Submitted:
30 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Sample Preparation
2.2. Methods for Testing Melting Properties
2.3. Methods for Testing Crystallization Properties
2.4. Methods for Analyzing Microstructure
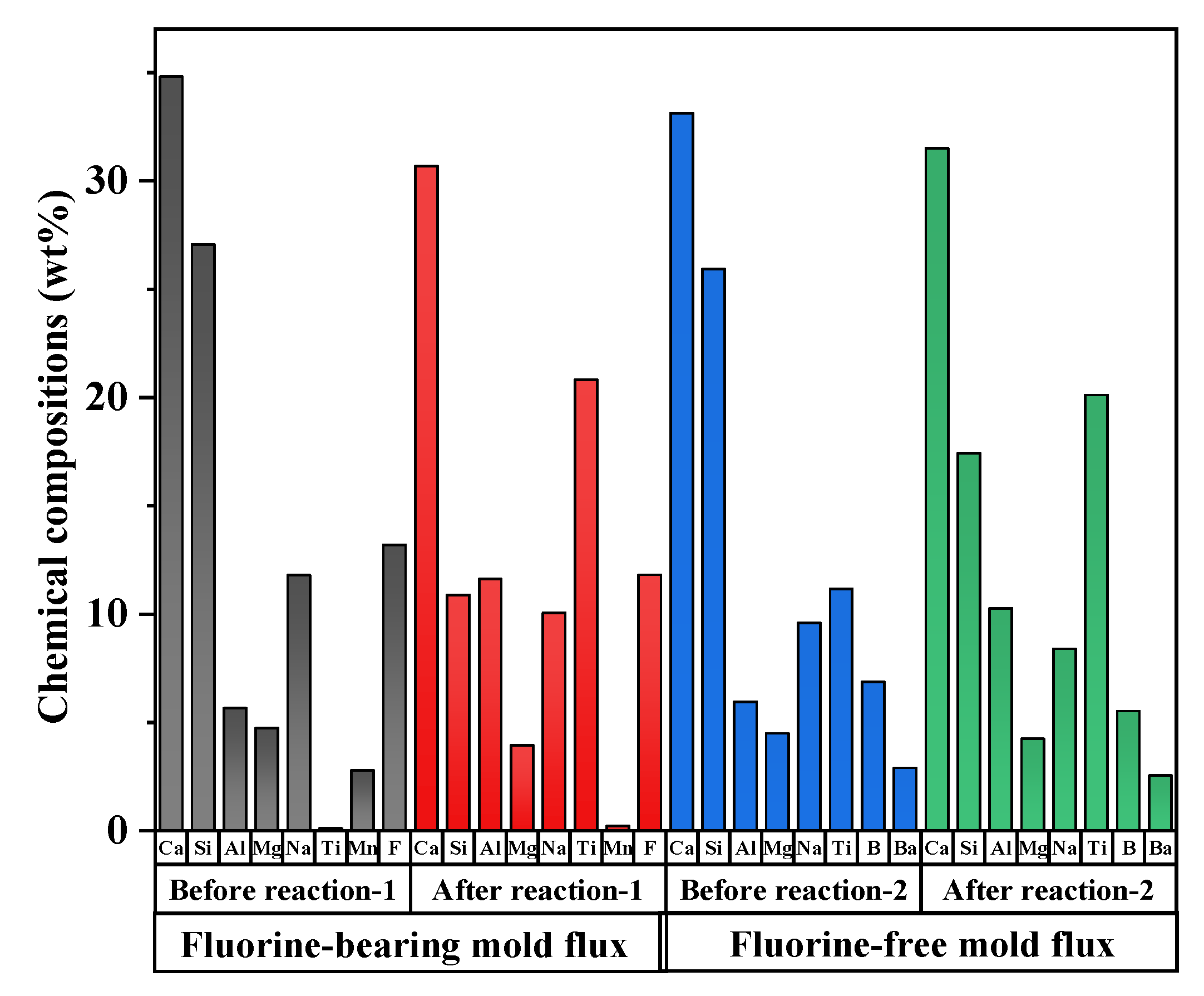
3. Results and Discussion
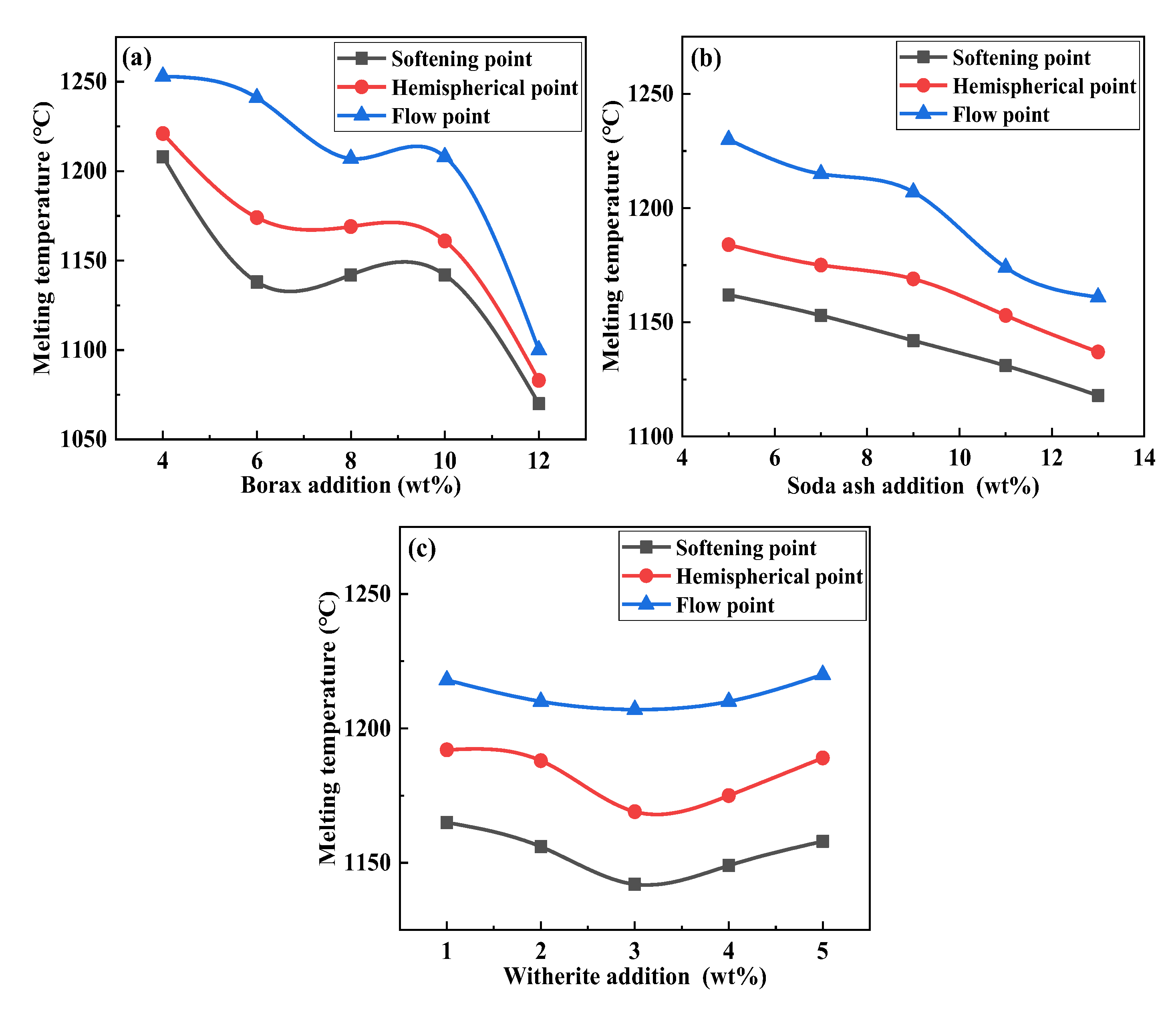
3.1. Effect of Mineral Raw Materials on Melting Temperature of Fluorine-Free Mold Flux
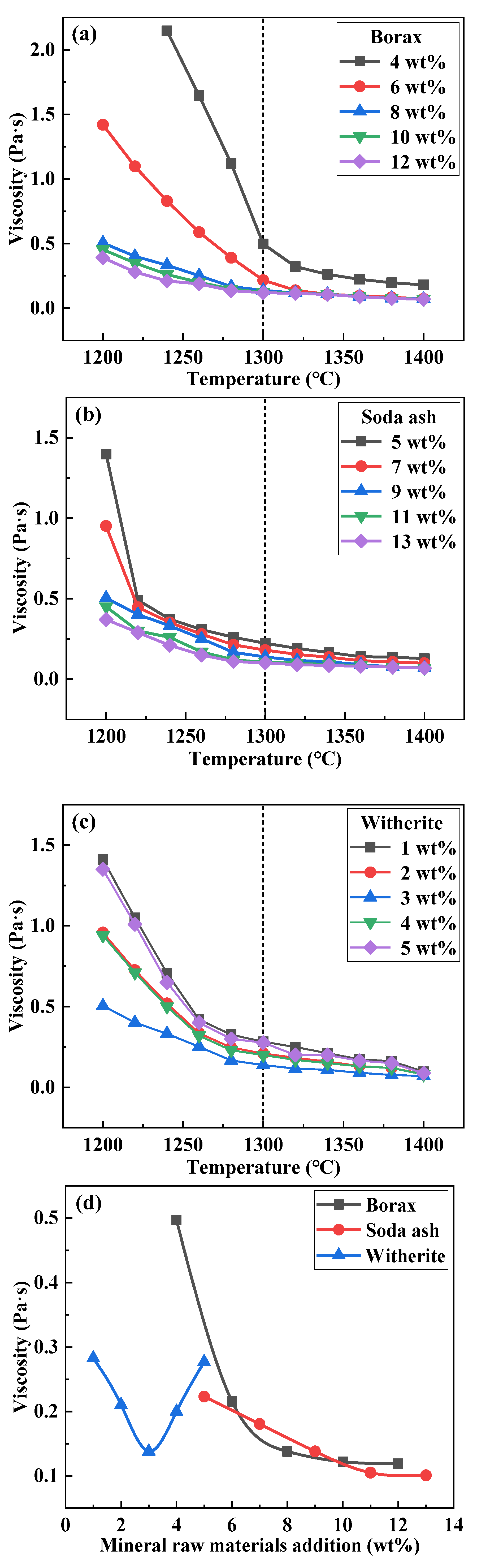
3.2. Effect of Mineral Raw Materials on the Viscosity of Fluorine-Free Mold Flux
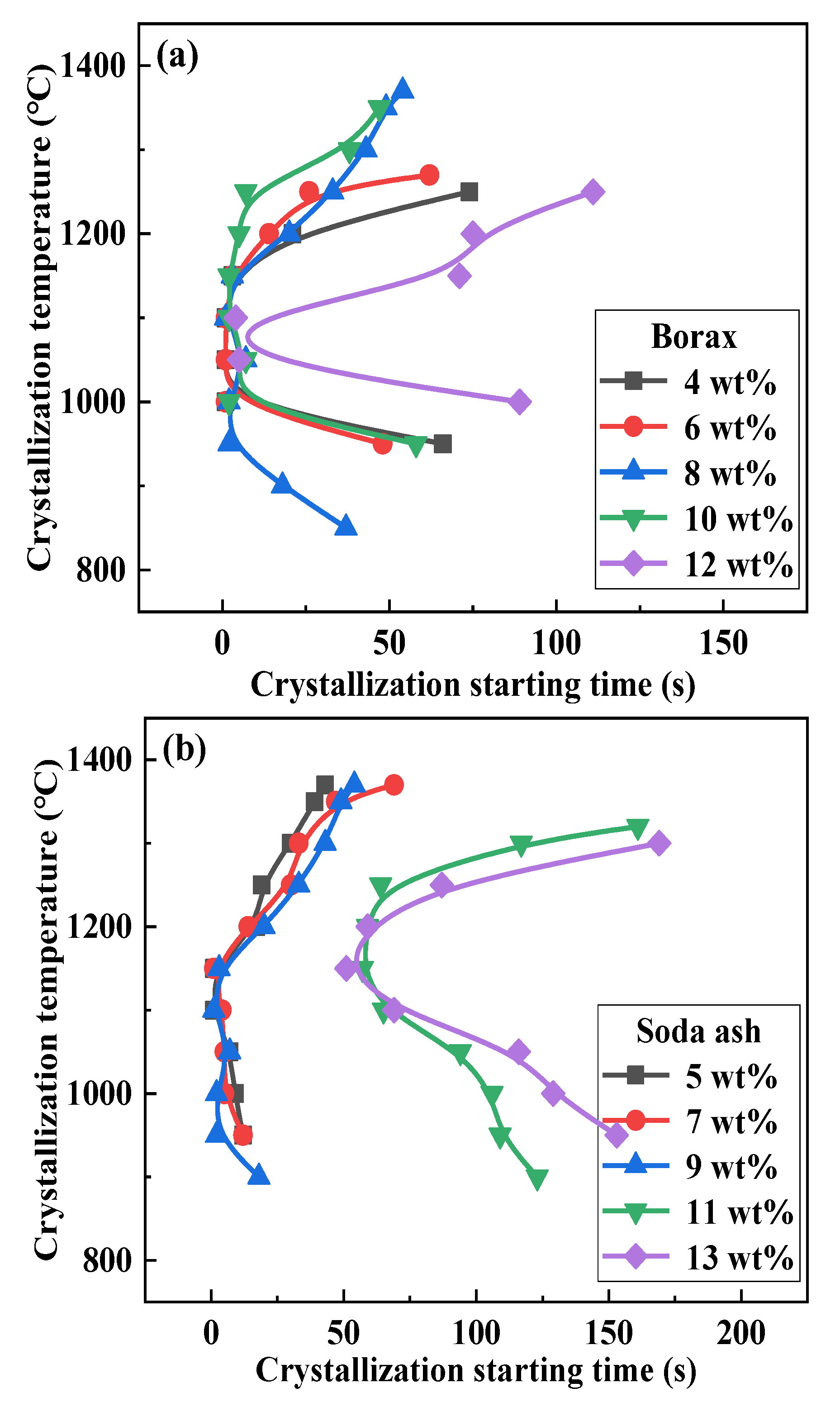
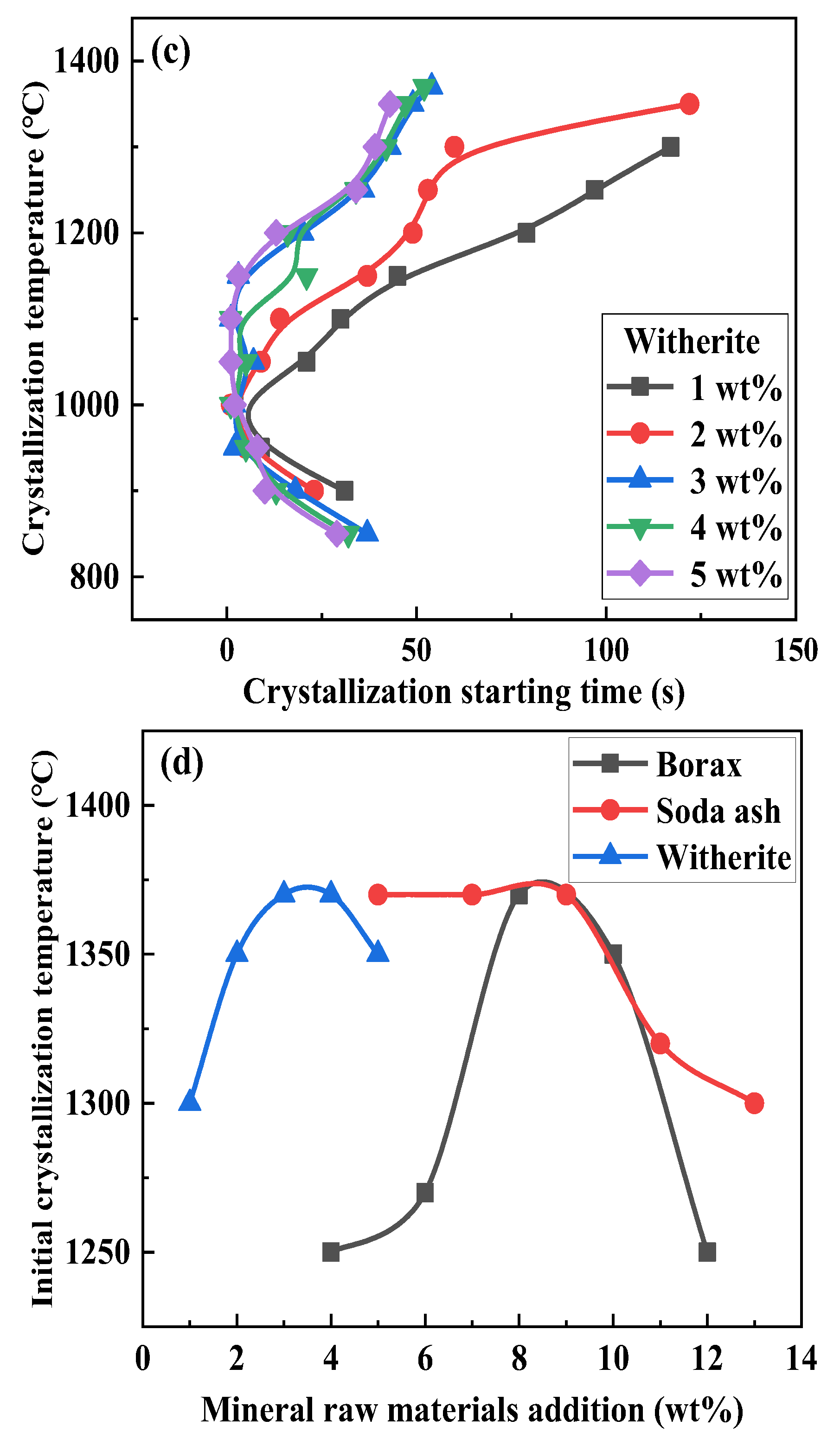
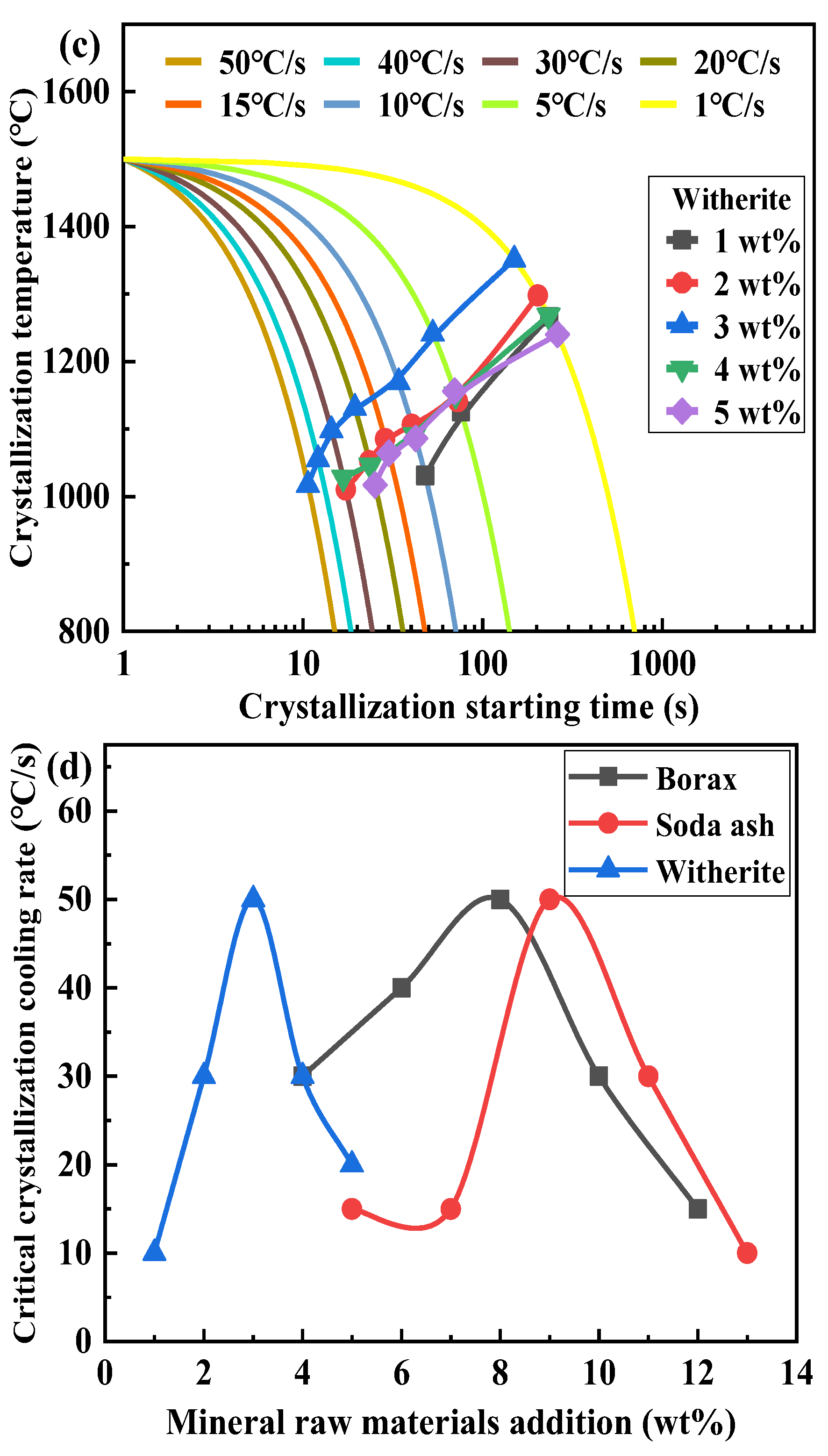
3.3. Effect of Mineral Raw Materials on the Initial Crystallization Temperature of Fluorine-Free Mold Flux
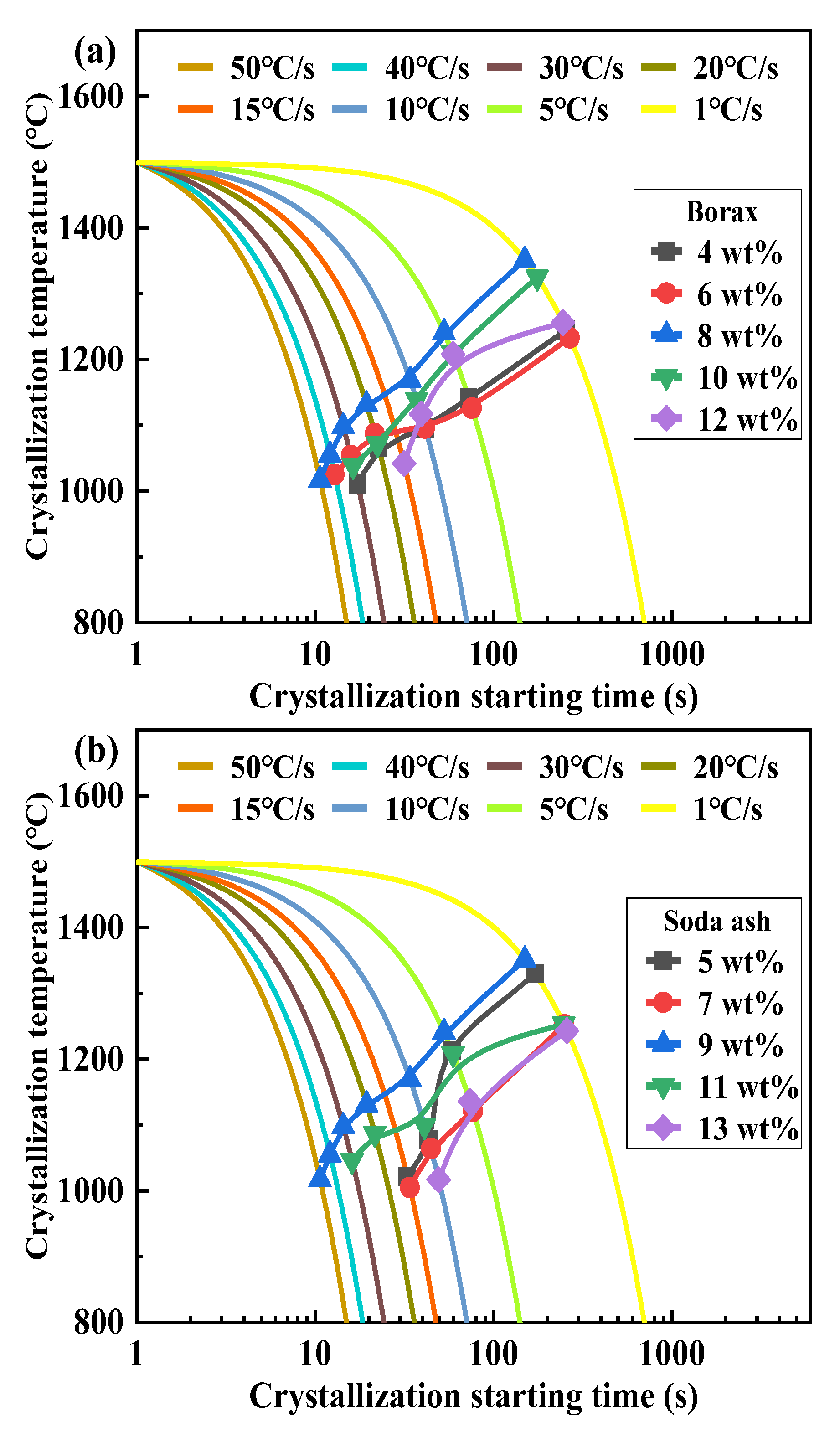
3.4. Effect of Mineral Raw Materials on the Critical Crystallization Cooling Rate of Fluorine-Free Mold Flux
3.5. Effect of Mineral Raw Materials on Crystalline Phases and Crystallization Ratio of Fluorine-Free Mold Flux
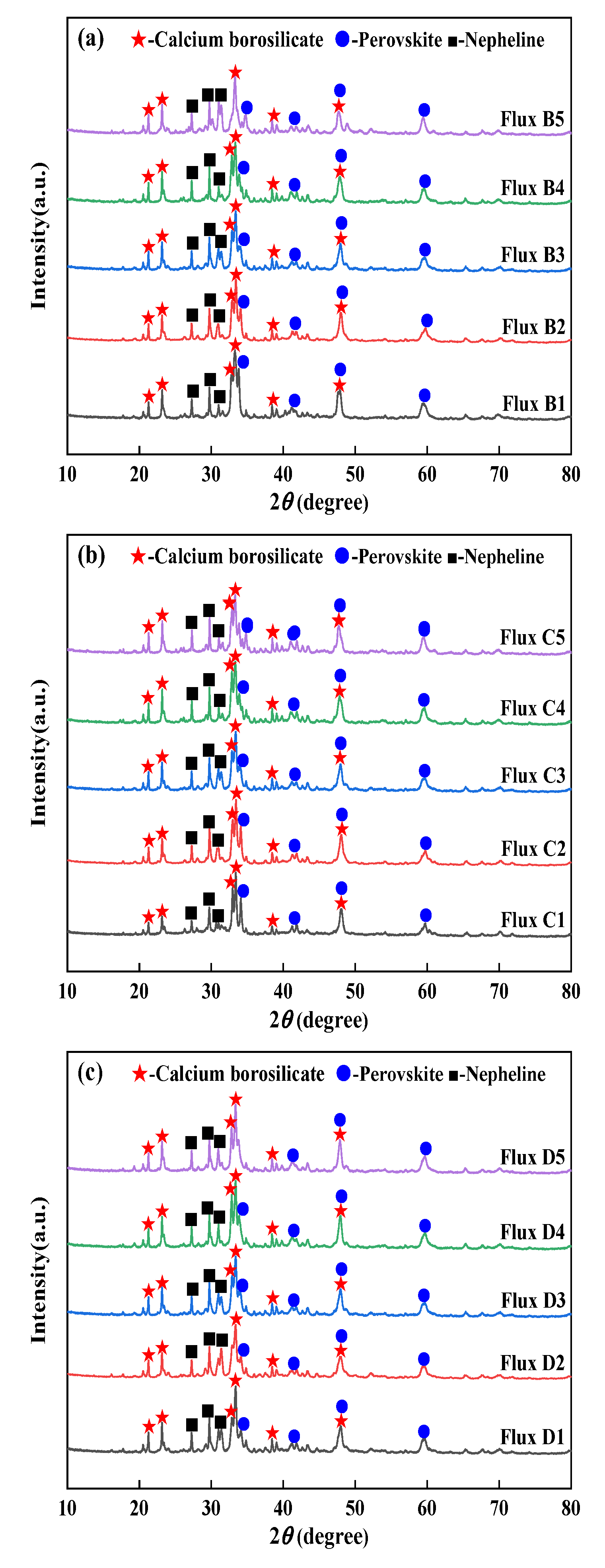
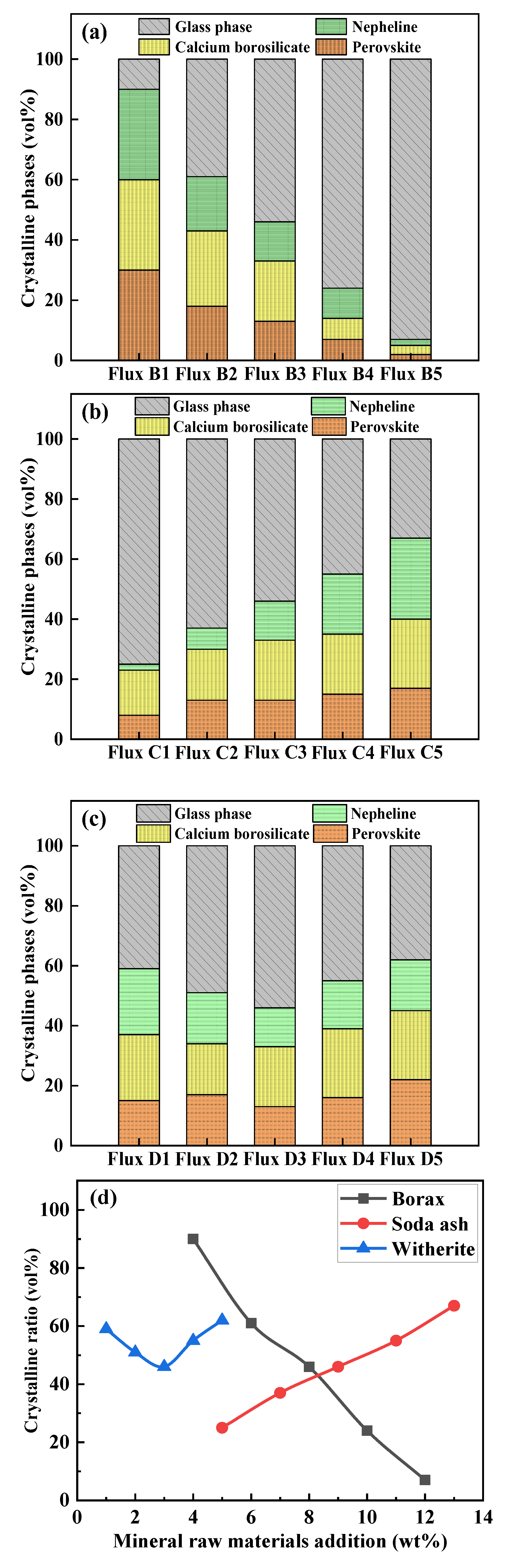

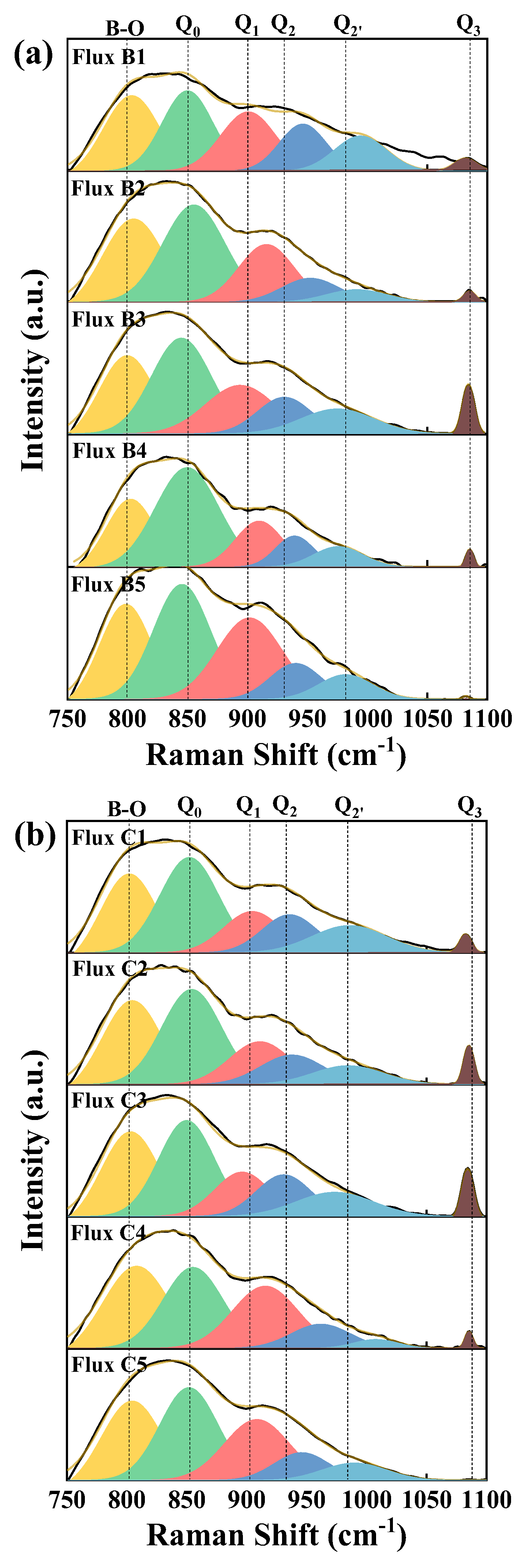
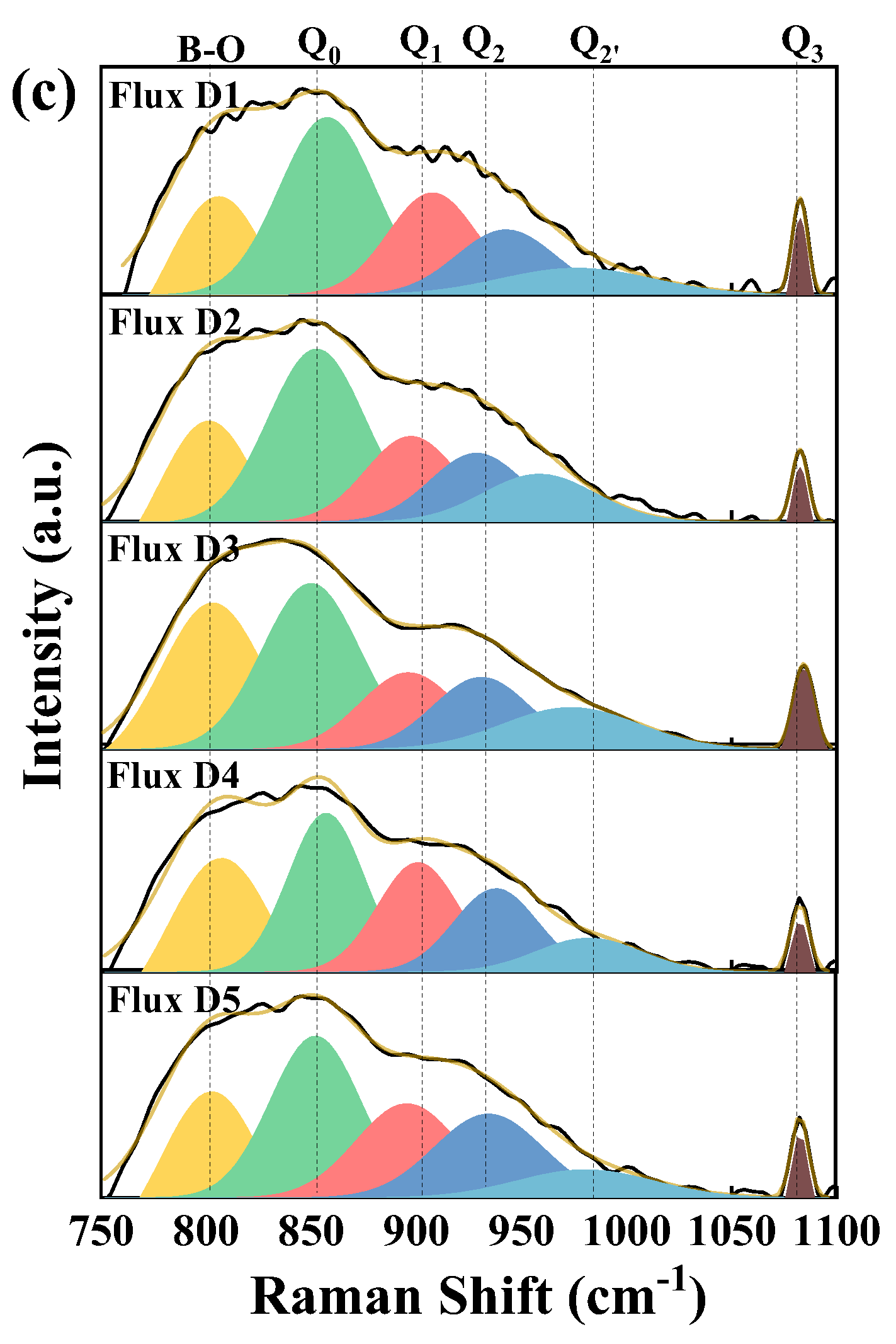
3.6. Effect of Mineral Raw Materials on the Microstructure of Fluorine-Free Mold Flux
