Submitted:
30 April 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Advantages and Disadvantages of Hydrogels in Wound Treatment, with Specific Emphasis on Radiation Wounds
3. Hydrogel Classification Based on Their Mechanism of Action in the Pathophysiology of Damage
3.1. Hydrogels Preventing Oxidative Stress
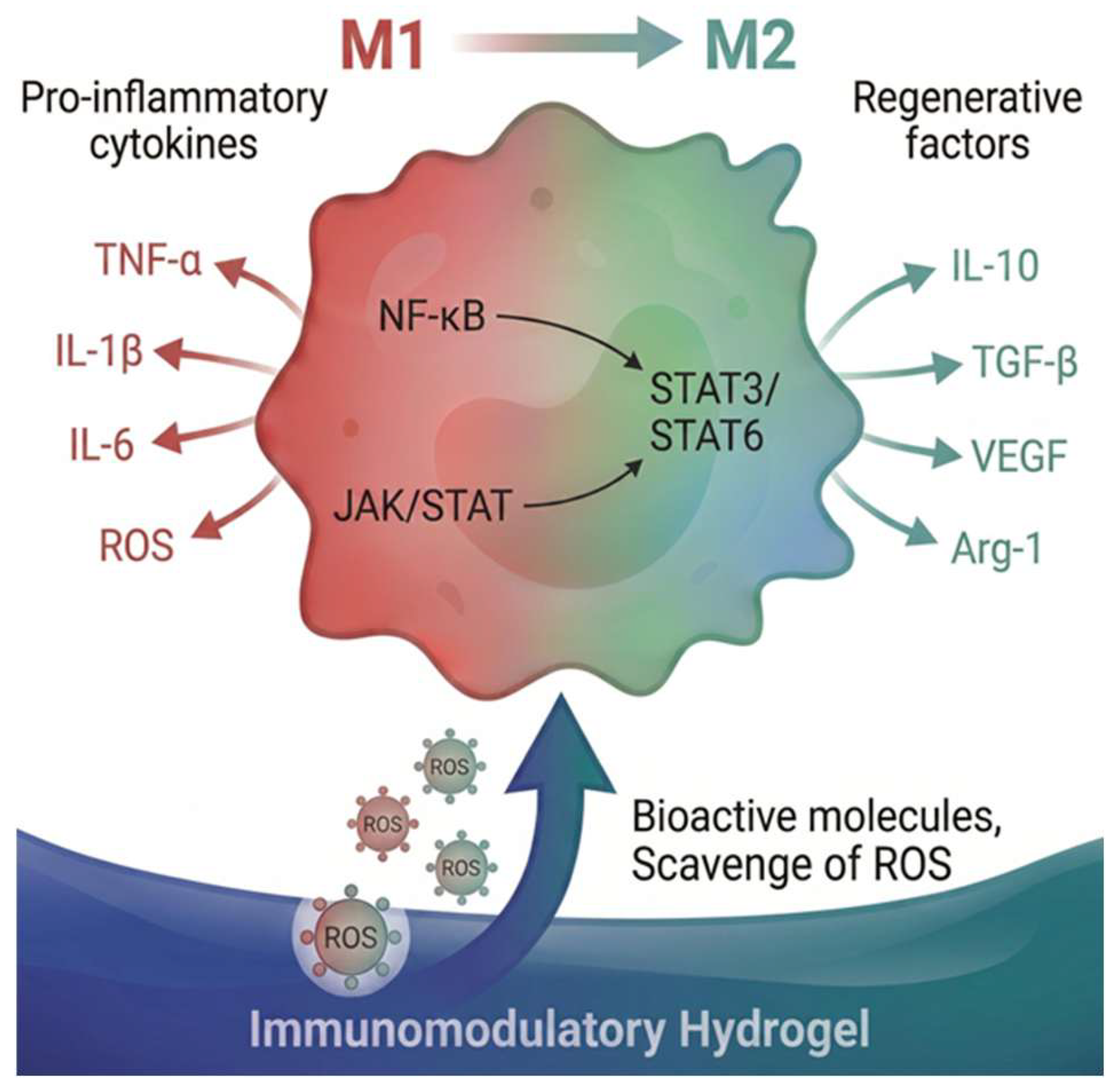
3.2. Immunomodulatory Hydrogels
3.3. Proangiogenic Hydrogels
3.4. Hydrogels Promoting Regeneration and Remodeling
4. Hydrogel-Based Strategies in the Treatment of Radiation-Induced Injuries Across Different Tissues and Organ Systems
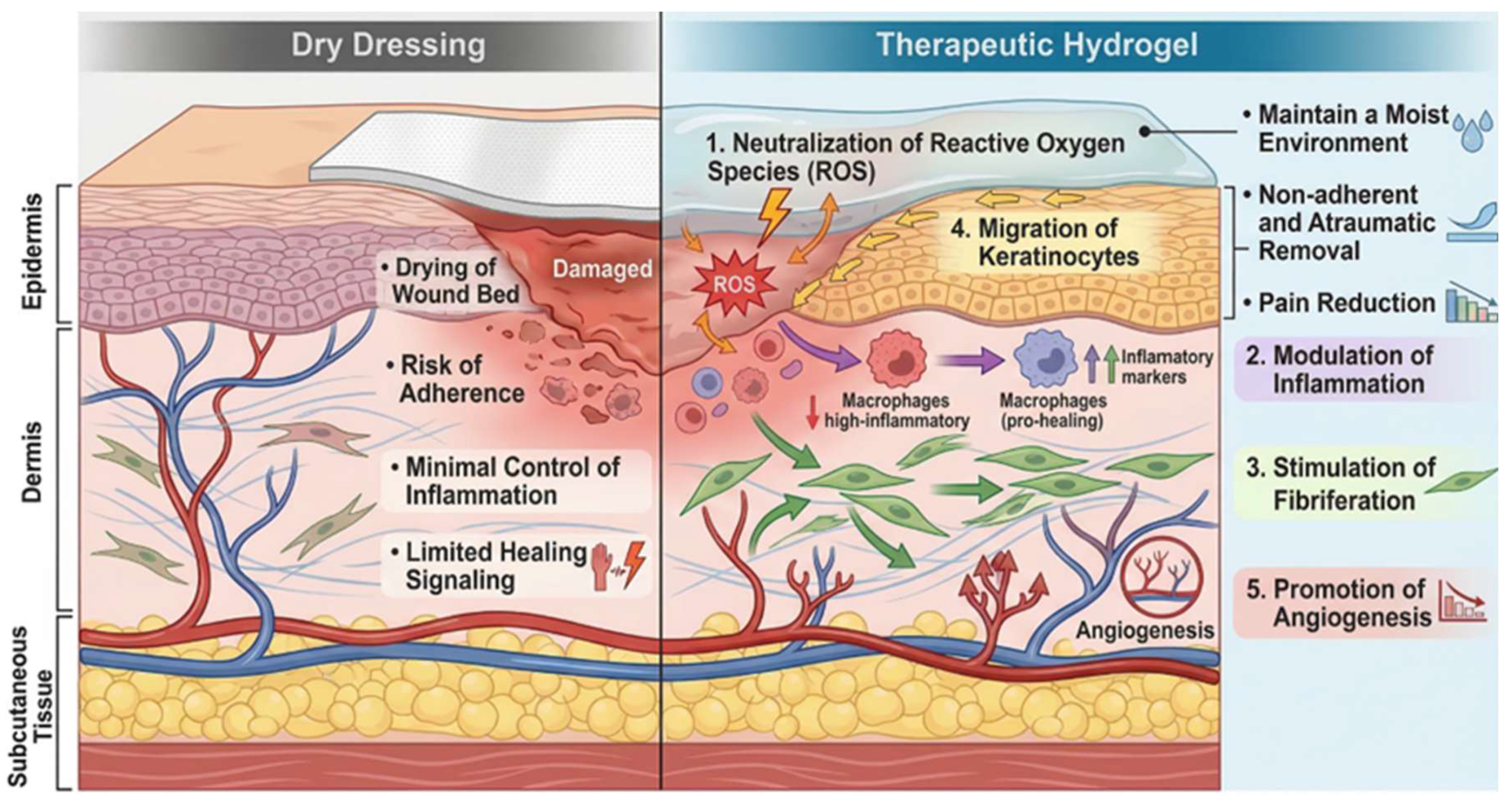
4.1. Skin
4.2. Mucosa
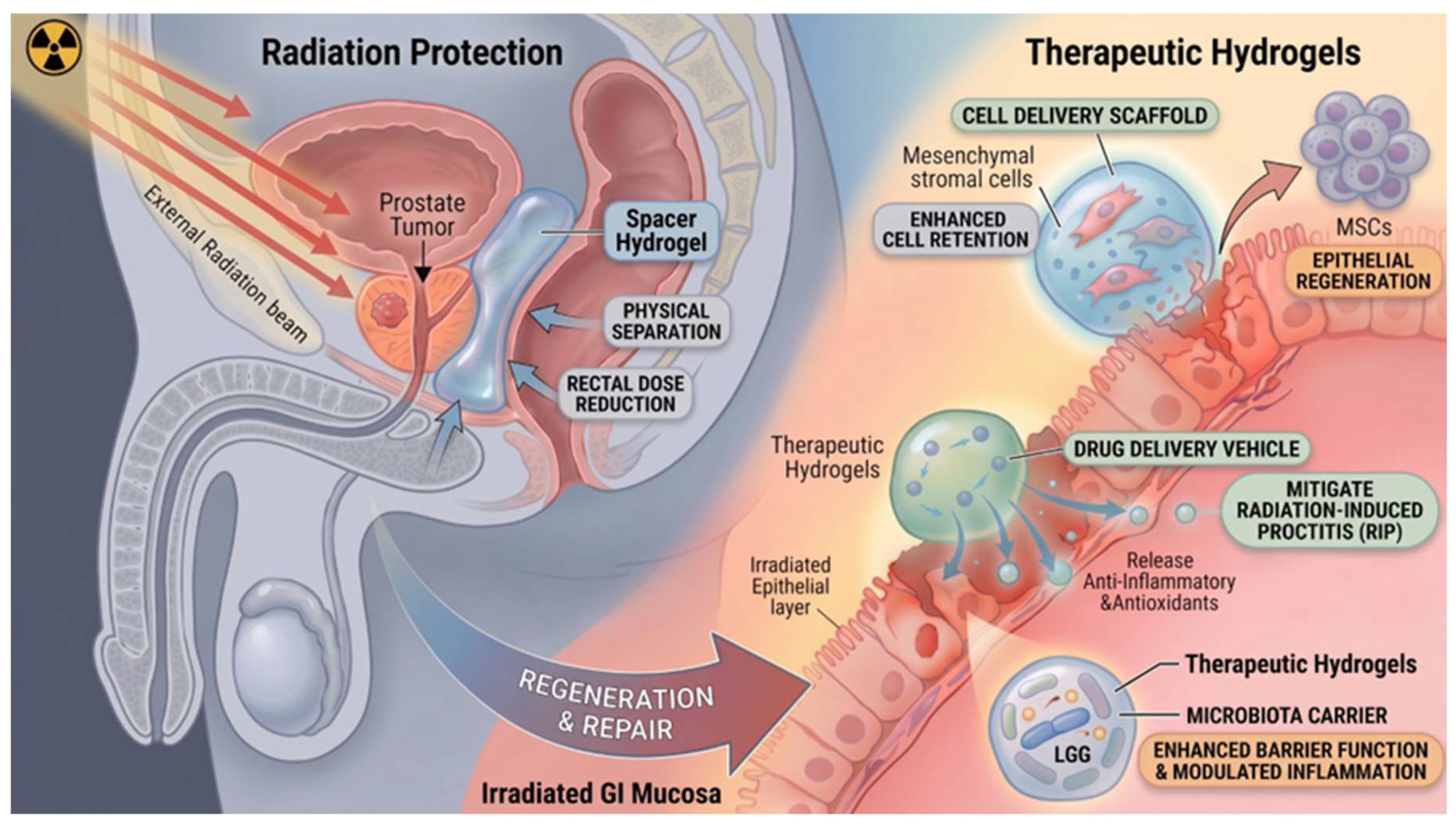
4.3. Gastrointestinal Tract
4.4. Lungs
4.5. Heart
4.6. Bones
5. Summary
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADSC | Adipose-derived stem cell |
| eNOS | Endothelial nitric oxide synthase |
| ECM | Extracellular matrix |
| EMT | Epithelial-to-mesenchymal transition |
| GF | Growth factor |
| IL | Interleukin |
| JAK/STAT | Janus kinase/signal transducer and activator of transcription |
| MSC | Mesenchymal stem cell |
| NF-kB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| PEG | Polyethylene glycol |
| RIHD | Radiation-induced heart disease |
| RILI | Radiation-induced lung injury |
| RIOM | Radiation-induced oral mucositis |
| ROS | Reactive oxygen species |
| TGF | Transforming growt factor |
| TNF | Tumor necrosis factor |
| VEGF | Vascular endothelial growth factor |
References
- Ho, T.-C.; Chang, C.-C.; Chan, H.-P.; Chung, T.-W.; Shu, C.-W.; Chuang, K.-P.; Duh, T.-H.; Yang, M.-H.; Tyan, Y.-C. Hydrogels: Properties and Applications in Biomedicine. Molecules 2022, 27, 2902. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Wichterle, O.; Lím, D. Hydrophilic Gels for Biological Use. Nature 1960, 185, 117–118. [Google Scholar] [CrossRef]
- Xiang, J.; Shen, L.; Hong, Y. Status and future scope of hydrogels in wound healing: Synthesis, materials and evaluation. Eur. Polym. J. 2020, 130. [Google Scholar] [CrossRef]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal Transduct. Target. Ther. 2021, 6, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Song, A.; Rane, A.A.; Christman, K.L. Antibacterial and cell-adhesive polypeptide and poly(ethylene glycol) hydrogel as a potential scaffold for wound healing. Acta Biomater. 2012, 8, 41–50. [Google Scholar] [CrossRef]
- Gounden, V.; Singh, M. Hydrogels and Wound Healing: Current and Future Prospects. Gels 2024, 10, 43. [Google Scholar] [CrossRef]
- Britto, E.J.; Nezwek, T.A.; Popowicz, P.; Robins, M. Wound Dressings 2024. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Macmillan, M.S.; Wells, M.; MacBride, S.; Raab, G.M.; Munro, A.; MacDougall, H. Randomized Comparison of Dry Dressings Versus Hydrogel in Management of Radiation-Induced Moist Desquamation. Int. J. Radiat. Oncol. 2007, 68, 864–872. [Google Scholar] [CrossRef]
- Demeter, M.; Scărișoreanu, A.; Călina, I. State of the Art of Hydrogel Wound Dressings Developed by Ionizing Radiation. Gels 2023, 9, 55. [Google Scholar] [CrossRef]
- Su, Y.; Cui, H.; Yang, C.; Li, L.; Xu, F.; Gao, J.; Zhang, W. Hydrogels for the treatment of radiation-induced skin and mucosa damages: An up-to-date overview. Front. Mater. 2022, 9. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, H.; He, Y.; Li, M.; Gao, J.; Han, Z.; Zhou, J.; Li, J. Hydrogel-Based Strategies for the Prevention and Treatment of Radiation-Induced Skin Injury: Progress and Mechanistic Insights. Biomimetics 2025, 10, 758. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Salman, S.; Khan, S.A.; Amin, A.; Rahman, Z.U.; Al-Ghamdi, Y.O.; Akhtar, K.; Bakhsh, E.M.; Khan, S.B. Versatility of Hydrogels: From Synthetic Strategies, Classification, and Properties to Biomedical Applications. Gels 2022, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Ullah, F.; Othman, M.B.H.; Javed, F.; Ahmad, Z.; Akil, H.M. Classification, processing and application of hydrogels: A review. Mater. Sci. Eng. C 2015, 57, 414–433. [Google Scholar] [CrossRef]
- Xue, J.; Yu, C.; Tang, Y.; Mo, W.; Tang, Z.; Sheng, W.; Jiao, Y.; Zhu, W.; Cao, J. NF-E2-Related Factor 2 (Nrf2) Ameliorates Radiation-Induced Skin Injury. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Averbeck, D.; Rodriguez-Lafrasse, C. Role of Mitochondria in Radiation Responses: Epigenetic, Metabolic, and Signaling Impacts. Int. J. Mol. Sci. 2021, 22, 11047. [Google Scholar] [CrossRef]
- Azzam, E.I.; Jay-Gerin, J.-P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett. 2012, 327, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wei, J.; Wang, X.; Zhao, Q.; Lv, J.; Tan, Z.; Xin, Y.; Jiang, X. Radiation-induced skin reactions: oxidative damage mechanism and antioxidant protection. Front. Cell Dev. Biol. 2024, 12, 1480571. [Google Scholar] [CrossRef]
- Tang, R.; Yin, J.; Liu, Y.; Xue, J. FLASH radiotherapy: A new milestone in the field of cancer radiotherapy. Cancer Lett. 2024, 587, 216651. [Google Scholar] [CrossRef]
- Calaf, G.M.; Crispin, L.A.; Muñoz, J.P.; Aguayo, F.; Narayan, G.; Roy, D. Cell Adhesion Molecules Affected by Ionizing Radiation and Estrogen in an Experimental Breast Cancer Model. Int. J. Mol. Sci. 2022, 23, 12674. [Google Scholar] [CrossRef]
- Melia, E.; Parsons, J.L. DNA damage and repair dependencies of ionising radiation modalities. Biosci. Rep. 2023, 43. [Google Scholar] [CrossRef]
- Dent, P.; Yacoub, A.; Fisher, P.B.; Hagan, M.P.; Grant, S. MAPK pathways in radiation responses. Oncogene 2003, 22, 5885–5896. [Google Scholar] [CrossRef]
- Liermann-Wooldrik, K.T.; Kosmacek, E.A.; McDowell, J.A.; Takkar, S.; Murthy, D.; Singh, P.K.; Schott, M.B.; Ponnusamy, M.P.; Oberley-Deegan, R.E. Radiation Promotes Acute and Chronic Damage to Adipose Tissue. Int. J. Mol. Sci. 2025, 26, 5626. [Google Scholar] [CrossRef]
