Submitted:
29 April 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Sample Analysis
2.3. Statistical Analysis
2.3.1. Bayesian Beta Regression Models
2.3.2. Prior Specification
2.3.3. Model Fitting and Diagnostics
2.3.4. Interpretation Framework
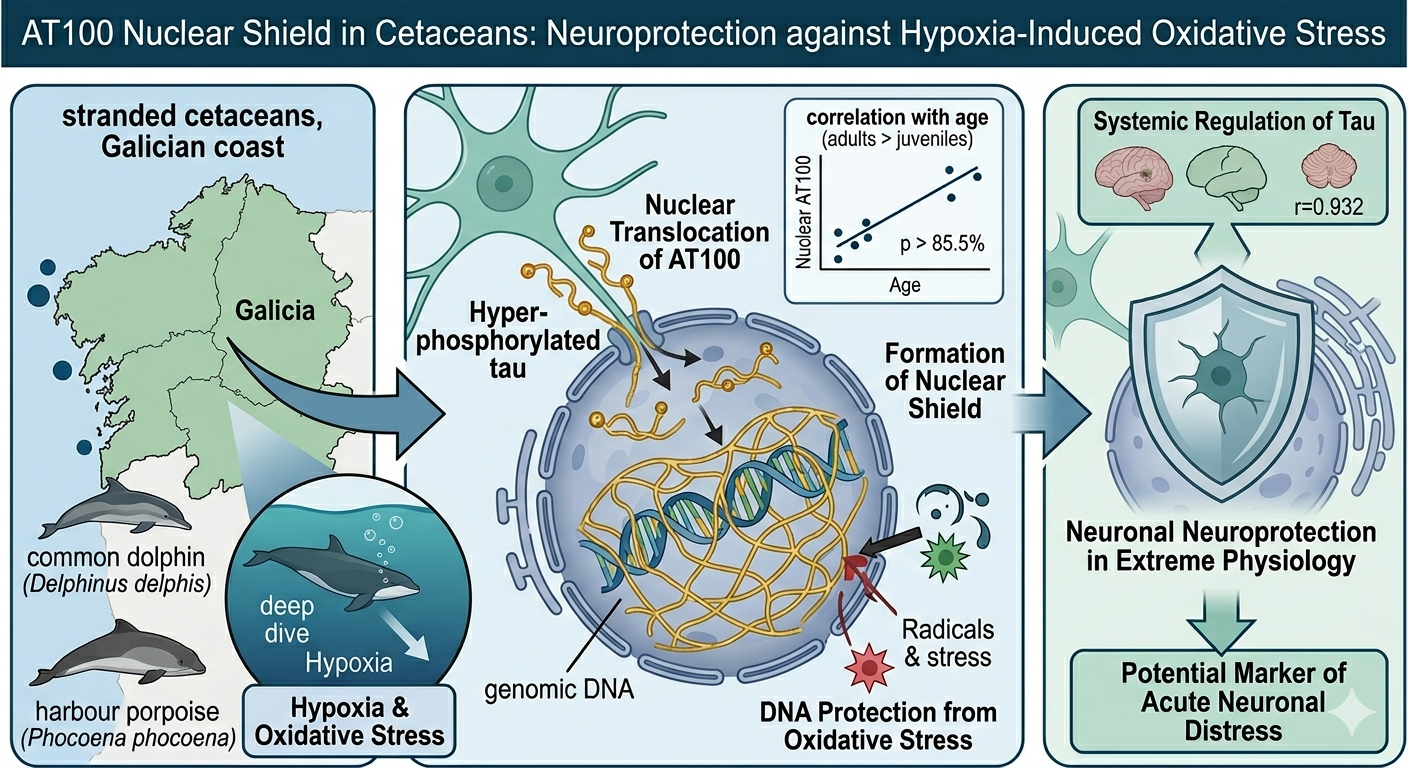
3. Results
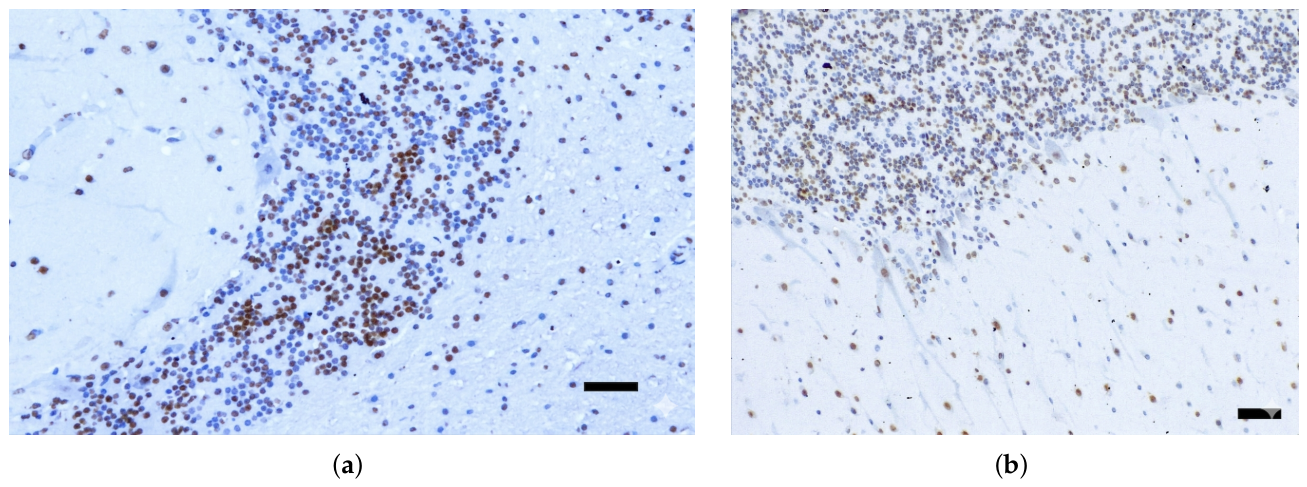
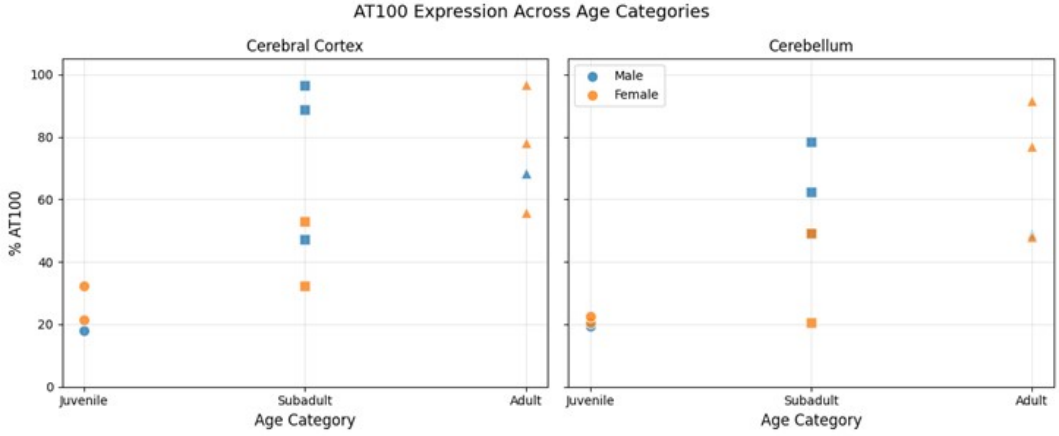
3.1. Immunohistochemical Analysis and AT100 Quantification
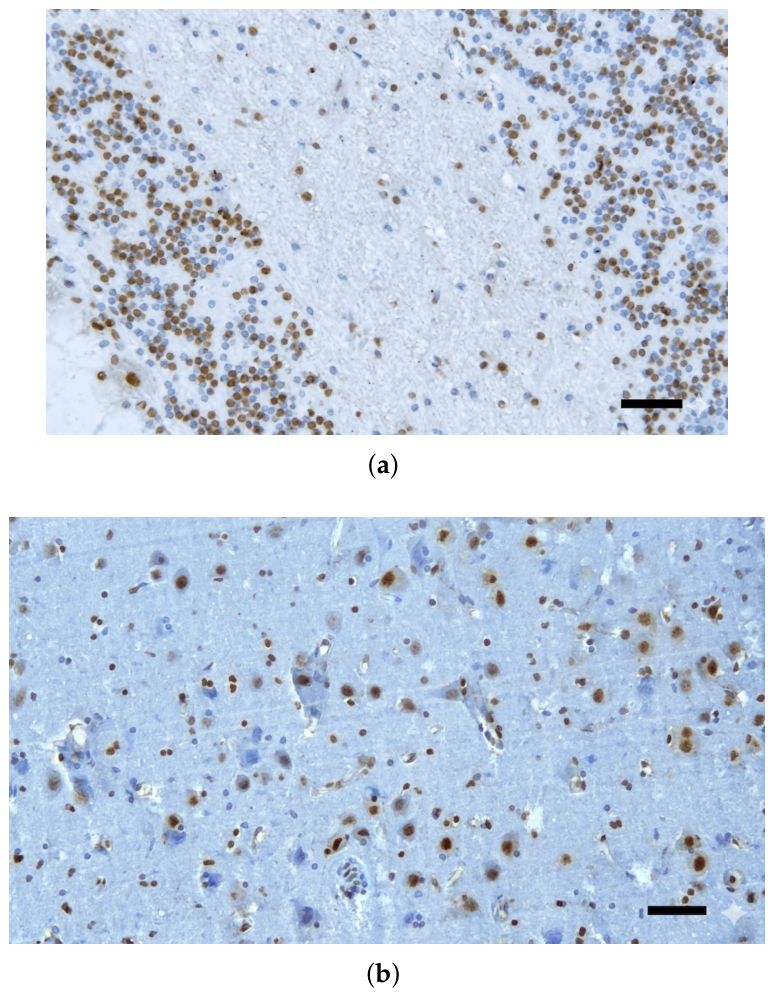
3.2. Atypical AT100 Expression in Two Subadult D. delphis
3.3. Descriptive Statistics
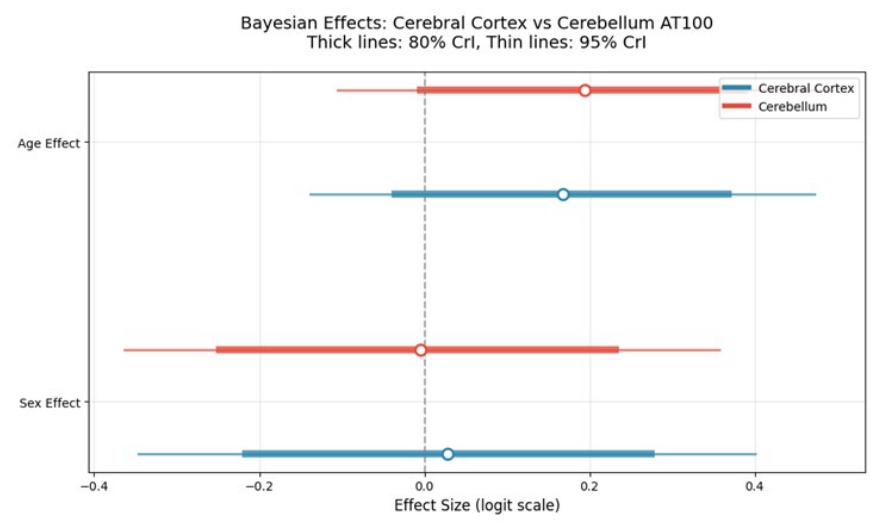
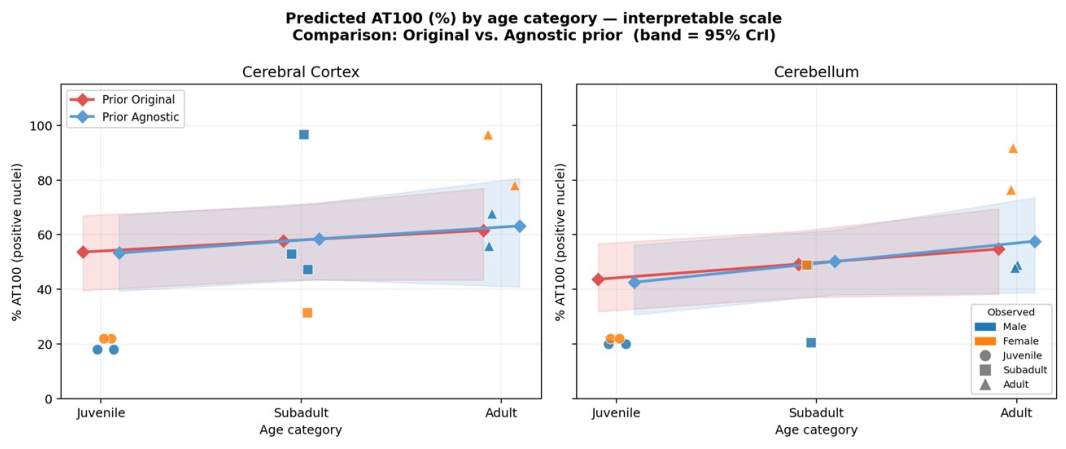
3.4. Bayesian Regression Results
3.4.1. Model Convergence and Fit
3.4.2. Age Effects on AT100 Expression
3.4.3. Sex Effects on AT100 Expression
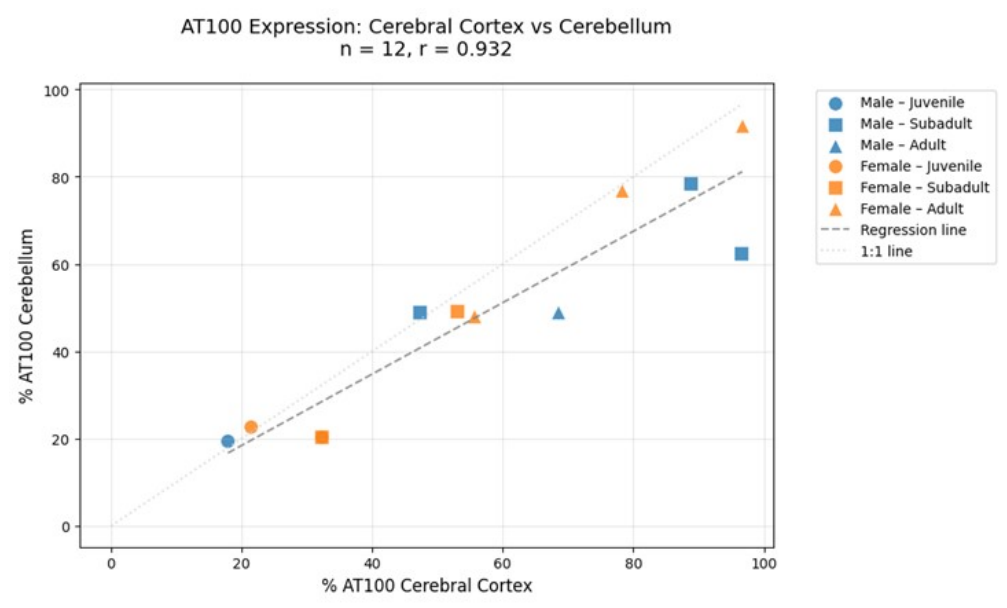
3.5. Regional Comparison
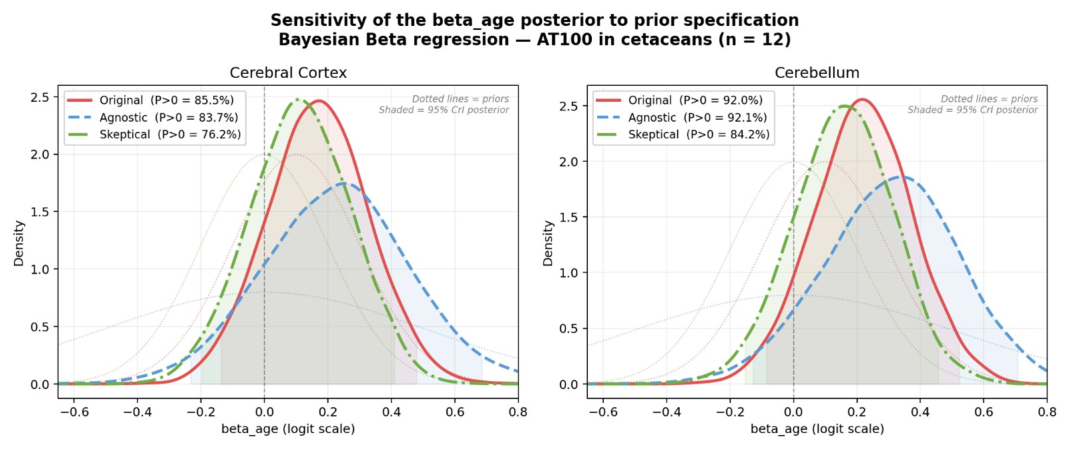
3.6. Sensitivity Analysis for Prior Specification
4. Discussion
5. Conclusions
Abbreviations
| AT100 | Phospho-Tau (Thr212/Ser214) monoclonal antibody clone AT100 |
| CC | Cerebral cortex |
| Cb | Cerebellum |
| CEMMA | Coordinadora para o Estudo dos Mamíferos Mariños |
| CrI | Credible interval |
| DAB | 3,3′-Diaminobenzidine |
| ESS | Effective sample size |
| GLG | Growth Layer Group |
| NUTS | No-U-Turn Sampler |
| pp | Percentage points |
| ROS | Reactive oxygen species |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
References
