Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Electrode Preparation and PbO₂ Electrodeposition
2.3. Surface Characterization and Electrochemical Measurements
3. Results and Discussion
3.1. Morphology of PbO₂ Coatings
3.2. Tafel Analysis and Kinetics of the Anodic Process
| Electrode | a(V) | b (V/Dec) | R² |
| Graphite | 2.129 | 0.323 | 0.984 |
| Ti/ PbO ₂ | 2.153 | 0.315 | 0.988 |
| PLA (7% mCNT)/ PbO ₂ | 2.182 | 0.331 | 0.992 |
| PLA (10% mCNT)/ PbO ₂ | 2.175 | 0.327 | 0.991 |
| PLA (Graphite) / PbO ₂ | 1.619 | 0.195 | 0.993 |
3.3. Operating Potential and Energy Efficiency
3.4. Specific Charge and Ohmic Resistance
3.5. Stability and Reproducibility over CV Cycles
3.6. Comprehensive Comparative Assessment of the Electrodes
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| mCNT | Multi-walled carbon nanotubes |
| PLA | Polylactic Acid |
| CV | Cyclic voltammetry |
References
- Cui, J.; Zhang, Y.; Wang, Y.; Li, Z.; Liu, B.; Zhao, Y. A Comprehensive Review of PbO₂ Electrodes in Electrocatalytic Degradation of Organic Pollutants: Recent Advances, Challenges and Prospects. Environmental Research 2025, 279, 121885. [Google Scholar] [CrossRef]
- Guo, H.; Hu, W.; Xu, Z.; Guo, S.; Qiao, D.; Wang, X.; Xu, H.; Yan, W. How to Improve Lead Dioxide Anodes Performance in Organic Wastewater Treatment: Review and Prospect. Process Safety and Environmental Protection 2022, 161, 599–621. [Google Scholar] [CrossRef]
