Submitted:
28 April 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Preparation of Plant Samples and Hydrodistillation
2.3. GC/MS -GC/FID
2.4. Statistical Analysis
3. Results
3.1. Weight of Dry Flower Heads
3.2. Essential Oil Content
3.3. Dependence of Essential Oil Content on the Weight of Chamomile Inflorescences
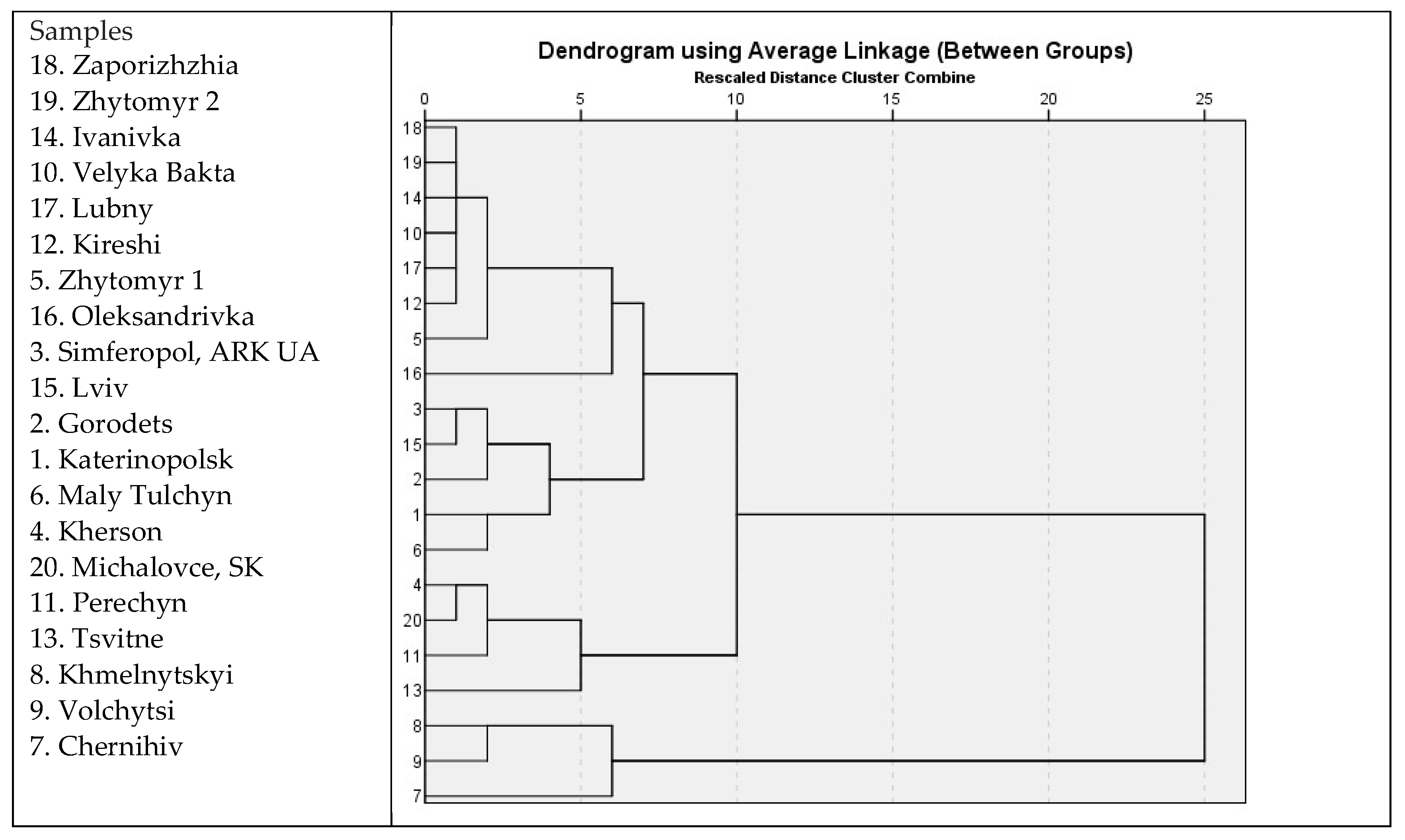
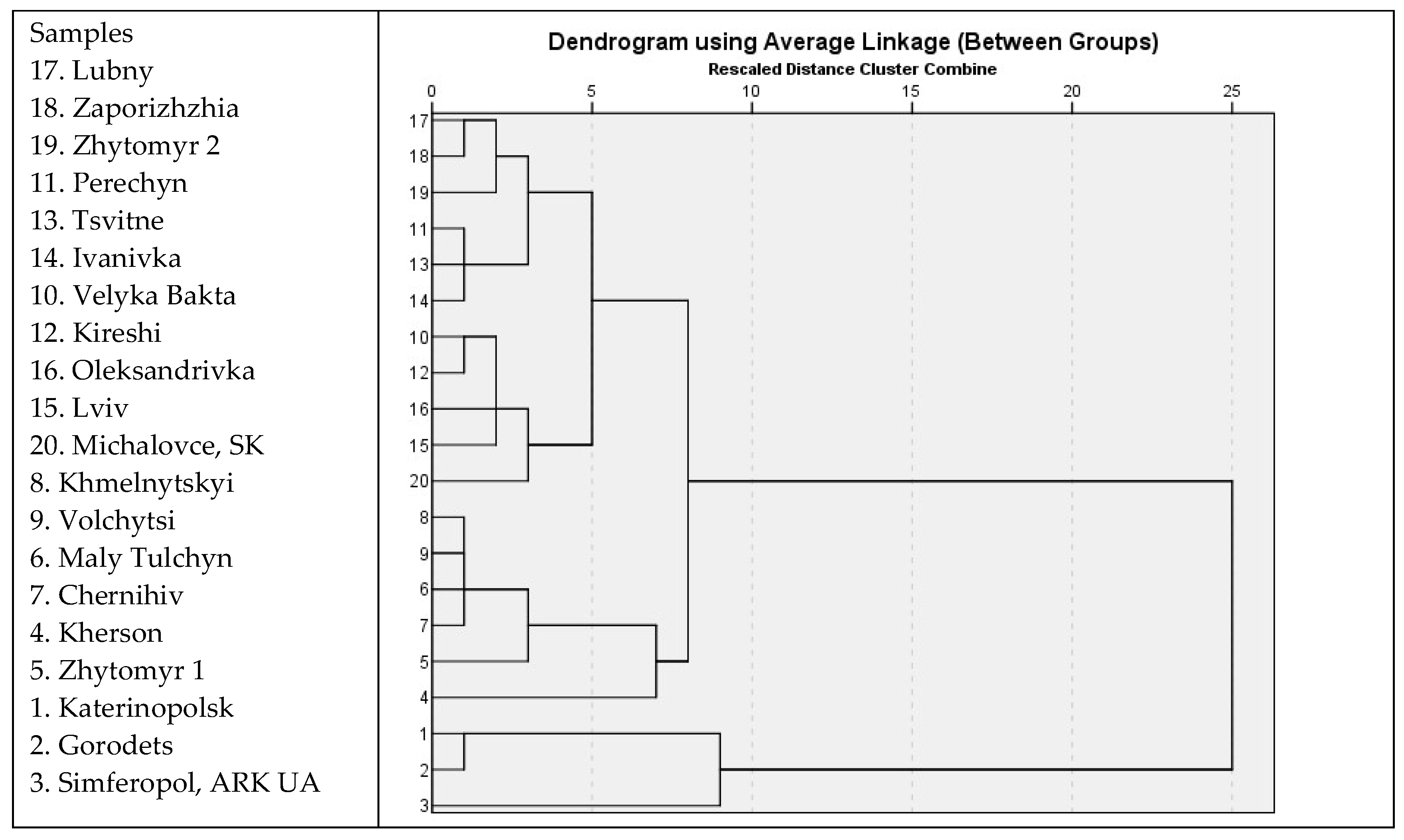
3.4. Chamomile Chemotypes in Ukraine
3.5. Chamomile Large-Scale Cultivation
3.6. Herbal Teas
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McKay, D. L.; Blumberg, J. B. A review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phyther. Res. 2006, 20(7), 519–530. [Google Scholar] [CrossRef]
- El Mihyaoui, A.; Esteves da Silva, J. C. G.; Charfi, S.; Candela Castillo, M. E.; Lamarti, A.; Arnao, M. B. Chamomile (Matricaria chamomilla L.): A review of ethnomedicinal use, phytochemistry and pharmacological uses. Life 2022, 12(4), 479. [Google Scholar] [CrossRef] [PubMed]
- Koshovyi, O.; Sepp, J.; Jakštas, V.; Žvikas, V.; Kireyev, I.; Karpun, Y.; Odyntsova, V.; Heinämäki, J.; Raal, A. German chamomile (Matricaria chamomilla L.) flower extract, its amino acid preparations and 3D-printed dosage forms: Phytochemical, pharmacological, technological, and molecular docking study. Int. J. Mol. Sci. 2024, 25(15), 8292. [Google Scholar] [CrossRef] [PubMed]
- Kozlowska, W.; Wagner, C.; Moore, E. M.; Matkowski, A.; Komarnytsky, S. Botanical provenance of traditional medicines from Carpathian Mountains at the Ukrainian-Polish border. Front. Pharmacol. 2018, 9, 295. [Google Scholar] [CrossRef]
- Sõukand, R.; Kalle, R.; Pieroni, A. Plants used for making recreational tea in Europe: a review based on specific research sites. J. Ethnobiol. Ethnomedicine 2013, 9, 58. [Google Scholar] [CrossRef]
- Srivastava, J. K.; Shankar, E.; Gupta, S. Chamomile: A herbal medicine of the past with a bright future. Mol. Med. Rep. 2010, 3(6), 895–901. [Google Scholar] [CrossRef]
- Lososová, Z.; Chytrý, M.; Cimalová, Š.; Otýpková, Z.; Pyšek, P.; Tichý, L. Classification of weed vegetation of arable land in the Czech Republic and Slovakia. Folia Geobot. 2006, 41(3), 259–273. [Google Scholar] [CrossRef]
- Lososová, Z.; Chytrý, M.; Kühn, I.; Hájek, O.; Horáková, V.; Pyšek, P.; Tichý, L. Plant trait patterns in annual vegetation of Central European human-made habitats. Perspect. Plant Ecol. Evol. Syst. 2006, 8(2), 69–81. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, 2007. [Google Scholar]
- Palaniswamy, U. R.; Palaniswamy, K. M. Handbook of statistics for teaching and research in plant and crop science, 1st ed.; Food Products Press/The Haworth Reference Press, 2006; ISBN 978-1-56022-293-4. [Google Scholar]
