Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Legal Restrictions on Nitrogen Oxide Emissions
3. Simulation Model and Validation Procedure
4. Modeling of NOx Emissions in Exhaust Gases of Marine Four-Stroke Medium-Speed Engines
4.1. Nitrogen Oxides
4.1.1. Thermal Nitrogen Monoxide (NO) Generation
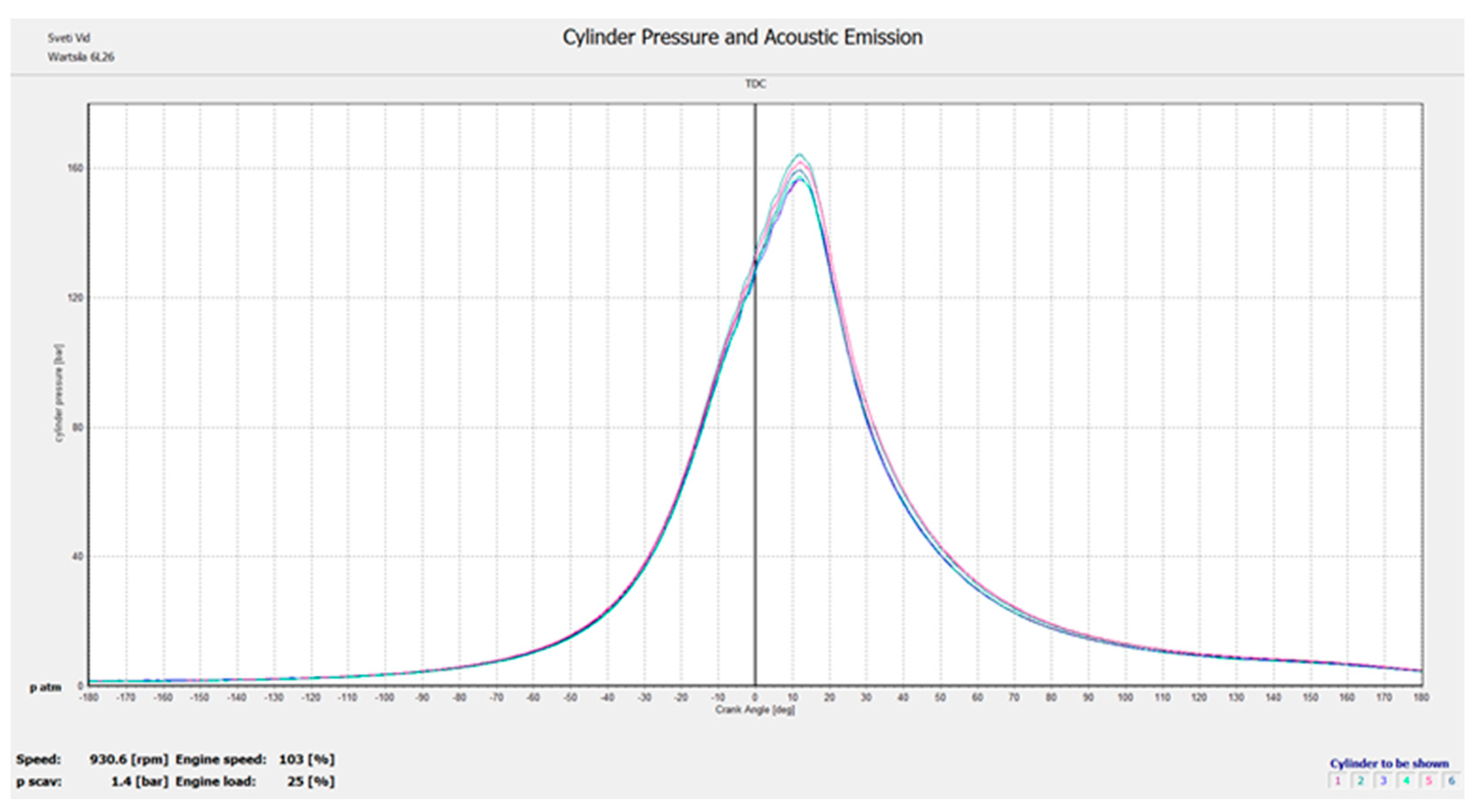
4.2. Conducting an Experiment
- Type: four-stroke medium-speed diesel engine with direct injection;
- Number of cylinders: 6, in-line engine;
