Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
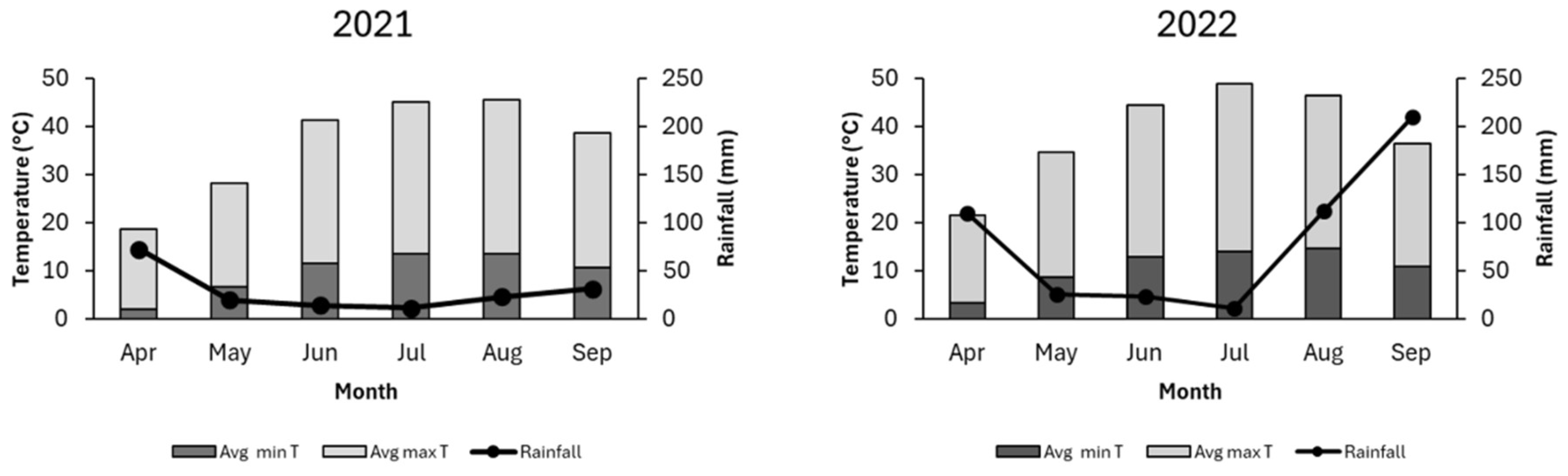
2.1. Experimental Site and Plant Growth Conditions
2.2. Micronized Biochar and Experimental Trial
2.3. Analysis of Soil Chemical Properties
2.4. Soil Enzymatic Activities
2.5. Plant Biomass and Biometric Parameters
2.6. Leaf Gas Exchange Parameters
2.7. Leaf Chlorophyll (Chl) and Flavonol (Flav) Content
2.8. Concentration of N, P, K, Ca and Mg in Leaves
2.9. Statistical Analysis
3. Results
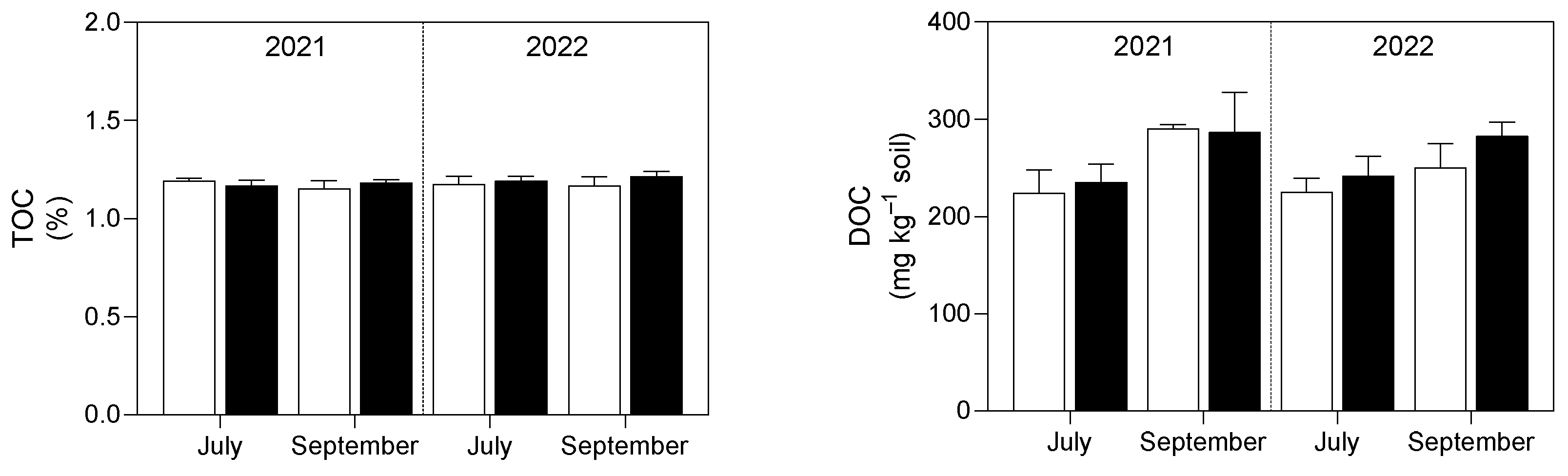
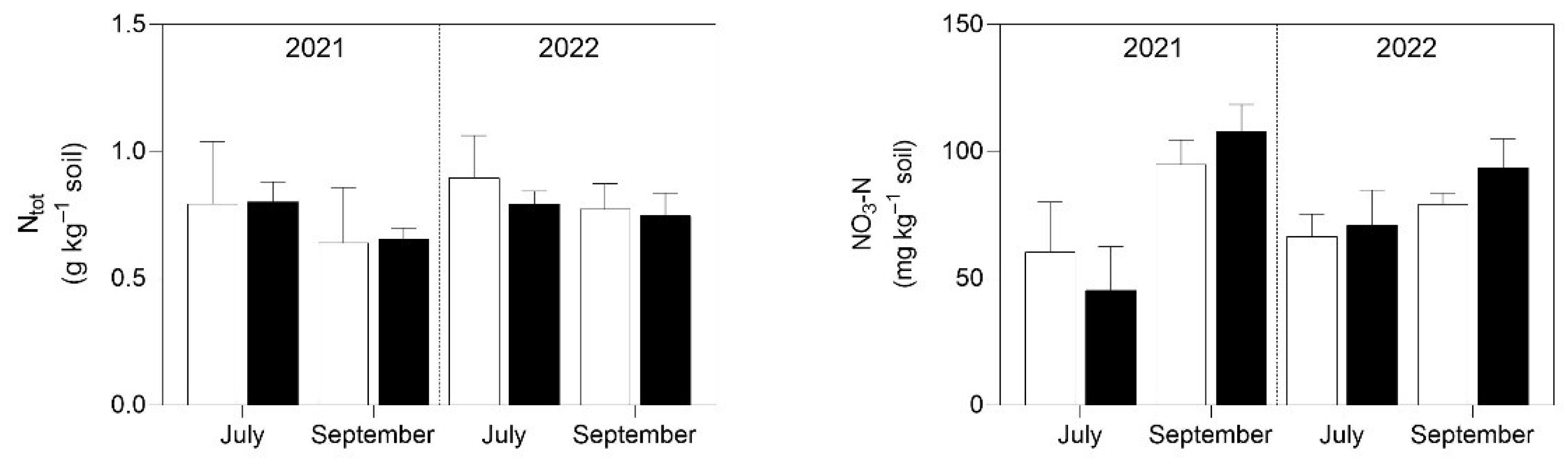
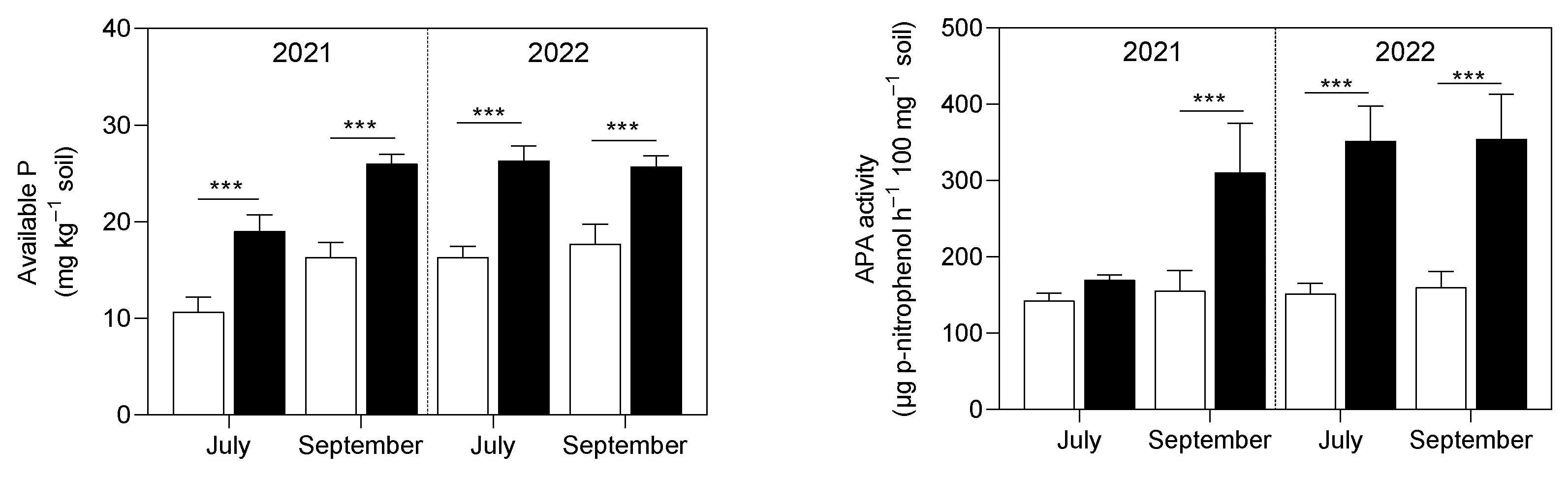
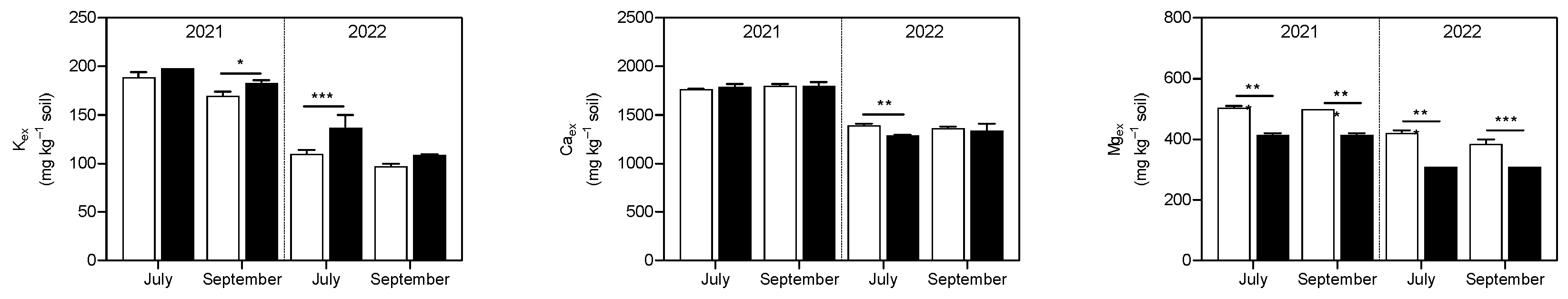
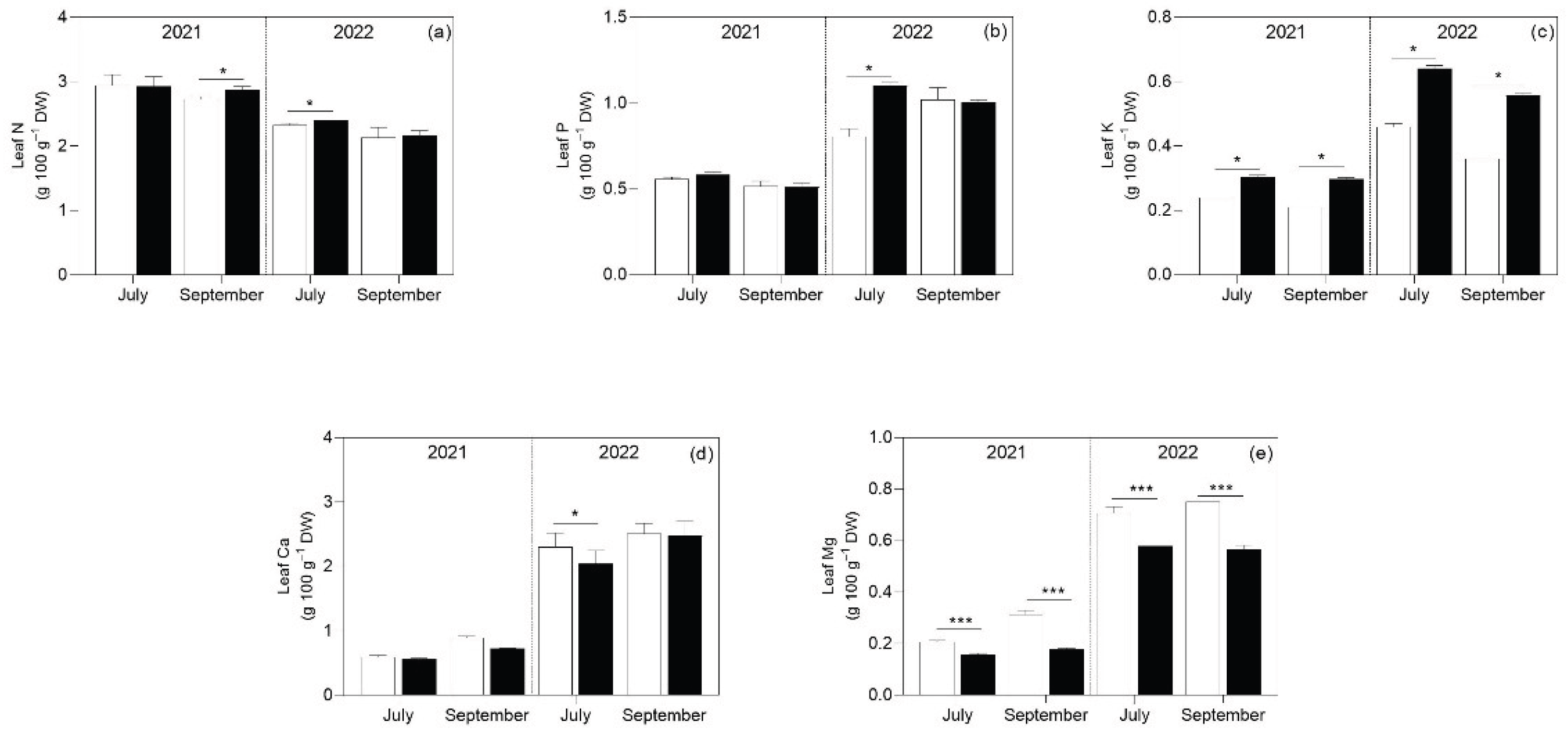
3.1. Soil Analysis
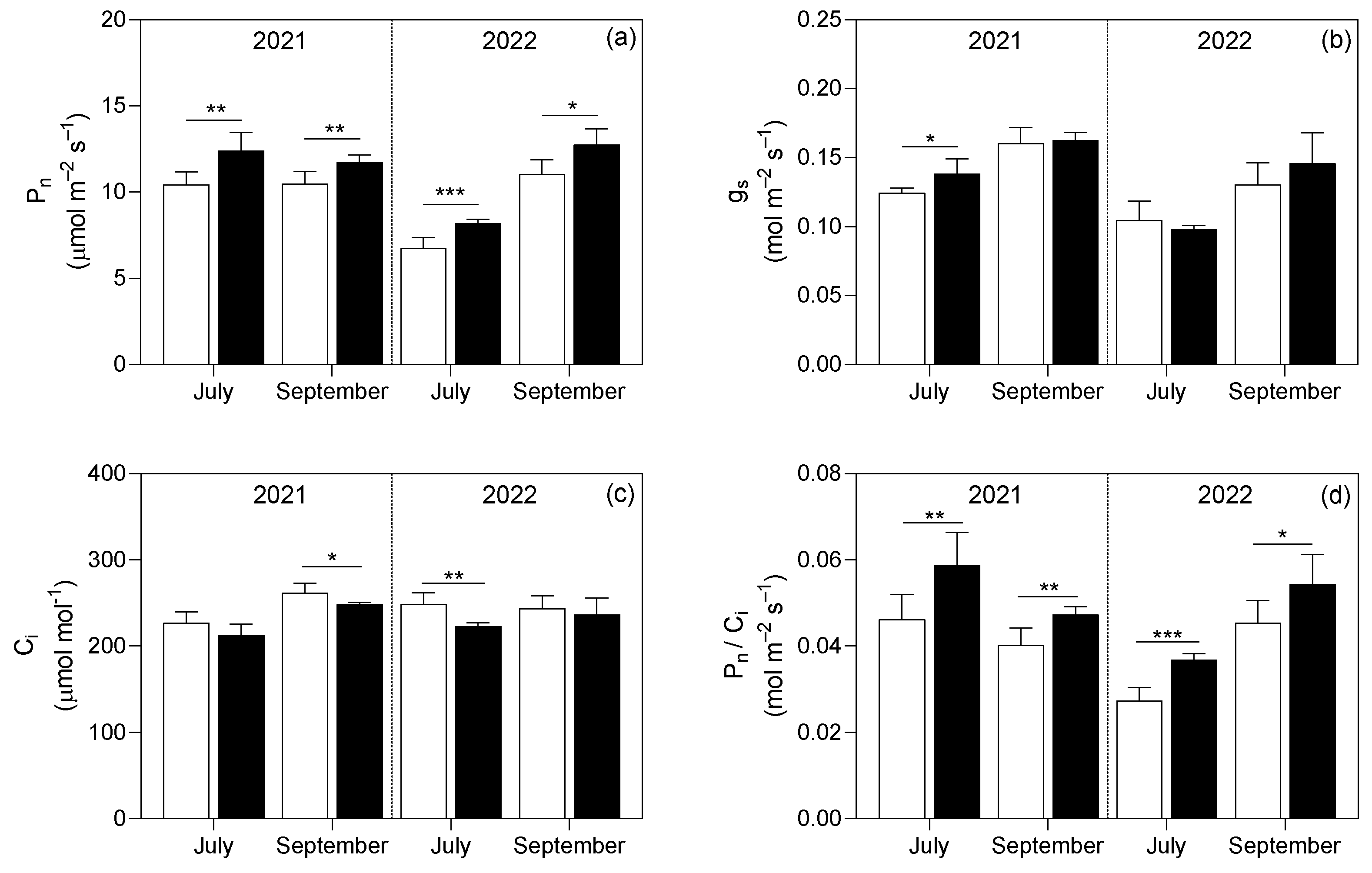
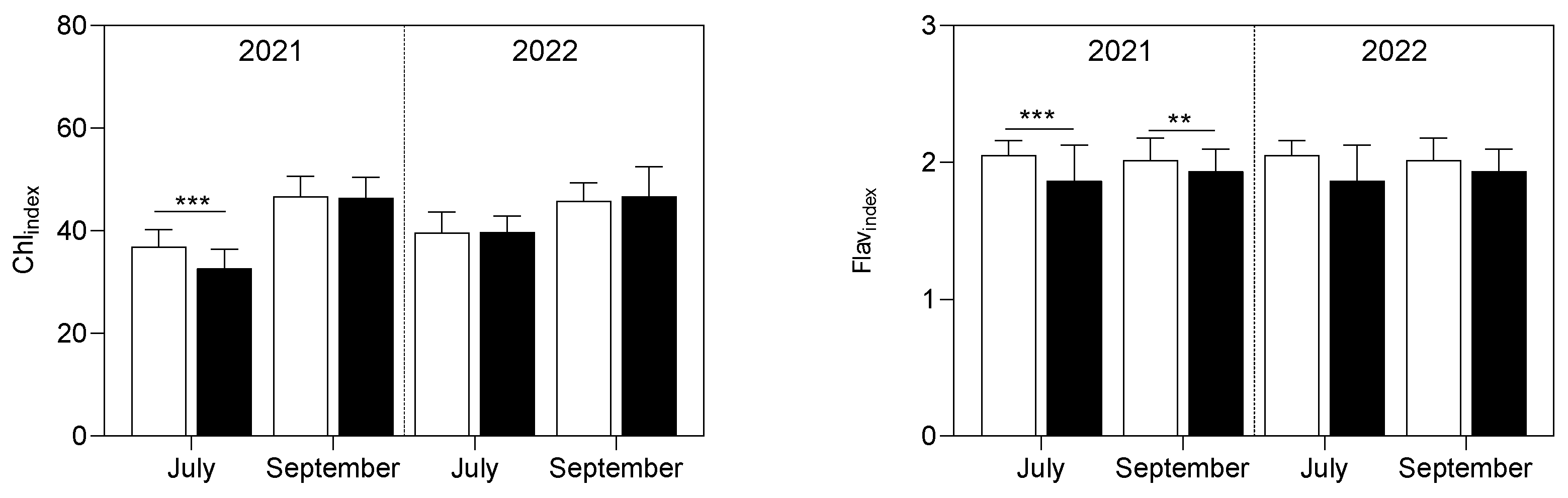
3.2. Plant Analyses
4. Discussion
4.1. Soil P and K Availability and Soil Enzymatic Activity Were Enhanced by the Application of Micronized Biochar
4.2. Leaf Biomass and Net Photosynthetic Process Were Improved by Continuous B Application in Both Experimental Years
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| APA | Alkaline Phosphatase Activities |
| available P | Available Phosphorous |
| B | Micronized Biochar |
| Caex | Exchangeable Ca |
| CEC | Cation Exchange Capacity |
| Chlindex | Chlorophyll Index |
| Ci | Intercellular CO2 Concentration |
| CTR | Control |
| DHA | Dehydrogenase Activities |
| DOC | Dissolved Organic Carbon |
| EC | Electric Conductivity |
| Flavindex | Flavonol Index |
| gs | Stomatal Conductance to Water Vapor |
| Kex | Exchangeable K |
| L | Length |
| Mgex | Exchangeable Mg |
| MPI | Metabolic Potential Index |
| NO3--N | Nitrates |
| Ntot | Total Nitrogen |
| Pn | Net Photosynthetic |
| Pn/Ci | Apparent Carboxylation Efficiency |
| SOM | Soil Organic Matter |
| TOC | Total Organic Carbon |