- IBM Corp. IBM SPSS Statistics 25 Command Syntax Reference. IBM Corporation, Armonk, NY, USA. 2017. Available online: https://knowledgezone.home.blog/wp-content/uploads/2019/05/wp-1558033893715.pdf.
- European Directorate for the Quality of Medicines; HealthCare (EDQM). European Pharmacopoeia (Ph. Eur.), 10th ed.; Council of Europe: Strasbourg, France, 2022; p. 3856 pp. [Google Scholar]
- State Enterprise Ukrainian Scientific Pharmacopoeia Center for the Quality of Medicinal Products. State Pharmacopoeia of Ukraine, 2nd ed.; Ukrainian Scientific Pharmacopoeia Center for the Quality of Medicinal Products: Kharkiv, Ukraine, 2014; Vol. 3. [Google Scholar]
- Ramazani, E.; Akaberi, M.; Emami, S. A.; Tayarani-Najaran, Z. Pharmacological and biological effects of alpha-bisabolol: An updated review of the molecular mechanisms. Life Sci. 2022, 304, 120728. [Google Scholar] [CrossRef]
- Arunachalam, S.; Nagoor Meeran, M. F.; Azimullah, S.; Kumar Jha, N.; Saraswathiamma, D.; Albawardi, A.; Beiram, R.; Ojha, S. α-Bisabolol Attenuates NF-κB/MAPK Signaling Activation and ER-Stress-Mediated Apoptosis by Invoking Nrf2-Mediated Antioxidant Defense Systems against Doxorubicin-Induced Testicular Toxicity in Rats. Nutrients 2022, 14(21), 4648. [Google Scholar] [CrossRef]
- Herbal drugs and phytopharmaceuticals: a handbook for practice on a scientific basis; Wichtl, M., Ed.; CRC press, 2004; p. 370 p. Available online: https://books.google.com.ua/books?hl=uk&lr=&id=7PRyMWo5e28C&oi=fnd&pg=PR5&ots=iT2VbrzvSw&sig=cP35Dd5IfZhMu1YjtVWO83Ppv0Y&redir_esc=y#v=onepage&q&f=false.
- Ramadan, Mai; Goeters, Susanne; Watzer, Bernhard; Krause, Eva; Lohmann, Klaus; Bauer, Rudolf; Hempel, Bernd; Imming, Peter. Chamazulene Carboxylic Acid and Matricin: A Natural Profen and Its Natural Prodrug, Identified through Similarity to Synthetic Drug Substances. J. Nat. Prod. 2006, 69, 1041–5. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M. F. B.; Rocha, J. E.; Coutinho, H. D. M.; Sharifi-Rad, J. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef]
- Gabbanini, S.; Neba, J. N.; Matera, R.; Valgimigli, L. Photochemical and Oxidative Degradation of Chamazulene Contained in Artemisia, Matricaria and Achillea. Essential Oils and Setup of Protection Strategies. Molecules 2024, 29(11), 2604. [Google Scholar] [CrossRef]
- Isaac, O. Pharmacological investigations with compounds of chamomile: I. On the pharmacology of (-)-α-bisabolol and bisabolol oxides. Planta Medica 1979, 35(02), 118–124. Available online: https://scispace.com/pdf/pharmacological-investigations-with-compounds-of-chamomile-i-2zpmbp1fro.pdf. [CrossRef]
- BenchChem. literature review on the biological activities of bisabolol oxide A. BenchChem. 2026. Available online: https://www.benchchem.com/product/b1251270#literature-review-on-the-biologicalactivities-of-bisabolol-oxide-a.