- Wei, J.; Wang, B.; Wang, H.; Meng, L.; Zhao, Q.; Li, X.; Xin, Y.; Jiang, X. Radiation-Induced Normal Tissue Damage: Oxidative Stress and Epigenetic Mechanisms. Oxidative Med. Cell. Longev. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Cheng, J.; Dong, J.; Fang, Y.; Zhang, X.; Dang, X. Radiation-induced skin injury: a review of pathophysiology, assessment, management, and re-irradiation protocols. Front. Oncol. 2026, 15. [Google Scholar] [CrossRef]
- Cui, J.; Wang, T.-J.; Zhang, Y.-X.; She, L.-Z.; Zhao, Y.-C. Molecular biological mechanisms of radiotherapy-induced skin injury occurrence and treatment. Biomed. Pharmacother. 2024, 180, 117470. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhu, J.; Liu, Y.; Zhou, P.; Gu, Y. Mechanisms of radiation-induced tissue damage and response. Medcomm 2024, 5, e725. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.T.; Vulin, A.; Hendry, J.H. Human epidermal stem cells: Role in adverse skin reactions and carcinogenesis from radiation. 2016, 770, 349–368. [Google Scholar] [CrossRef]
- Huang, C.; Huangfu, C.; Bai, Z.; Zhu, L.; Shen, P.; Wang, N.; Li, G.; Deng, H.; Ma, Z.; Zhou, W.; et al. Multifunctional carbomer based ferulic acid hydrogel promotes wound healing in radiation-induced skin injury by inactivating NLRP3 inflammasome. J. Nanobiotechnology 2024, 22, 1–18. [Google Scholar] [CrossRef]
- Shen, J.; Jiao, W.; Yang, J.; Zhuang, B.; Du, S.; Wu, Y.; Huang, G.; Zhang, Y.; Wang, Y.; Xu, C.; et al. In situ photocrosslinkable hydrogel treats radiation-induced skin injury by ROS elimination and inflammation regulation. Biomaterials 2024, 314, 122891. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Sun, M.; Li, D.; Zhang, T.; Li, J.; Zhou, D. An IFI6-based hydrogel promotes the healing of radiation-induced skin injury through regulation of the HSF1 activity. J. Nanobiotechnology 2022, 20, 1–14. [Google Scholar] [CrossRef]
- Liu, Z.; Gu, J.; Gao, Y.; Hu, H.; Jiang, H. ADSC-derived exosomes mitigate radiation-induced skin injury by reducing oxidative stress, inflammation and cell death. Front. Public Heal. 2025, 13, 1603431. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Ding, A. Nonresolving Inflammation. Cell 2010, 140, 871–882. [Google Scholar] [CrossRef]
- Peña, O.A.; Martin, P. Cellular and molecular mechanisms of skin wound healing. Nat. Rev. Mol. Cell Biol. 2024, 25, 599–616. [Google Scholar] [CrossRef]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef]
- Au, N.P.B.; Wu, T.; Kumar, G.; Jin, Y.; Li, Y.Y.T.; Chan, S.L.; Lai, J.H.C.; Chan, K.W.Y.; Yu, K.N.; Wang, X.; et al. Low-dose ionizing radiation promotes motor recovery and brain rewiring by resolving inflammatory response after brain injury and stroke. Brain Behav. Immun. 2023, 115, 43–63. [Google Scholar] [CrossRef]
- Chen, J.; Liu, X.; Zeng, Z.; Li, J.; Luo, Y.; Sun, W.; Gong, Y.; Zhang, J.; Wu, Q.; Xie, C. Immunomodulation of NK Cells by Ionizing Radiation. Front. Oncol. 2020, 10, 874. [Google Scholar] [CrossRef]
- Nakashima, H.; Nakashima, M.; Kinoshita, M.; Ikarashi, M.; Miyazaki, H.; Hanaka, H.; Imaki, J.; Seki, S. Activation and increase of radio-sensitive CD11b+ recruited Kupffer cells/macrophages in diet-induced steatohepatitis in FGF5 deficient mice. Sci. Rep. 2016, 6, 34466. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Vuckovic, I.; Zhang, S.; Xiong, Y.; Carlson, B.L.; Jacobs, J.; Olson, I.; Petterson, X.-M.; Macura, S.I.; Sarkaria, J.; et al. Radiation Induced Metabolic Alterations Associate With Tumor Aggressiveness and Poor Outcome in Glioblastoma. Front. Oncol. 2020, 10, 535. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Li, T.; Niu, M.; Zhang, H.; Wu, Y.; Wu, K.; Dai, Z. Targeting cytokine and chemokine signaling pathways for cancer therapy. Signal Transduct. Target. Ther. 2024, 9, 1–48. [Google Scholar] [CrossRef]
- Mai, Y.; Wang, H.; Lu, J.; Shi, S.; Cai, Y.; Zhang, W.; Xie, S.; Huang, R.; Ji, S.; Qu, X. Catalyst-modulated hydrogel dynamics for decoupling viscoelasticity and directing macrophage fate for diabetic wound healing. Bioact. Mater. 2025, 52, 878–895. [Google Scholar] [CrossRef]
- Chou, R.; Dana, T.; Bougatsos, C.; Blazina, I.; Starmer, A.J.; Reitel, K.; Buckley, D.I. Pressure Ulcer Risk Assessment and Prevention. Ann. Intern. Med. 2013, 159, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Rakotoarisoa, M.; Angelov, B.; Drechsler, M.; Nicolas, V.; Bizien, T.; Gorshkova, Y.E.; Deng, Y.; Angelova, A. Liquid crystalline lipid nanoparticles for combined delivery of curcumin, fish oil and BDNF: In vitro neuroprotective potential in a cellular model of tunicamycin-induced endoplasmic reticulum stress. Smart Mater. Med. 2022, 3, 274–288. [Google Scholar] [CrossRef]
- Huang, R.; Sun, W.; Li, W.; Hu, R.; Meng, R.; Peng, Z.; Yang, R.; Huang, T.; Du, J.; Shang, L.; et al. Immunomodulatory hydrogel patches loaded with curcumin and tannic acid assembled nanoparticles for radiation dermatitis repair and radioprotection. Chem. Eng. J. 2024, 500. [Google Scholar] [CrossRef]
- Fu, Y.; Shi, Y.; Wang, L.; Zhao, Y.; Wang, R.; Li, K.; Zhang, S.; Zha, X.; Wang, W.; Zhao, X.; et al. All-Natural Immunomodulatory Bioadhesive Hydrogel Promotes Angiogenesis and Diabetic Wound Healing by Regulating Macrophage Heterogeneity. Adv. Sci. 2023, 10. [Google Scholar] [CrossRef]
- Jin, C.; Liang, J.; Wu, J.; Han, X.; Zhou, Y.; Li, B.; Sun, W.; Su, J.; Sun, J.; Wan, S.; et al. Temporal Immunomodulatory Hydrogel Regulating the Immune-Osteogenic Cascade for Infected Bone Defects Regeneration. Adv. Mater. 2025, 38, e14419. [Google Scholar] [CrossRef]
- Yang, P.; Hu, Y.; Ju, Y.; Hsiung, N.; Ye, J.; Jian, A.; Lei, L.; Fang, B. DNA-Inspired Multi-Functional Double-Cross-Linking Self-Healing Hydrogel Promotes the Healing of Diabetic Wounds. Adv. Sci. 2025, 13, e13784. [Google Scholar] [CrossRef]
- Mehnath, S.; Karthikeyan, K.; Jeyaraj, M. Mechanical Force on Hydrogel Implication on Enhanced Drug Release, Antibacterial, and M2 Macrophage Polarization: New Insights Alleviate Diabetic Wound Healing. ACS Appl. Mater. Interfaces 2024. [Google Scholar] [CrossRef]
