Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
2.3. Peptide Derivative Identification in the Plasma of Rats Administered EP
2.4. Oral Glucose Tolerance Test
2.5. Effects of EP Intake on Hypertensive Kidney Failure
2.5.1. Animals
2.5.2. Histological Examination
2.5.3. Real-Time Polymerase Chain Reaction
2.5.4. Measurement of Nitric Oxide Release from the LRF-Rich Fraction
2.6. Statistical Analysis
2.7. Artificial Intelligence Declaration
3. Results
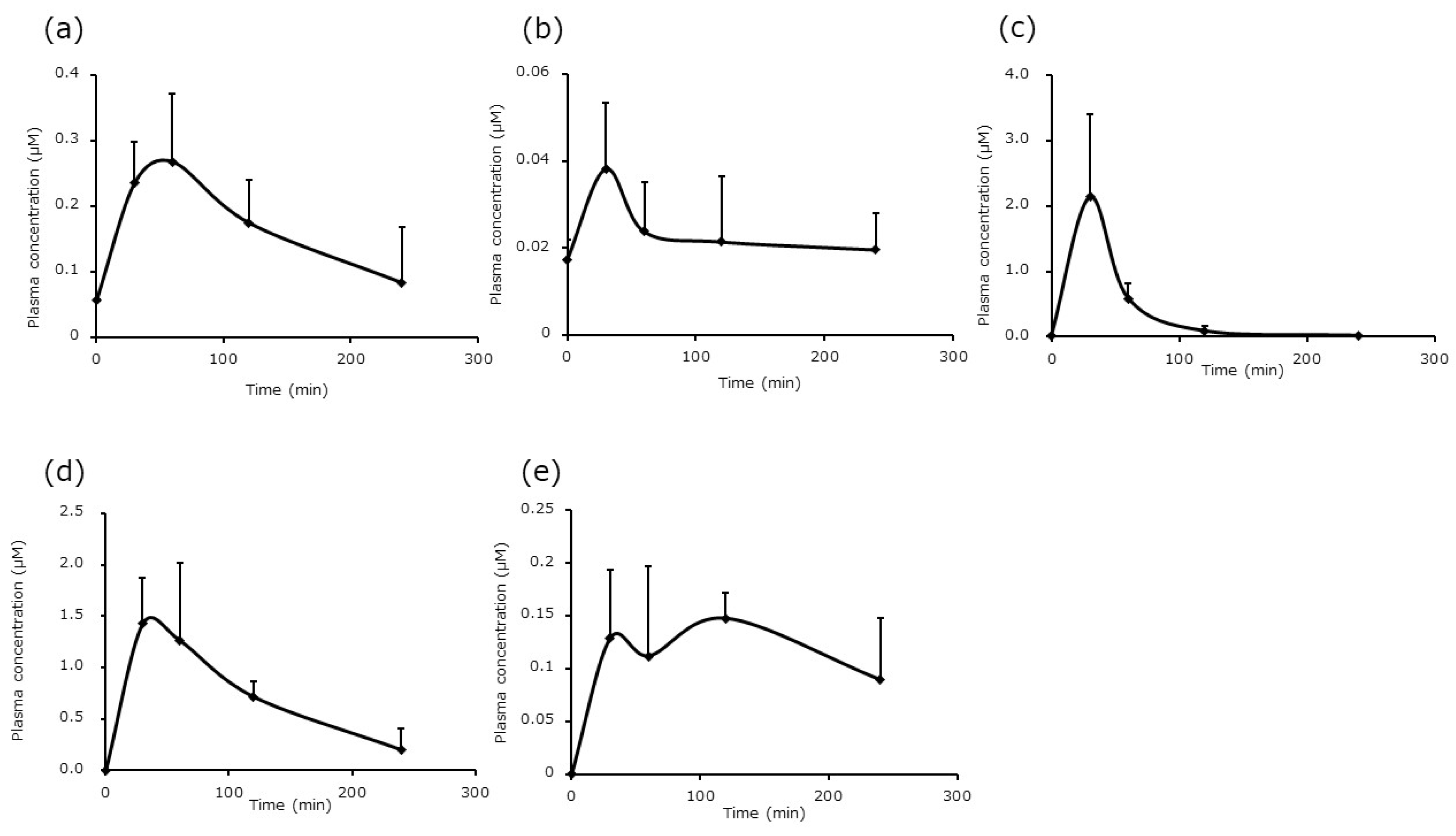
3.1. Quantification in Portal Vein Plasma Samples After EP Administration
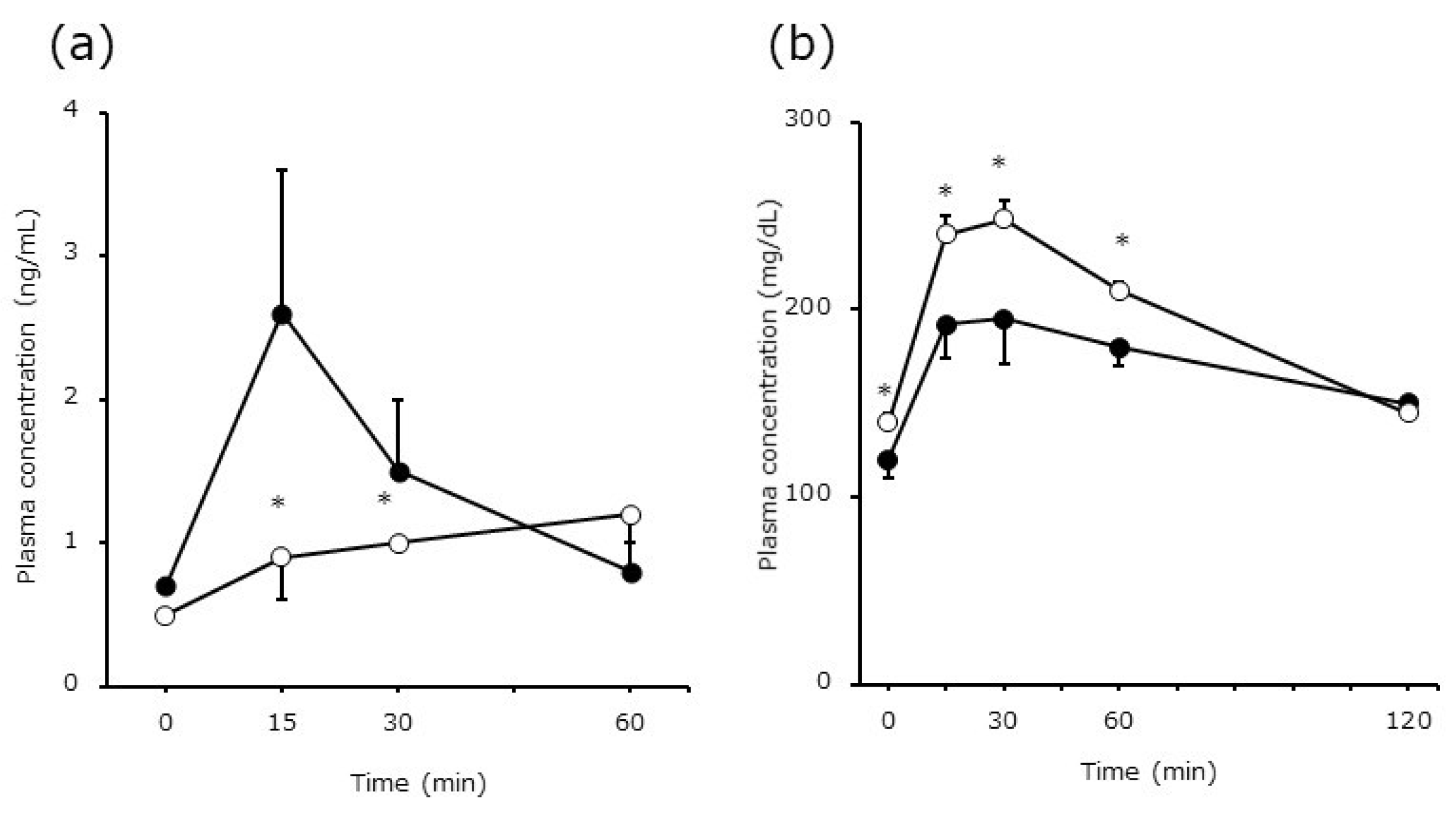
3.2. Comparison of Strain Differences in Insulin and Glucose Levels
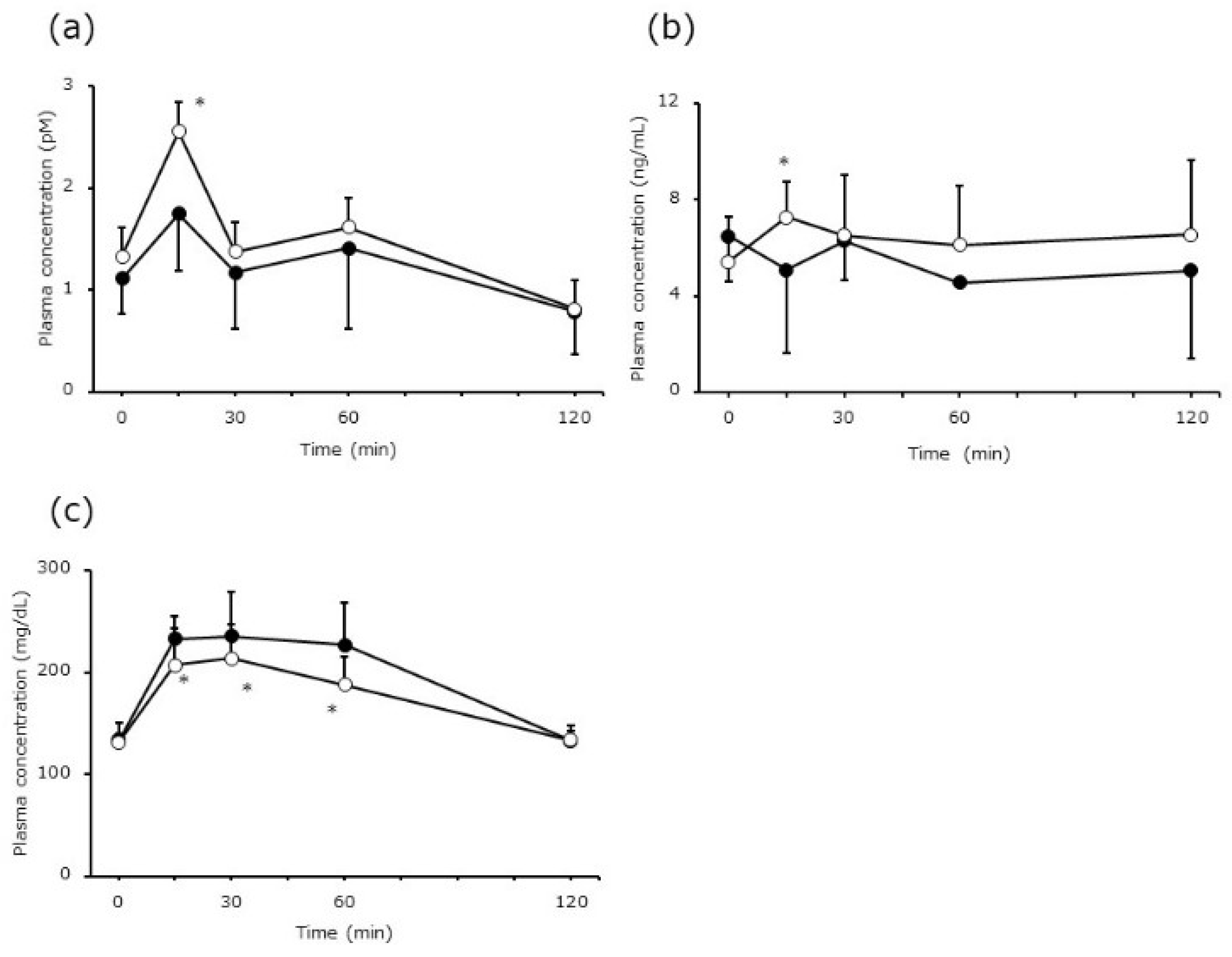
3.3. Effect of EPs on Glucose Metabolism
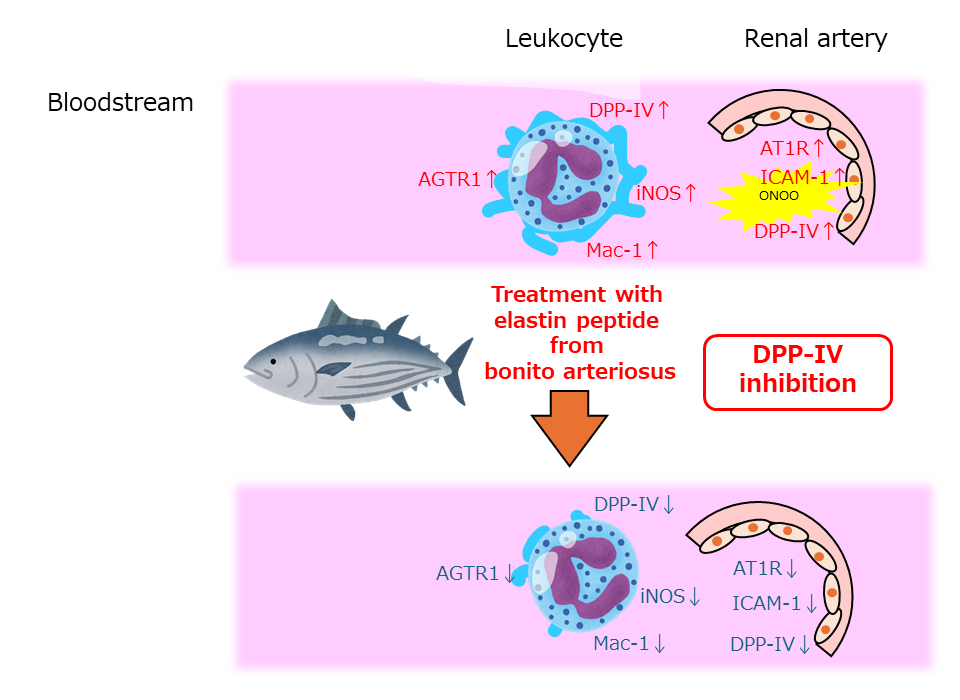
3.4. Effects of EP Intake on Hypertensive Kidney Failure
3.4.1. Chronological Changes in Blood Pressure

3.4.2. Organ Weight and Morphological Observations
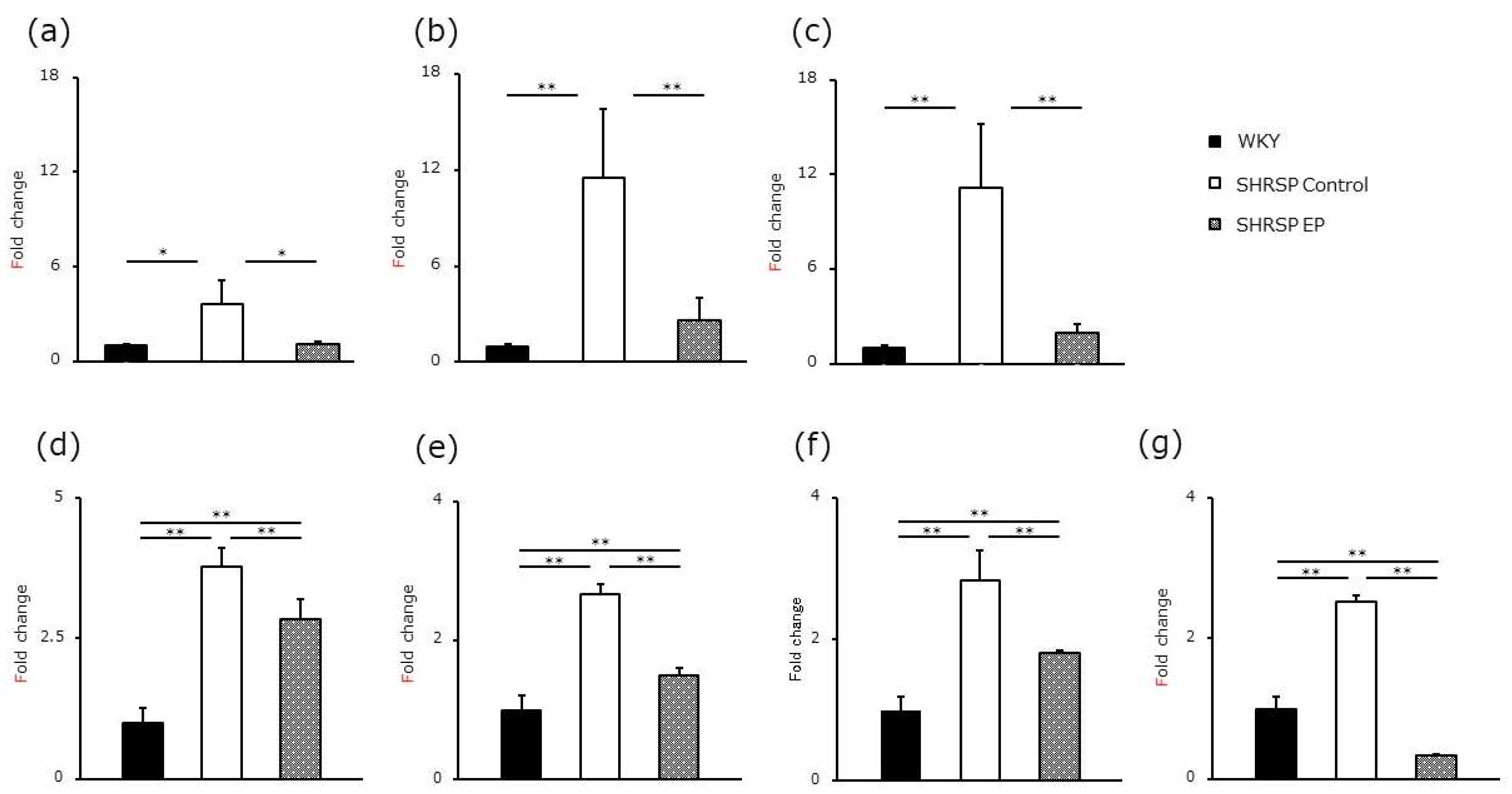
3.4.3. mRNA Expression of Dpp4, Icam-1, and Agtr1 in the Renal Glomerular Fraction
3.4.4. mRNA Expression of Dpp4, Mac-1, Agtr1, and iNOS from the LRF-Rich Fraction
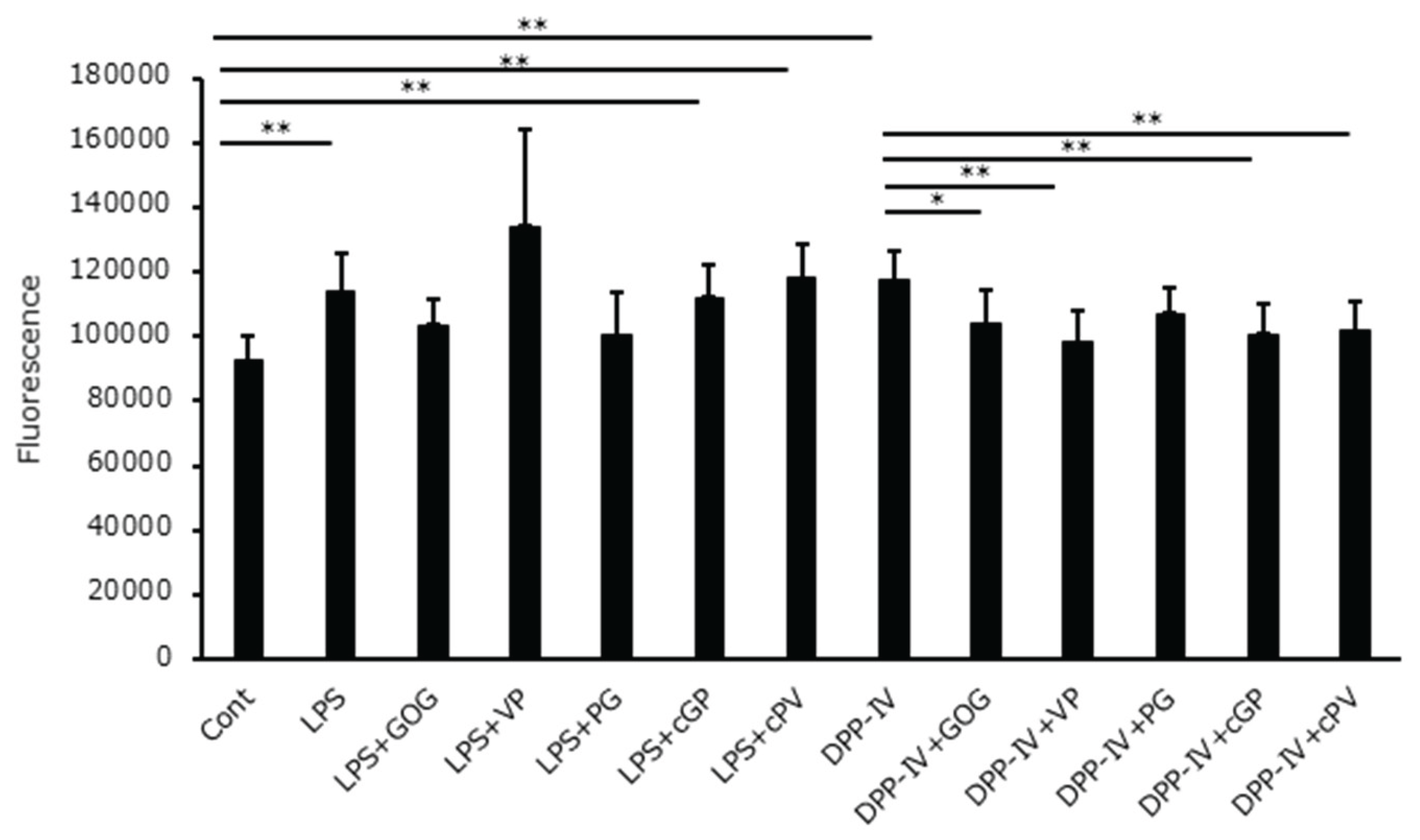
3.4.5. In Vitro Examination of Leukocyte NO Production
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AGTR1 | angiotensin receptor type 1 |
| cGP | cyclo(-Gly-Pro) |
| cPV | cyclo(L-Pro-L-Val) |