- Morrison, J.H.; Baxter, M.G. The ageing cortical synapse: Hallmarks and implications for cognitive decline. Nature 2012, 489, 229–235. [Google Scholar] [CrossRef]
- Park, D.C.; Reuter-Lorenz, P. The adaptive brain: Aging and neurocognitive scaffolding. Annu. Rev. Psychol. 2009, 60, 173–196. [Google Scholar] [CrossRef]
- Raz, N.; Lindenberger, U.; Rodrigue, K.M.; Kennedy, K.M.; Head, D.; Williamson, A.; Dahle, C.; Gerstorf, D.; Acker, J.D. Regional brain changes in aging healthy adults: General trends, individual differences and modifiers. Cereb. Cortex 2005, 15, 1676–1689. [Google Scholar] [CrossRef]
- Greenwood, P.M.; Parasuraman, R. Neuronal plasticity and genetic variation in dopamine and acetylcholine receptors as determinants of individual differences in cognitive aging. Neuropsychol. Rev. 2010, 20, 111–126. [Google Scholar]
- Marino, L. Convergence of complex cognitive abilities in cetaceans and primates. Brain Behav. Evol. 2002, 59, 21–32. [Google Scholar] [CrossRef]
- Marino, L.; Connor, R.C.; Fordyce, R.E.; Herman, L.M.; Hof, P.R.; Lefebvre, L.; Lusseau, D.; McCowan, B.; Nimchinsky, E.A.; Pack, A.A.; et al. Cetaceans have complex brains for complex lives. PLoS Biol. 2007, 5, e139. [Google Scholar] [CrossRef] [PubMed]
- Manger, P.R. An examination of cetacean brain structure with a novel hypothesis on the evolution of cetacean brains. Biol. Rev. 2006, 81, 293–338. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.; Dicke, U. Evolution of the brain and intelligence. Trends Cogn. Sci. 2005, 9, 250–257. [Google Scholar] [CrossRef]
