Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Isolation of Actinomycetes
2.2. Screening of Antifungal Actinomycetes
2.3. Identification of Antifungal Actinomycetes
2.4. Assay of Abroad-Spectrum Antifungal Activity of Strain 7-1
2.5. Preparation of Ethanol Extracts
2.6. Inhibition Percentage of Strain 7-1 Extracts on Mycelial Growth
2.7. Effects of Strain 7-1 Extracts on the Activity of Foc TR4
2.8. Effects of Strain 7-1 Extracts on Ultrastructure of Foc TR4
2.9. Effects of Crude Extracts on Foc TR4 Conidia
2.10. Effect of Crude Extracts on Foc TR4 Cell Membranes
2.11. Metabolic Changes During Co-Culture of Strain 7-1 and Foc TR4
2.12. Functional Verification of Key Bioactive Metabolites
2.13. Efficacy of Strain 7-1 in the Control of Banana Seedling Blight
2.14. Analysis of Inter-Root Microbial Communities
2.15. Effect of Streptomyces 7-1 on Root Defense Enzymes in Banana Seedlings
2.16. Preliminary Study on the Antifungal Mechanism of Key Bioactive Metabolites
2.17. Effects of Key Bioactive Metabolites on the Infectivity of Foc TR4 to Banana
2.18. Data Statistics and Analysis
3. Results
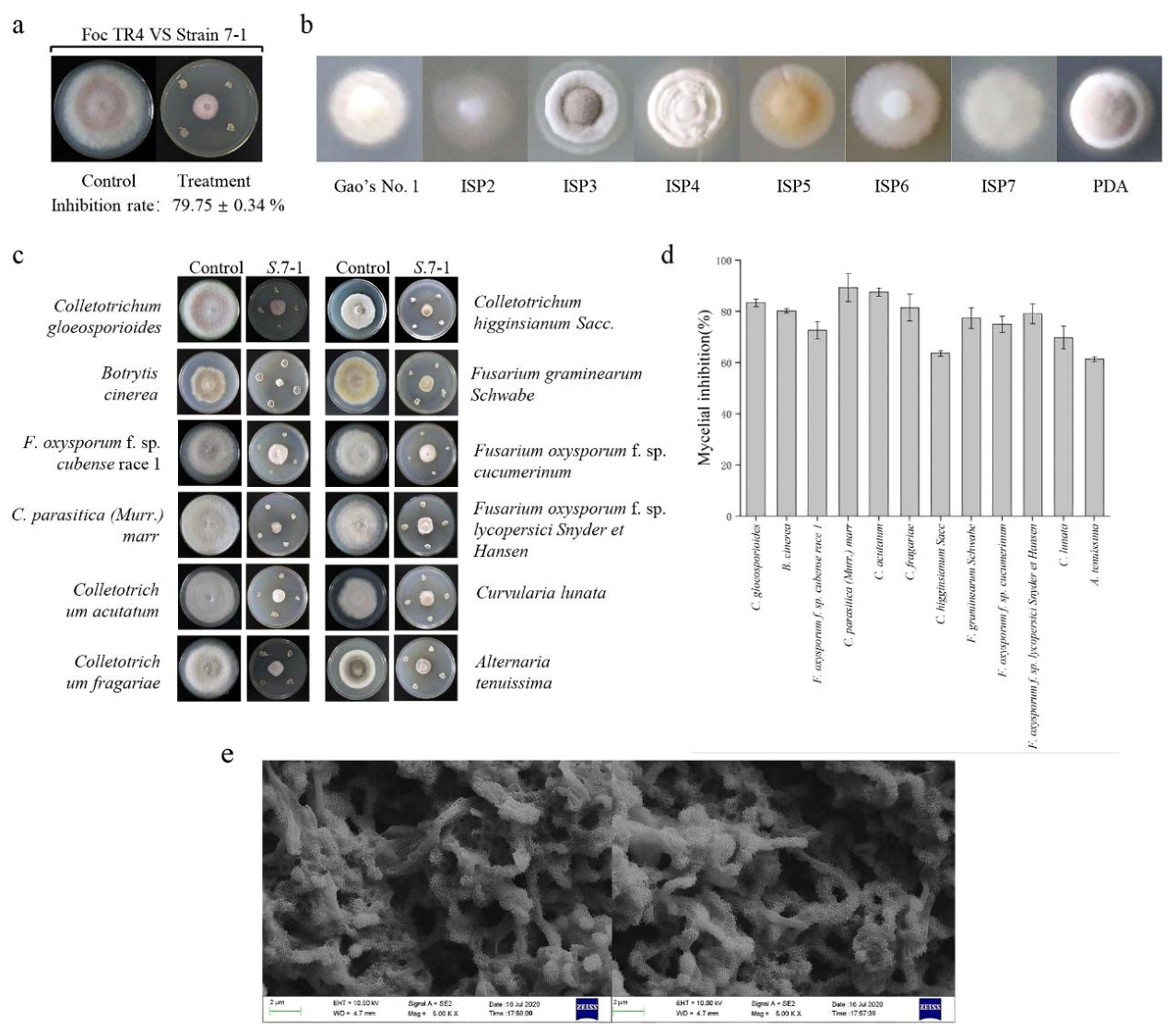
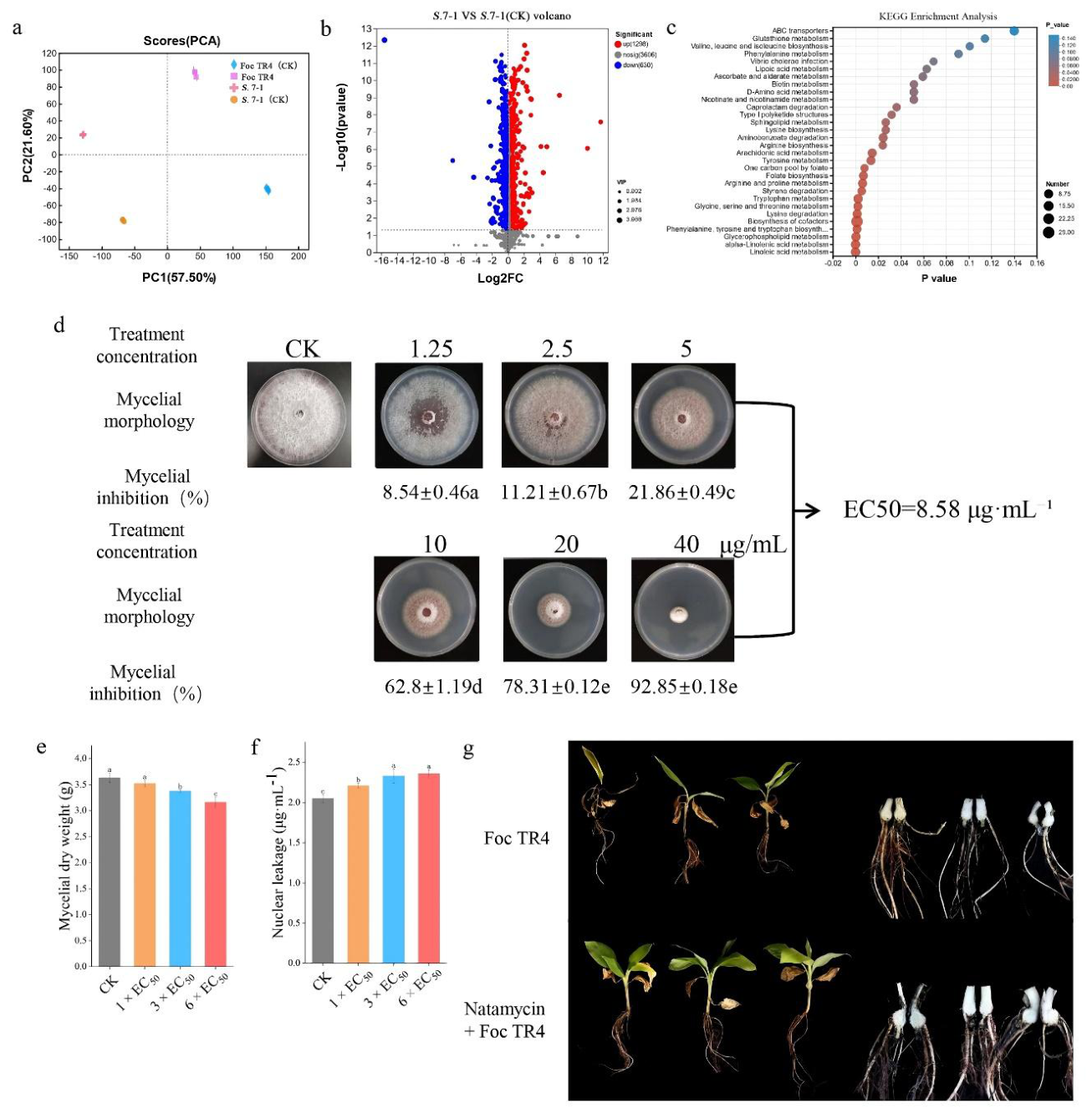
3.1. Streptomyces Strain 7-1 Exhibits Antifungal Activities Against Various Phytopathogenic Fungi
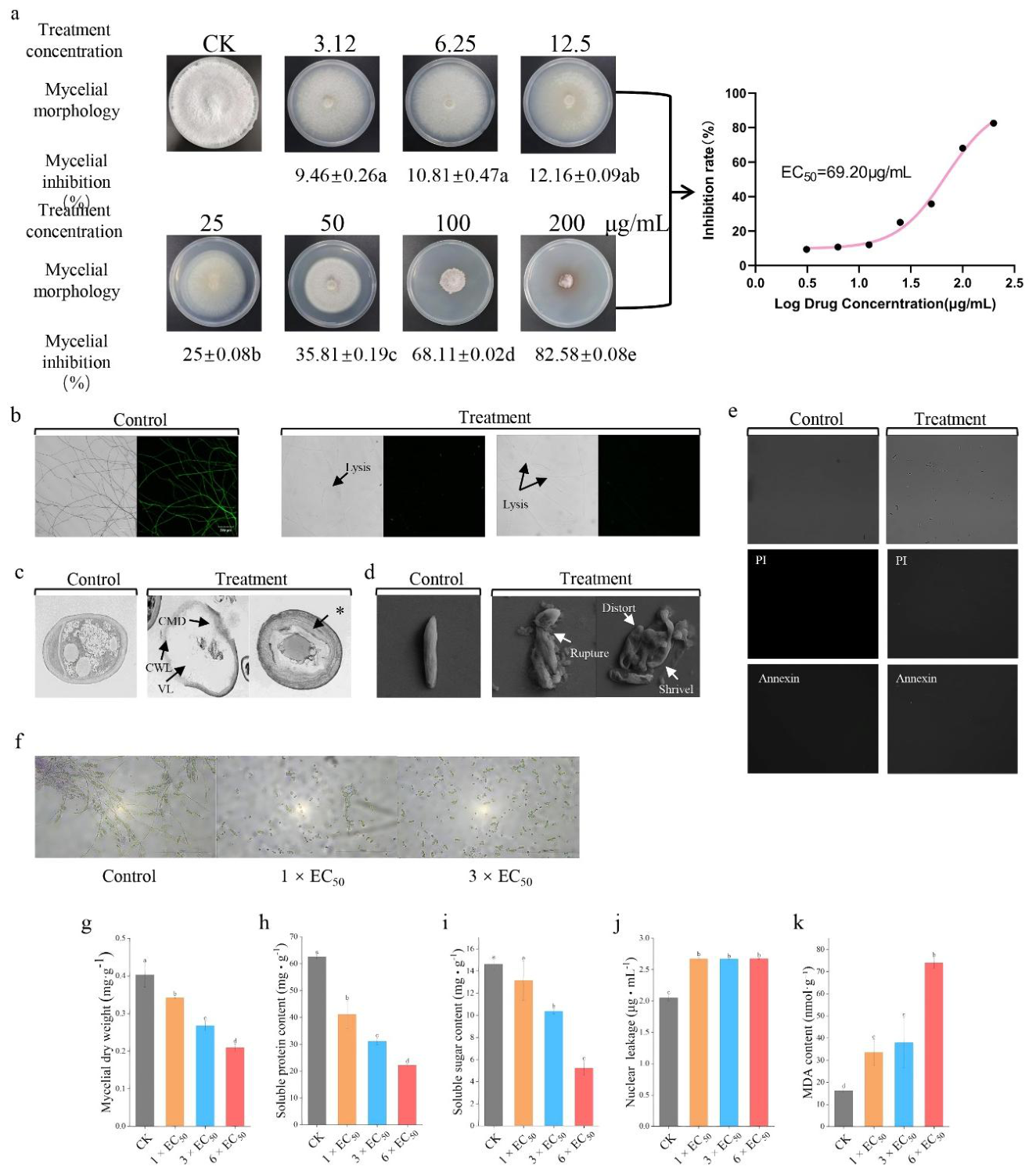
3.2. Streptomyces Strain 7-1 Extract Exhibits Antifungal Activity Against Foc TR4
3.3. Streptomyces Strain 7-1 Inhibited the Growth of Spores and Hyphae of Foc TR4
3.4. Physiological Mechanism Underlying Foc TR4 Inhibition by Streptomyces sp. 7-1 Crude Extract
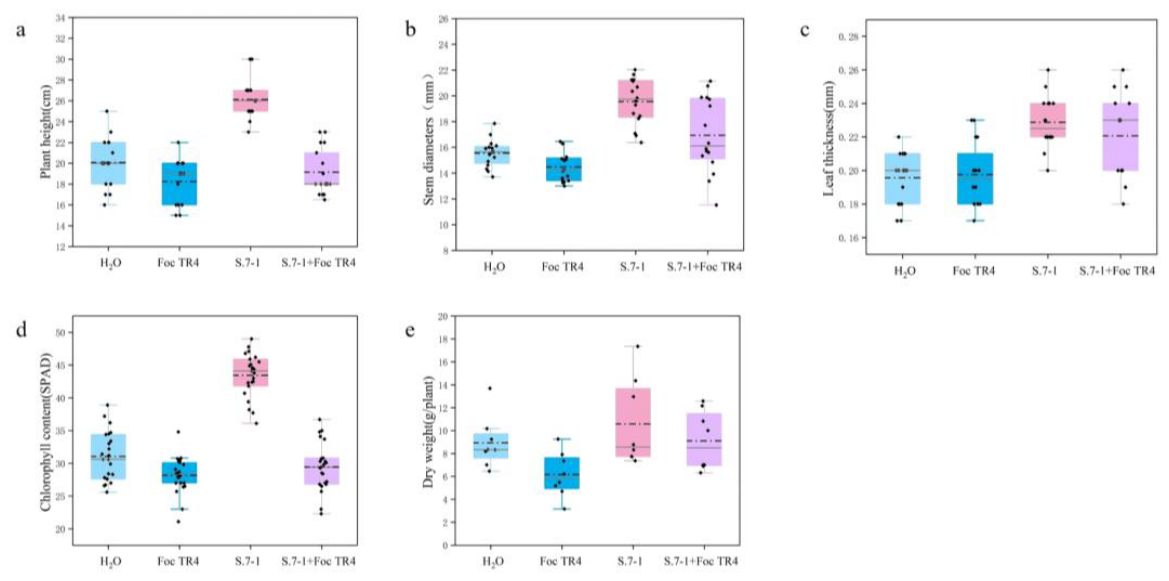
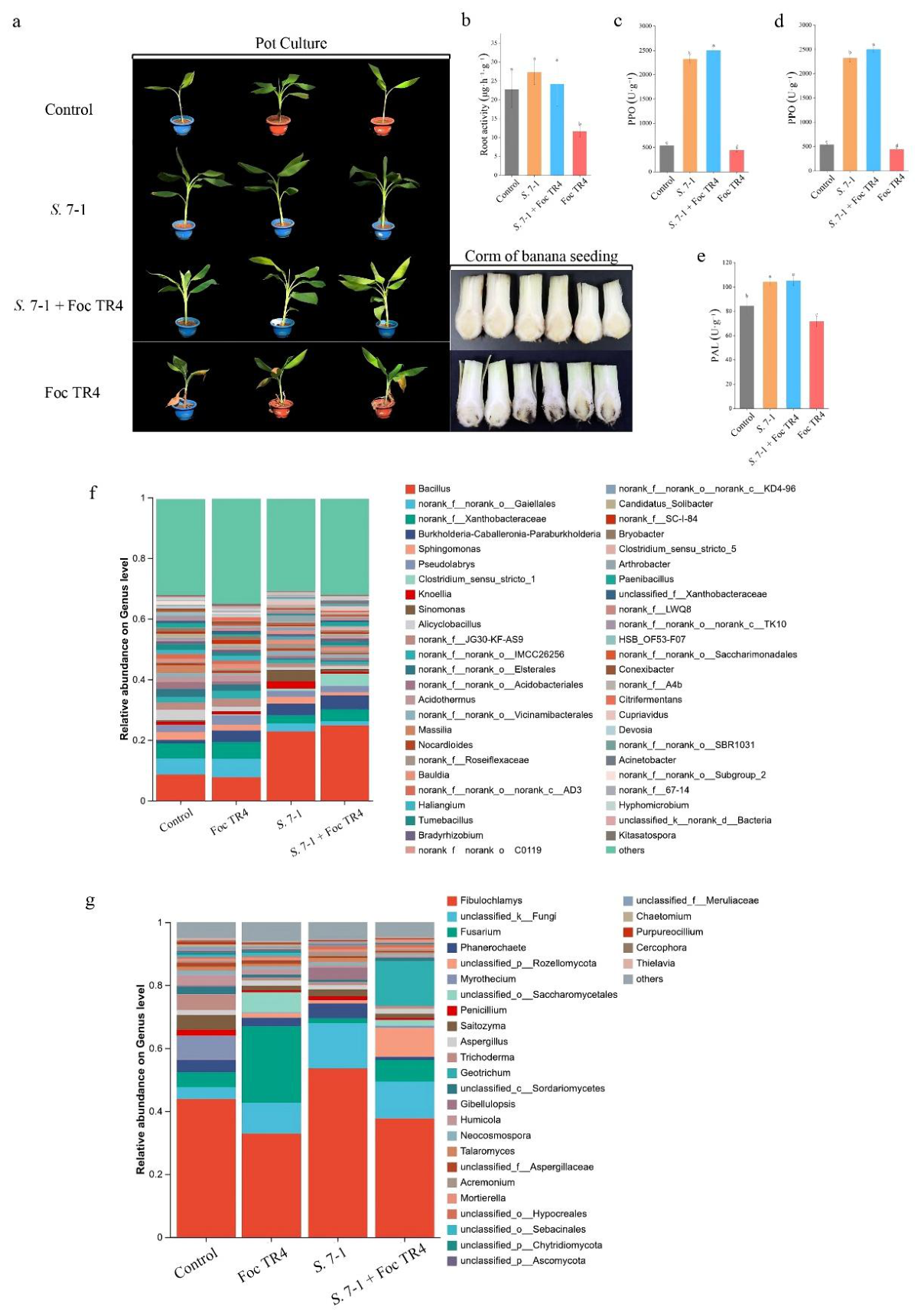
3.5. Streptomyces sp. 7-1 Improve the Plant Resistance Against Foc TR4
