Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Fungal Isolates
2.2. Fungicides
2.3. Mycelial Growth Inhibition Assay
2.4. Data Analysis
3. Results
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CLS | Olive cercospora leaf spot |
| EC50 | Effective concentration (mg L-1) that inhibits 50% of mycelial growth |
| QoIs | Strobilurins fungicides (quinone outside inhibitors) |
| DMIs | Triazoles fungicides (demethylation inhibitors) |
References
- Conde, P.; Montelongo, M.; Leoni, C. Enfermedades del Olivo. In Aceites de oliva: de la planta al consumidor, 1ra. ed.; INIA: Montevideo, Uruguay, 2013; Vol. 1, pp. 183–214. [Google Scholar]
- Ministerio de Ganadería; Agricultura y Pesca-Oficina de Estadísticas Agropecuarias (MGAP-DIEA). Anuario estadístico agropecuario 2025; MGAP-DIEA: Montevideo, Uruguay, 2025. Available online: https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/comunicacion/publicaciones/anuario-estadistico-agropecuario-2025.
- Ministerio de Ganadería; Agricultura y Pesca-Oficina de Estadísticas Agropecuarias (MGAP-DIEA). Censo nacional de olivos 2023; MGAP-DIEA: Montevideo, Uruguay, 2023. Available online: https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/sites/ministerio-ganaderia-agricultura-pesca/files/documentos/noticias/Comunicado_final.pdf.
- Bernaschina, Y.; Leoni, C.; Alaniz, S. Genetic diversity evidence a mixed reproduction mode in Venturia oleaginea populations in Uruguay. J. Plant Pathol. 2020, 102, 123–133. [Google Scholar] [CrossRef]
- Moreira, V.; Mondino, P.; Alaniz, S. Olive anthracnose caused by Colletotrichum in Uruguay: symptoms, species diversity and pathogenicity on flowers and fruits. Eur. J. Plant Pathol. 2021, 160, 663–681. [Google Scholar] [CrossRef]
- Trapero, A.; López, F.J.; Blanco, M.A. Enfermedades. In El cultivo del olivo, 7th ed.; Barranco, D., Fernández-Escobar, R., Rallo, L., Eds.; Mundi-Prensa: Madrid, Spain, 2017; pp. 733–798. [Google Scholar]
- Ávila, A.; Romero, J.; Agustí-Brisach, C.; Abdellatif, B.; Roca, L.F.; Trapero, A. Phenotypic and pathogenic characterization of Pseudocercospora cladosporioides, the causal agent of Cercospora leaf spot of olives. Eur. J. Plant Pathol. 2020, 156, 45–65. [Google Scholar] [CrossRef]
- Lombardo, P.; Leoni, C.; Alaniz, S.; Mondino, P. Cercospora leaf spot of olive in Uruguay. Phytopathol. Mediterr. 2023, 62(3), 413–426. [Google Scholar] [CrossRef]
- Lombardo, P.; Paredes, J.A.; Pugliese, B.D.; González, P.; Alaniz, S.; Mondino, P. Cercospora leaf spot of olive: inoculum production and infections timing throughout the year. Int. J. Pest. Manag. 2024, 71(1), 1–9. [Google Scholar] [CrossRef]
- Moral, J.; Agustí-Brisach, C.; Agalliu, G.; De Oliveira, R.; Pérez-Rodríguez, M.; Roca, L.F.; Romero, J.; Trapero, A. Preliminary selection and evaluation of fungicides and natural compounds to control olive anthracnose caused by Colletotrichum species. Crop Prot. 2018, 114, 167–176. [Google Scholar] [CrossRef]
- Nigro, F.; Antelmi, I.; Sion, V. Integrated control of aerial fungal diseases of olive. Acta Hortic. 2018, 1199, 327–332. Available online: https://<b>www</b>.actahort.org/books/1199/1199_51.htm. [CrossRef]
- Romero, J.; Ávila, A.; Agustí-Brisach, C.; Roca, L.F.; Trapero, A. Evaluation of fungicides and management strategies against Cercospora leaf spot of olive caused by Pseudocercospora cladosporioides. Agronomy 2020, 10, e271. [Google Scholar] [CrossRef]
- Moreira, V.; Carbone, M.J.; Ferronato, B.; Mondino, P.; Alaniz, S. Role of fungicides to control blossom blight and fruit rot the main symptoms of olive anthracnose in Uruguay. Int. J. Pest. Manag. 2024, 71(1), 24–41. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis: A Statistical Treatment of the Sigmoid Response Curve, 2nd ed.; Cambridge University Press: Cambridge, UK, 1952; Available online: https://www.cambridge.org/core/journals/journal-of-the-institute-of-actuaries/article/abs/probit-analysis-by-d-j-finney-ma-scd-2nd-ed-pp-xiv-318-cambridge-university-press-1952-35s/6A77C4EDFD67D3FD9269C796BCB74A0E.