| W | Width |
References
- Lazcano, C.; Decock, C.; Wilson, S.G. Defining and Managing for Healthy Vineyard Soils, Intersections With the Concept of Terroir. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- García-Jaramillo, M.; Meyer, K.M.; Phillips, C.L.; Acosta-Martínez, V.; Osborne, J.; Levin, A.D.; Trippe, K.M. Biochar Addition to Vineyard Soils: Effects on Soil Functions, Grape Yield and Wine Quality. Biochar 2021, 3, 565–577. [Google Scholar] [CrossRef]
- Giagnoni, L.; Maienza, A.; Baronti, S.; Vaccari, F.P.; Genesio, L.; Taiti, C.; Martellini, T.; Scodellini, R.; Cincinelli, A.; Costa, C.; et al. Long-Term Soil Biological Fertility, Volatile Organic Compounds and Chemical Properties in a Vineyard Soil after Biochar Amendment. Geoderma 2019, 344, 127–136. [Google Scholar] [CrossRef]
- Homet, P.; Gallardo-Reina, M.Á.; Aguiar, J.F.; Liberal, I.M.; Casimiro-Soriguer, R.; Ochoa-Hueso, R. Viticulture and the European Union’s Common Agricultural Policy (CAP): Historical Overview, Current Situation and Future Perspective. J. Sustain. Agric. Environ. 2024, 3, e12099. [Google Scholar] [CrossRef]
- Borsato, E.; Zucchinelli, M.; D’Ammaro, D.; Giubilato, E.; Zabeo, A.; Criscione, P.; Pizzol, L.; Cohen, Y.; Tarolli, P.; Lamastra, L.; et al. Use of Multiple Indicators to Compare Sustainability Performance of Organic vs Conventional Vineyard Management. Sci. Total. Environ. 2020, 711, 135081. [Google Scholar] [CrossRef]
- Hendgen, M.; Döring, J.; Stöhrer, V.; Schulze, F.; Lehnart, R.; Kauer, R. Spatial Differentiation of Physical and Chemical Soil Parameters under Integrated, Organic, and Biodynamic Viticulture. Plants 2020, 9, 1361. [Google Scholar] [CrossRef] [PubMed]
- Baronti, S.; Vaccari, F.P.; Miglietta, F.; Calzolari, C.; Lugato, E.; Orlandini, S.; Pini, R.; Zulian, C.; Genesio, L. Impact of Biochar Application on Plant Water Relations in Vitis Vinifera (L.). Eur. J. Agron. 2014, 53, 38–44. [Google Scholar] [CrossRef]
