Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
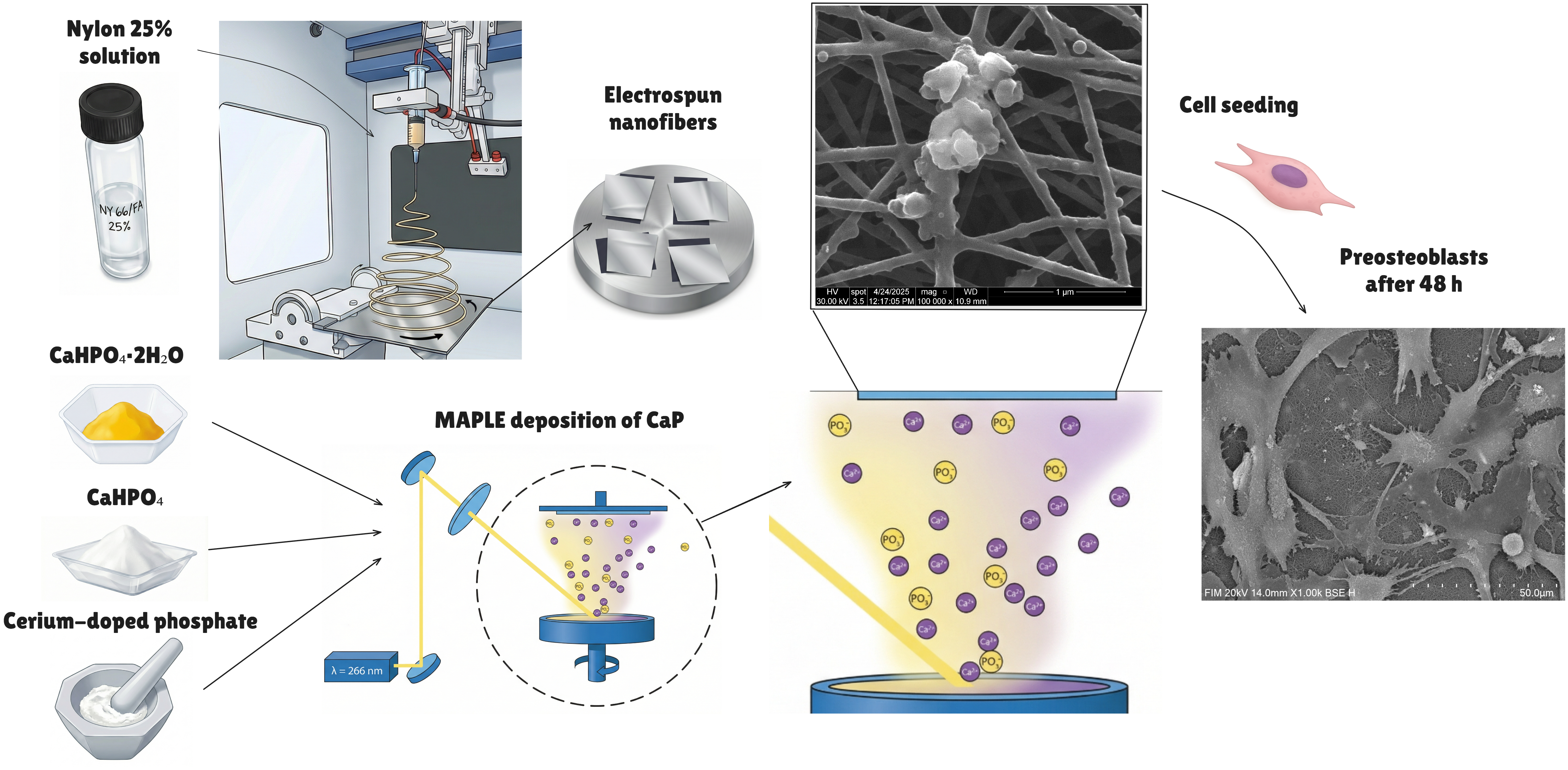
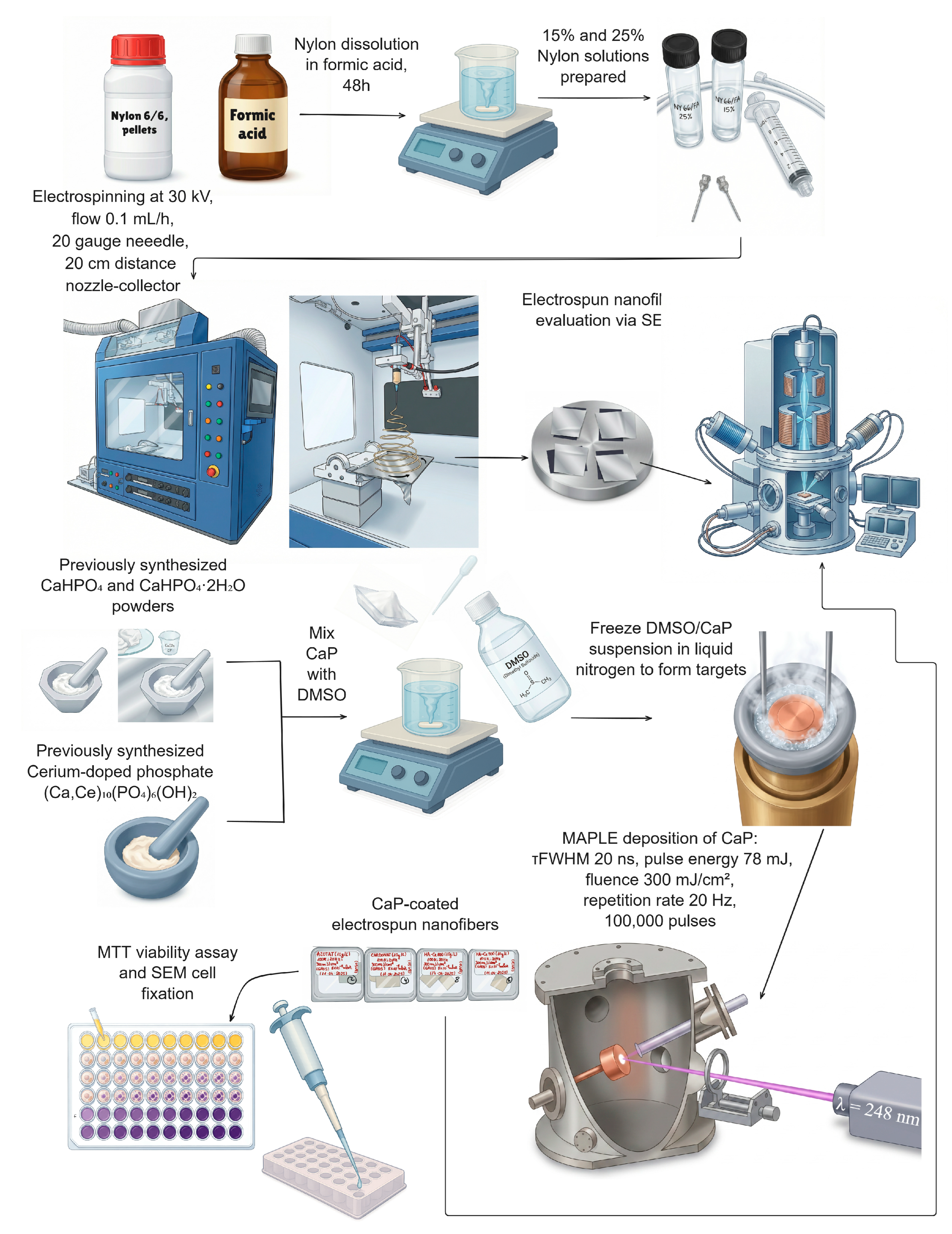
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Calcium Phosphates
2.3. Nylon Nanofibre Synthesis via Electrospinning
2.4. MAPLE Deposition of Calcium Phosphate Coatings
2.5. Characterization Methods
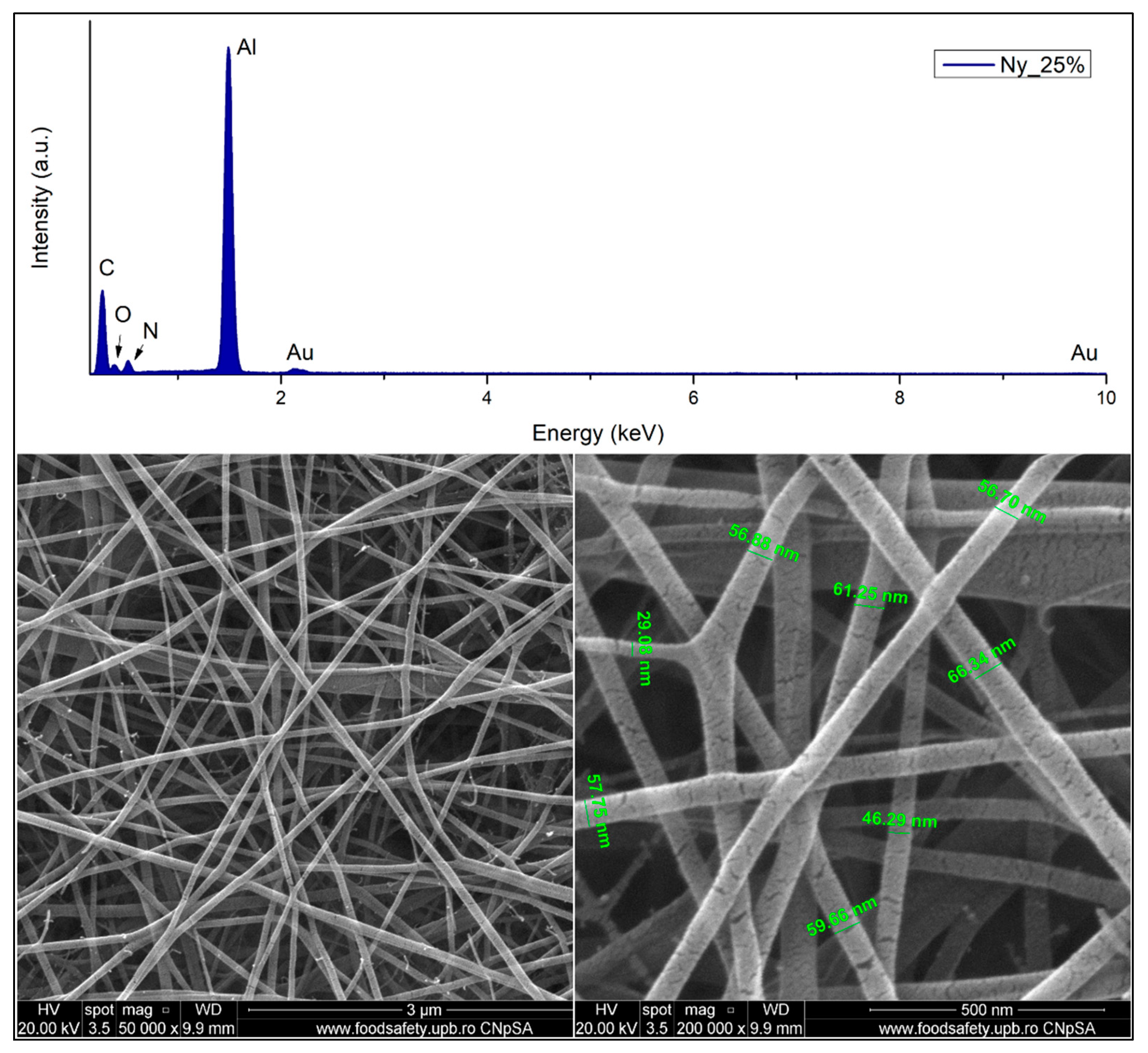
2.5.1. Scanning Electron Microscopy and Energy Dispersive X-Ray Spectroscopy
2.5.2. Fourier-Transform Infrared Spectroscopy
2.5.3. Fibre Diameter Measurements
2.5.4. Fibre Orientation Analysis
2.5.5. MC3T3-E1 Cell Culture
2.5.6. MTT Viability Assay
2.5.7. Cell Fixation for SEM
3. Results
3.1. Nylon Nanofibre and MAPLE Deposition Characterization
3.1.1. Scanning Electron Microscopy and X-Ray Energy Dispersive Spectroscopy
3.1.2. Fourier-Transform Infrared Spectroscopy
3.1.3. Electrospun Fibre Diameter
3.1.4. Electrospun Fibre Orientation
3.2. Cell Viabilty Evaluation
3.2.1. MTT Cell Viability Assay
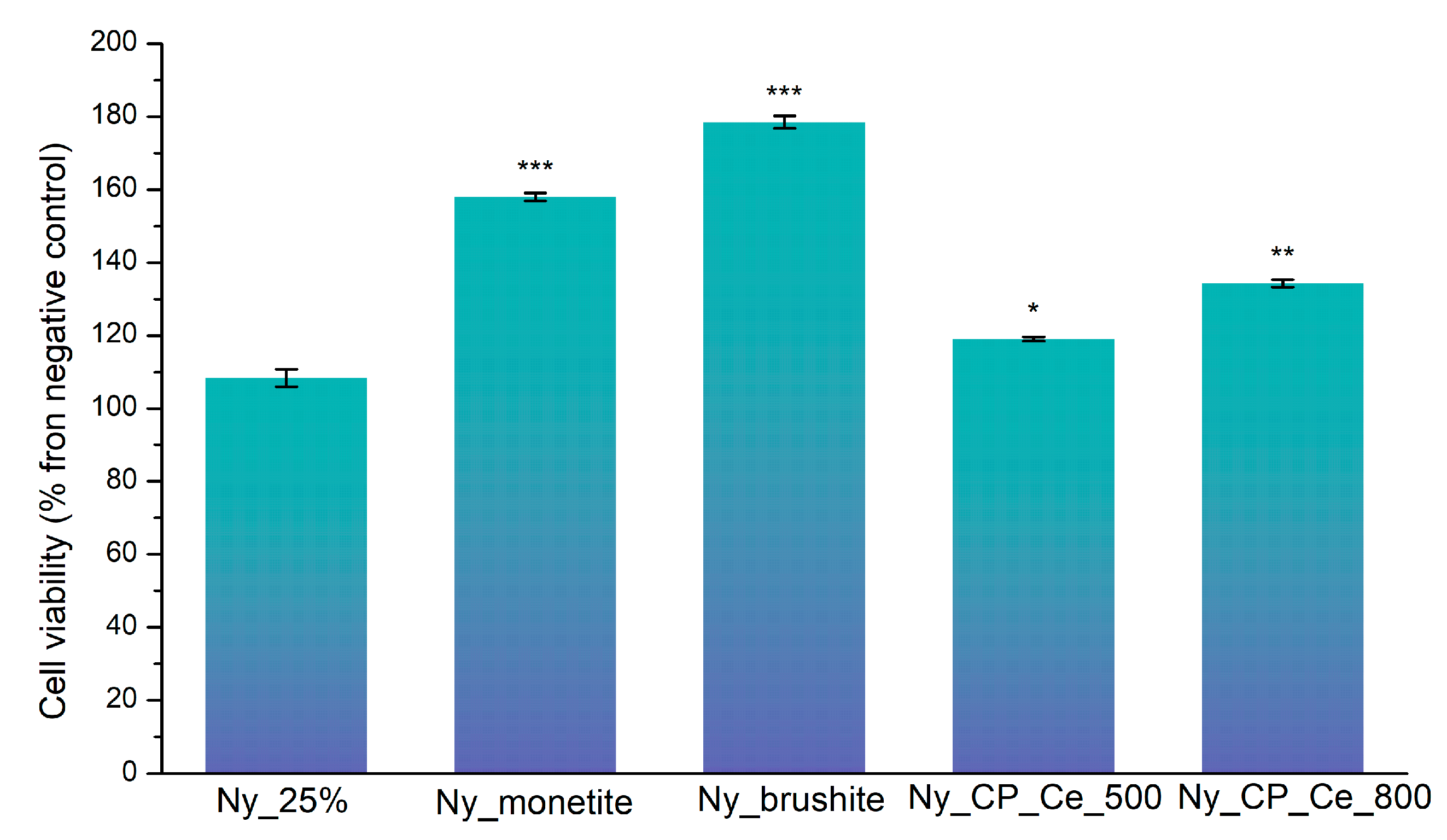
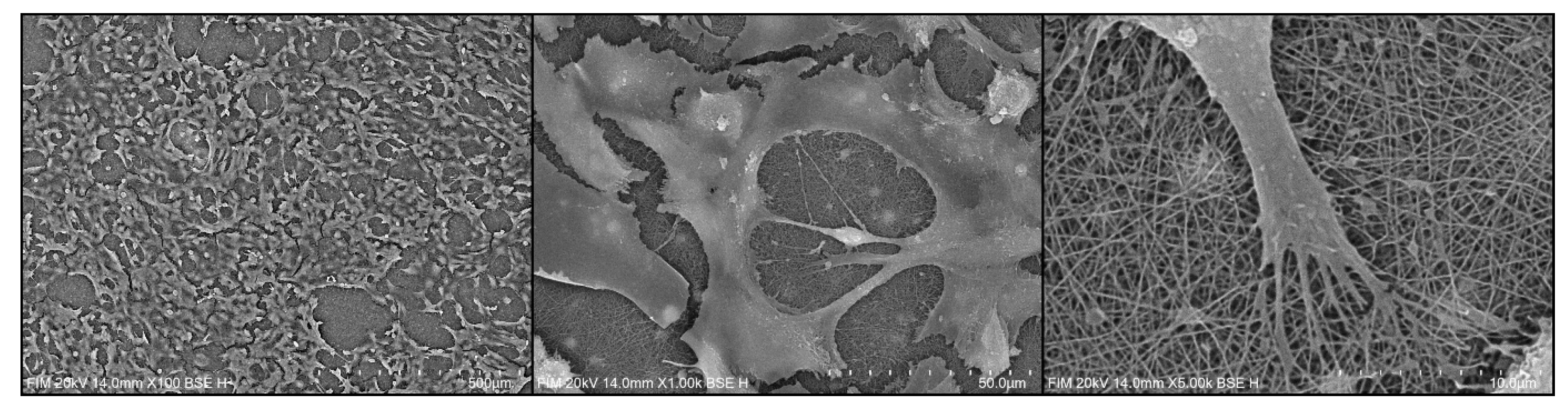
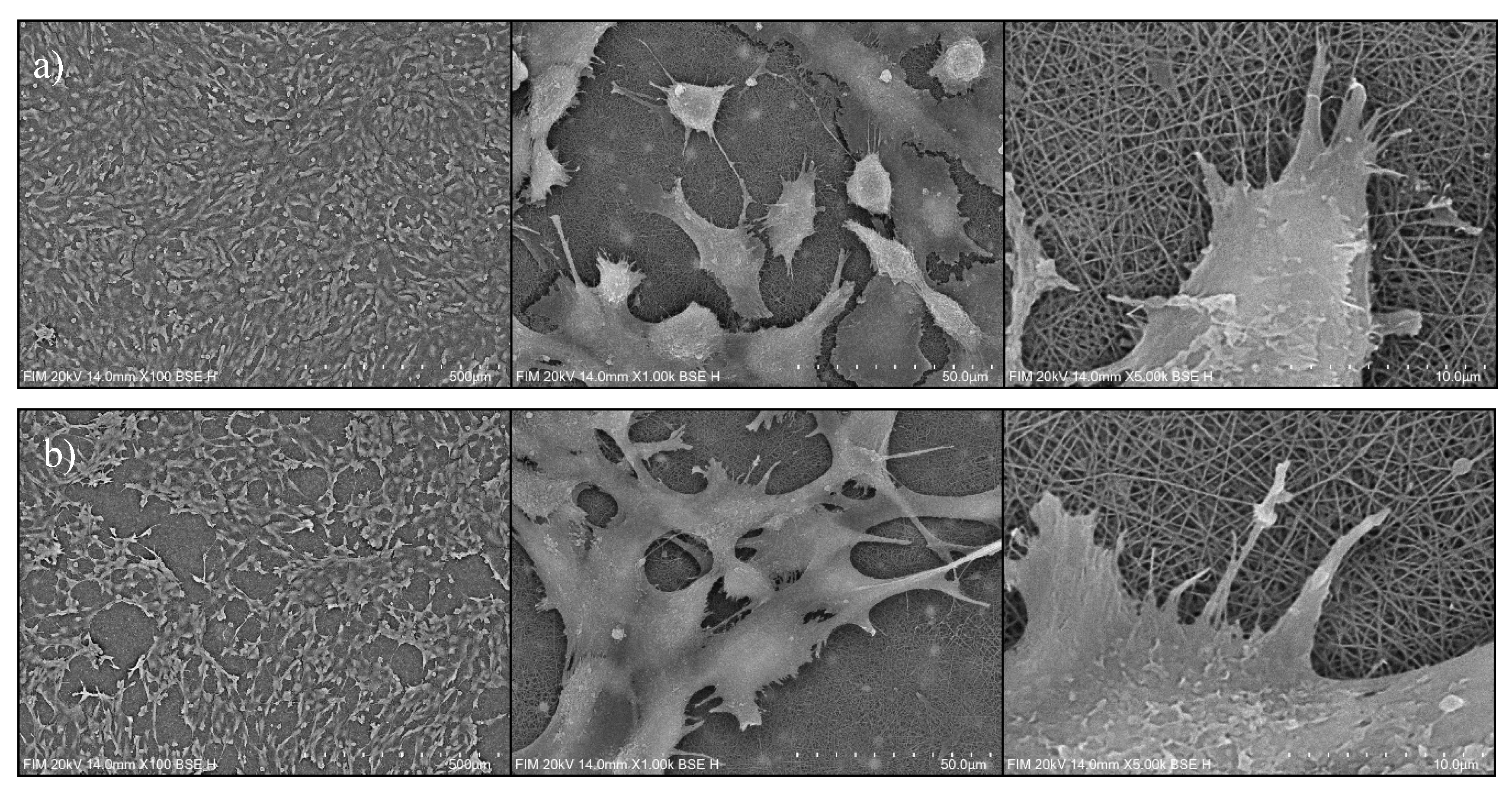
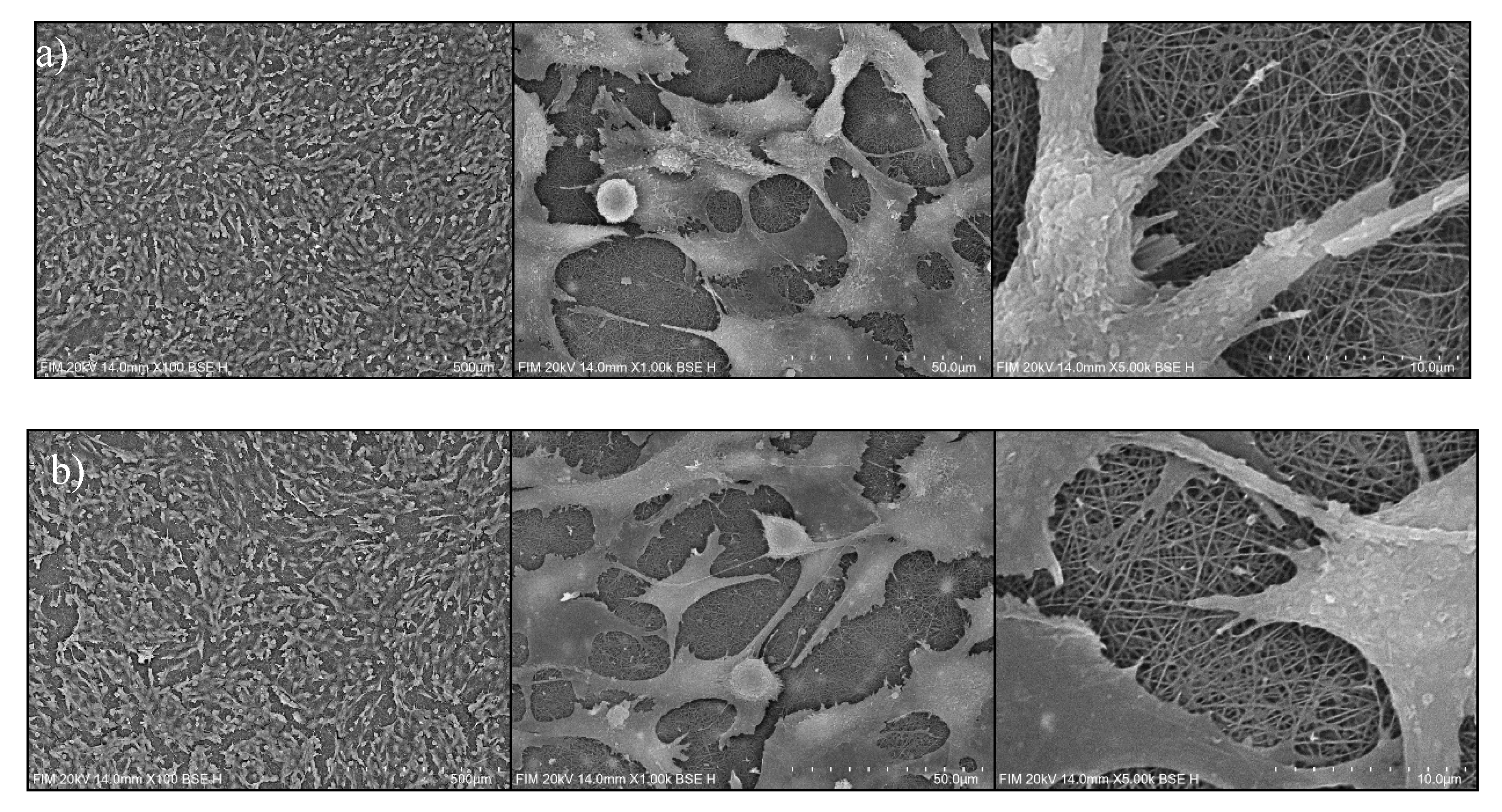
3.2.2. Cell Distribution via SEM Imaging
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MAPLE | Matrix-Assisted Pulsed Laser Evaporation |
| CaP | Calcium Phosphate |
| SEM | Scanning Electron Microscopy |
| EDS | Energy-dispersive X-ray Spectroscopy |
| FTIR | Fourier Transform Infrared Spectroscopy |
| PBS | Phosphate-buffered Saline |
| MTT | (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) |
| HDMS | Hexamethyldisilazane |
| DMSO | Dimethyl sulfoxide |
References
- Sun, J.; Chen, C.; Zhang, B.; Yao, C.; Zhang, Y. Advances in 3D-printed scaffold technologies for bone defect repair: materials, biomechanics, and clinical prospects. Biomed. Eng. OnLine 2025, vol. 24(no. 1), 51. [Google Scholar] [CrossRef]
- Baniameri, S.; et al. Tissue Engineering 3D-Printed Scaffold Using Allograft/Alginate/Gelatin Hydrogels Coated With Platelet-Rich Fibrin or Adipose Stromal Vascular Fraction Induces Osteogenesis In Vitro. J. Cell. Physiol. vol. 240(no. 1), e31497, 2025. [CrossRef]
