Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Chloroplasts are the primary sites of photosynthesis, but growing evidence highlights their broader role as central hubs that coordinate plant responses to environmental challenges. They retain a semi-autonomous genetic system and communicate extensively with the nucleus through anterograde and retrograde signalling pathways, enabling coordinated cellular regulation. Beyond energy conversion, chloroplasts host key biosynthetic pathways and dynamically adjust their metabolic and redox states in response to developmental and environmental cues. This review summarizes current knowledge of chloroplast functions in responses to abiotic and biotic stress, emphasizing their contribution to plant resilience, productivity and sustainability. Under abiotic stress, chloroplasts undergo structural, metabolic and redox reprogramming to maintain photosynthetic efficiency and metabolic homeostasis. During biotic stress, they act as a powerful signalling platforms that integrate immune responses with metabolic and redox regulation. These functions rely on overlapping signalling pathways that are differentially tuned to support acclimation or defence. By coordinating stress responses with photosynthetic activity and metabolic efficiency, chloroplasts play a central role in sustaining plant productivity and represent promising targets to enhance crop resilience and agricultural sustainability under climate change and increasing pathogen pressure.
Keywords:
1. Introduction
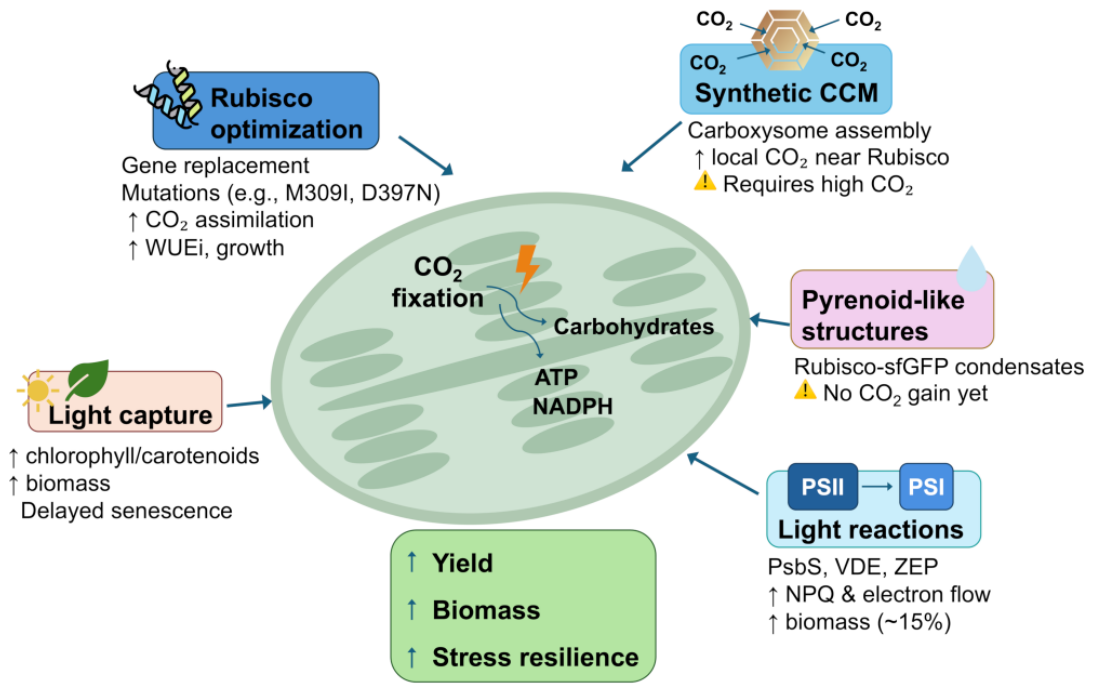
2. Photosynthesis
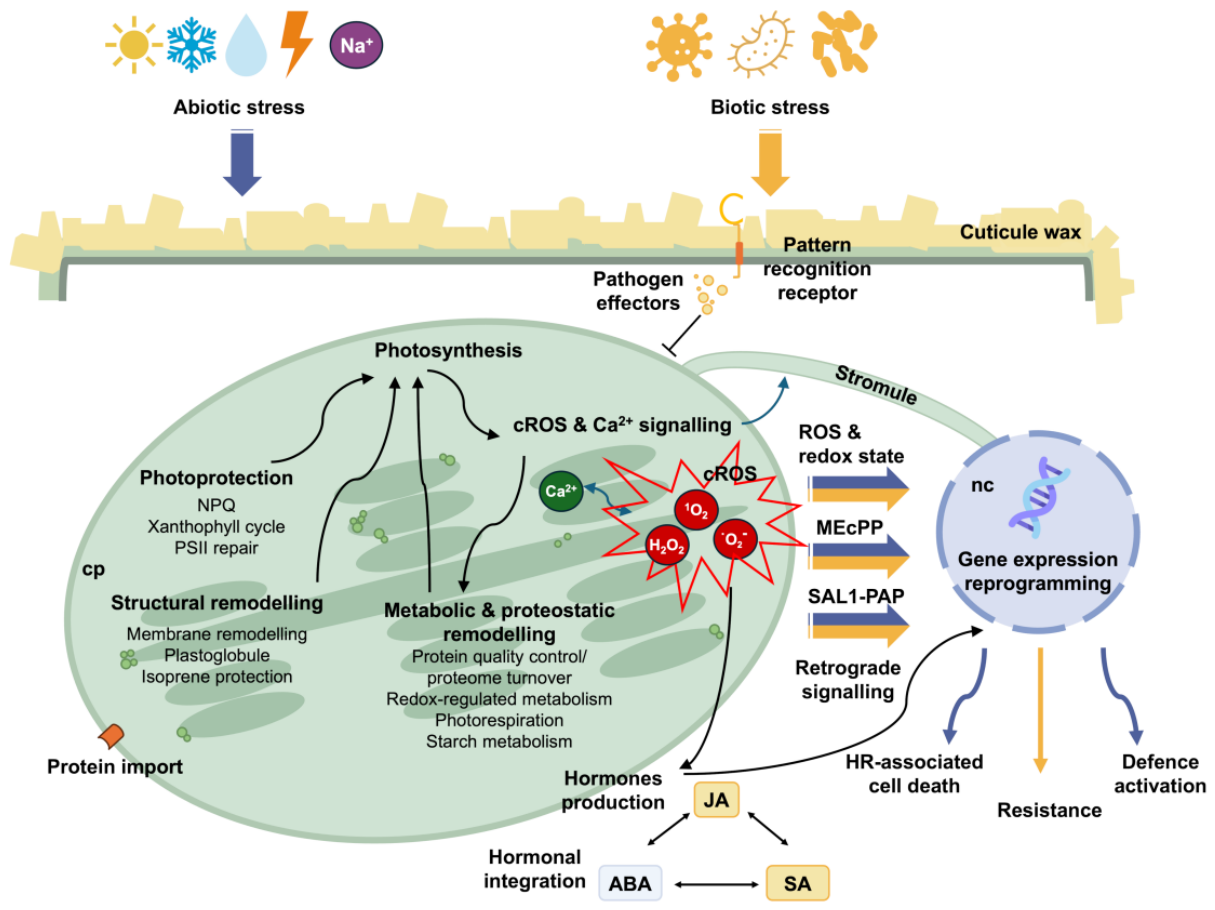
3. Abiotic Stress
4. Biotic Stress
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scotti, N.; Bellucci, M.; Cardi, T. The Chloroplasts as Platform for Recombinant Proteins Production. In Translation in Mitochondria and Other Organelles; Duchêne, A.-M., Ed.; Springer: Berlin, Heidelberg, 2013; pp. 225–262. ISBN 978-3-642-39426-3. [Google Scholar]
- Scotti, N.; Rigano, M.M.; Cardi, T. Production of Foreign Proteins Using Plastid Transformation. Biotechnol. Adv. 2012, 30, 387–397. [Google Scholar] [CrossRef]
- Bali, S.; Mohapatra, S.; Michael, R.; Arora, R.; Dogra, V. Plastidial Metabolites and Retrograde Signaling: A Case Study of MEP Pathway Intermediate MEcPP That Orchestrates Plant Growth and Stress Responses. Plant Physiol. Biochem. 2025, 222, 109747. [Google Scholar] [CrossRef]
- Jarvis, P.; López-Juez, E. Biogenesis and Homeostasis of Chloroplasts and Other Plastids. Nat. Rev. Mol. Cell. Biol. 2013, 14, 787–802. [Google Scholar] [CrossRef] [PubMed]
- Liebers, M.; Cozzi, C.; Uecker, F.; Chambon, L.; Blanvillain, R.; Pfannschmidt, T. Biogenic Signals from Plastids and Their Role in Chloroplast Development. J. Exp. Bot. 2022, 73, 7105–7125. [Google Scholar] [CrossRef]
- Pfannschmidt, T.; Terry, M.J.; Van Aken, O.; Quiros, P.M. Retrograde Signals from Endosymbiotic Organelles: A Common Control Principle in Eukaryotic Cells. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2020, 375, 20190396. [Google Scholar] [CrossRef]
- Renna, L.; Papini, A.; Mancuso, S.; Brandizzi, F.; Stefano, G. Plant Plastids: From Evolutionary Origins to Functional Specialization and Organelle Interactions. J. Exp. Bot. 2026, 77, 63–85. [Google Scholar] [CrossRef]
- Sierra, J.; Escobar-Tovar, L.; Leon, P. Plastids: Diving into Their Diversity, Their Functions, and Their Role in Plant Development. J. Exp. Bot. 2023, 74, 2508–2526. [Google Scholar] [CrossRef] [PubMed]
- Tiller, N.; Bock, R. The Translational Apparatus of Plastids and Its Role in Plant Development. Mol. Plant 2014, 7, 1105–1120. [Google Scholar] [CrossRef]