| DAF | diaminofluorescein |
| DPP-IV | dipeptidyl peptidase-IV |
| EP | elastin peptide |
| GLP-1 | glucagon-like peptide-1 |
| GOG | H-Gly-Hyp-Gly-OH |
| IC50 | half maximal inhibitory concentration |
| ICAM-1 | intercellular adhesion molecule 1 |
| iNOS | inducible NO synthase |
| LPS | lipopolysaccharide |
| LRF | leukocyte-rich fraction |
| NO | nitric oxide |
| PBS | phosphate-buffered saline |
| PG | H-Pro-Gly-OH |
| SHRSP | stroke-prone spontaneously hypertensive rats |
| VP | H-Val-Pro-OH |
| WKY/KPO | male Wistar-Kyoto rats |
References
- Hanafusa, N.; Abe, M.; Joki, N.; Hoshino, J.; Taniguchi, M.; Kikuchi, K.; Hasegawa, T.; Goto, S.; Ogawa, T.; Kanda, E.; Nakai, S.; Naganuma, T.; Miura, K.; Wada, A.; Takemoto, Y. Annual dialysis data report 2022, Japanese Society for Dialysis Therapy (JSDT) renal data registry. Ren. Replace. Ther. 2025, 11, 54. [Google Scholar] [CrossRef]
- Wakasugi, M.; Narita, I. Trends in the incidence of renal replacement therapy by type of primary kidney disease in Japan, 2006–2020. Nephrology 2023, 28, 119–129. [Google Scholar] [CrossRef]
- Costantino, V.V.; Gil Lorenzo, A.F.; Bocanegra, V.; Vallés, P.G. Molecular mechanisms of hypertensive nephropathy: renoprotective effect of losartan through Hsp70. Cells 2021, 10, 3146. [Google Scholar] [CrossRef] [PubMed]