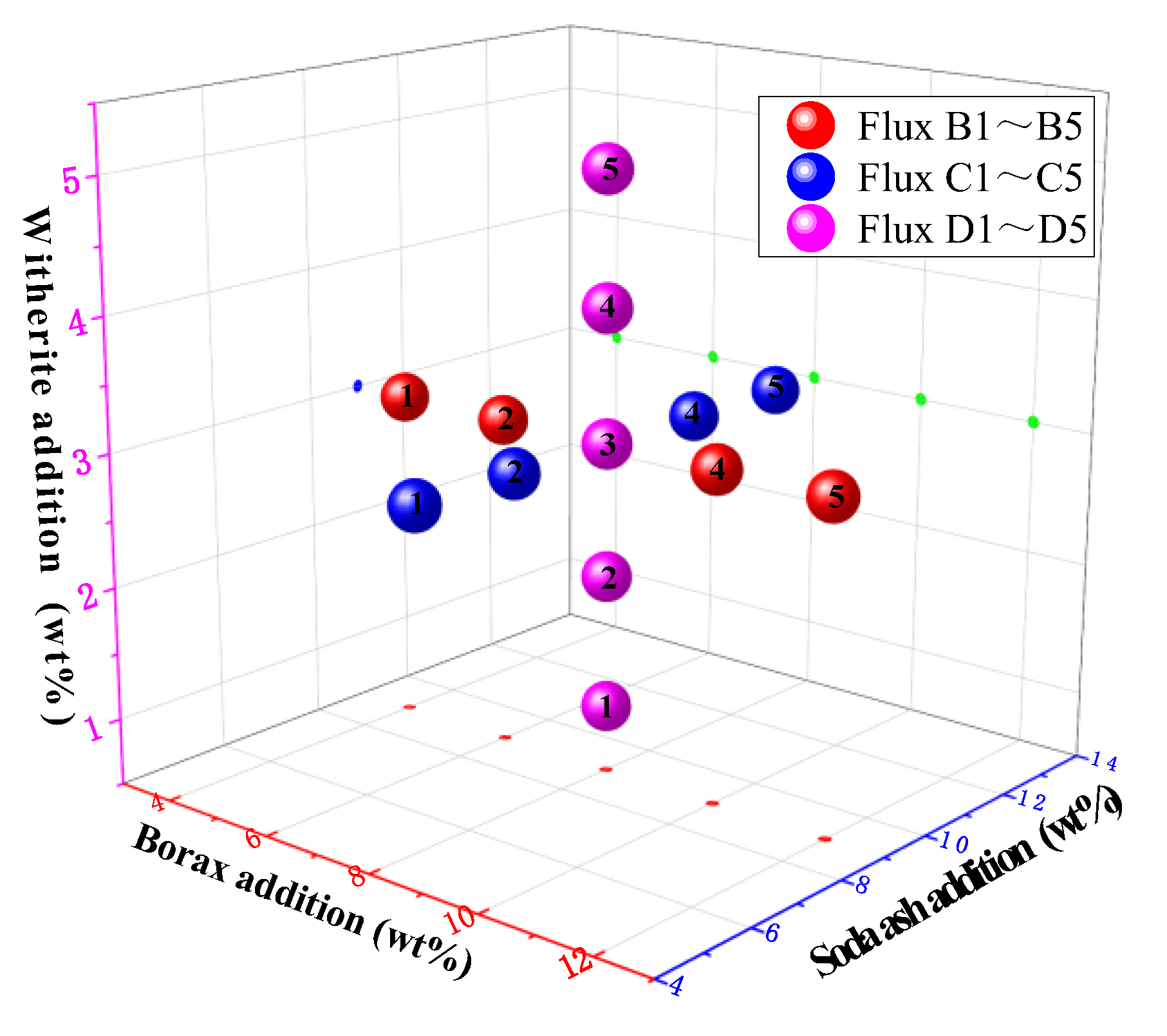
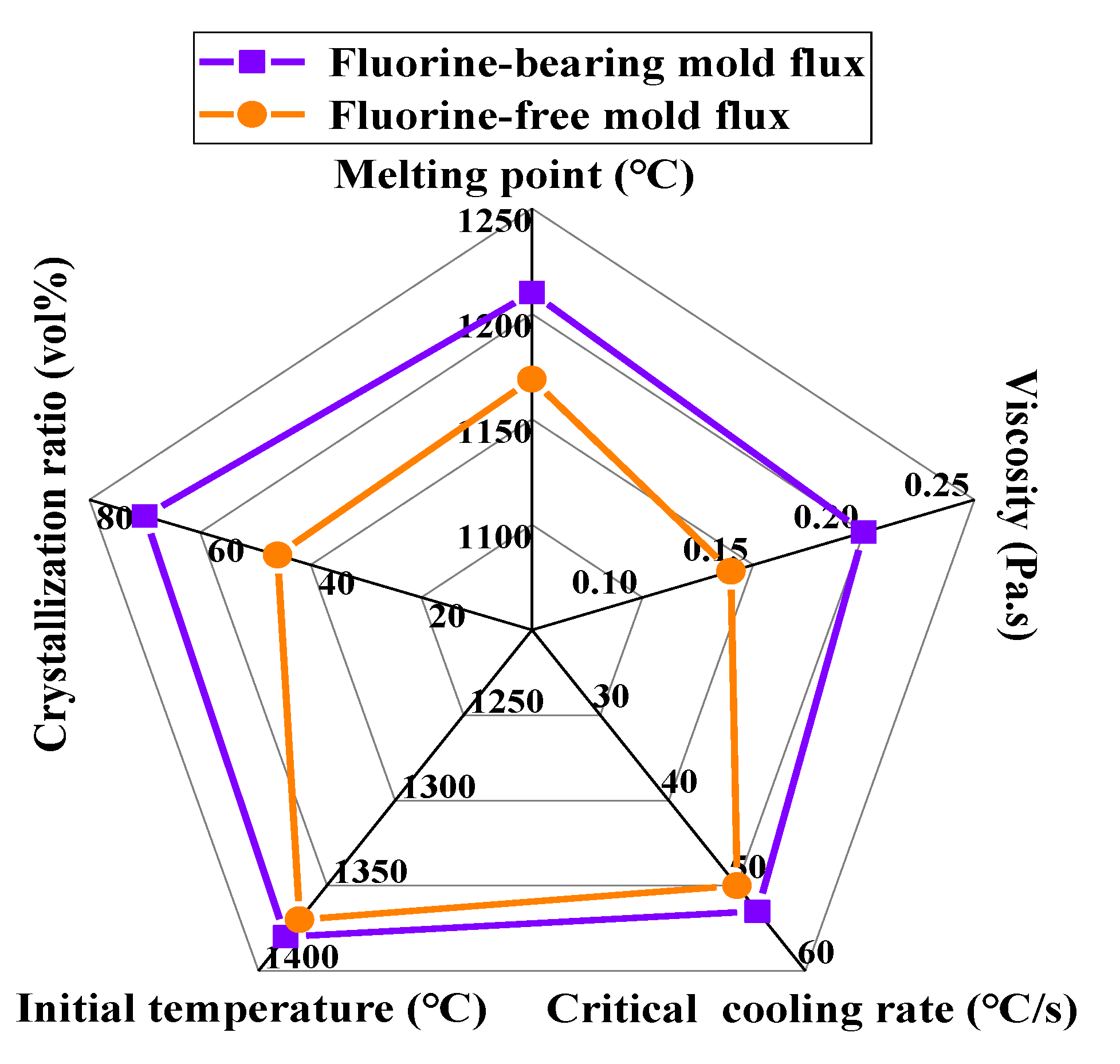
3.7. Optimized Design of Mold Flux for High-Titanium Steel Continuous Casting
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, S.; Gao, Z.; Wu, G.; Mao, X. Titanium microalloying of steel: A review of its effects on processing, microstructure and mechanical properties. Int. J. Miner. Metall. Mater. 2022, 29, 645. [Google Scholar] [CrossRef]
- Yang, P. H.; Yang, J. L.; Xu, J. L.; La, P.; Pan, G.; Cheng, J.; Huang, L.; Fu, H. Investigation of the microstructure and properties on high titanium wear-resistant steels. J. Mater. Res. Technol. 2025, 35, 1926–1940. [Google Scholar] [CrossRef]
- Gao, W.; Wang, S.; Zhang, H.; Wang, Q.; Liu, H.; Yu, H.; Sun, D. Low-Cost Application Strategies of Marine Titanium Alloys and Ti/Fe Dissimilar Materials. Metals 2025, 15, 1205. [Google Scholar] [CrossRef]
- Li, X.; Li, F.; Cui, Y.; Xiao, B.; Wang, J. The effect of manganese content on mechanical properties of high titanium microalloyed steels. Mater. Sci. Eng. A 2016, 677, 340. [Google Scholar] [CrossRef]
- Mills, K. C.; Fox, A. B. The role of mould fluxes in continuous casting—so simple yet so complex. ISIJ Int. 2003, 43, 1479. [Google Scholar] [CrossRef]