- Jaswal, S.; Srivastava, A.K.; Ballal, A.; Sandur, S.K. Chloroplast Engineering for Enhancing Photosynthetic Efficiency and Agronomic Traits. Trends Biotechnol. 2025. [Google Scholar] [CrossRef]
- Kamran, M.; Burdiak, P.; Karpiński, S. Crosstalk Between Abiotic and Biotic Stresses Responses and the Role of Chloroplast Retrograde Signaling in the Cross-Tolerance Phenomena in Plants. Cells 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, A.; Li, X.; Lu, C. The Role of Chloroplast Gene Expression in Plant Responses to Environmental Stress. IJMS 2020, 21, 6082. [Google Scholar] [CrossRef] [PubMed]
- Cornic, G.; Fresneau, C. Photosynthetic Carbon Reduction and Carbon Oxidation Cycles Are the Main Electron Sinks for Photosystem II Activity During a Mild Drought. Ann. Bot. 2002, 89, 887–894. [Google Scholar] [CrossRef]
- Gao, Y.; Li, X.; Han, C.; Huang, Q.; Wu, R.; Zhao, C.; She, K. Photosystem Vulnerabilities under Compound Abiotic Stresses: Mechanisms, Diagnostics, and Engineering for Resilient Crops. Plant Stress 2026, 19, 101193. [Google Scholar] [CrossRef]
- Saini, S.; Kaur, N.; Pati, P.K. Phytohormones: Key Players in the Modulation of Heavy Metal Stress Tolerance in Plants. Ecotoxicol. Environ. Saf. 2021, 223, 112578. [Google Scholar] [CrossRef] [PubMed]
- Croce, R.; Carmo-Silva, E.; Cho, Y.B.; Ermakova, M.; Harbinson, J.; Lawson, T.; McCormick, A.J.; Niyogi, K.K.; Ort, D.R.; Patel-Tupper, D.; et al. Perspectives on Improving Photosynthesis to Increase Crop Yield. Plant Cell. 2024, 36, 3944–3973. [Google Scholar] [CrossRef]
- Pesaresi, P.; Bono, P.; Corn, S.; Crosatti, C.; Daniotti, S.; Jensen, J.D.; Frébort, I.; Groli, E.; Halpin, C.; Hansson, M.; et al. Boosting Photosynthesis Opens New Opportunities for Agriculture Sustainability and Circular Economy: The BEST-CROP Research and Innovation Action. Plant J. 2025, 121, e17264. [Google Scholar] [CrossRef]
- Long, S.P.; Zhu, X.-G.; Naidu, S.L.; Ort, D.R. Can Improvement in Photosynthesis Increase Crop Yields? Plant Cell. Environ. 2006, 29, 315–330. [Google Scholar] [CrossRef]
- Alonso, H.; Blayney, M.J.; Beck, J.L.; Whitney, S.M. Substrate-Induced Assembly of Methanococcoides Burtoniid-Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase Dimers into Decamers*. J. Biol. Chem. 2009, 284, 33876–33882. [Google Scholar] [CrossRef]
- Kanevski, I.; Maliga, P.; Rhoades, D.F.; Gutteridge, S. Plastome Engineering of Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase in Tobacco to Form a Sunflower Large Subunit and Tobacco Small Subunit Hybrid1. Plant Physiol. 1999, 119, 133–142. [Google Scholar] [CrossRef]
- Whitney, S.M.; Sharwood, R.E.; Orr, D.; White, S.J.; Alonso, H.; Galmés, J. Isoleucine 309 Acts as a C4 Catalytic Switch That Increases Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase (Rubisco) Carboxylation Rate in Flaveria. PNAS 2011, 108, 14688–14693. [Google Scholar] [CrossRef]
- Whitney, S.M.; Kane, H.J.; Houtz, R.L.; Sharwood, R.E. Rubisco Oligomers Composed of Linked Small and Large Subunits Assemble in Tobacco Plastids and Have Higher Affinities for CO2 and O2. Plant Physiol. 2009, 149, 1887–1895. [Google Scholar] [CrossRef]
- Andrews, T.; Whitney, S.M. Manipulating Ribulose Bisphosphate Carboxylase/Oxygenase in the Chloroplasts of Higher Plants. Arch. Biochem. Biophys. 2003, 414, 159–169. [Google Scholar] [CrossRef]
- Zhang, X.-H.; Webb, J.; Huang, Y.-H.; Lin, L.; Tang, R.-S.; Liu, A. Hybrid Rubisco of Tomato Large Subunits and Tobacco Small Subunits Is Functional in Tobacco Plants. Plant Sci. 2011, 180, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Yamori, W.; Nakazato, I.; Yuchen, Q.; Sanga, Y.; Miyata, T.; Uehara, R.; Noto, Y.; Namba, K.; Fukayama, H.; Matsumura, H.; et al. Chloroplast Genome Editing of Rubisco Boosts Photosynthesis and Plant Growth 2025, 2025.01.02.631008.
