Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
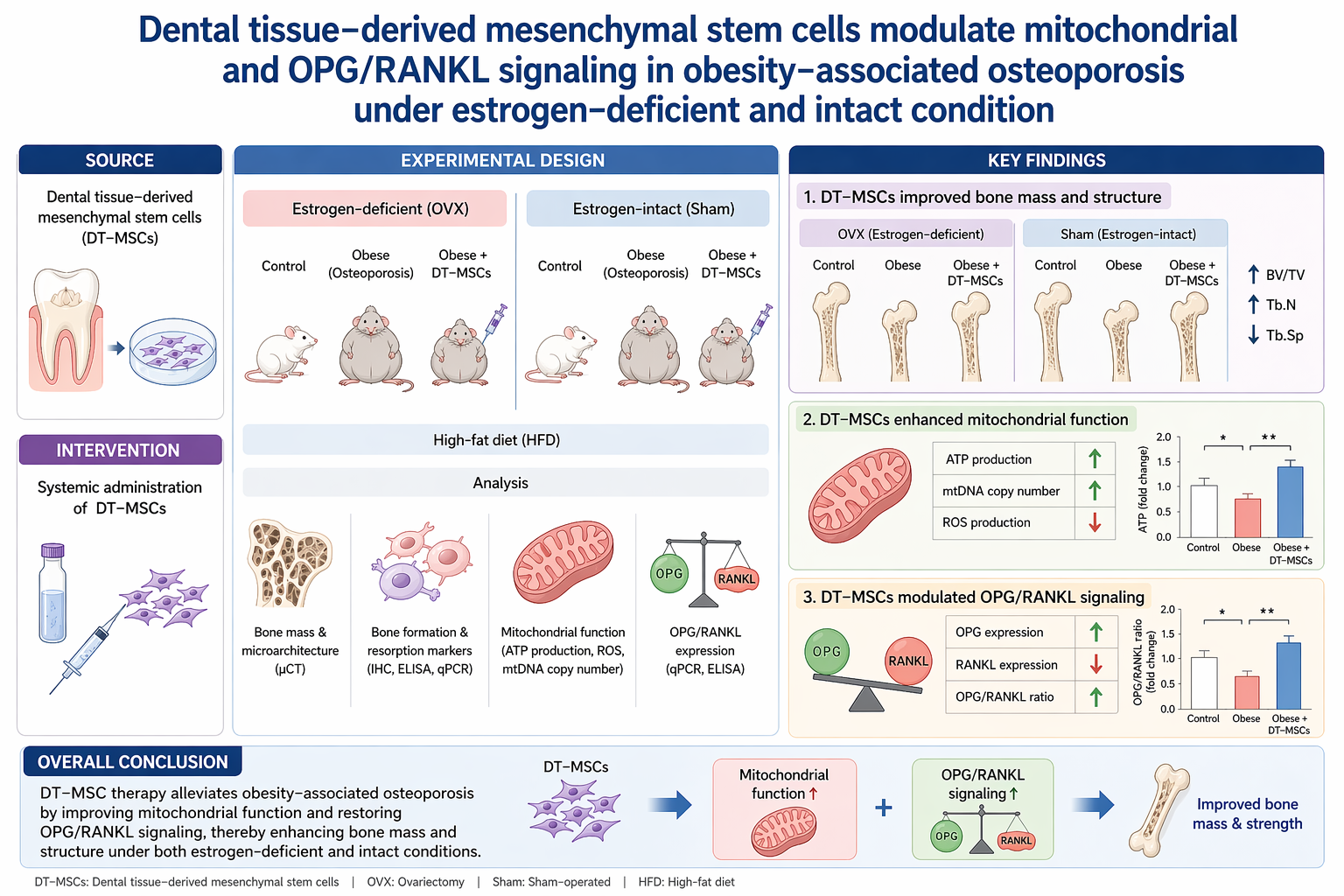
Keywords:
1. Introduction
2. Materials and Methods
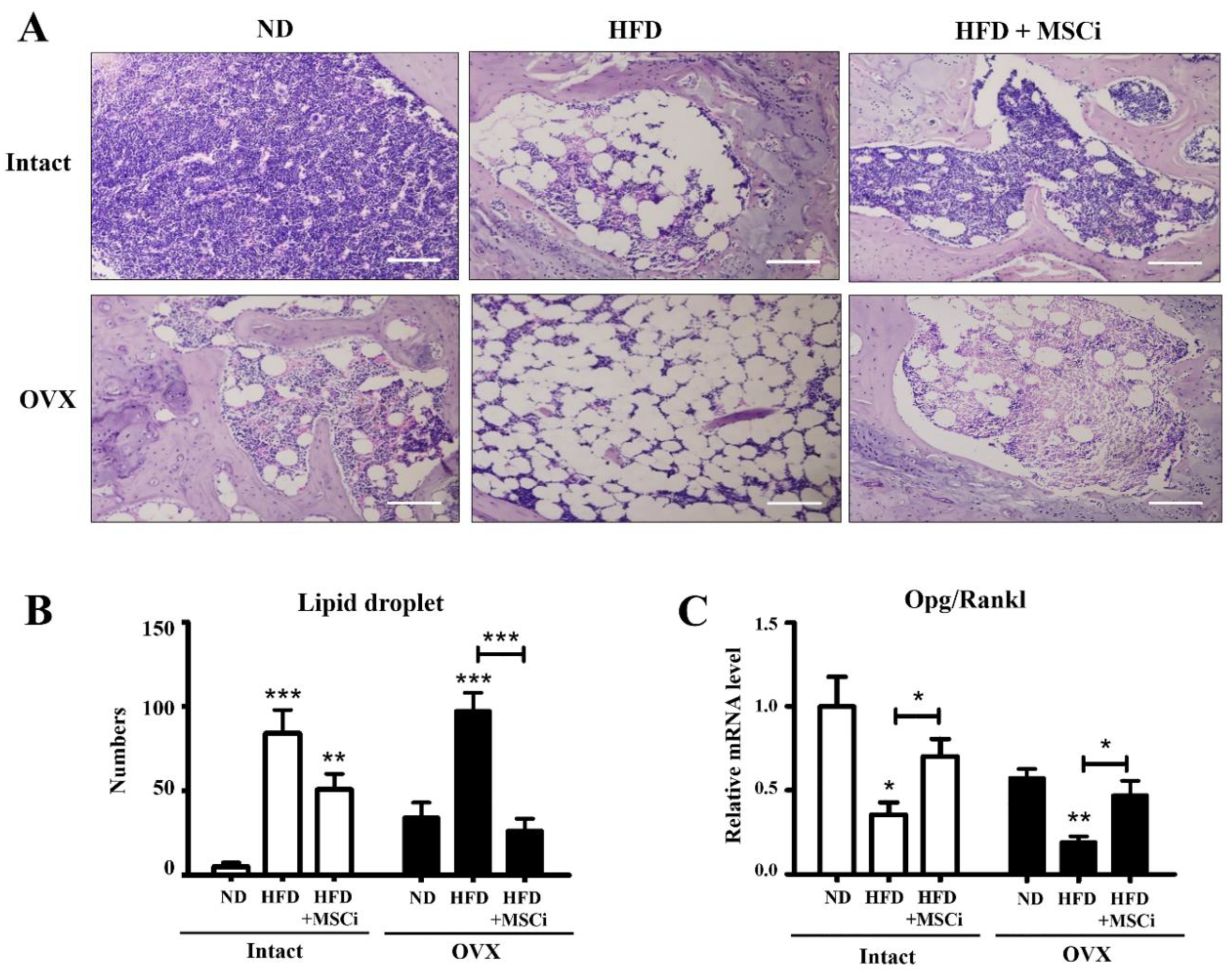
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| A-DMEM | Advanced Dulbecco’s Modified Eagle’s Medium |
| ALP | Alkaline phosphatase |
| D-MSCs | Dental tissue–derived mesenchymal stem cells |
| D-PBS | Dulbecco’s phosphate-buffered saline |
| ER | Estrogen receptor |
| ALP | Estrogen receptor alpha |
| D-MSCs | Estrogen receptor beta |
| D-PBS | Fetal bovine serum |
| ER | Fibroblast growth factor |
| ERα | High-fat diet |
| ERβ | Insulin-like growth factor-1 |
| FBS | Interleukin-1 beta |
| FGF | Mesenchymal stem cells |
| HFD | Alkaline phosphatase |
| IGF1 | Dental tissue–derived mesenchymal stem cells |
| Il-1β | Dulbecco’s phosphate-buffered saline |
| MSCs | Estrogen receptor |
| ND | Normal diet |
| OPG | Osteoprotegerin |
| OVX | Ovariectomized |
| Pgc1α | Peroxisome proliferator-activated receptor gamma coactivator-1 alpha |
| Pgc1β | Peroxisome proliferator-activated receptor gamma coactivator-1 beta |
| RANK | Receptor activator of nuclear factor-κB |
| RANKL | Receptor activator of nuclear factor-κB ligand |
| TGFβ | Transforming growth factor beta |
| Tnf-α | Tumor necrosis factor alpha |
References
- Ji, M.X.; Yu, Q. Primary osteoporosis in postmenopausal women. Chronic Dis. Transl. Med. 2015, 1, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D.; Rizzo, J.; Harris, E.; Cauley, J.; Ensrud, K.; Bauer, D.C.; Orwoll, E. Osteoporosis and fractures in postmenopausal women using estrogen. Arch. Intern. Med. 2002, 162, 2278–2284. [Google Scholar] [CrossRef] [PubMed]
