Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design Overview
2.2. Human Skin Source and Ethics
2.3. Explant Preparation and Culture Conditions
2.4. Topical Treatments with 1-LGVTY
2.5. Biomarkers Panel and Analytical Methods
2.6. Image Acquisition and Quantification
2.7. Statistical Analysis (Exploratory)
3. Results
3.1. Environmental Stress Challenge Induces Measurable Responses in Ex Vivo Skin Explants
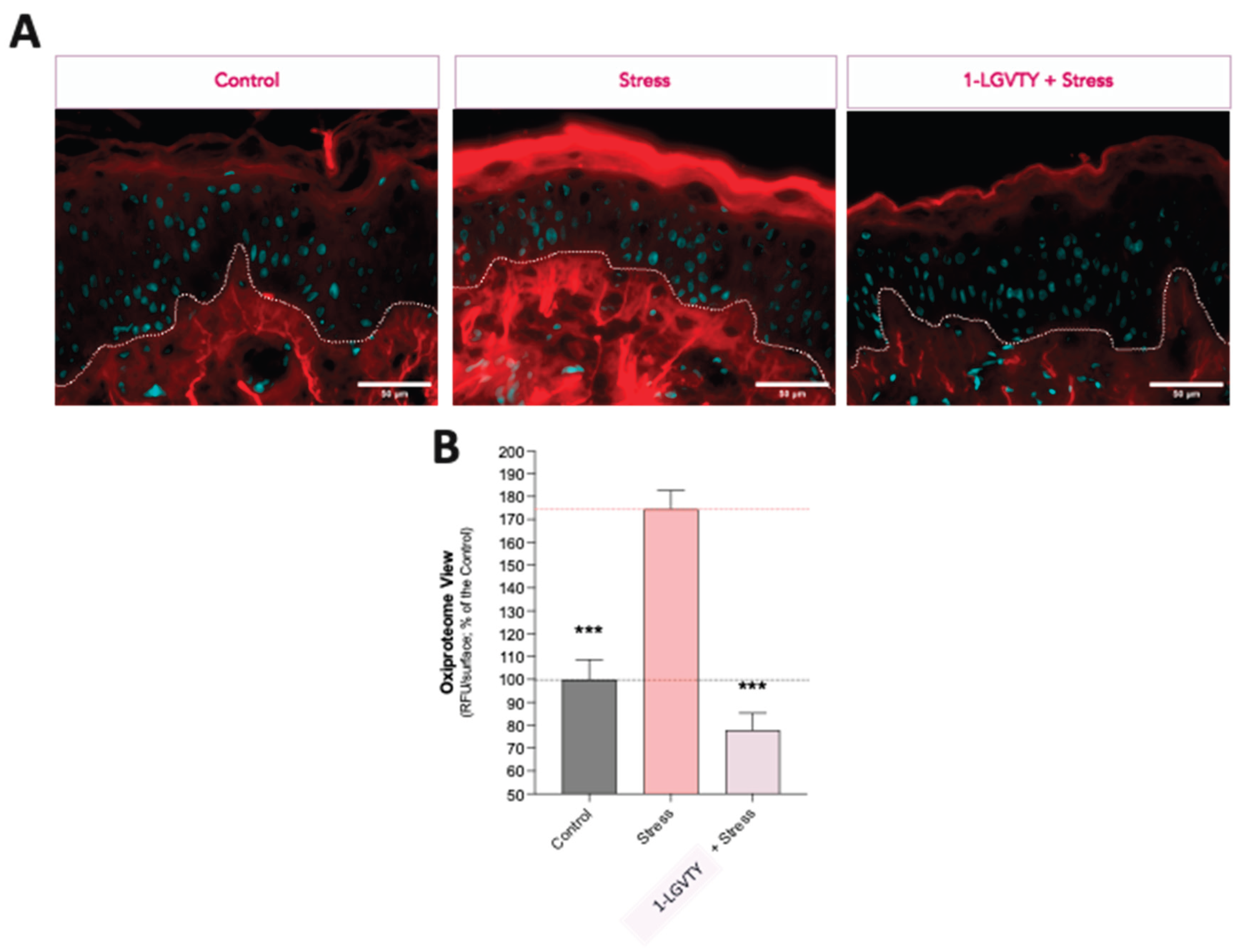
3.2. Oxidative Stress Responses in Skin Explants Exposed to Environmental Stressors
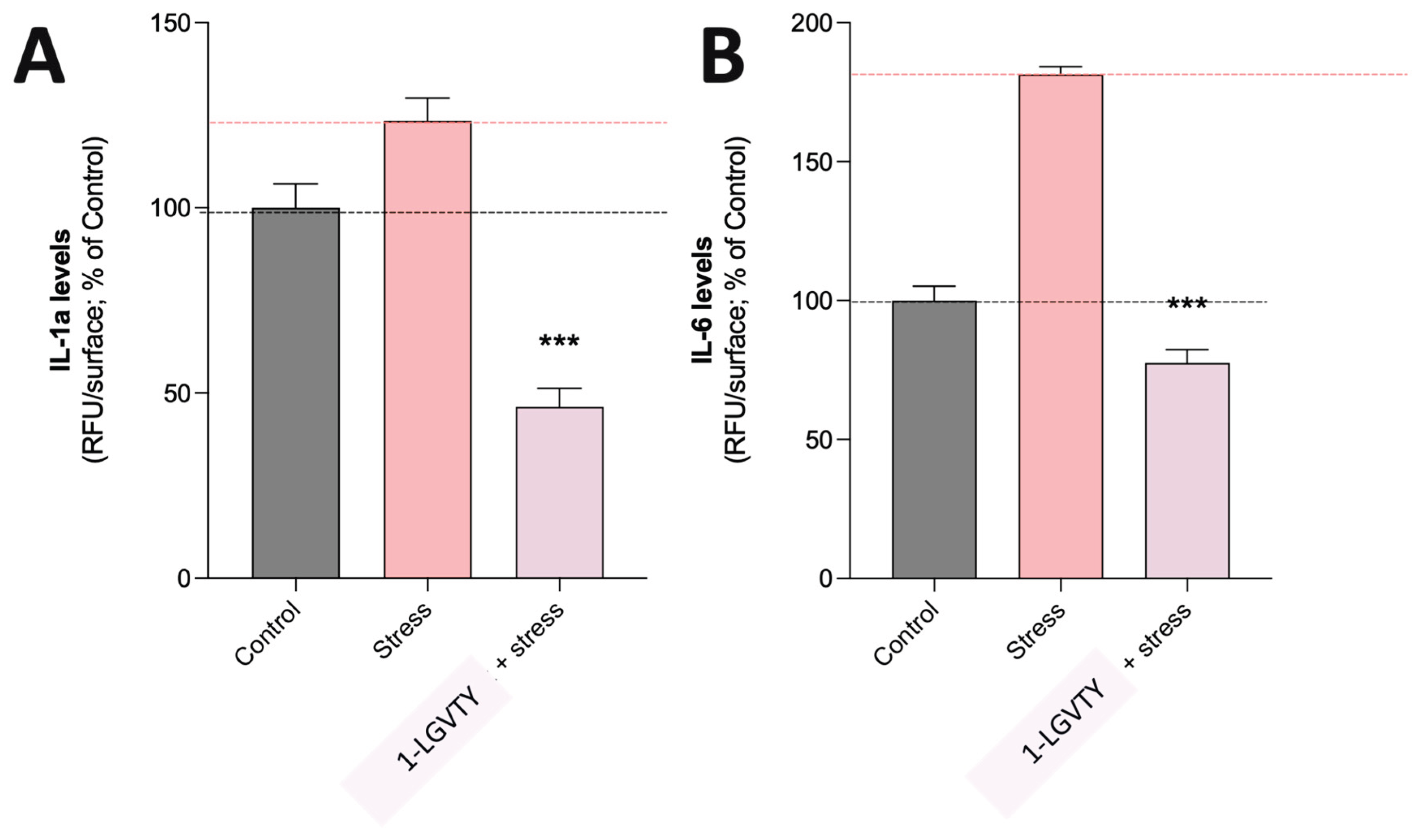
3.3. Modulation of Inflammatory Biomarkers
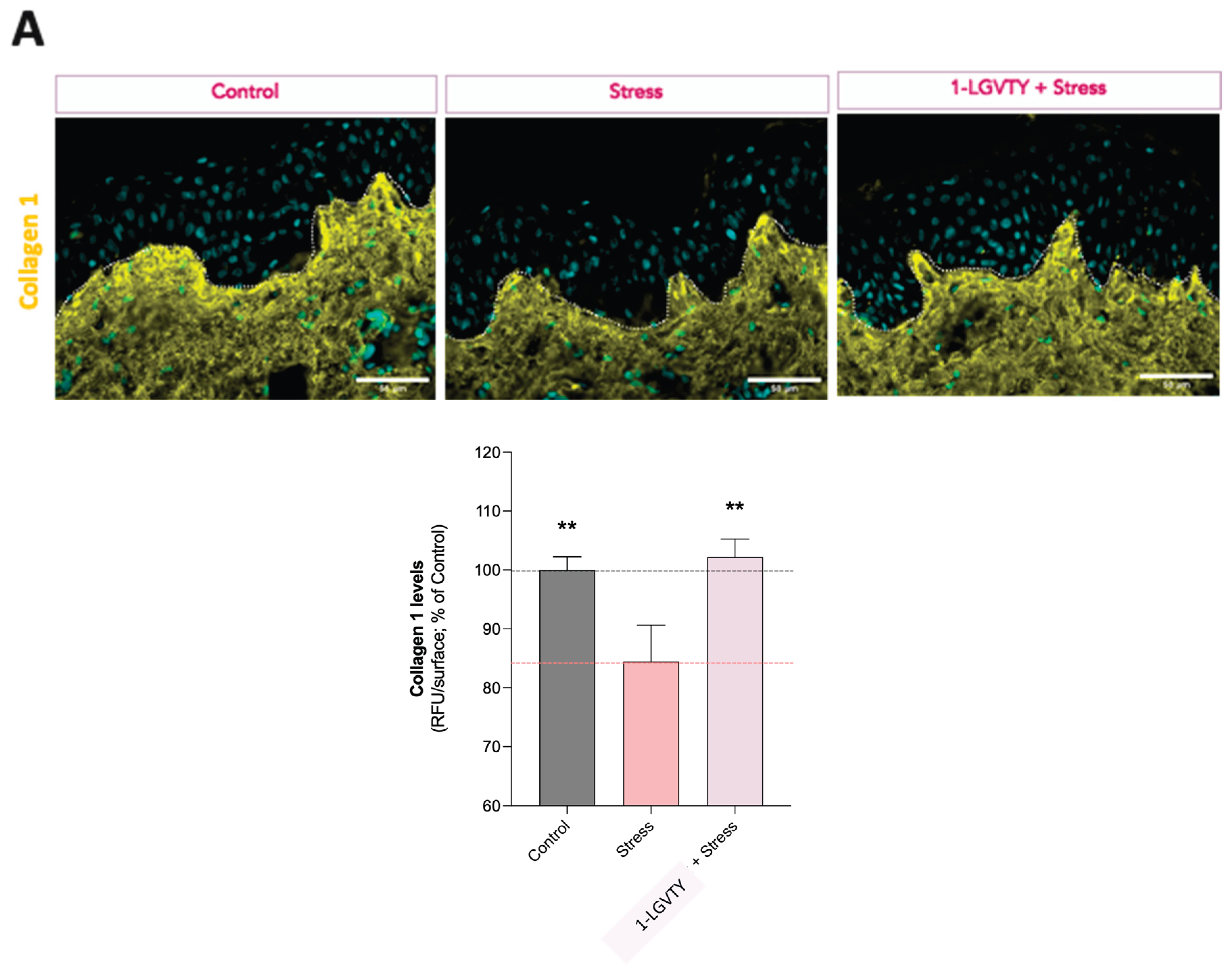
3.4. Extracellular Matrix Remodelling Markers
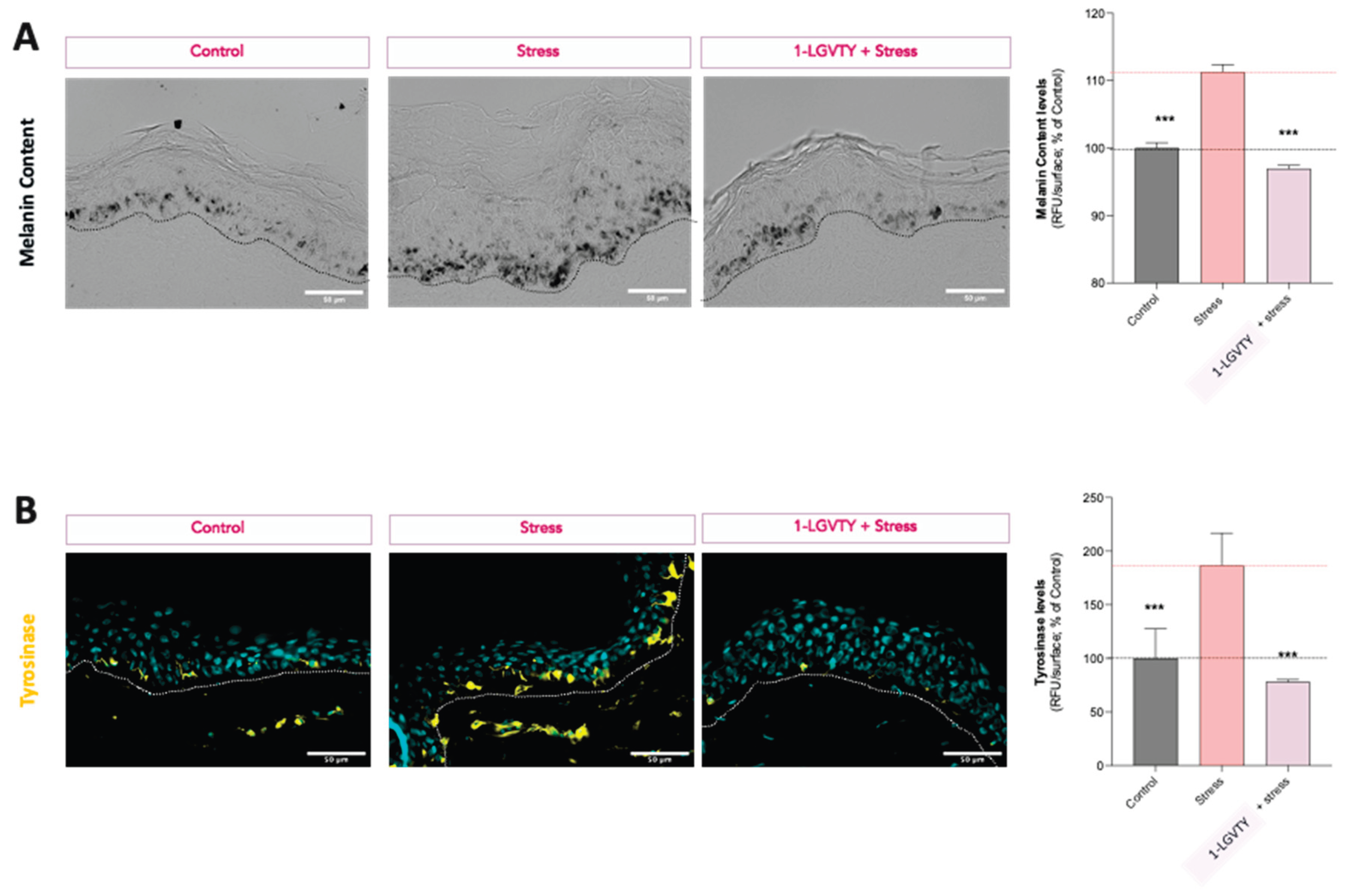
3.5. Pigmentation-Related Endpoints
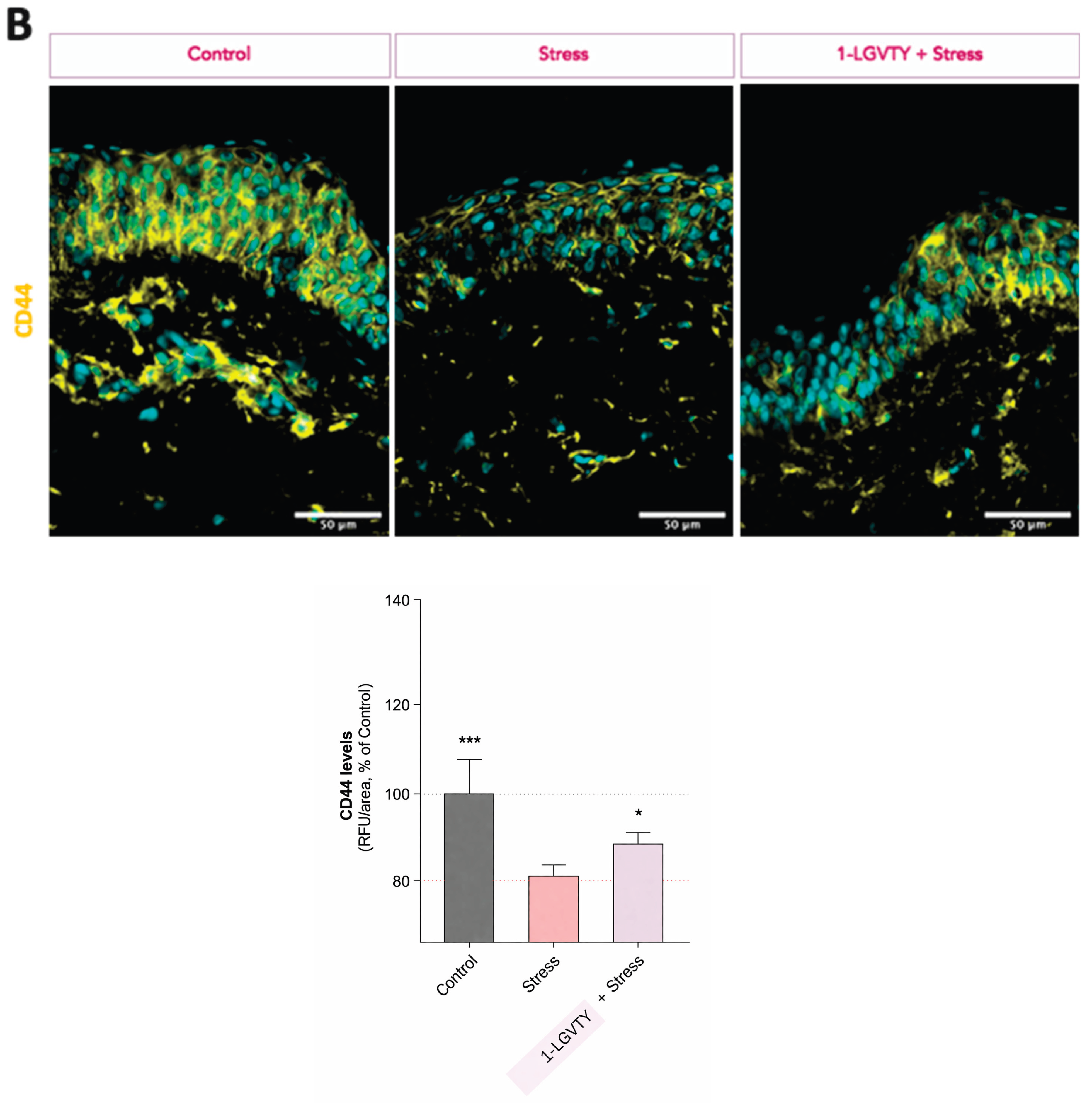
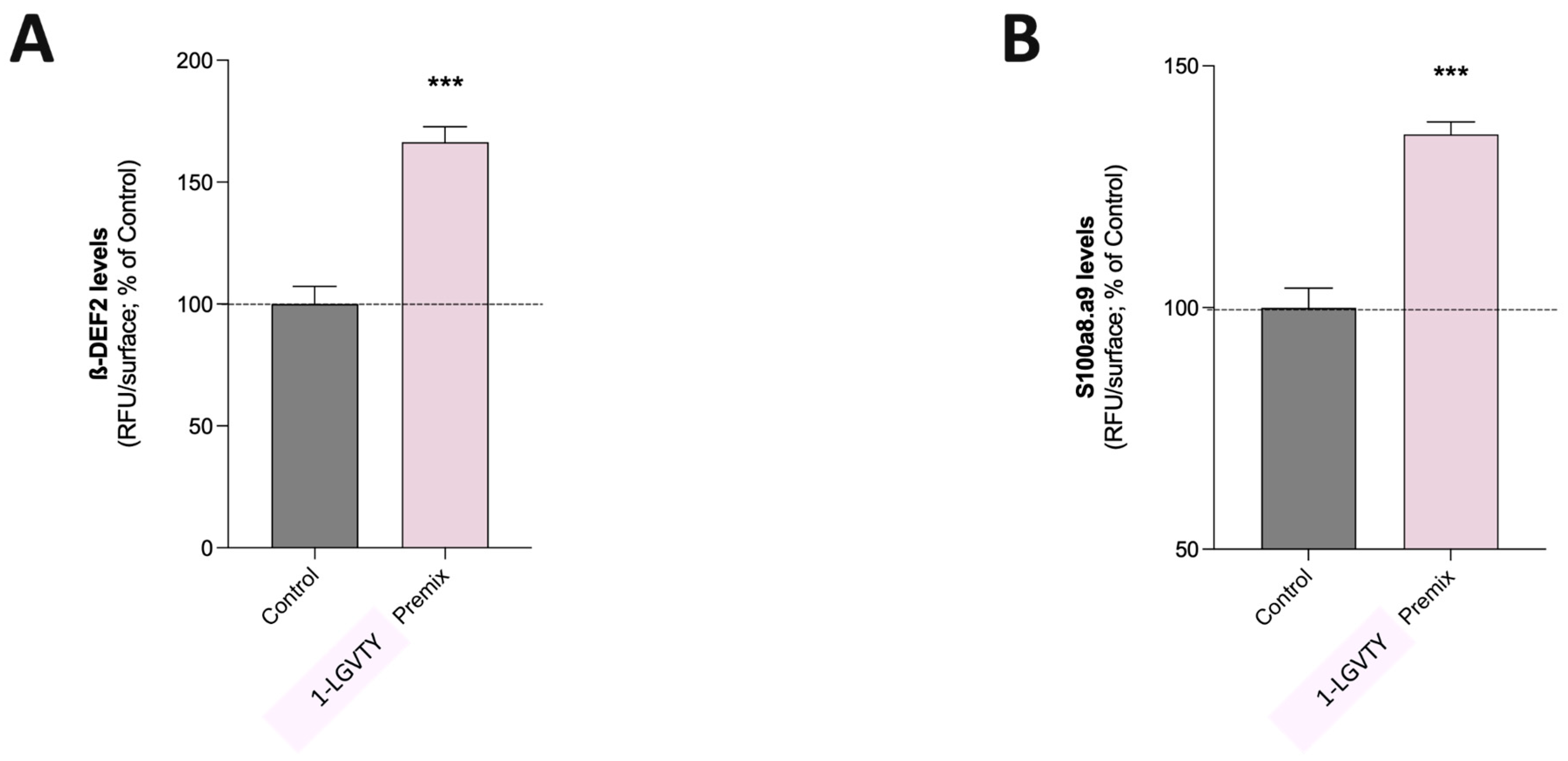
3.6. Skin Innate Immune Barrier Under Basal Conditions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 1-LGVTY | One Longevity Ingredient Combination |
| PM10 | Particulate Matter 10 |
| UV-A | Ultraviolet A |
| IL-1α | Interleukine 1α |
| IL-6 | Interleukine 6 |
| ECM | Extra Cellular Matrix |
| ROS | Reactive Oxidative Species |
| MMPs | Matrix Metallo Proteinases |
| HA | Hyaluronic Acid |
| AMPs | Anti-Microbial Peptides |
| PAH | Polycyclic Aromatic Hydrocarbons |
References
- Fisher, G.J.; Wang, Z.Q.; Datta, S.C.; Varani, J.; Kang, S.; Voorhees, J.J. Pathophysiology of premature skin aging induced by ultraviolet light. N Engl. J. Med. 1997, 337(20), 1419–28. [Google Scholar] [CrossRef]
