Earth Sciences 2010, 29, 619–626. [Google Scholar] [CrossRef]
- Cordan, W. Popol Vuh. Das Buch des Rates; Mythos und Geschichte der Maya. Diederichs, Düsseldorf, 1962. [Google Scholar]
- Garcia-Castellanos, D.; Villaseñor, A. Messinian salinity crisis regulated by competing tectonics and erosion at the Gibraltar arc. Nature 2011, 480, 359–365. [Google Scholar] [CrossRef]
- Castañeda, I.S.; Mulitza, S.; Schefuß, E.; Lopes dos Santos, R.A.; Sinninghe Damsté, J.S.; Schouten, S. Wet phases in the Sahara/Sahel region and human migration patterns in North Africa. Proceedings of the National Academy of Sciences of the United States of America 2009, 106, 20159–20163. [Google Scholar] [CrossRef]
- Cerling, T.; Wynn, J.; Andanje, S.; et al. Woody cover and hominin environments in the past 6 million years. Nature 2011, 476, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, C. P.; Winnick, M.J.; Mix, H.T.; Chamberlain, S.D.; Maher, K. The impact of neogene grassland expansion and aridification on the isotopic composition of continental precipitation. Global Biogeochem. Cycles 2014, 28, 992–1004. [Google Scholar] [CrossRef]
- Cheng, H.; Fleitmann, D.; Edwards, R.L.; Wang, X.; Cruz, F.W.; Auler, A.S.; Mangini, A.; Wang, Y.; Kong, X.; Burns, S.J.; Matter, A. Timing and structure of the 8.2 kyr B.P. event inferred from δ18O records of stalagmites from China, Oman, and Brazil. Geology 2009, 37, 1007–1010. [Google Scholar] [CrossRef]
- Chocian, E. Wherever it rains, we go there: Afar nomadic architecture: tradition and challenge. Diploma Thesis, Technische Universität Wien, 2017. [Google Scholar] [CrossRef]
- Darwin, C. The Origin of Species by Means of Natural Selection; Hurst & Co.; New York, 1911. [Google Scholar]
- Darwin, C. The Descent of Man; Penguin Books; London, 2004. [Google Scholar]
- Deacon, T.W. The Symbolic Species. The Co-evolution of Language and the Brain; W.W. Norton & Company; New York, London, 1997. [Google Scholar]
- Deino, A.L. Harrison, T., Ed.; 40Ar/39Ar Dating of Laetoli, Tanzania. In Paleontology and Geology of Laetoli: Human Evolution in Context. Vertebrate Paleobiology and Paleoanthropology Series.; Springer; Dordrecht, 2011. [Google Scholar] [CrossRef]
- De Sousa, A.; Wood, B. 4.18 – The Hominin Fossil Record and the Emergence of the Modern Human Central Nervous System. Evolution of Nervous Systems 2007, 4, 291–336. [Google Scholar] [CrossRef]
- deMenocal, P.B. Climate and Human Evolution. Science 2011, 331, 540–542. [Google Scholar] [CrossRef]
- Diamond, J. Why Is Sex Fun? The Evolution of Human Sexuality (Science Masters); BasicBooks; New York, 1997. [Google Scholar]
- Dingle, H.; Drake, V.A. BioScience 2007, 57, 113–121. [CrossRef]
- Dirsh, V.M. Genus Schistocerca (Acridomorpha, Insecta); Kluwer; Dordrecht, 1974. [Google Scholar]
- Donges, J.F.; Donner, R.V.; Trauth, M.H.; Kurths, J. Nonlinear detection of paleoclimate-variability transitions possibly related to human evolution. PNAS 2011, 108, 20422–20427. [Google Scholar] [CrossRef]
- Dowsett, H.J.; Barron, J.A.; Poore, R.Z.; Thompson, R.S.; Cronin, T.M.; Ishman, S.E.; Willard, D.A. Middle Pliocene Paleoenvironmental Reconstruction: PRISM2. U.S. GEOLOGICAL SURVEY OPEN FILE REPORT 99-535. 1999. Available online: https://pubs.usgs.gov/of/1999/of99-535/ (accessed on 31 Mar 2026).