- Tella, S.H.; Gallagher, J.C. Prevention and treatment of postmenopausal osteoporosis. J. Steroid Biochem. Mol. Biol. 2014, 142, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Baek, Y.; Park, K.; Lee, S.; Jang, E. The prevalence of general and abdominal obesity according to sasang constitution in Korea. BMC Complement. Altern. Med. 2014, 14, 298. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhou, W.; Gu, T.; Zhu, D.; Bi, Y. Association between obesity and cardiovascular risk. Sci. Rep. 2018, 8, 5806. [Google Scholar] [CrossRef] [PubMed]
- Bracht, J.R.; Vieira-Potter, V.J.; De Souza Santos, R.; Öz, O.K.; Palmer, B.F.; Clegg, D.J. The role of estrogens in the adipose tissue milieu. Ann. N. Y. Acad. Sci. 2020, 1461, 127–143. [Google Scholar] [CrossRef] [PubMed]
- van Gastel, N.; Carmeliet, G. Metabolic regulation of skeletal cell fate. Nat. Metab. 2021, 3, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Zhou, H.; Zeng, X.; Feng, S. Estrogen stimulates osteoprotegerin expression via the suppression of miR-145 expression in MG-63 cells. Mol. Med. Rep. 2017, 15, 1539–1546. [Google Scholar] [CrossRef]
- Schoppet, M.; Preissner, K.T.; Hofbauer, L.C. RANK ligand and osteoprotegerin. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Al-Masaoodi, R.A.; Al-Sallami, A.S.M.; Al-Baseesee, H. The relation between the RANKL and resistin in menopausal women with osteoporosis. AIP Conf. Proc. 2019, 2144, 040012. [Google Scholar] [CrossRef]
- Eghbali-Fatourechi, G.; Khosla, S.; Sanyal, A.; Boyle, W.J.; Lacey, D.L.; Riggs, B.L. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J. Clin. Invest. 2003, 111, 1221–1230. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, N.K.; Lee, S.Y. RANK signaling in osteoclast differentiation. Mol. Cells 2017, 40, 706–713. [Google Scholar] [CrossRef]
- Yu, B.; Huo, L.; Liu, Y.; Deng, P.; Szymanski, J.; Li, J.; Luo, X.; Hong, C.; Lin, J.; Wang, C.Y. PGC-1α controls skeletal stem cell fate. Cell. Stem Cell. 2018, 23, 193–209. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.X.; Sui, B.D.; Qiu, X.Y.; Hu, C.H.; Jin, Y. Mitochondrial regulation of stem cells. Trends Mol. Med. 2020, 26, 89–104. [Google Scholar] [CrossRef]
