Submitted:
30 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Site and Environmental Conditions
2.2. Biological and Chemical Materials Sourcing
2.3. Seed Pre-Treatment and Enhancement Protocol
2.4. Data Analysis
2.4.1. Experimental Design and Statistical Analysis
2.5. Seed Germination and Seedling Vigor Analysis
2.6. Seedling Morphological and Biomass Assessment
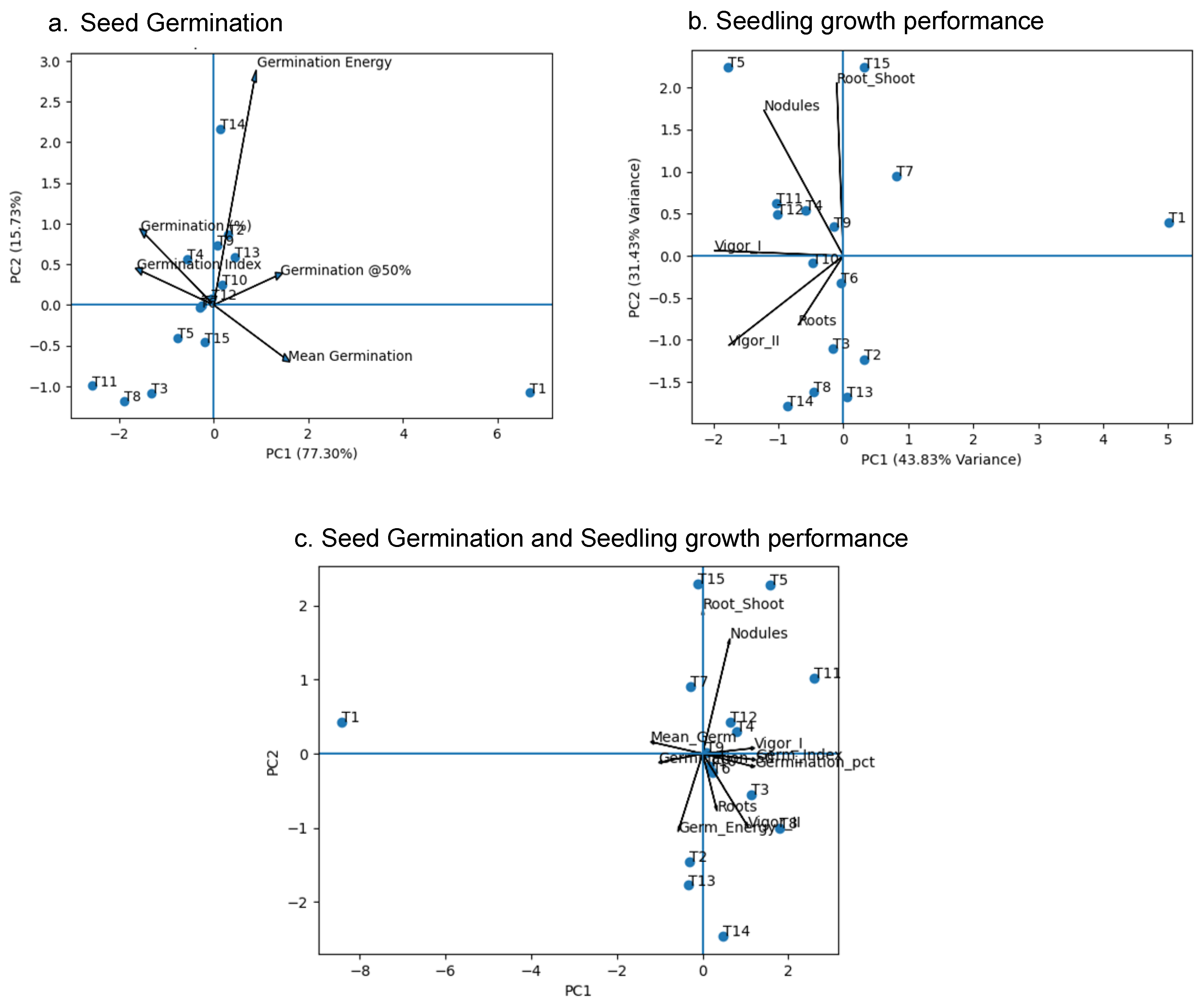
3. Results and Discussion
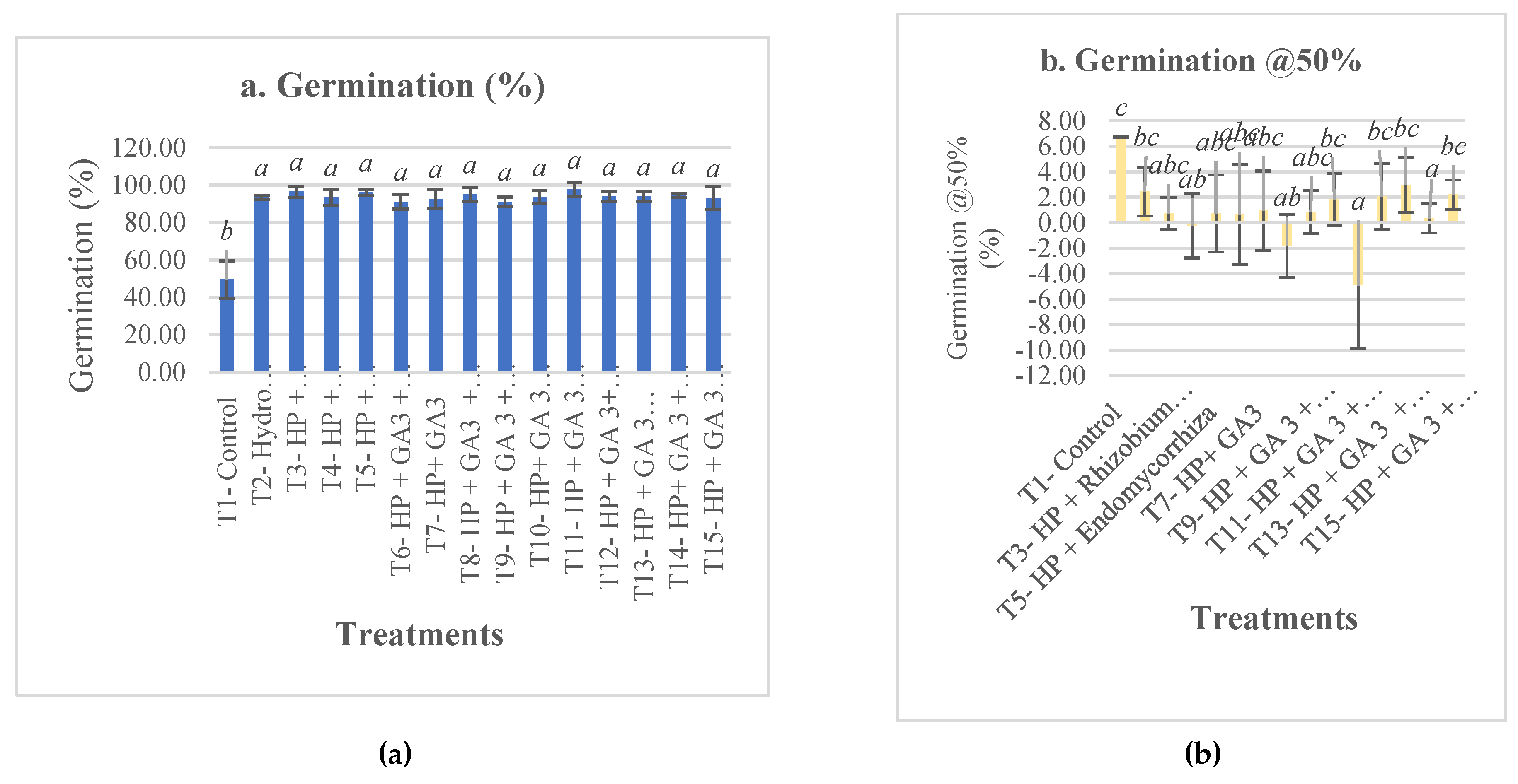
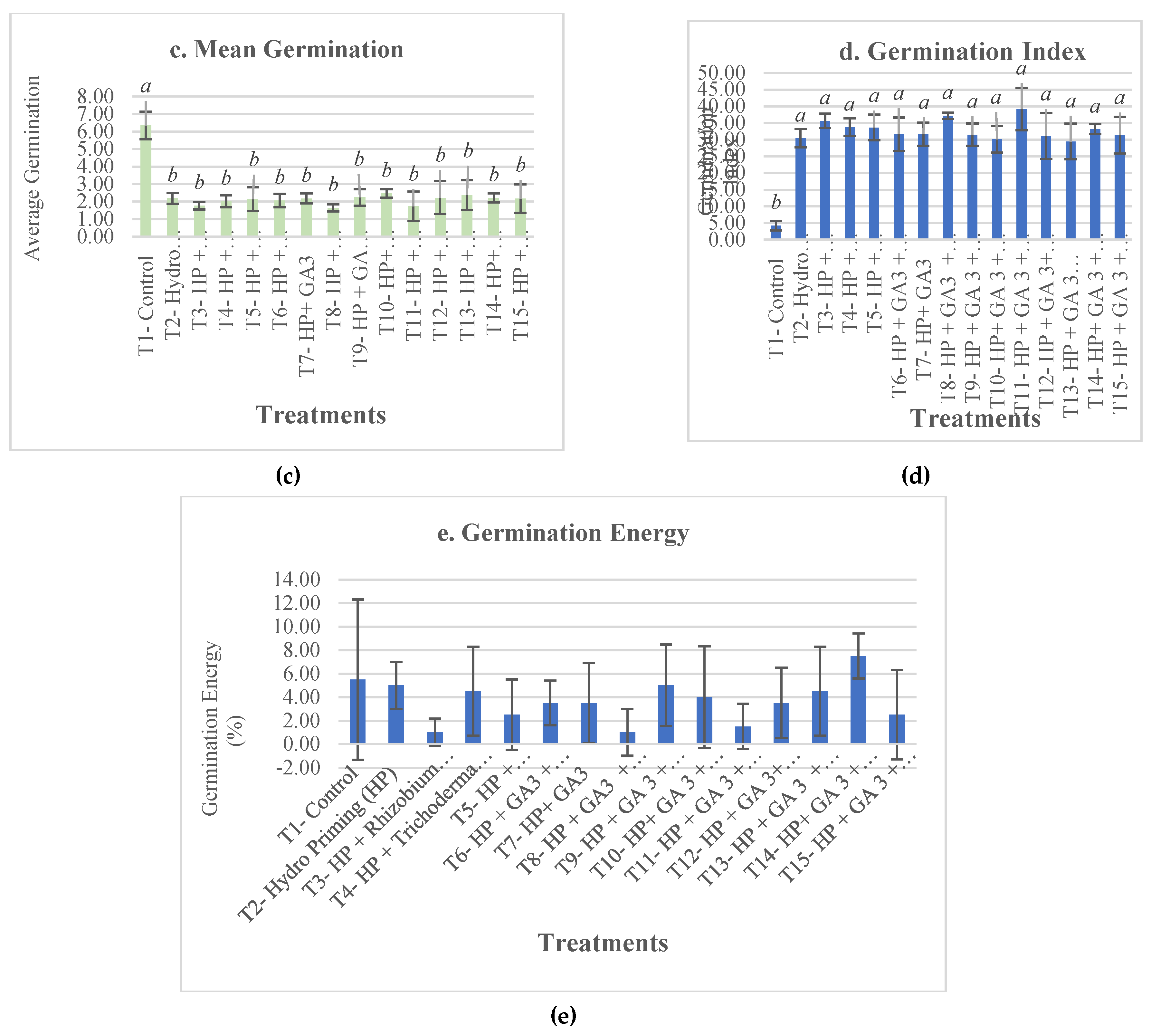
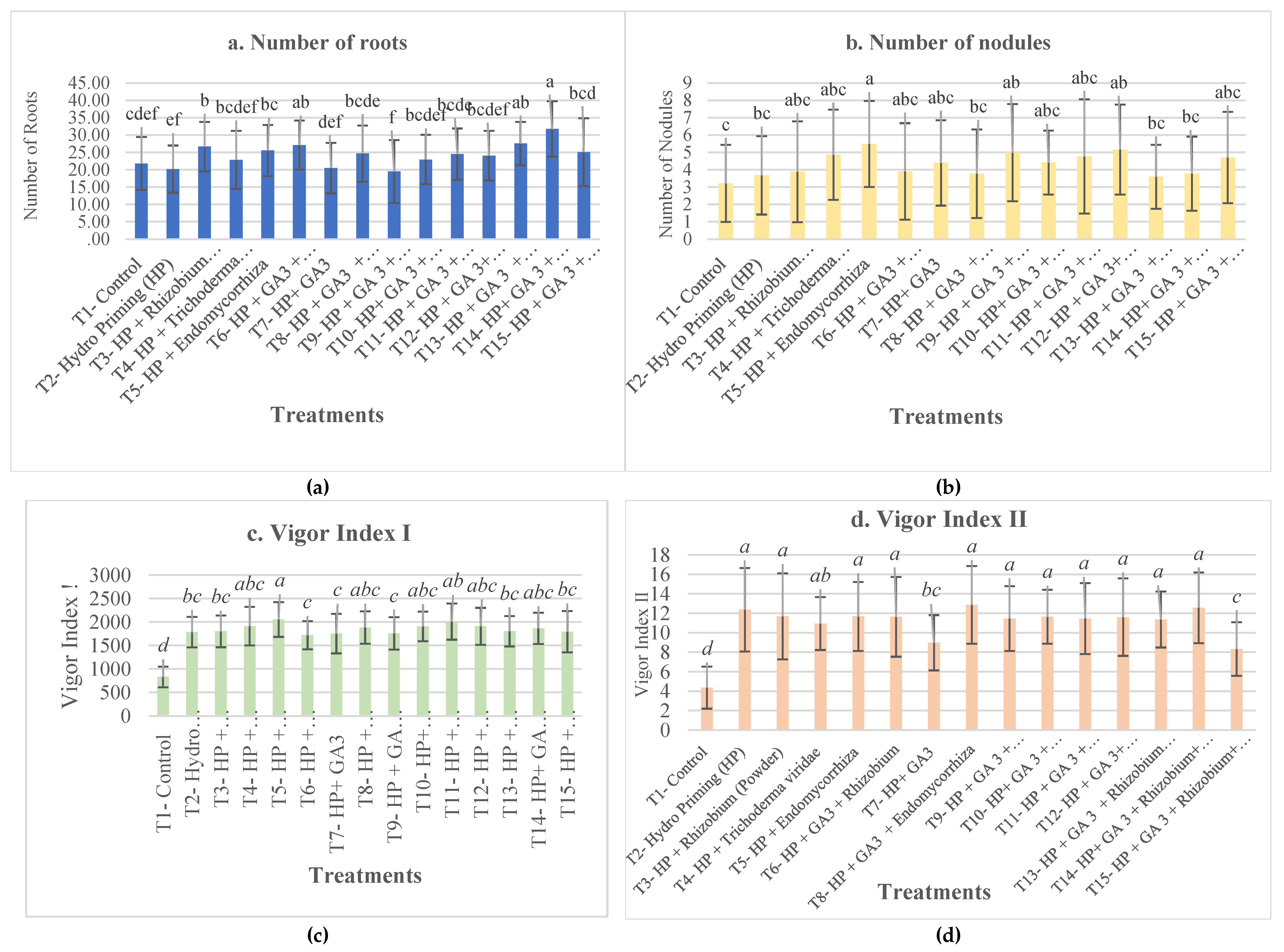
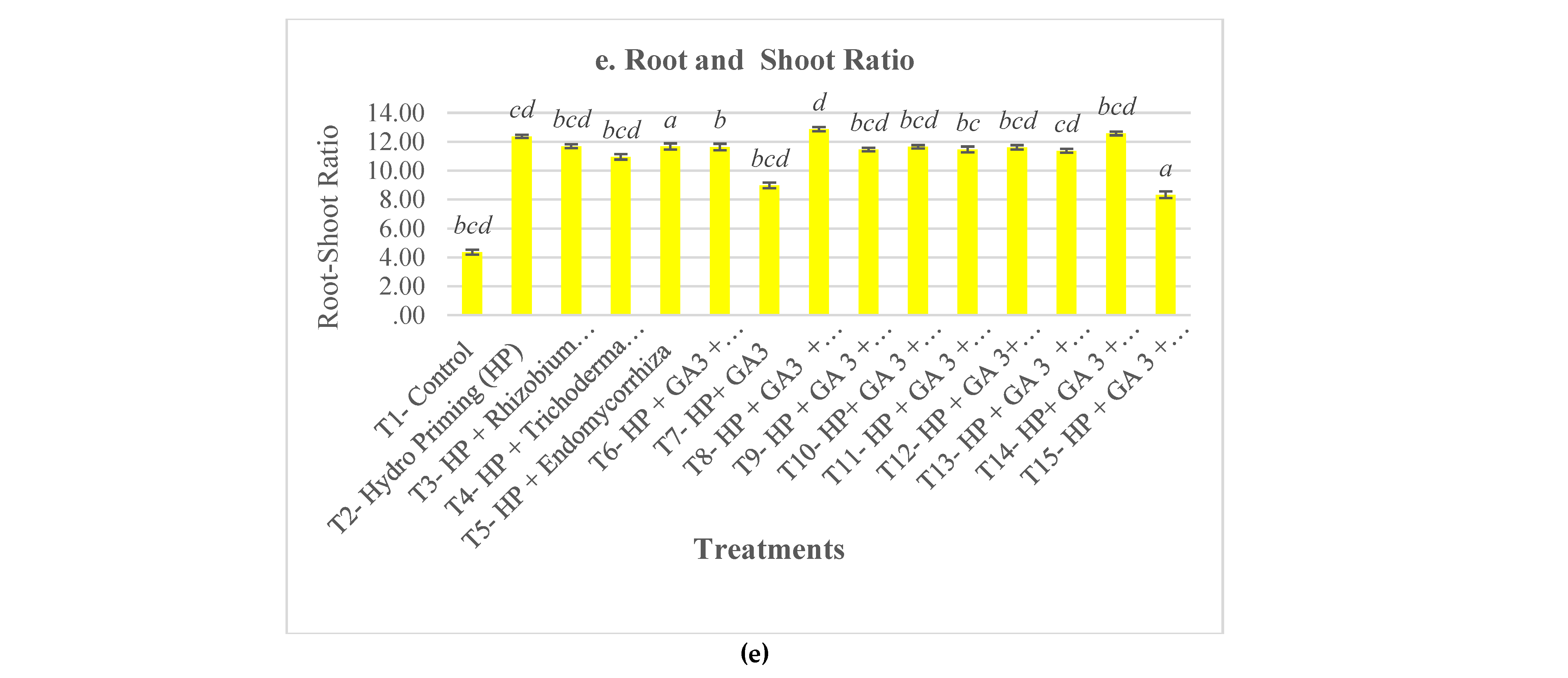
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nurheni Wijayanto, S.H.T. Evaluation of an Agroforestry System: The Growth of 14-Month-Old Solomon Sengon (Falcataria moluccana) and Rice (Oryza sativa). J. Sylva Lestari 2022, 10, 254–266. [Google Scholar] [CrossRef]
- Naufal Madani, H.; Wijayanto, N.; Hartoyo, A.; Solikhin, A. Slow-release Fertilizer Application on Silk (Falcataria moluccana Miq.) and Rice (Oryza sativa L.) Plant Growth and Yield in Agroforestry System. HAYATI J. Biosci. 2024, 31, 750–767. [Google Scholar] [CrossRef]
- Ambas, A.; Khatta, A.; Julius, K.; Kamu, A.; Chiang, L.; Terhem, R.; Awang Besar, N.; Hassan, A. Falcataria moluccana (Miq.) root distribution seedlings in response to nitrogen concentrations and tillage. Folia Oecol. 2024, 51, 47–55. [Google Scholar] [CrossRef]
- Paquit, J.C.L.-T.; Coritico, A. J. M.F. P. Hot water pre-treatment improves germination of Falcataria falcata. Bangladesh J. Bot. 2024, 53. [Google Scholar] [CrossRef]