- Arciero; Buonvino, S.; Melino, S. Slow H2S-Releasing Donors and 3D Printable Arrays Cellular Models in Osteo-Differentiation of Mesenchymal Stem Cells for Personalized Therapies. Biomolecules 2024, vol. 14(no. 11), 1380. [Google Scholar] [CrossRef]
- Laubach, M. Lost in translation: the lack of agreement between surgeons and scientists regarding biomaterials research and innovation for treating bone defects. BMC Med. 2024, vol. 22(no. 1), 517. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Sathiyaseelan, A.; Wang, M.-H. Wolfiporia extensa Extract-Loaded Carboxymethyl Cellulose-Stabilized Cerium Oxide-Doped Hydroxyapatite Nanocomposites as Advanced Antioxidant Platform for Bone Tissue Regeneration. Biomacromolecules 2025, vol. 26(no. 7), 4069–4083. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; et al. Multi-scale hierarchical scaffolds with aligned micro-fibers for promoting cell alignment. Biomed. Mater. 2021, vol. 16(no. 4), 045047. [Google Scholar] [CrossRef] [PubMed]
- Morales, M. A.; Ruiz-Salgado, S.; Agustín-Serrano, R.; Zenteno-Mateo, B.; Rodríguez-Mora, J. I. Design and mathematical modeling of polymeric phases to obtain controlled microporosity materials by 3D printing. Prog. Addit. Manuf. 2023, vol. 8(no. 6), 1701–1710. [Google Scholar] [CrossRef]
- Liu, J.; Yang, L.; Liu, K.; Gao, F. Hydrogel scaffolds in bone regeneration: Their promising roles in angiogenesis. Front. Pharmacol. 2023, vol. 14. [Google Scholar] [CrossRef]
- Nguyen, M. N.-H.; et al. Fabrication of 3-Dimensional-Printed Bilayered Scaffold Carboxymethyl Chitosan/Oxidized Xanthan Gum, Biphasic Calcium Phosphate for Osteochondral Regeneration. Biomater. Res. 2025, vol. 29, 0186. [Google Scholar] [CrossRef]
- Yu, S.; Castellani, R.; Pereira, A.; Valette, R.; Budtova, T. Additive-free 3D-printed nanostructured carboxymethyl cellulose aerogels. Int. J. Biol. Macromol. 2025, vol. 300, 140277. [Google Scholar] [CrossRef]