- Ogata-Ikeda, I.; Seo, H.; Kawanai, T.; Hashimoto, E.; Oyama, Y. Cytotoxic action of bisabololoxide A of German chamomile on human leukemia K562 cells in combination with 5-fluorouracil. Phytomed. Int. J. Phyther. Phytopharm. 2011, 18(5), 362–365. [Google Scholar] [CrossRef]
- Achterrath-Tuckermann, U.; Kunde, R.; Flaskamp, E.; Isaac, O.; Thiemer, K. Thiemer Pharmakologische Untersuchungen von Kamillen-Inhaltsstoffen. Planta Med. 1980, 39(5), 38–50. [Google Scholar] [CrossRef] [PubMed]
- Petrychenko, V. F.; Lykhochvor, V. V. Plantation New technologies for growing field crops: a textbook, 5th ed.; Lviv. Ukrainian Technologies Fund, 2021; p. 808. [Google Scholar] [CrossRef]
- Padalko, T.O.; Ovcharuk, V.I. Features of growth and development of Matricaria chamomilla L. plants depending on the autumn sowing period in the conditions of the Right-Bank Forest-Steppe of Ukraine. Foothill Mt. Agric. Stock. 2023, 75(1), 100–111. [Google Scholar] [CrossRef] [PubMed]
- Padalko, T.O.; Bakhmat, M.I.; Ovcharuk, O.V.; Horodyska, O.P. Quality of raw materials from camomile inflorescences depending on technological factors. Ukr. J. Ecol. 2021, 11(1), 234–240. Available online: https://www.ujecology.com/articles/quality-of-raw-material-from-camomile-inflorescences-depending-on-technological-factors.pdf. [CrossRef]
- Hendawy, S. F.; Khalid, K. A. Effect of chemical and organic fertilizers on yield and essential oil of chamomile flower heads. Med. Aromat. Plant Sci. Biotechnol. 2011, 5(1), 43–48. [Google Scholar]
- Kwiatkowski, C. A.; Harasim, E.; Feledyn-Szewczyk, B.; Stalenga, J.; Jańczak-Pieniążek, M.; Buczek, J.; Nnolim, A. Productivity and Quality of Chamomile (Chamomilla recutita (L.) Rausch.) Grown in an Organic System Depending on Foliar Biopreparations and Row Spacing. Agriculture 2022, 12(10), 1534. [Google Scholar] [CrossRef]
- Chamomile: Industrial profiles; Franke, R., Schilcher, H., Eds.; CRC Press, 2005. [Google Scholar] [CrossRef]
- Eapen, A. S.; Bhosale, Y. K.; Roy, S. A Review on Novel Techniques Used for Drying Medicinal Plants and Its Applications. Int. J. Biomater. 2025, 2025, 4533070. [Google Scholar] [CrossRef]
- Eddin, L. B.; Jha, N. K.; Goyal, S. N.; Agrawal, Y. O.; Subramanya, S. B.; Bastaki, S. M. A.; Ojha, S. Health Benefits, Pharmacological Effects, Molecular Mechanisms, and Therapeutic Potential of α-Bisabolol. Nutrients 2022, 14(7), 1370. [Google Scholar] [CrossRef] [PubMed]
- Şahin, Hasan; TUNÇ, Ramazan; Şekerciler, Fatoş. Chamazulene Content and Hypoglycemic Potential of Matricaria chamomilla L. Samples Collected from Six Different Localities of Diyarbakır/Türkiye. Commagene J. Biol. 2023, 7, 107–112. [Google Scholar] [CrossRef]
- Schilcher, H. Neuere Erkenntnisse bei der Qualitätsbeurteilung von Kamillenblüten bzw. Kamillenöl. 2. Qualitative Beurteilung des ätherischen Oles in flores chamomillae. Aufteilung der Handelskammillen in view bzw. fünf chemische Typen [Recent knowledge in quality evaluation of camomile blossoms respectively camomile oil. 2. Quality evaluation of the volatile oil in Flores Chamomillae. Grading of commercial camomiles into 4 respectively 5 chemical types]. Planta Medica 1973, 23(2), 132–144. [Google Scholar] [CrossRef] [PubMed]
- Circella, G.; De Mastro, G.; D'Andrea, L.; Nano, G.M. Comparison of Chamomile biotypes (Chamomilla recutita (L.) Rauschert). Acta Hortic. 1993, 330, 209–210. [Google Scholar] [CrossRef]
- Esmail, A.; Hassan, A.; Almuntaser, K.; Alashari, N.; Alkadry, N.; Alnahi, R.; Alhagwa, H.; Hamza, R.; Alhubaishy, N.; Al-Farga, A.; Alnuzaili, T. Phytochemical composition and antimicrobial activity of Matricaria chamomilla ethanolic extracts against clinical bacterial isolates in Ibb City, Yemen. Sci. Rep. 2026, 16(1), 7098. [Google Scholar] [CrossRef]
- Salamon, I. Production of Chamomile, Chamomilla recutita (L.) Rauschert, in Slovakia. J. Herbs Spices Med. Plants 1992, 1(1–2), 37–45. [Google Scholar] [CrossRef]
- Hameed, I. H.; Mohammed, G. J.; Kamal, S. A. A Review: Uses and Pharmacological Activity of Matricaria chamomilla. Indian J. Public Health Res. Dev. 2018, 9(3), 200–205. [Google Scholar] [CrossRef]
- Prasher, P.; Sharma, M.; Fatima, R.; Setzer, W. N.; Sharifi-Rad, J. Bisabolol as a natural anticancer agent: molecular insights and therapeutic potential in oncology. Med. Oncol. 2025, 42(11), 485. [Google Scholar] [CrossRef] [PubMed]
- Šalamon, I. Slovak chamomile varieties and their comparison of natural components. Curr. Perspect. Med. Aromat. Plants (CUPMAP) 2019, 2(2), 59–65. [Google Scholar] [CrossRef]
- Šalamon, I. The Slovak gene pool of German chamomile (Matricaria recutita L.) and comparison in its parameters. Hortic. Sci. 2004, 31(2), 70–75. [Google Scholar] [CrossRef]
- Šalamon, I. Chamomile biodiversity of the essential oil qualitative-quantitative characteristics. In Innovations in Chemical Biology; Şener, B., Ed.; Springer: Dordrecht, 2009; pp. 83–90. [Google Scholar] [CrossRef]
- Orav, A.; Raal, A.; Arak, E. Content and composition of the essential oil of Chamomilla recutita (L.) Rauschert from some European countries. Nat. Product. Res. 2010, 24(1), 48–55. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.; Khanam, Z.; Misra, N.; Srivastava, M.K. Chamomile (Matricaria chamomilla L.): An overview. Pharmacogn. Rev. 2011, 5(9), 82–95. [Google Scholar] [CrossRef]
- Shalaby, A. S.; Hendawy, S. F.; Khalil, M. Y. Evaluation of Some Chamomile Cultivars Introduced and Adapted in Egypt. J. Essent. Oil Bear. Plants 2010, 13(6), 655–669. [Google Scholar] [CrossRef]
- Salamon, I.; Ghanavati, M.; Khazaei, H. Chamomile biodiversity and essential oil qualitative-quantitative characteristics in Egyptian production and Iranian landraces. Emir. J. Food Agric. 2010, 22(1), 59–64. [Google Scholar] [CrossRef]
- Farhoudi, R. Chemical Constituents and Antioxidant Properties of Matricaria recutita and Chamaemelum nobile Essential Oil Growing Wild in the South West of Iran. J. Essent. Oil Bear. Plants 2013, 16(4), 531–537. [Google Scholar] [CrossRef]
- Salamon, I.; Ibraliu, A.; Kryvtsova, M. Essential Oil Content and Composition of the Chamomile Inflorescences (Matricaria recutita L.) Belonging to Central Albania. Horticulturae 2023, 9(1), 47. [Google Scholar] [CrossRef]
- Gosztola, B.; Sárosi, S.; Németh, E. Variability of the essential oil content and composition of chamomile (Matricaria recutita L.) affected by weather conditions. Nat. Product. Commun. 2010, 5(3), 465–470. [Google Scholar] [CrossRef]
- Nikolova, A. M.; Jankulov, J. K.; Kozhuharova, K. S. Chemical composition of chamomile populations in Bulgaria. Int. J. Appl. Eng. Res. 2011, 6(16), 1961–1967. Available online: https://www.researchgate.net/publication/287857706_Chemical_composition_of_chamomile_populations_in_Bulgaria.