- Yodfiatfinda, Y.; Fathiya Azzahra, P.; Rukavina, B. Risk Analysis of Sengon (Falcataria moluccana Miq.) Seedling Production Using Fault Tree Method. J. Ilmu Kehutan. 2023, 17. [Google Scholar] [CrossRef]
- Zhang, Y.; Du, Jianjun; Huang, Guanmin; Zhao, Yue; Man, Peng; Song, Anran; Zhao, Yanxin; Men, Qingmei; Wang, Chuanyu; Guo, Minkun; Guo, Xinyu; Zhao, Chunjiang. Integrating AI in seed science: Toward an intelligent design paradigm. Plant Commun. 2026, 7. [Google Scholar] [CrossRef]
- Baskorowati, L.; Wirabuana, P.Y.A.P.; Hendrati, R.L.; Nurtjahjaningsih, I.L.G.; Setiadi, D.; Mashudi, M.; Susanto, M.; Pudjiono, S.; Sumardi, S.; Santoso Sulistiadi, H.B.; et al. Early boosting of genetically improved Falcataria moluccana with mycorrhiza for better growth and disease tolerance. For. Sci. Technol. 2024, 20, 124–135. [Google Scholar] [CrossRef]
- Singh, P.; Vaishnav, A.; Liu, H.; Xiong, C.; Singh, H.B.; Singh, B.K. Seed biopriming for sustainable agriculture and ecosystem restoration. Microb. Biotechnol. 2023, 16, 2212–2222. [Google Scholar] [CrossRef]
- Miljaković, D.; Marinković, J.; Tamindžić, G.; Milošević, D.; Ignjatov, M.; Karačić, V.; Jakšić, S. Bio-Priming with Bacillus Isolates Suppresses Seed Infection and Improves the Germination of Garden Peas in the Presence of Fusarium Strains. J. Fungi 2024, 10, 358. [Google Scholar] [CrossRef]
- dos Reis, G.A.; Martínez-Burgos, W.J.; Pozzan, R.; Pastrana Puche, Y.; Ocán-Torres, D.; de Queiroz Fonseca Mota, P.; Rodrigues, C.; Lima Serra, J.; Scapini, T.; Karp, S.G.; et al. Comprehensive Review of Microbial Inoculants: Agricultural Applications, Technology Trends in Patents, and Regulatory Frameworks. Sustainability 2024, 16, 8720. [Google Scholar] [CrossRef]
