Submitted:
24 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
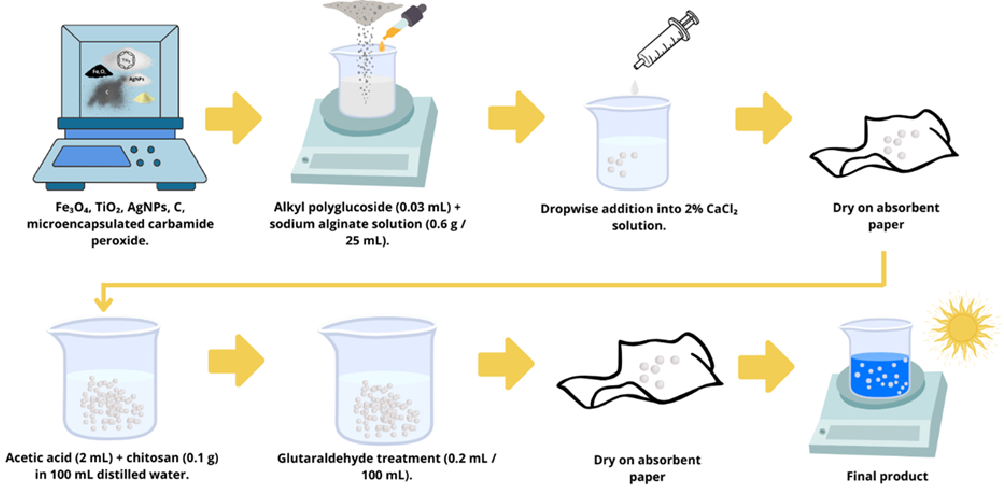
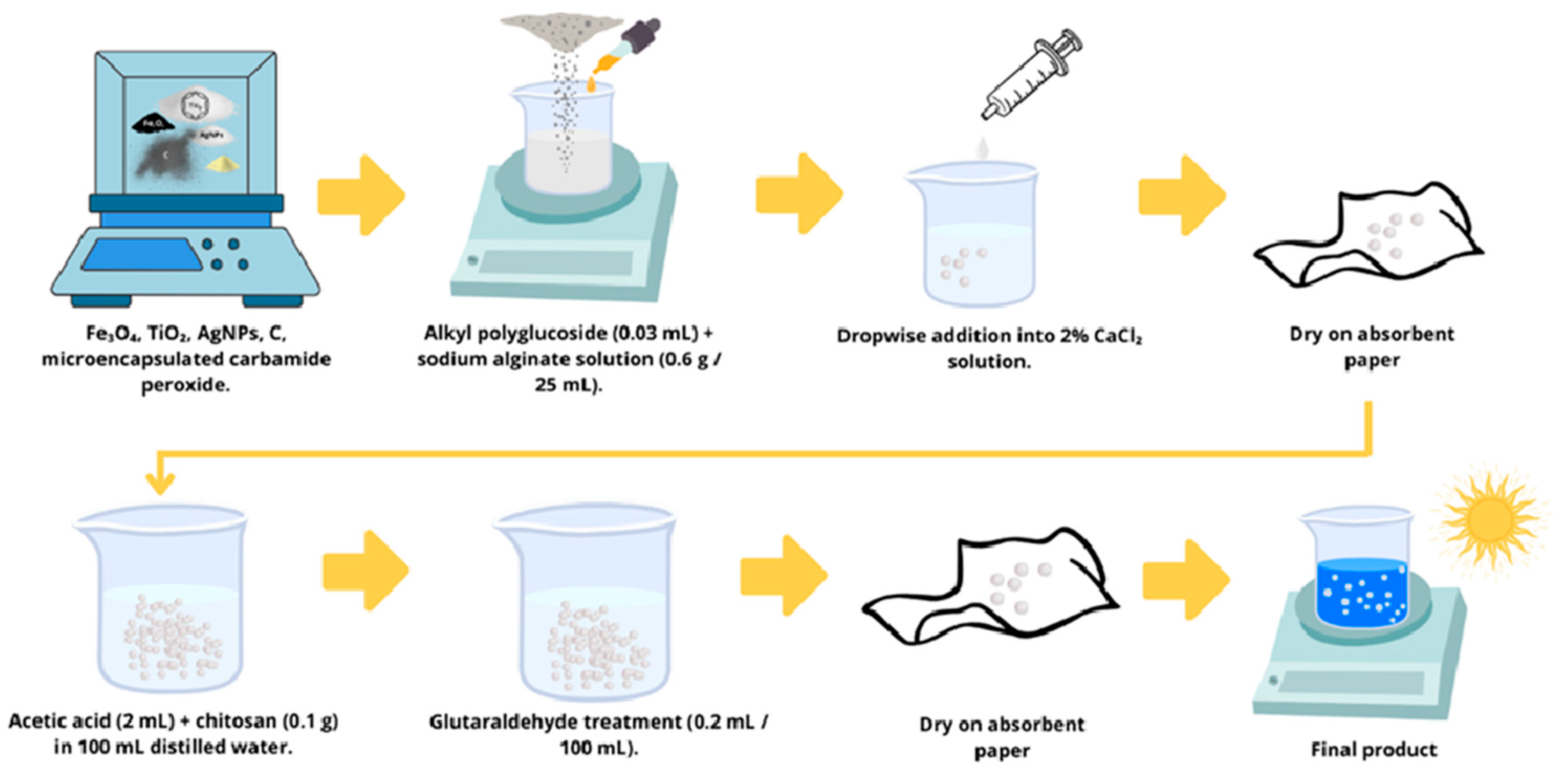
2.1. Spheres Manufacturing Process