- Idbella, M.; Baronti, S.; Vaccari, F.P.; Abd-ElGawad, A.M.; Bonanomi, G. Long-Term Application of Biochar Mitigates Negative Plant–Soil Feedback by Shaping Arbuscular Mycorrhizal Fungi and Fungal Pathogens. Microorganisms 2024, 12, 810. [Google Scholar] [CrossRef]
- Schmidt, H.-P.; Kammann, C.; Niggli, C.; Evangelou, M.W.H.; Mackie, K.A.; Abiven, S. Biochar and Biochar-Compost as Soil Amendments to a Vineyard Soil: Influences on Plant Growth, Nutrient Uptake, Plant Health and Grape Quality. Agric. Ecosyst. Environ. 2014, 191, 117–123. [Google Scholar] [CrossRef]
- Gao, Y.; Shao, G.; Yang, Z.; Zhang, K.; Lu, J.; Wang, Z.; Wu, S.; Xu, D. Influences of Soil and Biochar Properties and Amount of Biochar and Fertilizer on the Performance of Biochar in Improving Plant Photosynthetic Rate: A Meta-Analysis. Eur. J. Agron. 2021, 130, 126345. [Google Scholar] [CrossRef]
- Baronti, S.; Magno, R.; Maienza, A.; Montagnoli, A.; Ungaro, F.; Vaccari, F.P. Long Term Effect of Biochar on Soil Plant Water Relation and Fine Roots: Results after 10 Years of Vineyard Experiment. Sci. Total. Environ. 2022, 851, 158225. [Google Scholar] [CrossRef]
- He, Y.; Yao, Y.; Ji, Y.; Deng, J.; Zhou, G.; Liu, R.; Shao, J.; Zhou, L.; Li, N.; Zhou, X.; et al. Biochar Amendment Boosts Photosynthesis and Biomass in C3 but Not C4 Plants: A Global Synthesis. GCB Bioenergy 2020, 12, 605–617. [Google Scholar] [CrossRef]
- Lo Piccolo, E.; Becagli, M.; Lauria, G.; Cantini, V.; Ceccanti, C.; Cardelli, R.; Massai, R.; Remorini, D.; Guidi, L.; Landi, M. Biochar as a Soil Amendment in the Tree Establishment Phase: What Are the Consequences for Tree Physiology, Soil Quality and Carbon Sequestration? Sci. Total. Environ. 2022, 844, 157175. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, J.; Wang, Y.; Yang, Q.; Chen, T.; Chen, Y.; Chi, D.; Xia, G.; Siddique, K.H.M.; Wang, T. Photosynthesis, Chlorophyll Fluorescence, and Yield of Peanut in Response to Biochar Application. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Violante, P. Metodi di analisi chimica del suolo; Franco Angeli, 2000; ISBN 978-88-464-2240-8. [Google Scholar]
- Tabatabai, M. a. Soil Enzymes. In Methods of Soil Analysis; John Wiley & Sons, Ltd, 1994; pp. 775–833. ISBN 978-0-89118-865-0. [Google Scholar]
- Riffaldi, R.; Saviozzi, A.; Levi-Minzi, R.; Cardelli, R. Biochemical Properties of a Mediterranean Soil as Affected by Long-Term Crop Management Systems. Soil. Tillage Res. 2002, 67, 109–114. [Google Scholar] [CrossRef]
- Eivazi, F.; Tabatabai, M.A. Phosphatases in Soils. Soil. Biol. Biochem. 1977, 9, 167–172. [Google Scholar] [CrossRef]
- Buttaro, D.; Rouphael, Y.; Rivera, C.M.; Colla, G.; Gonnella, M. Simple and Accurate Allometric Model for Leaf Area Estimation in Vitis Vinifera L. Genotypes. Photosynthetica 2015, 53, 342–348. [Google Scholar] [CrossRef]
- Benini, O.; Saba, A.; Ferretti, V.; Gianfaldoni, D.; Kalantar-Zadeh, K.; Cupisti, A. Development and Analytical Evaluation of a Spectrophotometric Procedure for the Quantification of Different Types of Phosphorus in Meat Products. J. Agric. Food Chem. 2014, 62, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Hagemann, N.; Joseph, S.; Schmidt, H.-P.; Kammann, C.I.; Harter, J.; Borch, T.; Young, R.B.; Varga, K.; Taherymoosavi, S.; Elliott, K.W.; et al. Organic Coating on Biochar Explains Its Nutrient Retention and Stimulation of Soil Fertility. Nat. Commun. 2017, 8, 1089. [Google Scholar] [CrossRef]
- Paustian, K.; Lehmann, J.; Ogle, S.; Reay, D.; Robertson, G.P.; Smith, P. Climate-Smart Soils. Nature 2016, 532, 49–57. [Google Scholar] [CrossRef]
