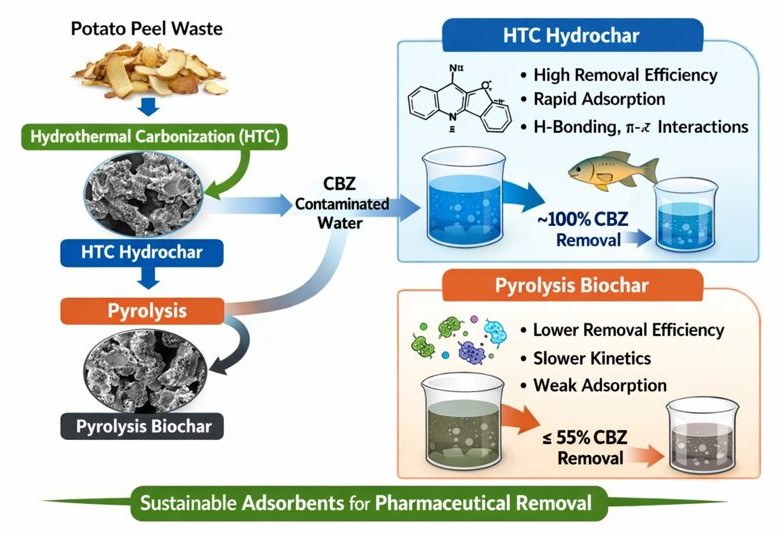
The persistence of pharmaceutical contaminants such as carbamazepine (CBZ) in aquatic environments presents a growing challenge for conventional wastewater treatment processes. In this study, potato peel waste was valorised into carbonaceous adsorbents via hydrothermal carbonization (HTC) and conventional pyrolysis, and their performance for CBZ removal from water was systematically compared. Hydrochars were prepared at 200 °C under varying residence times and biomass-to-water ratios, while biochars were produced at 400 °C using KOH activation under different reaction times and impregnation ratios. The materials were characterised using BET surface area analysis, CHNS elemental analysis, and FTIR spectroscopy. Adsorption experiments revealed that HTC-derived hydrochars achieved outstanding CBZ removal efficiencies (up to ~100%) and high uptake capacities (~50 mg g⁻¹) within one minute of contact, despite relatively low surface areas (< 2 m² g⁻¹). In contrast, pyrolysis biochars exhibited significantly lower removal efficiencies (7–55%) and slower, less stable adsorption behaviour. Correlation analysis demonstrated that CBZ removal was strongly associated with surface chemistry—particularly carbon, hydrogen, and nitrogen content and N/C ratio—rather than BET surface area or pore diameter. FTIR analysis indicated that π–π interactions, hydrogen bonding, and pore filling collectively govern CBZ adsorption, with oxygen- and nitrogen-containing functional groups playing a dominant role in rapid uptake. These findings highlight hydrothermal carbonization as an effective, low-severity route for producing high-performance adsorbents from food waste and demonstrate the potential of potato peel–derived hydrochars for rapid pharmaceutical remediation in water treatment applications.