- Zhou, P.; Yan, B.; Wei, B.; Fu, L.; Wang, Y.; Wang, W.; Zhang, L.; Mao, Y. Quercetin-solid lipid nanoparticle-embedded hyaluronic acid functionalized hydrogel for immunomodulation to promote bone reconstruction. Regen. Biomater. 2023, 10, rbad025. [Google Scholar] [CrossRef]
- Abe, J.-I.; Allen, B.G.; Beyer, A.M.; Lewandowski, D.; Mapuskar, K.A.; Subramanian, V.; Tamplin, M.R.; Grumbach, I.M. Radiation-Induced Macrovessel/Microvessel Disease. Arter. Thromb. Vasc. Biol. 2024, 44, 2407–2415. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.-H.; Oh, D.; Na, K.; Kim, H.; Choi, D.; Jung, Y.H.; Ahn, J.; Kim, J.; Kim, C.-H.; Chung, S. Radiation induces acute and subacute vascular regression in a three-dimensional microvasculature model. Front. Oncol. 2023, 13, 1252014. [Google Scholar] [CrossRef]
- Lee, M.-O.; Song, S.-H.; Jung, S.; Hur, S.; Asahara, T.; Kim, H.; Kwon, S.-M.; Cha, H.-J. Effect of Ionizing Radiation Induced Damage of Endothelial Progenitor Cells in Vascular Regeneration. Arter. Thromb. Vasc. Biol. 2012, 32, 343–352. [Google Scholar] [CrossRef]
- Lee, W.H.; Cho, H.J.; Sonntag, W.E.; Lee, Y.W. Radiation Attenuates Physiological Angiogenesis by Differential Expression of VEGF, Ang-1, Tie-2 and Ang-2 in Rat Brain. Radiat. Res. 2011, 176, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Kurtman, C.; Özbilgin, M.K.; Tek, I.; Toprak, S.K. An Experimental Study of Radiation Effect on Normal Tissue: Analysis of HIF-1α, VEGF, eIF2, TIA-1, and TSP-1 Expression. Turk. J. Hematol. 2013, 30, 371–378. [Google Scholar] [CrossRef]
- Müller-Seubert, W.; Ostermaier, P.; Horch, R.E.; Distel, L.; Frey, B.; Erber, R.; Arkudas, A. The Influence of Different Irradiation Regimens on Inflammation and Vascularization in a Random-Pattern Flap Model. J. Pers. Med. 2023, 13, 1514. [Google Scholar] [CrossRef]
- Ria, R.; Cirulli, T.; Giannini, T.; Bambace, S.; Serio, G.; Portaluri, M.; Ribatti, D.; Vacca, A.; Dammacco, F. Serum levels of angiogenic cytokines decrease after radiotherapy in non-Hodgkin lymphomas. Clin. Exp. Med. 2008, 8, 141–145. [Google Scholar] [CrossRef]
- Heissig, B.; Rafii, S.; Akiyama, H.; Ohki, Y.; Sato, Y.; Rafael, T.; Zhu, Z.; Hicklin, D.J.; Okumura, K.; Ogawa, H.; et al. Low-dose irradiation promotes tissue revascularization through VEGF release from mast cells and MMP-9–mediated progenitor cell mobilization. J. Exp. Med. 2005, 202, 739–750. [Google Scholar] [CrossRef]
- Veith, A.P.; Henderson, K.; Spencer, A.; Sligar, A.D.; Baker, A.B. Therapeutic strategies for enhancing angiogenesis in wound healing. Adv. Drug Deliv. Rev. 2019, 146, 97–125. [Google Scholar] [CrossRef]
- Lee, C.; Shim, S.; Jang, H.; Myung, H.; Lee, J.; Bae, C.-H.; Myung, J.K.; Kim, M.-J.; Lee, S.B.; Jang, W.-S.; et al. Human umbilical cord blood–derived mesenchymal stromal cells and small intestinal submucosa hydrogel composite promotes combined radiation-wound healing of mice. Cytotherapy 2017, 19, 1048–1059. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Qian, S.; Chen, J.; Jian, T.; Wang, X.; Zhu, X.; Dong, Y.; Fan, G. Photo-Crosslinked Pro-Angiogenic Hydrogel Dressing for Wound Healing. Int. J. Mol. Sci. 2024, 25, 9948. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Wu, B.; Xiong, Y.; Tao, R.; Panayi, A.C.; Chen, L.; Tian, W.; Xue, H.; Shi, L.; Zhang, X.; et al. Cryogenic 3D printed hydrogel scaffolds loading exosomes accelerate diabetic wound healing. Chem. Eng. J. 2021, 426. [Google Scholar] [CrossRef]
- Siebert, L.; Luna-Cerón, E.; García-Rivera, L.E.; Oh, J.; Jang, J.; Rosas-Gómez, D.A.; Pérez-Gómez, M.D.; Maschkowitz, G.; Fickenscher, H.; Oceguera-Cuevas, D.; et al. Light-Controlled Growth Factors Release on Tetrapodal ZnO-Incorporated 3D-Printed Hydrogels for Developing Smart Wound Scaffold. Adv. Funct. Mater. 2021, 31, 2007555. [Google Scholar] [CrossRef]
- Tao, B.; Lin, C.; Qin, X.; Yu, Y.; Guo, A.; Li, K.; Tian, H.; Yi, W.; Lei, D.; Chen, Y.; et al. Fabrication of gelatin-based and Zn2+-incorporated composite hydrogel for accelerated infected wound healing. Mater. Today Bio 2022, 13, 100216. [Google Scholar] [CrossRef]
- Li, W.; Xie, H.; Gou, L.; Zhou, Y.; Wang, H.; Li, R.; Zhang, Y.; Liu, S.; Liu, J.; Lu, Y.; et al. DNA-Based Hydrogels with Multidrug Sequential Release for Promoting Diabetic Wound Regeneration. JACS Au 2023, 3, 2597–2608. [Google Scholar] [CrossRef]
- Mottareale, R.; Frascogna, C.; La Verde, G.; Arrichiello, C.; Muto, P.; Netti, P.A.; Fusco, S.; Panzetta, V.; Pugliese, M. Impact of ionizing radiation on cell-ECM mechanical crosstalk in breast cancer. Front. Bioeng. Biotechnol. 2024, 12, 1408789. [Google Scholar] [CrossRef]
- Zhu, Y.; Ma, Z.; Kong, L.; He, Y.; Chan, H.F.; Li, H. Modulation of macrophages by bioactive glass/sodium alginate hydrogel is crucial in skin regeneration enhancement. Biomaterials 2020, 256, 120216. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Cheng, Z.; Yu, M.; Liu, K.; Duan, H.; Zhang, Y.; Huang, X.; Li, M.; Li, C.; Hu, Y.; et al. Injectable ECM-mimetic dynamic hydrogels abolish ferroptosis-induced post-discectomy herniation through delivering nucleus pulposus progenitor cell-derived exosomes. Nat. Commun. 2025, 16, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Li, Y.; Yu, M.; Ren, D.; Han, C.; Guo, S. Advances of exosomes in diabetic wound healing. Burn. Trauma 2025, 13, tkae078. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Li, H.; Guo, J.; Wang, D.; Zhang, J.; Liu, J.; Yang, C.; Zhang, Y.; Li, G.; Liu, J. Bio-Inspired Antioxidant Heparin-Mimetic Peptide Hydrogel for Radiation-Induced Skin Injury Repair. Adv. Heal. Mater. 2023, 12, e2203387. [Google Scholar] [CrossRef]
- Cytlak, U.M.; Dyer, D.P.; Honeychurch, J.; Williams, K.J.; Travis, M.A.; Illidge, T.M. Immunomodulation by radiotherapy in tumour control and normal tissue toxicity. Nat. Rev. Immunol. 2021, 22, 124–138. [Google Scholar] [CrossRef]
