Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of NH₂-MIL-88B(Fe)
2.3. Preparation of Fe₃O₄@Porous Carbon
2.4. Synthesis of g-C₃N₄
2.5. Fabrication of Fe₃O₄@PC/g-C₃N₄ Nanocomposite
2.6. Diagrammatic Scheme of Synthesis
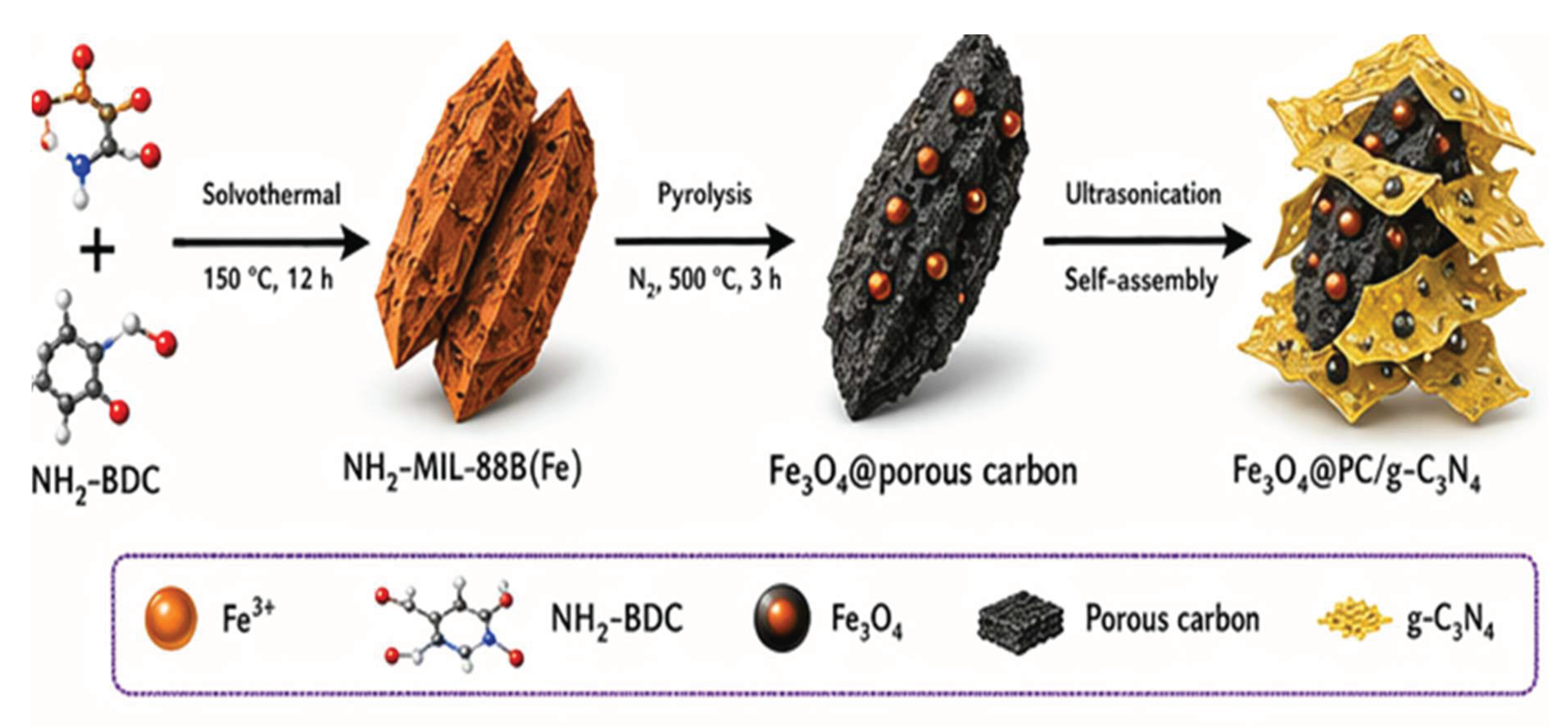
3. Characterization Techniques
4. Results and Discussion
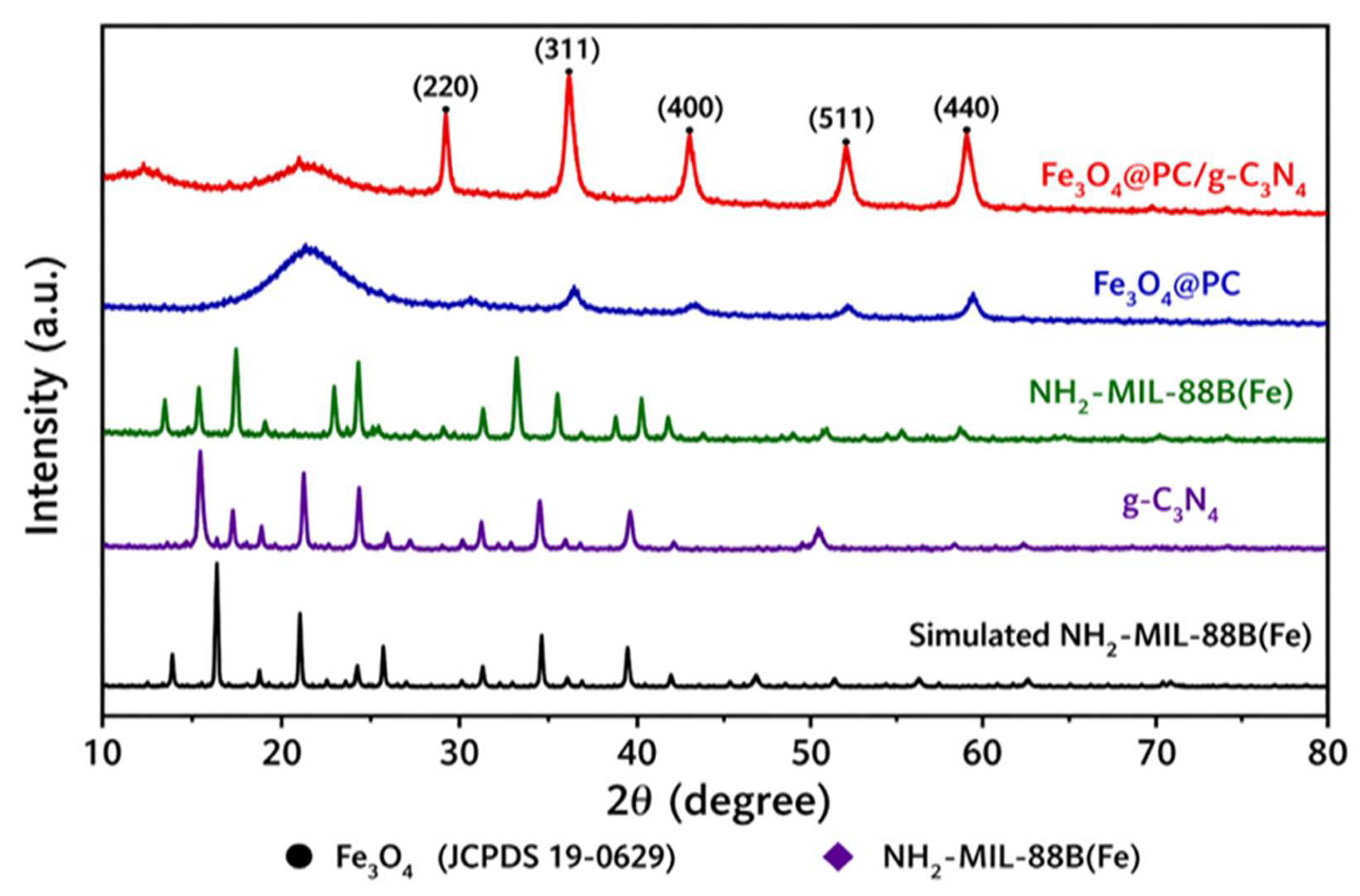