- Zhou, H.M.; Slominski, R.M.; Seymour, L.J.; Bell, M.C.; Dave, P.; Atumonye, J.; Wright W3rd Dawes, A.; Griesenauer, B.; Paczesny, S.; Kaplan, M.H.; Spandau, D.F.; Turner, M.J. Ex vivo culture of mouse skin activates an interleukin 1 alpha-dependent inflammatory response. Exp. Dermatol. 2020, 29(1), 102–106. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pittayapruek, P.; Meephansan, J.; Prapapan, O.; Komine, M.; Ohtsuki, M. Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis. Int. J. Mol. Sci. 2016, 17(6), 868. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rittié, L.; Fisher, G.J. UV-light-induced signal cascades and skin aging. Ageing Res. Rev. 2002, 1(4), 705–20. [Google Scholar] [CrossRef] [PubMed]
- Calikoglu, E.; Sorg, O.; Tran, C.; Grand, D.; Carraux, P.; Saurat, J.H.; Kaya, G. UVA and UVB decrease the expression of CD44 and hyaluronate in mouse epidermis, which is counteracted by topical retinoids. Photochem Photobiol. 2006, 82(5), 1342–7. [Google Scholar] [CrossRef]
- Siiskonen, H.; Törrönen, K.; Kumlin, T.; Rilla, K.; Tammi, M.I.; Tammi, R.H. Chronic UVR causes increased immunostaining of CD44 and accumulation of hyaluronan in mouse epidermis. J. Histochem Cytochem. 2011, 59(10), 908–17. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Byun, E.J.; Lee, J.D.; Kim, S.; Kim, H.S. Air Pollution, Autophagy, and Skin Aging: Impact of Particulate Matter (PM10) on Human Dermal Fibroblasts. Int. J. Mol. Sci. 2018, 19(9), 2727. [Google Scholar] [CrossRef]