- Papazoglou, V.; Anastassaki, T.; Demetzos, C.; Loukis, A. Composition of the Essential Oils of Wild Chamomilla recutita (L.) Rausch. Grown in Greece. J. Essent. Oil Res. 1998, 10(6), 635–636. Available online: https://pergamos.lib.uoa.gr/en/item/uoadl:3027364. [CrossRef]
- Tsivelika, N.; Sarrou, E.; Gusheva, K.; Pankou, C.; Koutsos, T.; Chatzopoulou, P.; Mavromatis, A. Phenotypic variation of wild chamomile (Matricaria chamomilla L.) populations and their evaluation for medicinally important essential oil. Biochem. Syst. Ecol. 2018, 80, 21–28. [Google Scholar] [CrossRef]
- Tirillini, B.; Pagiotti, R.; Menghini, L.; Pintore, G. Essential Oil Composition of Ligulate and Tubular Flowers and Receptacle from Wild Chamomilla recutita (L.) Rausch. Grown in Italy. J. Essent. Oil Res. 2006, 18(1), 42–45. [Google Scholar] [CrossRef]
- Barene, I.; Daberte, I.; Zvirgzdina, L.; Iriste, V. The complex technology on products of German chamomile. Medicina 2003, 39 (Suppl 2), 127–131. Available online: https://pubmed.ncbi.nlm.nih.gov/14617873/. [PubMed]
- Orav, A.; Kailas, T.; Ivask, K. Volatile constituents of Matricaria recutita L. from Estonia. Proc. Est. Acad. Sci. Chem. 2001, 50(1), 39–45. [Google Scholar] [CrossRef]
- Kniaziuk, O. V.; Kreshun, R. A. The influence of sowing dates and row spacing on the formation of productivity of medicinal chamomile (Matricaria chamomilla L.) plants. Agrobiology 2015, 2, 107–110. Available online: https://agrobiologiya.btsau.edu.ua/sites/default/files/visnyky/agrobiologiya/agrobiology_2_121_2015.pdf.
- Zhuravel, S.; Trembitska, O.; Klymenko, T.; Kropyvnytskyi, R.; Stoliar, S.; Nigorodova, S.; Diachenko, M.; Kravchuk, M.; Polishchuk, V. Innovative technologies of cultivating medicinal plants; Polissia National University, 2023. [Google Scholar]
- Stevens, N. C. Chamomile. Eighty 2024, 12, 35–44. Available online: https://rami-tea.com/products/eighty-degrees-tea-magazine-issue-12.
- Lupak, O. M. Potentiometric determination of the integral antioxidant activity of inflorescences of Matricaria recutita L. plants under different cultivation condition. Sci. Biol. Sci. 2018, 2(11), 16–19. [Google Scholar] [CrossRef]
- Salamon, I.; Sudimakova, I. Qualitaty of chamomile teas – essential oil content and its composition. Acta Hortic. 2007, 749, 181–186. [Google Scholar] [CrossRef]
- Salamon, I.; Kryvtsova, M.; Stricik, M.; Otepka, P. Significance of medicinal plants in Medzibodrozie Region, East-southern Slovakia, for the socio-economic stability of rural areas. In Medicinal Plants, Sustainable Development and Biodiversity; Ekiert, H. M., et al., Eds.; Springer Nature Switzerland AG, 2021; Volume 28, pp. 849–868. [Google Scholar] [CrossRef]
- Dai, Y. L.; Li, Y.; Wang, Q.; Niu, F. J.; Li, K. W.; Wang, Y. Y.; Wang, J.; Zhou, C. Z.; Gao, L. N. Chamomile: A Review of Its Traditional Uses, Chemical Constituents, Pharmacological Activities and Quality Control Studies. Molecules 2022, 28(1), 133. [Google Scholar] [CrossRef] [PubMed]