3.6. Strain 7-1 Extracts Activates Defense Response and Secondary Metabolite Biosynthesis in Banana
3.7. Streptomyces sp. 7-1 Enriches Beneficial Rhizosphere Microbiota
3.8. Identification of Key Active Metabolites of Streptomyces sp. 7-1 Inhibiting Foc TR4 Growth
3.9. Effects of Key Active Metabolites on Foc TR4 Infection in Banana
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1
| Medium type | Substrate mycelium | Aerial mycelium | Soluble pigment | Growth status |
| Gao’s No. 1 | pale yellow | white | None | +++ |
| ISP2 | pale yellow | white | None | +++ |
| ISP3 | pale yellow | white | None | +++ |
| ISP4 | pale yellow | white | None | +++ |
| ISP5 | yellow | white | None | ++ |
| ISP6 | pale yellow | white | None | +++ |
| ISP7 | pale yellow | white | None | +++ |
| PDA | pale yellow | white | None | ++ |
| Carbon sources | Results | Nitrogen sources | Results | Indicator Name | Results |
| Sucrose | ++ | Tryptophan | + | Catalase | + |
| Rhamnose | + | Tyrosine | + | Oxidase | - |
| Mannose | + | Histidine | +++ | MR (Methyl Red) | - |
| Raffinose | - | Asparagine | +++ | VP (Voges-Proskauer) | - |
| Glucose | + | Phenylalanine | ++ | Nitrate reduction | + |
| Arabinose | - | Anhydrous Creatine | - | Gelatin liquefaction | + |
| Mannitol | - | Glutamic Acid | - | Starch hydrolysis | + |
| α-Lactose | - | Valine | - | Urease | + |
| D-Fructose | +++ | Methionine | - | Tryptophan | + |
| Maltose | - | L-Hydroxyproline | + | Lipase-20 | + |
| Inositol | ++ | Glycine | +++ | Lipase-40 | + |
| Galactose | +++ | Arginine | ++ | Lipase-80 | + |
| Sorbose | - | Cysteine | + | Chitinase | + |
| Sorbitol | +++ | Potassium Nitrate | +++ | β-1,3-Glucanase | + |
| D-Cellobiose | +++ | Peptone | +++ | Hydrogen sulfide (H₂S) | - |