- Puri, P.; Nandar, S.K.; Kathuria, S.; Ramesh, V. Effects of air pollution on the skin: A review. Indian J. Dermatol. Venereol. Leprol. 2017, 83(4), 415–423. [Google Scholar] [CrossRef]
- Chung, J.H.; Kang, S.; Varani, J.; Lin, J.; Fisher, G.J.; Voorhees, J.J. Decreased extracellular-signal-regulated kinase and increased stress-activated MAP kinase activities in aged human skin in vivo. J. Investig. Dermatol. 2000, 115(2), 177–182. [Google Scholar] [CrossRef] [PubMed]
- Sander, C. S.; Chang, H.; Salzmann, S.; Müller, C. S.; Ekanayake-Mudiyanselage, S.; Elsner, P.; Thiele, J. J. Photoaging is associated with protein oxidation in human skin in vivo. J. Investig. Dermatol. 2002, 119(2), 536–543. [Google Scholar] [CrossRef]
- Gniadecka, M.; Wessel, S.; Heidenheim, M.; Wulf, H.C.; Nielsen, O.F.; Christensen, D.H. Water and protein structure in photoaged and chronically aged skin. J. Investig. Dermatol. 1998, 111(6), 1129–1133. [Google Scholar] [CrossRef]
- Quan, T.; He, T.; Kang, S.; Voorhees, J.J.; Fisher, G.J. Solar ultraviolet irradiation reduces collagen in photoaged human skin by blocking transforming growth factor-β type II receptor/Smad signaling. Am. J. Pathol. 2004, 165(3), 741–751. [Google Scholar] [CrossRef] [PubMed]