- Arjmand, B.; Sarvari, M.; Alavi-Moghadam, S.; Payab, M.; Goodarzi, P.; Gilany, K.; Mehrdad, N.; Larijani, B. Prospect of stem cell therapy and regenerative medicine in osteoporosis. Front. Endocrinol. 2020, 11, 430. [Google Scholar] [CrossRef]
- George, E.L.; Lin, Y.L.; Saunders, M.M. Bisphosphonate-related osteonecrosis of the jaw: A mechanobiology perspective. Bone Rep. 2018, 8, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Kennel, K.A.; Drake, M.T. Adverse effects of bisphosphonates: Implications for osteoporosis management. Mayo Clin. Proc. 2009, 84, 632–638. [Google Scholar] [CrossRef]
- Lechner, J.; von Baehr, V.; Zimmermann, B. Osteonecrosis of the jaw beyond bisphosphonates. Clin. Cosmet. Investig. Dent. 2021, 13, 21–37. [Google Scholar] [CrossRef]
- Vinogradova, Y.; Coupland, C.; Hippisley-Cox, J. Hormone replacement therapy and cancer risk. BMJ 2020, 371, m3873. [Google Scholar] [CrossRef] [PubMed]
- Barekzai, J.; Petry, F.; Zitzmann, J.; Czermak, P.; Salzig, D. Bioprocess development for human mesenchymal stem cell therapy products. New Adv. Ferment. Process. 2020, 1–25. [Google Scholar] [CrossRef]
- Liang, X.; Ding, Y.; Zhang, Y.; Tse, H.F.; Lian, Q. Paracrine mechanisms of mesenchymal stem cell-based therapy. Cell. Transplant. 2014, 23, 1045–1059. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, Z.; Jiang, D.; Liang, X.; Liao, S.; Zhang, Z.; Yue, W.; Li, X.; Chiu, S.M.; Chai, Y.H.; et al. Mitochondrial transfer by MSCs. Stem Cell. Rep. 2016, 7, 749–763. [Google Scholar] [CrossRef]
- Volponi, A.A.; Pang, Y.; Sharpe, P.T. Stem cell-based biological tooth repair. Trends Cell. Biol. 2010, 20, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.; Liu, Y.; Cui, D.; Pan, Y.; Zheng, L.; Wan, M. Dental tissue-derived human mesenchymal stem cells and their potential in therapeutic application. Stem Cells Int. 2020, 1–17. [Google Scholar] [CrossRef]