- Cylinder diameter: 260 mm;
- Piston stroke: 320 mm;
- Firing order: 1-5-3-6-2-4;
- Maximum continuous power MCR: 1669 kW;
- Maximum continuous speed: 999 rpm;
- Fuel injection time: 14 before top dead center;
- Maximum mean indicated pressure: 19.7 bar;
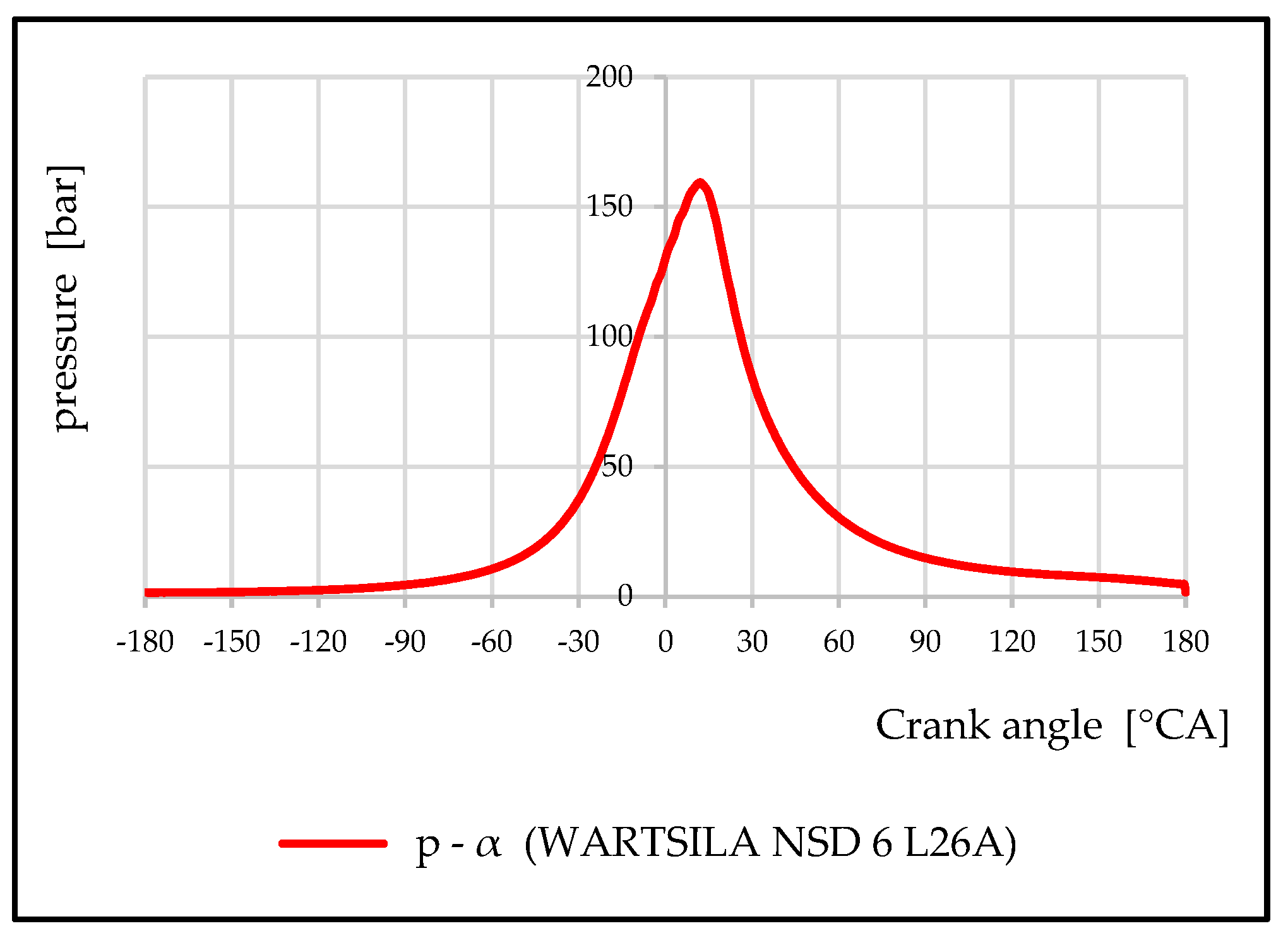
- Maximum combustion pressure: 164 bar;
- Brake specific fuel consumption BSFC: 199.5 g/kWh @ 100% MCR;
- Compression ratio: 17.5;
- Crankshaft radius to connecting rod length ratio: 0.2461.