- Chandrika, K.S.V.P.; Prasad, R.D.; Prasanna, S.L.; Shrey, B.; Kavya, M. Impact of biopolymer-based Trichoderma harzianum seed coating on disease incidence and yield in oilseed crops. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Adebisi, M. A.; Ajala, O.F.S.; Kehinde, M. O.; Daniel, T. O.; Ajani, I. O.O. O. Evaluation of Variations in Seed Vigour Characters of West African Rice (Oryza sativa L.) Genotypes Using Multivariate Technique. Am. J. Plant Sci. 2013, 4, 356–363. [Google Scholar] [CrossRef]
- Niang-Diop, F.; Sambou, B.; Lykke, A.M. Contraintes de régénération naturelle de Prosopis africana: facteurs affectant la germination des graines. Int. J. Biol. Chem. Sci. 2010, 4. [Google Scholar] [CrossRef]
- Nkemnkeng, F.J.M.; Tacham, G. A.W. N. Effects of seed pre-treatments on the germination and early growth of Echinops giganteus C.D Adam. J. Hortic. For. 2018, 10. [Google Scholar] [CrossRef]
- Salehzade, H.; Shishvan, M.I.; Ghiyasi, M.; Forouzin, F.; Siyahjani, A.A. Effect of seed priming on germination and seedling growth of wheat (Triticum aestivum L.). Res. J. Biol. Sci. 2009, 4, 629–631. [Google Scholar]
- Zhang, T.; Fan, S.; Xiang, Y.; Zhang, S.; Wang, J.; Sun, Q. Non-destructive analysis of germination percentage, germination energy and simple vigour index on wheat seeds during storage by Vis/NIR and SWIR hyperspectral imaging. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 239, 118488. [Google Scholar] [CrossRef]
- Association, I.S.T. International rules for seed testing. Seed Sci. Technol. 1999. [Google Scholar]