- Chen, L.; Rodriguez-Heredia, M.; Hanke, G.T.; Ruban, A.V. Distinct Features of PsbS Essential for Mediating Plant Photoprotection. Plant Commun. 2025, 6, 101179. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhang, S.; Du, K.; Kang, X. Genome-Wide Identification, Characterization, and Expression Analysis of CCT Transcription Factors in Poplar. Plant Physiol. Biochem. 2023, 204, 108101. [Google Scholar] [CrossRef]
- De Souza, A.P.; Burgess, S.J.; Doran, L.; Hansen, J.; Manukyan, L.; Maryn, N.; Gotarkar, D.; Leonelli, L.; Niyogi, K.K.; Long, S.P. Soybean Photosynthesis and Crop Yield Are Improved by Accelerating Recovery from Photoprotection. Science 2022, 377, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Feyissa, B.A.; de Becker, E.M.; Salesse-Smith, C.E.; Shu, M.; Zhang, J.; Yates, T.B.; Xie, M.; De, K.; Gotarkar, D.; Chen, M.S.S.; et al. An Orphan Gene BOOSTER Enhances Photosynthetic Efficiency and Plant Productivity. Dev. Cell. 2025, 60, 723–734.e7. [Google Scholar] [CrossRef]
- Kromdijk, J.; Głowacka, K.; Leonelli, L.; Gabilly, S.T.; Iwai, M.; Niyogi, K.K.; Long, S.P. Improving Photosynthesis and Crop Productivity by Accelerating Recovery from Photoprotection. Science 2016, 354, 857–861. [Google Scholar] [CrossRef]
- Wang, J.; Shan, Q.; Yuan, Q.; Pan, L.; Wang, M.; Zhao, P.; Yu, F.; Dai, L.; Xie, L.; Wang, Z.; et al. The Transcription Factor CaBBX10 Promotes Chlorophyll and Carotenoid Pigment Accumulation in Capsicum Annuum Fruit. Plant Physiol. 2025, 197, kiae592. [Google Scholar] [CrossRef]
- Wei, B.; Liu, Y.; Lyu, M.; Chen, R.; Ma, J.; Yin, C.; Huang, J.; Peng, L.; Fu, Z.; Song, Q.; et al. Natural Variation in CHELATASE SUBUNIT I-A Increases Grain Weight and Enhances Wheat Yield. Plant Cell. 2025, 37, koaf212. [Google Scholar] [CrossRef]
- Simkin, A.J.; McAusland, L.; Lawson, T.; Raines, C.A. Overexpression of the RieskeFeS Protein Increases Electron Transport Rates and Biomass Yield. Plant Physiol. 2017, 175, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Crosatti, C.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Cattivelli, L. Harden the Chloroplast to Protect the Plant. Physiol. Plant. 2013, 147, 55–63. [Google Scholar] [CrossRef]
- Lenzoni, G.; Knight, M.R. Increases in Absolute Temperature Stimulate Free Calcium Concentration Elevations in the Chloroplast. Plant Cell. Physiol. 2019, 60, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Sohail; Zaman, S.; Li, G.; Fu, M. Adaptive Responses of Plants to Light Stress: Mechanisms of Photoprotection and Acclimation. A Review. Front. Plant Sci. 2025, 16. [Google Scholar] [CrossRef]
- Razi, K.; Muneer, S. Drought Stress-Induced Physiological Mechanisms, Signaling Pathways and Molecular Response of Chloroplasts in Common Vegetable Crops. Cr. Rev. Biotechnol. 2021, 41, 669–691. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Z.; Sui, N. Sensitivity and Responses of Chloroplasts to Salt Stress in Plants. Front. Plant Sci. 2024, 15. [Google Scholar] [CrossRef]
- Li, J.; Liu, L.-N.; Meng, Q.; Fan, H.; Sui, N. The Roles of Chloroplast Membrane Lipids in Abiotic Stress Responses. Plant Signal. Behav. 2020, 15, 1807152. [Google Scholar] [CrossRef]
- Qiao, M.; Hong, C.; Jiao, Y.; Hou, S.; Gao, H. Impacts of Drought on Photosynthesis in Major Food Crops and the Related Mechanisms of Plant Responses to Drought. Plants 2024, 13, 1808. [Google Scholar] [CrossRef]
- Zhao, W.; Chen, X.; Wang, J.; Cheng, Z.; Ma, X.; Zheng, Q.; Xu, Z.; Zhang, F. Emerging Mechanisms of Plant Responses to Abiotic Stress. Plants 2025, 14, 3445. [Google Scholar] [CrossRef] [PubMed]
- Falcone, D.L.; Ogas, J.P.; Somerville, C.R. Regulation of Membrane Fatty Acid Composition by Temperature in Mutants of Arabidopsis with Alterations in Membrane Lipid Composition. BMC Plant Biol. 2004, 4, 17. [Google Scholar] [CrossRef]
- Upchurch, R.G. Fatty Acid Unsaturation, Mobilization, and Regulation in the Response of Plants to Stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef]
- Ying, S. Get the Ball Rolling: Update and Perspective on the Role of Chloroplast Plastoglobule-Associated Proteins under Abiotic Stress. J. Exp. Bot. 2025, 76, 4735–4744. [Google Scholar] [CrossRef]
- Yamamoto, Y. Quality Control of Photosystem II: The Mechanisms for Avoidance and Tolerance of Light and Heat Stresses Are Closely Linked to Membrane Fluidity of the Thylakoids. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Z.; Weraduwage, S.M.; Huang, T.; Sharkey, T.D. How Volatile Isoprenoids Improve Plant Thermotolerance. Trends Plant Sci. 2025, 30, 1237–1250. [Google Scholar] [CrossRef]
- Hameed, A.; Ahmed, M.Z.; Hussain, T.; Aziz, I.; Ahmad, N.; Gul, B.; Nielsen, B.L. Effects of Salinity Stress on Chloroplast Structure and Function. Cells 2021, 10, 2023. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Hodges, A.K.; Polutchko, S.K.; Adams, W.W. Zeaxanthin and Other Carotenoids: Roles in Abiotic Stress Defense with Implications for Biotic Defense. Plants 2025, 14, 2703. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Tuteja, N. Reactive Oxygen Species and Antioxidant Machinery in Abiotic Stress Tolerance in Crop Plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Wang, Q.-L.; Chen, J.-H.; He, N.-Y.; Guo, F.-Q. Metabolic Reprogramming in Chloroplasts under Heat Stress in Plants. IJMS 2018, 19, 849. [Google Scholar] [CrossRef]
- Shi, Y.; Ke, X.; Yang, X.; Liu, Y.; Hou, X. Plants Response to Light Stress. J. Genet. Genom. 2022, 49, 735–747. [Google Scholar] [CrossRef]