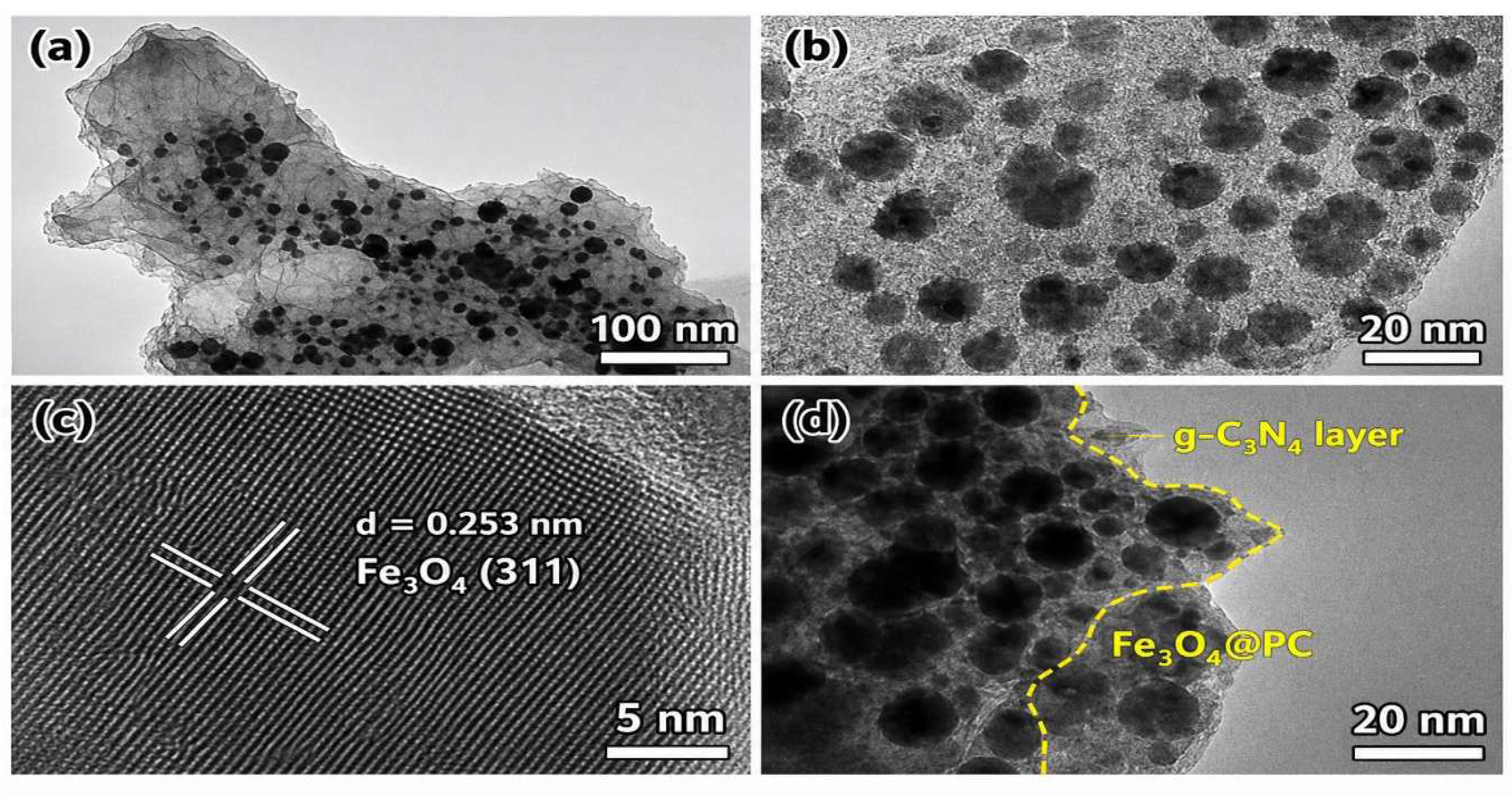
4.1. Structural and Morphological Analysis
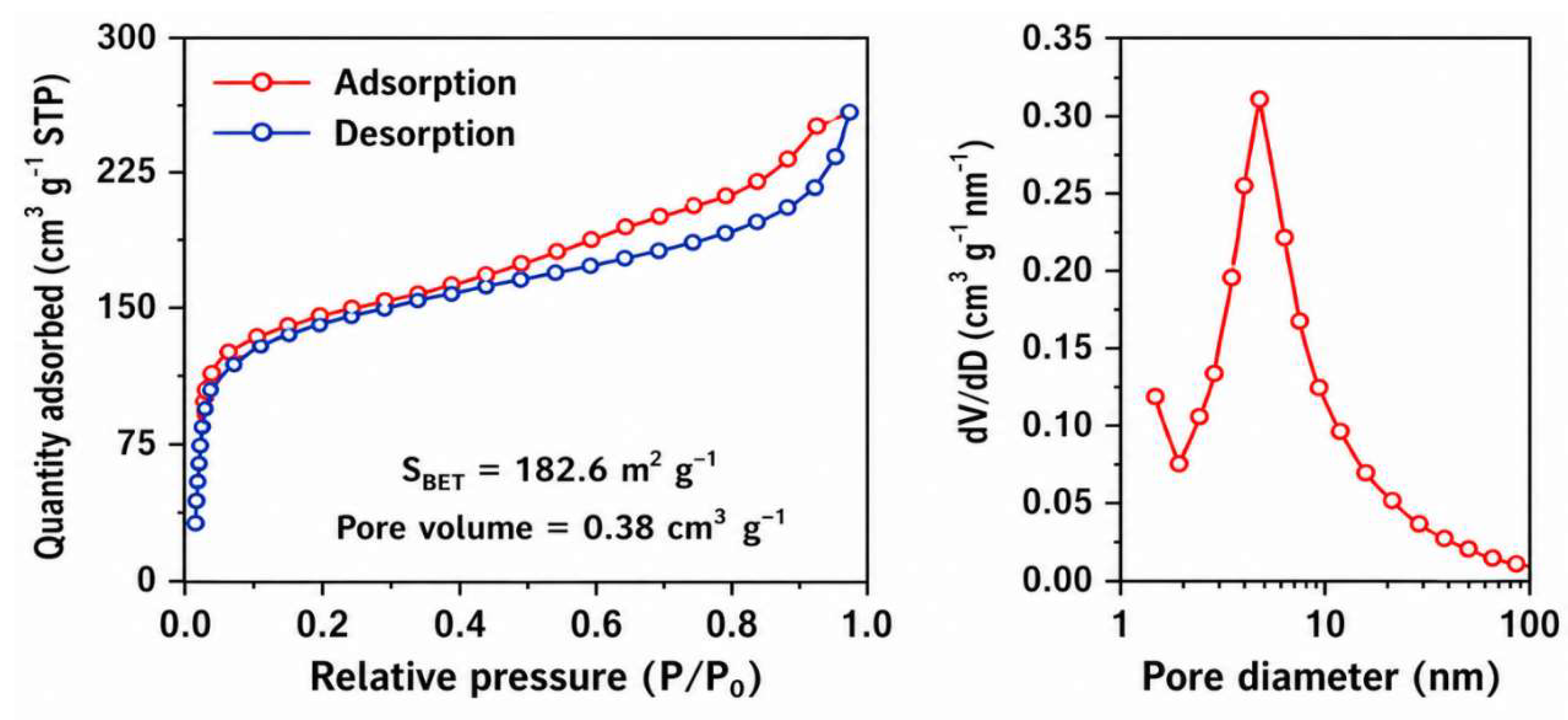
4.2. Surface Area and Porosity
4.3. Optical Properties
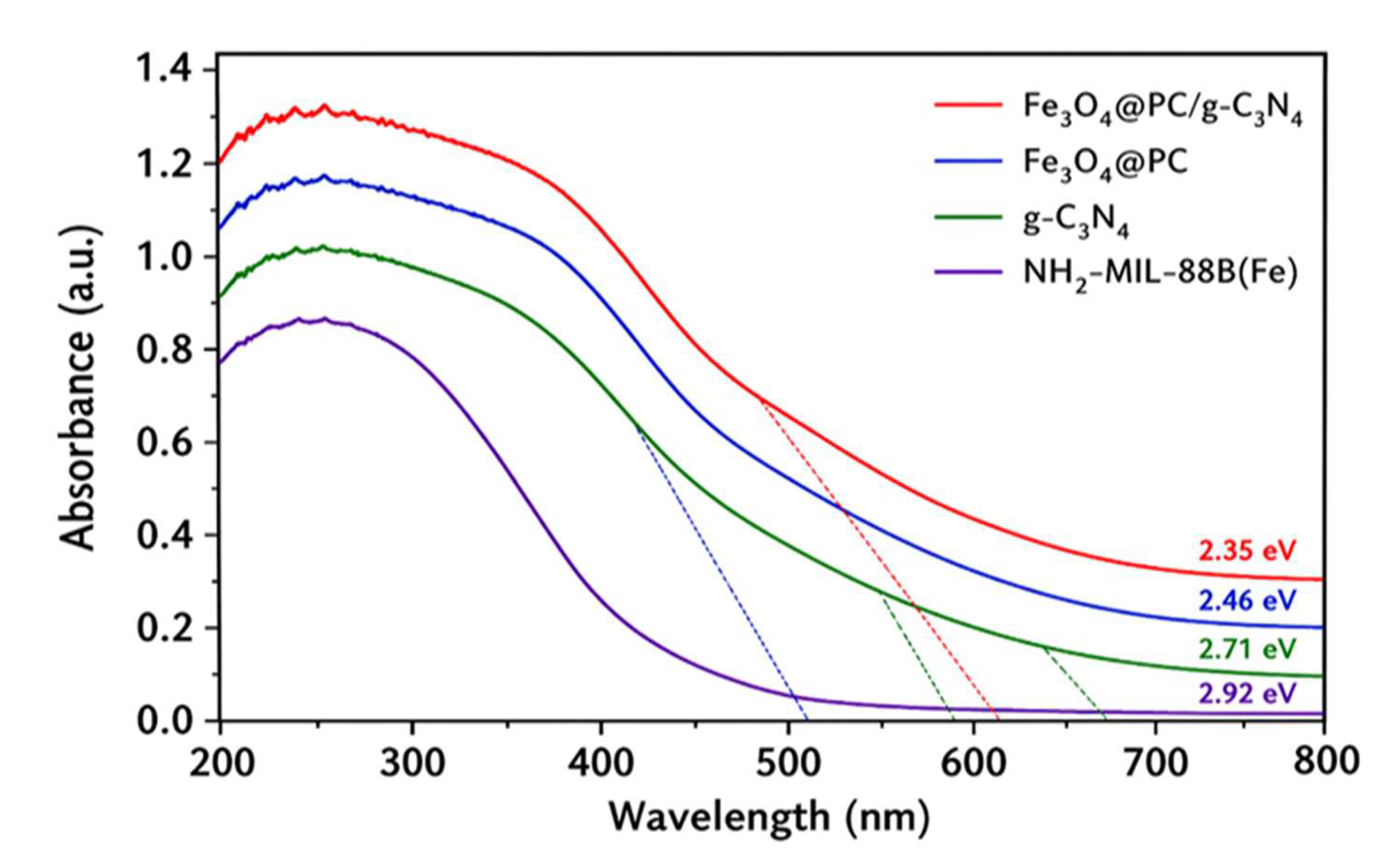
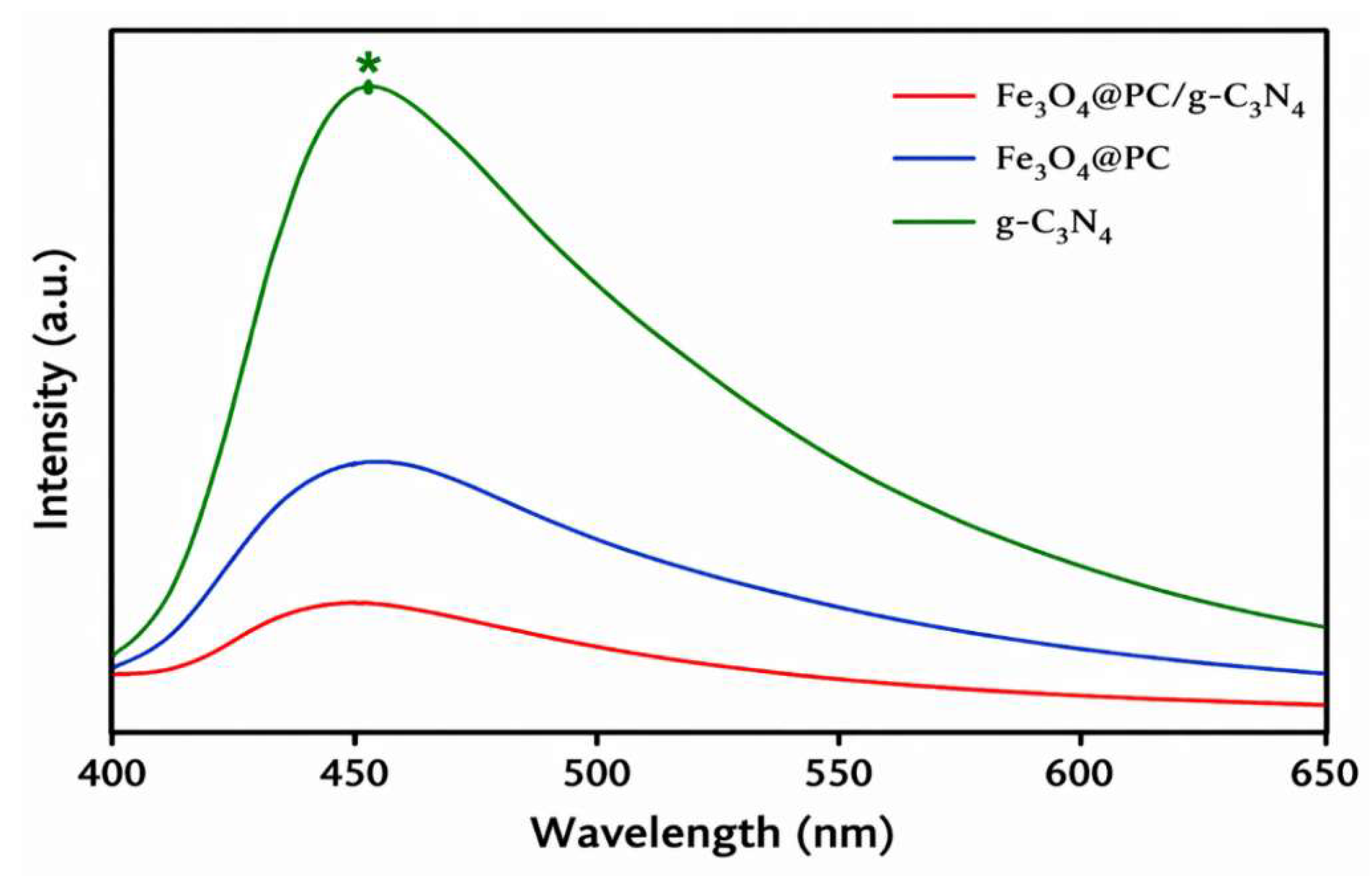
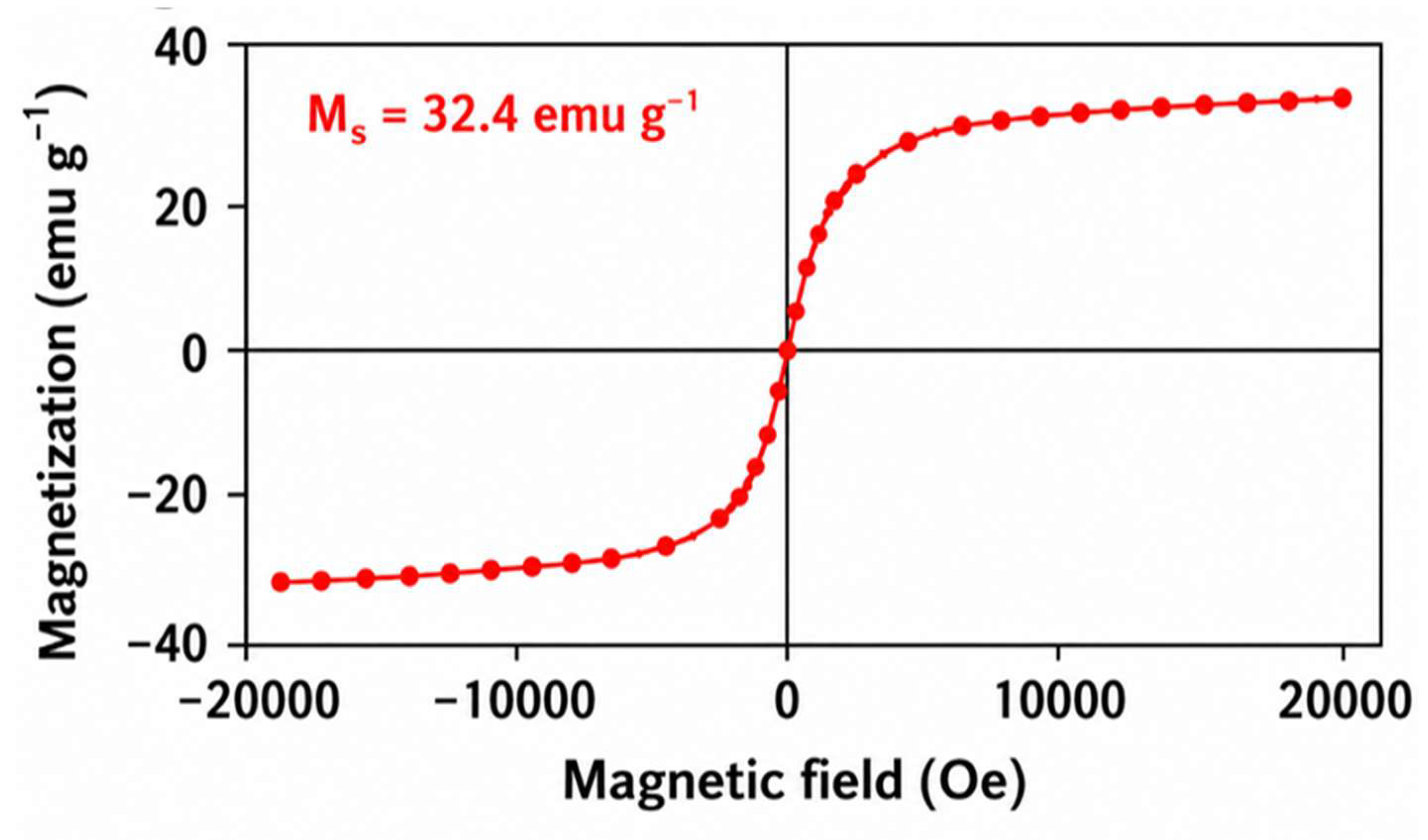
4.4. Magnetic Properties
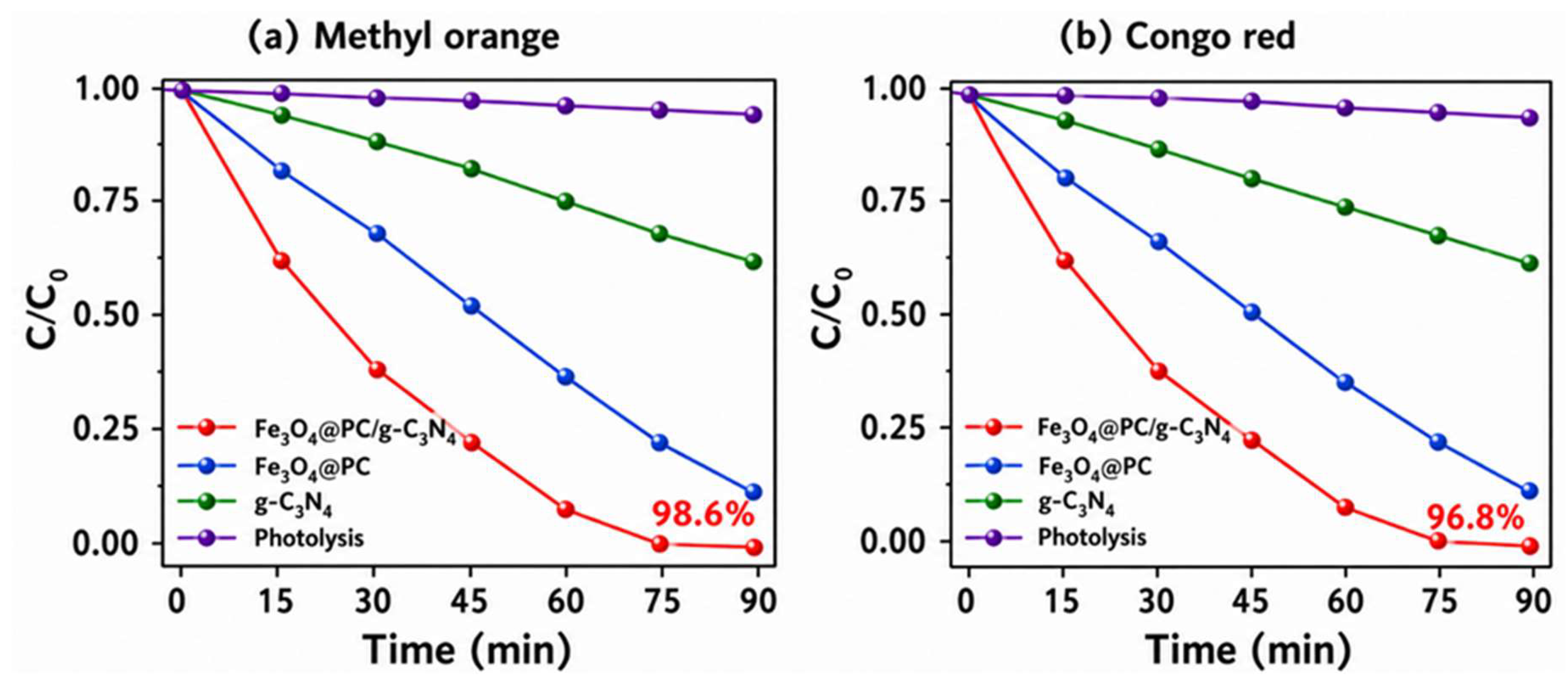