- Salvo, N.; Barnes, E.; van Draanen, J.; Stacey, E.; Mitera, G.; Breen, D.; Giotis, A.; Czarnota, G.; Pang, J.; De Angelis, C. Prophylaxis and Management of Acute Radiation-Induced Skin Reactions: A Systematic Review of the Literature. Curr. Oncol. 2010, 17, 94–112. [Google Scholar] [CrossRef]
- Nie, S.; Ren, C.; Liang, X.; Cai, H.; Sun, H.; Liu, F.; Ji, K.; Wang, Y.; Liu, Q. Supramolecular Hydrogel-Wrapped Gingival Mesenchymal Stem Cells in Cutaneous Radiation Injury. Cells 2022, 11, 3089. [Google Scholar] [CrossRef]
- Cui, X.; Wang, J.; Xu, X.; Cao, X.; Zhou, Y.; Guo, J. Progress and Application of Multifunctional Hydrogel in Radioactive Skin Injury. Adv. Mater. Interfaces 2025, 12. [Google Scholar] [CrossRef]
- Han, X.; Zhou, C.; Xu, R.; Jia, Z.; Liu, Y.; Chen, S.; Tang, W.; Li, X.; Zhou, L.; Sun, Y. Functionalized hydrogel sequentially deliver tannic acid and bioactive probiotics for radiation-induced skin injury. Mater. Today Bio 2025, 36, 102753. [Google Scholar] [CrossRef]
- Zhu, M.; Shi, G.; Chen, R.; Li, Z.; Wang, J.; Wu, Z.; Guo, L.; Wei, Y.; Li, J. A multifunctional parathyroid hormone-related supramolecular peptide-loaded dual network hydrogel for radiation-induced wound repair. Mater. Today Bio 2025, 32, 101807. [Google Scholar] [CrossRef]
- Liu, X.; Guo, T.; Huang, Z.; Chen, S.; Chen, L.; Li, C.; Tian, T.; Qian, Y.; Yang, L.; Xiang, J.; et al. Acellular dermal matrix hydrogels promote healing of radiation-induced skin injury in a rat model. J. Mater. Chem. B 2024, 12, 11218–11229. [Google Scholar] [CrossRef] [PubMed]
- DeCostanza, L.; Grogan, G.M.; Bruce, A.C.; Peachey, C.M.; Clark, E.A.; Atkins, K.; Tylek, T.; Solga, M.D.; Spiller, K.L.; Peirce, S.M.; et al. Decellularized porcine dermal hydrogel enhances implant-based wound healing in the setting of irradiation. Acta Biomater. 2024, 191, 260–275. [Google Scholar] [CrossRef]
- Chinnapaka, S.; Yang, K.S.; Surucu, Y.; Bengur, F.B.; Arellano, J.A.; Tirmizi, Z.; Malekzadeh, H.; Epperly, M.W.; Hou, W.; Greenberger, J.S.; et al. Human adipose ECM alleviates radiation-induced skin fibrosis via endothelial cell-mediated M2 macrophage polarization. iScience 2023, 26, 107660. [Google Scholar] [CrossRef]
- Chen, X.; Zhai, D.; Wang, B.; Hao, S.; Song, J.; Peng, Z. Hair keratin promotes wound healing in rats with combined radiation-wound injury. J. Mater. Sci. Mater. Med. 2020, 31, 1–9. [Google Scholar] [CrossRef]
- Johnson, M.B.; Pang, B.; Gardner, D.J.; Niknam-Benia, S.; Soundarajan, V.; Bramos, A.; Perrault, D.P.; Banks, K.; Lee, G.K.; Baker, R.Y.; et al. Topical Fibronectin Improves Wound Healing of Irradiated Skin. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.-T.; Gao, C.-H.; Tan, L.-F.; Chen, L.-X.; Fan, J.-X.; Xiong, W.; Cheng, K.; Zhao, Y.-D.; Liu, B. Gene-engineered polypeptide hydrogels with on-demand oxygenation and ECM-cell interaction mimicry for diabetic wound healing. Biomaterials 2024, 316, 122984. [Google Scholar] [CrossRef]
- Zhu, W.; Jia, L.; Chen, G.; Zhao, H.; Sun, X.; Meng, X.; Zhao, X.; Xing, L.; Yu, J.; Zheng, M. Epigallocatechin-3-gallate ameliorates radiation-induced acute skin damage in breast cancer patients undergoing adjuvant radiotherapy. Oncotarget 2016, 7, 48607–48613. [Google Scholar] [CrossRef]
- Nam, S.; Smith, D.M.; Dou, Q.P. Ester Bond-containing Tea Polyphenols Potently Inhibit Proteasome Activity in Vitro and in Vivo. J. Biol. Chem. 2001, 276, 13322–13330. [Google Scholar] [CrossRef]
- Wang, J.; Gao, L.; Song, J.; Li, S. Study of EGCG composite hydrogel for the treatment of radiation-induced skin injuries. J. Appl. Biomater. Funct. Mater. 2023, 21. [Google Scholar] [CrossRef]
- Dong, L.; Jia, R.; Liu, Z.; Aiyiti, W.; Shuai, C.; Li, Z.; Fu, Q.; Li, X. Tannic acid based multifunctional hydrogels with mechanical stability for wound healing. Colloids Surf. B Biointerfaces 2024, 243, 114127. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.; Chen, S.; Wu, C.; Zuo, J.; Wang, W.; Zhang, Y.; Liu, J.; Feng, H.; Chu, W.; Jin, Y. In situ photocrosslinking ROS-adaptive caffeoyl chitosan/boronic acid-grafted gelatin hydrogels for treatment of combined radiation-burn injury. Mater. Sci. Eng. C 2026, 183, 214760. [Google Scholar] [CrossRef]
- Wen, Y.; Wang, Y.; Zhao, C.; Zhao, B.; Wang, J. The Pharmacological Efficacy of Baicalin in Inflammatory Diseases. Int. J. Mol. Sci. 2023, 24, 9317. [Google Scholar] [CrossRef]
- Liu, X.; Shu, W.; Zhong, Q.; Zeng, A.; Zeng, Y.; Gu, H.; Chen, P.; Li, X. A Baicalin Liposome-Based Temperature-Sensitive Hydrogel for Treating Ultraviolet-Induced Skin Damage. Int. J. Nanomed. 2025, ume 20, 7935–7951. [Google Scholar] [CrossRef] [PubMed]
- Dalcin, A.J.F.; Roggia, I.; Felin, S.; Vizzotto, B.S.; Mitjans, M.; Vinardell, M.P.; Schuch, A.P.; Ourique, A.F.; Gomes, P. UVB photoprotective capacity of hydrogels containing dihydromyricetin nanocapsules to UV-induced DNA damage. Colloids Surf. B Biointerfaces 2021, 197, 111431. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Lang, Y.; He, J.; Cui, J.; Liu, X.; Yuan, H.; Li, L.; Zhou, M.; Wang, S. Phycocyanin-based multifunctional microspheres for treatment of infected radiation-induced skin injury. Biomaterials 2024, 317, 123061. [Google Scholar] [CrossRef]
- Andrade, T.d.A.; Heimfarth, L.; dos Santos, D.M.; dos Santos, M.R.V.; de Albuquerque-Júnior, R.L.C.; dos Santos-Neto, A.G.; de Araujo, G.R.S.; Lira, A.A.M.; Matos, S.S.; Frank, L.A.; et al. Hesperetin-Based Hydrogels Protect the Skin against UV Radiation-Induced Damage. Aaps Pharmscitech 2022, 23, 1–14. [Google Scholar] [CrossRef]
- Schultze-Mosgau, S.; A Blaese, M.; Grabenbauer, G.; Wehrhan, F.; Kopp, J.; Amann, K.; Rodemann, H.; Rödel, F. Smad-3 and Smad-7 expression following anti-transforming growth factor beta 1 (TGFβ1)-treatment in irradiated rat tissue. Radiother. Oncol. 2004, 70, 249–259. [Google Scholar] [CrossRef]