| D-Xylose | +++ | Urea | + | Cellulose hydrolysis | - |
| D-Ribose | - | Ammonium Sulfate | ++ | NaCl tolerance | <7% |
| Xylan | + | Proteose Peptone | +++ | Optimum pH | 7 |
| Trehalose | +++ | Yeast Extract | +++ | ||
| Melezitose | - | Casein | +++ |
| Antibiotic susceptibility | Content (µg/tablet) | Result | Antibiotic susceptibility | Content (µg/tablet) | Result |
| Penicillin | 100 | R | Ampicillin | 10 | R |
| Erythromycin | 15 | S | Oxacillin | 1 | R |
| Minocycline | 30 | R | Piperacillin | 100 | R |
| Kanamycin | 30 | S | Cefradine | 30 | R |
| Gentamicin | 10 | S | Cefuroxime | 30 | R |
| Cefoperazone | 75 | R | Amikacin | 30 | S |
| Ceftriaxone | 30 | R | Neomycin | 30 | S |
| Ceftazidime | 30 | R | Tetracycline | 30 | R |
| Cefazolin | 30 | R | Doxycycline | 30 | R |
| Cephalexin | 30 | R | Midecamycin | 30 | S |
| Carbenicillin | 100 | S | Ofloxacin | 50 | R |
Appendix B
References
- Cheng, C.; Wu, S.; Deng, G.; Sheng, O.; Yi, G.; Yang, Q. Recent Advances and Future Directions in Banana Molecular Biology and Breeding. Mol. Hortic. 2024, 4, 42. [CrossRef]
- Chen, Y.; Zhou, D.; Qi, D.; Gao, Z.; Xie, J.; Luo, Y. Growth Promotion and Disease Suppression Ability of a Streptomyces sp. CB-75 from Banana Rhizosphere Soil. Front. Microbiol. 2018, 8, 2704. [CrossRef]
- Jing, T.; Zhou, D.; Zhang, M.; Yun, T.; Qi, D.; Wei, Y.; et al. Newly Isolated Streptomyces sp. JBS5-6 as a Potential Biocontrol Agent to Control Banana Fusarium Wilt: Genome Sequencing and Secondary Metabolite Cluster Profiles. Front. Microbiol. 2020, 11, 602591. [CrossRef]
- Tao, C.; Wang, Z.; Liu, S.; Lv, N.; Deng, X.; Xiong, W.; et al. Additive Fungal Interactions Drive Biocontrol of Fusarium Wilt Disease. New Phytol. 2023, 238, 1198–1214. [CrossRef]