- El-Waki; Elgharieb, A.; Abdrabouh, A. Chamomile tea as a supplement therapy relieves thinner induced-lung injury in rats through inhibition inflammation, oxidative stress and P53-dependant apoptotic pathways. Beni-Suef Univ. J. Basic Appl. Sci. 2025, 14, 127–132. [Google Scholar] [CrossRef]
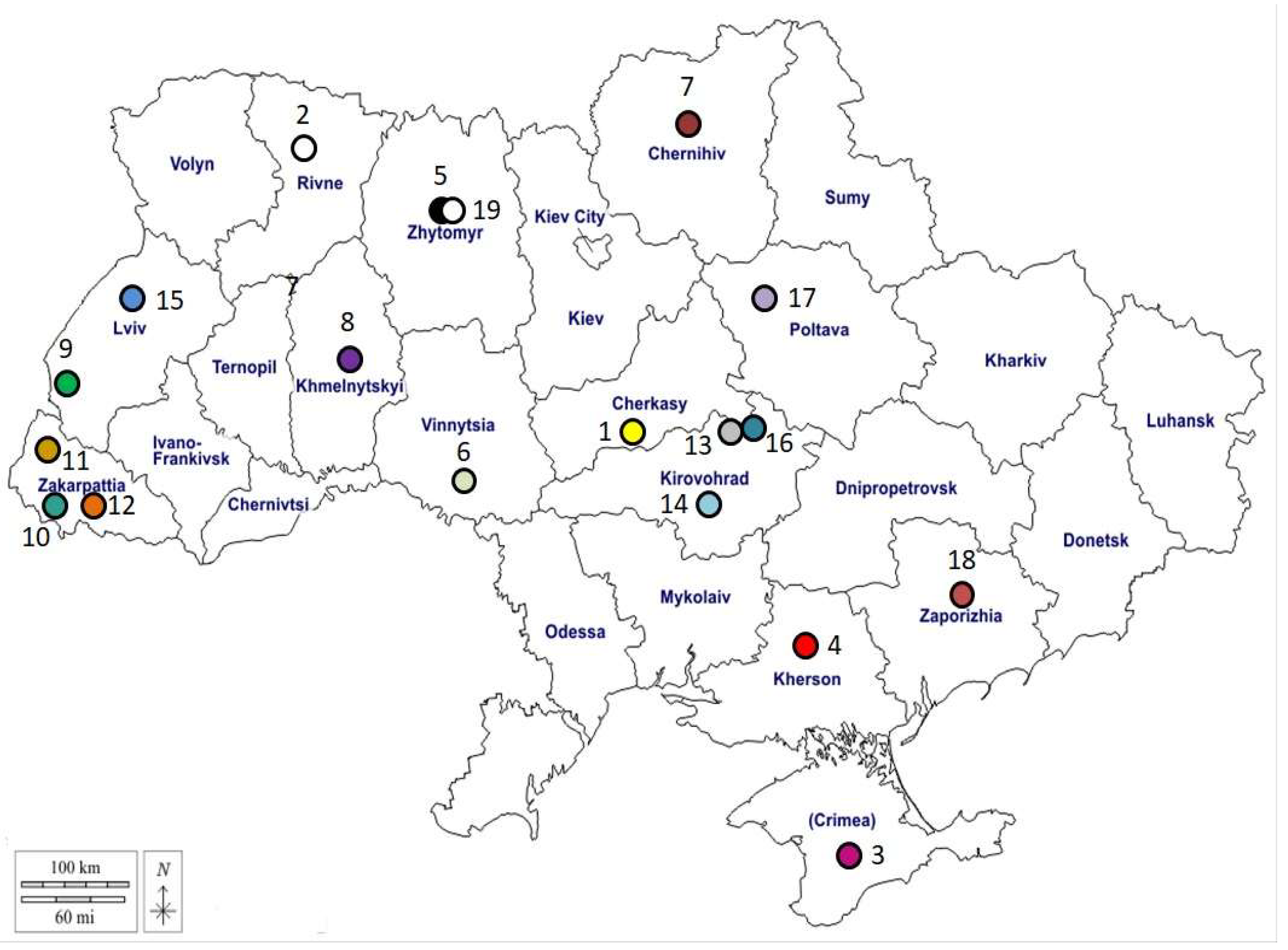


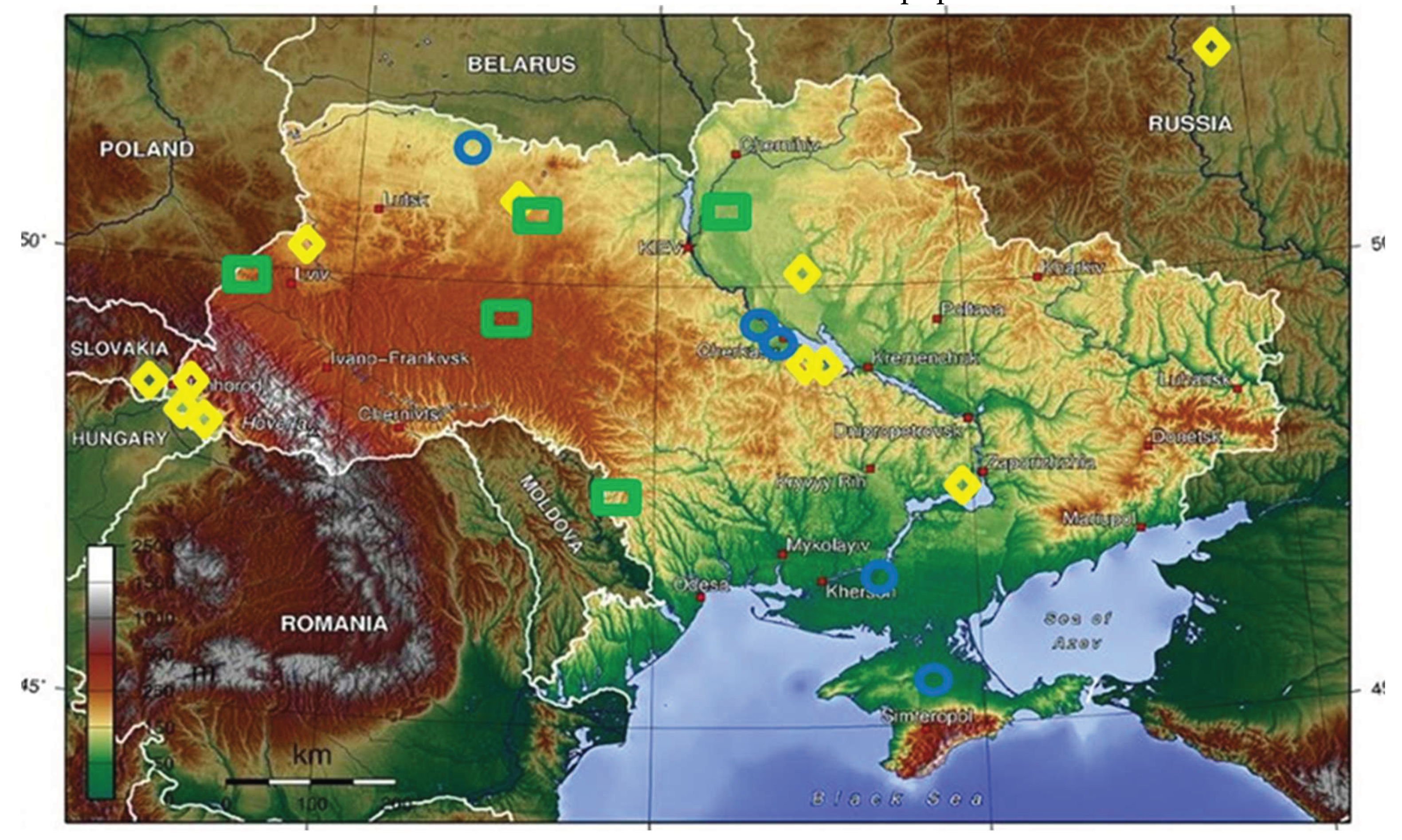
| Location | Latitude | Longitude | Altitude [m] | Exposure | ||
| 01 | Katerinopolsk, CHERKASY, ChR UA | N 48o56ʹ06" | E 0o54ʹ40" | 139 | center | 15° |
| 02 | Gorodets, RIVNE, RR UA | N 51°16'50" | E 26o20ʹ00" | 160 | north-west | 10° |
| 03 | Simferopol, CRIMEA, ARK UA | N 44°56'53" | E 34°06'15" | 350 | south | 5° |
| 04 | Kherson, KHERSON, CR, UA | N 46o38ʹ01" | E 32o36ʹ01" | 47 | south | 5° |
| 05 | Zhytomyr 1, ZHYTOMYR, ŽR, UA | N 50°15′16" | E 28°39′28" | 221 | north-west | 2° |
| 06 | Maly Tulchyn, VINNYTSIA, VR, UA | N 48o40ʹ28" | E 28o50ʹ59" | 208 | north-west | 12° |
| 07 | Chernihiv, CHERNIHIV, CheR UA | N 51o30ʹ00" | E 31o18ʹ00" | 124 | north | 5° |
| 08 | Khmelnytskyi, KHMELNYTSKYI, KhR, UA | N 49o25ʹ12" | E 27o00ʹ00" | 295 | west | 0° |
| 09 | Volchytsi, LVIV, LR, UA | N 49o12ʹ40" | E 22o54ʹ34" | 564 | west | 16° |
| 10 | Velyka Bakta, ZAKARPATTIA, ZR, UA | N 48°09'39" | E 22°39'50" | 118 | west | 10° |
| 11 | Perechyn, ZAKARPATTIA, ZR, UA | N 29°27'13" | E 30°34'51" | 176 | north | 0° |
| 12 | Kireshi, ZAKARPATTIA, ZR, UA | N 48°11'20" | E 23°21'09" | 164 | west | 16° |
| 13 | Tsvitne, KIROVOHRAD, KhR, UA | N 48o57ʹ32" | E 32 o30ʹ00" | 221 | center | 4° |
| 14 | Ivanivka, KIROVOHRAD, KhR, UA | N 48°11'36" | E 32°52'07" | 104 | center | 1° |
| 15 | Lviv, LVIV, LR, UA | N 49°50'00" | E 24°00'00" | 289 | west | 11° |
| 16 | Oleksandrivka, KIROVOHRAD, KR, UA | N 48°43'35" | E 33°18'08" | 133 | center | 7° |
| 17 | Lubny, POLTAVA, PR, UA | N 50°01'07" | E 32°59'13" | 158 | north | 3° |
| 18 | Zaporizhzhia, ZAPORIZHZHIA, ZR, UA | N 47°50'16" | E 35°08'18" | 105 | south-east | 13° |
| 19 | Zhytomyr 2, ZHYTOMYR, ZR UA | N 50°15'16" | E 28°39'28" | 221 | north-west | 2° |
| 20 | Michalovce, KOSICE, KSK, SK | N 39o10ʹ49" | E 22o45ʹ38" | 115 | south-east | 24° |
| № | Location | Region |
[g] |
σ |
[g] |
Sample weight [g] | |
| 1 | Katerinopolsk | ČO, UA | 2,567 | 0,043 | 0,017 | 2,57± 0,12 | 42,00 |
| 2 | Gorodec | RO, UA | 1,958 | 0,014 | 0,006 | 1,96 ± 0,04 | 45,91 |
| 3 | Simferopol | ARK, UA | 2,384 | 0,300 | 0,124 | 2,39± 0,80 | 38,50 |
| 4 | Kherson | CO, UA | 2,989 | 0,022 | 0,008 | 2,99± 0,06 | 38,05 |
| 5 | Zhytomyr | ŽO, UA | 1,701 | 0,078 | 0,032 | 1,70± 0,20 | 48,70 |
| 6 | Maly Tulchyn | VO, UA | 2,890 | 0,035 | 0,014 | 2,89± 0,10 | 32,20 |
| 7 | Chernihiv | ČeO, UA | 3,484 | 0,056 | 0,023 | 3,48± 0,17 | 30,10 |
| 8 | Khmelnitsk | KO, UA | 2,870 | 0,048 | 0,019 | 2,87± 0,13 | 34,52 |
| 9 | Volchytsi | LO, UA | 3,310 | 0,086 | 0,035 | 3,31± 0,24 | 54,23 |
| 10 | Velyka Bakta | ZO, UA | 2,182 | 0,407 | 0,166 | 2,19± 1,10 | 40,00 |
| 11 | Perechyn | ZO, UA | 3,116 | 0,092 | 0,037 | 3,12± 0,27 | 54,10 |
| 12 | Kireshi | ZO, UA | 2,328 | 0,076 | 0,076 | 2,33± 0,21 | 57,00 |