- Plocon, C. Development and Characterization of Electrospun Composites Built on Polycaprolactone and Cerium-Containing Phases. Int. J. Mol. Sci. 2023, vol. 24(no. 18), 14201. [Google Scholar] [CrossRef]
- Fayemi, E.; Letsholo, P. B.; Shoyiga, H. O.; Ahia, C. C.; Meyer, E. L. Investigation of the effects of concentration and voltage on the physicochemical properties of Nylon 6 nanofiber membrane. Sci. Rep. 2025, vol. 15(no. 1), 10865. [Google Scholar] [CrossRef]
- Li, T.; et al. Parametric modeling and simulation analysis of porous materials prepared by 3D printing technology. Int. J. Simul. Multidiscip. Des. Optim. vol. 16(p. 3), 2025. [CrossRef]
- Oruc, M. E.; Evcimen Duygulu, N.; Onder, B.; Yelkenci, A.; Ustündag, C. B.; Ciftci, F. Nano-Hydroxyapatite/Poly(methyl methacrylate) Composite Bone Scaffold: Surfactant Surface Effects. Polymers 2025, vol. 17(no. 9), 1148. [Google Scholar] [CrossRef] [PubMed]
- Tharakan, S. 3D Printed Osteoblast–Alginate/Collagen Hydrogels Promote Survival, Proliferation and Mineralization at Low Doses of Strontium Calcium Polyphosphate. Pharmaceutics 2022, vol. 15(no. 1), 11. [Google Scholar] [CrossRef]
- Ameli, S.; Nourani, M.; Bakhshi, N.; Salemi, B.; Assadpour, E.; Jafari, S. M. Alginate-gelatin composite hydrogels for encapsulating Aloe vera extract; optimization, characterization, and release kinetics. Carbohydr. Polym. Technol. Appl. 2025, vol. 9, 100717. [Google Scholar] [CrossRef]
- Ding, W.; Ge, Y.; Zhang, T.; Zhang, C.; Yin, X. Advanced construction strategies to obtain nanocomposite hydrogels for bone repair and regeneration. NPG Asia Mater. 2024, vol. 16(no. 1), 14. [Google Scholar] [CrossRef]
- Duan, G.; Ding, M.; Yang, X.; Wu, Q.; Jiang, S. Optimizations of Electrospun Nylon Fibers Toward Potential Application in Artificial Heart Valve. J. Appl. Polym. Sci. vol. 142(no. 25), e57053, 2025. [CrossRef]
- Holec, P.; Jirkovec, R.; Kalous, T.; Baťka, O.; Brožek, J.; Chvojka, J. The Potential for the Direct and Alternating Current-Driven Electrospinning of Polyamides. Nanomaterials 2022, vol. 12(no. 4), 665. [Google Scholar] [CrossRef]