- Durinx, C.; Lambeir, A.M.; Bosmans, E.; Falmagne, J.B.; Berghmans, R.; Haemers, A.; Scharpé, S.; De Meester, I. Molecular characterization of dipeptidyl peptidase activity in serum: soluble CD26/dipeptidyl peptidase IV is responsible for the release of X-Pro dipeptides. Eur. J. Biochem. 2000, 267, 5608–5613. [Google Scholar] [CrossRef] [PubMed]
- Elmansi, A.M.; Awad, M.E.; Eisa, N.H.; Kondrikov, D.; Hussein, K.A.; Aguilar-Pérez, A.; Herberg, S.; Periyasamy-Thandavan, S.; Fulzele, S.; Hamrick, M.W.; McGee-Lawrence, M.E.; Isales, C.M.; Volkman, B.F.; Hill, W.D. What doesn’t kill you makes you stranger: dipeptidyl peptidase-4 (CD26) proteolysis differentially modulates the activity of many peptide hormones and cytokines generating novel cryptic bioactive ligands. Pharmacol. Ther. 2019, 198, 90–108. [Google Scholar] [CrossRef]
- Klemann, C.; Wagner, L.; Stephan, M.; von Hörsten, S. Cut to the chase: a review of CD26/dipeptidyl peptidase-4’s (DPP4) entanglement in the immune system. Clin. Exp. Immunol. 2016, 185, 1–21. [Google Scholar] [CrossRef]
- Campbell, J.E.; Drucker, D.J. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell. Metab. 2013, 17, 819–837. [Google Scholar] [CrossRef]
- Deacon, C.F. A review of dipeptidyl peptidase-4 inhibitors. Hot topics from randomized controlled trials. Diabetes Obes. Metab. 2018, 20, 34–46. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, Q.; Zhong, J.; Liu, C.; Zheng, B.; Gong, Q. DPP-4 inhibitors as potential candidates for antihypertensive therapy: improving vascular inflammation and assisting the action of traditional antihypertensive drugs. Front. Immunol. 2019, 10, 1050. [Google Scholar] [CrossRef]