- Dullo, T.T.; Kalyanapu, A.J. Floods in Ethiopia: A Review; Natural Hazards Research, 2025. [Google Scholar] [CrossRef]
- Dumanot, H.J. The Nile: Origin, environments, limnology and human use; Springer Science & Business; Berlin, Heidelberg, 2009. [Google Scholar]
- Ebeling, W.; Feistel, R. Physik der Selbstorganisation und Evolution; Akademie-Verlag; Berlin, Germany, 1982. [Google Scholar]
- Ebeling, W.; Feistel, R. Chaos und Kosmos. In Prinzipien der Evolution; Spektrum-Verlag; Heidelberg, Germany, 1994. [Google Scholar]
- Eigen, M. Selforganization of matter and the evolution of biological macromolecules. Naturwissenschaften 1971, 58, 465–523. [Google Scholar] [CrossRef]
- Elawa, A.M.T. Migration of Organisms. In Climate. Geography. Ecology.; Springer; Berlin, Heidelberg, 2005. [Google Scholar] [CrossRef]
- Emera, D.; Romero, R.; Wagner, G. The evolution of menstruation: A new model for genetic assimilation. Bioessays 2011, 34, 26–35. [Google Scholar] [CrossRef]
- Engels, F. Heid, H., Rodax, K., Hoff, E.H., Eds.; Anteil der Arbeit an der Menschwerdung des Affen. In Ökologische Kompetenz. VS Verlag für Sozialwissenschaften; Wiesbaden, 2000. [Google Scholar] [CrossRef]
- Facchini, F. Die Ursprünge der Menschheit; Konrad Theiss; Stuttgart, 2006. [Google Scholar]
- Feakins, S.J.; Levin, N.E.; Liddy, H.M.; Sieracki, A.; Eglinton, T.I.; Bonnefille, R. Northeast African vegetation change over 12 m.y.; Geological Society of America; Data Repository, 2013; p. item 2013079. [Google Scholar] [CrossRef]
- Fedorov, A.V.; Brierley, C.M.; Lawrence, K.T.; Liu, Z.; Dekens, P.S.; Ravelo, A.C. Patterns and mechanisms of early Pliocene warmth. Nature 2013, 496, 43–49. [Google Scholar] [CrossRef]
- Feistel, R. Beschreibung von Evolutionsprozessen bei diploider Sequenzreplikation. Wiss. Z. WPU Rostock 1977, 26, 655–662. Available online: https://www.researchgate.net/publication/266685247_Zur_Beschreibung_von_Evolutionsprozessen_bei_diploider_Sequenzreplikation (accessed on 10 Apr 2026).
- Feistel, R. On the Evolution of Symbols and Prediction Models. Biosemiotics 2023b, 16, 311–371. [Google Scholar] [CrossRef]
- Feistel, R. Origin of Life: A Symmetry-Breaking Physical Phase Transition. Symmetry 2024, 16, 1611. [Google Scholar] [CrossRef]
- Feistel, R. Bipedalism, Childhood, and Ritualisation of Human Sexual Behaviour: A Hominin Model Scenario of Ontogenetic Selection. Biosystems 2025, 257, 105598, Update (2026). [Google Scholar] [CrossRef] [PubMed]
- Feistel, R.; Ebeling, W. Deterministic and Stochastic Theory of Sustained Oscillations in Autocatalytic Reaction Systems. Physica A 1978, 93, 114–137. [Google Scholar] [CrossRef]
- Feistel, R.; Ebeling, W. Evolution of Complex Systems; Kluwer; Dordrecht, 1989. [Google Scholar]
- Feistel, R.; Ebeling, W. Physics of Self-Organization and Evolution; Wiley VCH; Weinheim, 2011. [Google Scholar]
- Feistel, R.; Feistel, S. Locust Phase Transitions. Preprint 2015. [Google Scholar] [CrossRef]
- Foubert, A.; Keir, D.; Atnafu, B.; Kidane, T. and the ADD-ON Workshop Consortium (2024): Workshop report: Afar Dallol Drilling – ONset of sedimentary processes in an active rift basin (ADD-ON). Scientific Drilling 33, 207–218. [CrossRef]
- Fryxell, J.M.; Holt, R.D. Environmental change and the evolution of migration. Ecology 2013, 94, 1274–1279. [Google Scholar] [CrossRef]
- Galdikas, B.M.F.; Wood, J.W. Birth spacing patterns in humans and apes. American Journal of Biological Anthropology 1990, 83, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Castellanos, D.; Villaseñor, A. Messinian salinity crisis regulated by competing tectonics and erosion at the Gibraltar arc. Nature 2011, 480, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, D.H.; Konopka, G. Neuroscience: Genes and human brain evolution. Nature 2012, 486, 481–482. [Google Scholar] [CrossRef]
- Gibbons, A. A New Kind of Ancestor: Ardipithecus Unveiled. Science 2009, 326, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Glansdorff, P.; Prigogine, I. Thermodynamic Theory of Structure, Stability and Fluctuations; Wiley-Interscience; London – New York – Sydney - Toronto, 1971. [Google Scholar]
- Goodall, J. Wilde Schimpansen. In Verhaltensforschung am Gombe-Strom; Rowohlt; Reinbek, 1991. [Google Scholar]
- Green, D.J.; Alemseged, Z. Australopithecus afarensis Scapular Ontogeny, Function, and the Role of Climbing in Human Evolution. Science 2012, 338, 514–517. [Google Scholar] [CrossRef]
- Gregoire, L.J.; Payne, A.J.; Valdes, P.J. (1012): Deglacial rapid sea level rises caused by ice-sheet saddle collapses. Nature 487, 219–222. [CrossRef]
- Griswold, C.K.; Taylor, C.M.; Norris, D.R. The evolution of migration in a seasonal environment. Proc. Biol. Sci. 2010, 277, 2711–2720. [Google Scholar] [CrossRef]
- Gruss, L.T.; Schmitt, D. The evolution of the human pelvis: changing adaptations to bipedalism, obstetrics and thermoregulation. Phil. Trans. R. Soc. B 2015, 370, 20140063. [Google Scholar] [CrossRef]
- Gursky, S.L.; Nekaris, K.A.I. Primate Anti-Predator Strategies; Springer; New York, 2007; Available online: https://link.springer.com/content/pdf/bfm:978-0-387-34810-0/1.pdf (accessed on 31 Mar 2026).