| 13 | Tsvitne | KO, UA | 3,642 | 0,045 | 0,018 | 3,65± 0,11 | 40,23 |
| 14 | Ivanivka | KO, UA | 1,989 | 0,057 | 0,023 | 1,99± 0,16 | 40,30 |
| 15 | Lviv | LO, UA | 2,293 | 0,124 | 0,124 | 2,30± 0,38 | 48,30 |
| 16 | Oleksandrivka | KO, UA | 1,479 | 0,161 | 0.066 | 1,48± 0,42 | 56,10 |
| 17 | Lubny | PO, UA | 2,022 | 0,138 | 0,056 | 2,03± 0,38 | 45,23 |
| 18 | Zaporizhzhia | ZO, UA | 2,021 | 0,100 | 0,041 | 2,02± 0,29 | 42,50 |
| 19 | Zhytomyr | ŽO, UA | 1,847 | 0,104 | 0,042 | 1,85± 0,30 | 64,00 |
| 20 | Michalovce | KSK, SK | 2,733 | 0,051 | 0,021 | 2,74± 0,13 | 40,00 |
| № | Location/Sample | Extractable substances with 60% ethanol [ % ] | Essential oil yield [ % ] (v/w, expressed on a dry weight basis) |
| 01 | Katerinopolsk, ChR UA | 33± 1 | 0,35± 0,05 |
| 02 | Gorodets, RR UA | 38± 1 | 0,55± 0,05 |
| 03 | Simferopol, ARK UA | 38± 1 | 0,45± 0,05 |
| 04 | Kherson, CR, UA | 35± 1 | 0,20± 0,05 |
| 05 | Zhytomyr 1, ŽR, UA | 35± 1 | 0,30± 0,05 |
| 06 | Maly Tulchyn, VR, UA | 27± 1 | 0,45± 0,05 |
| 07 | Chernihiv, CheR UA | 42± 1 | 0,85± 0,10 |
| 08 | Khmelnytskyi, KhR, UA | 30± 1 | 0,70± 0,05 |
| 09 | Volchytsi, LR, UA | 34± 1 | 0,60± 0,05 |
| 10 | Velyka Bakta, ZR, UA | 21± 1 | 0,25± 0,02 |
| 11 | Perechyn, ZR, UA | 23± 1 | 0,30± 0,05 |
| 12 | Kireshi, ZR, UA | 33± 1 | 0,30± 0,05 |
| 13 | Tsvitne, KR, UA | 37± 1 | 0,35± 0,05 |
| 14 | Ivanivka, KhR, UA | 28± 1 | 0,20± 0,05 |
| 15 | Lviv, LR, UA | 34± 1 | 0,50± 0,05 |
| 16 | Oleksandrivka, KhR, UA | 38± 1 | 0,45± 0,05 |
| 17 | Lubny, PR, UA | 25± 1 | 0,30± 0,05 |
| 18 | Zaporizhzhia, ZR, UA | 27± 1 | 0,25± 0,05 |
| 19 | Zhytomyr 2, ZR UA | 30± 1 | 0,25± 0,05 |
| 20 | Michalovce, KSK, SK | 25± 1 | 0,25± 0,05 |
| Location | Samples [№] |
[g] | Inflorescence mass, % | Essential oil yield [%] |
Essential oil content [%] |
Altitude [m] | Exposure | |
| High mass and high essential oil content | ||||||||
| Chernihiv, CheR UA | 7 | 3,48± 0,17 | 96 | 0,85± 0,10 | 100 | 124 | north | 5° |
| Khmelnytskyi, KhR, UA | 8 | 2,87± 0,13 | 79 | 0,70± 0,05 | 82 | 295 | west | 0° |
| Volchytsi, LR, UA | 9 | 3,31± 0,24 | 91 | 0,60± 0,05 | 71 | 564 | west | 16° |
| High mass and low essential oil content | ||||||||
| Kherson, CR, UA | 4 | 2,99± 0,06 | 82 | 0,20± 0,05 | 24 | 47 | south | 5° |
| Perechyn, ZR, UA | 11 | 3,12± 0,27 | 86 | 0,30± 0,05 | 35 | 176 | west | 0° |
| Tsvitne, KR, UA | 13 | 3,65± 0,11 | 100 | 0,35± 0,05 | 41 | 221 | centre | 4° |
| Michalovce, KSK, SK | 20 | 2,74± 0,13 | 75 | 0,25± 0,05 | 29 | 115 | south- west |
24° |
| Medium mass and medium essential oil content | ||||||||
| Katerinopolsk, ChR UA | 1 | 2,57± 0,12 | 70 | 0,35± 0,05 | 41 | 139 | centre | 15° |
| Gorodets, RR UA | 2 | 1,96 ± 0,04 | 54 | 0,55± 0,05 | 65 | 160 | north-west | 10° |
| Simferopol, ARK UA | 3 | 2,39± 0,80 | 65 | 0,45± 0,05 | 53 | 350 | south | 5° |
| Lviv, LR, UA | 15 | 2,30± 0,38 | 63 | 0,50± 0,05 | 59 | 289 | west | 11° |
| Maly Tulchyn, VR, UA | 6 | 2,89± 0,10 | 79 | 0,45± 0,05 | 53 | 208 | north-west | 12° |
| Low mass and low essential oil content | ||||||||
