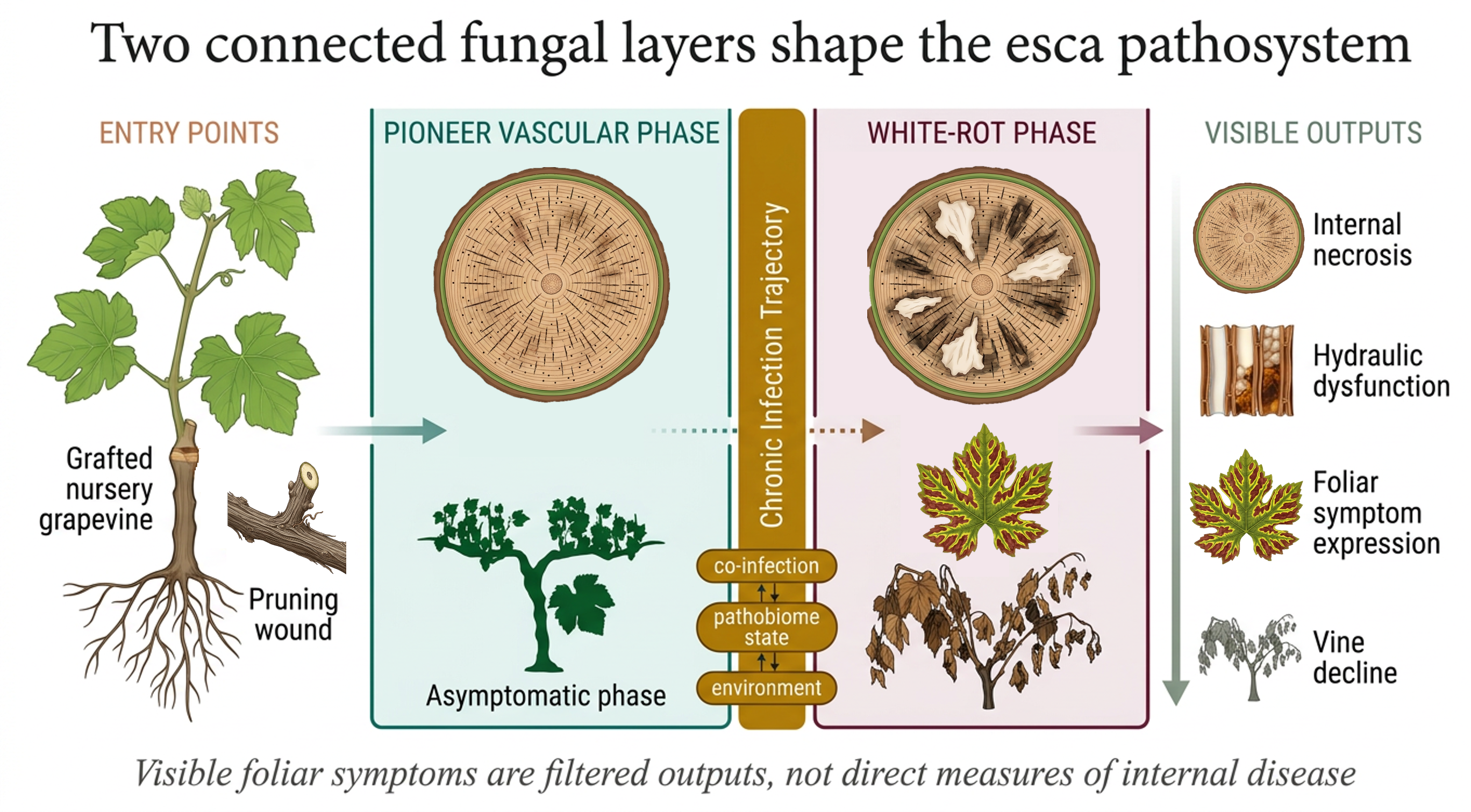
Esca remains one of the most conceptually challenging disease syndromes of grapevine because it cannot be reduced to a single pathogen, lesion type or external phenotype. Foliar symptoms are erratic, internal wood damage is heterogeneous, and fungi associated with the syndrome may persist in asymptomatic vines. Much of this difficulty stems from treating esca-associated fungi as functionally equivalent, even though they occupy different niches and contribute differently to disease development. Here, we revisit grapevine esca through a fungal-biology-centred framework focused on Phaeomoniella chlamydospora, Phaeoacremonium minimumand white-rot basidiomycetes, especially Fomitiporia mediterranea. We argue that the esca pathosystem is best understood as the interaction between two biologically connected but non-equivalent fungal layers: a pioneer vascular phase and a later white-rot phase. Within this perspective, Petri disease and esca proper are interpreted as stages along a continuous host-fungus trajectory shaped by colonization strategy, tissue occupation, decay biology and pathobiome context. This distinction clarifies why host phenotypes are so difficult to interpret. Current evidence supports partially independent layers of host response, including resistance to pioneer colonization, tolerance to hydraulic dysfunction, resistance to wood decay and resilience under chronic infection. We therefore propose that “esca resistance” should be interpreted not as a unitary host trait, but as a multidimensional consequence of fungal functional differentiation, host physiology and environmental modulation.