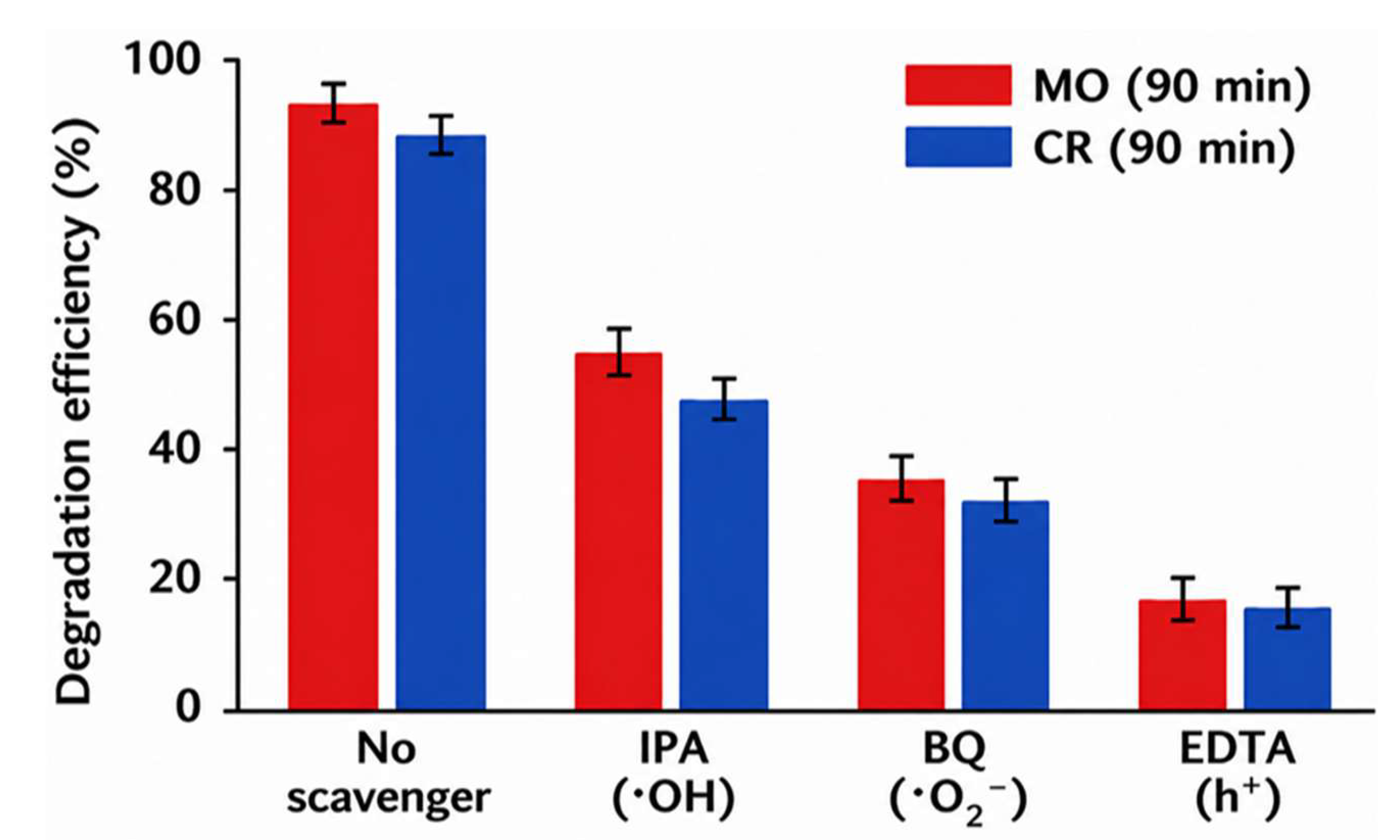
4.5. Photocatalytic Performance
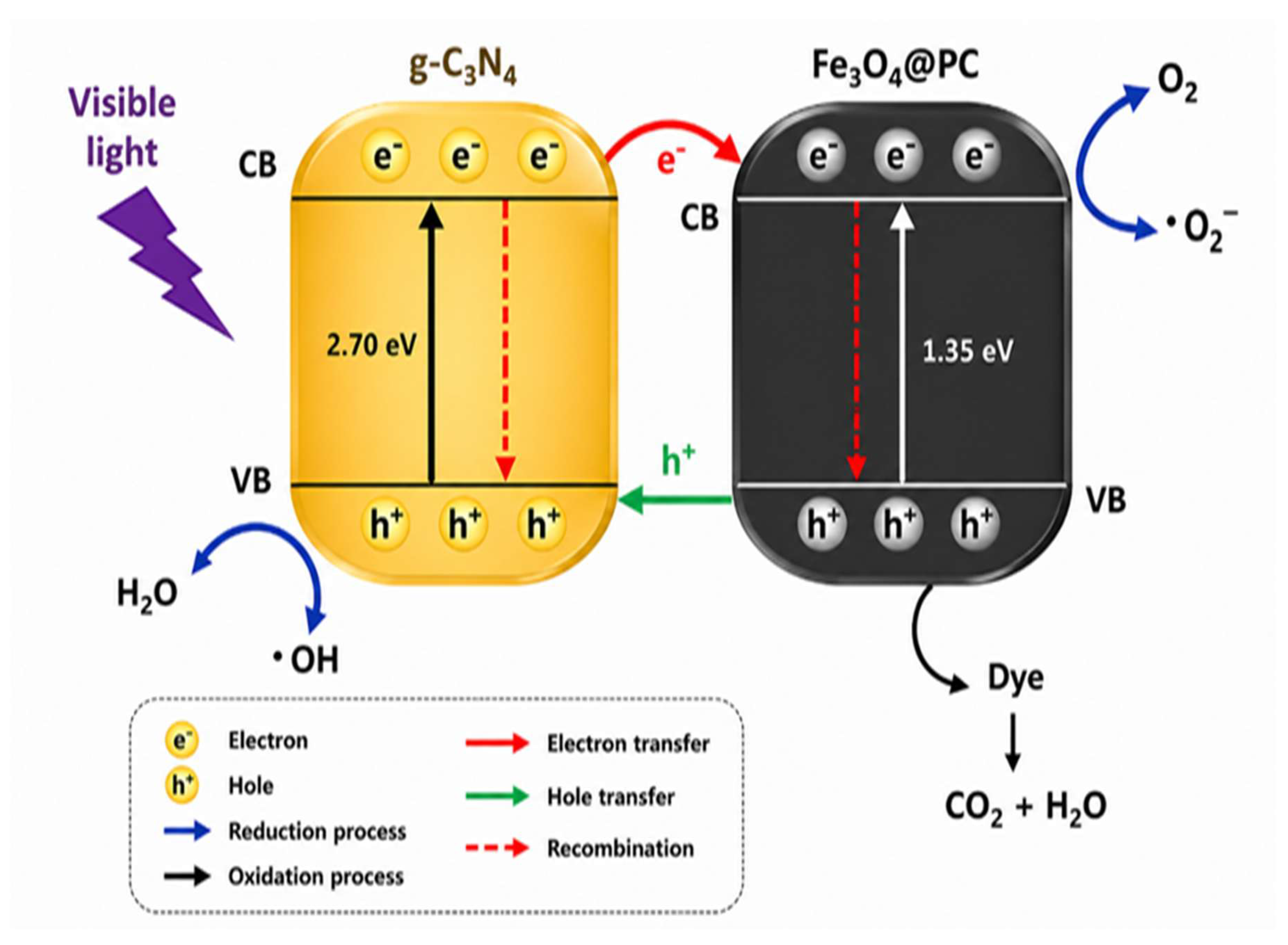
4.6. Mechanism Study
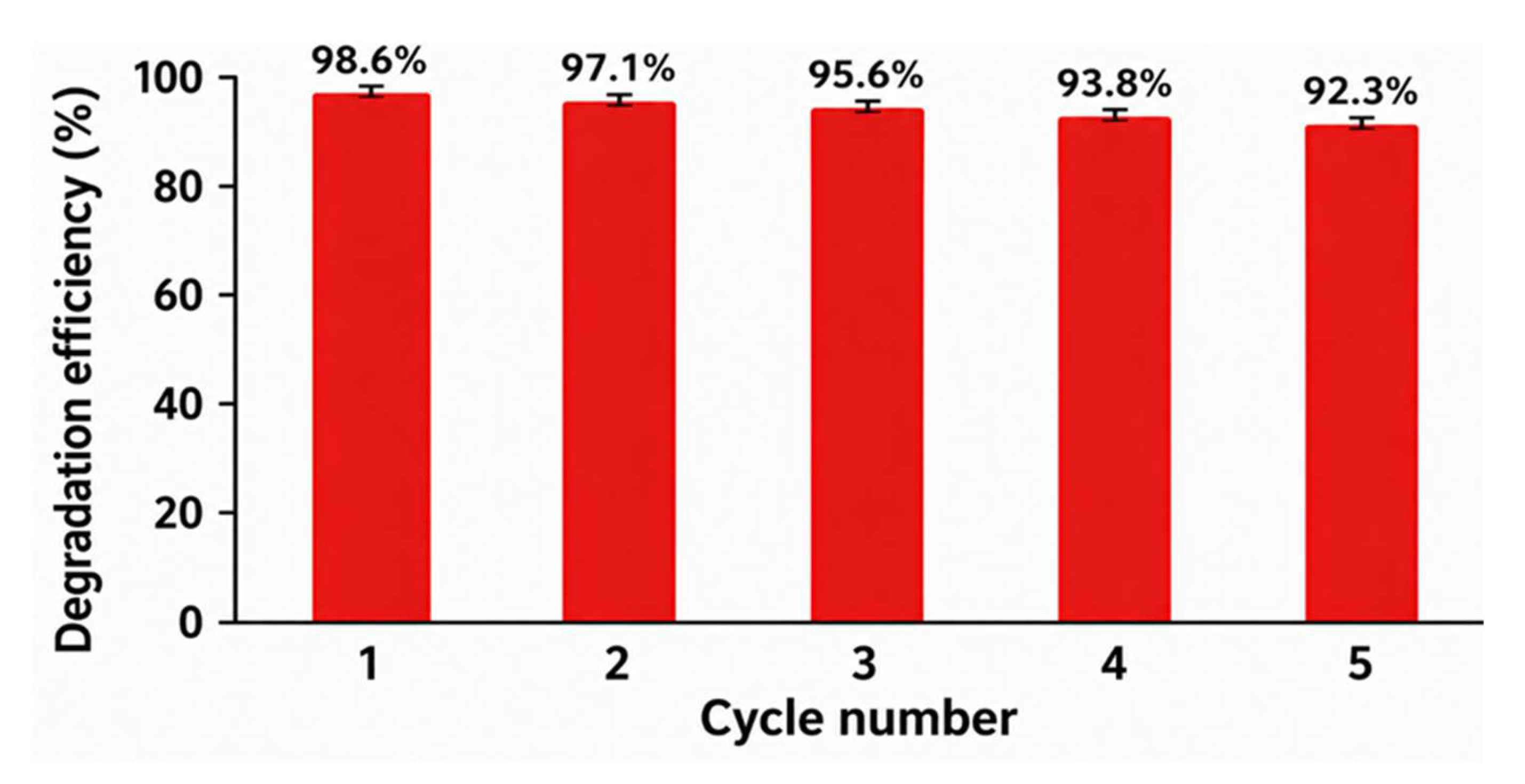
4.7. Reusability and Stability
5. Conclusion
Conflicts of Interest
References
- Khan, W.U.; Ahmed, S.; Dhoble, Y.; Madhav, S. A critical review of hazardous waste generation