2.2. Powder Characterization
2.3. Absorbance Measurements
3. Results
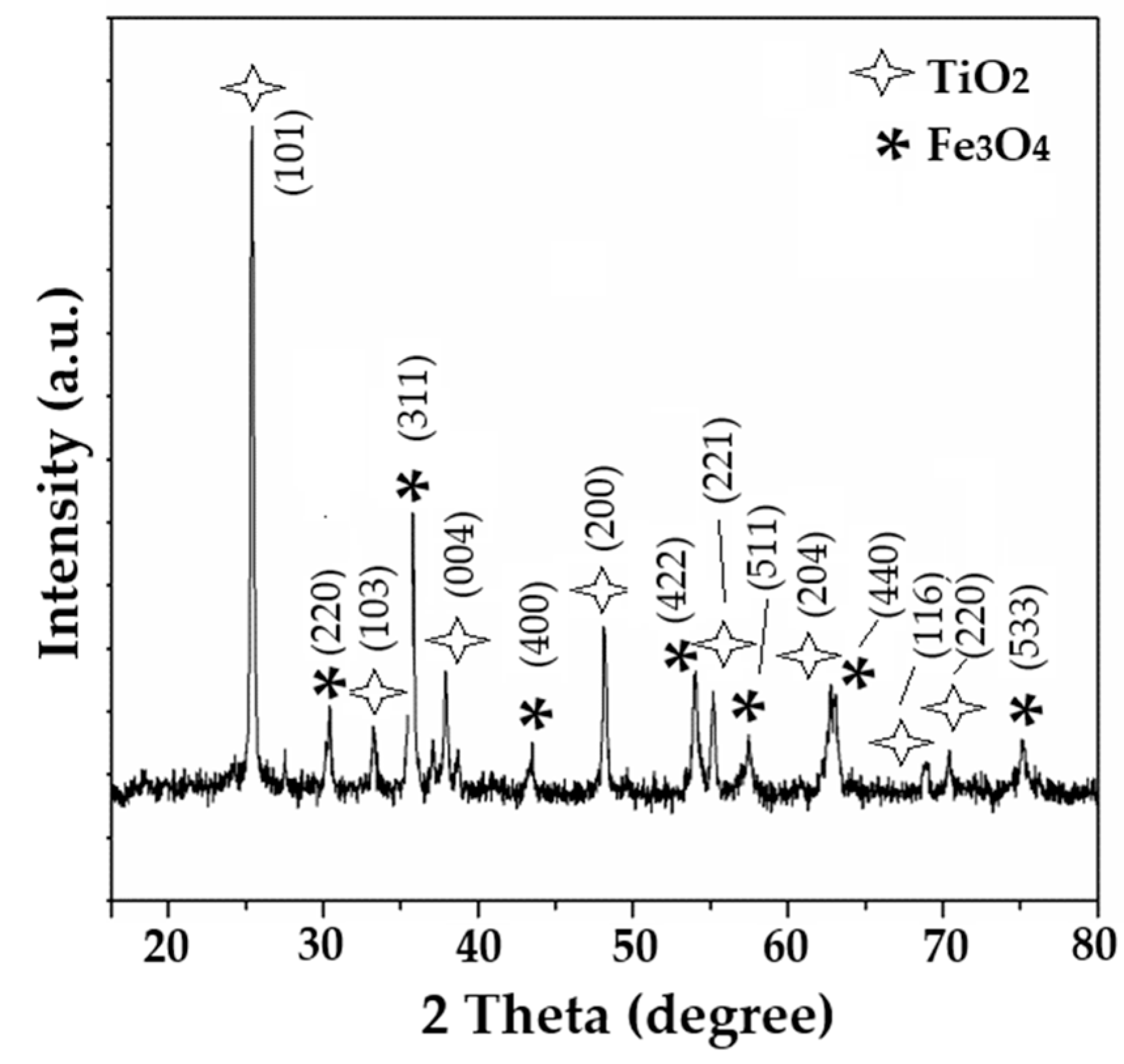
3.1. Structure X-Ray Diffraction
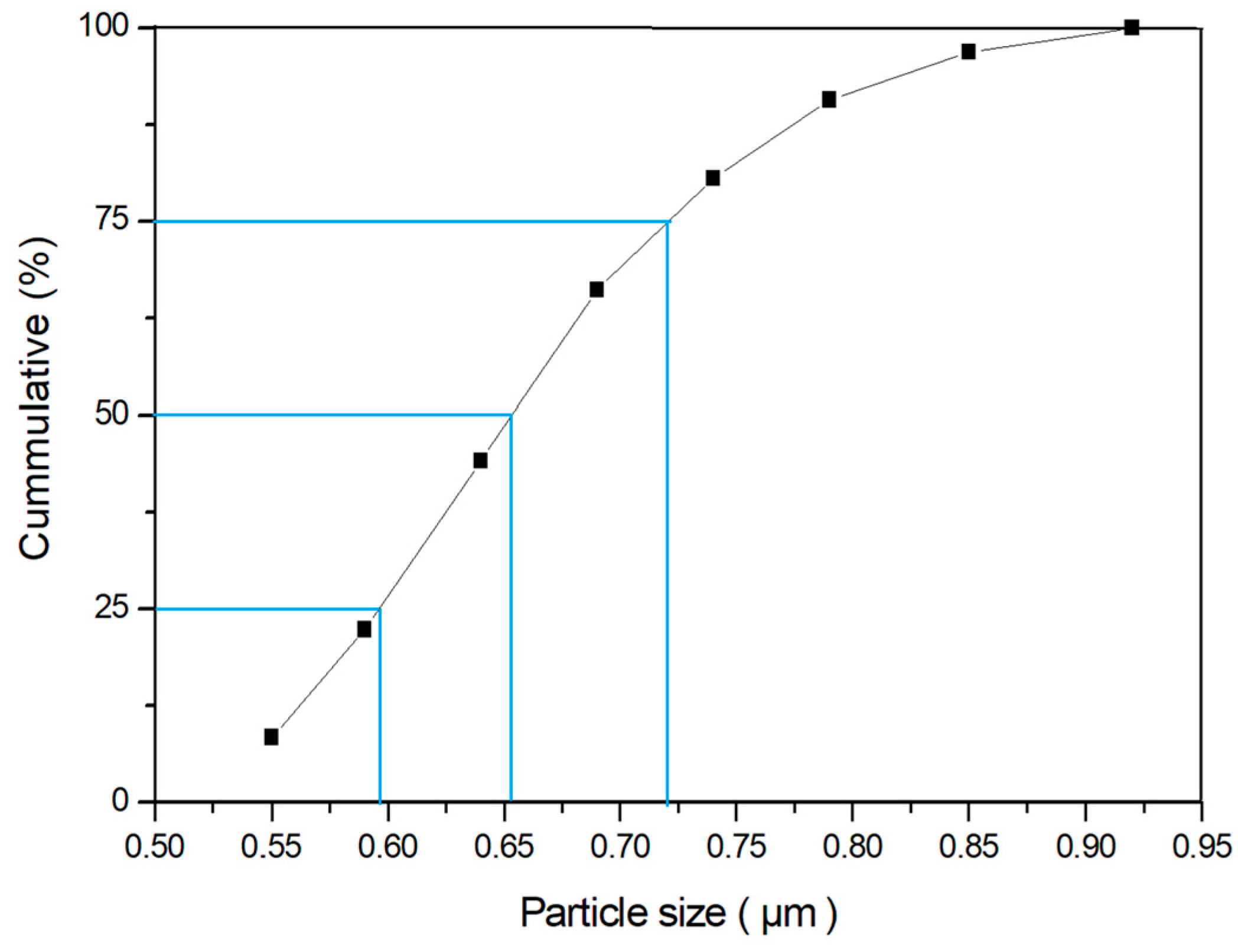
3.2. Particle Size Distribution