- Hof, P.R.; Van der Gucht, E. Structure of the cerebral cortex of the humpback whale, Megaptera novaeangliae (Cetacea, Mysticeti, Balaenopteridae). Anat. Rec. 2007, 290, 1331–1359. [Google Scholar] [CrossRef]
- Striedter, G.F. Principles of Brain Evolution; Sinauer Associates: Sunderland, MA, USA, 2005. [Google Scholar]
- Whitehead, H.; Rendell, L. The Cultural Lives of Whales and Dolphins; University of Chicago Press: Chicago, IL, USA, 2015. [Google Scholar]
- George, J.C.; Bada, J.; Zeh, J.; Scott, L.; Brown, S.E.; O’Hara, T.; Suydam, R. Age and growth estimates of bowhead whales (Balaena mysticetus) via aspartic acid racemization. Can. J. Zool. 1999, 77, 571–580. [Google Scholar] [CrossRef]
- Povlishock, J.T.; Christman, C.W.; Ellis, E.F. The cytoskeleton in health and disease. J. Neuropathol. Exp. Neurol. 1999, 58, 715–732. [Google Scholar]
- Panegyres, P.K.; Zafiris-Toufexis, K. The biology of tau protein. J. Clin. Neurosci. 2002, 9, 6–12. [Google Scholar]
- Gil, L.; Hernández-Ortega, K.; García-Magro, N.; Martínez-Valbuena, I.; Al-Shahrour, F.; Ávila, J.; Rabano, A.; Hernández, F. AT100-tau immunoreactivity in the neuronal nucleus: An aging-related marker in the human brain. Front. Cell. Neurosci. 2017, 11, 352. [Google Scholar]
- Wei, Y.; Qu, M.H.; Wang, X.S.; Chen, L.; Wang, D.L.; Liu, Y.; Hua, Q.; He, R.Q. Binding to the minor groove of DNA by cytoskeletal protein tau at low pH. J. Biol. Chem. 2008, 283, 36390–36398. [Google Scholar]