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Mächler, M.; Bolker, B.M. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R. J. 2017, 9(2), 378–400. Available online: https://journal.r-project.org/articles/RJ-2017-066/index.html. [CrossRef]
- Hartig, F. DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.4.7, 2024. Available online: https://CRAN.R-project.org/package=DHARMa (accessed on 28 April 2026).
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Package 'emmeans'. R package version 1.3.2. 2019. [Google Scholar]
- Wickham, H. Ggplot2; Springer International Publishing: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/.
- Nigro, F.; Ferrara, M. Olive cercosporiosis. In Olive Diseases and Disorders; Scherna, L., Agosteo, G.E., Cacciola, S.O., Eds.; Transworld Research Network: Kerala, India, 2011; pp. 247–258. [Google Scholar]
- Moral, J.; Xavier, C.; Roca, L.F.; Romero, J.; Moreda, W.; Trapero, A. Olive anthracnosis and its effect on oil quality. Grasas Aceites 2014, 65(2), e028. [Google Scholar] [CrossRef]
- Roca, L.F.; Moral, J.; Viruega, J.R.; Ávila, A.; Oliveira, R.; Trapero, A. Copper fungicides in the control of olive diseases. Olea 2007a, 26, 48–50. Available online: https://www.researchgate.net/publication/284371685_Copper_fungicides_in_the_control_of_olive_diseases.
- Roca, L.; Viruega, J.R.; Ávila, A.; Oliveira, R.; Marchal, F.; Moral, J.; Trapero, A. Los fungicidas cúpricos en el control de las enfermedades del olivo. Vida Rural 2007b, 256, 52–56. Available online: https://www.researchgate.net/publication/309761862_Los_fungicidas_cupricos_en_el_control_de_las_enfermedades_del_olivo.
- Roca, L.F.; Viruega, J.R.; Ávila, A.; Moral, J.; Marchal, F.; Romero, J.; Agustí, C.; Trapero, A. El cobre: materia activa básica en el control de las enfermedades del olivo. In Proceedings of the 14 Symposium Sanidad Vegetal: hacia el cambio, Sevilla, Spain, 25–27 January 2017; pp. 233–239. Available online: https://www.researchgate.net/publication/317004250_EL_COBRE_MATERIA_ACTIVA_BASICA_EN_EL_CONTROL_DE_LAS_ENFERMEDADES_DEL_OLIVO.
- EUR-Lex. Commission Implementing Regulation (EU) 2018/1981. Off. J. Eur. Union. 2018. Available online: https://eur-lex.europa.eu/eli/reg_impl/2018/1981/oj/eng.
- Fungicide Resistance Action Committee (FRAC). FRAC Code List: Fungal control agents sorted by cross-resistance pattern and mode of action, 2025. Available online: https://www.frac.info/media/ljsi3qrv/frac-code-list-2025.pdf.
- Carmona, M.; Gally, M.; Sautua, F.; Abello, A.; López, P. Uso de mezclas de azoxistrobina y triazoles para controlar enfermedades de fin de ciclo de la soja. Summa Phytopathol. 2011, 37(2), 134–139. [Google Scholar] [CrossRef]
- Manicom, B.Q.; Schoeman, M.H. Fungicides for Pseudocercospora control on avocados: a review, a trial and observations. S. Afr. Avocado Grow. Assoc. Yearb. 2008, 31, 37–43. Available online: https://www.avocadosource.com/Journals/SAAGA/SAAGA_2008/SAAGA_2008_31_PGS_37-43_Manicom.pdf.