- Haile-Selassie, Y.; Latimer, B.M.; Alene, M.; Deino, A.L.; Gibert, L.; Melillo, S.M.; Saylor, B.Z.; Scott, G.R.; Lovejoy, C.O. An early Australopithecus afarensis postcranium from Woranso-Mille, Ethiopia. Proceedings of the National Academy of Sciences of the United States of America 2010, 107, 12121–12126. [Google Scholar] [CrossRef]
- Haken, H. Synergetics. An Introduction. Nonequilibrium Phase Transitions and Self-Organization in Physics, Chemistry and Biology; Springer; Berlin/Heidelberg, Germany; New York, NY, USA, 1977. [Google Scholar]
- Hancock, G.; Pankhurst, R.; Willetts, D. Under ETHIOPIAN Skies; H&L Communications; London, 1983. [Google Scholar]
- Haug, G.H.; Ganopolski, A.; Sigman, D.M.; Rosell-Mele, A.; Swann, G.E.A.; Tiedemann, R.; Jaccard, S.L.; Bollmann, J.; Maslin, M.A.; Leng, M.J.; Eglinton, G. North Pacific seasonality and the glaciation of North America 2.7 million years ago. Nature 2005, 433, 821–825. [Google Scholar] [CrossRef]
- Hawks, J.; Hunley, K.; Lee, S.H.; Wolpoff, M. Population Bottlenecks and Pleistocene Human Evolution. Molecular Biology and Evolution 2000, 17, 2–22. [Google Scholar] [CrossRef]
- Henke, W.; Rothe, H. Menschwerdung; Avus Buch & Medien; Köln, 2003. [Google Scholar]
- Henry, A.; Ungar, P.; Passey, B.; et al. The diet of Australopithecus sediba. Nature 2012, 487, 90–93. [Google Scholar] [CrossRef]
- Hernández-Molina, F.J.; Stow, D.A.V.; Alvarez-Zarikian, C.A.; Acton, G.; Bahr, A.; Balestra, B.; Ducassou, E.; Flood, R.; Flores, J.-A.; Furota, S.; Grunert, P.; Hodell, D.; Jimenez-Espejo, F.; Kim, J.K.; Krissek, L.; Kuroda, J.; Li, B.; Llave, E.; Lofi, J.; Lourens, L.; Miller, M.; Nanayama, F.; Nishida, N.; Richter, C.; Roque, C.; Pereira, H.; Sanchez Goñi, M.F.; Sierro, F.J.; Singh, A.D.; Sloss, C.; Takashimizu, Y.; Tzanova, A.; Voelker, A.; Williams, T.; Xuan, C. Onset of Mediterranean outflow into the North Atlantic. Science 2014, 344, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, G. The genetic legacy of the Quaternary ice ages. Nature 2000, 405, 907–913. [Google Scholar] [CrossRef]
- Hill, A.; Ward, S. Origin of the Hominidae: The Record of African Large Hominoid Evolution Metween 14 My and 4 My. American Journal of Physical Anthropology 1988, 31, fmi, ii, 1–229. [Google Scholar] [CrossRef]
- Hoffmann, D.; Rogerson, M.; Spötl, C.; et al. Timing and causes of North African wet phases during the last glacial period and implications for modern human migration. Scientific Reports 2016, 6, 36367. [Google Scholar] [CrossRef] [PubMed]
- Holloway, R.L.; Broadfield, D.C.; Yuan, M.S. Schwartz, J.H., Tattersall, I., Holloway, R.L., Broadfield, D.C., Yuan, M.S., Eds.; Appendix 1: Endocranial Volumes of the Fossil Hominids; The Human Fossil Record, 2004. [Google Scholar] [CrossRef]
- Hoorn, C.; Wesselingh, F.P.; ter Steege, H.; Bermudez, M.A.; Mora, A.; Sevink, J.; Sanmartín, I.; Sanchez-Meseguer, A.; Anderson, C.L.; Figueiredo, J.P.; Jaramillo, C.; Riff, D.; Negri, F.R.; Hooghiemstra, H.; Lundberg, J.; Stadler, T.; Särkinen, T.; Antonelli, A. Amazonia Through Time: Andean Uplift, Climate Change, Landscape Evolution, and Biodiversity. Science 2010, 330, 927–931. [Google Scholar] [CrossRef]
- Horowitz, A.; Swets; Zeitlinger, B.V. The Jordan Rift Valley; Lisse, 2001. [Google Scholar]
- Huxley, J. (1914): The Courtship-Habits of the Great Crested Grebe (Podiceps cristatus); with an Addition to the Theory of Sexual Selection. Proceedings of the Zoological Society of London 1914, 491–562. Available online: https://dn790006.ca.archive.org/0/items/biostor-107422/biostor-107422.pdf (accessed on 05 June 2025). [CrossRef]
- Jakobus, F. Bipedalism: The Evolutionary Leap that Shaped Humanity. Journal of Evolutionary Medicine 2024, 12, 146158. [Google Scholar] [CrossRef]
- Johanson, D.C.; Edey, M. Lucy: the Beginnings of Humankind; Simon & Schuster; New York, 1990. [Google Scholar]
- Johanson, D.C.; White, T.D.; Coppens, Y. A New Species of the Genus Australopithecus (Primates: Hominidae) from the Pliocene of Eastern Africa. Kirtlandia 1978, 28, 1–14. Available online: https://biostor.org/reference/193077.