- Liu, L.; Han, X.L.; Li, M.D.; Zhang, D. Influence of Mineralogical Structure of Mold Flux Film on Heat Transfer and Longitudinal Cracks of Slabs. Materials 2022, 15, 2980. [Google Scholar]
- Wang, W. L.; Blazek, K.; Cramb, A. A study of the crystallization behavior of a new mold flux used in the casting of transformation-induced-plasticity steels. Metall. Mater. Trans. B 2008, 39, 66. [Google Scholar] [CrossRef]
- Mo, R. Z.; Ren, Y.; Zhang, L. F. Dynamics of mold flux composition in high-titanium steel continuous casting: modeling and prediction. J. Iron Steel Res. Int. 2025, 32, 4240. [Google Scholar] [CrossRef]
- Yan, W.; Hao, Z.Q.; Chen, W.Q.; Li, J. Mixing effect of slag compositions and additives on crystallization of mold flux for high-Ti steel continuous casting. J. Mater. Res. Technol. 2020, 10, 882. [Google Scholar] [CrossRef]
- Wang, W. L.; Cai, D.; Zhang, L.; et al. Effect of TiO2 and TiN on the viscosity, fluidity, and crystallization of fluorine-free mold fluxes for casting Ti-bearing steels. Steel Res. Int. 2021, 92, 2000314. [Google Scholar] [CrossRef]
- Geng, X.; Li, B.Y.; Jiang, Z.H. Effect of TiO₂ on viscosity and structure for CaO–SiO₂–Al₂O₃-based mold flux. Ceram. Int. 2023, 81, 102545. [Google Scholar]