- Violet, M.; Delattre, L.; Dewachter, I.; Lefebvre, B.; Galas, M.C. A major role for tau in neuronal DNA and RNA protection in vivo under physiological and hyperthermic conditions. Nucleic Acids Res. 2014, 42, 2961–2978. [Google Scholar] [CrossRef]
- Rossi, G.; Dalprà, L.; Crosti, F.; Lissoni, S.; Sciacca, F.L.; Catania, M.; Di Fede, G.; Tagliavini, F. Tau protein binds to DNA and chromatin and its chromosomal distribution is dependent on its phosphorylation state. J. Biol. Chem. 2008, 283, 30332–30342. [Google Scholar]
- Hua, Q.; He, R.Q. Tau protein binds to the distal end of the microtubule. FEBS Lett. 2003, 540, 95–99. [Google Scholar]
- Avila, J.; Lucas, J.J.; Pérez, M.; Hernández, F. Role of tau protein in both physiological and pathological conditions. Physiol. Rev. 2004, 84, 361–403. [Google Scholar] [CrossRef]
- Wang, Y.; Mandelkow, E. Tau in physiology and pathology. Nat. Rev. Neurosci. 2016, 17, 5–21. [Google Scholar] [CrossRef]
- Goedert, M.; Jakes, R.; Vanmechelen, E. Monoclonal antibody AT100 recognizes a conformational epitope of highly phosphorylated tau from Alzheimer’s disease brain. Biochem. J. 1994, 301, 871–877. [Google Scholar] [CrossRef]
- Gärtner, U.; Holzer, M.; Heumann, R.; Arendt, T. Induction of Alzheimer-like phosphorylation of the microtubule-associated protein tau in rat brain and liver by the inhibition of protein phosphatase 2A. Neuroscience 1998, 87, 567–575. [Google Scholar]
- Hernández-Ortega, K.; Ferrera, P.; Blanco-Alvarez, V.M.; Arias, C. Subcellular distribution of tau protein in the human brain: Nuclear tau as a hallmark of neuronal health. J. Alzheimers Dis. 2015, 45, 1169–1181. [Google Scholar]