- Kalb, J.E.; Froehlich, D.J.; Bell, G.L. Shoshani, J., Tassy, P., Eds.; Palaeobiogeography of late Neogene African and Eurasian Elephantoidea. In The Proboscidea. Evolution and palaeoecology of the Elephants and their relatives.; Oxford, New York, Tokyo, 1996; pp. 117–123. [Google Scholar] [CrossRef]
- Kashchiev, D. Nucleation. In Basic Theory with Applications; Butterworth-Heinemann; Oxford, 2000. [Google Scholar] [CrossRef]
- Kennedy, G.E. From the ape’s dilemma to the weanling’s dilemma: early weaning and its evolutionary context. Journal of Human Evolution 2005, 48, 123–145. [Google Scholar] [CrossRef]
- Klix, F. Erwachendes Denken; Deutscher Verlag der Wissenschaften; Berlin, 1980. [Google Scholar]
- Knutz, P.C.; Newton, A.M.W.; Hopper, J.R.; et al. Eleven phases of Greenland Ice Sheet shelf-edge advance over the past 2.7 million years. Nat. Geosci. 2019, 12, 361–368. [Google Scholar] [CrossRef]
- Koeberl, C.; Pittarello, L.; Reimold, W.U.; Raschke, U.; Brigham-Grette, J.; Melles, M.; Minyuk, P.; Spray, J. El’gygytgyn impact crater, Chukotka, Arctic Russia: Impact cratering aspects of the 2009 ICDP drilling project. Meteoritics & Planetary Science 2013, 48, 1108–1129. [Google Scholar] [CrossRef]
- Kröpelin, S. Revisiting the Age of the Sahara Desert. Science 2006, 312, 1138. [Google Scholar] [CrossRef] [PubMed]
- Kunkelova, T.; Crocker, A. J.; Wilson, P.A.; Schepanski, K. Dust source activation frequency in the Horn of Africa. Journal of Geophysical Research: Atmospheres 2024, 129, e2023JD039694. [Google Scholar] [CrossRef]
- Landau, L.D.; Lifschitz, E.M. Lehrbuch der Theoretischen Physik Band V, Statistische Physik; Akademie-Verlag; Berlin, Germany, 1966. [Google Scholar]
- Larsen, H.; Saunders, A.; Clift, P.; Beget, J.; Wei, W.; Spezzaferri, S.; Ali, J.; Cambray, H.; Demant, A.; Fitton, G.; Fram, M.; Fukuma, K.; Gieskes, J.; Holmes, M.; Hunt, J.; Lacasse, C.; Larsen, L.; Lykke-Andersen, H.; Meltser, A.; Morrison, M.; Nemoto, N.; Okay, N.; Saito, S.; Sinton, C.; Stax, R.; Vallier, T.; Vandamme, D.; Werner, R. Seven million years of glaciation in Greenland. Science 1994, 264, 952–955. [Google Scholar] [CrossRef]
- Last, G.C. A Geography of Ethiopia; Ministry of Education; Addis Ababa, Ethiopia, 1965. [Google Scholar]
- Lee-Thorp, J.; Likius, A.; Mackaye, H.T.; Vignaud, P.; Sponheimer, M.; Brunet, M. Isotopic evidence for an early shift to C4 resources by Pliocene hominins in Chad. PNAS 2012, 109, 20369–20372. [Google Scholar] [CrossRef]
- Lehninger, A.L. Biochemistry; Worth Publishers; New York, 1972. [Google Scholar]
- Lunt, D.J.; Foster, G.L.; Haywood, A.M.; Stone, E.J. Late Pliocene Greenland glaciation controlled by a decline in atmospheric CO2 levels. Nature 2008, 454, 1102–1105. [Google Scholar] [CrossRef]
- Margulis, L.; Sagan, D. What is Life? The Eternal Enigma; University of California Press; Berkeley, CA, USA; Los Angeles, CA, USA, 1995. [Google Scholar]
- Marijuan, P.C.; Igamberdiev, A.U.; Deacon, T.W.; Iurato, G. Anthropogenesis and the origin of human consciousness. BioSystems 2026, 259, 105668. [Google Scholar] [CrossRef]
- McCleave, J.D.; Arnold, G.P.; Dodson, J.J.; Neill, W.H. Mechanisms of Migration in Fishes; Springer; New York, 1984. [Google Scholar] [CrossRef]
- McPherron, S.P.; Alemseged, Z.; Marean, C.W.; Wynn, J.G.; Reed, D.; Geraads, D.; Bobe, R.; Béarat, H.A. Evidence for stone-tool-assisted consumption of animal tissues before 3.39 million years ago at Dikika, Ethiopia. Nature 2010, 466, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Meijer, P.T.; Krijgsman, W. A quantitative analysis of the desiccation and re-filling of the Mediterranean during the Messinian Salinity Crisis. Earth and Planetary Science Letters 2005, 240, 510–520. [Google Scholar] [CrossRef]