- Silva, T.S.; Da Fonseca, L.F.; Yamada, J.K.; Pontes, N.C. Flutriafol and azoxystrobin: an efficient combination to control fungal leaf diseases in corn crops. Crop Prot. 2021, 140, 105394. [Google Scholar] [CrossRef]
- Willis, A.; Duvenhage, J.A. Evaluation of alternative fungicides for control of Cercospora spot on 'Fuerte'. Proceedings V World Avocado Congress (Actas V Congreso Mundial del Aguacate), 2003; pp. 579–583. Available online: https://www.avocadosource.com/WAC5/Papers/WAC5_p579.pdf.
- Oliveira, T.Y.K.; Silva, T.C.; Moreira, S.I.; Christiano, F.S., Jr.; Gasparoto, M.C.G.; Fraaije, B.A.; Ceresini, P.C. Evidence of resistance to QoI fungicides in contemporary populations of Mycosphaerella fijiensis, M. musicola and M. thailandica from banana plantations in southeastern Brazil. Agronomy 2022, 12(12), 2952. [Google Scholar] [CrossRef]
- Oliver, J.E.; Lewis, K.J.; Aktaruzzaman, M.; Ali, M.E. First report of resistance to pyraclostrobin in Pseudocercospora spp. from blackberry (Rubus fruticosus) in Georgia, United States. Plant Health Prog. 2023, 24(3), 394–396. Available online: https://apsjournals.apsnet.org/doi/pdf/10.1094/PHP-12-22-0127-BR. [CrossRef]
- Akinsanmi, O.A.; Miles, A.K.; Drenth, A. Alternative fungicides for controlling husk spot caused by Pseudocercospora macadamiae in macadamia. Australas. Plant Pathol. 2008, 37, 141–147. Available online: https://era.dpi.qld.gov.au/id/eprint/764/1/AlternativeFungicides-Akinsanmi-sec.pdf. [CrossRef]
- Kaewjan, P.; Sittikul, C.; Cheewangkoon, R.; Hyde, K.; Akarapisan, A. Mycosphaerella and other fungi associated with banana leaf diseases from Northern Thailand. Chiang Mai Univ. J. Nat. Sci. 2012, 11(1), 25–32. Available online: https://www.thaiscience.info/journals/Article/CMUJ/10887422.pdf.