- Smith; Read, S.D.J. Mycorrhizal Symbiosisthird; 2008. [Google Scholar]
- Khan, M.; Hussain, F. Palatability and animal preferences of plants in Tehsil Takht-e-Nasrati, District Karak, Pakistan. Afr. J. Agric. Res. 2012, 7, 5858–5872. [Google Scholar] [CrossRef]
- Silva, J.M.; Pereira, C.V.; Mano, F.; Silva, E.; Castro, V.n.I.; Sá-Nogueira, I.; Reis, R.L.; Paiva, A.; Matias, A.A.; Duarte, A.R.C. Therapeutic role of deep eutectic solvents based on menthol and saturated fatty acids on wound healing. ACS Appl. Bio Mater. 2019, 2, 4346–4355. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, M.G.; Ascough, G.D.; Van Staden, J. Effects of Foliar Applications of Smoke-Water and a Smoke-isolated Butenolide on Seedling Growth of Okra and Tomato. HortScience 2007, 42, 179–182. [Google Scholar] [CrossRef]
- Kalsa, K.K.; Abebie, B. Influence of seed priming on seed germination and vigor traits of Vicia villosa ssp. dasycarpa (Ten.). Afr. J. Agric. Res. 2012, 7, 3202–3208. [Google Scholar] [CrossRef]
- Hameed, A.; Hussain, S.; Nisar, F.; Rasheed, A.; Shah, S.Z. Seed Priming as an Effective Technique for Enhancing Salinity Tolerance in Plants: Mechanistic Insights and Prospects for Saline Agriculture with a Special Emphasis on Halophytes. Seeds 2025, 4, 14. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seeds: ecology, biogeography, and, evolution of dormancy and germination; Academic press, 2000. [Google Scholar]
- Glick, B.R. Plant growth-promoting bacteria: mechanisms and applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef] [PubMed]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma species — opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef]
