Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
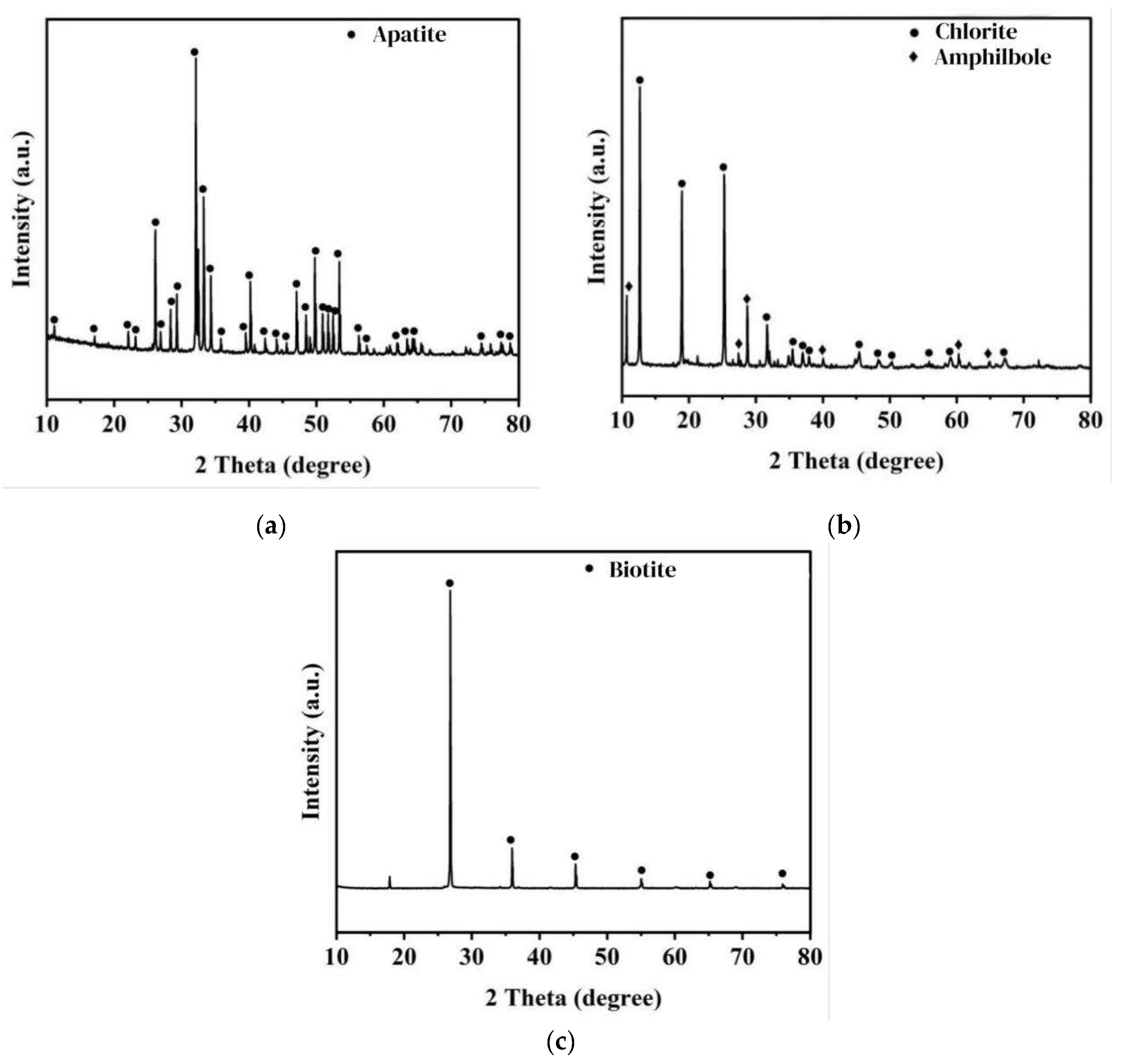
2.1. Test Samples and Reagents
| Name | CaO | P2O5 | F | SiO2 | SO3 | Fe2O3 | SrO | MgO | MnO |
|---|---|---|---|---|---|---|---|---|---|
| Content | 56.51 | 38.58 | 1.53 | 1.10 | 0.85 | 0.09 | 0.07 | 0.04 | 0.04 |
| Name | SiO2 | Al2O3 | Fe2O3 | MgO | CaO | Na2O | K2O | MnO | TiO2 |
|---|---|---|---|---|---|---|---|---|---|
| Content | 32.54 | 18.32 | 9.91 | 26.87 | 1.82 | 0.14 | 0.01 | 0.06 | 0.33 |
| Name | IL | Al2O3 | SiO2 | Fe2O3 | CaO | MgO | K2O | Na2O | TiO2 |
|---|---|---|---|---|---|---|---|---|---|
| Content | 2.06 | 17.10 | 40.72 | 13.95 | 0.18 | 15.88 | 8.82 | 0.10 | 0.89 |
2.2. Experimental Methods
2.2.1. Single Mineral Flotation Tests
2.2.2. Reagent Mechanism Detection and Analysis
3. Results and Discussion
3.1. Flotation Tests
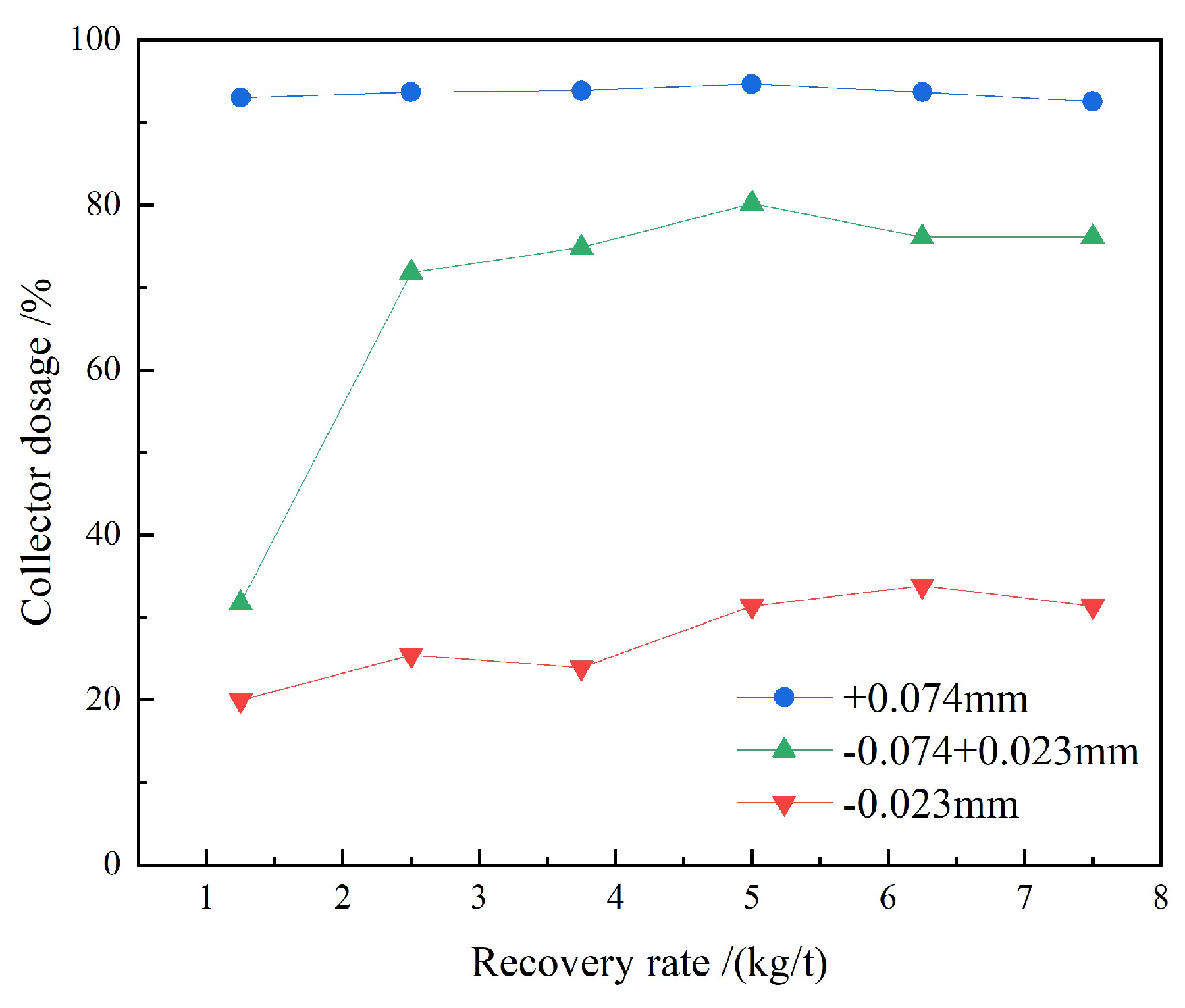
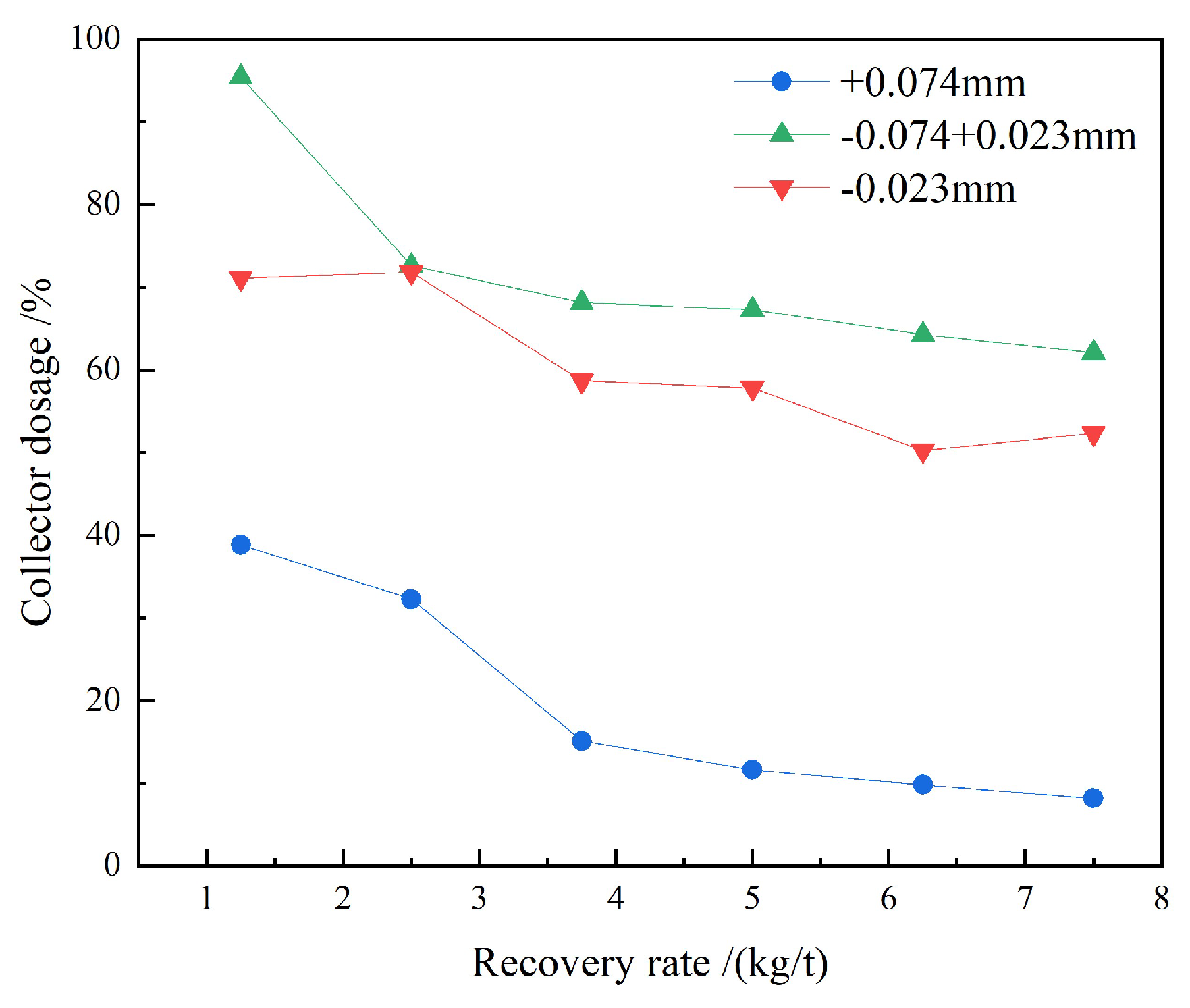