- Rodríguez-Callejas, J.D.; Fuchs, E.; Pérez-Cruz, C. Evidence of tau hyperphosphorylation and dystrophic microglia in the common marmoset as a naturally occurring model for brain aging and tauopathy. Front. Aging Neurosci. 2016, 8, 214. [Google Scholar] [CrossRef] [PubMed]
- Gil, L.; Ávila, J.; Hernández, F. Nuclear tau in Alzheimer’s disease and other tauopathies. Neurobiol. Aging 2021, 100, 1–12. [Google Scholar]
- Mansuroglu, Z.; Hummel, A.; Thiry, M.; Sergeant, N.; Delattre, L.; Dewachter, I.; Galas, M.C.; Lefebvre, B. Loss of nuclear tau in Alzheimer’s disease: A contributing factor to neurodegeneration? Front. Mol. Neurosci. 2016, 9, 146. [Google Scholar]
- Baquero, M.; Andreu-Reinón, M.E. The brain aging process. Front. Aging Neurosci. 2019, 11, 304. [Google Scholar]
- Lockyer, C. A review of factors involved in the efficiency of age determination from teeth of odontocetes. Rep. Int. Whal. Comm. 1995, 16, 95–109. [Google Scholar]
- Hohn, A.A.; Scott, M.D.; Wells, R.S.; Sweeney, J.C.; Irvine, A.B. Growth layers in teeth from known-age free-ranging bottlenose dolphins. Mar. Mammal Sci. 1989, 5, 315–342. [Google Scholar] [CrossRef]
- Perrin, W.F.; Myrick, A.C. Age Determination of Toothed Whales and Sirenians; IWC Special Issue 3; International Whaling Commission: Cambridge, UK, 1980. [Google Scholar]
- Perrin, W.F.; Reilly, S.B. Reproductive parameters of dolphins and small whales of the family Delphinidae. Rep. Int. Whal. Comm. 1984, 6, 97–133. [Google Scholar]