- Kang, Y.H.; Lee, H.J.; Jang, S.J.; Byun, J.H.; Lee, J.S.; Lee, H.C.; Park, W.U.; Lee, J.H.; Rho, G.J.; Park, B.W. Immunomodulatory properties and in vivo osteogenesis of human dental stem cells from fresh and cryopreserved dental follicles. Differentiation 2015, 90, 48–58. [Google Scholar] [CrossRef]
- Park, B.W.; Jang, S.J.; Byun, J.H.; Kang, Y.H.; Choi, M.J.; Park, W.U.; Lee, W.J.; Rho, G.J. Cryopreservation of human dental follicle tissue. J. Tissue Eng. Regen. Med. 2014, 11, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Kameda, T.; Mano, H.; Yuasa, T.; Mori, Y.; Miyazawa, K.; Shiokawa, M.; Nakamaru, Y.; Hiroi, E.; Hiura, K.; Kameda, A.; et al. Estrogen inhibits bone resorption by directly inducing apoptosis of osteoclasts. J. Exp. Med. 1997, 186, 489–495. [Google Scholar] [CrossRef]
- Bener, A.; Hammoudeh, M.; Zirie, M.; Heller, R.F. Is obesity a protective factor for osteoporosis? APLAR J. Rheumatol. 2005, 8, 32–38. [Google Scholar] [CrossRef]
- Cao, J.J. Effects of obesity on bone metabolism. J. Orthop. Surg. Res. 2011, 6, 30. [Google Scholar] [CrossRef]
- Zhao, L.J.; Liu, Y.J.; Liu, P.Y.; Hamilton, J.; Recker, R.R.; Deng, H.W. Relationship of obesity with osteoporosis. J. Clin. Endocrinol. Metab. 2007, 92, 1640–1646. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Kuhn, G.; Schirmer, M.; Müller, R.; Ruffoni, D. Impaired bone formation in ovariectomized mice reduces implant integration. PLoS ONE 2017, 12, e0184837. [Google Scholar] [CrossRef]
- Mathavan, N.; Turunen, M.J.; Tägil, M.; Isaksson, H. Characterising bone structure in ovariectomized rat model. Calcif. Tissue Int. 2015, 97, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Bi, Y.N.; Zhang, P.Y.; Yuan, X.M.; Liu, Y.; Zhang, Y.; Huang, J.Y.; Zhou, K. Optimization of the time window in ovariectomized mice. Biomed. Res. Int. 2017, 1–10. [Google Scholar] [CrossRef]