- Foyer, C.H.; Hanke, G. ROS Production and Signalling in Chloroplasts: Cornerstones and Evolving Concepts. Plant J. 2022, 111, 642–661. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Ascorbate and Glutathione: The Heart of the Redox Hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef]
- Li, M.; Kim, C. Chloroplast ROS and Stress Signaling. Plant Commun. 2022, 3, 100264. [Google Scholar] [CrossRef] [PubMed]
- Estavillo, G.M.; Crisp, P.A.; Pornsiriwong, W.; Wirtz, M.; Collinge, D.; Carrie, C.; Giraud, E.; Whelan, J.; David, P.; Javot, H.; et al. Evidence for a SAL1-PAP Chloroplast Retrograde Pathway That Functions in Drought and High Light Signaling in Arabidopsis. Plant Cell. 2011, 23, 3992–4012. [Google Scholar] [CrossRef]
- Tee, E.E.; Fairweather, S.J.; Vo, H.M.; Zhao, C.; Breakspear, A.; Kimura, S.; Carmody, M.; Wrzaczek, M.; Bröer, S.; Faulkner, C.; et al. SAL1-PAP Retrograde Signaling Orchestrates Photosynthetic and Extracellular Reactive Oxygen Species for Stress Responses. Plant J. 2025, 122, e70271. [Google Scholar] [CrossRef] [PubMed]
- Bowerman, A.F.; Moore, M.; Yadav, A.; Zhang, J.; Mortimer, M.D.; Plšková, Z.; Tee, E.E.; Au, E.K.; Collinge, D.P.; Estavillo, G.M.; et al. Modulation of SAL Retrograde Signalling Promotes Yield and Water Productivity Responses in Dynamic Field Environments. New Phytol. 2026, 249, 1219–1233. [Google Scholar] [CrossRef]
- van Veen, E.; Küpers, J.J.; Gommers, C.M.M. Plastids in a Pinch: Coordinating Stress and Developmental Responses Through Retrograde Signalling. Plant Cell. Environ. 2025, 48, 6897–6911. [Google Scholar] [CrossRef]
- Woodson, J.D. Chloroplast Quality Control – Balancing Energy Production and Stress. New Phytol. 2016, 212, 36–41. [Google Scholar] [CrossRef]
- Chan, K.X.; Phua, S.Y.; Crisp, P.; McQuinn, R.; Pogson, B.J. Learning the Languages of the Chloroplast: Retrograde Signaling and Beyond. Annu. Rev. 2016, 67, 25–53. [Google Scholar] [CrossRef]
- Pollastri, S.; Sukiran, N.A.; Jacobs, B.C.I.C.; Knight, M.R. Chloroplast Calcium Signalling Regulates Thermomemory. J. Plant Physiol. 2021, 264, 153470. [Google Scholar] [CrossRef]
- Hochmal, A.K.; Schulze, S.; Trompelt, K.; Hippler, M. Calcium-Dependent Regulation of Photosynthesis. BBA-Bioenerg. 2015, 1847, 993–1003. [Google Scholar] [CrossRef]
- Voss, I.; Sunil, B.; Scheibe, R.; Raghavendra, A.S. Emerging Concept for the Role of Photorespiration as an Important Part of Abiotic Stress Response. Plant Biol. 2013, 15, 713–722. [Google Scholar] [CrossRef]
- Smith, A.M.; Stitt, M. Coordination of Carbon Supply and Plant Growth. Plant Cell. Environ. 2007, 30, 1126–1149. [Google Scholar] [CrossRef] [PubMed]
- Thalmann, M.; Pazmino, D.; Seung, D.; Horrer, D.; Nigro, A.; Meier, T.; Kölling, K.; Pfeifhofer, H.W.; Zeeman, S.C.; Santelia, D. Regulation of Leaf Starch Degradation by Abscisic Acid Is Important for Osmotic Stress Tolerance in Plants. Plant Cell. 2016, 28, 1860–1878. [Google Scholar] [CrossRef]
- Lee, K.; Kang, H. Roles of Organellar RNA-Binding Proteins in Plant Growth, Development, and Abiotic Stress Responses. IJMS 2020, 21, 4548. [Google Scholar] [CrossRef]
- Lee, K.; Lee, H.J.; Kim, D.H.; Jeon, Y.; Pai, H.-S.; Kang, H. A Nuclear-Encoded Chloroplast Protein Harboring a Single CRM Domain Plays an Important Role in the Arabidopsis Growth and Stress Response. BMC Plant Biol. 2014, 14, 98. [Google Scholar] [CrossRef] [PubMed]
- Cottage, A.; Mott, E.K.; Kempster, J.A.; Gray, J.C. The Arabidopsis Plastid-Signalling Mutant Gun1 (Genomes Uncoupled1) Shows Altered Sensitivity to Sucrose and Abscisic Acid and Alterations in Early Seedling Development. J. Exp. Bot. 2010, 61, 3773–3786. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Xu, T.; Lee, K.; Lee, K.H.; Kang, H. A Chloroplast-Localized DEAD-Box RNA helicaseAtRH3 Is Essential for Intron Splicing and Plays an Important Role in the Growth and Stress Response in Arabidopsis Thaliana. Plant Physiol. Biochem. 2014, 82, 309–318. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, H.; Yang, Y.; Fish, T.; Lyi, S.M.; Thannhauser, T.W.; Zhang, L.; Li, L. Plastid Ribosomal Protein S5 Is Involved in Photosynthesis, Plant Development, and Cold Stress Tolerance in Arabidopsis. J. Exp. Bot. 2016, 67, 2731–2744. [Google Scholar] [CrossRef]
- Tan, J.; Tan, Z.; Wu, F.; Sheng, P.; Heng, Y.; Wang, X.; Ren, Y.; Wang, J.; Guo, X.; Zhang, X.; et al. A Novel Chloroplast-Localized Pentatricopeptide Repeat Protein Involved in Splicing Affects Chloroplast Development and Abiotic Stress Response in Rice. Mol. Plant 2014, 7, 1329–1349. [Google Scholar] [CrossRef]
- Watson, S.J.; Sowden, R.G.; Jarvis, P. Abiotic Stress-Induced Chloroplast Proteome Remodelling: A Mechanistic Overview. J. Exp. Bot. 2018, 69, 2773–2781. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Zhang, H.; Cheng, H.; Sowden, R.G.; Cai, W.; Jarvis, R.P.; Ling, Q. Selective Autophagy Regulates Chloroplast Protein Import and Promotes Plant Stress Tolerance. EMBO J. 2023, 42, EMBJ2022112534. [Google Scholar] [CrossRef]
- Thomson, S.M.; Pulido, P.; Jarvis, R.P. Protein Import into Chloroplasts and Its Regulation by the Ubiquitin-Proteasome System. Biochem. Soc. Trans. 2020, 48, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Tamburino, R.; Sannino, L.; Palomba, F.; Russo, E.; Esposito, M.C.; Sacco, A.; Ruocco, M.; Scotti, N. Genotype-Specific Transcriptomic Response to Drought Stress in Potato Cultivars Modulated by Microbial Biostimulants. BMC Plant Biol. 2025, 25, 1777. [Google Scholar] [CrossRef]