- Foroozandeh, et al. Electrospun nylon 6/hyaluronic acid/chitosan bioactive nanofibrous composite as a potential antibacterial wound dressing. J. Biomed. Mater. Res. B Appl. Biomater. 2024, vol. 112(no. 1), e35370. [Google Scholar] [CrossRef]
- Ragab, H.; Mettwally, B. S.; Mubarak, M. F.; Al-Ghamdi, A.; Hemdan, M. Eco-friendly Electrospinning of Recycled Nylon 6,12 Waste for High-Performance Nonwoven Nanofibers in Sustainable Textile Applications. J. Inorg. Organomet. Polym. Mater. 2024, vol. 34(no. 4), 1491–1505. [Google Scholar] [CrossRef]
- Khademi; Khandan, A.; Iranmanesh, P.; Heydari, M. Development of a 3D Bioprinted Alginate-Gelatin Hydrogel Scaffold Loaded with Calcium Phosphates for Dental Pulp Tissue Regeneration. Iran. J. Chem. Chem. Eng. 2025, vol. 44(no. 1), 1–16. [Google Scholar] [CrossRef]
- Takabatake, K. Effect of Scaffold Geometrical Structure on Macrophage Polarization during Bone Regeneration Using Honeycomb Tricalcium Phosphate. Materials 2024, vol. 17(no. 16), 4108. [Google Scholar] [CrossRef] [PubMed]
- Trifan, et al. Effect of Phosphate Phase Incorporation on 3D-Printed Hydrogel Scaffolds: Towards Customizable Bone Graft Materials. Gels 2025, vol. 11(no. 8), 665. [Google Scholar] [CrossRef] [PubMed]
- Boanini, E.; Pagani, S.; Tschon, M.; Rubini, K.; Fini, M.; Bigi, A. Monetite vs. Brushite: Different Influences on Bone Cell Response Modulated by Strontium Functionalization. J. Funct. Biomater. 2022, vol. 13(no. 2, Art. no. 2). [Google Scholar] [CrossRef] [PubMed]
- Issa, K.; et al. “Brushite: Synthesis, Properties, and Biomedical Applications,” in Crystallization and Applications; IntechOpen, 2022. [Google Scholar] [CrossRef]
- Ciobanu, S. Novel Dextran Coated Cerium Doped Hydroxyapatite Thin Films. Polymers 2022, vol. 14(no. 9), 1826. [Google Scholar] [CrossRef]
- Gradișteanu-Pircalabioru, G.; et al. Enhancing orthopaedic implant efficacy: the development of cerium-doped bioactive glass and polyvinylpyrrolidone composite coatings via MAPLE technique. Biomed. Mater. 2024, vol. 20(no. 1), 015019. [Google Scholar] [CrossRef]