- Gallet, P.; Phulpin, B.; Merlin, J.-L.; Leroux, A.; Bravetti, P.; Mecellem, H.; Tran, N.; Dolivet, G. Long-Term Alterations of Cytokines and Growth Factors Expression in Irradiated Tissues and Relation with Histological Severity Scoring. PLoS ONE 2011, 6, e29399. [Google Scholar] [CrossRef] [PubMed]
- Maria, O.M.; Eliopoulos, N.; Muanza, T. Radiation-Induced Oral Mucositis. Front. Oncol. 2017, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Jiang, Z.; Xu, S.; He, L.; Jiang, T.; Yang, Y.; Xie, X.; Lei, L. Designing adhesive hydrogels for oral diseases treatment. Mater. Today Bio 2026, 37, 102911. [Google Scholar] [CrossRef]
- Mndlovu, H.; du Toit, L.C.; Kumar, P.; Choonara, Y.E.; Marimuthu, T.; Kondiah, P.P.D.; Pillay, V. Bioplatform Fabrication Approaches Affecting Chitosan-Based Interpolymer Complex Properties and Performance as Wound Dressings. Molecules 2020, 25, 222. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Han, X.; Xiao, W.; Wang, P.; Wang, J.; Zou, D.; Luo, X.; Shi, L.; Wu, J.; Guo, L.; et al. Mussel-inspired adhesive drug-loaded hydrogels for oral ulcers treatment. Acta Biomater. 2024, 187, 98–109. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, D.; Gao, J.; Wu, X.; Hu, W.; Han, L. Surface-engineered hydrophobic hydrogels via cholesterol micelle rearrangement for robust wet adhesion and oral mucositis therapy. Mater. Today Bio 2025, 34, 102126. [Google Scholar] [CrossRef]
- Wu, Y.; Jiang, L.; Li, K.; Liu, J.; Chen, P.; Xu, J.; Zhang, J. Hyaluronic acid-based composite hydrogels embedded with core-shell microgels with properties of mucosal adhesion and combined drug administration for chemoradiotherapy induced oral mucositis. Int. J. Biol. Macromol. 2025, 322, 146575. [Google Scholar] [CrossRef]
- Shi, M.; Huang, C.; Peng, Y.; Yang, C.; Sun, M. Multifunctional Hydrogel with Dual Functions of ROS Scavenging and Responsive Antibiotic Release for Synergistic Oral Mucositis Treatment. ACS Omega 2026, 11, 11961–11970. [Google Scholar] [CrossRef]
- Yuan, R.; Du, S.; Pan, S.; Lin, Z.; Zhang, N.; Zhang, C.; Zeng, Q.; Wei, Y.; Wu, Y.; Tao, L. Multifunctional hydrogel encapsulated with baicalin for full-layer regeneration of drug-resistant bacteria-infected wounds after radiotherapy. Bioact. Mater. 2025, 53, 20–31. [Google Scholar] [CrossRef]
- Ding, Z.; Hu, X.; Liang, W.; Zheng, S.; Luo, X.; Zhao, H. Correction: Dual-functional guanosine-based hydrogel: high-efficiency protection in radiation-induced oral mucositis. J. Mater. Chem. B 2025, 14, 374–375. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, X.; Mao, R.; Li, H.; Hao, Y.; Zhang, J.; Wang, W.; Zhang, Y.; Liu, J. Multifunctional Glycopeptide-Based Hydrogel via Dual-Modulation for the Prevention and Repair of Radiation-Induced Skin Injury. ACS Biomater. Sci. Eng. 2024, 10, 5168–5180. [Google Scholar] [CrossRef]
- Moussa, L.; Pattappa, G.; Doix, B.; Benselama, S.-L.; Demarquay, C.; Benderitter, M.; Sémont, A.; Tamarat, R.; Guicheux, J.; Weiss, P.; et al. A biomaterial-assisted mesenchymal stromal cell therapy alleviates colonic radiation-induced damage. Biomaterials 2017, 115, 40–52. [Google Scholar] [CrossRef]
- Mathieu, E.; Lamirault, G.; Toquet, C.; Lhommet, P.; Rederstorff, E.; Sourice, S.; Biteau, K.; Hulin, P.; Forest, V.; Weiss, P.; et al. Intramyocardial Delivery of Mesenchymal Stem Cell-Seeded Hydrogel Preserves Cardiac Function and Attenuates Ventricular Remodeling after Myocardial Infarction. PLoS ONE 2012, 7, e51991. [Google Scholar] [CrossRef]
- Kim, I.G.; Cho, H.; Shin, J.; Cho, J.H.; Cho, S.-W.; Chung, E.-J. Regeneration of irradiation-damaged esophagus by local delivery of mesenchymal stem-cell spheroids encapsulated in a hyaluronic-acid-based hydrogel. Biomater. Sci. 2021, 9, 2197–2208. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.H.; Yoo, J.H.; Yoo, I.K.; Kwon, C.I.; Hong, S.P. Effects of Mesenchymal Stem Cells Treatment on Radiation-Induced Proctitis in Rats. Yonsei Med. J. 2023, 64, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Dunn, C.M.; Kameishi, S.; Grainger, D.W.; Okano, T. Strategies to address mesenchymal stem/stromal cell heterogeneity in immunomodulatory profiles to improve cell-based therapies. Acta Biomater. 2021, 133, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Ai, S.; Hong, Q.; Zhang, C.; Song, Y.; Wang, X.; Wang, X.; Cui, S.; Li, Z.; Zhu, H.; et al. A supramolecular hydrogel based on the combination of YIGSR and RGD enhances mesenchymal stem cells paracrine function via integrin α2β1 and PI3K/AKT signaling pathway for acute kidney injury therapy. Chem. Eng. J. 2022, 436. [Google Scholar] [CrossRef]
- Chen, G.; Han, Y.; Zhang, H.; Tu, W.; Zhang, S. Radiotherapy-Induced Digestive Injury: Diagnosis, Treatment and Mechanisms. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Sharma, R.; Lewis, S.; Wlodarski, M.W. DNA Repair Syndromes and Cancer: Insights Into Genetics and Phenotype Patterns. Front. Pediatr. 2020, 8, 570084. [Google Scholar] [CrossRef]
- Moussa, L.; Usunier, B.; Demarquay, C.; Benderitter, M.; Tamarat, R.; Sémont, A.; Mathieu, N. Bowel Radiation Injury: Complexity of the Pathophysiology and Promises of Cell and Tissue Engineering. Cell Transplant. 2016, 25, 1723–1746. [Google Scholar] [CrossRef]
- Usunier, B.; Benderitter, M.; Tamarat, R.; Chapel, A. Management of Fibrosis: The Mesenchymal Stromal Cells Breakthrough. Stem Cells Int. 2014, 2014, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.D.; Coquia, S.; De Jong, R.; Gourin, C.; Page, B.; Latronico, D.; Dah, S.; Su, L.; Clarke, S.; Schultz, J.; et al. Effects of biodegradable hydrogel spacer injection on contralateral submandibular gland sparing in radiotherapy for head and neck cancers. 15th International Wolfsberg Meeting on Molecular Radiation Biology/Oncology; LOCATION OF CONFERENCE, COUNTRYDATE OF CONFERENCE; pp. 96–99.