- Hong, S.; Jv, H.; Lu, M.; Wang, B.; Ruan, Y. Significant Decline in Banana Fusarium Wilt Disease Is Associated with Soil Microbiome Reconstruction under Chilli Pepper-Banana Rotation. Eur. J. Soil Biol. 2020, 97, 103154. [CrossRef]
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N. CRISPR/Cas9-Based Genome Editing of Banana for Disease Resistance. Curr. Opin. Plant Biol. 2020, 56, 118–126. [CrossRef]
- Zapata-Sarmiento, D.H.; Palacios-Pala, E.F.; Rodríguez-Hernández, A.A.; Melchor, D.L.M.; Rodríguez-Monroy, M.; Sepúlveda-Jiménez, G. Trichoderma asperellum, a Potential Biological Control Agent of Stemphylium vesicarium on Onion (Allium cepa L.). Biol. Control 2019, 140, 104105. [CrossRef]
- Bubici, G.; Kaushal, M.; Prigigallo, M.I.; Gómez-Lama Cabanás, C.; Mercado-Blanco, J. Biological Control Agents against Fusarium Wilt of Banana. Front. Microbiol. 2019, 10, 616. [CrossRef]
- Thangavelu, R.; Gopi, M. Combined Application of Native Trichoderma Isolates Possessing Multiple Functions for the Control of Fusarium Wilt Disease in Banana cv. Grand Naine. Biocontrol Sci. Technol. 2015, 25, 1147–1164. [CrossRef]
- Li, C.; Cheng, P.; Zheng, L.; Li, Y.; Yu, G. Comparative Genomics Analysis of Two Banana Fusarium Wilt Biocontrol Endophytes Bacillus subtilis R31 and TR21 Provides Insights into Their Differences on Phytobeneficial Trait. Genomics 2021, 113, 900–909. [CrossRef]
- Xu, Z.; Wang, M.; Du, J.; Huang, T.; Chen, Y. Isolation of Burkholderia sp. HQB-1, a Promising Biocontrol Bacteria to Protect Banana against Fusarium Wilt through Phenazine-1-Carboxylic Acid Secretion. Front. Microbiol. 2020, 11, 605152. [CrossRef]
- Chen, Y.; Li, X.; Zhou, D.; Wei, Y.; Feng, J.; Cai, B.; et al. Streptomyces-Secreted Fluvirucin B6 as a Potential Bio-Fungicide for Managing Banana Fusarium Wilt and Mycotoxins and Modulating the Soil Microbial Community Structure. J. Agric. Food Chem. 2024, 72, 17890–17902. [CrossRef]