- Yousefzadeh, N.; Kashfi, K.; Jeddi, S.; Ghasemi, A. Ovariectomised rat model of osteoporosis. EXCLI J. 2020, 19, 89–107. [Google Scholar] [CrossRef]
- Dong, S.; Qi, M.; Wang, Y.; Chen, L.; Weaver, J.C.; Krilis, S.A.; Giannakopoulos, B. β2GPI exerts an anti-obesity effect in female mice by inhibiting lipogenesis and promoting lipolysis. Oncotarget 2017, 8, 92652–92666. [Google Scholar] [CrossRef]
- Totten, M.S.; Pierce, D.M.; Erikson, K.M. Diet-induced obesity disrupts gene expression. Nutrients 2020, 12, 1401. [Google Scholar] [CrossRef]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the obese gene. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Vellapandian, C.; Sukumaran, E. S.; Sivasubramanian, L. R.; Rajabatar Vetrivelan, V. A comparative study of actinidia deliciosa and garcinia mangostana in ovariectomy- induced osteoporosis in female Wistar rats. In BioMed Research International; 2017; Volume 2017, pp. 1–10. [Google Scholar] [CrossRef]
- Boyce, B.F.; Xing, L. The RANKL/RANK/OPG pathway. Curr. Osteoporos. Rep. 2007, 5, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Tobeiha, M.; Moghadasian, M.H.; Amin, N.; Jafarnejad, S. RANKL/RANK/OPG pathway. Biomed. Res. Int. 2020, 1–11. [Google Scholar] [CrossRef]
- Chen, T.Y.; Zhang, Z.M.; Zheng, X.C.; Wang, L.; Huang, M.J.; Qin, S.; Chen, J.; Lai, P.L.; Yang, C.L.; Liu, J.; et al. Endogenous n-3 polyunsaturated fatty acids mitigate ovariectomy-induced bone loss by attenuating bone marrow adipogenesis in FAT1 transgenic mice. Drug. Des. Dev. Ther. 2013, 7, 545–552. [Google Scholar] [CrossRef]
- Fan, C.M.; Su, Y.W.; Howe, P.R.; Xian, C.J. Long chain omega-3 polyunsaturated fatty acid supplementation protects against chemotherapy-induced bone marrow damage in female rats. Int. J. Mol. Sci. 2018, 19, 484. [Google Scholar] [CrossRef]