3.3. Powders Morphology
3.5. Mappings
3.4. Irradiation System
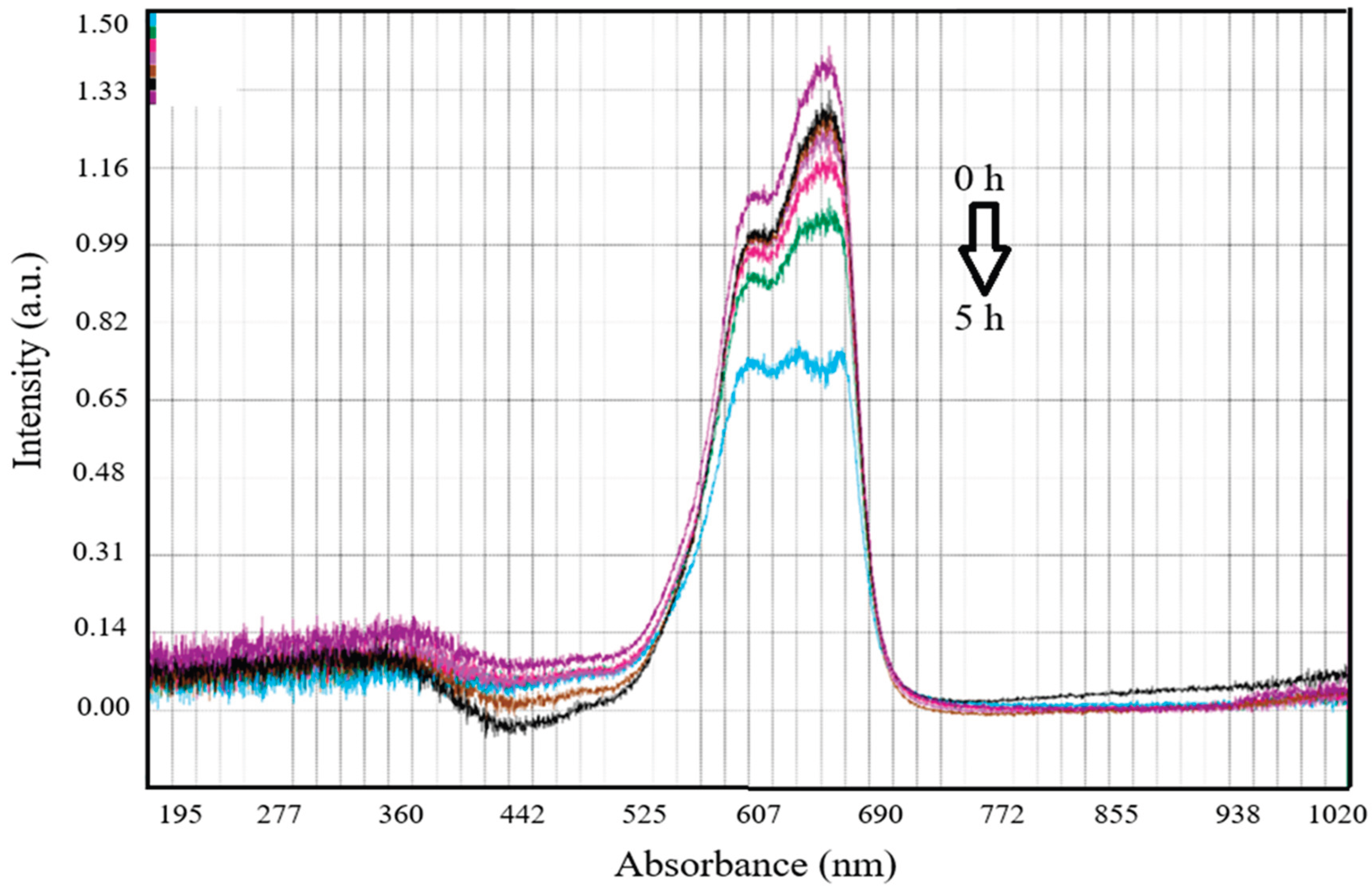
3.5. Photocatalytic Activity
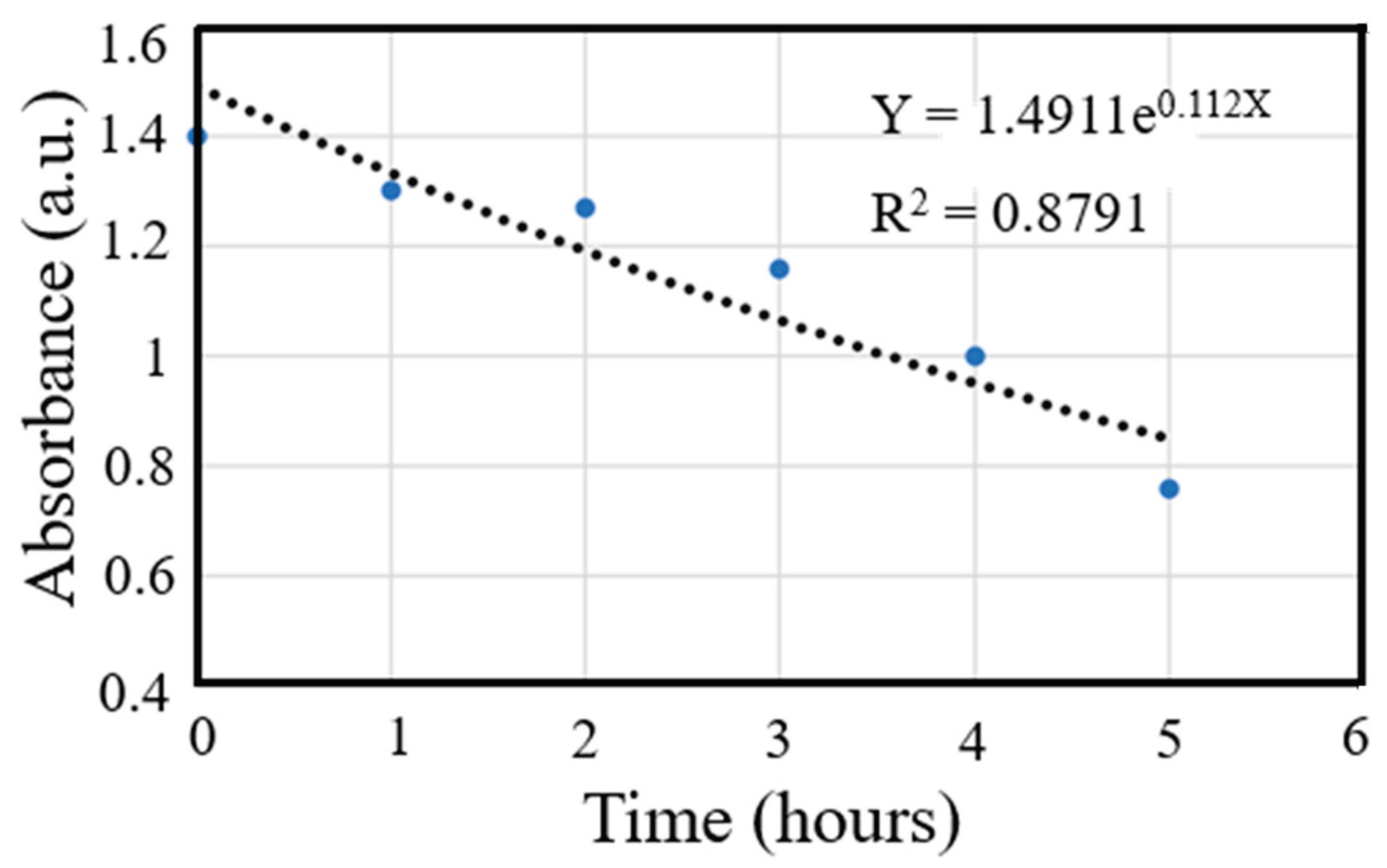
3.6. Absorbance Rate
3.7. Photocatalytic Antibacterial Activity
4. Discussion
5. Conclusions
- Multifunctional spheres containing AgNPs-doped TiO2-Fe3O4 were successfully synthesized through ionic gelation and biopolymer coating, integrating photocatalytic, adsorptive, antimicrobial and magnetic recovery functionalities in a single reusable material with structural stability and environmental safety.