- Jefferson, T.A.; Webber, M.A.; Pitman, R.L. Marine Mammals of the World: A Comprehensive Guide to Their Identification; Academic Press: San Diego, CA, USA, 2015. [Google Scholar]
- Gelman, A.; Carlin, J.B.; Stern, H.S.; Dunson, D.B.; Vehtari, A.; Rubin, D.B. Bayesian Data Analysis, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- McElreath, R. Statistical Rethinking: A Bayesian Course with Examples in R and Stan, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Ferrari, S.; Cribari-Neto, F. Beta regression for modelling rates and proportions. J. Appl. Stat. 2004, 31, 799–815. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Gelman, A. The No-U-Turn Sampler: Adaptively setting path lengths in Hamiltonian Monte Carlo. J. Mach. Learn. Res. 2014, 15, 1593–1623. [Google Scholar]
- Orekhova, K.; et al. Amyloid-beta and tau pathology in Mediterranean cetaceans. Sci. Rep. 2024, 14, 4521. [Google Scholar]
- Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [CrossRef]
- Ittner, L.M.; Götz, J. Amyloid-β and tau — a toxic pas de deux in Alzheimer’s disease. Nat. Rev. Neurosci. 2011, 12, 65–72. [Google Scholar] [CrossRef]
- Pérez-Cruz, C.; Rodríguez-Callejas, J.D. Non-human primate models of Alzheimer’s disease. Methods Mol. Biol. 2023, 2561, 145–160. [Google Scholar]
- Schindowski, K.; Belarbi, K.; Buée, L. Neurogenesis: Reduction in Alzheimer’s disease or during physiological aging? Am. J. Pathol. 2008, 172, 1373–1386. [Google Scholar]