- Drevet, R.; Fauré, J.; Benhayoune, H. Bioactive Calcium Phosphate Coatings for Bone Implant Applications: A Review. Coatings 2023, vol. 13(no. 6), 1091. [Google Scholar] [CrossRef]
- Prodana, M. In-Depth Characterization of Two Bioactive Coatings Obtained Using MAPLE on TiTaZrAg. Materials 2024, vol. 17(no. 12), 2989. [Google Scholar] [CrossRef]
- Caricato, P. MAPLE deposition of nanomaterials. Appl. Surf. Sci. 2014, vol. 302, 92–98. [Google Scholar] [CrossRef]
- Niemczyk. Solvent/non-solvent-based approach in MAPLE deposition of EVA coatings. Appl. Surf. Sci. 2023, vol. 615, 156336. [Google Scholar] [CrossRef]
- Dinu, M.; Bita, B.; Parau, A. C.; Ristoscu, C.; Negut, I. Bioactive Glass and Melittin Thin Films Deposited by MAPLE for Titanium Implant Functionalization. Materials 2025, vol. 18(no. 10), 2410. [Google Scholar] [CrossRef] [PubMed]
- Alfe, M.; Minopoli, G.; Tartaglia, M.; Gargiulo, V.; Ausanio, G. Biocompatible Hybrid Graphenic Thin Coatings on Flexible Substrates through Matrix-Assisted Pulsed Laser Evaporation (MAPLE). ACS Appl. Mater. Interfaces 2024, vol. 16(no. 30), 38956–38967. [Google Scholar] [CrossRef] [PubMed]
- Negut, et al. Multifunctional Bioactive Coatings Based on Bioglass, Vitamin D3, and Melittin Deposited via MAPLE for Enhanced Osseointegration, Antibacterial Activity, and Corrosion Resistance of Titanium Implants. Adv. Mater. Interfaces 2025, vol. 12(no. 22), e00505. [Google Scholar] [CrossRef]
- Schindelin, J. Fiji: an open-source platform for biological-image analysis. Nat. Methods 2012, vol. 9(no. 7), 676–682. [Google Scholar] [CrossRef]
- BIG > OrientationJ. Available online: https://bigwww.epfl.ch/demo/orientation/ (accessed on Feb. 24 2026).
- Satheesan, S.; Gehrig, J.; Thomas, L. S. V. Virtual Orientation Tools (VOTj): Fiji plugins for object centering and alignment. MicroPublication Biol. vol. 2024. [CrossRef]
- Illustrae - Professional Scientific Illustration Platform. Available online: https://illustrae.co/dashboard (accessed on Feb. 18 2026).