- Melles, M.; Brigham-Grette, J.; Minyuk, P.; Koeberl, C.; Andreev, A.; Cook, T.; Fedorov, G.; Gebhardt, C.; Haltia-Hovi, E.; Kukkonen, M.; Nowaczyk, N.; Schwamborn, G.; Wennrich, B. El’gygytgyn Scientific Party (2011): The Lake El’gygytgyn Scientific Drilling Project – Conquering Arctic Challenges through Continental Drilling. Scientific Drilling 11, 29–40. [CrossRef]
- Mix, H.T.; Winnick, M.J.; Mulch, A.; Chamberlain, C.P. Grassland expansion as an instrument of hydrologic change in Neogene western North America. Earth and Planetary Science Letters 2013, 377–378, 73–83. [Google Scholar] [CrossRef]
- Mologni, C.; Revel, M.; Chaumillon, E.; Malet, E.; Coulombier, T.; Sabatier, P.; Brigode, P.; Hervé, G.; Develle, A.-L.; Schenini, L.; Messous, M.; Davtian, G.; Carré, A.; Bosch, D.; Volto, N.; Ménard, C.; Khalidi, L.; Arnaud, F. 50-year seasonal variability in East African droughts and floods recorded in central Afar lake sediments (Ethiopia) and their connections with the El Niño–Southern Oscillation. Climate odell Past 2024, 20, 1837–1860. [Google Scholar] [CrossRef]
- Morgan, T.J.H., Uomini, N.T., Rendell, L.E., Chouinard-Thuly, L., Street, S.E., Lewis, H.M. Cross, C.P., Evans, C., Kearney, R., de la Torre, I., Whiten, A., Laland, K.N. (2015): Experimental evidence for the co-evolution of hominin tool-making teaching and language. Nature Communications 6, 6029. [CrossRef]
- Müller, J.; Massé, G.; Stein, R.; Belt, S.T. Variability of sea-ice conditions in the Fram Strait over the past 30,000 years. Nature Geoscience 2009, 2, 772–776. [Google Scholar] [CrossRef]
- Nicolis, G.; Prigogine, I. Die Erforschung des Komplexen; Piper; München, 1987. [Google Scholar]
- Nigel, P. Africa’s Great Rift Valley; Harry N. Abrams; New York, 2001. [Google Scholar]
- Organ, C.; Nunn, C.L.; Machanda, Z.; Wrangham, R.W. Phylogenetic rate shifts in feeding time during the evolution of Homo. PNAS 2011, 108, 14555–14559. [Google Scholar] [CrossRef]
- Oparin, A.I. Das Leben, seine Natur, Herkunft und Entwicklung; Gustav Fischer; Jena, 1963. [Google Scholar]
- Orlando, L.; Ginolhac, A.; Zhang, G.; Froese, D.; Albrechtsen, A.; Stiller, M.; Schubert, M.; Cappellini, E.; Petersen, B.; Moltke, I.; Johnson, P.L.F.; Fumagalli, M.; Vilstrup, J.T.; Raghavan, M.; Korneliussen, T.; Malaspinas, A.-S.; Vogt, J.; Szklarczyk, D.; Kelstrup, C.D.; Vinther, J.; Dolocan, A.; Stenderup, J.; Velazquez, A.M.V.; Cahill, J.; Rasmussen, M. Recalibrating Equus evolution using the genome sequence of an early Middle Pleistocene horse. Nature 2013, 499, 74–78. [Google Scholar] [CrossRef]
- Otto, S.P.; Day, T. A biologists guide to mathematical modeling in ecology and evolution; Princeton University Press; Princeton, NJ, 2007. [Google Scholar]
- Owen Lovejoy, C. The Origin of Man. Science 1981, 211, 341–350. [Google Scholar] [CrossRef]
- Oxtoby, D.W. Nucleation of First-Order Phase Transitions. Accounts of Chemical Research 1998, 31, 91–97. [Google Scholar] [CrossRef]
- Palombit, R.A. Kappeler, P.M., Silk, J.B., Eds.; Chapter 3. Conflict and Bonding Between the Sexes. In Mind the Gap; Springer; Berlin Heidelberg, 2010. [Google Scholar] [CrossRef]
- Pawłowski, B.; Żelaźniewicz, A. The evolution of perennially enlarged breasts in women: a critical review and a novel hypothesis. Biological reviews of the Cambridge Philosophical Society 2021, 96, 2794–2809. [Google Scholar] [CrossRef]
- Pennisi, E. The Burdens of Being a Biped. Science 2012, 336, 974. [Google Scholar] [CrossRef]
- Pilbeam, D.R.; Lieberman, D.E. Muller, M.N., Wrangham, R.W., Pilbeam, D.R., Eds.; Reconstructing the Last Common Ancestor of Chimpanzees and Humans. In Chimpanzees and Human Evolution; Belknap Press of Harvard University Press; Cambridge, MA, 2017; pp. 22–141. [Google Scholar]