from textile industries and associated ecological impacts. J. Indian Chem. Soc. 2023, 100(1), 100829. [Google Scholar] [CrossRef]
- Osemba, M.O. Electrochemical Degradation and Chemical Assessment of Azo Dyes in the Textile Waste Water. PhD Thesis] [Internet]. PhD Thesis, Pwani University, 2019. Available online: https://elibrary. https://www.researchgate.net/profile/Martin-Osemba/publication/395242671_ELECTROCHEMICAL_DEGRADATION_AND_CHEMICAL_ASSESSMENT_OF_AZO_DYES_IN_THE_TEXTILE_WASTE_WATER_MARTIN_OUMA_OSEMBA/links/68b8f078360112563e103c4b/ELECTROCHEMICAL-DEGRADATION-AND-CHEMICAL-ASSESSMENT-OF-AZO-DYES-IN-THE-TEXTILE-WASTE-WATER-MARTIN-OUMA-OSEMBA.pdf.
- Alzain, H.; Kalimugogo, V.; Hussein, K.; Karkadan, M. A review of environmental impact of azo dyes. Int. J. Res. Rev. 2023, 10(6), 673–89. [Google Scholar] [CrossRef]
- Mutuku, D.; Osemba, M.O.; Thoruwa, T.; Keheze, F. Effect of chitosan coating on the efficiency and degradation of solar panels. Mount Kenya University [Internet]. 2025. Available online: https://www.academia.edu/download/128364799/Chitosan_coating.pdf.