- Li, X.; Cui, Y.; Feng, Y.; Xie, Z.; Li, Y. Application of Lead Oxide Electrodes in Wastewater Treatment: A Review. Science of the Total Environment 2022, 808, 152110. [Google Scholar] [CrossRef]
- Yu, B.; Xu, R.; Wang, X.; He, S.; Chen, B. Electrodeposition of MnO ₂ Doped Pb–0.6%Sb/ α PbO ₂/ β PbO₂ Novel Composite Energy Saving Anode for Zinc Electrowinning. Journal of Energy Storage 2023, 61, 106264. [Google Scholar] [CrossRef]
- Feng, Z.; Yang, L.; Liu, J.; Wang, Y.; Chen, X. Electrochemical Technologies for Wastewater Treatment and Resource Reclamation. Environmental Science: Water Research & Technology 2016, 2, 800–831. [Google Scholar] [CrossRef]
- Alon Davidy CFD Simulation of H2S Desulfurization Using Ionic Liquids and Graphene Oxide Membrane // Fuels (MDPI) — 2023. [CrossRef]
- Velichenko, A.B.; Amadelli, R.; Gruzdeva, E.V.; Luk'yanenko, T.V.; Danilov, F. I. Electrodeposition of Lead Dioxide from Methanesulfonate Solutions. Journal of Power Sources 2009, 191, 103–110. [Google Scholar] [CrossRef]
- Martínez -Huitle, C.A.; Rodrigo, M.A.; Sires, I.; Scialdone, O. Single and Coupled Electrochemical Processes and Reactors for the Abatement of Organic Water Pollutants: A Critical Review. Chemical Reviews 2015, 115, 13362–13407. [Google Scholar] [CrossRef]
- Chen, G. Electrochemical Technologies in Wastewater Treatment. Separation and Purification Technology 2004, 38, 11–41. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G. Fabrication and Electrochemical Treatment Application of a Novel Lead Dioxide Anode with Superhydrophobic Surfaces, High Oxygen Evolution Potential, and Oxidation Capability. Environmental Science & Technology 2010, 44, 1754–1759. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Dai, Q.; Lei, L.; Ma, C.; Yu, P. Long Life Modified Lead Dioxide Anode for Organic Wastewater Treatment: Electrochemical Characteristics and Degradation Mechanism. Environmental Science & Technology 2005, 39, 363–370. [Google Scholar] [CrossRef]
- Electrochemistry for the Environment; Comninellis, C., Chen, G., Eds.; Springer: New York, NY, USA, 2010; p. 578. [Google Scholar] [CrossRef]
- Vassiliev, V. P.; Lysenko, V. A. A New Approach for the Study of Thermodynamic Properties of Lanthanide Compounds. Electrochimica Acta 2016, 222, 1770–1777. [Google Scholar] [CrossRef]
- Liang, Z.; Liu, J.; Huang, W.; Wang, Y.; Xu, H. Electrochemical Oxidation of Sulfide on a Ti/PbO₂Electrode in Alkaline Solution. Electrochimica Acta 2011, 56, 8851–8856. [Google Scholar] [CrossRef]
- Amadelli, R.; Armelao, L.; Velichenko, A.B.; Girenko, D.V.; Kovalyov, S.; Franchuk, A.G.; Danilov, F.I. Oxygen and Ozone Evolution at Fluoride Modified Lead Dioxide Electrodes. Electrochimica Acta 1999, 45, 713–720. [Google Scholar] [CrossRef]
- Chen, S.; Chu, X.; Wu, L.; Foord, J. S.; Hu, J.; Hou, H.; Yang, J. Three-Dimensional PbO2-Modified Carbon Felt Electrode for Efficient Electrocatalytic Oxidation of Phenol Characterized with In Situ ATR-FTIR. The Journal of Physical Chemistry C 2022, 126(2), 912–921. [Google Scholar] [CrossRef]