- Ambient temperature: 13 °C;
- Atmospheric pressure: 750 mmHg / 9999.15 mbar;
- Relative humidity: 35%.
5. Conclusion
Abbreviations
| MCR IMO ACR AERC 3D-CFD MARPOL MEPC ECA EGCS MGO DNV NK |
Maximum Continuous Rating International Maritime Organization Arrhenius Pre-exponential Factor Arrhenius Activation Energy Factor 3D Computational Fluid Dynamics Međunarodnu konvenciju o sprječavanju onečišćenja s brodova Marine Environment Protection Committee Emission Control Areas Exhaust Gas Cleaning System Marine Gas Oil Det Norske Veritas Nippon Kaiji Kyokai |
| NOx NO k1,f MCR IMO ACR AERC 3D-CFD NO2 O N n rpm AO C H S O Pi pi dc cm p vm s mair,deliv mair,stoich T V vair R Rcp N2O N2O3 N2O5 k2,f k3,f N2 x N2 x O2 KC KP xNO xN2 xO2 |
Nitrogen oxide emission concentration, g/kWh Nitrogen monoxide, g/kWh; ppm Forvard rate constant of the first Zeldovich reaction, cm3/mol s Maximum Continuous Rating International Maritime Organization Arrhenius Pre-exponential Factor Arrhenius Activation Energy Factor 3D Computational Fluid Dynamics Nitrogen dioxide Oxygen Nitrogen Revolution Revolution per minute, 1/min Stoichiometric amount of air, kgair/kgfuel Carbon Hydrogen Sulfur Oxygen Indicated engine power, kW Mean indicated pressure, bar Cylinder diameter, m Mean piston speed, m/s Pressure, bar Mean piston speed, m/s Piston stroke, m Mass of delivered air, kg/kWh Mass of air required for stoichiometric combustion, kg/kWh Temperature, K Cylinder volume, m3 Scavenging air mass delivered Universal gas constant, J/kmolK Gas constant of combustion products, J/kmolK Nitrous oxide Dinitrogen trioxide Dinitrogen pentoxide Forvard rate constant of the second Zeldovich reaction, cm3/mol s Forvard rate constant of the third Zeldovich reaction, cm3/mol s molecular nitrogen (elemental nitrogen) Mole fraction of nitrogen Mole fraction of oxygen Equilibrium concentration constant Equilibrium gas constant Nitrogen monoxide content Nitrogen content Oxygen content |
| τ λm α αair |
Number of strokes per engine cycle (for two-stroke cycle 2, for four-stroke cycle 4 Ratio of crank radius to the connecting rod length Crankshaft angle, °CA Excess air ratio |
| λs | Wavelength of light, nm |
| TLA | Three letter acronyms |
| LD | Linear dichroism |
References
- Tsitsilonis E., Theotokatos G. A digital twin framework for marine diesel engines health assessment. Elsevier, Ap-plied Soft Computing, Vol 121, 2022. [CrossRef]
- Hebbar G. S. NOx from diesel engine emission and control strategies - a review. International Journal of Mechanical Engineering Robotics Research, Vol. 3, No.4, str. 471-482 October 2014.
- Wang, F.; Zhao, J.; Li, T.; Guan, P.; Liu, S.;Wei, H.; Zhou, L. Research on NOx Emissions Testing and Optimization Strategies for Diesel Engines Under Low-Load Cycles. Atmosphere. 2025, 16, 190. [CrossRef]
- Xiao, H.; Ying,W.; Chen, A.; Chen, G.; Liu, Y.; Lyu, Z.; Qiao, Z.; Li, J.; Zhou, Z.; Deng, X. Study on the Impact of Ammonia–Diesel Dual-Fuel Combustion on Performance of a Medium-Speed Diesel Engine. J. Mar. Sci. Eng. 2024, 12, 806. (accessed on 01 March 2026). [CrossRef]
- Jo K.S.; Kong K.J.; Han S.H. Comparative Analysis of Combustion Characteristics and Emission Formation in Marine Diesel Engines Using Biofuels: Chemical Mechanism Analysis and Computational Fluid Dynamics Simulation. J. Mar. Sci. Eng. 2025, 13, 1098. (accessed on 05 February 2026). [CrossRef]
- Lalić, B. Low Speed Marine Diesel Engine Modeling for NOx Prediction in Exhaust Gases. Ph.D. Thesis, Faculty of Electrical Engineering, Mechanical Engineering and Naval Architecture, University of Split, Split, Croatia, 2020.