- Pizzari, T.; Snook, R.R. Perspective: Sexual conflict and sexual selection: Chasing away paradigm shifts. Evolution 2003, 57, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- Plummer, T.W.; et al. Expanded geographic distribution and dietary strategies of the earliest Oldowan hominins and Paranthropus. Science 2023, 379, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Prado-Martinez, J., Sudmant, P.H., Kidd, J.M., Li, H., Kelley, J.L., Lorente-Galdos, B., Veeramah, K.R., Woerner, A.E., O’Connor, T.D., Santpere, G., Cagan, A., Theunert, C., Casals, F., Laayouni, H., Munch, K., Hobolth, A., Halager, A.E., Malig, M., Hernandez-Rodriguez, J., Hernando-Herraez, I., Prüfer, K., Pybus, M., Johnstone, L., Lachmann, M., Alkan, C., Twigg, D., Petit, N., Baker, C., Hormozdiari, F., Fernandez-Callejo, M., Dabad, M., Wilson, M.L., Stevison, L., Camprubi, C., Carvalho, T., Ruiz-Herrera, A., Vives, L., Mele, M., Abello, T., Kondova, I., Bontrop, R.E., Pusey, A., Lankester, F., Kiyang, J.A., Bergl, R.A., Lonsdorf, E., Myers, S., Ventura, M., Gagneux, P., Comas, D., Siegismund, H., Blanc, J., Agueda-Calpena, L., Gut, M., Fulton, L., Tishkoff, S.A., Mullikin, J.C., Wilson, R.K., Gut, I.G., Gonder, M.K., Ryder, O.A., Hahn, B.H., Navarro, A., Akey, J.M., Bertranpetit, J., Reich, D., Mailund, T., Schierup, M.H., Hvilsom, C., Andres, A.M., Wall, J.D., Bustamante, C.D., Hammer, M.F., Eichler, E.E., Tomas Marques-Bonet, T. (2013): Great ape genetic diversity and population history. Nature 499, 471-475. [CrossRef]
- Prigogine, I.; Nicolis, G.; Babloyantz, A. Thermodynamics of Evolution. Physics Today 1972, 25, 23–28. [Google Scholar] [CrossRef]
- Prigogine, I.; Stengers, I. Dialog mit der Natur; Piper; München, 1981. [Google Scholar]
- Prum, R.O. The Evolution of Beauty; Doubleday; New York, USA, 2017. [Google Scholar]
- Reichholf, J.H. Das Rätsel der Menschwerdung. Die Entstehung des Menschen im Wechselspiel der Natur; Dtv; München, 2004. [Google Scholar]
- Richmond, B.G.; Jungers, W.L. Orrorin tugenensis Femoral Morphology and the Evolution of Hominin Bipedalism. Science 2008, 319, 1662–1665. [Google Scholar] [CrossRef]
- Rime, V.; Negga, H.; Fentimen, R.; Rüggeberg, A.; El Korh, A.; Pirkenseer, C.; Schaegis, J.-C.; Hajdas, I.; Adatte, T.; Atnafu, B.; Kidane, T.; Foubert, A. Nature and significance of Late Pleistocene to Holocene thick evaporite deposits of the Danakil Depression, Afar, Ethiopia. Sedimentology 2025, 72, 475–506. [Google Scholar] [CrossRef]
- Roach, N.T.; Venkadesan, M.; Rainbow, M.J.; Lieberman, D.E. Elastic energy storage in the shoulder and the evolution of high-speed throwing in Homo. Nature 2013, 498, 483–486. [Google Scholar] [CrossRef]
- Robert, C.; Diester-Haass, L.; Paturel, J. Clay mineral assemblages, siliciclastic input and paleoproductivity at ODP Site 1085 off Southwest Africa: A late Miocene–early Pliocene history of Orange river discharges and Benguela current activity, and their relation to global sea level change. Marine Geology 2009, 216, 221–238. [Google Scholar] [CrossRef]
- Roberts, A. Evolution. The Human Story; Dorling Kindersley; London, 2011. [Google Scholar]
- Rosales-Reynoso, M.A.; Juárez-Vázquez, C.I.; Barros-Núñez, P. Evolution and genomics of the human brain. Neurología 2018, 33, 211–282. [Google Scholar] [CrossRef]
- Sandel, A.A.; et al. Lethal conflict after group fission in wild chimpanzees. Science 2026, 392, 216–220. [Google Scholar] [CrossRef]
- Schmelzer, J.W.P. Nucleation Theory and Applications; Wiley VCH; Weinheim, 2005. [Google Scholar] [CrossRef]
- Schrenk, F.; Kullmer, O.; Sandrock, O.; Bromage, T.G. Early Hominid diversity, age and biogeography of the Malawi-Rift. Human Evolution 2002, 17, 113–122. [Google Scholar] [CrossRef]
- Schrödinger, E. (1944): What is Life? The Physical Aspect of the Living Cell; Cambridge University Press; Cambridge, UK.