- Duan, X.; Ning, Z.; Wang, W.; et al. Y-mediated optimization of 3DG-PbO2 anode for electrochemical degradation of PFOS. BMC Chemistry 2023, 17, 146. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, H.; Wang, Y.; Li, X.; Zhang, J. Electrochemical Oxidation Treatment of Coal Tar Wastewater with Lead Dioxide Anodes. Water Science and Technology 2020, 81, 1820–1829. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Application of Diamond Electrodes to Electrochemical Processes. Electrochimica Acta 2005, 51, 191–199. [Google Scholar] [CrossRef]
- Neeraj, V.S.; Yashwanth, M.; Pavan Kalyan, B.; Appusamy, Arunagiri; Muthukumar, Karuppan. Improved kitchen wastewater treatment using PbO2-coated graphite electrode. Journal of the Indian Chemical Society 2024, Volume 101(Issue 11), 101453. [Google Scholar] [CrossRef]
- Bi, Qiang; Sun, Yifei; Yang, Bo; Zhao, Yilin; Zhang, Zekun; Xue, Juanqin. Preparation of an ultra-long-life porous bilayer Ti/Sb-SnO2 electrode modified by nano- TiC for degradation of phenol. Materials Today Communications, Volume 2023, Volume 35, 106307. [Google Scholar] [CrossRef]
- Contreras-Naranjo, J. E.; Pérez-González, V. H.; Mata-Gómez, M.A.; Aguilar, O. 3D Printed HybridCarbonBased Electrodes for Electroanalytical Sensing Applications. Electrochimica Acta 2021, 388, 138611. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Y.; Liu, Q.; Li, Y. Affordable PbO₂ Anode on Conductive Polymer–Carbon Composite Material for Wastewater Treatment. Journal of Electroanalytical Chemistry 2020, 877, 114610. [Google Scholar] [CrossRef]