- Lalić B.; Poljak A.; Radica G.; Mišura A. Low-Speed Marine Diesel Engine Modeling for NOx Prediction in Exhaust Gases. Energies. 2021, 14, 4442. (accessed on 15 February 2026). [CrossRef]
- M. Tadros, Boulougouris E. Calibration Procedures for NOx Emissions Model of a High-Speed Marine Diesel Engine Using Optimization Procedures. J. Mar. Sci. Eng. 2025, 13, 1585. (accessed on 16 February 2026). [CrossRef]
- Zeldovich, Y.B. Oxidation of Nitrogen in Combustion and Explosions. Acta Physicochim. URSS 1946, 21, 577–628.
- Bowman C. T. Kinetics of Pollutant Formation and Destruction on Combustion. Progress in Energy and Combustion Science, Volume 1, Issue 1, Pages 33-45, 1975.
- Zajemska M.; Poskart A.; Musiai D. The kinetics of nitrogen oxides formation in the flame gas. Czestochowa University of Technology, Poland, Economic and Environmental Studies EES, Vol. 15, No. 4, str. 445-460, 2015.
- Merker G.P.; Hohlbaum B.; Rauscher M. Two-Zone Model for Calculation of Nitrogen-Oxide Formation in Direct Injection Diesel Engines. SAE 1993 Trans. 1993, 2043–2050.
- G. A. Weisser. Modelling of Combustion and Nitric Oxide Formation for Medium-Speed DI Diesel Engines: A Comparative Evaluation of Zero and Thre-Dimensional Approaches. Doctoral Thesis, ETH Swiss Federal Institute of Technology, Zürich, 2001.
- Tomić M.; Z. Jovanović Z.; Knežević D.; Masončić Z.; Dragutinović S. The Simple Method for IC Engine Heat Release Evaluation from Cylinder Pressure Record. 2015 International Conference on Advance in Software, Control and Mechanical Engineering (ICSCME'2015), Antalya, Turkey, Sept. 7-8, str. 1-8.
- Petrović S.; Jankov R.; Tomić M.; Filipi Z.; Pusonja M.; Jovanović Z. Combustion Modelling in SI engines. Faculty of. Mech. Eng. University of Belgrade, Belgrade, 1995.
- Isenburg, R.; Creping, J.; Beer, A. Diesel-Speichereinspritzsystem Common-Rail; Bosch: Stuttgart, Germany, 1998.
- Krugler C. Validierungeines 1D-Spraymodells zur Simulation der Gemischbildung in direkteinspritzenden Dieselmotoren. Dissertation, Technischen Hochschule Aachen, 2001.
- Hanson, R.K.; Salimian, S. Survey of Rate Constants in the N/H/O System in Combustion Chemistry, Chapter 6. Springer: New York, NY, USA, pp. 361–421, 1984.
- Wang, Z.; Yang, X. NOx Formation Mechanism and Emission Prediction in Turbulent Combustion: A Review. Appl. Sci. 14, 6104, 2024. (accessed on 26 February 2026). [CrossRef]
- Zajemska, M.; Poskart, A.; Musial, D. The kinetics of nitrogen oxides formation in the flame gas. J. Econ. Environmental Studies EES, 15, 445–460, 2015.
- Wysocki O.; Witkowski K. Determining the NOx emission from an auxiliary marine engine in the conditions of changing engine load and speed. Scientific Journals of the Maritime University of Szczecin, Vol. 73 (145), page, 45–53, 2023. [CrossRef]
- American LUNG Association-Nitrogen Dioxide. https://www.lung.org/our-initiatives/healthy-air/outdoor/air-pollution/nitrogen-dioxide.html.
- https://www.lr.org/en/knowledge/class-news/05-25/ (accessed on 27 February 2026).
- https://wwwcdn.imo.org/localresources/en/OurWork/Environment/Documents/annex/resolution%20MEPC%20361(79).pdf (accessed on 05 March 2026).