- Becagli, M.; Santin, M.; Cardelli, R. Co-Application of Wood Distillate and Biochar Improves Soil Quality and Plant Growth in Basil (Ocimum Basilicum). J. Plant Nutr. Soil. Sci. 2022, 185, 120–131. [Google Scholar] [CrossRef]
- Schulz, H.; Glaser, B. Effects of Biochar Compared to Organic and Inorganic Fertilizers on Soil Quality and Plant Growth in a Greenhouse Experiment. J. Plant Nutr. Soil. Sci. 2012, 175, 410–422. [Google Scholar] [CrossRef]
- Hailegnaw, N.S.; Mercl, F.; Pračke, K.; Száková, J.; Tlustoš, P. Mutual Relationships of Biochar and Soil pH, CEC, and Exchangeable Base Cations in a Model Laboratory Experiment. J. Soils Sediments 2019, 19, 2405–2416. [Google Scholar] [CrossRef]
- Koga, N.; Shimoda, S.; Iwata, Y. Biochar Impacts on Crop Productivity and Greenhouse Gas Emissions from an Andosol. J. Environ. Qual. 2017, 46, 27–35. [Google Scholar] [CrossRef]
- Pokharel, P.; Ma, Z.; Chang, S.X. Biochar Increases Soil Microbial Biomass with Changes in Extra- and Intracellular Enzyme Activities: A Global Meta-Analysis. Biochar 2020, 2, 65–79. [Google Scholar] [CrossRef]
- Ouyang, L.; Tang, Q.; Yu, L.; Zhang, R. Effects of Amendment of Different Biochars on Soil Enzyme Activities Related to Carbon Mineralisation. Soil. Res. 2014, 52, 706–716. [Google Scholar] [CrossRef]
- Carril, P.; Becagli, M.; Celletti, S.; Fedeli, R.; Loppi, S.; Cardelli, R. Biofertilization with Liquid Vermicompost-Activated Biochar Enhances Microbial Activity and Soil Properties. Soil. Syst. 2024, 8, 54. [Google Scholar] [CrossRef]
- Sorrenti, G.; Ventura, M.; Toselli, M. Effect of Biochar on Nutrient Retention and Nectarine Tree Performance: A Three-Year Field Trial. J. Plant Nutr. Soil. Sci. 2016, 179, 336–346. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, Y.; Liu, B.; Amonette, J.E.; Lin, Z.; Liu, G.; Ambus, P.; Xie, Z. How Does Biochar Influence Soil N Cycle? A Meta-Analysis. Plant Soil. 2018, 426, 211–225. [Google Scholar] [CrossRef]