- Sultan, A.; Nesslany, F.; Violet, M.; Bégard, S.; Loyens, A.; Tulasne, D.; Buée-Scherrer, V.; Ittner, L.M.; Buée, L.; Galas, M.C. Nuclear tau, a key player in neuronal DNA protection. J. Biol. Chem. 2011, 286, 4566–4575. [Google Scholar] [CrossRef]
- Ávila, J.; Llorens-Martín, M.; Pallas-Bazarra, N.; Hernández, F. Role of tau protein in both physiological and pathological conditions. Front. Aging Neurosci. 2012, 4, 23. [Google Scholar]
- Domingo, M.; Visa, J.; Pumarola, M.; Marco, A.J.; Ferrer, L.; Llobera, R.; Kennedy, S. Pathologic and immunocytochemical studies of morbillivirus infection in striped dolphins (Stenella coeruleoalba). Vet. Pathol. 1992, 29, 1–10. [Google Scholar] [CrossRef]
- Di Guardo, G.; Marruchella, G.; Agrimi, U.; Kennedy, S. Morbillivirus infections in marine mammals: A review. Vet. Pathol. 2005, 42, 105–123. [Google Scholar]
- Guzmán-Verri, C.; González-Barrientos, R.; Hernández-Mora, G.; Morales, J.A.; Baquero-Calvo, E.; Chaves-Olarte, E.; Moreno, E. Brucella ceti and brucellosis in cetaceans. Emerg. Infect. Dis. 2012, 18, 175–176. [Google Scholar] [CrossRef]
- Dubey, J.P.; Zarnke, R.; Thomas, N.J.; Wong, S.K.; Van Bonn, W.; Reed, M.; Dierauf, L.; Stephens, I.D. Toxoplasmosis in marine mammals. J. Parasitol. 2003, 89, 752–757. [Google Scholar]
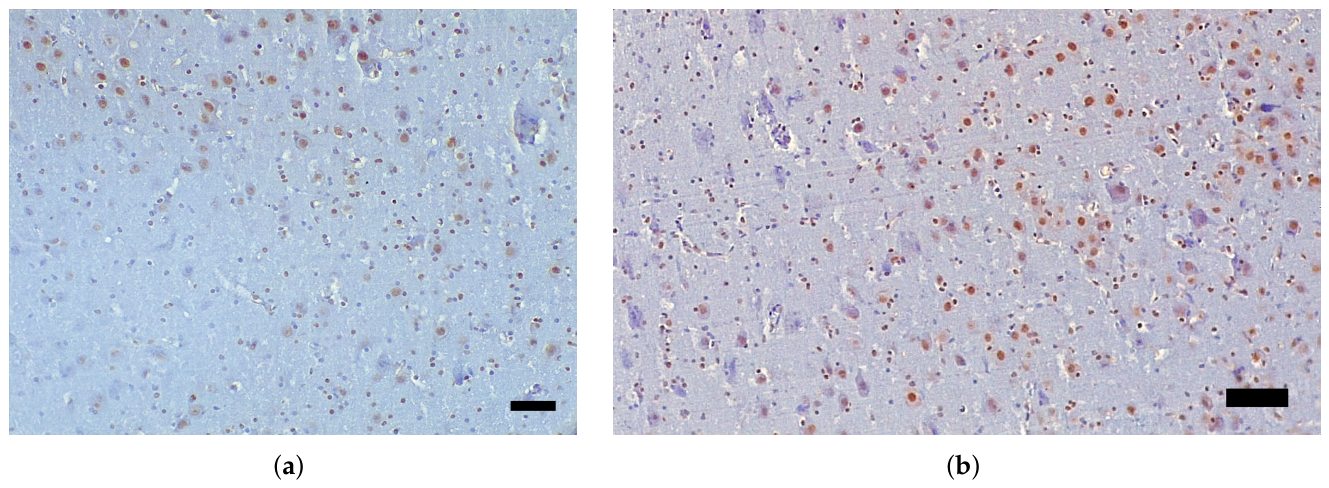
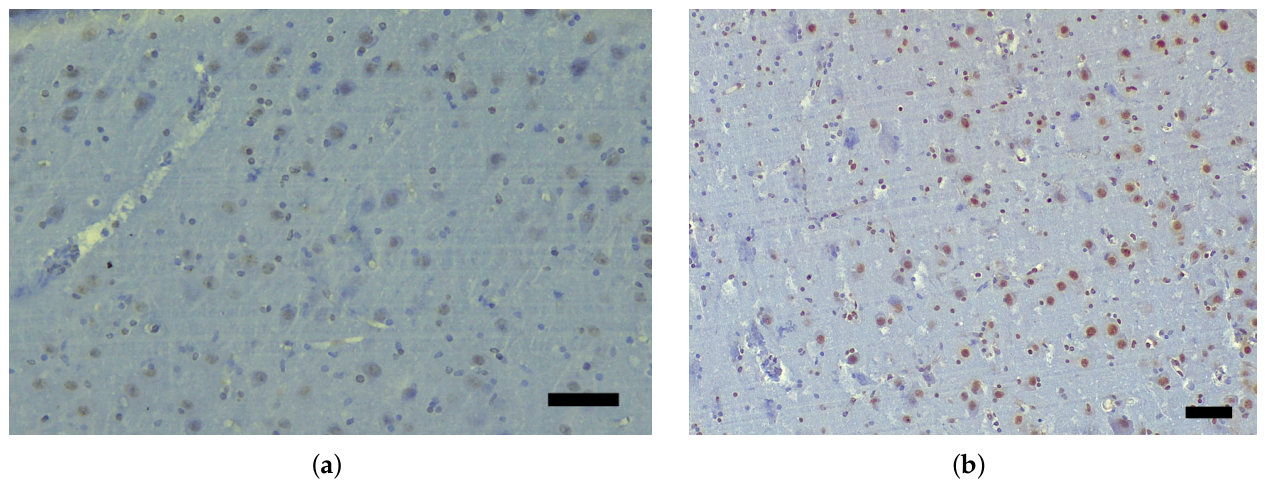
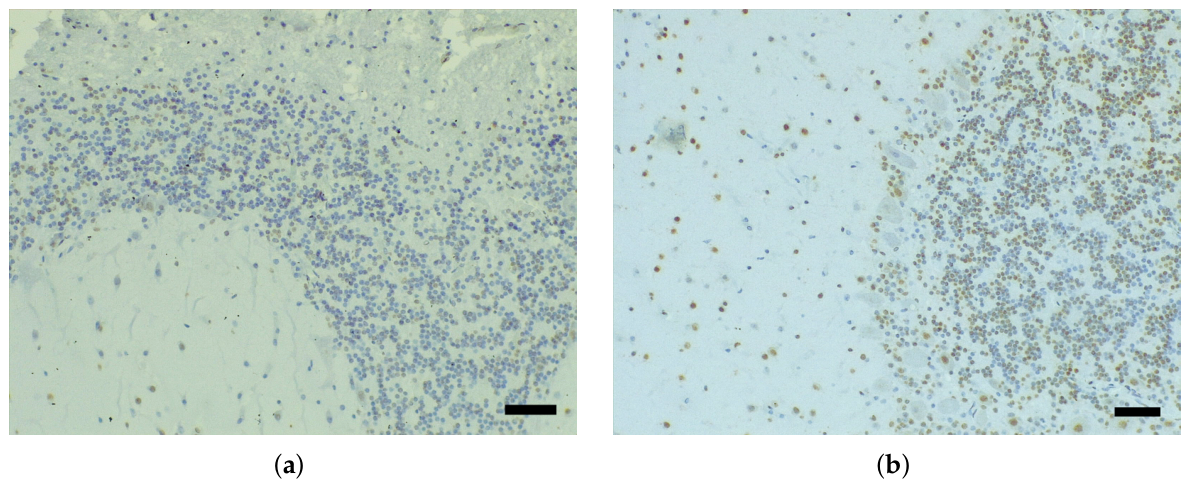
| Species | Age class * | Sex | Length (cm) | Weight (kg) |
|---|---|---|---|---|
| Delphinus delphis | Adult | M | 218 | 95.2 |