- Jones, L.; Jones, A.M. Suspected adverse drug reactions of the type 2 antidiabetic drug class dipeptidyl-peptidase IV inhibitors (DPP4i): can polypharmacology help explain? Pharmacol. Res. Perspect. 2022, 10, e01029. [Google Scholar] [CrossRef]
- Takemori, K.; Yamamoto, E.; Ito, H.; Kometani, T. Prophylactic effects of elastin peptide derived from the bulbus arteriosus of fish on vascular dysfunction in spontaneously hypertensive rats. Life Sci. 2015, 120, 48–53. [Google Scholar] [CrossRef]
- Takemori, K.; Yamamoto, E.; Chikugo, T.; Shiratsuchi, E.; Kometani, T. DPP-IV inhibition by solubilized elastin peptides from yellowtail bulbus arteriosus suppresses ultraviolet-B-induced photoaging in hairless mice. Nutraceuticals 2024, 4, 683–694. [Google Scholar] [CrossRef]
- Shiratsuchi, E.; Nakaba, M.; Shigemura, Y.; Yamada, M.; Sato, K. Fish-elastin hydrolysate: development and impact on the skin and blood vessels. In Marine Proteins and Peptides: Biological Activities and Applications; Kim, S.K., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2013; pp. 467–486. [Google Scholar]