- Varani, J.; Damek-Poprawa, M.; Fligiel, S. E.; Wise, J. A.; Kang, S.; Fisher, G. J.; Voorhees, J. J. Decreased collagen production in chronologically aged skin: roles of age-associated alterations in fibroblast function and TGF-β signaling. Am. J. Pathol. 2006, 168(5), 1861–1868. [Google Scholar] [CrossRef]
- Bernstein, E.F.; Chen, Y.Q.; Kopp, J.B.; Fisher, L.; Brown, D.B.; Hahn, P.J.; Robey, F.A.; Lakkakorpi, J.; Uitto, J. Long-term sun exposure alters the collagen of the papillary dermis: Comparison of sun-protected and photoaged skin by Northern analysis, immunohistochemical staining, and confocal laser scanning microscopy. J. Am. Acad. Dermatol. 1996, 34(2), 209–218. [Google Scholar] [CrossRef]
- Naylor, E.C.; Watson, R.E.; Sherratt, M.J. Molecular aspects of skin ageing. Maturitas 2011, 69(3), 249–256. [Google Scholar] [CrossRef]
- Fisher, G. J.; Varani, J.; Voorhees, J. J. Looking older: fibroblast collapse and therapeutic implications. Arch. Dermatol. 2008, 144(5), 666–672. [Google Scholar] [CrossRef] [PubMed]
- Heinz, A. Elastic fibers during aging and disease. Ageing Res. Rev. 2021, 66, 101255. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, J.; Gilaberte, Y.; Prudkin, L.; Piquero-Casals, J. Pollution, a Relevant Exposome Factor in Skin Aging and the Role of Multi-benefit Photoprotection. Actas Dermosifiliogr. 2025, S0001-7310(25)00100-0. [Google Scholar] [CrossRef]
- Bernerd, F.; Passeron, T.; Castiel, I.; Marionnet, C. The Damaging Effects of Long UVA (UVA1) Rays: A Major Challenge to Preserve Skin Health and Integrity. Int. J. Mol. Sci. 2022, 23(15), 8243. [Google Scholar] [CrossRef]