- Chen, X.; Yang, S.-H.; Jiang, Z.-W.; Ding, J.; Sun, X. Biochar as a Tool to Reduce Environmental Impacts of Nitrogen Loss in Water-Saving Irrigation Paddy Field. J. Clean. Prod. 2021, 290, 125811. [Google Scholar] [CrossRef]
- Xiu, L.; Sun, Y.; Han, X. Effects of Biochar Amendment on Potassium Supply Capacity and Potassium Accumulation in Soybean Across Diverse Soils. Plants 2025, 14, 1959. [Google Scholar] [CrossRef]
- Butnan, S.; Deenik, J.L.; Toomsan, B.; Vityakon, P. Biochar Properties Affecting Carbon Stability in Soils Contrasting in Texture and Mineralogy. Agr. Nat. Resour. 2017, 51, 492–498. [Google Scholar] [CrossRef]
- Lützow, M. v.; Kögel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of Organic Matter in Temperate Soils: Mechanisms and Their Relevance under Different Soil Conditions – a Review. Eur. J. Soil. Sci. 2006, 57, 426–445. [Google Scholar] [CrossRef]
- Sadowska, U.; Domagała-Świątkiewicz, I.; Żabiński, A. Biochar and Its Effects on Plant–Soil Macronutrient Cycling during a Three-Year Field Trial on Sandy Soil with Peppermint (Mentha Piperita L.). Part I: Yield and Macro Element Content in Soil and Plant Biomass. Agronomy 2020, 10, 1950. [Google Scholar] [CrossRef]
- Liao, J.X.; So, P.S.; Bordoloi, S.; Li, D.N.; Yuan, H.R.; Chen, Y.; Xin, L.Q. Plant Performance and Soil–Plant Carbon Relationship Response to Different Biochar Types. Biochar 2024, 6, 75. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, Q.; Chen, Y.; Lu, H. Effects of Biochar on Plant Growth and Hydro-Chemical Properties of Recycled Concrete Aggregate. Sci. Total. Environ. 2023, 882, 163557. [Google Scholar] [CrossRef]