- Miceli, V.; Bulati, M.; Iannolo, G.; Zito, G.; Gallo, A.; Conaldi, P.G. Therapeutic properties of MSCs. Int. J. Mol. Sci. 2021, 22, 2400. [Google Scholar] [CrossRef]
- Morsczeck, C.; Götz, W.; Schierholz, J.; Zeilhofer, F.; Kühn, U.; Möhl, C.; Sippel, C.; Hoffmann, K.H. Isolation of precursor cells from human dental follicle. Matrix Biol. 2005, 24, 155–165. [Google Scholar] [CrossRef]
- Ding, G.; Liu, Y.; An, Y.; Zhang, C.; Shi, S.; Wang, W.; Wang, S. Suppression of T cell proliferation by root apical papilla stem cells in vitro. Cells Tissues Organs 2010, 191, 357–364. [Google Scholar] [CrossRef]
- Yamaza, T.; Kentaro, A.; Chen, C.; Liu, Y.; Shi, Y.; Gronthos, S.; Wang, S.; Shi, S. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell. Res. Ther.;Stem Cell. Res. Ther. 2010, 1 1(1), 5.us t. [Google Scholar] [CrossRef] [PubMed]
- Del Angel-Mosqueda, C.; Gutiérrez-Puente, Y.; López-Lozano, A.P.; Romero-Zavaleta, R.E.; Mendiola-Jiménez, A.; Medina-De la Garza, C.E.; Márquez, M.; De la Garza-Ramos, M.A. Epidermal growth factor enhances osteogenic differentiation of dental pulp stem cells in vitro. Head. Face Med. 2015, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Navabazam, A.R.; Nodoshan, F.S.; Sheikhha, M.H.; Miresmaeili, S.M.; Solcimani, M.; Fesahat, F. Characterization of mesenchymal stem cells from human dental pulp, preapical follicle and periodontal ligament. Iran. J. Reprod. Med. 2013, 11, 235–242. [Google Scholar] [PubMed]
- Lee, R. H.; Kim, B. C.; Choi, I. S.; Kim, H.; Choi, H. S.; Suh, K. T.; Bae, Y. C.; Jung, J. S. Characterization and expression analysis of mesenchymal stem cells from human bone marrow and adipose tissue. Cell. Physiol. Biochem. 2004, 14(4–6), 311–324. [Google Scholar] [CrossRef]