- Wang, X.J.; Jin, H.B.; Zhu, L.G.; Xu, Y.; Liu, R.; Piao, Z.L.; Qu, S. Effect of CaF₂ on the viscosity and microstructure of CaO–SiO₂–Al₂O₃ based continuous casting mold flux. Metals 2019, 9, 871. [Google Scholar]
- Yang, J.; Zhang, J.Q.; Ostrovski, O.; Zhang, C.; Cai, D.X. Effects of fluorine on solidification, viscosity, structure, and heat transfer of CaO–Al₂O₃-based mold fluxes. Metall. Mater. Trans. B 2019, 50, 1694. [Google Scholar]
- Nakada, H.; Nagata, K. Crystallization of CaO–SiO₂–CaF₂ slag as a candidate for mold flux. ISIJ Int. 2006, 46, 441. [Google Scholar] [CrossRef]
- Park, J.Y.; Ko, E.Y.; Choi, J.; Sohn, N. Characteristics of medium carbon steel solidification and mold flux crystallization using the multi-mold simulator. Met. Mater. Int. 2014, 20, 1103. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Zhang, J.Q.; Ostrovski, O.; Sun, S.Y. Qualitative and mechanism research of volatiles in the most commonly used CaO-SiO₂-CaF₂-Na₂O slag during casting process. ISIJ Int. 2013, 53, 1784. [Google Scholar]
- Schulz, T.; Lychatz, B.; Haustein, N.; Janke, D. Structurally based assessment of the influence of fluorides on the characteristics of continuous casting powder slags[J]. Metall. Mater. Trans. B 2013, 44, 317. [Google Scholar] [CrossRef]