- Cardoso, R. M.; Silva, P.R.L.; Lima, A.P.; Rocha, D.P.; Richter, E.M.; Muñoz, R.A.A. 3D Printed Graphene/Polylactic Acid Electrode for Bioanalysis: Biosensing of Glucose and Simultaneous Determination of Uric Acid and Nitrite. Sensors and Actuators B: Chemical 2020, 307, 127621. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Sheridan, E.; Gilmartin, N.; Killard, A.D. Mixed Graphite/Carbon Black Recycled PLA Conductive Additive Manufacturing Filament for the Electrochemical Detection of Oxalate. Analytical Chemistry 2023, 95, 14753–14761. [Google Scholar] [CrossRef]
- Vaněčková, Eva; Bouša, Milan; Nováková Lachmanová, Stěpánka; Rathouský, Jiří; Gál, Miroslav; Sebechlebská, Táňa; Kolivoška, Viliam. 3D printed polylactic acid/carbon black electrodes with nearly ideal electrochemical behavior. Journal of Electroanalytical Chemistry 2020, Volume 857, 113745. [Google Scholar] [CrossRef]
- Richter, E. M.; Rocha, D.P.; Cardoso, R. M.; Tormin, T.F.; de Oliveira, A.P.; Muñoz, R.A.A. Complete Additively Manufactured (3D Printed) Electrochemical Sensing Platform. Analytical Chemistry 2019, 91, 12844–12851. [Google Scholar] [CrossRef] [PubMed]
- Filimonova, A.A.; Vlasova, A.Yu.; Kamalieva, R.F.; Mayorov, E.S.; Filimonov, A.A. Creation Electricity supply Composite with Polymers Materials for 3D Printing Basis Surface Modifications. Bulletin Voronezhsky State University Engineering Tekhnologiy 2025, 87, 241–248. [Google Scholar] [CrossRef]