| Oleksandrivka, KhR, UA | 16 | 1,48± 0,42 | 41 | 0,45± 0,05 | 53 | 133 | centre-east | 7° |
| Zhytomyr 1, ŽR, UA | 5 | 1,70± 0,20 | 47 | 0,30± 0,05 | 35 | 221 | north-west | 2° |
| Zaporizhzhia, ZR, UA | 18 | 2,02± 0,29 | 55 | 0,25± 0,05 | 29 | 105 | south | 13° |
| Zhytomyr 2, ZR UA | 19 | 1,85± 0,30 | 51 | 0,25± 0,05 | 29 | 221 | north- west |
2° |
| Ivanivka, KhR, UA | 14 | 1,99± 0,16 | 55 | 0,20± 0,05 | 24 | 104 | centre | 1° |
| Velyka Bakta, ZR, UA | 10 | 2,19± 1,10 | 60 | 0,25± 0,02 | 29 | 118 | west | 10° |
| Lubny, PR, UA | 17 | 2,03± 0,38 | 56 | 0,30± 0,05 | 35 | 158 | north | 3° |
| Kireshi, ZR, UA | 12 | 2,33± 0,21 | 64 | 0,30± 0,05 | 35 | 164 | west | 16° |
| Samples [№] | Location | Basic composition of chamomile essential oil in % | |||||||||||
| Fa * | Bo | Ch | BoA | BoB | c-.t-Dc | ||||||||
| ** | *** | ||||||||||||
| 1 | Katerinopolsk, ChR | 8,0±0.61 | 13 | 55,2±2,01 | 92 | trace | 0 | 1,8±0,26 | 3 | 2,5±0,53 | 4 | 21,1±2,41 | 35 |
| 2 | Gorodets, RR | 10,3±0,87 | 17 | 54,0±2,22 | 90 | trace | 0 | 5,2±1,22 | 9 | 2,1±0,41 | 3 | 21,0±2,29 | 35 |
| 3 | Simferopol, ARK, UA | 4,4±0,24 | 7 | 41,0±1,87 | 68 | 20,2±1,41 | 34 | 11,0±1,32 | 18 | 9,3±1,21 | 15 | 4,1±0,67 | 7 |
| 4 | Kherson, ChR | 7,1±0,64 | 12 | 33,0±2,78 | 55 | 0,8±0,22 | 1 | 22,0±2,81 | 37 | 2,5±0,54 | 12 | 18,1±1,67 | 55 |
| Samples [№] |
Location | Basic composition of chamomile essential oil in % | |||||||||||
| Fa * ** *** |
Bo | Ch | BoA | BoB | c-.t-Dc | ||||||||
| 8 | Khmelnytskyi, KhR | 8,1 ± 1,56 | 13 | 10,1 ± 1,98 | 17 | 9,4 ± 1,76 | 16 | 22,6 ± 2,78 | 38 | 17,0 ± 1,32 | 28 | 25,2 ± 2,11 | 42 |
| 9 | Volchytsi, LR | 8,2 ± 1,61 | 14 | 9,3 ± 1,42 | 15 | 10,0 ± 1,88 | 17 | 23,2 ± 1,95 | 39 | 15,1 ± 1,79 | 25 | 26,1 ± 1,92 | 43 |
| 6 | Maly Tulchyn, VR | 10,1 ± 1,98 | 17 | 11,1 ± 1,78 | 18 | 7,1 ± 1,12 | 12 | 23,2 ± 2,52 | 39 | 12,1 ± 1,32 | 20 | 28,1 ± 2,77 | 47 |
| 7 | Chernihiv, CheR | 5,2 ± 1,32 | 9 | 10,2 ± 1,76 | 17 | 11,3 ± 1,96 | 19 | 22,1 ± 2,21 | 37 | 12,4 ± 1,61 | 21 | 32,1 ± 2,98 | 53 |
| 5 | Zhytomyr 1, ZR | 8,1 ± 1,67 | 13 | 20,0 ± 2,21 | 33 | 9,3 ± 1,42 | 15 | 17,6 ± 1,65 | 29 | 16,2 ± 2,12 | 27 | 13,1 ± 1,43 | 22 |
| Samples [№] |
Location | Basic composition of chamomile essential oil in % | |||||||||||
| Fa * ** *** |
Bo | Ch | BoA | BoB | c-.t-Dc | ||||||||
| 17 | Lubny, PR | 5,4 ± 1,76 | 9 | 7,3 ± 1,78 | 12 | 1,5 ± 0,23 | 2 | 60,2 ± 2,53 | 100 | 9,2 ± 1,23 | 15 | 12,2 ± 1,2 | 20 |
| 18 | Zhytomyr 2, ŽR | 5,6 ± 1,78 | 9 | 7,5 ± 1,76 | 12 | 1,0 ± 0,29 | 2 | 60,1 ± 2,23 | 100 | 9,0 ± 1,32 | 15 | 13,6 ± 1,4 | 23 |
| 19 | Zaporizhzhia, ZR | 5,1 ± 1,45 | 8 | 10,2 ± 1,71 | 17 | 6,0 ± 1,54 | 1 | 46,2 ± 2,12 | 77 | 14,1 ± 2,22 | 23 | 12,2 ± 1,2 | 20 |
| 10 | Velyka Bakta, ZR | 10,3 ± 1,23 | 17 | 11,0 ± 1,37 | 18 | 10,8 ± 1,45 | 18 | 39,0 ± 1,21 | 65 | 16,4 ± 1,78 | 27 | 5,5 ± 0,51 | 9 |
| 11 | Perechyn, ZR | 6,5 ± 1,89 | 11 | 6,1 ± 1,67 | 10 | 4,8 ± 1,25 | 8 | 50,1 ± 1,45 | 83 | 5,61 ± 1,78 | 9 | 21,1 ± 1,9 | 35 |
| 12 | Kireshi, ZR | 5,5 ± 1,69 | 9 | 13,4 ± 1,98 | 22 | 7,1 ± 1,45 | 12 | 34,7 ± 1,41 | 58 | 22,0 ± 2,73 | 37 | 10,1 ± 1,5 | 17 |