- Structural and morphological analyses confirmed the coexistence of nanocrystalline anatase TiO2 and magnetite phases with finely dispersed Ag nanoparticles and a predominantly submicrometric particle size distribution, providing high surface area and favorable interfacial charge transfer for photocatalytic processes.
- Photocatalytic experiments under simulated solar irradiation demonstrated effective degradation of methylene blue following pseudo-first-order kinetics (kapp ~0.112 h-1), evidencing enhanced visible-light activity and reduced electron-hole recombination due to the synergistic interaction of AgNPs, TiO2 and Fe3O4.
- The spheres exhibited strong antibacterial performance, achieving reductions of 87% in total coliforms and >93% in fecal coliforms after 5 h of irradiation, confirming their dual capability for organic contaminant removal and solar-driven water disinfection.
- The incorporation of magnetite enabled rapid magnetic recovery and reuse of the spheres without functional loss, supporting the feasibility of AgNPs-doped TiO2-Fe3O4 spheres as an efficient, green and low-cost and sustainable platform for solar water treatment applications.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AgNPs | Silver nanoparticles |
| •OH | Hydroxyl radicals |
| O₂•⁻ | superoxide anion |
| SEM | Scanning electron microscopy |
| XRD | X-ray diffraction |
| kapp | Apparent rate constant |
| ROS | Reactive oxygen species |
References
- UNESCO. The United Nations World Water Development Report 2023: Partnerships and Cooperation for Water; UNESCO: Paris, France, 2023. [Google Scholar]
- Hübner, U.; Spahr, S.; Lutze, H.; Wieland, A.; Rüting, S.; Gernjak, W.; Wenk, J. Advanced oxidation processes for water and wastewater treatment – Guidance for systematic future research. Helyon 2024, 10, e30402. [Google Scholar] [CrossRef] [PubMed]
- Chong, M.N.; Jin, B.; Chow, C.W.K.; Saint, C. Current Status and Future Prospects of Photocatalytic Technology for Water Sterilization. Catalysts 2026, 16(1), 40. [Google Scholar] [CrossRef]
- Nematov, D. Titanium Dioxide and Photocatalysis: a Detailed Overview of the Synthesis, Applications, Challenges, Advances and Prospects for Sustainable Development. J. Mod. Green. Energy 2024, 3, 6. [Google Scholar] [CrossRef]
- Gatou, M.A.; Syrrakou, A.; Lagopati, N.; Pavlatou, E.A. Photocatalytic TiO2-Based Nanostructures as a Promising Material for Diverse Environmental Applications: A Review. Reactions 2024, 5(1), 135–194. [Google Scholar] [CrossRef]
- Jabbar, Z.H.; Graimed, B.H.; Ammar, S.H.; Sabit, D.A.; Najim, A.A.; Radeef, A.Y.; Taher, A.G. Environmental The latest progress in the design and application of semiconductor photocatalysis systems for degradation of environmental pollutants in wastewater: Mechanism insight and theoretical calculations. Mat. Sci. Semicond. Proc. 2024, 173(2024), 108153. [Google Scholar] [CrossRef]
- Wang, J.; Tang, J. Fe-based Fenton-like catalysts for water treatment: Catalytic mechanisms and applications. J. Mol. Liq. 2021, 332(416), 115755. [Google Scholar] [CrossRef]
- Liou, Y.; Yang, Z.; Wang, J. Fenton/Fenton-like processes with in-situ production of hydrogen peroxide/hydroxyl radical for degradation of emerging contaminants: Advances and prospects. J. Hazard. Mater. 2021, 404 Pt B, 124191. [Google Scholar] [CrossRef] [PubMed]