- Lee, W. J.; Hah, Y. S.; Ock, S. A.; Lee, J. H.; Jeon, R. H.; Park, J. S.; Lee, S. Il; Rho, N. Y.; Rho, G. J.; Lee, S. L. Cell source-dependent in vivo immunosuppressive properties of mesenchymal stem cells derived from the bone marrow and synovial fluid of minipigs. Exp. Cell. Res. 2015, 333(2), 273–288. [Google Scholar] [CrossRef]
- Campos, J. M.; Sousa, A. C.; Caseiro, A. R.; Pedrosa, S. S.; Pinto, P. O.; Branquinho, M. V.; Amorim, I.; Santos, J. D.; Pereira, T.; Mendonça, C. M.; Afonso, A.; Atayde, L. M.; Maurício, A. C. Dental pulp stem cells and Bonelike® for bone regeneration in ovine model. Regen. Biomater. 2019, 6(1), 49–59. [Google Scholar] [CrossRef]
- Fujii, Y.; Kawase-Koga, Y.; Hojo, H.; Yano, F.; Sato, M.; Chung, U. Il; Ohba, S.; Chikazu, D. Bone regeneration by human dental pulp stem cells using a helioxanthin derivative and cell-sheet technology. Stem Cell. Res. Ther. 2018, 9(1), 6–7. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Magdaleno DDS, M.; Isabel Romo-Tobías DDS, A.; Romo-Ramírez DDS, F.; María Escobar DDS, D.; Flores-Reyes DDS, H.; Pozos-Guillén DDS, A. Behavior of mesenchymal stem cells obtained from dental tissues: A review of the literature Comportamiento de células mesenquimales obtenidas de tejid. Rev. Lit.-ODOVTOS-Int. J. Dent. Sc. 2018, 21(1), 31–40. [Google Scholar] [CrossRef]
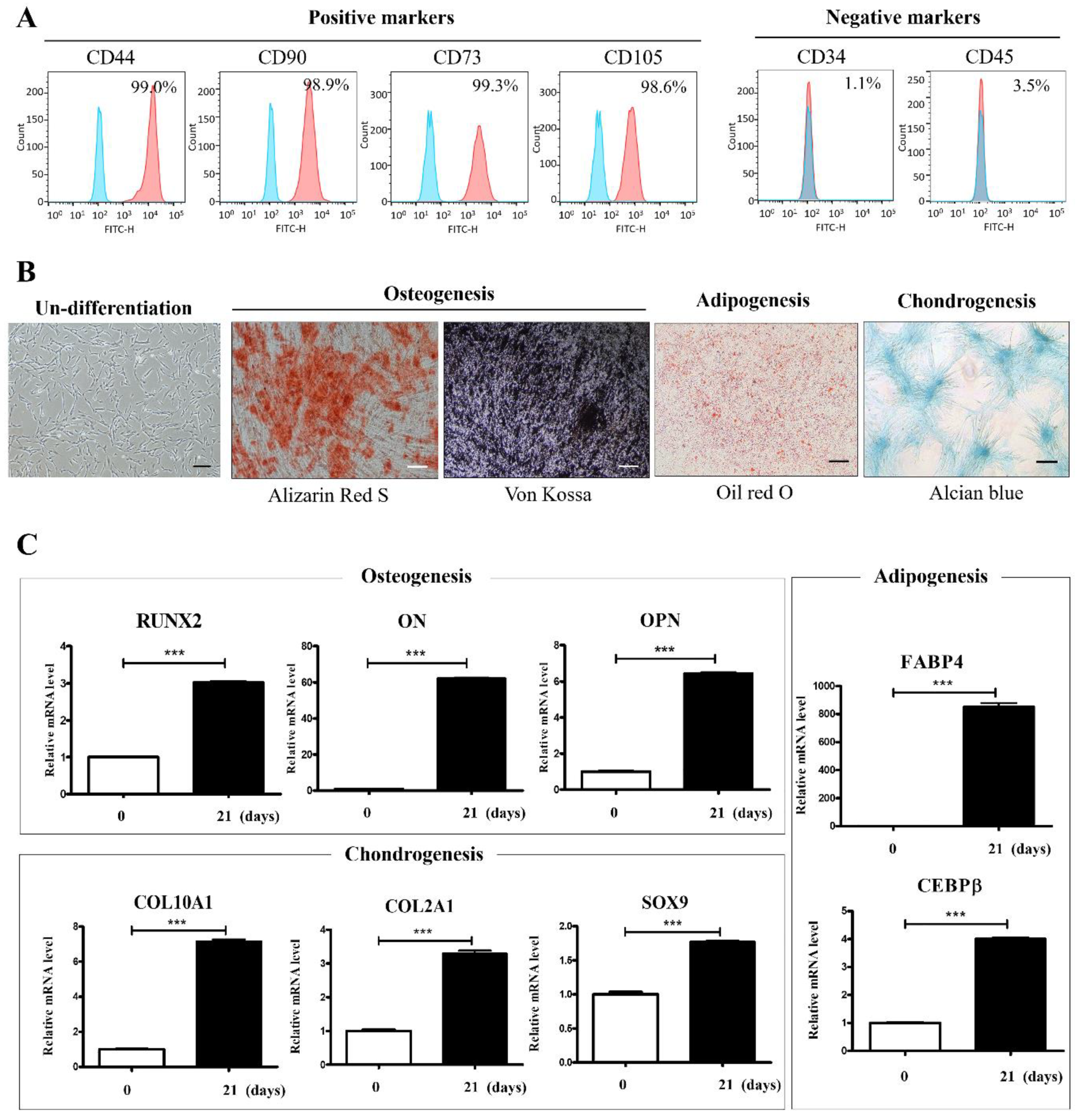
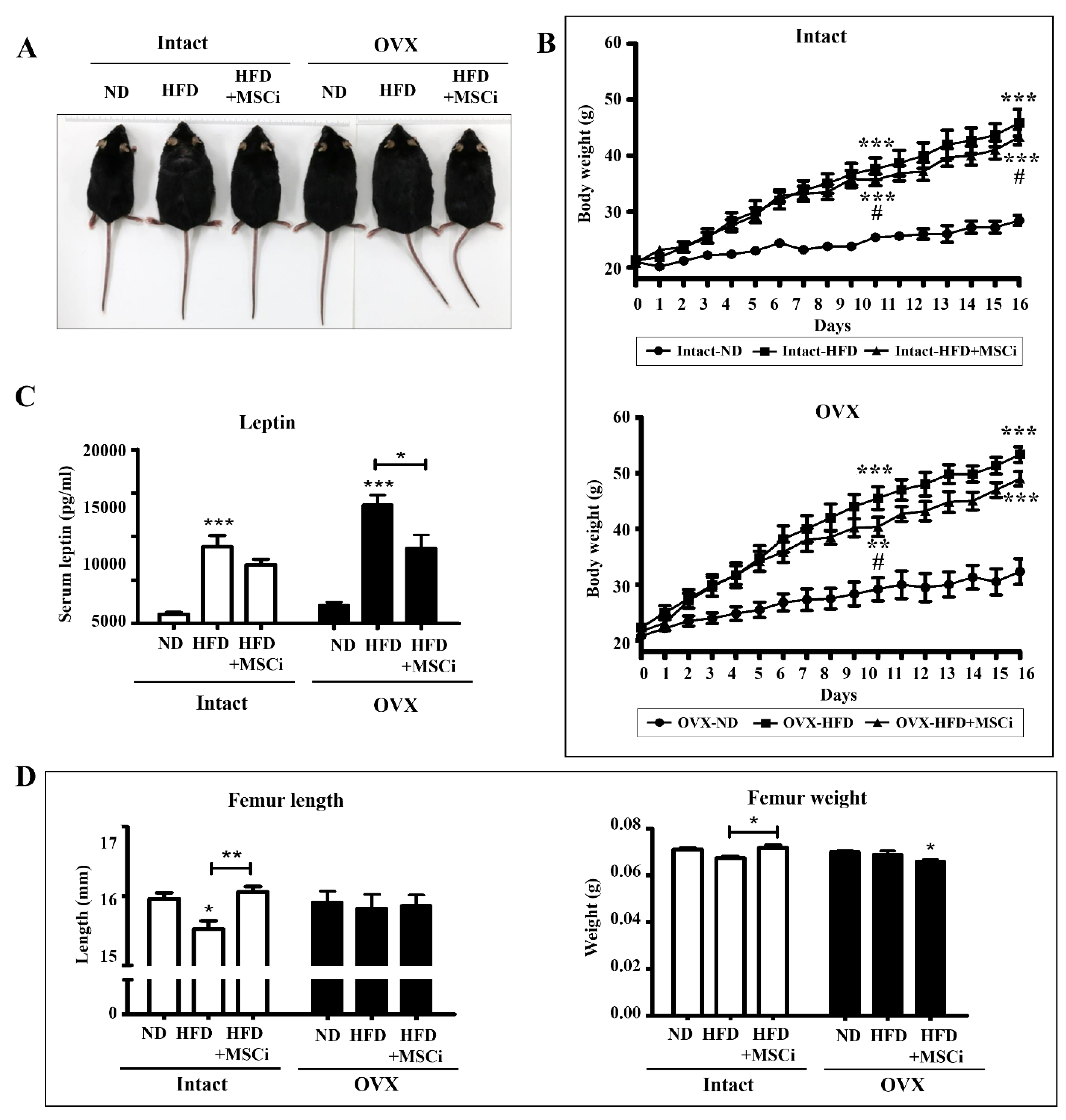
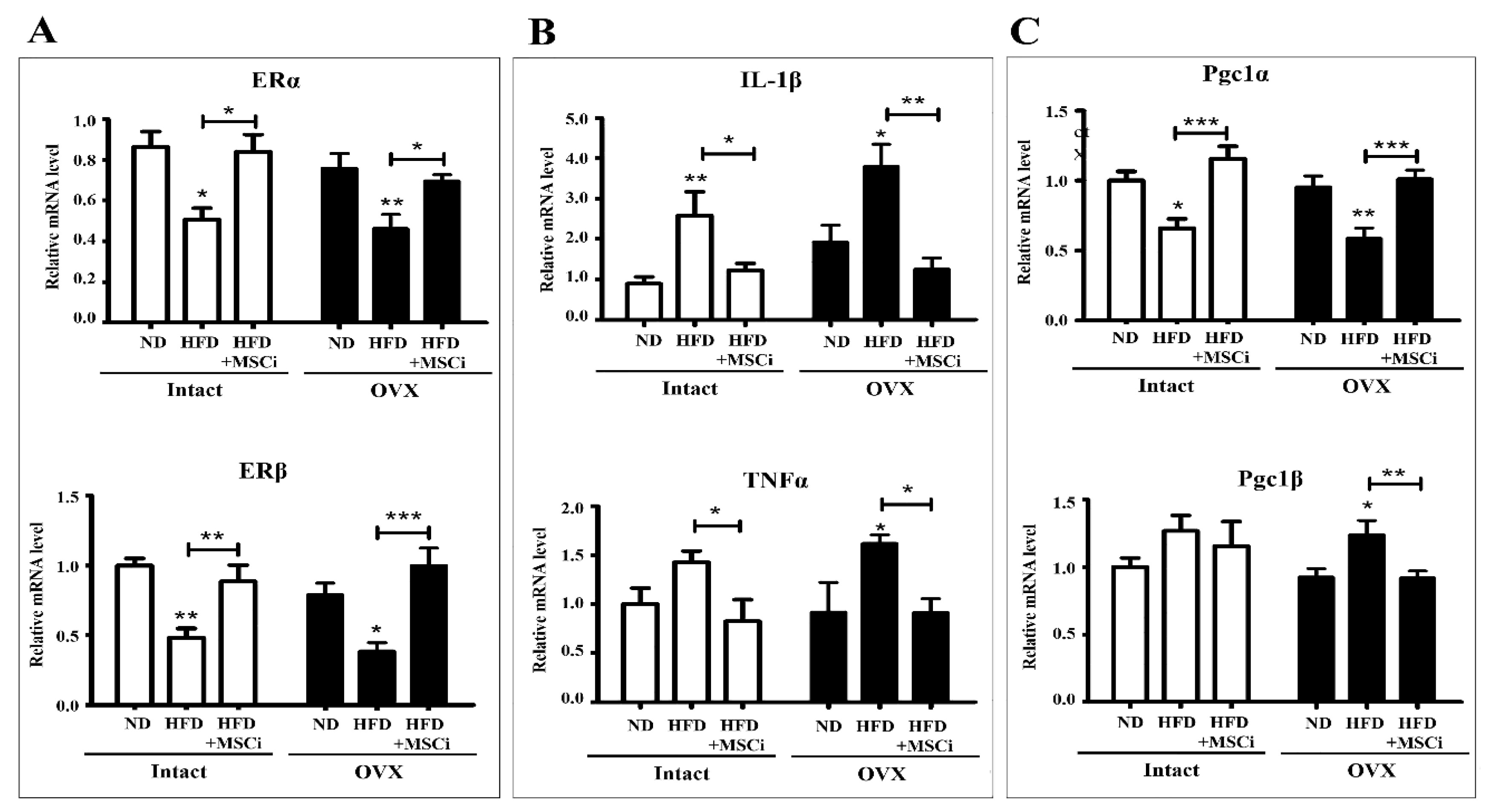
| Antibody | Company | Amount | Dilution |
| FITC mouse anti-human CD34 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| FITC mouse anti-human CD45 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| FITC rat anti-human CD44 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| FITC mouse anti-human CD90 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| APC mouse anti-human CD73 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| APC mouse anti-human CD105 | BD PharmingenTM | 0.5 mg/ml | 1:100 |