| 13 | Tsvitne, KR | 10,9 ± 2,06 | 18 | 5,2 ± 1,98 | 9 | 4,0 ± 1,65 | 7 | 43,3 ± 2,22 | 72 | 3,5 ± 0,59 | 6 | 22,0 ± 2,2 | 37 |
| 14 | Ivanivka, KhR | 2,5 ± 0,55 | 4 | 4,5 ± 0,59 | 7 | 2,5 ± 0,55 | 4 | 42,0 ± 2,48 | 70 | 5,0 ± 1,78 | 8 | 20,1 ± 2,4 | 33 |
| 15 | Lviv, LR | 10,3 ± 1,98 | 17 | 7,3 ± 1,65 | 12 | 16,2 ± 1,43 | 27 | 38,0 ± 2,32 | 63 | 9,1 ± 1,67 | 15 | 14,0 ± 1,2 | 23 |
| 16 | Oleksandrivka, KhR | 8,9 ± 1,94 | 15 | 3,0 ± 0,54 | 5 | 7,3 ± 1,51 | 12 | 40,0 ± 2,32 | 66 | 20,4 ± 2,86 | 34 | 12,1 ± 1,4 | 20 |
| 20 | Michalovce, KSK, SK | 0,4 ± 0,01 | 1 | 12,4 ± 0,57 | 21 | 15,6 ± 0,51 | 15 | 33,8 ± 3,21 | 56 | 17,2 ± 0,51 | 29 | 20,1 ± 0,5 | 33 |
| Tea/Producer – company name and its production location |
Extractable substances with 60% ethanol |
Essential oil content |
||
| [ % ] | mg. g-1 | [ % ] | ||
| 01 | Romack Kvitky (Chamomile Flower Tea/ Lubipharm, Co., Lubny, UA) | 30 ± 1 | 2.5 ± 0.5 | 0.25 ± 0.05 |
| 02 | Romashka Kvitky (Matricariae Flos/ Viola, Co., Zaporozie, UA) | 39 ± 1 | 3.6 ± 0.5 | 0.36 ± 0.05 |
| 03 | Dietna Dobavka Romashka (Dietary Supplement, Ronfarm, Ltd., Kyev, UA) | 21 ± 1 | 1.0 ± 0.5 | 0.10 ± 0.02 |
| 04 | Karpatskyj Caj – Romashka (Karpatian Tea – Chamomile, Ecoprodukt, Ltd., Ivano-Frankivsk, UA) |
21 ± 1 | 0.8 ± 0.2 | 0.08 ± 0.02 |
| 05 | Romashka Kvitky (Chamomile Flower Tea/Keys of Health, Ltd., Kharkov, UA) | 29 ± 1 | 3.0 ± 0.5 | 0.30 ± 0.05 |
| 06 | Romashka Kvitky (Chamomilae Flos/Keys of Health, Ltd., Kharkov, UA) | 23 ± 1 | 2.0 ± 0.2 | 0.20 ± 0.02 |
| 07 | Romashka Caj (Chamomile Tea/ Galka-Caj, Co., Lviv, UA) | 24 ± 1 | 1.7 ± 0.2 | 0.17 ± 0.15 |
| Tea/Manufacturer | The basic composition of chamomile essential oil in % | ||||||
| Fa | Bo | Ch | BoA | BoB | c-,t-Dc | ||
| 01 | Romashka Kvitky/ Lubipharm, Co. |
7.1±1.23 | 7.0±1.20 | 0.9±0.10 | 53.0±4.8 | 10.0±1,58 | 8.0±1,10 |
| 02 | Romashka Kvitky/ Viola, Co. |
5.3±0.5 | 7.0±0.50 | 0.7±0.21 | 53.0±5.4 | 9.0±1,27 | 11.0±1.6 |
| 03 | Dietna Dobavka Romashka/ Ronfarm, Ltd. |
9.0±1.78 | 9.0±1.2 | 1.0±0.15 | 45.0±3.43 | 10.0±2.2 | 7.0±0.92 |
| 04 | Karpatskyj Čaj – Romashka/ Ecoprodukt, Ltd. |
6.6±0.50 | 11.0±1.70 | 4.1±0.20 | 23.0±2.05 | 18.0±1.0 | 11.0±1.0 |
| 05 | Romashka Kvitky/ Keys of Health, Ltd. |
15.0±2,55 | 14.1±1,55 | 10.0±1.05 | 15.0±1.88 | 22.0±2.7 | 11.0±1.2 |
| 06 | Romashka Kvitky/ Keys of Health |
19.0±1,98 | 11.1±1.6 | 7.3±0.5 | 9.0±0.50 | 19.1±1,90 | 11.0±1.5 |
| 07 | Romashka Caj/ Galka-Caj, Co. |
1.0±0.32 | 1.6±0.3 | 16.3±3.51 | 29.2±2.32 | 34.50±4.86 | 2.5±0.91 |
| Essential oil | type A | type B | type C | type D |
| /-/α-bisabololoxide A | 4,74-15,68 | 31,07-52,25 | 2,13-18,50 | 9,62-25,83 |
| /-/-α-bisabolol | 4,37-15,41 | 8,81-12,92 | 24,18-77.21 | 8,49-19,58 |
| /-/-α-bisabololoxide B | 22,43-58,85 | 5,27-8,79 | 3,17-34,46 | 10,43-24,20 |
| en-in-dicycloethers | 2,61-11,27 | 4,08-9,90 | 1,92-12,00 | 5,51-10,68 |
| chamazulene | 2,70-17,69 | 5,40-7,95 | 1,45-14,90 | 1,91-7,89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