- Martínez- Huitle, C.A.; Brillas, E. Decontamination of Wastewaters Containing Synthetic Organic Dyes by Electrochemical Methods: A General Review. Applied Catalysis B: Environmental 2009, 87, 105–145. [Google Scholar] [CrossRef]
- Farkhutdinov, M.M.; Fetisov, L.V. Materialy, Primenyaemye for 3D Printing in Electrical Engineering Devices. Modern Innovatsii 2020, 2(30), 11–17. [Google Scholar] [CrossRef]
- Brillas, E.; Sires, I.; Oturan, M.A. Electro Fenton Process and Related Electrochemical Technologies Based on Fenton's Reaction Chemistry. Chemical Reviews 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Yu, B.; Xu, R.; Wang, X.; Wang, W.; Feng, S. Study of simultaneously electrodepositing α/β-PbO2 coating materials in methanesulfonic acid and its application in novel flow battery. Renewable Energy 2020, 159, 885–892. [Google Scholar] [CrossRef]
- He, Z.; Hayat, M. D.; Huang, S.; Wang, X.; Cao, P. Physicochemical Characterization of PbO2 Coatings Electrosynthesized from a Methanesulfonate Electrolytic Solution. Journal of The Electrochemical Society 2018, 165(14), D670–D675. [Google Scholar] [CrossRef]
- Filimonova, A.A.; Chichirov, A.A.; Khairutdinov, A.M. Study of the Influence of Electrode Material and Electrolyte Composition on Cathode and Anodic Overvoltage during Electrolysis. Surf. Engin. Appl.Electrochem 2025, 61, 832–841. [Google Scholar] [CrossRef]