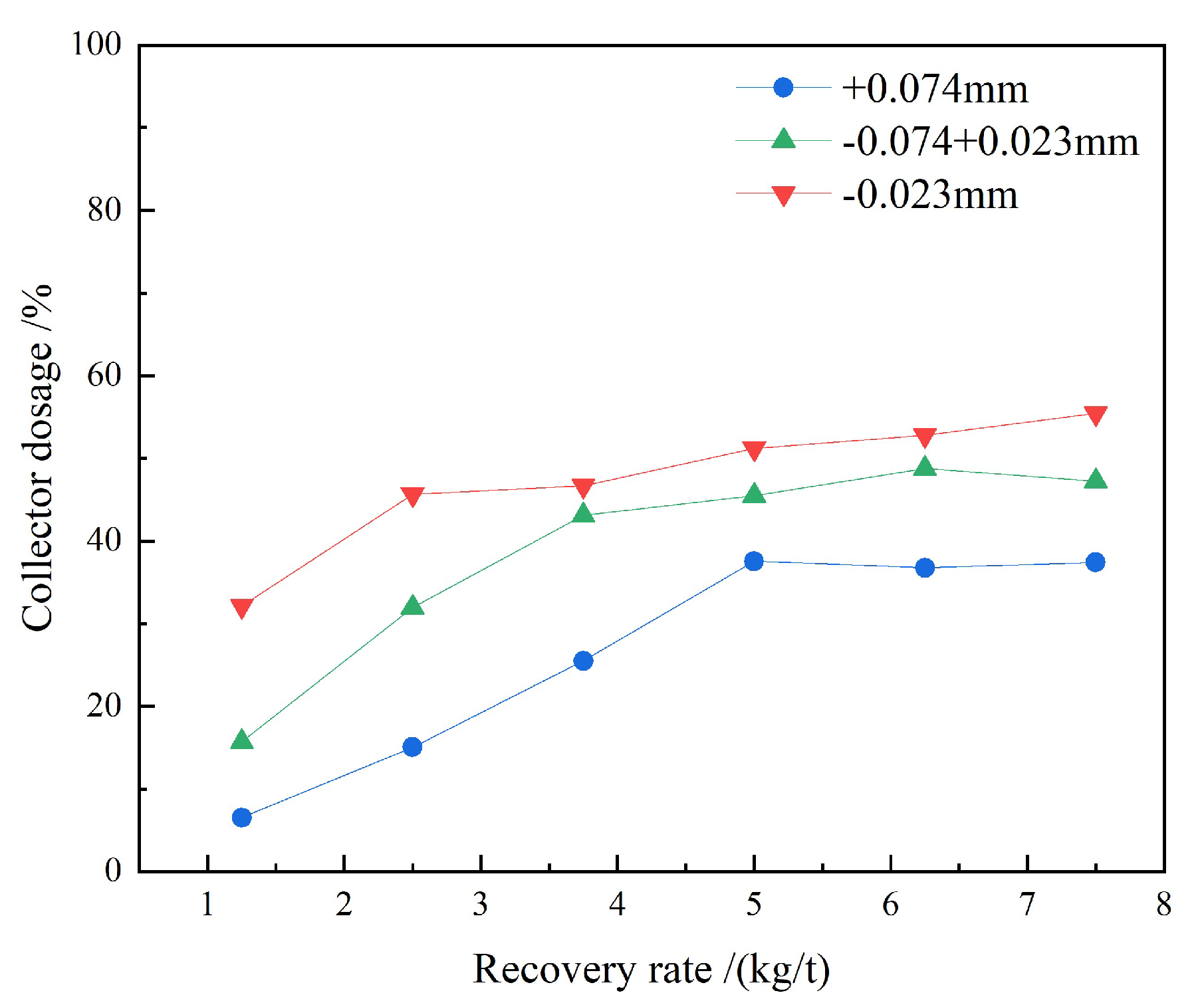
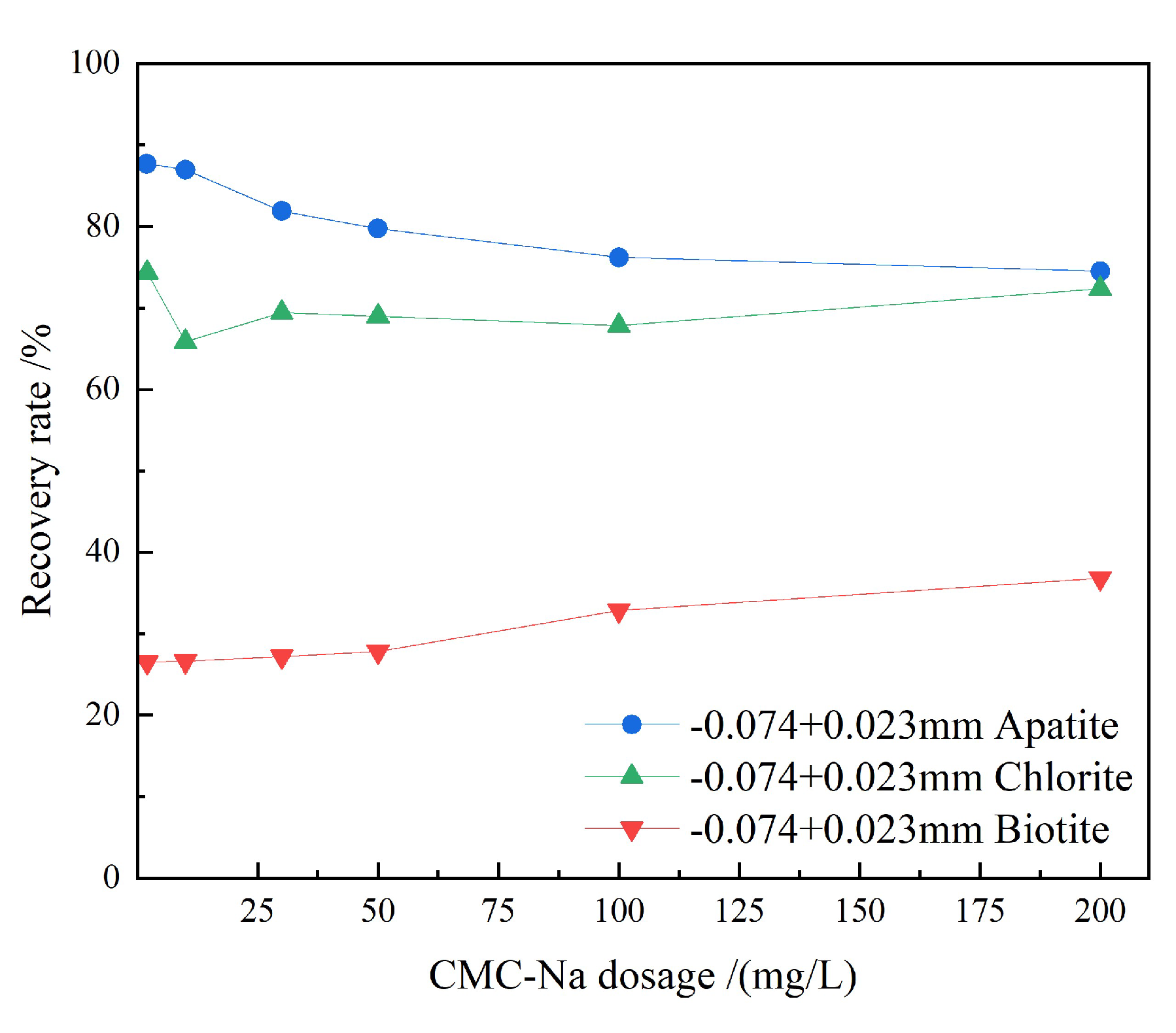
3.1.1. Collector Dosage Tests
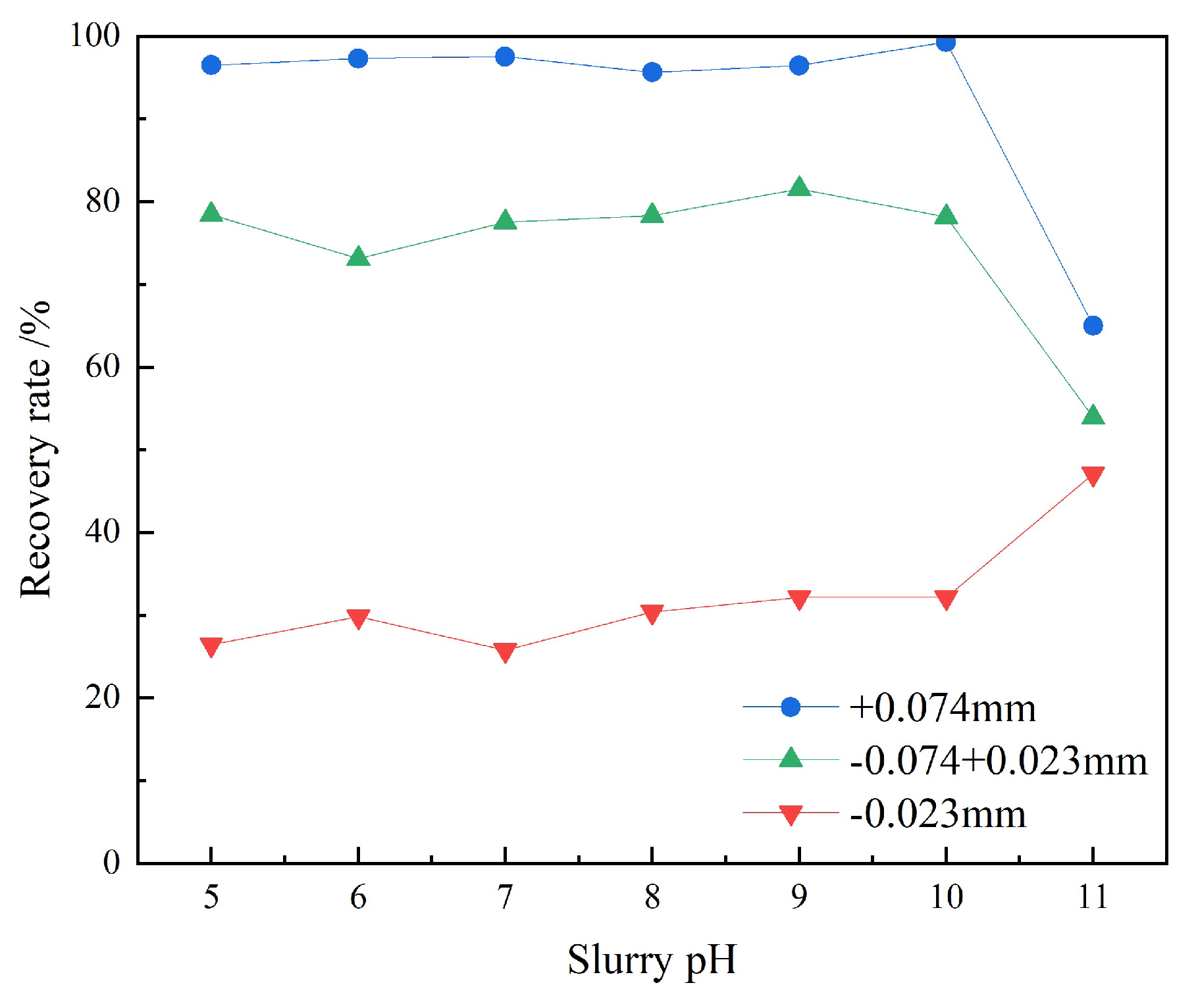
3.1.2. pH Determination