- RRID | Resource Report ( RRID: RRID:CVCL_0409). Available online: https://rrid.site/data/record/SCR_013869-1/RRID:CVCL_0409/resolver?q=CVCL_0409&i=rrid:cvcl_0409-0 (accessed on Feb. 26 2026).
- MC3T3-E1 Subclone 4 - CRL-2593 | ATCC. Available online: https://www.atcc.org https://www.atcc.org/products/crl-2593 (accessed on Feb. 26 2026).
- Ghasemi, M.; Turnbull, T.; Sebastian, S.; Kempson, I. The MTT Assay: Utility, Limitations, Pitfalls, and Interpretation in Bulk and Single-Cell Analysis. Int. J. Mol. Sci. 2021, vol. 22(no. 23), 12827. [Google Scholar] [CrossRef]
- Kokubo, T.; Kushitani, H.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W3. J. Biomed. Mater. Res. 1990, vol. 24(no. 6), 721–734. [Google Scholar] [CrossRef]
- Trandaș, A.-N.; et al. Properties of europium and silver doped bioglass thin films obtained by two deposition methods: Biointerfaces for bioinert implants. Mater. Chem. Phys. 2023, vol. 309, 128396. [Google Scholar] [CrossRef]
- Chen, X.; Li, H.; Ma, Y.; Jiang, Y. Calcium Phosphate-Based Nanomaterials: Preparation, Multifunction, and Application for Bone Tissue Engineering. Molecules 2023, vol. 28(no. 12), 4790. [Google Scholar] [CrossRef]
- Liu, X.-L.; et al. Nacre-mimetic cerium-doped nano-hydroxyapatite/chitosan layered composite scaffolds regulate bone regeneration via OPG/RANKL signaling pathway. J. Nanobiotechnology 2023, vol. 21(no. 1), 259. [Google Scholar] [CrossRef]
- Yokoi, T. Synthesis of Octacalcium Phosphate Containing Glutarate Ions with a High Incorporation Fraction. Materials 2023, vol. 16(no. 1, Art. no. 1). [Google Scholar] [CrossRef]
- Ng, P.; et al. Fabrication of Antibacterial, Osteo-Inductor 3D Printed Aerogel-Based Scaffolds by Incorporation of Drug Laden Hollow Mesoporous Silica Microparticles into the Self-Assembled Silk Fibroin Biopolymer. Macromol. Biosci. 2022, vol. 22(no. 4), e2100442. [Google Scholar] [CrossRef]