- Sires, I.; Brillas, E.; Oturan, M.A.; Rodrigo, M.A.; Panizza, M. Electrochemical Advanced Oxidation Processes: Today and Tomorrow. Environmental Science and Pollution Research 2014, 21, 8336–8367. [Google Scholar] [CrossRef]
- Radjenovic, J.; Sedlak, D. L. Challenges and Opportunities for Electrochemical Processes as Next Generation Technologies for the Treatment of Contaminated Water. Environmental Science & Technology 2015, 49, 11292–11302. [Google Scholar] [CrossRef]
- Anglada, B.; Urtiaga, A.; Ortiz, I. Contributions of Electrochemical Oxidation to Wastewater Treatment: Fundamentals and Review of Applications. Journal of Chemical Technology and Biotechnology 2009, 84, 1747–1755. [Google Scholar] [CrossRef]
- Manzanares- Palenzuela, C.L.; Kadara, R.O.; Jenkinson, N.; Rawson, F. J. 3D Printed Graphene/Polylactic Acid Electrodes Promise High Sensitivity in Electroanalysis. Analytical Chemistry 2018, 90, 5753–5759. [Google Scholar] [CrossRef]
- Foster, C. W.; Down, M.P.; Zhang, Y.; Ji, X.; Rowley-Neale, S. J.; Smith, G. C.; Kelly, P.J.; Banks, C.E. 3D Printed Graphene Based Energy Storage Devices. Electroanalysis 2017, 29, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Bi, H.; Yu, C.; Gao, W.; Cao, P. Physicochemical Characterization of Electrosynthesized PbO2Coatings: The Effect of Pb2+Concentration and Current Density. Journal of The Electrochemical Society 2014, 161(6), D327–D332. [Google Scholar] [CrossRef]