- Okamoto, K.; Yamori, Y.; Nagaoka, A. Establishment of the stroke-prone spontaneously hypertensive rat (SHR). Circ. Res. 1974, 34–35, 143–153. [Google Scholar]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; Emerson, M.; Garner, P.; Holgate, S.T.; Howells, D.W.; Karp, N.A.; Lazic, S.E.; Lidster, K.; MacCallum, C.J.; Macleod, M.; Pearl, E.J.; Petersen, O.H.; Rawle, F.; Reynolds, P.; Rooney, K.; Sena, E.S.; Silberberg, S.D.; Steckler, T.; Würbel, H. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. Br. J. Pharmacol. 2020, 177, 3617–3624. [Google Scholar] [CrossRef]
- Nagao, A.; Nakamoto, Y.; Miyauchi, S.; Sato, K. Presence of modified peptides with high bioavailability and angiotensin-converting enzyme inhibitory activity in Japanese fermented soybean paste (Miso). J. Agric. Food Chem. 2024, 72, 18942–18956. [Google Scholar] [CrossRef]
- Yamamoto, T. Isolation and enrichment of glomeruli using sieving techniques. In Renal and Urinary Proteomics: Methods and Protocols; Thongboonkerd, V., Ed.; John Wiley & Sons: Chichester, 2009; pp. 1–7. [Google Scholar]
- Takemori, K.; Ito, H.; Suzuki, T. Effects of the AT1 receptor antagonist on adhesion molecule expression in leukocytes and brain microvessels of stroke-prone spontaneously hypertensive rats. Am. J. Hypertens. 2000, 13, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Kojima, H.; Nakatsubo, N.; Kikuchi, K.; Kawahara, S.; Kirino, Y.; Nagoshi, H.; Hirata, Y.; Nagano, T. Detection and imaging of nitric oxide with novel fluorescent indicators: diaminofluoresceins. Anal. Chem. 1998, 70, 2446–2453. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Sato, M.; Katakura, Y.; Sugawara, Y.; Shigemura, Y. Increase in blood-transferable linear and cyclic dipeptides in human plasma following ingestion of elastin hydrolysate. Food Hydrocoll. Health 2024, 6, 100188. [Google Scholar] [CrossRef]
- Nongonierma, A.B.; FitzGerald, R.J. Features of dipeptidyl peptidase IV (DPP-IV) inhibitory peptides from dietary proteins. J. Food Biochem. 2019, 43, e12451. [Google Scholar] [CrossRef]
- Trivedi, K.; Dolinsky, V. Role of endogenous incretin hormones, GLP-1 and GIP, in cardiovascular physiology. Can. J. Physiol. Pharmacol. 2026, 104, 1–13. [Google Scholar] [CrossRef]
- Taga, Y.; Iwasaki, Y.; Tometsuka, C.; Funato, N.; Shigemura, Y.; Kusubata, M.; Mizuno, K. Identification of a highly stable bioactive 3-hydroxyproline-containing tripeptide in human blood after collagen hydrolysate ingestion. NPJ Sci. Food 2022, 6, 29. [Google Scholar] [CrossRef]
- Pérez-Vielma, N.M.; Gómez-López, M.; Maldonado, J.; Correa-Basurto, J.; Martínez-Godínez, M.L.Á.; Miliar-García, Á. Recognition of the interaction between the bioactive peptide Val–Pro–Pro and the minimal promoter region of genes SOD and CAT using QCM-D and docking studies. Anal. Methods 2023, 15, 2979–2988. [Google Scholar] [CrossRef] [PubMed]