- Rucinski, A.; Brons, S.; Richter, D.; Habl, G.; Debus, J.; Bert, C.; Haberer, T.; Jäkel, O. Ion therapy of prostate cancer: daily rectal dose reduction by application of spacer gel. Radiat. Oncol. 2015, 10, 56–56. [Google Scholar] [CrossRef] [PubMed]
- van Wijk, Y.; Vanneste, B.G.; Walsh, S.; van der Meer, S.; Ramaekers, B.; van Elmpt, W.; Pinkawa, M.; Lambin, P. Development of a virtual spacer to support the decision for the placement of an implantable rectum spacer for prostate cancer radiotherapy: Comparison of dose, toxicity and cost-effectiveness. Radiother. Oncol. 2017, 125, 107–112. [Google Scholar] [CrossRef]
- Rao, A.D.; Feng, Z.; Shin, E.J.; He, J.; Waters, K.M.; Coquia, S.; DeJong, R.; Rosati, L.M.; Su, L.; Li, D.; et al. A Novel Absorbable Radiopaque Hydrogel Spacer to Separate the Head of the Pancreas and Duodenum in Radiation Therapy for Pancreatic Cancer. Int. J. Radiat. Oncol. 2017, 99, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Pinkawa, M.; Berneking, V.; Schlenter, M.; Krenkel, B.; Eble, M.J. Quality of Life After Radiation Therapy for Prostate Cancer With a Hydrogel Spacer: 5-Year Results. Int. J. Radiat. Oncol. 2017, 99, 374–377. [Google Scholar] [CrossRef]
- Chao, M.; Ho, H.; Chan, Y.; Tan, A.; Pham, T.; Bolton, D.; Troy, A.; Temelcos, C.; Sengupta, S.; McMillan, K.; et al. Prospective analysis of hydrogel spacer for patients with prostate cancer undergoing radiotherapy. BJU Int. 2018, 122, 427–433. [Google Scholar] [CrossRef]
- Armstrong, N.; Bahl, A.; Pinkawa, M.; Ryder, S.; Ahmadu, C.; Ross, J.; Bhattacharyya, S.; Woodward, E.; Battaglia, S.; Binns, J.; et al. SpaceOAR Hydrogel Spacer for Reducing Radiation Toxicity During Radiotherapy for Prostate Cancer. A Systematic Review. Urology 2021, 156, e74–e85. [Google Scholar] [CrossRef]
- van Gysen, K.; Kneebone, A.; Alfieri, F.; Guo, L.; Eade, T. Feasibility of and rectal dosimetry improvement with the use of SpaceOAR® hydrogel for dose-escalated prostate cancer radiotherapy. J. Med. Imaging Radiat. Oncol. 2014, 58, 511–516. [Google Scholar] [CrossRef]
- Uhl, M.; Herfarth, K.; Eble, M.J.; Pinkawa, M.; van Triest, B.; Kalisvaart, R.; Weber, D.C.; Miralbell, R.; Song, D.Y.; DeWeese, T.L. Absorbable hydrogel spacer use in men undergoing prostate cancer radiotherapy: 12 month toxicity and proctoscopy results of a prospective multicenter phase II trial. Radiat. Oncol. 2014, 9, 96–96. [Google Scholar] [CrossRef]
- Ardekani, M.A.; Ghaffari, H. Optimization of prostate brachytherapy techniques with polyethylene glycol–based hydrogel spacers: A systematic review. Brachytherapy 2020, 19, 13–23. [Google Scholar] [CrossRef]
- Kundu, P.; Lin, E.Y.; Yoon, S.M.; Parikh, N.R.; Ruan, D.; Kishan, A.U.; Lee, A.; Steinberg, M.L.; Chang, A.J. Rectal Radiation Dose and Clinical Outcomes in Prostate Cancer Patients Treated With Stereotactic Body Radiation Therapy With and Without Hydrogel. Front. Oncol. 2022, 12, 853246. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, M.H.; Siavashpour, Z.; Sina, S. Tissue spacers in brachytherapy: A systematic review and meta-analysis. Crit. Rev. Oncol. 2025, 213, 104790. [Google Scholar] [CrossRef]
- Folkert, M.R.; Zelefsky, M.J.; Hannan, R.; Desai, N.B.; Lotan, Y.; Laine, A.M.; Kim, D.W.N.; Neufeld, S.H.; Hornberger, B.; Kollmeier, M.A.; et al. A Multi-Institutional Phase 2 Trial of High-Dose SAbR for Prostate Cancer Using Rectal Spacer. Int. J. Radiat. Oncol. 2021, 111, 101–109. [Google Scholar] [CrossRef]
- Mahal, B.A.; O'LEary, M.P.; Nguyen, P.L. Hydrogel Spacing for Radiotherapy of Prostate Cancer: A Review of the Literature. Urol. Pr. 2014, 1, 79–85. [Google Scholar] [CrossRef]
- Hatiboglu, G.; Pinkawa, M.; Vallée, J.; Hadaschik, B.; Hohenfellner, M. Application technique: placement of a prostate–rectum spacer in men undergoing prostate radiation therapy. BJU Int. 2012, 110, E647–E652. [Google Scholar] [CrossRef] [PubMed]
- Pinkawa, M.; Piroth, M.D.; Holy, R.; Escobar-Corral, N.; Caffaro, M.; Djukic, V.; Klotz, J.; Eble, M.J. Spacer stability and prostate position variability during radiotherapy for prostate cancer applying a hydrogel to protect the rectal wall. Radiother. Oncol. 2013, 106, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Ding, K.; Rao, A.; He, J.; Bhutani, M.S.; Herman, J.M.; Narang, A.; Shin, E.J. EUS-guided hydrogel microparticle injection in a cadaveric model. J. Appl. Clin. Med. Phys. 2021, 22, 83–91. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Shen, S.-H.; Wang, Y.-P.; Chang, N.-W.; Lee, P.-C.; Li, C.-P.; Lan, K.-L.; Shiau, C.-Y.; Hu, Y.-W.; Huang, P.-I.; et al. Feasibility estimation of injected hydrodissection before definitive radiotherapy of pancreatic adenocarcinoma. J. Chin. Med. Assoc. 2024, 87, 511–515. [Google Scholar] [CrossRef]
- Cirillo, G.; Spizzirri, U.G.; Curcio, M.; Nicoletta, F.P.; Iemma, F. Injectable Hydrogels for Cancer Therapy over the Last Decade. Pharmaceutics 2019, 11, 486. [Google Scholar] [CrossRef]
- Pang, L.; Tian, P.; Cui, X.; Wu, X.; Zhao, X.; Wang, H.; Wang, D.; Pan, H. In Situ Photo-Cross-Linking Hydrogel Accelerates Diabetic Wound Healing through Restored Hypoxia-Inducible Factor 1-Alpha Pathway and Regulated Inflammation. ACS Appl. Mater. Interfaces 2021, 13, 29363–29379. [Google Scholar] [CrossRef]
- Pandey, M.; Choudhury, H.; Singh, S.K.D.S.; Annan, N.C.; Bhattamisra, S.K.; Gorain, B.; Amin, M.C.I.M. Budesonide-Loaded Pectin/Polyacrylamide Hydrogel for Sustained Delivery: Fabrication, Characterization and In Vitro Release Kinetics. Molecules 2021, 26, 2704. [Google Scholar] [CrossRef]