| Target gene | Sequence (5′-3′) | Product size (bp) | Anneal. Tm. (°C) | Reference |
|
Estrogen receptor-α |
F-AAGCGTCAGAGAGATGACTTGG R-CAGGGCTATTCTTCTTAGTGTGC |
118 | 60 | LC260510.1 |
|
Estrogen receptor-β |
F-CAACTCGTTTCGCATTCCTACC R-AGTGACCACATTCAGACAGACC |
184 | 60 | NM_207707.1 |
| Il-1β | F-ATGACCTGTTCTTTGAAGTTGACG R-CCTGAAGCTCTTGTTGATGTGC |
128 | 60 | BC011437.1 |
| Tnf-α | F- ATGAGCACAGAAAGCATGATCC R-ATGAGAAGAGGCTGAGACATAGG |
112 | 60 | NM_001278601.1 |
| Pgc1α | F-TCTTCCTTTAACTCTCCGTGTCG R-TGACCTGGAATATGGTGATCGG |
138 | 60 | BC066868.1 |
| Pgc1β |
F-GGACTGAGTTCTCTATCCTAAGGG R-GTGTGAGGGAAGCATAGACAGG |
102 | 60 | NM_133249.3 |
| OPG | F-TGGACATCATTGAATGGACAAC R-TATAAGAGTGGTCAGGGCAAG |
174 | 60 | U94331.1 |
| RANKL | F-CCGAGCTGGTGAAGAAATTAG R-TCTATGTCCTGAACTTTGAAAG |
102 | 60 | AF019048.1 |
| TBP | F-AGTGAAGAACAATCCAGACTAG R-TATAGGGAACTTCACATCACA |
129 | 60 | NM_013684.3 |
| Target gene | Sequence (5′-3′) | Product size (bp) | Anneal. Tm. (°C) | Reference |
| RUNX2 | F-CTCTACTATGGCACTTCGTCAGG R-TTTAATAGCGTGCTGCCATTCG |
119 | 60 | NM_001015051.4 |
| ON | F-GTGCAGAGGAAACCGAAGAG R-AAGTGGCAGGAAGAGTCGAA |
130 | 60 | NM_03118.2 |
| OPN | F- TTGCAGCCTTCTCAGCCAA R-GGAGGCAAAAGCAAATCACTG |
102 | 60 | NM_001040058 |
| FABP4 | F- GGAAAGTCAAGAGCACCATAACC R-CATTCCACCACCAGTTTATCATCC |
118 | 60 | NM_001442.2 |
| CEBPβ | F-TTTGTCCAAACCAACCGCACAT R-CAGAGGGAGAAGCAGAGAGTTTA |
110 | 60 | NM_001285879.1 |
| COL10A1 |
F-AACAGGCAACAGCATTATGACC R-AAACATGAGTCCCTTTCACATGC |
103 | 60 | NM_000493.4 |
| COL2A2 | F-AGGAATTCGGTGTGGACATAGG R-GGAAAGTACTTGGGTCCTTTGG |
100 | 60 | NM_033150.2 |
| SOX9 | F-GACCTTTGGGCTGCCTTATATTG R-CTCCCTCACTCCAAGAGAAGATG |
116 | 60 | NM_000346.3 |
| YWHAZ | F-CGAAGCTGAAGCAGGAGAAG R-TTTGTGGGACAGCATGGATG |
111 | 60 | NM_003406.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