- Magnani, N.D.; Muresan, X.M.; Belmonte, G.; Cervellati, F.; Sticozzi, C.; Pecorelli, A.; Miracco, C.; Marchini, T.; Evelson, P.; Valacchi, G. Skin Damage Mechanisms Related to Airborne Particulate Matter Exposure. Toxicol. Sci. 2016, 149(1), 227–36. [Google Scholar] [CrossRef]
- Jadwiga, R.; Ruzgas, T.; Engblom, J.; Holefors, A. The impact of pollution on skin and proper efficacy testing for anti-pollution claims. Cosmetics 2018, 5(no. 1), 4. [Google Scholar] [CrossRef]
- Pillai, S.; Oresajo, C.; Hayward, J. Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation - a review. Int. J. Cosmet. Sci. 2005, 27(1), 17–34. [Google Scholar] [CrossRef]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol. Sci. 2017, 85(3), 152–161. [Google Scholar] [CrossRef] [PubMed]
- Vierkötter, A.; Schikowski, T.; Ranft, U.; Sugiri, D.; Matsui, M.; Krämer, U.; Krutmann, J. Airborne particle exposure and extrinsic skin aging. J. Investig. Dermatol. 2010, 130(12), 2719–2726. [Google Scholar] [CrossRef]
- Hüls, A.; Vierkötter, A.; Gao, W.; Krämer, U.; Yang, Y.; Ding, A.; Stolz, S.; Matsui, M.; Kan, H.; Wang, S.; Jin, L.I. Traffic-related air pollution contributes to development of facial lentigines: further epidemiological evidence from Caucasians and Asians. J. Investig. Dermatol. 2016, 136(5), 1053–1056. [Google Scholar] [CrossRef]
- Schauber, J.; Gallo, R.L. Antimicrobial peptides and the skin immune defense system. J. Allergy Clin. Immunol. 2008, 122(2), 261–6. [Google Scholar] [CrossRef]