- Melo, I.S.; Candeia-Medeiros, N.; Ferro, J.N.S.; Cavalcante-Araújo, P.M.; Oliveira, T.L.; Santos, C.E.A.; Cardoso-Sousa, L.; Aguiar, E.M.G.; Wutke Oliveira, S.; Castro, O.W.; Alves-Balvedi, R.P.; Rodrigues, L.P.; Hickmann, J.M.; Alves, D.A.; Santos, I.A.; Jardim, A.C.G.; Siqueira, W.L.; Pipi, A.R.F.; Goulart, L.R.; Barreto, E.O.; Sabino-Silva, R. Restoration of Cyclo-Gly–Pro-induced salivary hyposecretion and submandibular composition by naloxone in mice. PLoS ONE 2020, 15, e0229761. [Google Scholar] [CrossRef]
- Martins, F.L.; Ribeiro-Silva, J.C.; Nistala, R.; Girardi, A.C.C. Bidirectional relation between dipeptidyl peptidase 4 and angiotensin II type I receptor signaling. Am. J. Physiol. Cell. Physiol. 2024, 326, C1203–C1211. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Nakagawa, S.; Wu, Y.; Kawabata, Y.; Numabe, A.; Yanagi, Y.; Tamaki, Y.; Uehara, Y.; Araie, M. A high-salt diet enhances leukocyte adhesion in association with kidney injury in young Dahl salt-sensitive rats. Hypertens. Res. 2017, 40, 912–920. [Google Scholar] [CrossRef]
- Takemori, K.; Inoue, T.; Ito, H. Possible role of nitric oxide generated by leukocytes in the pathogenesis of hypertensive cerebral edema in stroke-prone spontaneously hypertensive rats. Brain Res. 2011, 1417, 137–145. [Google Scholar] [CrossRef]