- Feng, W.; Yang, F.; Cen, R.; Liu, J.; Qu, Z.; Miao, Q.; Chen, H. Effects of Straw Biochar Application on Soil Temperature, Available Nitrogen and Growth of Corn. J. Environ. Manag. 2021, 277, 111331. [Google Scholar] [CrossRef]
- Genesio, L.; Miglietta, F.; Baronti, S.; Vaccari, F.P. Biochar Increases Vineyard Productivity without Affecting Grape Quality: Results from a Four Years Field Experiment in Tuscany. Agric. Ecosyst. Environ. 2015, 201, 20–25. [Google Scholar] [CrossRef]
- Jaiswal, A.K.; Alkan, N.; Elad, Y.; Sela, N.; Philosoph, A.M.; Graber, E.R.; Frenkel, O. Molecular Insights into Biochar-Mediated Plant Growth Promotion and Systemic Resistance in Tomato against Fusarium Crown and Root Rot Disease. Sci. Rep. 2020, 10, 13934. [Google Scholar] [CrossRef] [PubMed]
- Tanazawa, Y.; Tomotsune, M.; Suzuki, T.; Koizumi, H.; Yoshitake, S. Photosynthetic Response of Young Oaks to Biochar Amendment in Field Conditions over 3 Years. J. For. Res. 2021, 26, 116–126. [Google Scholar] [CrossRef]
- Wang, Y.; Pan, F.; Wang, G.; Zhang, G.; Wang, Y.; Chen, X.; Mao, Z. Effects of Biochar on Photosynthesis and Antioxidative System of Malus hupehensis Rehd. Seedlings under Replant Conditions. Sci. Hortic. 2014, 175, 9–15. [Google Scholar] [CrossRef]
- Zhang, K.; Han, X.; Fu, Y.; Khan, Z.; Zhang, B.; Bi, J.; Hu, L.; Luo, L. Biochar Coating Promoted Rice Growth under Drought Stress through Modulating Photosynthetic Apparatus, Chloroplast Ultrastructure, Stomatal Traits and ROS Homeostasis. Plant Physiol. Biochem. 2024, 216, 109145. [Google Scholar] [CrossRef] [PubMed]
- Bozzolo, A.; Pizzeghello, D.; Cardinali, A.; Francioso, O.; Nardi, S.; Bozzolo, A.; Pizzeghello, D.; Cardinali, A.; Francioso, O.; Nardi, S. Effects of Moderate and High Rates of Biochar and Compost on Grapevine Growth in a Greenhouse Experiment. AIMS agric. Food 2017, 2, 113–128. [Google Scholar] [CrossRef]