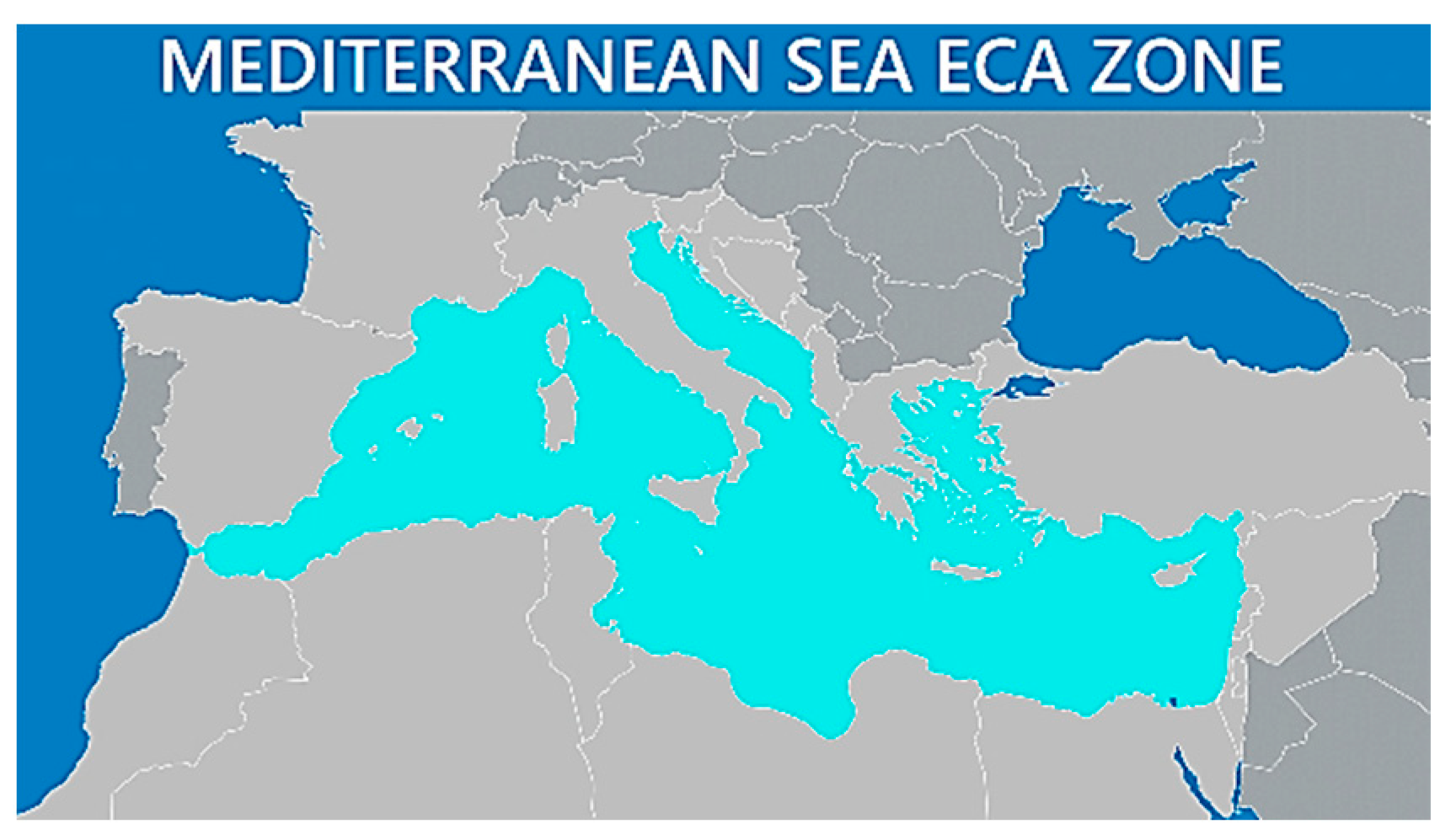
- https://public.axsmarine.com/blog/navigating-the-mediterranean-sea-eca (accessed on 20 March 2026).
- Škifić N. Analiza utjecajnih parametara opreme na značajke dizelskog motora. Ph.D. Thesis, University of Rijeka, Faculty of Engineering, Rijeka, 2003.
- Hebbar G.S. NOx from diesel engine emission and control strategies - a review. International Jurnal of Mechanical Engineering Robotics Research, Vol. 3, No.4, str. 471-482, October 2014.