- AbdElgawad, H.; Abuelsoud, W.; Madany, M.M.Y.; Selim, S.; Zinta, G.; Mousa, A.S.M.; et al. Actinomycetes Enrich the Soil Rhizosphere and Improve Seed Quality as Well as Productivity of Legumes by Boosting Nitrogen Availability and Metabolism. Biomolecules 2020, 10, 1675. [CrossRef]
- Bhatti, A.A.; Haq, S.; Bhat, R.A. Actinomycetes Benefaction Role in Soil and Plant Health. Microb. Pathog. 2017, 111, 458–467. [CrossRef]
- Bu, Q.T.; Li, Y.P.; Xie, H.; Li, J.F.; Li, Y.Q. Rational Engineering Strategies for Achieving High-Yield, High-Quality and High-Stability of Natural Product Production in Actinomycetes. Metab. Eng. 2021, 67, 198–215. [CrossRef]
- Dede, A.; Güven, K.; Ahn, N. Isolation, Plant Growth-Promoting Traits, Antagonistic Effects on Clinical and Plant Pathogenic Organisms and Identification of Actinomycetes from Olive Rhizosphere. Microb. Pathog. 2020, 143, 104134. [CrossRef]
- Li, X.; Li, K.; Zhou, D.; Zhang, M.; Qi, D.; Jing, T.; Zang, X.; Qi, C.; Wang, W.; Xie, J. Biological Control of Banana Wilt Disease Caused by Fusarium oxysporum f. sp. Cubense Using Streptomyces sp. H4. Biol. Control 2021, 155, 104524. [CrossRef]
- Cao, M.; Cheng, Q.; Cai, B.; Chen, Y.; Wei, Y.; Qi, D.; Li, Y.; Yan, L.; Li, X.; Long, W.; Liu, Q.; Xie, J.; Wang, W. Antifungal Mechanism of Metabolites from Newly Isolated Streptomyces sp. Y1-14 against Banana Fusarium Wilt Disease Using Metabolomics. J. Fungi, 2022, 8(12), 1291. [CrossRef]
- Ou, Y.; Penton, C.R.; Geisen, S.; Shen, Z.; Shen, Q. Deciphering Underlying Drivers of Disease Suppressiveness Against Pathogenic Fusarium oxysporum. Front. Microbiol. 2019, 10, 2535. [CrossRef]