- Zhao, W.; Xing, J.; Chen, D.; Shen, J. Comparative studies on the performance of porous Ti/Sno2-Sb2O3/Pbo2 enhanced by CNT and Bi Co-doped electrodes for methyl orange oxidation. Journal of Advanced Oxidation Technologies 2017, 20(1). [Google Scholar] [CrossRef]
- Filimonova, A.A.; Vlasova, A.Yu.; Mayorov, E.S.; Filimonov, A.A.; Kamalieva, R.F.; Khayrutdinov, A.M. Sozdanie elektroprovodyashchego kompozita na osnove materialov dlya 3D-pechati. Khimicheskaya Promyshlennost' Segodnya 2026, 1, 33–43. [Google Scholar]

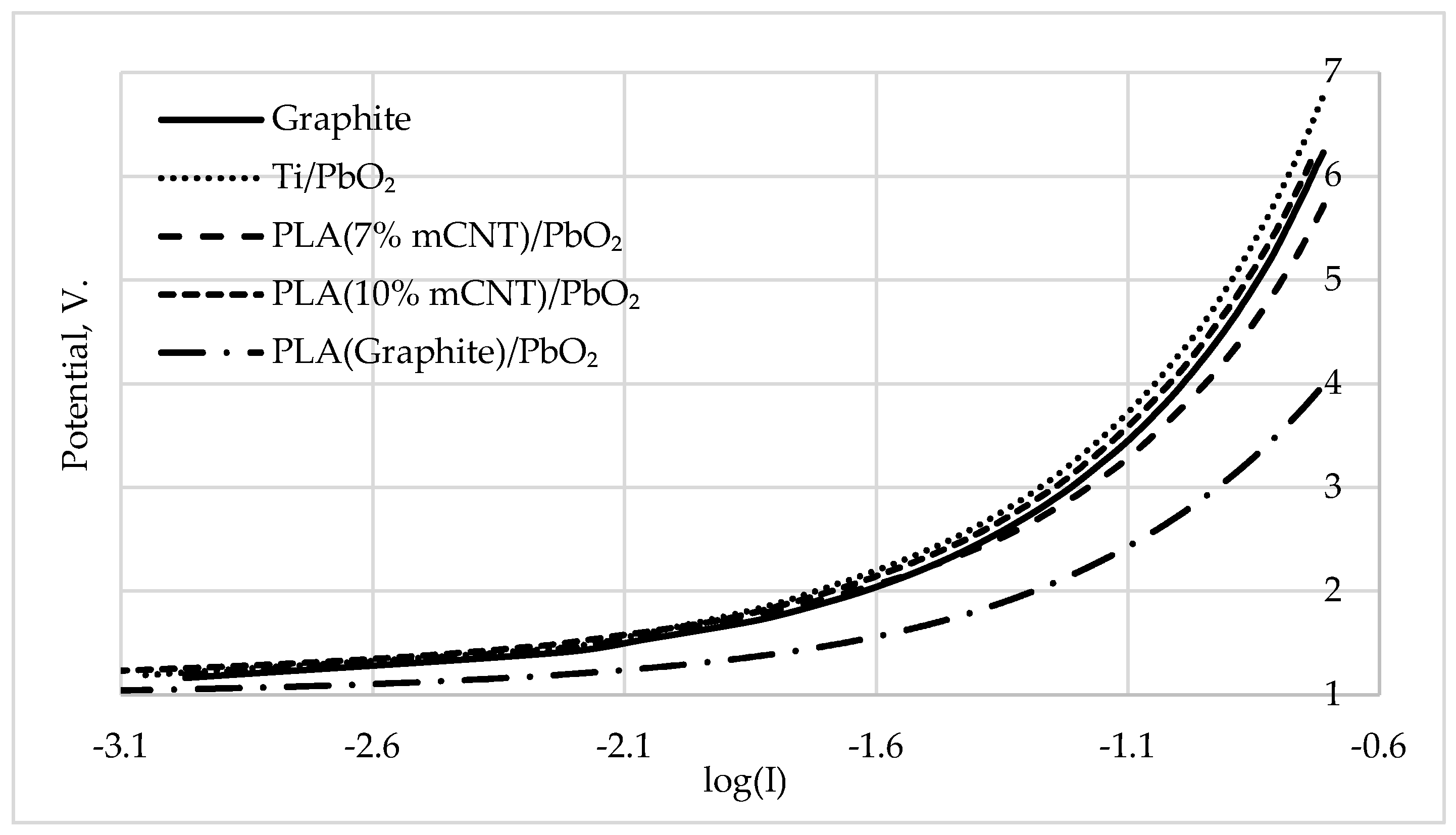
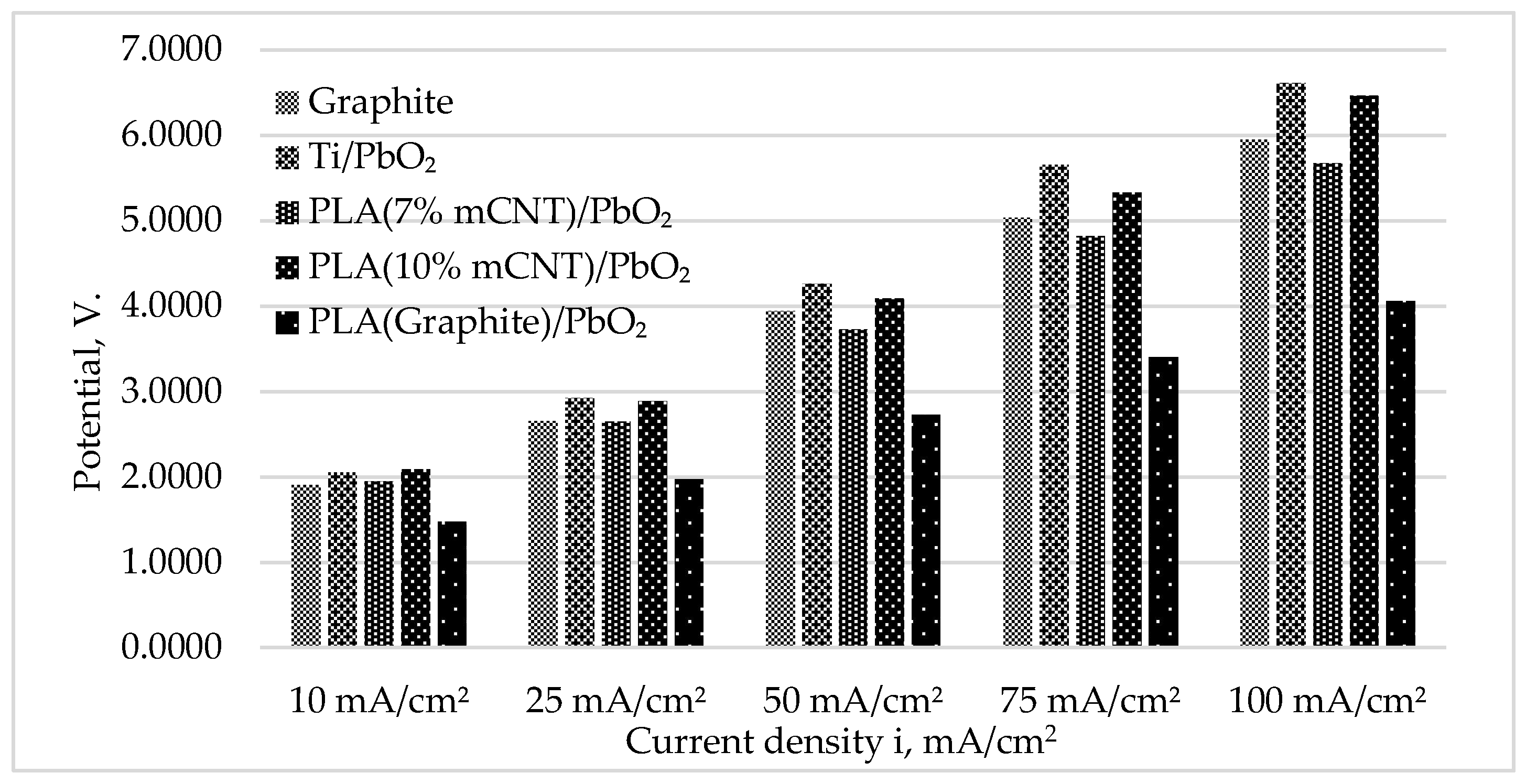
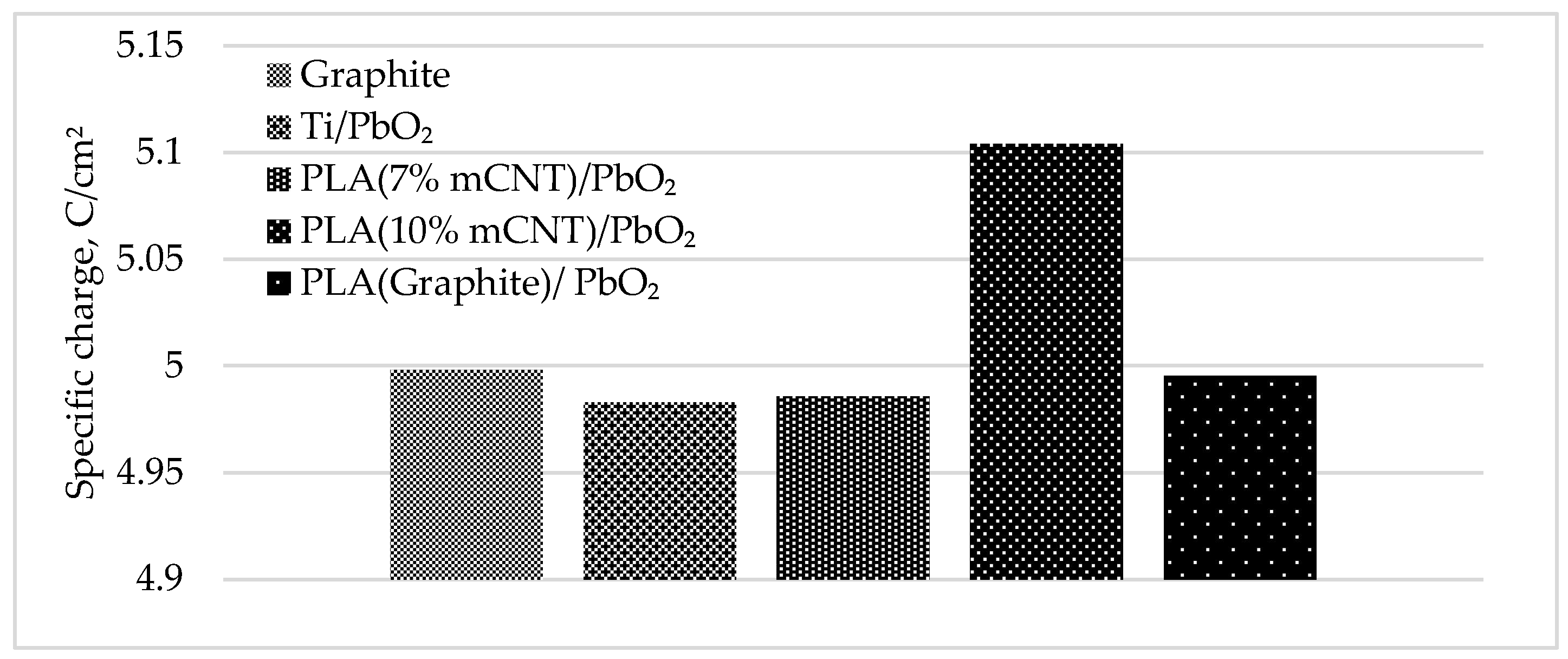
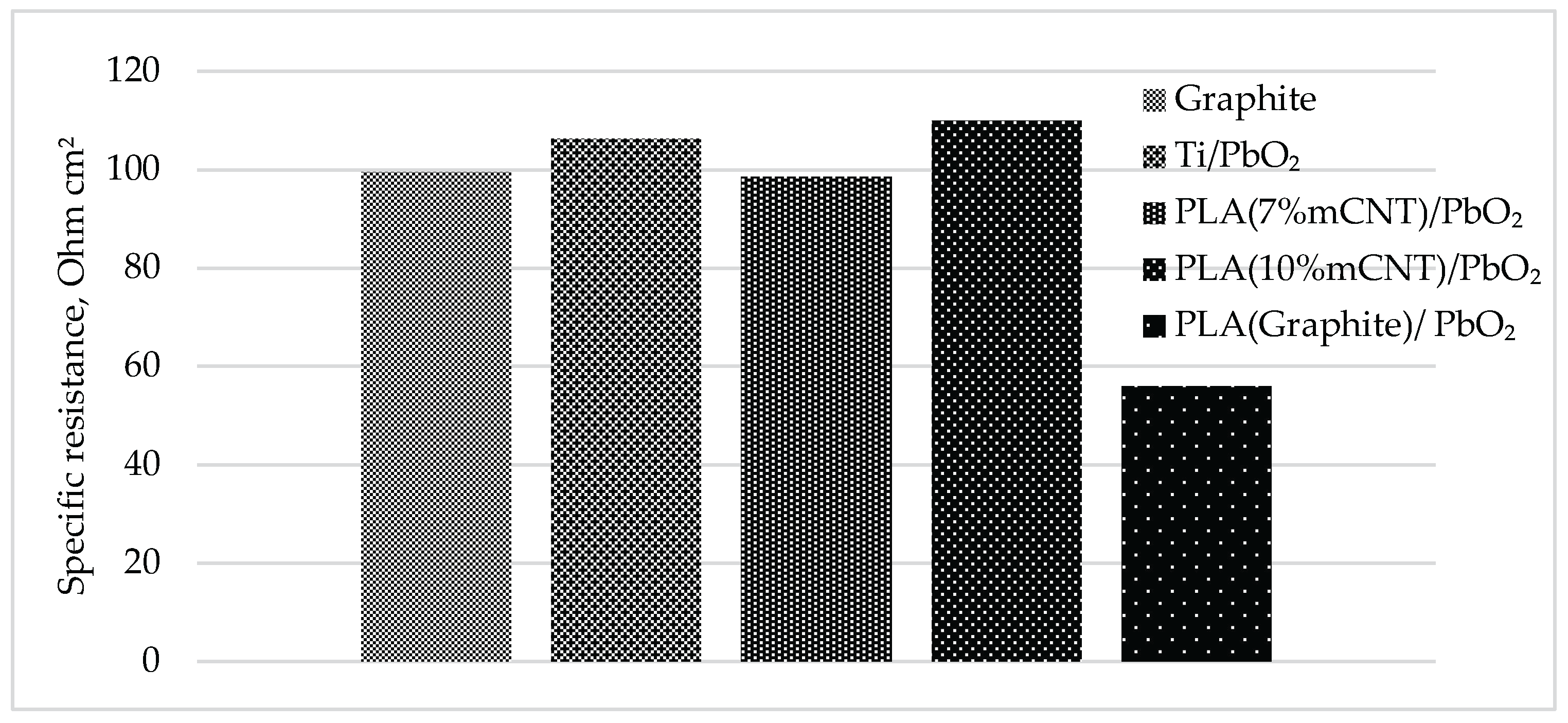
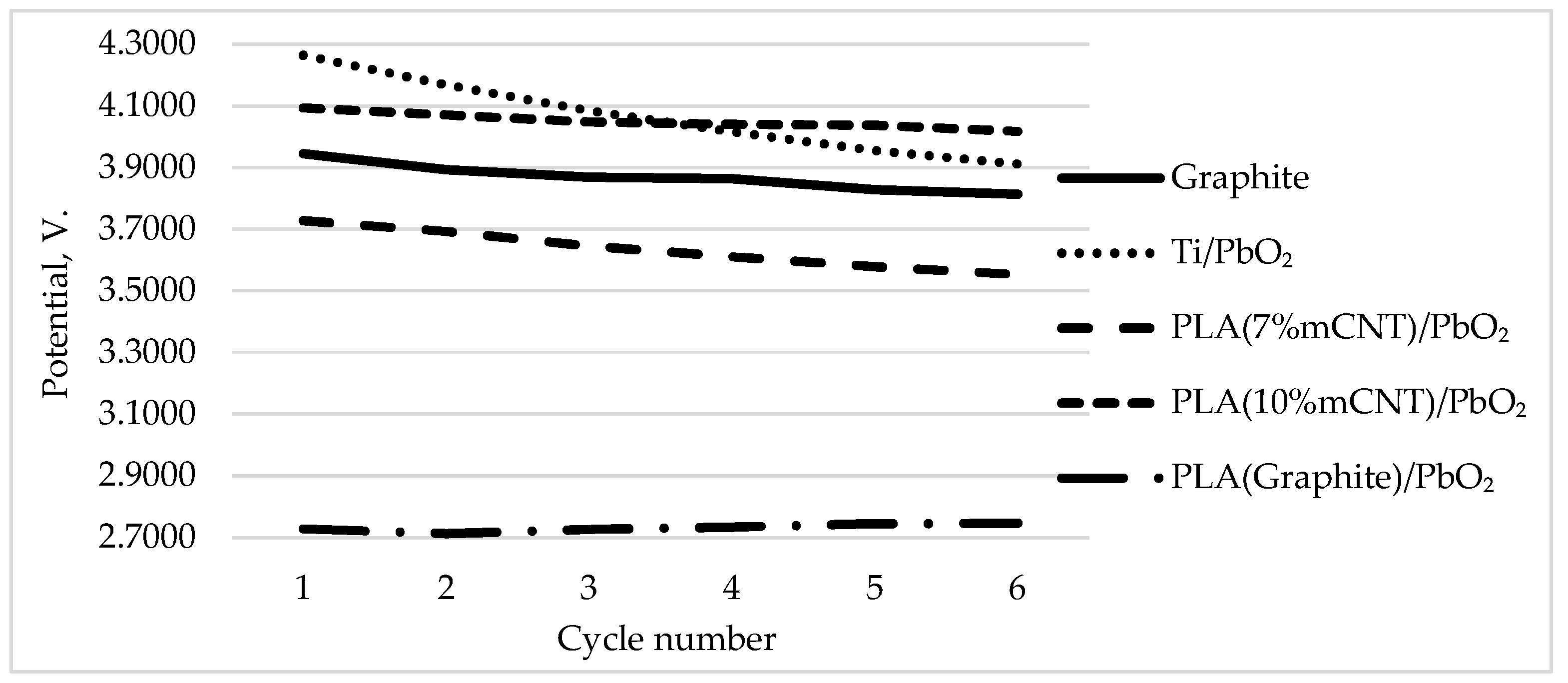
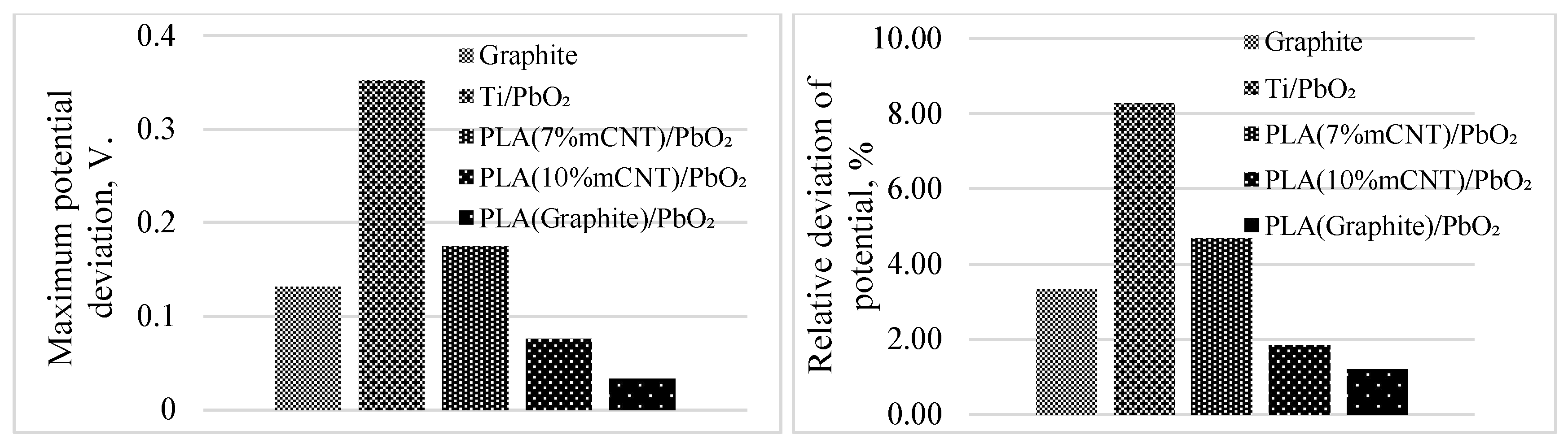
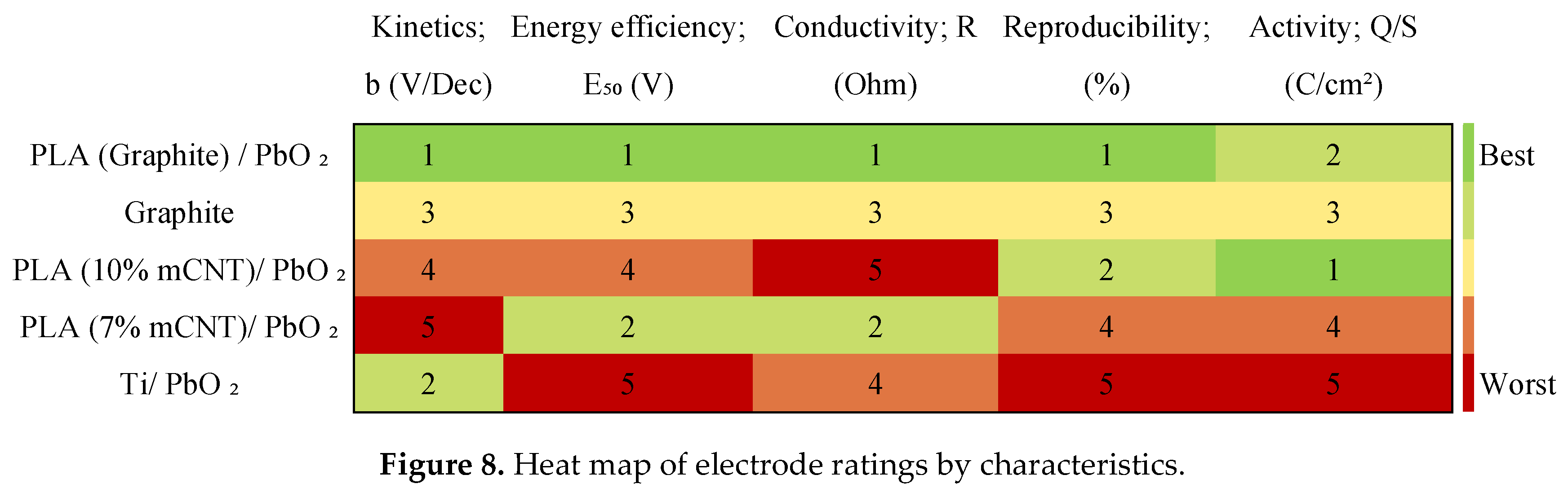
| Electrode | a (V) | b (V/Dec) | R² | E₅₀ (V) | Q/S (C/cm²) | R (Ohm) | Potential deviation (%) | Potential deviation (V) |
| Graphite | 2.1290 | 0.3230 | 0.9228 | 3.8683 | 4.9982 | 49.8000 | 3.3391 | 0.1317 |
| Ti/PbO ₂ | 2.1530 | 0.3150 | 0.9242 | 4.0664 | 4.9829 | 53.2000 | 8.2724 | 0.3527 |
| PLA (7% mCNT)/PbO ₂ | 2.1820 | 0.3310 | 0.9498 | 3.6344 | 4.9857 | 49.3000 | 4.6799 | 0.1744 |
| PLA (10% mCNT)/PbO ₂ | 2.1750 | 0.3270 | 0.9719 | 4.0508 | 5.1039 | 55.0000 | 1.8589 | 0.0761 |
| PLA (Graphite)/PbO ₂ | 1.6190 | 0.1950 | 0.9932 | 2.7321 | 4.9953 | 28.0000 | 1.2160 | 0.0334 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