- Tamburino, R.; Vitale, M.; Ruggiero, A.; Sassi, M.; Sannino, L.; Arena, S.; Costa, A.; Batelli, G.; Zambrano, N.; Scaloni, A.; et al. Chloroplast Proteome Response to Drought Stress and Recovery in Tomato (Solanum Lycopersicum L.). BMC Plant Biol. 2017, 17, 40. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, G.-Z. Chloroplast ATP-Dependent Metalloprotease FtsH5/VAR1 Confers Cold-Stress Tolerance through Singlet Oxygen and Salicylic Acid Signaling. Plant Commun. 2025, 6, 101353. [Google Scholar] [CrossRef]
- Zhuang, Y.; Wei, M.; Ling, C.; Liu, Y.; Amin, A.K.; Li, P.; Li, P.; Hu, X.; Bao, H.; Huo, H.; et al. EGY3 Mediates Chloroplastic ROS Homeostasis and Promotes Retrograde Signaling in Response to Salt Stress in Arabidopsis. Cell. Rep. 2021, 36, 109384. [Google Scholar] [CrossRef]
- Anee, T.I.; Sewelam, N.A.; Bautista, N.S.; Hirayama, T.; Suzuki, N. Roles of ROS and NO in Plant Responses to Individual and Combined Salt Stress and Waterlogging. Antioxidants 2025, 14, 1455. [Google Scholar] [CrossRef]
- Crawford, T.; Lehotai, N.; Strand, Å. The Role of Retrograde Signals during Plant Stress Responses. J. Exp. Bot. 2018, 69, 2783–2795. [Google Scholar] [CrossRef]
- Wang, S.; Ma, Q.; Li, C.; Zhang, S.; Liu, X. Chloroplast Responses to Drought: Integrative Mechanisms and Mitigation Strategies. IJMS 2025, 26, 11872. [Google Scholar] [CrossRef]
- Rahikainen, M.; Berkowitz, O.; Whelan, J.; Kangasjärvi, S.; Pascual, J. Role of Aconitase in Plant Stress Response and Signaling. Physiol. Plant. 2025, 177, e70128. [Google Scholar] [CrossRef]
- Wang, Q.; Yue, J.; Yan, J. Research Progress on Maintaining Chloroplast Homeostasis under Stress Conditions: A Review. Acta Biochim. Biophys. Sin. 2023, 55, 173–182. [Google Scholar] [CrossRef]
- Dangl, J.L.; Horvath, D.M.; Staskawicz, B.J. Pivoting the Plant Immune System from Dissection to Deployment. Science 2013, 341, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gong, P.; Lu, R.; Lozano-Durán, R.; Zhou, X.; Li, F. Chloroplast Immunity: A Cornerstone of Plant Defense. Mol. Plant 2024, 17, 686–688. [Google Scholar] [CrossRef]
- Dodds, P.N.; Rathjen, J.P. Plant Immunity: Towards an Integrated View of Plant–Pathogen Interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Geng, R.; Li, X.; Huang, J.; Zhou, W. The Chloroplast Singlet Oxygen-Triggered Biosynthesis of Salicylic Acid and Jasmonic Acid Is Mediated by EX1 and GUN1 in Arabidopsis. Plant Cell. Environ. 2024, 47, 2852–2864. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhang, J.; Zheng, X.; Xia, N.; Diao, Z.; Wang, X.; Chen, Z.; Tang, D.; Li, S. OsMPK12 Positively Regulates Rice Blast Resistance via OsEDC4-Mediated Transcriptional Regulation of Immune-Related Genes. Plant Cell. Environ. 2024, 47, 3712–3731. [Google Scholar] [CrossRef]
- Manna, M.; Rengasamy, B.; Sinha, A.K. Revisiting the Role of MAPK Signalling Pathway in Plants and Its Manipulation for Crop Improvement. Plant Cell. Environ. 2023, 46, 2277–2295. [Google Scholar] [CrossRef]
- Littlejohn, G.R.; Breen, S.; Smirnoff, N.; Grant, M. Chloroplast Immunity Illuminated. New Phytol. 2021, 229, 3088–3107. [Google Scholar] [CrossRef]
- Ershova, N.; Sheshukova, E.; Kamarova, K.; Arifulin, E.; Tashlitsky, V.; Serebryakova, M.; Komarova, T. Nicotiana Benthamiana Kunitz Peptidase Inhibitor-like Protein Involved in Chloroplast-to-Nucleus Regulatory Pathway in Plant-Virus Interaction. Front. Plant Sci. 2022, 13. [Google Scholar] [CrossRef]
- Sun, Q.; Hou, S.; Gao, H. Chloroplasts Orchestrate Multilayered Defences Against Plant Pathogens. Plant Cell. Environ. 2025, 48, 8502–8517. [Google Scholar] [CrossRef] [PubMed]
- Rui, L.; Yang, S.-Q.; Zhou, X.-H.; Wang, W. The Important Role of Chloroplasts in Plant Immunity. Plant Commun. 2025, 6, 101420. [Google Scholar] [CrossRef] [PubMed]
- Kachroo, P.; Burch-Smith, T.M.; Grant, M. An Emerging Role for Chloroplasts in Disease and Defense. Annu. Rev. 2021, 59, 423–445. [Google Scholar] [CrossRef] [PubMed]
- Caplan, J.L.; Kumar, A.S.; Park, E.; Padmanabhan, M.S.; Hoban, K.; Modla, S.; Czymmek, K.; Dinesh-Kumar, S.P. Chloroplast Stromules Function during Innate Immunity. Dev. Cell. 2015, 34, 45–57. [Google Scholar] [CrossRef]
- Meier, N.D.; Seward, K.; Caplan, J.L.; Dinesh-Kumar, S.P. Calponin Homology Domain Containing Kinesin, KIS1, Regulates Chloroplast Stromule Formation and Immunity. Sci. Adv. 2023, 9, eadi7407. [Google Scholar] [CrossRef]
- Zurbriggen, M.D.; Carrillo, N.; Tognetti, V.B.; Melzer, M.; Peisker, M.; Hause, B.; Hajirezaei, M.-R. Chloroplast-Generated Reactive Oxygen Species Play a Major Role in Localized Cell Death during the Non-Host Interaction between Tobacco and Xanthomonas Campestris Pv. Vesicatoria. Plant J. 2009, 60, 962–973. [Google Scholar] [CrossRef]
- Göhre, V.; Jones, A.M.E.; Sklenář, J.; Robatzek, S.; Weber, A.P.M. Molecular Crosstalk Between PAMP-Triggered Immunity and Photosynthesis. MPMI 2012, 25, 1083–1092. [Google Scholar] [CrossRef]
- Nomura, H.; Komori, T.; Uemura, S.; Kanda, Y.; Shimotani, K.; Nakai, K.; Furuichi, T.; Takebayashi, K.; Sugimoto, T.; Sano, S.; et al. Chloroplast-Mediated Activation of Plant Immune Signalling in Arabidopsis. Nat. Commun. 2012, 3, 926. [Google Scholar] [CrossRef]
- Mignolet-Spruyt, L.; Xu, E.; Idänheimo, N.; Hoeberichts, F.A.; Mühlenbock, P.; Brosché, M.; Van Breusegem, F.; Kangasjärvi, J. Spreading the News: Subcellular and Organellar Reactive Oxygen Species Production and Signalling. J. Exp. Bot. 2016, 67, 3831–3844. [Google Scholar] [CrossRef]