- Kumar, A.; van Duijnen, R.; Delory, B.M.; Reichel, R.; Brüggemann, N.; Temperton, V.M. Barley shoot biomass responds strongly to N:P stoichiometry and intraspecific competition, whereas roots only alter their foraging. Plant Soil. 2020, 453, 515–528. [Google Scholar] [CrossRef]
- Mašková, T.; Herben, T. Root: shoot ratio in developing seedlings: How seedlings change their allocation in response to seed mass and ambient nutrient supply. Ecol. Evol. 2018, 8, 7143–7150. [Google Scholar] [CrossRef]
- Hikosaka, K.T.; Kabeya, T.; Hirose, D.; Kamata, T.N. Biomass Allocation and Leaf Chemical Defence in Defoliated Seedlings of Quercus serrata with Respect to Carbon–Nitrogen Balance. Ann. Bot. 2005, 95, 1025–1032. [Google Scholar] [CrossRef]
- Qi, X.; Li, Z.; Akami, M.; Mansour, A.; Niu, C. Fermented crop straws by Trichoderma viride and Saccharomyces cerevisiae enhanced the bioconversion rate of Musca domestica (Diptera: Muscidae). Environ. Sci. Pollut. Res. 2019, 26, 29388–29396. [Google Scholar] [CrossRef]
- Wang, X.; Pan, Q.; Chen, F.; Yan, X.; Liao, H. Effects of co-inoculation with arbuscular mycorrhizal fungi and rhizobia on soybean growth as related to root architecture and availability of N and P. Mycorrhiza 2011, 21, 173–181. [Google Scholar] [CrossRef]
- Poorter, H.; Nagel, O. The role of biomass allocation in the growth response of plants to different levels of light, CO 2, nutrients and water: a quantitative review. Aust. J. Plant Physiol. 2000, 27, 595–607. [Google Scholar] [CrossRef]