- Jacinto, M.; Ferreira, L.F.; Silva, V.C. Magnetic materials for photocatalytic applications-a review. J. Sol.-Gel Sci. Technol. 2020, 96, 4–5. [Google Scholar] [CrossRef]
- Mallah, S.; Mchaouri, M.E.; Boumya, W.; Machrouhi, A.; Elmoubarki, R.; Barka, N.; Elhalil, A. Recent advances and modification strategies of TiO2 semiconductors for the photocatalytic remediation of water-containing organic pollutants. Euro-Mediterr. J. Environ. Integr. 2025, 10, 4849–4884. [Google Scholar] [CrossRef]
- Qiu, P.; Xiong, J.; Lu, M.; Liu, L.; Li, W.; Wen, Z.; Li, W.; Chen, R.; Cheng, G. Integrated p-n/Schottky junctions for efficient photocatalytic hydrogen evolution upon Cu@TiO2-Cu2O ternary hybrids with steering charge transfer. J. Colloid Interface Sci. 2022, 622, 924–937. [Google Scholar] [CrossRef] [PubMed]
- Dube, E.; Okuthe, G.E. Silver Nanoparticle-Based Antimicrobial Coatings: Sustainable Strategies for Microbial Contamination Control. Microbiol. Res. 2025, 16(6), 110. [Google Scholar] [CrossRef]
- López-Herrera, A.; Gómez-Merino, F.C.; Zavaleta-Mancera, H.A.; Avalos-Borja, M.; García-Nava, J.R.; Trejo-Téllez, L.-I. Effect of Silver Nanoparticles (AgNPs) on Aquatic and Wetland Plants. Environm 2024, 11(12), 297. [Google Scholar] [CrossRef]
- Bansal, R.C.; Goyal, M. Activated Carbon Adsorption; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar] [CrossRef]
- Aranaz, I.; Alcántara, A.R.; Civera, M.C.; Arias, C.; Elorza, B.; Heras-Caballero, A.; Acosta, N. Chitosan: An Overview of Its Properties and Applications. Polym. 2021, 13, 3256. [Google Scholar] [CrossRef]
- Deng, Y.; Yang, N.; Okoro, O.V.; Shavandi, A.; Nie, L. Alginate-Based Composite and Its Biomedical Applications. In Properties and Applications of Alginates; InTech Open: London, UK, 2021. [Google Scholar] [CrossRef]
- Alvarez-Ramirez, J.; Ochoa-Tapia, J.A.; Viveros, T. Langmuir–Hinshelwood kinetics: a singular value decomposition perspective on the quasi-steady state assumption. Reac. Kinet. Mech. Cat. 2026, 139, 71–97. [Google Scholar] [CrossRef]
- Sendão, R.M.S.; Algarra, M.; Lázaro-Martínez, J.; Brandão, A.T.S.C.; Gil, A.; Pereira, C.; Esteves da Silva, J.C.G.; Pinto da Silva, L. Visible-light-driven photocatalytic degradation of organic dyes using a TiO2 and waste-based carbon dots nanocomposite. Colloids Surf. A Physicochem. Eng. Asp. 2025, 713, 136475. [Google Scholar] [CrossRef]
- Pham, T.A.; Ping, Y.; Galli, G. Modelling heterogeneous interfaces for solar water splitting. In Nature Materials; 2019; pp. 1–38. Available online: https://www.osti.gov/servlets/purl/1844499.
- Monfort, O.; Wu, Y. Photocatalytic Processes for Environmental Applications. Processes 2021, 9(11), 2080. [Google Scholar] [CrossRef]
- Boudechiche, N.; Morante, N.; Sannino, D.; Monzillo, K.; Trari, M.; Sadaoui, Z. Enhanced Visible-Light Photocatalysis Activity of TiO2/Ag Nanocomposites Prepared by the Ultrasound-Assisted Sol–Gel Method: Characterization and Degradation–Mineralization of Cationic and Anionic Dyes. Catalysts 2024, 14, 883. [Google Scholar] [CrossRef]
- Keeley, M.; Kisslinger, K.; Adamson, C.; Furlan, P.Y. Magnetically Recoverable and Reusable Titanium Dioxide Nanocomposite for Water Disinfection. J. Mar. Sci. Eng. 2021, 9(9), 943. [Google Scholar] [CrossRef]





| Contaminant |
Original (NMP/100 mL) |
Treated (NMP/100 mL) |
| Total coliforms | 629 | 42 |
| Fecal coliforms | 53 | < 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