- Tiwari, K.K.; Raghav, R.; Pandey, R. Recent advancements in fluoride impact on human health: A critical review. Environ. Sustain. Indic. 2023, 20, 100305. [Google Scholar] [CrossRef]
- Makete, N.; Rizzu, M.; Seddaiu, G.; Gohole, L.; Otinga, A. Fluoride toxicity in cropping systems: mitigation, adaptation strategies and related mechanisms. A review. Sci. Total Environ. 2022, 833, 155129. [Google Scholar] [CrossRef]
- Zhang,L. Xie, X.F.; He,S.P.; Zhang,X.B.; Wang,Q.Q. Influence of TiO2/SiO2 and BaO/CaO ratios on viscosity, crystallization and structure of high-TiO2 mold slags for casting of high-Ti steel. Ceram. Int. 2024, 50, 27188. [CrossRef]
- Zhang,J.J.; Chen, Z.; Du,W. Zhou,J.; Qiang Li. Kinetics of nonisothermal crystallization of SiO2–TiO2–CaO–BaO–Al2O3-based fluorine-free mold flux for high-Ti steel. J. Am. Ceram. Soc. 2025, 109, 70396. [CrossRef]
- Chen, Z.; Zhang, J.J.; Li, X.T.; Du, W.T.; Ma, J.C.; Yang, J. A critical review of the challenges of developing continuous casting mold fluxes for high-Ti steels. Int. J. Miner. Metall. Mater. 2026, 33, 35. [Google Scholar] [CrossRef]
- Mo, R.; Zhang, X.; Ren, Y.; Hu, J.; Zhang, L. Influence of substituting B2O3 with Li2O on the viscosity, structure and crystalline phase of low-reactivity mold flux. Int. J. Miner. Metall. Mater. 2023, 30, 1320. [Google Scholar] [CrossRef]