- Janah, I.; Elhasnaoui, A.; Laouane, R. B.; Ait-El-Mokhtar, M.; Anli, M. Exploring seed priming as a strategy for enhancing abiotic stress tolerance in cereal crops. Stresses 2025, 5, 39. [Google Scholar] [CrossRef]
- Magsayo, B.M.T.; Aggangan, Nelly S; Gilbero, Dennis M; Amparado, Ruben F., Jr. Evaluating microbial biofertilizers for root colonization potential in narra (Pterocarpus indicus Willd.) and their efficacy in heavy metal remediation. Forests 2024, 15, 180. [Google Scholar] [CrossRef]
- de Dorlodot, S.; Forster, B.; Pagès, L.; Price, A.; Tuberosa, R.; Draye, X. Root system architecture: opportunities and constraints for genetic improvement of crops. Trends Plant Sci. 2007, 12, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Comas, L.; Becker, S.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4–2013. [Google Scholar] [CrossRef]
- Wang, J.X.; Zhenzhu. Plant response to and recovery from drought. Curr. Biol. 2026, 36, R343–R362. [Google Scholar] [CrossRef]
- Xue, D.Y.; Chen, W. F.; Yang, G. P.; Li, Y. G.; Zhang, J. J. Screening and application of highly efficient rhizobia for leguminous green manure Astragalus sinicus in lyophilized inoculants and seed coating. Plants 2025, 14, 2431. [Google Scholar] [CrossRef]
- Yadav, A.; Yadav, S.; Choudhary, R.; M K, S.; Anumantharaj, A.; Bharadwaj, R.; Yadav, R.; Yashpal; Saini, N.; Adhikari, S.; et al. Insights from multivariate analysis of seed vigor traits in rapeseed and mustard (Brassica spp.) genotypes. Turk. J. Agric. For. 2025, 49, 651–660. [Google Scholar] [CrossRef]
- Maji, A.S.A.A. Application of principal component analysis for rice germplasm characterization and evaluation. J. Plant Breed. Crop Sci. 2012, 4, 87–93. [Google Scholar] [CrossRef]