- Braff, M.H.; Bardan, A.; Nizet, V.; Gallo, R.L. Cutaneous defense mechanisms by antimicrobial peptides. J. Investig. Dermatol. 2005, 125(1), 9–13. [Google Scholar] [CrossRef]
- Murakami, M.; Ohtake, T.; Dorschner, R.A.; Gallo, R.L.; Schittek, B.; Garbe, C. Cathelicidin antimicrobial peptide expression in sweat, an innate defense system for the skin. J. Investig. Dermatol. 2002, 119(5), 1090–1095. [Google Scholar] [CrossRef] [PubMed]
- Braff, M.H.; Gallo, R.L. Antimicrobial peptides: an essential component of the skin defensive barrier. In Antimicrobial Peptides and Human Disease; 2006; pp. 91–110. [Google Scholar] [CrossRef]
- Coates, M.; Blanchard, S.; MacLeod, A.S. Innate antimicrobial immunity in the skin: A protective barrier against bacteria, viruses, and fungi. PLoS Pathog. 2018, 14(12), e1007353. [Google Scholar] [CrossRef]
- Inciarte-Mundo, J.; Frade-Sosa, B.; Sanmartí, R. From bench to bedside: Calprotectin (S100A8/S100A9) as a biomarker in rheumatoid arthritis. Front Immunol. 2022, 13, 1001025. [Google Scholar] [CrossRef] [PubMed]
- Abtin, A.; Eckhart, L.; Gläser, R.; Gmeiner, R.; Mildner, M.; Tschachler, E. The antimicrobial heterodimer S100A8/S100A9 (calprotectin) is upregulated by bacterial flagellin in human epidermal keratinocytes. J. Invest Dermatol. 2010, 130(10), 2423–30. [Google Scholar] [CrossRef]