- Gu, S.; Wen, G.; Ding, Z.; Tang, P.; Liu, Q. Effect of Shear Stress on Isothermal Crystallization Behavior of CaO-Al2O3-SiO2-Na2O-CaF2 Slags. Materials 2018, 11, 1085. [Google Scholar] [CrossRef]
- Zhang, D. Han, X.L.; Liu, L.; Guo, J.J.; Yang,Y.; Wu,L. Physical Properties of Mold Flux and Mineralogical Characteristics of Flux Film for Low-alloy Peritectic Steel Continuous Casting. Materials 2025, 18, 4298. [CrossRef]
- Chen, Z.; Zhang, J. J.; Li, X. T.; et al. A critical review of the challenges of developing continuous casting mold fluxes for high-Ti steels. Int. J. Miner. Metall. Mater. 2026, 33, 35. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, G.H.; Li, Jing. Viscosity and structure evolution of CaO–SiO2-based mold fluxes with involvement of CaO–Al2O3-based tundish fluxes[J]. Ceram. Int. 2020, 46, 14078. [Google Scholar] [CrossRef]
- Geng, X.; Tao, X.R.; Jiang, Z.H.; Zhen, P.L.; Liu, F.B.; Li, H.B. Effect of B2O3 on ESR Slag Viscosity for Remelting 9CrMoCoB Steel. ISIJ Int. 2022, 62, 1070. [Google Scholar] [CrossRef]
- Mo, R. Z.; He, W. Y.; Li, Z. T.; Ren, Y.; Zhang, L.F. Influence of BaO and MgO on viscosity, crystalline phase, and structure of CaO-Al2O3-10%SiO2-20%CaF2-based system[J]. Ceram. Int. 2023, 49, 27311. [Google Scholar] [CrossRef]