- McAllister S.; Chen J.; Fernandez-Pello A.C. Fundamentals of Combustion Processes. Springer Science+Business Media, ISBN 978-1-4419-7942-1. 2011. [CrossRef]
- Murić K. Modeling of NOx formation in heavy duty engines. Department of Automatic Control Lund University, ISSN 0280-5316, September 2011.
- Turns S.R. An introduction to combustion: concepts and applications 2nd edition. McGraw-Hill series in mechanical engineering, ISBN 0-07-116910-5, 1996.
- https://www.maridis.de/en/mobile-devices/marprime-marprime-ultra.html.
- Merker G.P.; Schwarz C.; Stiesch G.; Otto F. Simulation of combustion and pollutant formation for engine-development. Springer, 2004., ISBN 10 3-540-25161-8.
- Mellor A.M.; Mello J.P.; Duffy K.P.; Easley W.L.; Faulkner J.C. Skeletal Mechanism for NOx Chemistry in Diesel Engines, International Spring Fuels and Lubricants Meeting and Exposition. SAE Technical paper series 981450, 1998.
- Psota M.A.; Mellor A.M. Dynamic Application of a Skeletal Mechanism for DI Diesel NOx Emissions. SAE Technical Paper Series 2001-01-1984. 10.42712001-01-1984.
- Warnatz J.; Maas U.; Dibble R.W. Combustion, Physical and Chemical Fundamentals, Modeling and Simulation, Experiments, Pollutant Formation, 4th Edition. Springer-Verlag Berlin Heidelberg, ISBN-13978-3-540-25992-3, 2006.
- https://web.eng.ucsd.edu/mae/groups/combustion/mechanism.html.
- Bowman C.T. NOx Formation and Models, Encyclopedia of Automotive Engineering. John Wiley & Sons, Ltd. 2014. [CrossRef]
- Bowman C.T. Investigation of Nitric Oxide Formation Kinetics in Combustion Processes: The Hydrogen-Oxygen-Nitrogen Reaction. Combustion Science and Technology, 3:1, 37-45, 2007. (accessed on 05 February 2023). [CrossRef]
- Goswami M.; Volkov E.N.; Konnov A.A.; Bastiaans R.J.M; Goey L.P.H. Updated Kinetic Mechanism for NOx Prediction and Hydrogen Combustion. Energy-2008-6-clean coal technologies, Mechanical Engineering Technische Universiteit Eindhoven Eindhoven, The Netherlands, 2008.



| Tier | Ship construction date on or after | 130 < n < 2000 |
|---|---|---|
| I | 1 January 2000 | 45 · n-0,2, e.g. 1000 rpm= 11,3 g/kWh |
| II | 1 January 2011 | 44 · n-0,23, e.g. 1000 rpm = 8,98 g/kWh |
| III | 1 January 2016 | 9 · n-0,2, e.g. 1000 rpm = 2,26 g/kWh |
| Characteristic values | Unit | ISO-F-DMA |
| Kinematic viscosity @ 50° C | mm2/s | 2,913 |
| Density | kg/m3 | 834,3 |
| Net specific energy | kJ/kg | 42940 |
| Carbon | m/m | 85,86 |
| Hydrogen | m/m | 13,78 |
| Sulphur | m/m | 0,033 |
| Nitrogen | m/m | 0,0019 |
| Oxygen | m/m | 0,32 |
| Water | m/m | 0,0 |
| No. |
k1,f (mol/cm3s) |
NO (g/kWh) |
NO (ppm) |
Error (%) |
Ref. |
|---|---|---|---|---|---|
| 1 | 12,21 | 1013,59 | 14,01 | [9] | |
| 2 | 11,17 | 927,26 | +4,3 | [13] | |
| 3 | 10,38 | 864,68 | -3,07 | [18] | |
| 4 | 10,15 | 842,58 | -5,22 | [18] | |
| 5 | 10,38 | 861,68 | -3,07 | [28] | |
| 6 | 10,61 | 880,77 | -0,93 | [32] | |
| 7 | 10,57 | 877,45 | -1,30 | [33] | |
| 8 | 10,56 | 876,62 | +1,39 | [34] | |
| 9 | 10,88 | 903,18 | +1,60 | [35] | |
| 10 | 10,50 | 871,64 | +1,95 | [36] | |
| 11 | 10,18 | 845,07 | -4,94 | [37] | |
| 12 | 9,95 | 825,98 | -7,09 | [38] | |
| 13 | 10,15 | 842,58 | -5,22 | [39] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