- Hong, S.; Hwang, S.; Adriko, J.; Semalulu, O.; Nampamya, D.; Lee, J.W.; Park, T.S. Effect of Pseudocercospora spot disease control and soil moisture management on citrus production in Teso Region, Uganda. J. Korean Soc. Int. Agric. 2020, 32(3), 199–205. [Google Scholar] [CrossRef]
- Sarpkaya, K.; Erkılıç, A. Studies on biology, epidemiology and control of Pseudocercospora pistacina in pistachio. Acta Hortic. 2020, 1269, 33–40. [Google Scholar] [CrossRef]
- Kumar Vishwakarma, V.; Saxena, M.; Kumar Kashyap, V.; Jatav, R.B. In vitro evaluation of strobilurin and triazole fungicides against frogeye leaf spot caused by Cercospora sojina Hara. Int. J. Curr. Microbiol. Appl. Sci. 2021, 10(1), 794–798. [Google Scholar] [CrossRef]
- Ishii, H.; Bryson, P.K.; Kayamori, M.; Miyamoto, T.; Yamaoka, Y.; Schnabel, G. Cross-resistance to the new fungicide mefentrifluconazole in DMI-resistant fungal pathogens. Pestic. Biochem. Physiol. 2021, 171, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, A.P.; Ruth, S.; Satoh-Cruz, M.; Bublitz, D.; Hanson, L.E.; Willbur, J.F. Characterization of fungicide resistance in Cercospora beticola populations from sugarbeet (Beta vulgaris) in Michigan. PhytoFrontiers 2025, 5, 651–662. [Google Scholar] [CrossRef]
- Balaga, K.M.B.; Rey, J.D.; Dacones, L.S. Genetic basis of pathogen establishment and fungicide resistance of phylogenetically related Pseudocercospora spp. through comparative genomics. Philipp. J. Sci. 2022, 151(5), 1857–1876. Available online: https://philjournalsci.dost.gov.ph/genetic-basis-of-pathogen-establishment-and-fungicide-resistance-of-phylogenetically-relate-pseudocercospora-spp-through-comparative-genomics/. [CrossRef]
- Brito, F.S.D.; Santos, J.R.P.; Azevedo, V.C.R.; Peixouto, Y.S.; De Oliveira, S.A.; Ferreira, C.F.; Haddad, F.; Amorim, E.P.; Fraaije, B.; Miller, R.N.G. Genetic diversity and azole fungicide sensitivity in Pseudocercospora musae field populations in Brazil. Front. Microbiol. 2020, 11, e99. [Google Scholar] [CrossRef]