- Kuźniak, E.; Kopczewski, T. The Chloroplast Reactive Oxygen Species-Redox System in Plant Immunity and Disease. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Exposito-Rodriguez, M.; Laissue, P.P.; Yvon-Durocher, G.; Smirnoff, N.; Mullineaux, P.M. Photosynthesis-Dependent H2O2 Transfer from Chloroplasts to Nuclei Provides a High-Light Signalling Mechanism. Nat. Commun. 2017, 8, 49. [Google Scholar] [CrossRef]
- Robert-Seilaniantz, A.; Grant, M.; Jones, J.D.G. Hormone Crosstalk in Plant Disease and Defense: More Than Just JASMONATE-SALICYLATE Antagonism. Annu. Rev. 2011, 49, 317–343. [Google Scholar] [CrossRef] [PubMed]
- Ishiga, Y.; Watanabe, M.; Ishiga, T.; Tohge, T.; Matsuura, T.; Ikeda, Y.; Hoefgen, R.; Fernie, A.R.; Mysore, K.S. The SAL-PAP Chloroplast Retrograde Pathway Contributes to Plant Immunity by Regulating Glucosinolate Pathway and Phytohormone Signaling. MPMI 2017, 30, 829–841. [Google Scholar] [CrossRef] [PubMed]
- Stael, S.; Kmiecik, P.; Willems, P.; Van Der Kelen, K.; Coll, N.S.; Teige, M.; Van Breusegem, F. Plant Innate Immunity – Sunny Side Up? Trends Plant Sci. 2015, 20, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Gassmann, W. Cytoplasmic Regulation of Chloroplast ROS Accumulation during Effector-Triggered Immunity. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Cheaib, A.; Killiny, N. Photosynthesis Responses to the Infection with Plant Pathogens. Mol. Plant Microbe Interact. 2025, 38, 9–29. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhi, T.; Zou, J.; Chen, G. Transcriptome Analysis to Identify Genes Related to Programmed Cell Death Resulted from Manipulating of BnaFAH Ortholog by CRISPR/Cas9 in Brassica Napus. Sci. Rep. 2024, 14, 26389. [Google Scholar] [CrossRef]
- Pierella Karlusich, J.J.; Zurbriggen, M.D.; Shahinnia, F.; Sonnewald, S.; Sonnewald, U.; Hosseini, S.A.; Hajirezaei, M.-R.; Carrillo, N. Chloroplast Redox Status Modulates Genome-Wide Plant Responses during the Non-Host Interaction of Tobacco with the Hemibiotrophic Bacterium Xanthomonas Campestris Pv. Vesicatoria. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef]
- Arce, R.C.; Demarchi, M.; Figueroa, N.; Delprato, M.L.; Hajirezaei, M.-R.; Mayta, M.L.; Lodeyro, A.F.; Krapp, A.R.; Carrillo, N. Light and Chloroplast Redox State Modulate the Progression of Tobacco Leaf Infection by Pseudomonas Syringae Pv Tabaci. Plant Sci. 2025, 356, 112512. [Google Scholar] [CrossRef]
- Liu, P.; Myo, T.; Ma, W.; Lan, D.; Qi, T.; Guo, J.; Song, P.; Guo, J.; Kang, Z. TaTypA, a Ribosome-Binding GTPase Protein, Positively Regulates Wheat Resistance to the Stripe Rust Fungus. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Jan, M.; Liu, Z.; Rochaix, J.-D.; Sun, X. Retrograde and Anterograde Signaling in the Crosstalk between Chloroplast and Nucleus. Front. Plant Sci. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Walker, B.J.; He, S.Y.; Hu, J. The Role of Photorespiration in Plant Immunity. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Ciereszko, I.; Kuźniak, E. Photorespiratory Metabolism and Its Regulatory Links to Plant Defence Against Pathogens. IJMS 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Serrano, I.; Audran, C.; Rivas, S. Chloroplasts at Work during Plant Innate Immunity. J. Exp. Bot. 2016, 67, 3845–3854. [Google Scholar] [CrossRef]
- Mène-Saffrané, L.; Dubugnon, L.; Chételat, A.; Stolz, S.; Gouhier-Darimont, C.; Farmer, E.E. Nonenzymatic Oxidation of Trienoic Fatty Acids Contributes to Reactive Oxygen Species Management in Arabidopsis*. J. Biol. Chem. 2009, 284, 1702–1708. [Google Scholar] [CrossRef]
- Camejo, D.; Guzmán-Cedeño, Á.; Moreno, A. Reactive Oxygen Species, Essential Molecules, during Plant–Pathogen Interactions. Plant Physiol. Biochem. 2016, 103, 10–23. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, Y.; Fei, S.; Chen, Y.; Xu, Y.; Zhu, Z.; He, Y. Overexpression of Sly-miR398b Compromises Disease Resistance against Botrytis Cinerea through Regulating ROS Homeostasis and JA-Related Defense Genes in Tomato. Plants 2023, 12. [Google Scholar] [CrossRef]
- Qi, Y.; Wu, J.; Yang, Z.; Li, H.; Liu, L.; Wang, H.; Sun, X.; Wu, X.; Nie, J.; Zhou, J.; et al. Chloroplast Elongation Factors Break the Growth–Immunity Trade-off by Simultaneously Promoting Yield and Defence. Nat. Plan. 2024, 10, 1576–1591. [Google Scholar] [CrossRef]
- Srikakulam, N.; Guria, A.; Karanthamalai, J.; Murugesan, V.; Krishnan, V.; Sundaramoorthy, K.; Saha, S.; Singh, R.; Victorathisayam, T.; Rajapriya, V.; et al. An Insight Into Pentatricopeptide-Mediated Chloroplast Necrosis via microRNA395a During Rhizoctonia Solani Infection. Front. Genet. 2022, 13. [Google Scholar] [CrossRef]
- Mwaba, I.; Rey, M.E.C. Nitric Oxide Associated Protein 1 Is Associated with Chloroplast Perturbation and Disease Symptoms in Nicotiana Benthamiana Infected with South African Cassava Mosaic Virus. Virus Res. 2017, 238, 75–83. [Google Scholar] [CrossRef]
- Hossain, R.; Willems, G.; Wynant, N.; Borgolte, S.; Govaerts, K.; Varrelmann, M. Aphid-Mediated Beet Yellows Virus Transmission Initiates Proviral Gene Deregulation in Sugar Beet at Early Stages of Infection. PLoS ONE 2024, 19, e0311368. [Google Scholar] [CrossRef]
- de Torres Zabala, M.; Littlejohn, G.; Jayaraman, S.; Studholme, D.; Bailey, T.; Lawson, T.; Tillich, M.; Licht, D.; Bölter, B.; Delfino, L.; et al. Chloroplasts Play a Central Role in Plant Defence and Are Targeted by Pathogen Effectors. Nat. Plan. 2015, 1, 15074. [Google Scholar] [CrossRef]