| Delphinus delphis | Adult | M | 224 | 200.0 |
| Delphinus delphis | Adult | F | 194 | 110.3 |
| Delphinus delphis | Subadult | M | 159 | 58.3 |
| Delphinus delphis | Subadult | M | 196 | 75.6 |
| Delphinus delphis | Subadult | M | 196 | 74.8 |
| Delphinus delphis | Subadult | F | 170 | 53.2 |
| Delphinus delphis | Juvenile | M | 148 | 35.4 |
| Tursiops truncatus | Adult | F | 278 | 223.0 |
| Stenella coeruleoalba | Juvenile | F | 126 | 23.1 |
| Phocoena phocoena | Subadult | M | 164 | 52.6 |
| Phocoena phocoena | Juvenile | F | 116 | 29.0 |
| Scientific name | Age class | Sex | CC count | AT100 CC (%) | Cb count | AT100 Cb (%) |
|---|---|---|---|---|---|---|
| D. delphis | Adult | M | 156/122 | 78.2 | 156/120 | 76.9 |
| D. delphis | Adult | F | 140/96 | 68.5 | 159/78 | 49.0 |
| D. delphis | Adult | M | 140/78 | 55.7 | 156/75 | 48.0 |
| D. delphis | Juvenile | M | 140/25 | 17.9 | 154/30 | 19.5 |
| D. delphis | Subadult | M | 142/67 | 47.2 | 139/68 | 48.9 |
| D. delphis | **Subadult | M | 145/140 | 96.55* | 149/93 | 62.41* |
| D. delphis | **Subadult | M | 142/126 | 88.7* | 156/123 | 78.4* |
| D. delphis | Subadult | F | 147/47 | 32.2 | 158/32 | 20.5 |
| D. delphis | Juvenile | M | 140/25 | 17.9 | 154/30 | 19.5 |
| T. truncatus | Adult | F | 156/151 | 96.7 | 170/156 | 91.7 |
| S. coeruleoalba | Juvenile | F | 140/30 | 21.4 | 123/28 | 22.7 |
| P. phocoena | Subadult | M | 136/72 | 52.94 | 142/70 | 49.2 |
| P. phocoena | Juvenile | F | 147/47 | 32.2 | 156/32 | 20.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