- Chong, P.; Essoh, J.N.; Arango Isaza, R.E.; Keizer, P.; Stergiopoulos, I.; Seidl, M.F.; Guzman, M.; Sandoval, J.; Verweij, P.E.; Scalliet, G.; et al. A world-wide analysis of reduced sensitivity to DMI fungicides in the banana pathogen Pseudocercospora fijiensis. Pest. Manag. Sci. 2021, 77(7), 3273–3288. [Google Scholar] [CrossRef]
- Diaz-Trujillo, C.; Chong, P.; Stergiopoulos, I.; Cordovez, V.; Guzman, M.; De Wit, P.J.G.M.; Meijer, H.J.G.; Scalliet, G.; Sierotzki, H.; Peralta, L.E.; et al. A new mechanism for reduced sensitivity to demethylation-inhibitor fungicides in the fungal banana black Sigatoka pathogen Pseudocercospora fijiensis. Mol. Plant Pathol. 2018, 19(6), 1491–1503. [Google Scholar] [CrossRef]
- Martínez-Bolaños, L.; Téliz-Ortiz, D.; Rodríguez-Maciel, C.; Mora-Aguilera, A.; Nieto-Ángel, D.; Cortéz-Flores, J.I.; Mejía-Sánchez, D.; Nava-Diaz, C.; Silva-Aguayo, G. Resistencia a fungicidas en poblaciones de Mycosphaerella fijiensis del sureste mexicano. Agrociencia 2012, 46(7), 707–717. Available online: http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1405-31952012000700006&lng=es&tlng=es.
- Petrović, E.; Vrandečić, K.; Ćosić, J.; Godena, S. Chemical control of olive fungal diseases: strategies and risks. Poljoprivreda 2024, 30(1), 44–53. [Google Scholar] [CrossRef]
| Locality | Isolate | WGS84 a | Cultivarb |
|---|---|---|---|
| Salto, Olivares Salteños | E07 E12 |
S 31°19’19”, W 54°04’12 | Arbequina Arbequina |
| Salto, Puntas de Valentín | E19 E20 E27 |
S 31°32’55”, W 57°10’28” | n.d. n.d. n.d. |
| Rocha, Nuevo Manantiales | E31 E33 E40 |
S 34°16’26”, W 54°04’12” | Arbequina Arbequina Coratina |
| Maldonado, Agroland | E43 E48 E52 |