- Hnamte, L.; Vanlallawmzuali; Kumar, A.; Yadav, M.K.; Zothanpuia; Singh, P.K. An Updated View of Bacterial Endophytes as Antimicrobial Agents Against Plant and Human Pathogens. Curr. Res. Microbial Sci. 2024, 7, 100241. [CrossRef]
- Zou, N.; Zhou, D.; Chen, Y.; Lin, P.; Chen, Y.; Wang, W.; Xie, J.; Wang, M. A Novel Antifungal Actinomycete Streptomyces sp. Strain H3-2 Effectively Controls Banana Fusarium Wilt. Front. Microbiol. 2021, 12, 706647. [CrossRef]
- Wang, J.; Cai, B.; Li, K.; Zhao, Y.; Li, C.; Liu, S.; Xiang, D.; Zhang, L.; Xie, J.; Wang, W. Biological Control of Fusarium oxysporum f. sp. cubense Tropical Race 4 in Banana Plantlets Using Newly Isolated Streptomyces sp. WHL7 from Marine Soft Coral. Plant Dis. 2022, 106, 254–259. [CrossRef]
- Ahmad, M.S.; El-Gendy, A.O.; Ahmed, R.R.; Hassan, H.M.; El-Kabbany, H.M.; Merdash, A.G. Exploring the Antimicrobial and Antitumor Potentials of Streptomyces sp. AGM12-1 Isolated from Egyptian Soil. Front. Microbiol. 2017, 8, 438. [CrossRef]
- Patani, A.; Patel, M.; Islam, S.; Yadav, V.K.; Prajapati, D.; Yadav, A.N.; Sahoo, D.K.; Patel, A. Recent Advances in Bacillus-Mediated Plant Growth Enhancement: A Paradigm Shift in Redefining Crop Resilience. World J. Microbiol. Biotechnol. 2024, 40, 77. [CrossRef]
- Vimal, S.R.; Singh, J.S.; Kumar, A.; Prasad, S.M. The Plant Endomicrobiome: Structure and Strategies to Produce Stress Resilient Future Crop. Curr. Res. Microbial Sci. 2024, 6, 100236. [CrossRef]
- Liu-Xu, L.; González-Hernández, A.I.; Camañes, G.; Vicedo, B.; Scalschi, L.; Llorens, E. Harnessing Green Helpers: Nitrogen-Fixing Bacteria and Other Beneficial Microorganisms in Plant–Microbe Interactions for Sustainable Agriculture. Horticulturae 2024, 10, 621. [CrossRef]