- Kashiwaya, Y.; Cicutti, C. E.; Cramb, A. W.; Ishii, K. Development of Double and Single Hot Thermocouple Technique for in Situ Observation and Measurement of Mold Slag Crystallization. ISIJ Int. 1998, 38, 348. [Google Scholar] [CrossRef]
- Lei, Y.; Xie, B.; Ma, W.H. Analysis of crystallization behavior of mold fluxes containing TiO2 using single hot thermocouple technique. J. Iron Steel Res. Int. 2016, 23, 322. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, J.; Zhang, J. Q.; Ostrovski, O.; Zhang, C.; Cai, D.X. Effect of Na2O on properties, structure, and crystallization of CaO-Al2O3-based mold fluxes[J]. Steel Res. Int. 2022, 93, 2100193. [Google Scholar] [CrossRef]
- Chen, Y.; Li, M.; Wang, S. S.; Zhu, L. L.; Zhang, X. B.; He, S. P.; Wang, Q. Q. Effect of MgO on solidification and crystallization properties of ultrahigh-basicity mold flux. Mater. Chem. Phys. 2022, 276, 125403. [Google Scholar] [CrossRef]
- Maldonado, Y. G.; Humberto; Castillejos, E. A. A new method for estimating the isothermal devitrification and crystallization of mold powder slags from non-isothermal DSC data. Mater. Des. 2015, 83, 728. [Google Scholar] [CrossRef]