| Treatment | Seed Germination |
Seedling Growth Performance |
||||||||
| Germination (%) | Germination @50% | Mean Germination | Germination Index | Germination Energy | no. of roots | No. of nodules | Root: Shoot Ratio | Vigor Index I | Vigor Index II | |
| T1- Control | 49.5±9.98b | 6.73±0.05c | 6.34±0.79a | 4.23±1.42b | 5.5±6.81 | 21.83±7.63cdef | 3.22±2.22c | 0.46±0.17bcd | 832.48±219.09d | 4.36±2.16d |
| T2- Hydro Priming (HP) | 93.5±1a | 2.43±1.91bc | 2.19±0.31b | 30.43±2.77a | 5±2 | 20.2±6.81ef | 3.68±2.26bc | 0.37±0.11cd | 1784.7±325.32bc | 12.37±4.29a |
| T3- HP + Rhizobium sp | 96.5±3a | 0.73±1.23abc | 1.77±0.22b | 35.61±2.15a | 1±1.15 | 26.68±7.16b | 3.89±2.91abc | 0.41±0.13bcd | 1802.01±337.52bc | 11.69±4.42a |
| T4- HP + Trichoderma sp | 93.5±4.43a | -0.22±2.54ab | 2.01±0.34b | 33.73±2.6a | 4.5±3.79 | 22.88±8.36bcdef | 4.87±2.6abc | 0.44±0.18bcd | 1913.38±409.32abc | 10.95±2.72ab |
| T5- HP + Endomycorrhiza sp | 96±1.63a | 0.73±3.03abc | 2.13±0.68b | 33.62±3.83a | 2.5±3 | 25.57±7.37bc | 5.49±2.48a | 0.59±0.21a | 2055.57±370.84a | 11.67±3.55a |
| T6- HP + GA3 + Rhizobium sp | 91±3.83a | 0.66±3.94abc | 2.06±0.38b | 31.61±4.99a | 3.5±1.91 | 27.13±7.08ab | 3.91±2.78abc | 0.49±0.22b | 1720.86±299.59c | 11.64±4.1a |
| T7- HP+ GA3 | 92.5±5a | 0.94±3.12abc | 2.18±0.28b | 31.61±3.43a | 3.5±3.42 | 20.5±7.27def | 4.4±2.46abc | 0.47±0.19bcd | 1754.59±421.59c | 8.98±2.84bc |
| T8- HP + GA3 + Endomycorrhiza | 95±3.83a | -1.81±2.48ab | 1.64±0.2b | 37.15±0.97a | 1±2 | 24.69±8.12bcde | 3.78±2.55bc | 0.37±0.14d | 1883.81±343.54abc | 12.87±4a |