- Li, J.; Zhang, L.; Yao, G.; Zhu, L.; Lin, J.; Wang, C.; Du, B.; Ding, Y.; Mei, X. Synergistic Effect of Co-Culture Rhizosphere Streptomyces: A Promising Strategy to Enhance Antimicrobial Activity and Plant Growth-Promoting Function. Front. Microbiol. 2022, 13, 976484. [CrossRef]
- Pang, F.; Solanki, M.K.; Wang, Z. Streptomyces Can Be an Excellent Plant Growth Manager. World J. Microbiol. Biotechnol. 2022, 38, 209. [CrossRef]
- Sousa, J.A.d.J.; Olivares, F.L. Plant Growth Promotion by Streptomycetes: Ecophysiology, Mechanisms and Applications. Chem. Biol. Technol. Agric. 2016, 3, 22. [CrossRef]
- Liu, Z.; Zhang, J.; Fan, C.; Sun, S.; An, X.; Sun, Y.; et al. Influence of Bacillus subtilis strain Z-14 on microbial ecology of cucumber rhizospheric vermiculite infested with Fusarium oxysporum f. sp. cucumerinum. Pestic. Biochem. Physiol. 2024, 201, 105875. [CrossRef]
- Li, X.; Li, K.; Zhou, D.; Zhang, M.; Qi, D.; Jing, T.; et al. Biological control of banana wilt disease caused by Fusarium oxysporum f. sp. cubense using Streptomyces sp. H4. Biol. Control 2021, 155, 104524. [CrossRef]
- Lu, J.; Huang, Y.; Liu, R.; Liang, Y.; Zhang, H.; Shen, N.; et al. Antimicrobial mechanisms and antifungal activity of compounds generated by banana rhizosphere Pseudomonas aeruginosa Gxun-2 against Fusarium oxysporum f. sp. cubense. Front. Microbiol. 2024, 15, 1456847. [CrossRef]
- Cao, Y.; Zhang, X.; Song, X.; Li, W.; Ren, Z.; Feng, J.; Ma, Z.; Liu, X.; Wang, Y. Efficacy and toxic action of the natural product natamycin against Sclerotinia sclerotiorum. Pest Manage. Sci. 2023, 80, 1981–1990. [CrossRef]
- Juneja, V.K.; Dwivedi, H.P.; Yan, X. Novel Natural Food Antimicrobials. Annu. Rev. Food Sci. Technol. 2012, 3, 381–403. [CrossRef]