- Nistala, R.; Meuth, A.I.; Smith, C.; et al. DPP4 inhibition mitigates ANG II-mediated kidney immune activation and injury in male mice. Am. J. Physiol. Ren. Physiol. 2021, 320, F505–F517. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Sen, U. More than just an enzyme: Dipeptidyl peptidase-4 (DPP-4) and its association with diabetic kidney remodelling. Pharmacol. Res. 2019, 147, 104391. [Google Scholar] [CrossRef]
 : SHRSP EP. Mean ± SD, WKY n = 9, SHRSP Control n = 9, SHRSP EP n = 10, Tukey’s honest significant difference (HSD) test, *p < 0.05 WKY vs. SHRSP Control, +p < 0.05 WKY vs. SHRSP EP.
: SHRSP EP. Mean ± SD, WKY n = 9, SHRSP Control n = 9, SHRSP EP n = 10, Tukey’s honest significant difference (HSD) test, *p < 0.05 WKY vs. SHRSP Control, +p < 0.05 WKY vs. SHRSP EP.
 : SHRSP EP. Mean ± SD, WKY n = 9, SHRSP Control n = 9, SHRSP EP n = 10, Tukey’s honest significant difference (HSD) test, *p < 0.05 WKY vs. SHRSP Control, +p < 0.05 WKY vs. SHRSP EP.
: SHRSP EP. Mean ± SD, WKY n = 9, SHRSP Control n = 9, SHRSP EP n = 10, Tukey’s honest significant difference (HSD) test, *p < 0.05 WKY vs. SHRSP Control, +p < 0.05 WKY vs. SHRSP EP.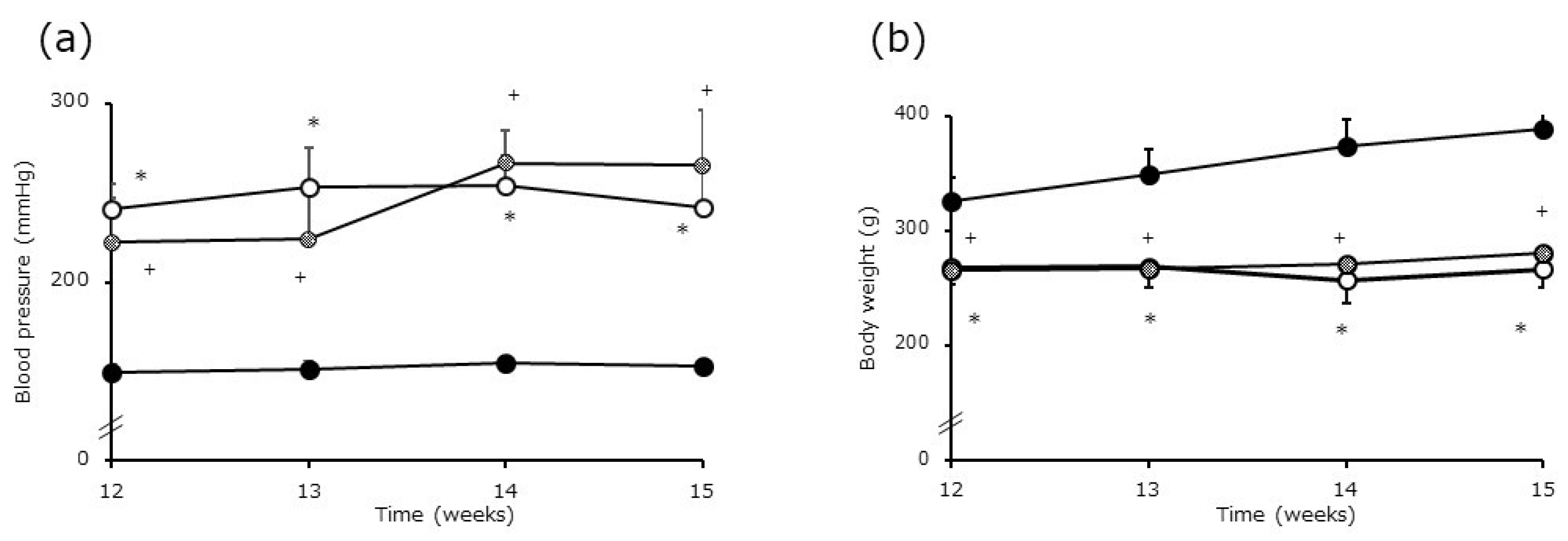
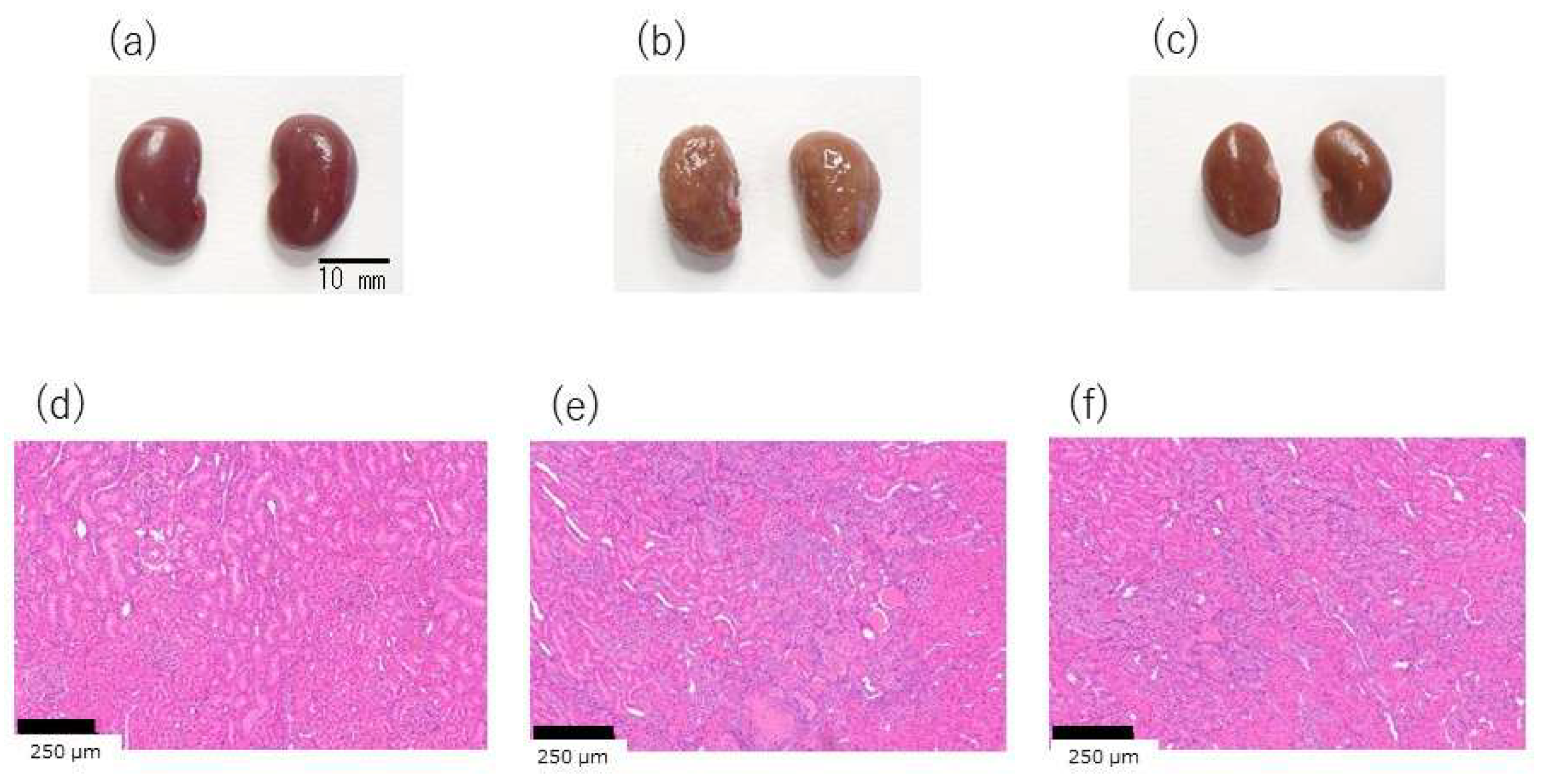
| Organ | WKY | SHRSP Control | SHRSP EP |
| Brain | 1.923±0.041 | 2.048±0.112 | 1.981±0.132 |
| Heart | 1.276±0.042 | 1.351±0.071 | 1.233±0.218 |
| Kidney (right) | 1.410±0.089 | 1.091±0.117** | 1.013±0.155** |
| Kidney (left) | 1.351±0.122 | 1.118±0.079** | 1.029±0.171** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