S 34°37’24”, W 54°36’56” | Manzanilla Leccino Coratina |
| Montevideo, ARU | E60 E62 |
S 34°43’00”, W 56°16’00” | Arbequina Pendolino |
| Canelones, INIA Las Brujas | E70 E74 |
S 34°40’05”, W 56°20’37” | Leccino Seggianese |
| Montevideo, FAgro | E76 | S 34°50’14”, W 56°13’40” | n.d. |
| Colonia, San Pedro | E78 E82 |
S 34°21’05”, W 57°47’50” S 34°21’30”, W 57°30’09” |
Arbequina Arbequina |
| Chemical group (FRAC)1 | Active ingredient | Trade name | Company | Formulation 2 |
|---|---|---|---|---|
| Inorganic Cupric (M 01) |
Copper calcium sulfate | Bordeles AR Caldo Bordeles F |
Agro Regional Fanaproqui |
74 % wp (Cu 20 %) 68 % wp (Cu 20 %) |
| Copper oxychloride | Oxicloruro Cu AR Fanavid 85 |
Agro Regional Fanaproqui |
85 % wp (Cu 50 %) | |
| Copper oxychloride flowable | Fanavid Flowable | Fanaproqui | 68 % SC (Cu 40 %) | |
| Cuprous oxide | Cuproxido 75 PM Cobre Nordox S75 | Agro Regional Lanafil S.A. |
85 % wp (Cu 75 %) 86 % wp (Cu 75 %) |
|
| Ditiocarbamate (M 03) |
Mancozeb | Mancozeb AR Sancozeb 80 pm |
Agro Regional Lanafil S.A. |
80 % wp |
| Ziram | Ziram Tafirel Ziram Granuflo |
La Forja S.A. Taminco Uy S.A. |
80 % dg 76 % dg |
|
| F-talimide (M04) | Captan | Captan 80 Dikebell Merpan 80 DF |
Agro Regional Lanafil S.A. |
80 % dg |
| Guanidine (U 12) |
Dodine | Dodin Flo Syllit 400 SC |
Agro Regional Lanafil S.A. |
40 % 410 g L-1 cs 39.4 % 400 g L-1 cs |
| Anilino-pyrimidine (9) | Pyrimethanil | Venthos SC | Ineplus S.A. (Proquimur S.A) | 28.3 % 300 g L-1 cs |
| Benzimidazol (1) | Carbendazim | Bencarb - L | Saudu | 45 % 500 g L-1 cs |
| Qol-fungicide Strobilurins 3 (11) |
Azoxystrobin * | Mirador 25 SC Amistar |
Lanafil S.A. Syngenta |
22,9 % 250 g L-1 cs 23.3 % 250 g L-1 cs |
| Pyraclostrobin * | Comet Classic |
Basf S.A. Lanafil S.A. |
23,8 % 250 g L-1 ec | |
| Kresoxim-methyl | Ardent 50 SC | Lanafil S.A. | 46,2 % 500 g L-1 cs | |