- Wang, Z.; Shu, Q. F.; Chou, K. C. Crystallization kinetics and structure of mold fluxes with SiO2 being substituted by TiO2 for casting of titanium-stabilized stainless steel. Metall. Mater. Trans. B 2013, 44, 606. [Google Scholar] [CrossRef]
- Zhao, F. H.; Xin, W. B.; Deng, Y. H.; Zhang, Jing.; Cao, Z. J.; Peng, J. Viscosity, structure, and crystallization behavior of CaO-SiO2-CeO2-La2O3-P2O5-CaF2 slag with different Al2O3 contents. Ceram. Int. 2026, 52, 6633. [Google Scholar] [CrossRef]
- Jiang, L. J.; Chou, K.C.; Shu, Q.F. Structure and viscosity of CaO-Al2O3-B2O3 based mould fluxes with varying CaO/Al2O3 ratio. ISIJ Int. 2020, 60, 51. [Google Scholar]
- Mahinroosta, M.; Allahverdi, A. Hazardous aluminum dross characterization and recycling strategies: A critical review. J. Environ. Manag. 2018, 223, 452. [Google Scholar] [CrossRef]
| Raw materials | CaO | SiO2 | Al2O3 | MgO | TiO2 | Na2O | B2O3 | CaCO3 | Na2CO3 | BaCO3 |
|---|---|---|---|---|---|---|---|---|---|---|
| Blast furnace slag | 26.71 | 24.74 | 11.87 | 8.96 | 22.31 | |||||
| quartz sand | 98.32 | |||||||||
| Limestone | >98 | |||||||||
| Borax | 30.49 | 68.51 | ||||||||
| Soda ash | >99 | |||||||||
| Witherite | >99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