- Li, Z.; Li, C.; Fu, S.; Liu, Y.; Xu, Y.; Wu, J.; Wang, Y.; Zhou, X. NSvc4 Encoded by Rice Stripe Virus Targets Host Chloroplasts to Suppress Chloroplast-Mediated Defense. Viruses 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zheng, L.; Jia, J.; Guo, J.; Zheng, M.; Zhao, J.; Shao, J.; Liu, X.; An, L.; Yu, F.; et al. Chloroplast Translation Elongation Factor EF-Tu/SVR11 Is Involved in Var2-Mediated Leaf Variegation and Leaf Development in Arabidopsis. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Zhang, R.; Wu, Y.; Qu, X.; Yang, W.; Wu, Q.; Huang, L.; Jiang, Q.; Ma, J.; Zhang, Y.; Qi, P.; et al. The RING-Finger Ubiquitin E3 Ligase TaPIR1 Targets TaHRP1 for Degradation to Suppress Chloroplast Function. Nat. Commun. 2024, 15, 6905. [Google Scholar] [CrossRef]
- Garavaglia, B.S.; Thomas, L.; Gottig, N.; Dunger, G.; Garofalo, C.G.; Daurelio, L.D.; Ndimba, B.; Orellano, E.G.; Gehring, C.; Ottado, J. A Eukaryotic-Acquired Gene by a Biotrophic Phytopathogen Allows Prolonged Survival on the Host by Counteracting the Shut-Down of Plant Photosynthesis. PLoS ONE 2010, 5, e8950. [Google Scholar] [CrossRef] [PubMed]
- Rui, L.; Cong, Z.; Zhou, X.; Yang, Q.; Wang, Z.; Wang, W. To Win the Battle: The Chloroplast Is a Key Battleground in Plant–Pathogen Interactions. Hortic. Res. 2026, 13, uhaf294. [Google Scholar] [CrossRef] [PubMed]
| Abiotic stress | Species studied | Chloroplast function/structure involved | Main chloroplast processes/ signals | Type of signal | Reference |
|---|---|---|---|---|---|
| Salt stress | Arabidopsis, rice, halophytes | Chloroplast ultrastructure | Thylakoid remodeling, plastoglobules | Lipid remodelling | [47] |
| Salt, high light, heat, drought | Arabidopsis; rice; maize; tomato | Plastoglobules | Lipid metabolism, photoprotection | Lipid remodelling / redox | [44] |
| Cold stress | Arabidopsis | Thylakoid membranes | Fatty acid desaturation | Lipid remodeling | [42] |
| Temperature stress | Arabidopsis | Thylakoid membrane lipids | Fatty acid unsaturation | Lipid remodeling | [43] |
| Heat stress | Populus, Nicotiana tabacum, Quercus spp. | Isoprenoid biosynthesis (MEP pathway) | Isoprene/monoterpenes, membrane stabilization, ROS reduction | Lipid/membrane stabilization | [46] |
| Heat / light stress | Arabidopsis | Thylakoid membrane fluidity | PSII protection and repair | Membrane stability | [45] |
| High light / fluctuating light | Arabidopsis thaliana | Thylakoids | NPQ, PSII repair, ROS generation and signalling | ROS/ photoprotection | [51] |
| High light | Arabidopsis thaliana | Pigment metabolism | Xanthophyll cycle, carotenoids, | Photoprotection | [36,48] |
| Cold stress | Arabidopsis thaliana | Thylakoid quality control | FtsH protease, PSII repair, ¹O₂ signalling | ROS signalling/ proteostasis | [77] |
| Multiple stresses | Arabidopsis | Chloroplast redox network | ROS-mediated signalling and proteostasis | ROS signalling | [52] |
| Multiple stresses | Arabidopsis | cROS network | ROS-mediated retrograde signalling and proteostasis | ROS signalling | [54] |
| General abiotic stress | Crops | Antioxidant systems | Ascorbate-glutathione cycle | Antioxidant / ROS detox | [53] |
| Salt stress | Arabidopsis | cROS network | H2O2 retrograde signalling | ROS signalling/retrograde | [78] |
| Combined stresses | ROS and NO signalling | Redox signalling networks | ROS/NO signalling | [79] | |
| High light, oxidative stress | Arabidopsis thaliana | Retrograde signalling | ROS, β-cyclocitral, MEcPP, PAP | Retrograde signalling | [80] |
| Drought, high light | Arabidopsis thaliana | Retrograde signalling | SAL1–PAP pathway, ROS/ABA coordination | [55,56] | |
| Drought stress | Chloroplast signalling network | ROS, Ca2+ oscillations, PAP and MEcPP retrograde signal | ROS / retrograde signalling | [81] | |
| Multiple stresses | Arabidopsis | Chloroplast Ca2+ network | CAS-mediated Ca2+ signalling | Ca2+ signalling | [62] |
| Multiple stresses | Arabidopsis | Chloroplast Ca2+ dynamics | Ca2+ oscillations | Ca2+ signalling | [35] |
| Multiple stresses | Arabidopsis | Thylakoid Ca2+ buffering | Ca2+-dependent signalling | Ca2+ signalling | [61] |
| Multiple stresses | Starch metabolism | Starch turnover | Metabolism | [64] | |
| Drought / metabolic stress | Chloroplast metabolism | Photorespiration | Metabolic acclimation | [63] | |
| Multiple stresses | Arabidopsis thaliana, Nicotiana tabacum | Metabolic/redox crosstalk | Aconitase-mediated retrograde signalling | Metabolic/redox signalling | [82] |
| Multiple stresses | Arabidopsis thaliana, crops | Chloroplast proteostasis | Protein import (TOC–TIC), turnover, quality control | Proteostasis | [72,83] |
| Heat/ UV-B stress | Arabidopsis | TOC complex | Autophagy-mediated regulation of chloroplast protein import | Proteostasis / protein import | [73] |
| Drought stress | Solanum lycopersicum | Chloroplast proteome | Proteome remodelling, redox enzymes, ABA-linked signalling | Proteostasis | [76] |
| Drought stress | Rice, maize, wheat | Chloroplast-associated metabolic responses | Photosynthesis and photorespiration | Metabolic acclimation | [40] |
| Drought stress | Arabidopsis thaliana, vegetable crops (e.g., bean, sugar beet) | Chloroplast-associated metabolic responses | Photosynthesis inhibition, redox imbalance, ABA integration | Metabolic signalling | [37] |