- Xiong, D.; Flexas, J. Leaf Anatomical Characteristics Are Less Important than Leaf Biochemical Properties in Determining Photosynthesis Responses to Nitrogen Top-Dressing. J. Exp. Bot. 2021, 72, 5709–5720. [Google Scholar] [CrossRef]
- Jindo, K.; Sonoki, T.; Matsumoto, K.; Canellas, L.; Roig, A.; Sanchez-Monedero, M.A. Influence of Biochar Addition on the Humic Substances of Composting Manures. Waste Manag. 2016, 49, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Riaz, M.; Liu, B.; Xia, H.; El-desouki, Z.; Jiang, C. Two-Year Study of Biochar: Achieving Excellent Capability of Potassium Supply via Alter Clay Mineral Composition and Potassium-Dissolving Bacteria Activity. Sci. Total. Environ. 2020, 717, 137286. [Google Scholar] [CrossRef]
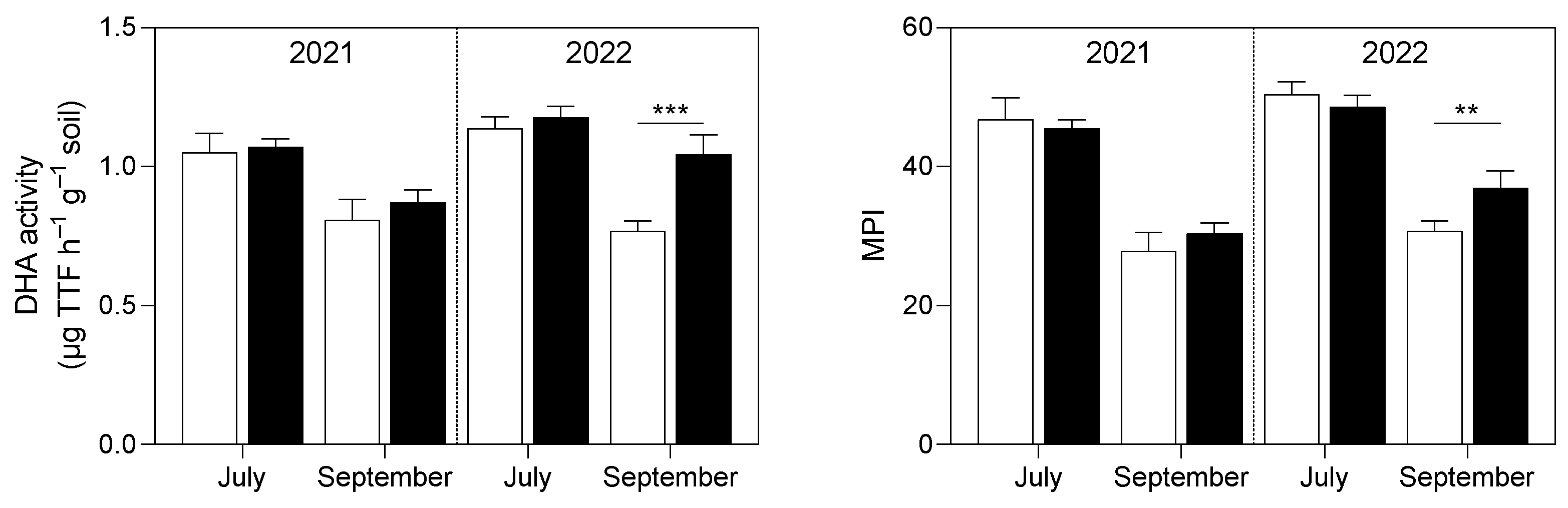
| Title 1 | Unit | Value |
|---|---|---|
| Sand | % | 69.0 |
| Silt | % | 25.0 |
| Clay | % | 6.0 |
| pH | 6.1 | |
| f | % | 35.0 |
| CEC | meq 100 g–1 | 38.0 |
| CaCO3 | % | <1.0 |
| TOC | % | 1.13 |
| Ntot | g kg–1 | 0.98 |
| C/N ratio | 11.5 | |
| Available P | mg kg–1 | 13.0 |
| Exchangeable K | mg kg–1 | 113.0 |
| Exchangeable Mg | mg kg–1 | 480.0 |
| Exchangeable Ca | mg kg–1 | 2111.0 |
| 2021 | 2022 | |||
|---|---|---|---|---|
| CTR | B | CTR | B | |
| Leaf biomass (g DW plant–1) | 20.1 ± 5.2 | 17.0 ± 2.0 | 20.5 ± 5.3 | 36.6 ± 5.7 * |
| Branches biomass (g DW plant–1) | 21.0 ± 2.7 | 25.0 ± 5.5 | 24.8 ± 3.1 | 30.1 ± 6.6 |
| Trunk biomass (g DW plant–1) | 40.6 ± 5.9 | 46.5 ± 9.9 | 46.7 ± 6.8 | 54.4 ± 11.6 |
| Total biomass (g DW plant–1) | 81.7 ± 12.5 | 88.5 ± 15.6 | 91.9 ± 11.1 | 121.1 ± 20.7 * |
| Leaf number (n° plant–1) | 88.4 ± 16.2 | 76.8 ± 19.2 | 116.0 ± 12.4 | 124.6 ± 9.9 |
| LA (cm2) | 40.1 ± 6.3 | 51.3 ± 7.1 * | 40.4 ± 0.5 | 58.6 ± 0.3 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