| Trifloxystrobin * | Flint 50 WG | Bayer S.A. | 50 % wg | |
| DMI-fungicide Triazole EBI 4 (3) |
Difenoconazole | Score 250 EC | Syngenta Agro Uruguay S.A. | 24 % 250 g L-1 ec |
| Mefentriflucona-zole | Cevya | Basf S.A. | 35 % 400 g L-1 cs | |
| Tebuconazole * | Tebuzate 43 SC | La Forja S.A. | 39,1 % 430 g L-1 cs | |
| Propiconazole * | Quick 250 EC | Saudu S.A. | 25,5 % 250 g L-1 ec |
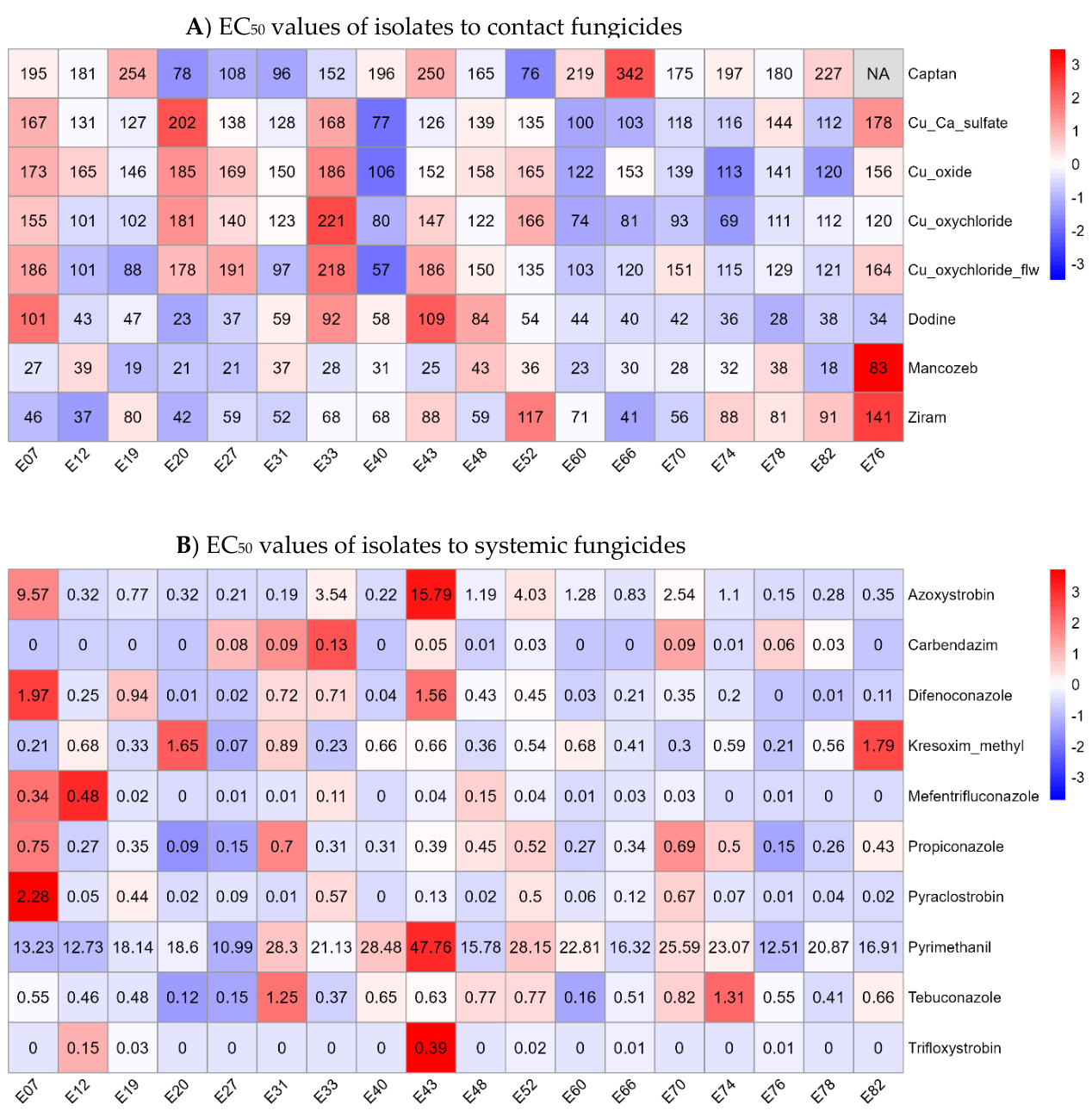
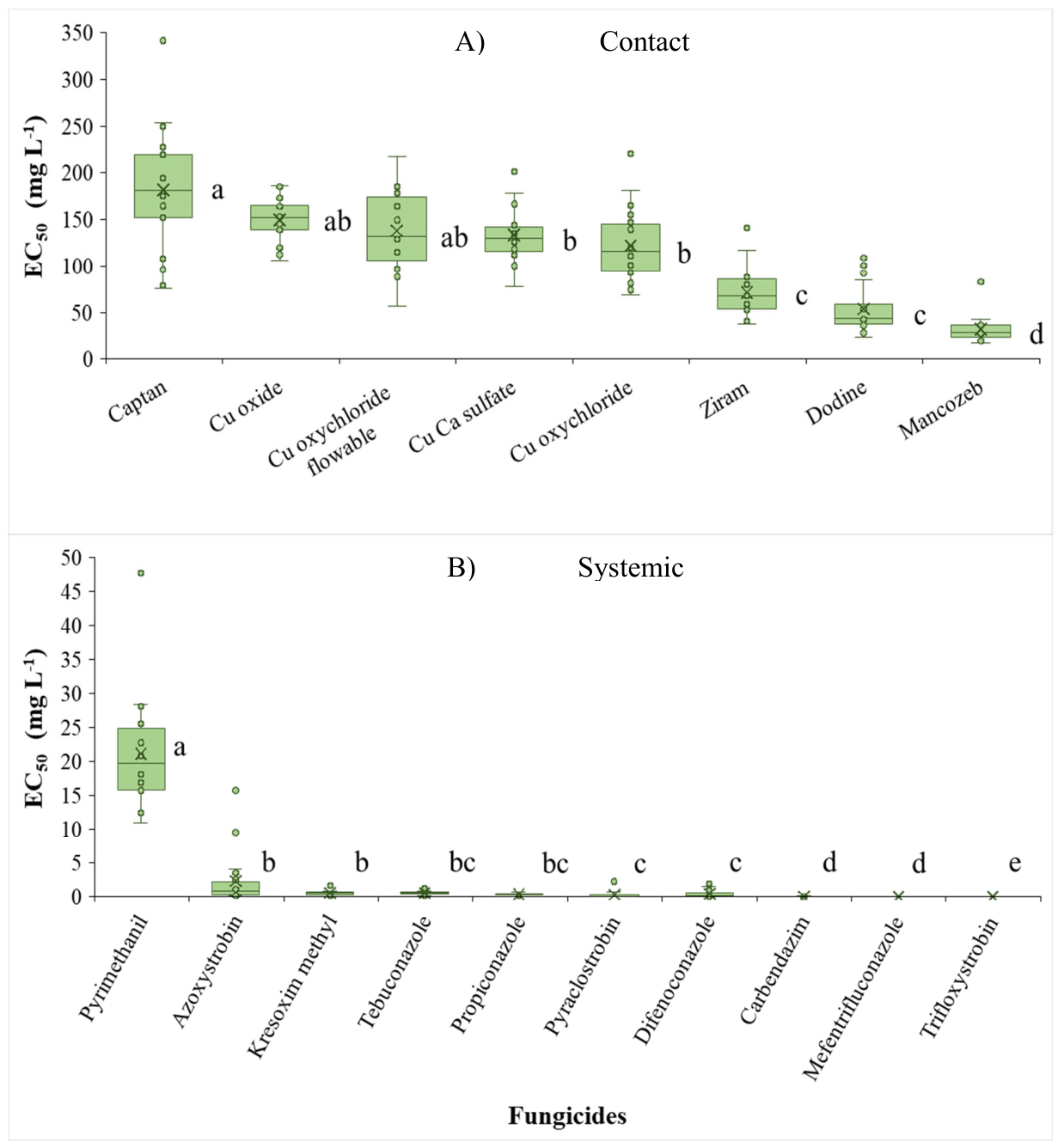
| Chemical group | Active ingredient | EC50 a,b ± SEc | EC50 range | ||
|---|---|---|---|---|---|
| Contact fungicides and dodine | |||||
| F-talimide | Captan | 185 | ± 19 | a | 76-342 |
| Cupric | Cuprous oxide | 150 | ± 15 | ab | 106-186 |
| Cupric | Copper oxychloride flowable | 138 | ± 14 | ab | 57-218 |
| Cupric | Copper calcium sulfate | 131 | ± 13 | b | 77-202 |
| Cupric | Copper oxychloride | 123 | ± 12 | b | 69-221 |
| Ditiocarbamate | Ziram | 71 | ± 7 | c | 37-141 |
| Guanidine | Dodine | 53 | ± 5 | c | 23-109 |
| Ditiocarbamate | Mancozeb | 30 | ± 3 | d | 27-83 |
| Systemic Fungicides | |||||
| Anilino-Pyrimidine | Pyrimethanil | 25.3 | ± 12.5 | a | 11.0-47.8 |
| Strobilurin | Azoxystrobin | 1.05 | ± 0.52 | b | 0.15-15.8 |
| Strobilurin | Kresoxim-methyl | 0.96 | ± 0.49 | b | 0.07-1.79 |
| Triazole | Tebuconazole | 0.56 | ± 0.28 | bc | 0.12-1.31 |
| Triazole | Propiconazole | 0.44 | ± 0.22 | bc | 0.09-0.75 |
| Strobilurin | Pyraclostrobin | 0.19 | ± 0.10 | c | 0.003-2.28 |
| Triazole | Difenoconazole | 0.19 | ± 0.10 | c | 0.003-1.97 |
| Benzimidazol | Carbendazim | 0.03 | ± 0.02 | d | <0.001-0.13 |
| Triazole | Mefentrifluconazole | 0.03 | ± 0.01 | d | <0.001-0.48 |
| Strobilurin | Trifloxystrobin | 0.006 | ± 0.003 | e | <0.001-0.39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