- Johnstone, K.F.; Wei, Y.; Bittner-Eddy, P.D.; Vreeman, G.W.; Stone, I.A.; Clayton, J.B.; Reilly, C.S.; Walbon, T.B.; Wright, E.N.; Hoops, S.L.; Boyle, W.S.; Costalonga, M.; Herzberg, M.C. Calprotectin (S100A8/A9) Is an Innate Immune Effector in Experimental Periodontitis. Infect. Immun89 2021. [Google Scholar] [CrossRef]
- Lavigne, E.G.; Cavagnino, A.; Steinschneider, R.; Breton, L.; Baraibar, M.A.; Jäger, S. Oxidative damage prevention in human skin and sensory neurons by a salicylic acid derivative. Free Radic. Biol. Med. 2022, 181, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Li, B.Q.; Fu, T.; Gong, W.H.; Dunlop, N.; Kung, H.; Yan, Y.; Kang, J.; Wang, J.M. The flavonoid baicalin exhibits anti-inflammatory activity by binding to chemokines. Immunopharmacology 2000, 49, 295–306. [Google Scholar] [CrossRef]
- Gao, Z.; Huang, K.; Yang, X.; Xu, H. Free radical scavenging and antioxidant activities of flavonoids extracted from the radix. Biochim Biophys. Acta 1472, 643–50. [CrossRef]
- Nakajima, T.; Imanishi, M.; Yamamoto, K.; Cyong, J.C.; Hirai, K. Inhibitory effect of baicalein, a flavonoid in scutellaria root, on eotaxin production by human dermal fibroblasts. Plan. Med. 2001, 67(2), 132–5. [Google Scholar] [CrossRef] [PubMed]
- Kono, Y.; Shibata, H.; Kodama, Y.; Sawa, Y. The suppression of the N-nitrosating reaction by chlorogenic acid. Biochem J. 1995, 312 Pt 3, 947–53. [Google Scholar] [CrossRef]
- Salvi, A.; Carrupt, P.A.; Tillement, J.P.; Testa, B. Structural damage to proteins caused by free radicals: assessment, protection by antioxidants, and influence of protein binding. Biochem. Pharmacol. 2001, 61:10, 1237–1242. [Google Scholar] [CrossRef]