- Qiao, Y.; Zhang, Q.; Wang, Q.; Li, Y.; Wang, L. Filament-anchored hydrogel layer on polypropylene hernia mesh with robust anti-inflammatory effects. Acta Biomater. 2021, 128, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Machado, V.S.; Camponogara, C.; Oliveira, S.M.; Baldissera, M.D.; Sagrillo, M.R.; Gundel, S.D.S.; Ourique, A.F.; Klein, B.; Wagner, R.; Santos, R.C.; et al. Topical hydrogel containing Achyrocline satureioides oily extract (free and nanocapsule) has anti-inflammatory effects and thereby minimizes irritant contact dermatitis. An. Da Acad. Bras. De Cienc. 2020, 92, e20191066. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-S.; Xu, B.-X.; Fan, K.-J.; Li, Y.-W.; Wu, J.; Wang, T.-Y. Dexamethasone-Loaded Thermosensitive Hydrogel Suppresses Inflammation and Pain in Collagen-Induced Arthritis Rats. Drug Des. Dev. Ther. 2020, ume 14, 4101–4113. [Google Scholar] [CrossRef] [PubMed]
- Kerdsirichairat, T.; Narang, A.K.; Thompson, E.; Kim, S.-H.; Rao, A.; Ding, K.; Shin, E.J. Feasibility of Using Hydrogel Spacers for Borderline-Resectable and Locally Advanced Pancreatic Tumors. Gastroenterology 2019, 157, 933–935. [Google Scholar] [CrossRef]
- Sémont, A.; François, S.; Mouiseddine, M.; François, A.; Saché, A.; Frick, J.; Thierry, D.; Chapel.
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in Regenerative Medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef]
- Moussa, L.; Demarquay, C.; Réthoré, G.; Benadjaoud, M.A.; Siñeriz, F.; Pattapa, G.; Guicheux, J.; Weiss, P.; Barritault, D.; Mathieu, N. Heparan Sulfate Mimetics: A New Way to Optimize Therapeutic Effects of Hydrogel-Embedded Mesenchymal Stromal Cells in Colonic Radiation-Induced Damage. Sci. Rep. 2019, 9, 164. [Google Scholar] [CrossRef]
- Jensen, M.M.; Jia, W.; Isaacson, K.J.; Schults, A.; Cappello, J.; Prestwich, G.D.; Oottamasathien, S.; Ghandehari, H. Silk-elastinlike protein polymers enhance the efficacy of a therapeutic glycosaminoglycan for prophylactic treatment of radiation-induced proctitis. J. Control. Release 2017, 263, 46–56. [Google Scholar] [CrossRef]
- Rimpy; Abhishek; Ahuja, M. Evaluation of carboxymethyl moringa gum as nanometric carrier. Carbohydr. Polym. 2017, 174, 896–903. [Google Scholar] [CrossRef]
- Abhishek; Rimpy; Ahuja, M. Moringa gum-g-poly(N-vinyl-2-pyrrolidone) – a potential buccoadhesive polymer. Int. J. Biol. Macromol. 2018, 109, 732–739. [Google Scholar] [CrossRef]
- Singh, B.; Kumar, A. Radiation-induced graft copolymerization of N-vinyl imidazole onto moringa gum polysaccharide for making hydrogels for biomedical applications. Int. J. Biol. Macromol. 2018, 120, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Acuña, R.; Quirós, M.; Farkas, E.A.; Dedhia, P.H.; Huang, S.; Siuda, D.; García-Hernández, V.; Miller, A.J.; Spence, J.R.; Nusrat, A.; et al. Synthetic hydrogels for human intestinal organoid generation and colonic wound repair. Nat. Cell Biol. 2017, 19, 1326–1335. [Google Scholar] [CrossRef]
- Gu, X.; Yu, L.; Wang, X.; Yin, S.; Zheng, Y.; Zheng, Z.; Zhang, Y.; Chen, K.; Zhang, Y.; Ding, Y. CS@LGG as a therapeutic biomaterial for acute radiation-induced bowel injury alleviation. Adv. Radiother. Nucl. Med. 2025, 025230026. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71(3), 209–249. [Google Scholar] [CrossRef]
- Chang, S.; Lv, J.; Wang, X.; Su, J.; Bian, C.; Zheng, Z.; Yu, H.; Bao, J.; Xin, Y.; Jiang, X. Pathogenic mechanisms and latest therapeutic approaches for radiation-induced lung injury: A narrative review. Crit. Rev. Oncol. 2024, 202, 104461. [Google Scholar] [CrossRef] [PubMed]
- Hanania, A.N.; Mainwaring, W.; Ghebre, Y.T.; Hanania, N.A.; Ludwig, M. Radiation-Induced Lung Injury. Chest 2019, 156, 150–162. [Google Scholar] [CrossRef]
- Giuranno, L.; Ient, J.; De Ruysscher, D.; Vooijs, M.A. Radiation-Induced Lung Injury (RILI). Front. Oncol. 2019, 9, 877. [Google Scholar] [CrossRef]
- Maier, P.; Hartmann, L.; Wenz, F.; Herskind, C. Cellular Pathways in Response to Ionizing Radiation and Their Targetability for Tumor Radiosensitization. Int. J. Mol. Sci. 2016, 17, 102. [Google Scholar] [CrossRef]
- Yan, Y.; Fu, J.; Kowalchuk, R.O.; Wright, C.M.; Zhang, R.; Li, X.; Xu, Y. Exploration of radiation-induced lung injury, from mechanism to treatment: a narrative review. Transl. Lung Cancer Res. 2022, 11, 307–322. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Z.; Huang, M.; Jin, Y.; Huang, Y.; Ji, P.; Ma, Z. Pathological Mechanisms of Radiation-Induced Lung Injury and Novel Nano-Drug Delivery Therapeutic Strategies. Int. J. Nanomed. 2025, ume 20, 12431–12465. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, S.; Yang, R.; Wang, Z.; Zhang, W.; Liu, H.; Huang, Y. ROS Promote Hypoxia-Induced Keratinocyte Epithelial-Mesenchymal Transition by Inducing SOX2 Expression and Subsequent Activation of Wnt/β-Catenin. Oxidative Med. Cell. Longev. 2022, 2022, 1084006. [Google Scholar] [CrossRef]
- Robert, S.; Gicquel, T.; Victoni, T.; Valenca, S.S.; Barreto, E.; Bailly-Maître, B.; Boichot, E.; Lagente, V. Involvement of matrix metalloproteinases (MMPs) and inflammasome pathway in molecular mechanisms of fibrosis. Biosci. Rep. 2016, 36. [Google Scholar] [CrossRef] [PubMed]
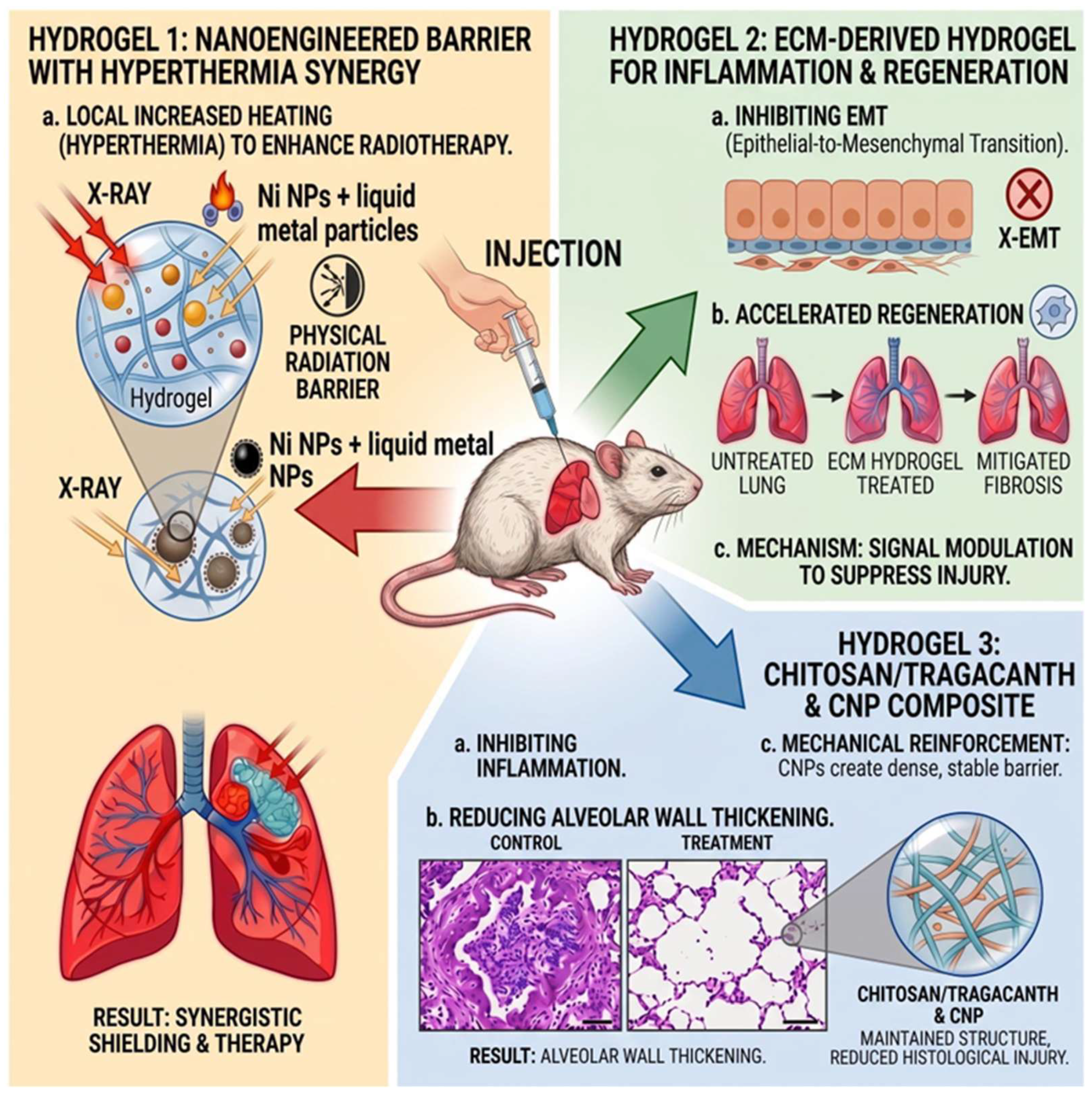
- Zhao, R.; Liu, C.; Luo, H.; Zhao, J.; Zhang, J.; He, Y.; Li, Z.; Yang, P.; Xu, L.; Wan, Y. Nanoengineered Injectable Hydrogel: An Advanced Radioprotective Barrier with Magnetic Hyperthermia Synergy. ACS Appl. Mater. Interfaces 2024, 16, 50188–50201. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wu, P.; Sun, H.; Zhou, H.; Zhang, Y.; Xiao, Z. Lung tissue extracellular matrix-derived hydrogels protect against radiation-induced lung injury by suppressing epithelial–mesenchymal transition. J. Cell. Physiol. 2019, 235, 2377–2388. [Google Scholar] [CrossRef]