- Semaw, S.; Rogers, M.J.; Quade, J.; Renne, P.R.; Butler, R.F.; Domínguez-Rodrigo, M.; Stout, D.; Hart, W.S.; Pickering, T.; Simpson, S.W. 2.6-Million-year-old stone tools and associated bones from OGS-6 and OGS-7, Gona, Afar, Ethiopia. Journal of Human Evolution 2003, 45, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Sen, S. A review of the Pleistocene dwarfed elephants from the Aegean islands, and their paleogeographic context. Fossil Imprint 2017, 73, 76–92. [Google Scholar] [CrossRef]
- Senut, B.; Pickford, M.; Ségalen, L. Neogene desertification of Africa. Comptes Rendus Geoscience 2009, 341, 591–602. [Google Scholar] [CrossRef]
- Shaw, A.K.; Couzin, I.D. Migration or Residency? The Evolution of Movement Behavior and Information Usage in Seasonal Environments. The American Naturalist 2013, 181, 114–124. [Google Scholar] [CrossRef]
- Smith, I.G.; Reynolds, S.C. Out of Afar: the first hominin migration? Long-term landscape changes in the Afar region and implications for hominin bipedalism. Earth ArXive 2025, preprint. [Google Scholar] [CrossRef]
- Song, H. On the origin of the desert locust Schistocerca gregaria (Forskål) (Orthoptera: Acrididae: Cyrtacanthacridinae). Proc. Biol. Sci. 2004, 271, 1641–1648. [Google Scholar] [CrossRef]
- Spray, J.; Elliott, R. Earth Impact Database. Planetary and Space Science Centre, University of New Brunswick, Fredericton, New Brunswick, Canada. 2013. Available online: http://www.passc.net/ (accessed on 05 Apr 2026).
- Stige, L.C.; Chan, K.-S.; Zhang, Z.; Stenseth, N.C. Thousand-year-long Chinese time series reveals climatic forcing of decadal locust dynamics. PNAS 2007, 104, 16188–16193. [Google Scholar] [CrossRef]
- Stinchcomb, G.E.; Quade, J.; Levin, N.E.; Iverson, N.; Dunbar, N.; McIntosh, W.; Arnold, L.J.; Demuro, M.; Duval, M.; Grün, R.; Zhao, J.; White, M.; Hynek, S.A.; Brown, F.H.; Rogers, M.J.; Semaw, S. Fluvial response to Quaternary hydroclimate in eastern Africa: Evidence from Gona, Afar, Ethiopia. Quaternary Science Reviews 2023, 309, 108083. [Google Scholar] [CrossRef]
- Stone, R. Battle for the Americas. Science 2013, 341, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Suhr, D. Das Mosaik der Menschwerdung; Springer; Heidelberg, 2018. [Google Scholar]
- Sutou, S. The Hairless Mutation Hypothesis: a Driving Force of Humanization by Enforcing Bipedalism to Hold a Baby, by Allowing Immature Baby Delivery to Enlarge the Brain after Birth, and by Making Use of Fire to Get Meat and to Cook Foods. Genes and Environment 2014, 36, 78–88. [Google Scholar] [CrossRef]
- Sylvestre, F.; Melles, M.; Wennrich, V.; et al. Decadal-scale droughts disrupted the African Humid Period in the Sahara. Nature 2026, 652, 365–370. [Google Scholar] [CrossRef]
- Thomas, E.R.; Wolff, E.W.; Mulvaney, R.; Steffensen, J.P.; Johnsen, S.J.; Arrowsmith, C.; White, J.W.C.; Vaughn, B.; Popp, T. The 8.2 kyr event from Greenland ice cores. Quaternary Science Reviews 2007, 26, 70–81. [Google Scholar] [CrossRef]
- Trauth, M.H.; Maslin, M.A.; Deino, A.; Strecker, M.R. (2005): Late Cenozoic Moisture History of East Africa. Science 309, 2051–2053. [CrossRef]
- Trauth, M.H.; Maslin, M.A.; Deino, A.L.; Junginger, A.; Lesoloyia, M.; Odada, E.O.; Olago, D.O.; Olaka, L.A.; Strecker, M.R.; Tiedemann, R. Human evolution in a variable environment: the amplifier lakes of Eastern Africa. Quaternary Science Reviews 2010, 29, 2981–2988. [Google Scholar] [CrossRef]
- Treves, A.; Palmqvist, P. Gursky, S.L., Nekaris, K.A.I., Eds.; Reconstructing Hominin Interactions with Mammalian Carnivores (6.0–1.8 Ma). In Primate Anti-Predator Strategies. Developments in Primatology: Progress and Prospects.; Springer; Boston, MA, 2007. [Google Scholar] [CrossRef]
- Uvarov, B.P. A revision of the genus Locusta, L. ( = Pachytylus, Fieb.), with a new theory as to the periodicity and migrations of locusts. Bulletin of Entomological Research 1921, 12, 135–163. [Google Scholar] [CrossRef]
- Uvarov, B.P. The aridity factor in the ecology of locusts and grasshoppers of the Old World. In Unesco Report NS/AZ/204; Paris, 1955. [Google Scholar]
- Van Soest, P.J. Nutritional ecology of the ruminant; Cornell University Press; Ithaca, 1994. [Google Scholar]