- Bouzoud, D.; Peno-Mazzarino, L.; Arnold, F.; Lati, E.; Luu, M.T.; Mercier, M. Prevention of Skin Aging by Plant Extracts Acting on the Glycation Process Ex-Vivo. Poster presented at The Society of Cosmetic Chemists - Annual Scientific Meeting, New York, 2010. [Google Scholar]
- Li, H.R.; Habasi, M.; Xie, L.Z.; Aisa, H.A. Effect of Chlorogenic Acid on Melanogenesis of B16 Melanoma Cells. Molecules 2014, 19(9), 12940–12948. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Liou, S.S.; Tzeng, T.F.; Lee, S.L.; Liu, I.M. Effect of Topical Application of Chlorogenic Acid on Excision Wound Healing in Rats. Plan. Medica 2013, 79(8). [Google Scholar] [CrossRef]
- Min, W.; Liu, X.; Qian, Q.; Lin, B.; Wu, D.; Wang, M.; Ahmad, I.; Yusuf, N.; Luo, D. Effects of baicalin against UVA-induced photoaging in skin fibroblasts. Am. J. Chin. Med. 2014, 42(3), 709–27. [Google Scholar] [CrossRef] [PubMed]
- Morita, A.; Torii, K.; Maeda, A.; Yamaguchi, Y. Molecular basis of tobacco smoke-induced premature skin aging. J. Investig. Dermatol. Symp. Proc. 2009, 14(1), 53–5. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; He, T.; Kang, S.; Voorhees, J.J.; Fisher, G.J. Connective tissue growth factor: expression in human skin in vivo and inhibition by ultraviolet irradiation. J. Invest Dermatol. 2002, 118(3), 402–8. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