- Wassif, R. K.; Shamma, R. N.; El-Hoffy, N. M.; El-Kayal, M. Recent Advances in the Local Drug Delivery Systems for Diabetic Wound Healing: A Comprehensive Review. AAPS PharmSciTech 2025, vol. 26(no. 6), 177. [Google Scholar] [CrossRef]

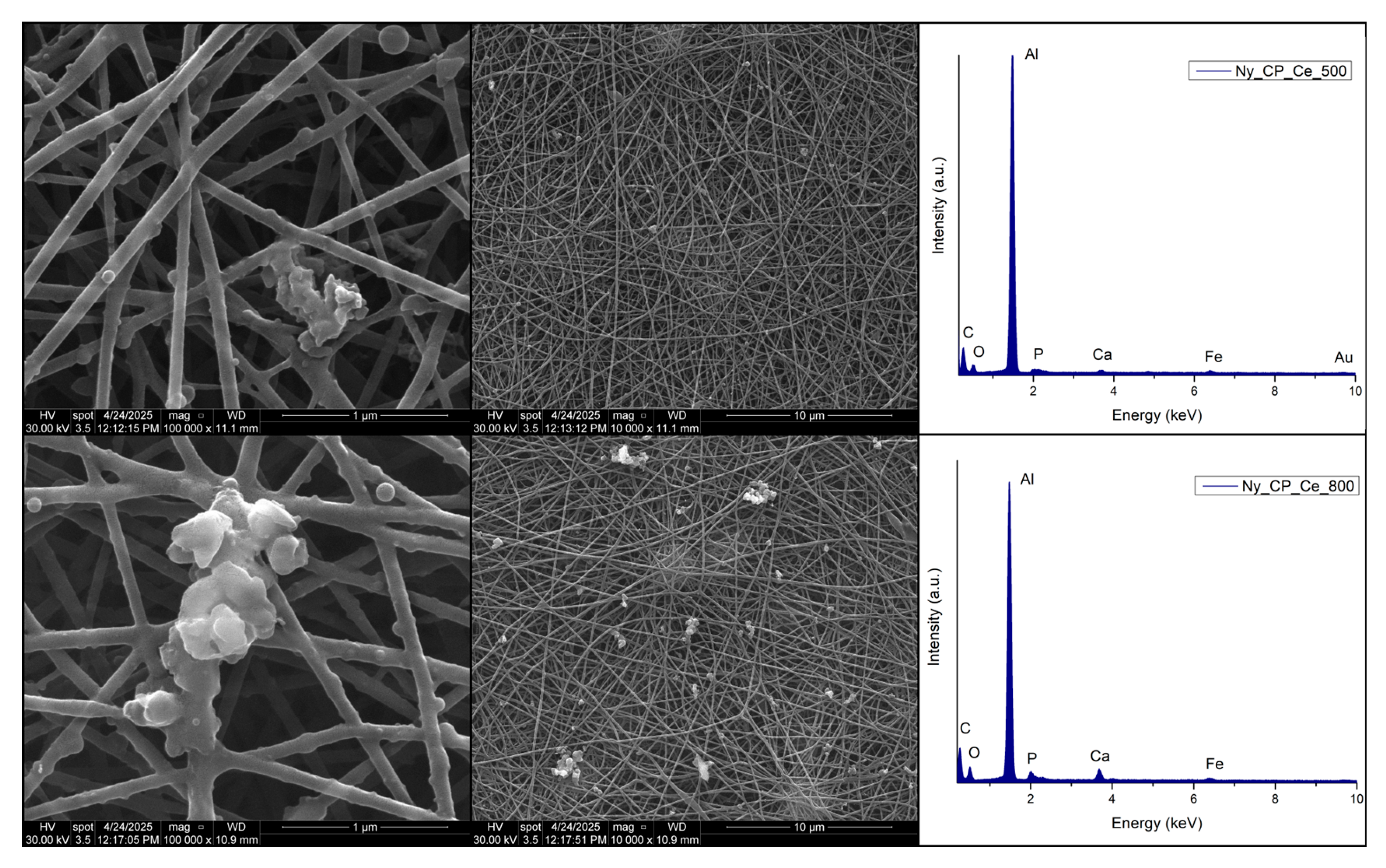
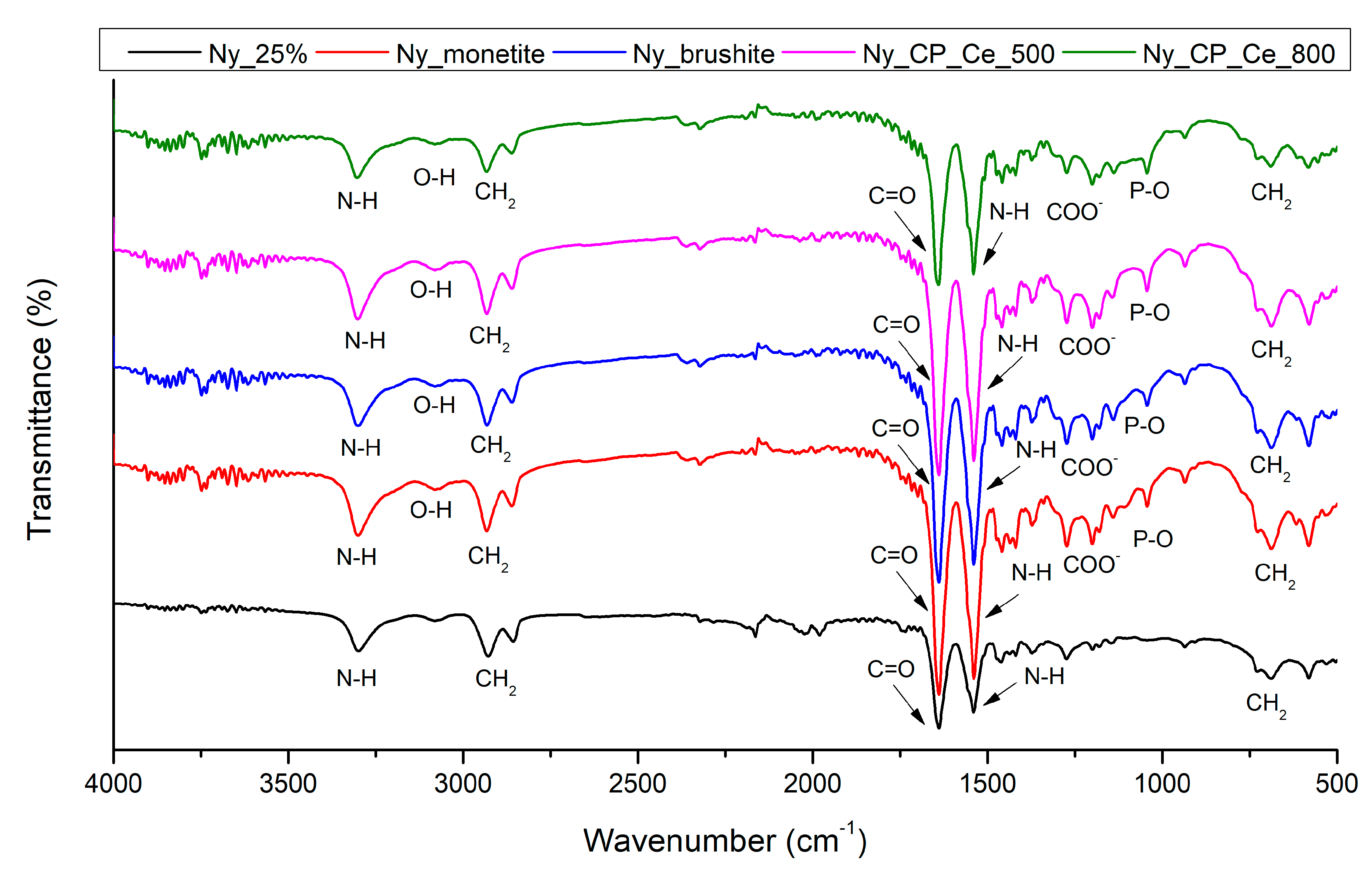
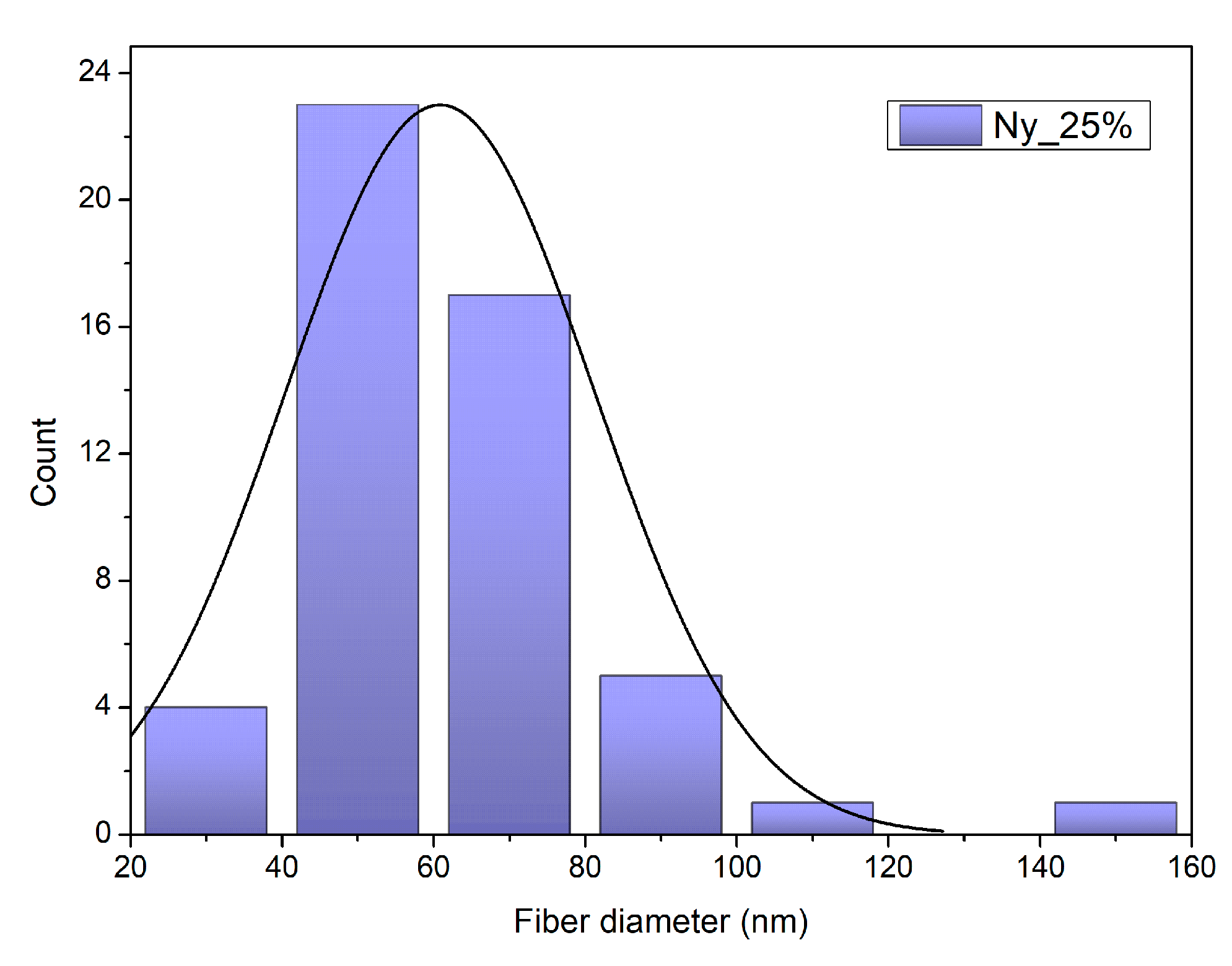
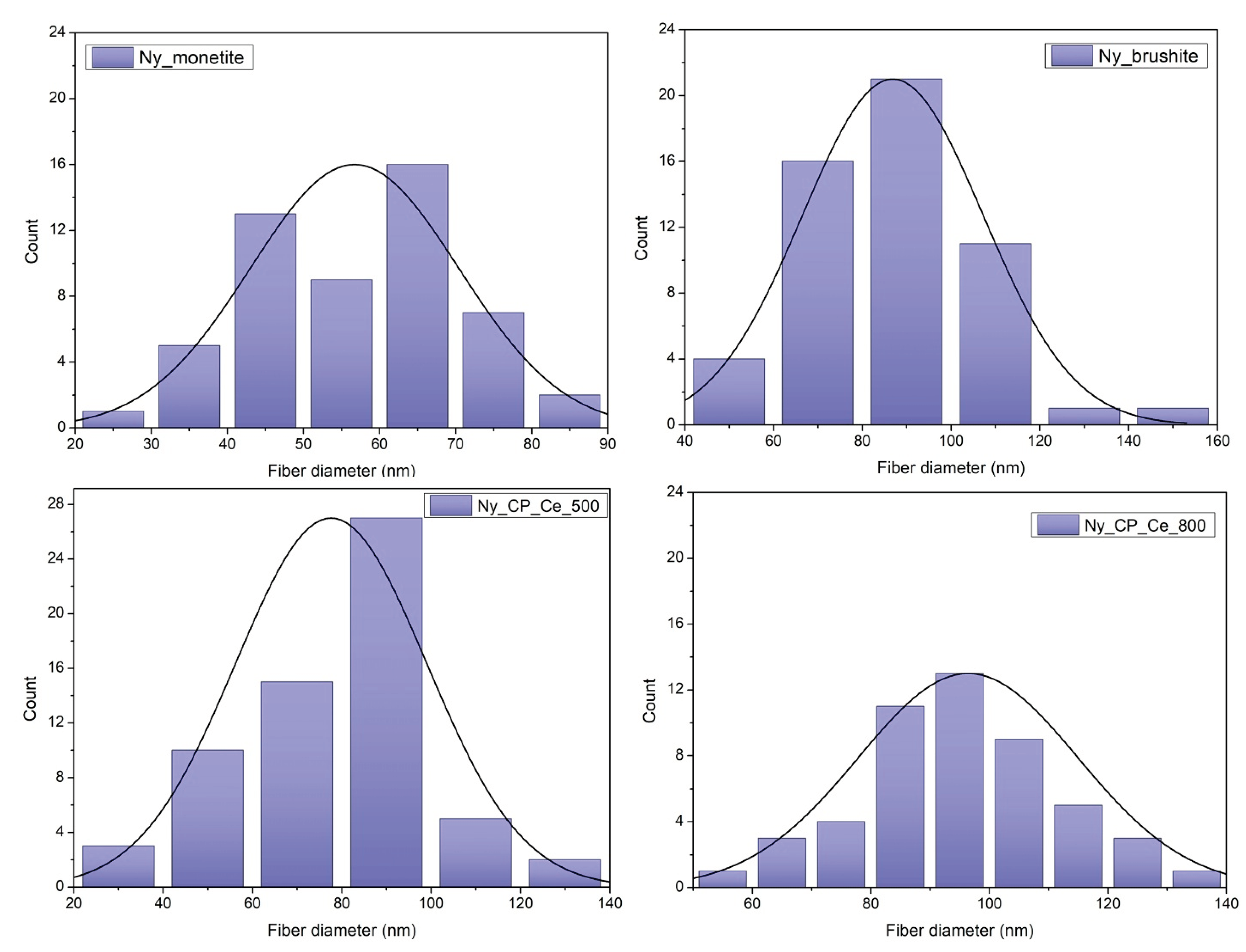
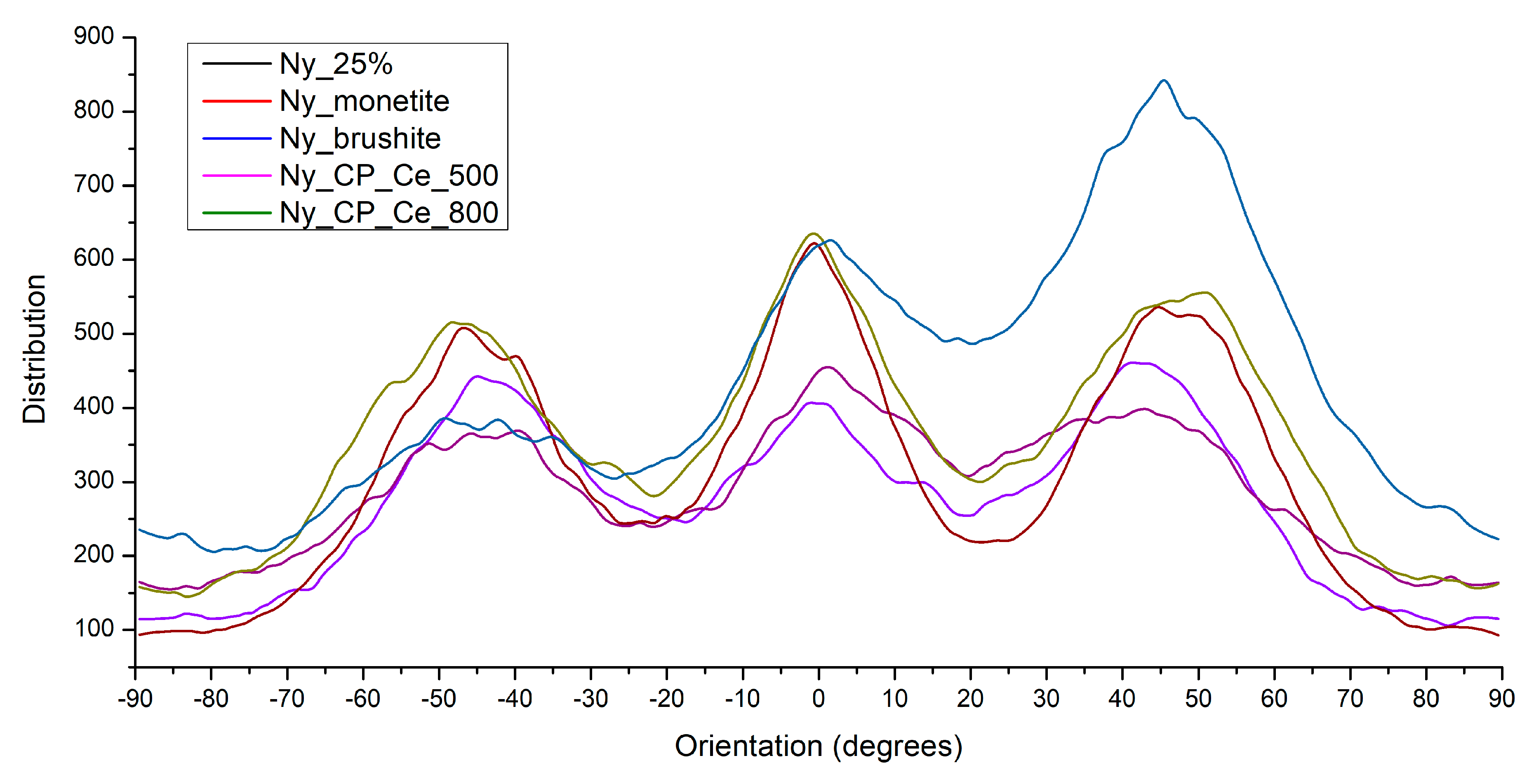
| Sample | Description |
| Ny_25% | Electrospun nylon scaffold (25 wt% solution), uncoated |
| Ny_monetite | Ny_25% scaffold coated by MAPLE with monetite (CaHPO₄) |
| Ny_brushite | Ny_25% scaffold coated by MAPLE with brushite (CaHPO₄·2H₂O) |
| Ny_CP_Ce_500 | Ny_25% scaffold coated by MAPLE with cerium-doped phosphate calcined at 500 °C |
| Ny_CP_Ce_800 | Ny_25% scaffold coated by MAPLE with cerium-doped phosphate calcined at 800 °C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