- Lasboo, S.A.; Eslami, H.; Razavi-Tousi, S.M.T.; Ansari, M.; Bazghaleh, A.A. The affinity of cellulose nanoparticle toward hydrogel based on chitosan/tragacanth for radiation protection: Study of pulmonary damages on rats. J. Polym. Sci. 2024, 62, 4456–4471. [Google Scholar] [CrossRef]
- Nikjoo, D.; van der Zwaan, I.; Brülls, M.; Tehler, U.; Frenning, G. Hyaluronic Acid Hydrogels for Controlled Pulmonary Drug Delivery—A Particle Engineering Approach. Pharmaceutics 2021, 13, 1878. [Google Scholar] [CrossRef]
- Tan, B.; He, Y. Amniotic extracelluar matrix microgels for attenuation of radiation-induced lung injury. Med. Hypotheses 2024, 186. [Google Scholar] [CrossRef]
- Wang, H.; Wei, J.; Zheng, Q.; Meng, L.; Xin, Y.; Yin, X.; Jiang, X. Radiation-induced heart disease: a review of classification, mechanism and prevention. Int. J. Biol. Sci. 2019, 15, 2128–2138. [Google Scholar] [CrossRef]
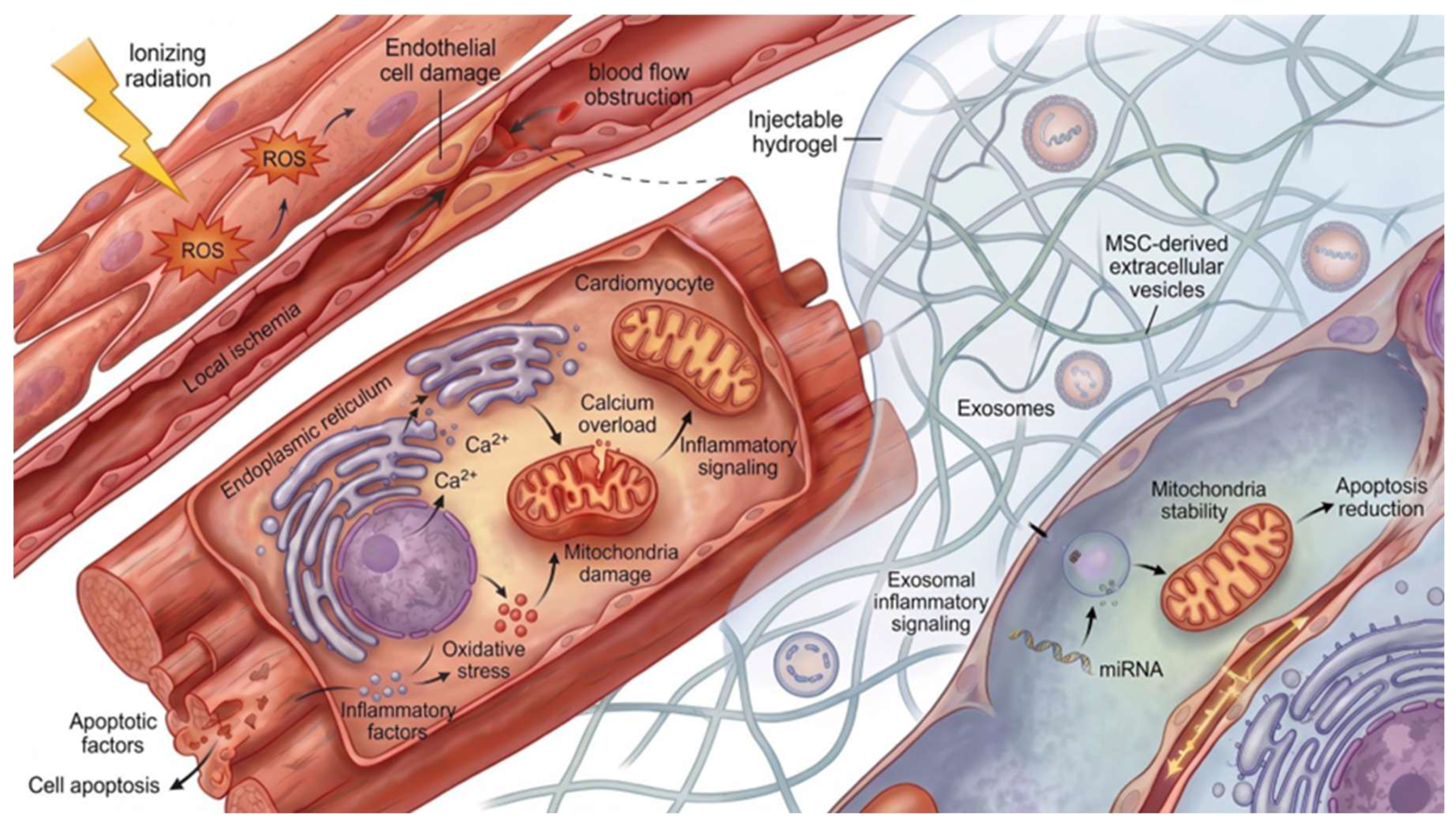
- Wang, L.; Zhao, L.-Y.; Wen, D.-W.; Guo, Y.-Y.; Wang, Z.-Y.; Li, S.-Q.; Wen, M.-L.; Liu, Y. Development of an injectable conductive silk sericin hydrogel loaded with exosomes for potential treatment of radiation-induced heart disease. Mater. Chem. Phys. 2025, 350. [Google Scholar] [CrossRef]
- Abouegylah, M.; Braunstein, L.Z.; El-Din, M.A.A.; Niemierko, A.; Salama, L.; Elebrashi, M.; Edgington, S.K.; Remillard, K.; Napolitano, B.; Naoum, G.E.; et al. Evaluation of radiation-induced cardiac toxicity in breast cancer patients treated with Trastuzumab-based chemotherapy. Breast Cancer Res. Treat. 2018, 174, 179–185. [Google Scholar] [CrossRef]
- Zhao, C.; Xu, S.; Yang, Y.; Shen, X.; Wang, J.; Xing, S.; Yu, Z. Intersection of Cardio-Oncology: An Overview of Radiation-Induced Heart Disease in the Context of Tumors. J. Am. Hear. Assoc. 2025, 14, e040937. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, R.; Takase, K.; Jingu, K.; Takanami, K.; Ota, H.; Kaneta, T.; Takeda, K.; Matsushita, H.; Ariga, H.; Takahashi, S.; et al. Evaluation of radiation-induced myocardial damage using iodine-123 -methyl-iodophenyl pentadecanoic acid scintigraphy. J. Radiat. Res. 2013, 54, 880–889. [Google Scholar] [CrossRef]
- Tarsitano, M.; Ming, C.L.C.; Idais, D.; Mahmodi, H.; Wyllie, K.; Isella, B.; Cox, T.R.; Kabakova, I.; Paolino, D.; Gentile, C. Sericin improves alginate-gelatin hydrogels’ mechanical properties, porosity, durability and viability of fibroblast in cardiac spheroids. Int. J. Bioprinting 2024, 5678. [Google Scholar] [CrossRef]
- Pu, X.; Ma, S.; Gao, Y.; Xu, T.; Chang, P.; Dong, L. Mesenchymal Stem Cell-Derived Exosomes: Biological Function and Their Therapeutic Potential in Radiation Damage. Cells 2020, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Bian, Y.; Liu, L.; Liu, L.; Liu, X.; Ma, S. Molecular pathways associated with oxidative stress and their potential applications in radiotherapy (Review). Int. J. Mol. Med. 2022, 49, 1–11. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Z.; Xu, S.; Jiang, J.; Xiao, J. Review of the Pathogenesis, Diagnosis, and Management of Osteoradionecrosis of the Femoral Head. Med. Sci. Monit. 2023, 29, e940264–e940264-13. [Google Scholar] [CrossRef]
- Zhai, J.; He, F.; Wang, J.; Chen, J.; Tong, L.; Zhu, G. Influence of radiation exposure pattern on the bone injury and osteoclastogenesis in a rat model. Int. J. Mol. Med. 2019, 44, 2265–2275. [Google Scholar] [CrossRef]
- Delanian, S.; Lefaix, J.-L. The radiation-induced fibroatrophic process: therapeutic perspective via the antioxidant pathway. Radiother. Oncol. 2004, 73, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Robbins, M.E. Inflammation and Chronic Oxidative Stress in Radiation-Induced Late Normal Tissue Injury: Therapeutic Implications. Curr. Med. Chem. 2009, 16, 130–143. [Google Scholar] [CrossRef]
- Wang, Y.; Turkstani, H.; Alfaifi, A.; Akintoye, S.O. Diagnostic and Therapeutic Approaches to Jaw Osteoradionecrosis. Diagnostics 2024, 14, 2676. [Google Scholar] [CrossRef] [PubMed]
- Topkan, E.; Kucuk, A.; Somay, E.; Yilmaz, B.; Pehlivan, B.; Selek, U. Review of Osteoradionecrosis of the Jaw: Radiotherapy Modality, Technique, and Dose as Risk Factors. J. Clin. Med. 2023, 12, 3025. [Google Scholar] [CrossRef]
- Chronopoulos, A.; Zarra, T.; Tröltzsch, M.; Mahaini, S.; Ehrenfeld, M.; Otto, S. Osteoradionecrosis of the mandible: A ten year single-center retrospective study. J. Cranio-Maxillofac. Surg. 2015, 43, 837–846. [Google Scholar] [CrossRef] [PubMed]
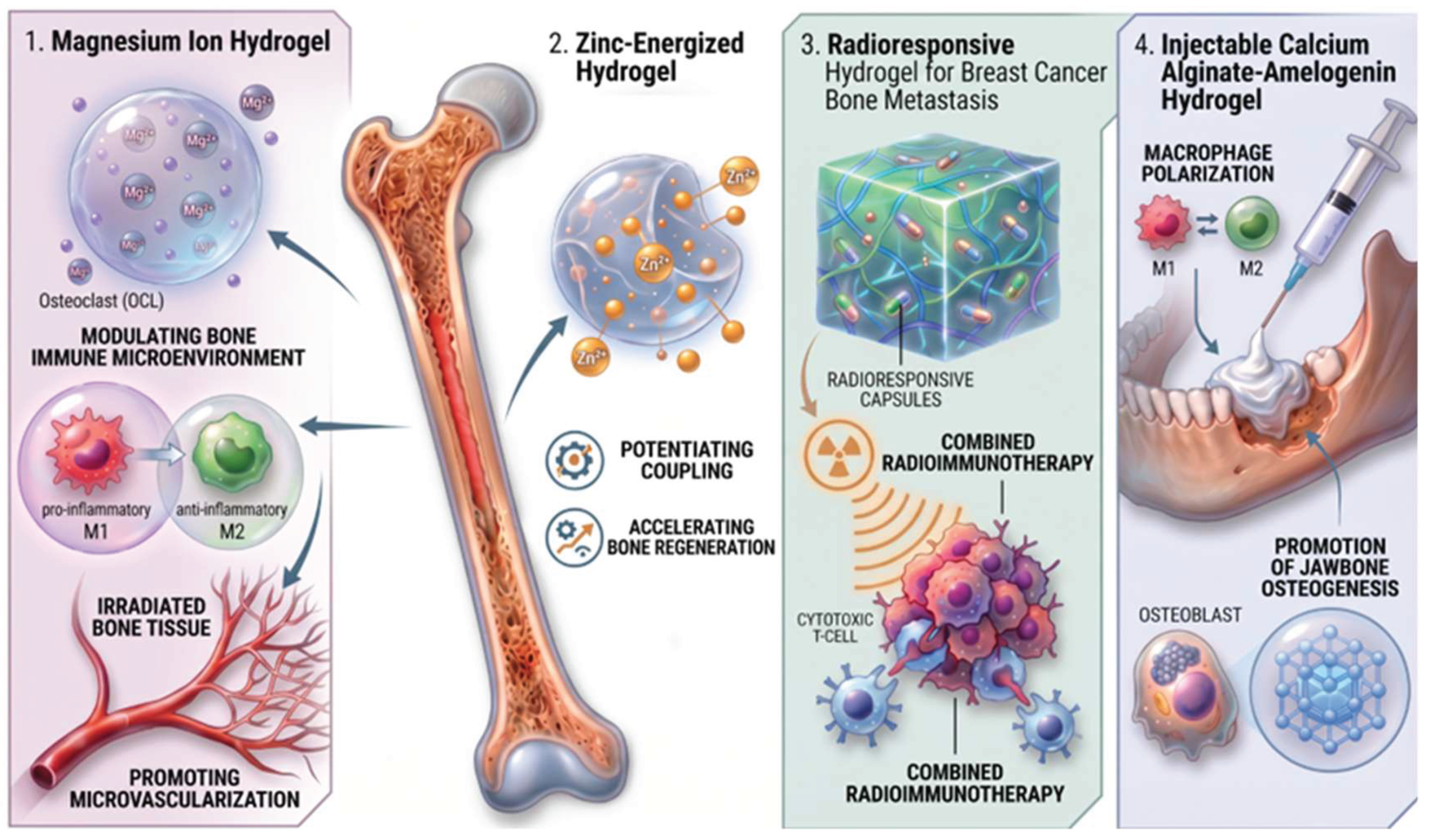
- Wang, Q.; Hu, X.; Xiao, Z.; Ye, K.; Li, J.; Tan, J.; Rao, N.; Zhang, D.; Sun, G.; Cai, M.; et al. Magnesium ion hydrogel enhances resistance to radiation-induced bone injury by modulating the bone immune microenvironment and promoting microvascularization. Regen. Biomater. 2025, 12. [Google Scholar] [CrossRef]
- Zhao, T.; Chen, L.; Yu, C.; He, G.; Lin, H.; Sang, H.; Chen, Z.; Hong, Y.; Sui, W.; Zhao, J. Effect of injectable calcium alginate–amelogenin hydrogel on macrophage polarization and promotion of jawbone osteogenesis. RSC Adv. 2024, 14, 2016–2026. [Google Scholar] [CrossRef]
- Li, L.; Gu, Z.; Wu, Y.; Lin, S.; Cheng, X.; Zhang, L.W.; Qin, J.; Dong, Q.; Wang, Y.; Wang, Y. Hyaluronic acid and polyvinyl alcohol-based radioresponsive hydrogel for combined radioimmunotherapy of breast cancer bone metastasis. Carbohydr. Polym. 2025, 368, 124108. [Google Scholar] [CrossRef] [PubMed]
- Lv, N.; Zhou, Z.; Hong, L.; Li, H.; Liu, M.; Qian, Z. Zinc-energized dynamic hydrogel accelerates bone regeneration via potentiating the coupling of angiogenesis and osteogenesis. Front. Bioeng. Biotechnol. 2024, 12, 1389397. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