| T9- HP + GA 3 + Endomycorrhiza + Trichoderma sp | 91±2.58a | 0.84±1.67abc | 2.24±0.47b | 31.5±3.37a | 5±3.46 | 19.5±9.07f | 4.99±2.8ab | 0.39±0.12bcd | 1758.6±346.25c | 11.46±3.32a |
| T10- HP+ GA 3 + Endomycorrhiza+ Trichoderma sp + Rhizobium sp | 93.5±3.42a | 1.84±2.05bc | 2.46±0.24b | 30.1±4.02a | 4±4.32 | 22.96±7.13bcdef | 4.42±1.84abc | 0.43±0.11bcd | 1907.27±316.46abc | 11.65±2.78a |
| T11- HP + GA 3 + Endomycorrhiza sp+ Trichoderma sp + Rhizobium sp + Polymer coat + Insecticide | 97.5±3.79a | -4.91±4.94a | 1.73±0.84b | 39.14±6.39a | 1.5±1.91 | 24.51±7.41bcde | 4.77±3.3abc | 0.48±0.2bc | 2011.09±385.59ab | 11.46±3.65a |
| T12- HP + GA 3+ Endomycorrhiza + Trichoderma sp + Rhizobium + Polymer coat + Nutrients | 94±2.83a | 2.06±2.6bc | 2.22±0.94b | 31.09±6.91a | 3.5±3 | 24.08±7.14bcdef | 5.17±2.59ab | 0.43±0.15bcd | 1909.51±393.55abc | 11.61±3.98a |
| T13- HP + GA 3 + Rhizobium sp+ Polymer coat + Nutrients + Fungicide | 94±2.83a | 2.96±2.14bc | 2.37±0.85b | 29.45±5.36a | 4.5±3.79 | 27.58±6.24ab | 3.6±1.85bc | 0.38±0.14cd | 1805.77±323.68bc | 11.37±2.88a |
| T14- HP+ GA 3 + Rhizobium sp+ Polymer coat + Fungicide | 94.5±1a | 0.35±1.16abc | 2.21±0.26b | 33.17±1.43a | 7.5±1.91 | 31.75±8.02a | 3.78±2.14bc | 0.41±0.13bcd | 1866.14±331.61abc | 12.57±3.63a |
| T15- HP + GA 3 + Rhizobium sp+ Polymer coat + Fungicide + Insecticide+ Nutrients | 93±6.22a | 2.21±1.15bc | 2.17±0.81b | 31.33±5.47a | 2.5±3.79 | 25.09±9.76bcd | 4.71±2.63abc | 0.6±0.23a | 1792.73±440.09bc | 8.33±2.75c |
| p-value | ** | ** | ** | ** | ns | ** | ** | ** | ** | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