- Villmoare, B.; Delezene, L.K.; Rector, A.L.; et al. New discoveries of Australopithecus and Homo from Ledi-Geraru, Ethiopia. Nature 2025, 650, 374–380. [Google Scholar] [CrossRef]
- Wasmund, N.; Siegel, H. Feistel, R., Nausch, G., Wasmund, N., Eds.; Chapter 15: Phytoplankton. In State and Evolution of the Baltic Sea 1952-2005; Wiley-Interscience; Hoboken, NJ, 2008. [Google Scholar] [CrossRef]
- Wei, G.B.; Hu, S.M.; Yu, K.F.; Hou, Y.M.; Li, X.; Jin, C.Z.; Wang, Y.; Zhao, J.X.; Wang, W.H. New materials of the steppe mammoth, Mammuthus trogontherii, with discussion on the origin and evolutionary patterns of mammoths. Science China Earth Sciences 2010, 53, 956–963. [Google Scholar] [CrossRef]
- Wei, Z.; Lee, X.; Aemisegger, F.; et al. A global database of water vapor isotopes measured with high temporal resolution infrared laser spectroscopy. Scientific Data 2019, 6, 180302. [Google Scholar] [CrossRef]
- Weninger, B.; Schulting, R.; Bradtmöller, M.; Clare, L.; Collard, M.; Edinborough, K.; Hilpert, J.; Jöris, O.; Niekus, M.; Rohling, E.J.; Wagner, B. The catastrophic final flooding of Doggerland by the Storegga Slide tsunami. Documenta Praehistorica 2008, 35, 1–24. [Google Scholar] [CrossRef]
- Weßling, B. Der Ruf der Kraniche: Expeditionen in eine geheimnisvolle Welt; Wilhelm Goldmann; München, 2020. [Google Scholar]
- Wilson, E.O.; Bossert, W.H. Einführung in die Populationsbiologie. In American original (1971): A Primer of Population Biology; Springer, Berlin-Heidelberg-New York; Sinauer Associates; Stamford, CT, 1973. [Google Scholar]
- Wiki Lucy. 2025a. Available online: https://de.wikipedia.org/wiki/Lucy (accessed on 01 Apr 2026).
- Wiki (2026a): Savannah hypothesis. Available online: https://en.wikipedia.org/wiki/Savannah_hypothesis (accessed on 30 Mar 2026).
- Wiki (2026b): Animal migration. Available online: https://en.wikipedia.org/wiki/Animal_migration (accessed on 01 Apr 2026).
- Wiki (2026c): Afar Triangle. Available online: https://en.wikipedia.org/wiki/Afar_Triangle (accessed on 06 Apr 2026).
- Wiki (2026d): 8.2-kiloyear event. Available online: https://en.wikipedia.org/wiki/8.2-kiloyear_event (accessed on 11 Apr 2026).
- Wiki (2026e): 2019–2022 locust infestation. Available online: https://en.wikipedia.org/wiki/2019%E2%80%932022_locust_infestation (accessed on 13 Apr 2026).
- Wiki (2026f): Hopf bifurcation. Available online: https://en.wikipedia.org/wiki/Hopf_bifurcation (accessed on 14 Apr 2026).
- Wiki (2026g): Ngorongoro Conservation Area. Available online: https://en.wikipedia.org/wiki/Ngorongoro_Conservation_Area (accessed on 28 Apr 2026).

| 1 | Cordan (1962): p. 30: Nicht Ruhm noch Größe wird sein, bis der Mensch erscheint, bis der Mensch geschaffen. |
| 2 | Original quoted text: „Das wahrscheinlich wichtigste Merkmal, das alle Vertreter der menschlichen Linie eint und das, nach derzeitigem Wissensstand, nicht parallel entstanden ist, ist der aufrechte Gang. Die zweibeinige Fortbewegung, auch Bipedie genannt, ist die eigentliche Revolution zu Beginn der menschlichen Evolution. … Selbst bei gut dokumentierten Skeletten, wie beispielsweise dem von »Lucy« (Australopithecus afarensis), gab es einen hitzigen wissenschaftliche Disput … um die Frage, inwieweit … eine modern zu nennende Zweibeinigkeit entwickelt war oder ob »Lucy« doch überwiegend auf Bäumen lebte.“ |
| 3 | Original quoted text: „Die Erfolgsgeschichte des Menschen begann offenbar damit, dass einige pliozäne Arten eine habituelle zweibeinige Lebensweise entwickelten, während die Verkürzung und Verkleinerung des Kauapparats und die typisch menschliche Hirnentfaltung erst viel später mit dem Auftreten der Gattung Homo folgten. … Erst vor rund 2,6 Millionen Jahren sind Populationen des frühen Homo – oder von Australopithecus – zu einer dauerhaften Bipedie übergegangen.“ |
| 4 | Original quoted text: „Legt man das heutige Verhältnis zwischen wandernden und ortsfesten (residenten) Großtieren in Ostafrika zugrunde, dann übertreffen die Wandernden die Residenten um das Zehnfache“ |
| 5 | Original quoted text: „Wenn Saisonalität als die alternierende Abfolge von Perioden der Nahrungsverknappung und des Nahrungsreichtums angesehen wird, dann kann Saisonalität als ein Faktor bezeichnet werden, der zu intensiverer Selektion und letztlich zu evolutionärem Wandel beiträgt“ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