| Heat stress | Arabidopsis thaliana, rice, wheat, maize, tomato | Chloroplast metabolism | Metabolic reprogramming, ROS production, acclimation | ROS/metabolism | [50] |
| Biotic stress / interaction | Species studied | Chloroplast function/structure involved | Main chloroplast processes / signals | Type of signal / response | Reference |
|---|---|---|---|---|---|
| General immune responses (bacterial, fungal, viral) | Arabidopsis, Nicotiana benthamiana, crops | Chloroplast as immune signalling hub | Integration of PTI/ETI outputs; coordination of redox, hormonal and retrograde signals | Immune integration / retrograde signalling | [102] |
| Early immune activation after PAMP perception | Arabidopsis | Photosynthetic apparatus / chloroplast-associated signalling | PTI-associated perturbation of photosynthesis; immune-linked chloroplast signalling | PTI / photosynthesis crosstalk | [98,123] |
| PTI and ETI | Arabidopsis | Chloroplast Ca²⁺ network (CAS) | Stromal Ca²⁺ signalling; activation of SA biosynthesis genes; transcriptional reprogramming of defence genes | Ca2+ signalling / immune regulation | [99] |
| ETI / avirulent bacterial interactions | Arabidopsis | Chloroplast ROS (cROS) network | cROS accumulation linked to HR-associated programmed cell death | Redox signalling / HR | [97,100] |
| PTI and ETI | Nicotiana benthamiana, Arabidopsis | Stromules and chloroplast repositioning | Stromule induction; perinuclear chloroplast clustering; enhanced chloroplast-nucleus communication | Organelle dynamics / retrograde signalling | [95,96] |
| Pathogen infection / retrograde signalling | Arabidopsis | SAL1–PAP pathway | Regulation of glucosinolate accumulation; modulation of SA- and JA-dependent pathways; contribution to immune competence | Metabolite retrograde signalling / hormone regulation | [104] |
| Biotrophic / hemibiotrophic defence | Arabidopsis and model plants | SA biosynthesis in chloroplasts | Isochorismate pathway; chloroplast-derived SA precursor synthesis; immune hormone integration | Hormonal signalling (SA) | [115] |
| Necrotrophic defence / wound-related immunity | Arabidopsis and crops | Chloroplast fatty acid metabolism | Lipid precursors for JA biosynthesis; oxylipin-mediated defence responses | Hormonal signalling (JA) / lipid signalling | [116] |
| Broad immune modulation during infection | Arabidopsis, crops | Hormone integration in chloroplasts | Coordination of SA, JA and ABA crosstalk during pathogen challenge | Hormone crosstalk / signalling integration | [92,103,117] |
| Photorespiration during immunity | Arabidopsis, multiple pathosystems | Photorespiration | H2O2 production; inter-organellar redox control; interaction with SA/JA signalling | Metabolic signalling / redox regulation | [113,114] |
| Non-host and virulent bacterial interactions | Tobacco | Chloroplast redox buffering | Flavodoxin-dependent modulation of cROS; defence-associated metabolic and transcriptional reprogramming; selective control of HR | Redox signalling / defence cost modulation | [109,110] |
| Stripe rust resistance | Wheat | Chloroplast-localized regulator | TaTypA-dependent cROS accumulation and HR intensity | Redox signalling / HR | [111] |
| Viral infection / chloroplast-to-nucleus signalling | Nicotiana benthamiana | Chloroplast retrograde signalling / PhANG regulation | KPILP-mediated repression of LHCB, HEMA1, RBCS1A; altered carbon partitioning | Retrograde signalling / transcriptional regulation | [91] |
| Viral infection | Nicotiana benthamiana | Chloroplast translation machinery | Repression of NOA1 and plastid translation factors; chloroplast dysfunction and pigment loss | Translation / plastid homeostasis | [121] |
| Viral infection | Sugar beet | Chloroplast-related gene expression | Downregulation of chloroplast-associated genes during early infection | Transcriptional regulation / plastid dysfunction | [122] |
| Oomycete infection | Potato | Plastid translation / elongation factors | Regulation of plastid translation; cROS production; stromule-associated immune competence; effector targeting | Translation / proteostasis / effector targeting | [119] |
| Fungal infection | Rice | Plastid integrity / post-transcriptional regulation | miRNA-mediated regulation of PPR targets linked to chloroplast degradation and susceptibility | RNA regulation / plastid homeostasis | [120] |
| Bacterial infection | Arabidopsis | NECGs1 / PSII | Effector-mediated suppression of photosynthesis; inhibition of cROS burst and CO2 assimilation | Effector targeting / photosynthesis-defence trade-off | [123] |
| Viral infection | Nicotiana benthamiana | Chloroplast-localized viral protein | NSvc4-mediated inhibition of cROS and chloroplast-mediated defence | Effector targeting / ROS suppression | [124] |
| Stripe rust susceptibility | Wheat | PhANG2 regulation / chloroplast function | TaPIR1-mediated suppression of PhANGs; reduced cROS; increased susceptibility | Effector-associated suppression / transcriptional control | [126] |
| Citrus canker | Citrus | Photosynthetic proteins / chloroplast proteome | Pathogen-mediated modulation of photosynthesis; maintenance of host tissue viability | Pathogen manipulation / metabolic reprogramming | [127] |
| General pathogen pressure | Arabidopsis, Nicotiana spp., crops | Chloroplast as effector target | Direct targeting of chloroplast proteins and signalling pathways by pathogen effectors | Effector targeting / immune suppression | [119,123] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