- Pan, H.; Zhong, C.; Xia, L.; Li, W.; Wang, Z.; Deng, L.; Li, L.; Long, C. Antifungal activity of natamycin against kiwifruit soft rot caused by Botryosphaeria dothidea and potential mechanisms. Scientia Hortic. 2022, 305, 111344. [CrossRef]
- Wang, F.; Saito, S.; Michailides, T.J.; Xiao, C.-L. Postharvest use of natamycin to control Alternaria rot on blueberry fruit caused by Alternaria alternata and A. arborescens. Postharvest Biol. Technol. 2021, 172, 111383. [CrossRef]
- Streekstra, H.; Verkennis, A.E.E.; Jacobs, R.; Dekker, A.; Stark, J.; Dijksterhuis, J. Fungal Strains and the Development of Tolerance against Natamycin. Int. J. Food Microbiol. 2016, 238, 15–22. [CrossRef]
- Gong, M.; Wang, J.D.; Zhang, J.; Yang, H.; Lu, X.F.; Pei, Y.; et al. Study of the antifungal ability of Bacillus subtilis strain PY-1 in vitro and identification of its antifungal substance (iturin A). Acta Biochim. Biophys. Sin. 2006, 38, 233–240. [CrossRef]
- Gu, S.; Wei, Z.; Shao, Z.; Friman, V.P.; Cao, K.; Yang, T.; et al. Competition for iron drives phytopathogen control by natural rhizosphere microbiomes. Nat. Microbiol. 2020, 5, 1002–1010. [CrossRef]
- Yun, T.; Zhang, M.; Zhou, D.; Jing, T.; Zang, X.; Qi, D.; et al. Anti-Foc RT4 Activity of a Newly Isolated Streptomyces sp. 5-10 from a Medicinal Plant (Curculigo capitulata). Front. Microbiol. 2021, 11, 610698. [CrossRef]
- Juraschek, L.M.; Kappenberg, A.; Amelung, W. Mycotoxins in Soil and Environment. Sci. Total Environ. 2022, 814, 152425. [CrossRef]
| Treatment | Foc TR4 | S. 7-1 + Foc TR4 |
| Disease index (%) | 63.87 | 23.54 |
| Control effect (%) | - | 63.15 |
| Treatment | Foc TR4 | Natamycin + Foc TR4 |
| Disease index (%) | 84.50 | 55.00 |
| Control effect (%) | - | 34.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