- Dutta, P.; Rabbi, M.; Sufian, M.; Mahjebin, S. Effects of textile dyeing effluent on the environment and its treatment: A review. Eng. Appl. Sci. Lett. (EASL) 2022, 5, 1–17. [Google Scholar] [CrossRef]
- Negi, A. Environmental impact of textile materials: challenges in fiber–dye chemistry and implication of microbial biodegradation. Polymers 2025, 17(7), 871. [Google Scholar] [CrossRef] [PubMed]
- Osemba, M.; Maghanga, J.; Ojwang, L. Green Synthesis of Indium Tin Oxide Nanoparticles from Herbal Extracts for Photocatalytic Dye Degradation [Internet]. 2025. Available online: https://www.researchgate.net/profile/Martin-Osemba/publication/399824967_Green_Synthesis_of_Indium_Tin_Oxide_Nanoparticles_from_Herbal_Extracts_for_Photocatalytic_Dye_Degradation/links/696a08c5abecff2489ec1de0/Green-Synthesis-of-Indium-Tin-Oxide-Nanoparticles-from-Herbal-Extracts-for-Photocatalytic-Dye-Degradation.pdf.
- Islam, T.; Repon, MdR; Islam, T.; Sarwar, Z.; Rahman, M.M. Impact of textile dyes on health and ecosystem: a review of structure, causes, and potential solutions. Env. Sci. Pollut. Res. 2022, 30(4), 9207–42. [Google Scholar] [CrossRef] [PubMed]
- Oguanobi, N.C.; Aniagor, C.O.; Okoronkwo, G.; Ude, C.N.; Onu, C.E.; Anike, E.N. Industrial dye effluent sources, generation, and value-added products. In Engineered Biocomposites for Dye Adsorption [Internet]; Elsevier, 2025 [cited 2026 Apr 26; pp. 1–10. Available online: https://www.sciencedirect.com/science/article/pii/B9780443298776000019.
- as Electrocatalyst SN. International Journal of Advanced Research [Internet]. 2024. Available online: https://www.academia.edu/download/128364783/electrochemical_removal.pdf.
- onto Indium SNEE. International Journal of Pure and Applied Chemistry [Internet]. 2026. Available online: https://www.researchgate.net/profile/Martin-Osemba/publication/399824133_Silver_Nanoparticle_Elecrocatalyst_Embedded_onto_Indium_Tin_Oxide_Electrodes_for_Degradation_of_Azo_Dyes/links/6969fcd9abecff2489ec167d/Silver-Nanoparticle-Elecrocatalyst-Embedded-onto-Indium-Tin-Oxide-Electrodes-for-Degradation-of-Azo-Dyes.pdf.
- Osemba, M.O. Microwave-assisted starch stabilization & Chitosan green synthesis of zinc oxide nanoparticles for the photocatalytic applications [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9131972/latest.
- Nidheesh, P.V.; Couras, C.; Karim, A.V.; Nadais, H. A review of integrated advanced oxidation processes and biological processes for organic pollutant removal. Chem. Eng. Commun. 2022, 209(3), 390–432. [Google Scholar] [CrossRef]
- Osemba, M.O.; Huerta, A.C. Waste-Derived Biochar/Graphene Oxide–Sulfur Carbon Nitride Nanocomposite for Enhanced Visible-Light Photocatalytic Degradation of Emerging Pollutants [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9209007/latest.
- Silva, J.A. Advanced oxidation process in the sustainable treatment of refractory wastewater: a systematic literature review. Sustainability 2025, 17(8), 3439. [Google Scholar] [CrossRef]
- OSEMBA, M.O. SYNTHESIS AND CHARACTERIZATION OF SILVER NANOPARTICLE ELECROCATALYST EMBEDDED ONTO INDIUM TIN OXIDE ELECTRODES FOR DEGRADATION OF AZO DYES [Internet]. 2025. Available online: https://www.academia.edu/download/131940347/MARTIN_OUMA_OSEMBA_THESIS_1_.pdf.
- Manna, M.; Sen, S. Advanced oxidation process: a sustainable technology for treating refractory organic compounds present in industrial wastewater. Env. Sci. Pollut. Res. 2022, 30(10), 25477–505. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, Y.; Zhang, H.; Duan, X.; Ma, J.; Sun, H.; et al. Carbonaceous materials in structural dimensions for advanced oxidation processes. Chem. Soc. Rev. 2025, 54(5), 2436–82. [Google Scholar] [CrossRef]
- Pandis, P.K.; Kalogirou, C.; Kanellou, E.; Vaitsis, C.; Savvidou, M.G.; Sourkouni, G.; et al. Key points of advanced oxidation processes (AOPs) for wastewater, organic pollutants and pharmaceutical waste treatment: A mini review. ChemEngineering 2022, 6(1), 8. [Google Scholar] [CrossRef]
- Osemba, M.O.; Huerta, A.C. Visible-Light-Driven Photocatalytic Degradation of Methylene Blue Using a Graphene Oxide/Sulfur Carbon Nitride (GO/SCN) Nanocomposite [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9207538/latest.
- Pavel, M.; Anastasescu, C.; State, R.N.; Vasile, A.; Papa, F.; Balint, I. Photocatalytic degradation of organic and inorganic pollutants to harmless end products: assessment of practical application potential for water and air cleaning. Catalysts 2023, 13(2), 380. [Google Scholar] [CrossRef]
- Osemba, M.O.; Huerta, A.C. Surface-Engineered Fe₃O₄/Graphene Oxide/Polymer Magnetic Nanocomposites for Efficient and Reusable Removal of Pb^2+ and Cd^2+ from Wastewater [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9248702/latest.
- Osemba, M.O.; Huerta, A.C.; Karenga, S.; Keru, G. Surface Functionalization and Interfacial Chemistry in PVDF/BaTiO₃/Graphene Nanocomposites for Enhanced Dielectric Performance [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9370521/latest.
- Rasool, B.S.; Abbas, A.K.; Haddad, R. Photocatalytic innovations in environmental remediation: mechanisms, materials, and challenges for persistent organic pollutant removal. Env. Monit. Assess. 2025, 197(10), 1086. [Google Scholar] [CrossRef]
- Roslan, N.N.; Lau, H.L.H.; Suhaimi, N.A.A.; Shahri, N.N.M.; Verinda, S.B.; Nur, M.; et al. Recent advances in advanced oxidation processes for degrading pharmaceuticals in wastewater—a review. Catalysts 2024, 14(3), 189. [Google Scholar] [CrossRef]
- Osemba, M.O. Recent Advances Of PVA/Chitosan/ITO Nanocomposites in Structural, Optical, Dielectric, And Nonlinear Optical Properties [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9131821/latest.
- Genius, G.M. Surface Functionalization and Plasmonic Interactions in PVA–TiO₂–Silver Nanoparticle Hybrid Nanocomposites [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9424010/latest.
- Jorge, A.M.; Athira, K.K.; Alves, M.B.; Gardas, R.L.; Pereira, J.F. Textile dyes effluents: A current scenario and the use of aqueous biphasic systems for the recovery of dyes. J. Water Process Eng. 2023, 55, 104125. [Google Scholar] [CrossRef]
- Patti, A.; Acierno, D. Towards the sustainability of the plastic industry through biopolymers: Properties and potential applications to the textiles world. Polymers 2022, 14(4), 692. [Google Scholar] [CrossRef]
- Osemba, M.O.; Huerta, A.C. Structural, Optical, Dielectric and Nonlinear Properties of PVA/Na-Alg/ITO Nanocomposite Films [Internet]. 2026. Available online: https://www.researchsquare.com/article/rs-9366243/latest.
- Islam, M.T.; Islam, T.; Islam, T.; Repon, M.R. Synthetic dyes for textile colouration: Process, factors and environmental impact. Text. Leather Rev. 2022, 5, 327–73. [Google Scholar] [CrossRef]
- Akter, T.; Protity, A.T.; Shaha, M.; Al Mamun, M.; Hashem, A. The Impact of Textile Dyes on the Environment. In Nanohybrid Materials for Treatment of Textiles Dyes [Internet]; Ahmad, A., Jawaid, M., Mohamad Ibrahim, M.N., Yaqoob, A.A., Alshammari, M.B., Eds.; Springer Nature Singapore: Singapore, 2023 [cited 2026 Apr 26; pp. 401–31. Available online: https://link.springer.com/10.1007/978-981-99-3901-5_17.
- Al-dolaimy, F.; Kzar, M.H.; Hussein, S.A.; Dakheel, A.H.; Ramadan, M.F.; Abdulwahid, A.S.; et al. Advancements in Exploring Metal-Organic Gels: Structure, Synthesis, and Characterization with a Focus on Preparation Conditions. J. Clust. Sci. 2024, 35(3), 741–63. [Google Scholar] [CrossRef]
- Li, D.; Yadav, A.; Zhou, H.; Roy, K.; Thanasekaran, P.; Lee, C. Advances and Applications of Metal-Organic Frameworks (MOFs) in Emerging Technologies: A Comprehensive Review. Glob. Chall. 2024, 8(2), 2300244. [Google Scholar] [CrossRef]
- Li, T.; Pan, Y.; Shao, B.; Zhang, X.; Wu, T.; He, Q.; et al. Covalent–Organic Framework (COF)-Core–Shell Composites: Classification, Synthesis, Properties, and Applications. Adv. Funct. Mater. 2023, 33(45), 2304990. [Google Scholar] [CrossRef]
- Chen, C.; Shen, L.; Wang, B.; Lu, X.; Raza, S.; Xu, J.; et al. Environmental applications of metal–organic framework-based three-dimensional macrostructures: a review. Chem. Soc. Rev. 2025, 54(5), 2208–45. [Google Scholar] [CrossRef]
- Cui, Y.; Zhao, Y.; Wu, J.; Hou, H. Heterogeneous Nanosized Metal (Metallic Compound)@Metal-Organic Framework Composites: Recent Advances in the Preparation and Applications. Adv. Funct. Mater. 2023, 33(36), 2302573. [Google Scholar] [CrossRef]
- Yao, Y.; Zhao, X.; Chang, G.; Yang, X.; Chen, B. Hierarchically Porous Metal–Organic Frameworks: Synthetic Strategies and Applications. Small Struct. 2023, 4(1), 2200187. [Google Scholar] [CrossRef]
- Liu, X.; Verma, G.; Chen, Z.; Hu, B.; Huang, Q.; Yang, H.; et al. Metal-organic framework nanocrystal-derived hollow porous materials: Synthetic strategies and emerging applications. The Innovation [Internet]. 2022, 3. Available online: https://www.cell.com/the-innovation/fulltext/S2666-6758(22)00077-7.
- Pathak, P.K.; Ghosh, A.; Dasgupta Ghosh, B. Metal–Organic Frameworks and Their Derived Materials for Energy Storage and Harvesting: Recent Advances and Future Perspectives. Energy Fuels 2026, 40(16), 8517–52. [Google Scholar] [CrossRef]
- Tsang, M.Y.; Sinelshchikova, A.; Zaremba, O.; Schöfbeck, F.; Balsa, A.D.; Reithofer, M.R.; et al. Multilength Scale Hierarchy in Metal-Organic Frameworks: Synthesis, Characterization and the Impact on Applications. Adv. Funct. Mater. 2024, 34(43), 2308376. [Google Scholar] [CrossRef]
- Xie, Y.; Zhang, T.; Wang, B.; Wang, W. The application of metal–organic frameworks in water treatment and their large-scale preparation: a review. Materials 2024, 17(9), 1972. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Wang, Y.; Shi, D.; Han, S.; Xiong, L. A review on shaped monolithic MOFs for photocatalytic environmental remediation and fuel and energy production. Ionics 2026, 32(4), 3791–828. [Google Scholar] [CrossRef]
- Gautam, J.; Kale, A.M.; Rawal, J.; Varma, P.; Lee, S.J.; Lee, S.; et al. Biomass-Derived Carbon Photocatalysts for Organic Pollutant Degradation: Strategies and Perspectives. Carbon Neutraliz. 2026, 5(1), e70109. [Google Scholar] [CrossRef]
- Bibi, F.; Zubair, M.; Kalsoom, A.; Hossain, I.; Yasmeen, K.; Aldosari, E.; et al. Development of a high-performance La/Cu Co-doped BiFeO₃/g-C₃N₄ nanocomposite for efficient degradation of moxifloxacin and crystal violet dye for environmental applications. J. Sol.-Gel Sci. Technol. 2025, 116(3), 3030–53. [Google Scholar] [CrossRef]
- Haris, F.F.P.; Rajeev, A.; Poyil, M.M.; Kelappan, N.K.; Sasi, S. Development of a MOF-5/g-C3N4 nanocomposite: an effective type 2 heterojunction photocatalyst for rhodamine B dye degradation. Env. Sci. Pollut. Res. 2024, 31(50), 60298–313. [Google Scholar] [CrossRef]
- Roushree, R.R.; Haimbodi, R. Recent Advances in ZnO-Based Nanocomposites for Amoxicillin Photocatalytic Degradation and Adsorption in Wastewater: A Review. J. Inorg. Organomet. Polym. 2026, 36(1), 26–63. [Google Scholar] [CrossRef]
- Zeinali, A.; Moradi, L. Rational design and fabrication of g-C₃N₄/CS/MOF-5 nanocomposite as a green, recyclable and efficient catalyst for the rapid synthesis of novel benzo [a] pyrano [2, 3-c] phenazine derivatives. Scientific Reports [Internet]. 2025. Available online: https://www.nature.com/articles/s41598-025-32332-z.
- Jallouli, N.; Mansouri, F.; Roy, S.; Ahmaruzzaman, Md; Cheema, W.; Ksibi, M. Photocatalytic membranes for pharmaceutical removal: advancements and future directions. Emergent Mater. 2026, 9(1–3), 51. [Google Scholar] [CrossRef]
- Ramalingam, G.; Arunkumar, P.; Alqahtani, M.D.; Elgarahy, A.M. Photocatalytic activity of microwave-assisted synthesized MoS₂/g-C₃N₄ composite for efficient decomposition of aniline green dye. J. Clust. Sci. 2025, 36(5), 171. [Google Scholar] [CrossRef]
- Li, S.; Huang, X.; Teng, C.; Xue, X.; Ma, H.; Zhang, B.; et al. Multifunctional Ti₃C₂/g-C₃N₄-PAO hydrogel (PTCH) for closed-loop uranium recycling: Synergistic adsorption, self-reporting sensing, and solar-driven photocatalytic recovery. Chem. Eng. J. 2025, 167131. [Google Scholar] [CrossRef]
- Bissenova, M.; Idrissov, N.; Kuspanov, Z.; Umirzakov, A.; Daulbayev, C. Hybrid adsorption–photocatalysis composites: a sustainable route for efficient water purification. Mater. Renew. Sustain Energy 2025, 14(2), 44. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