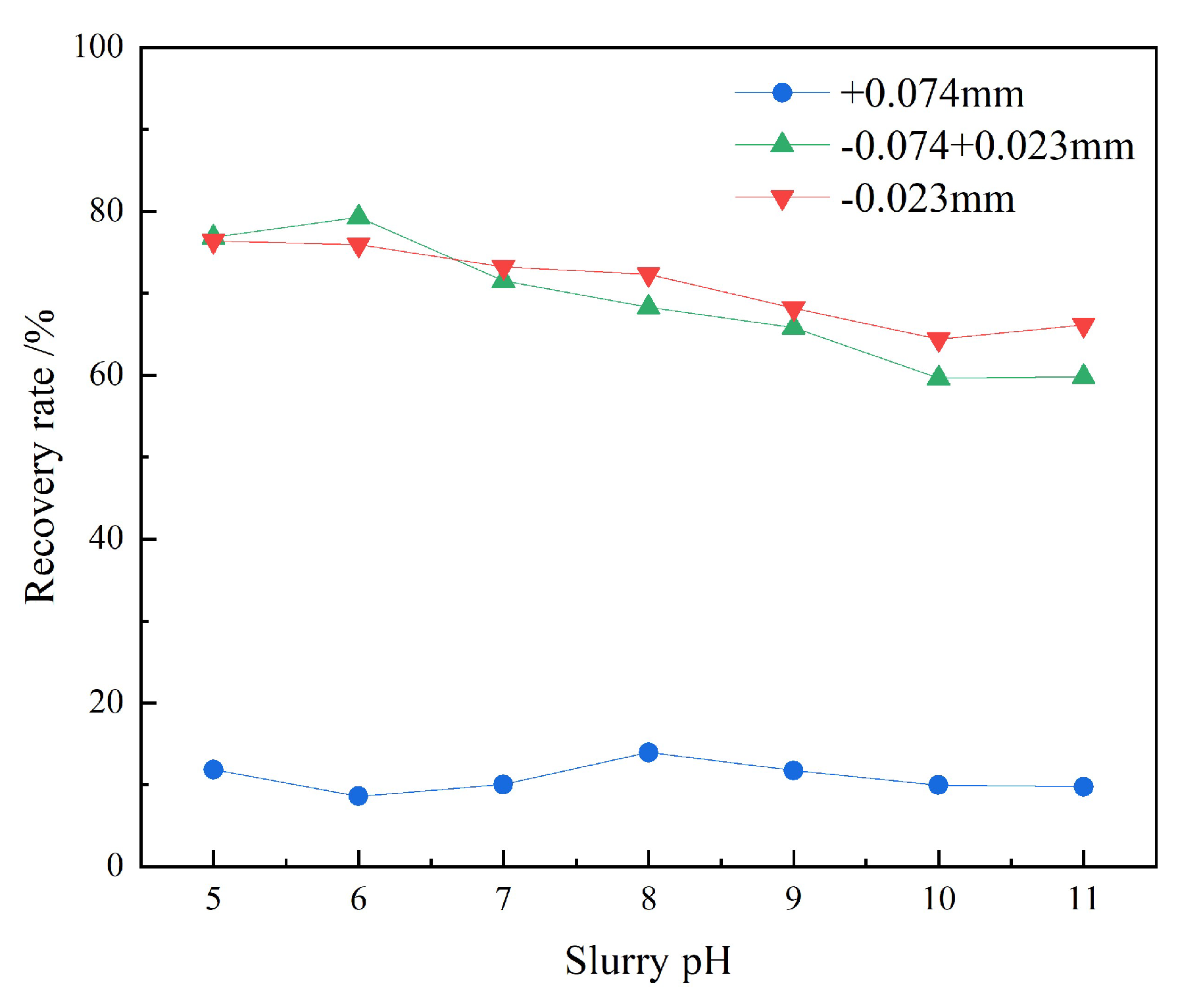
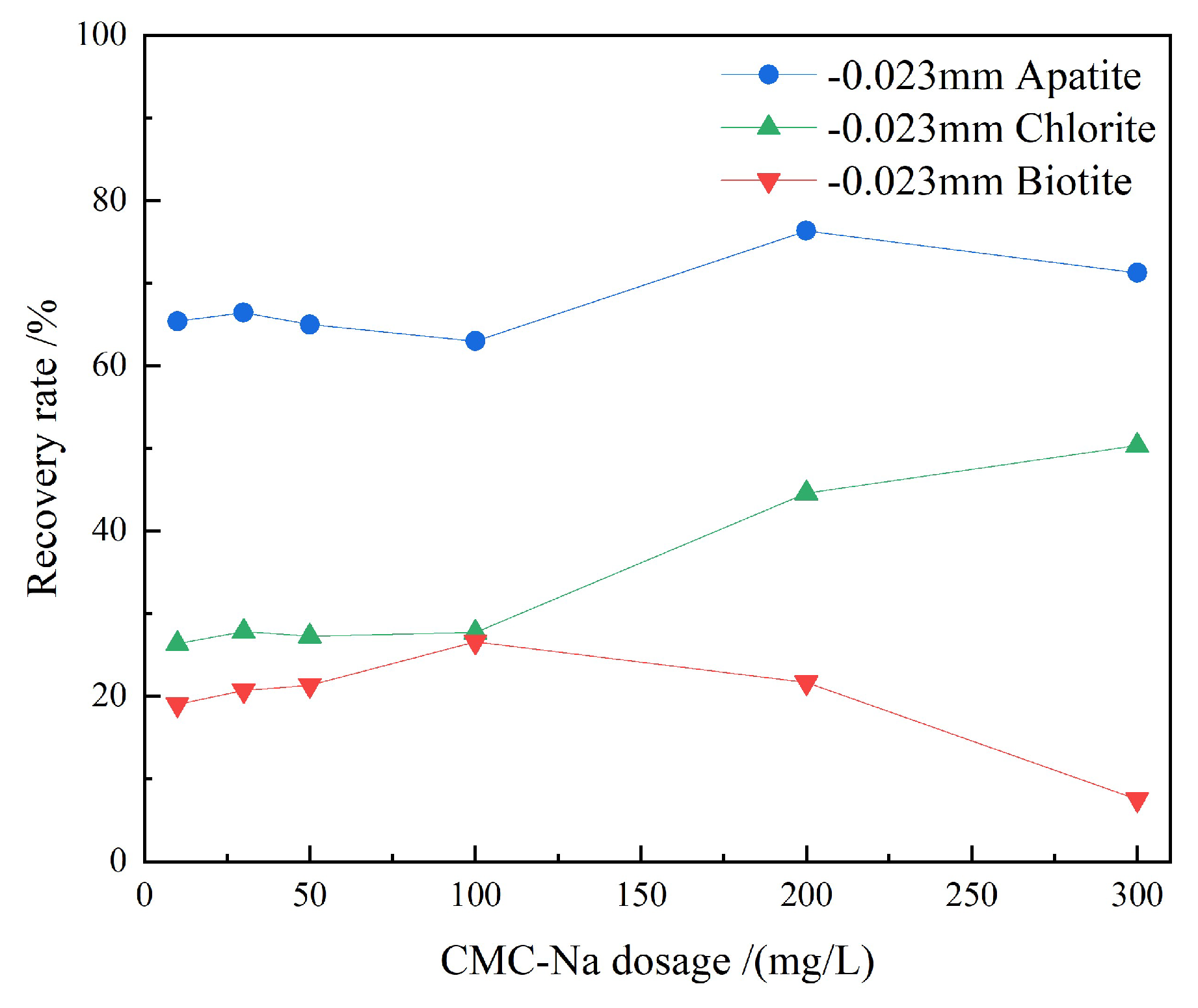
3.1.3. CMC-Na Dosage Tests
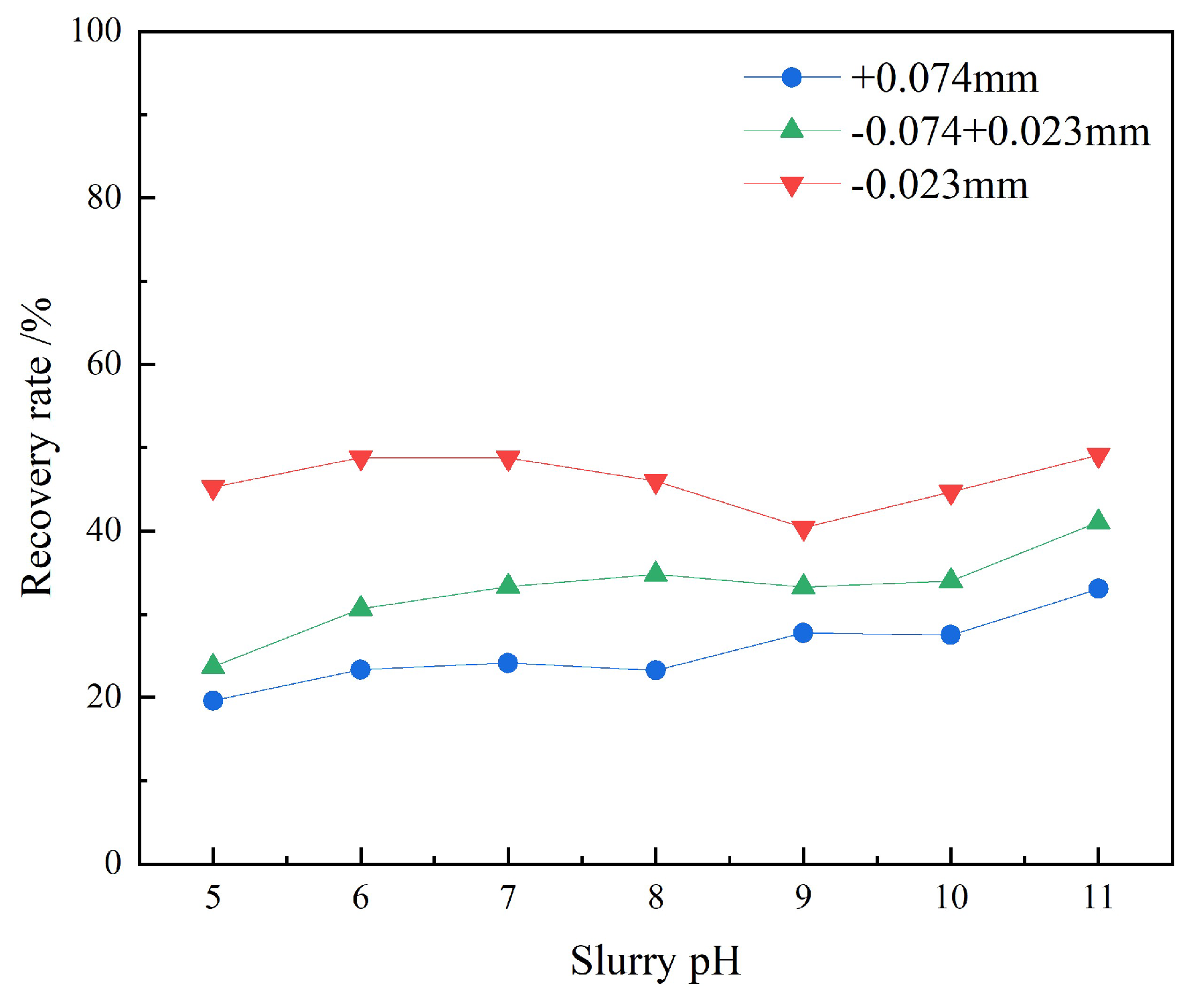
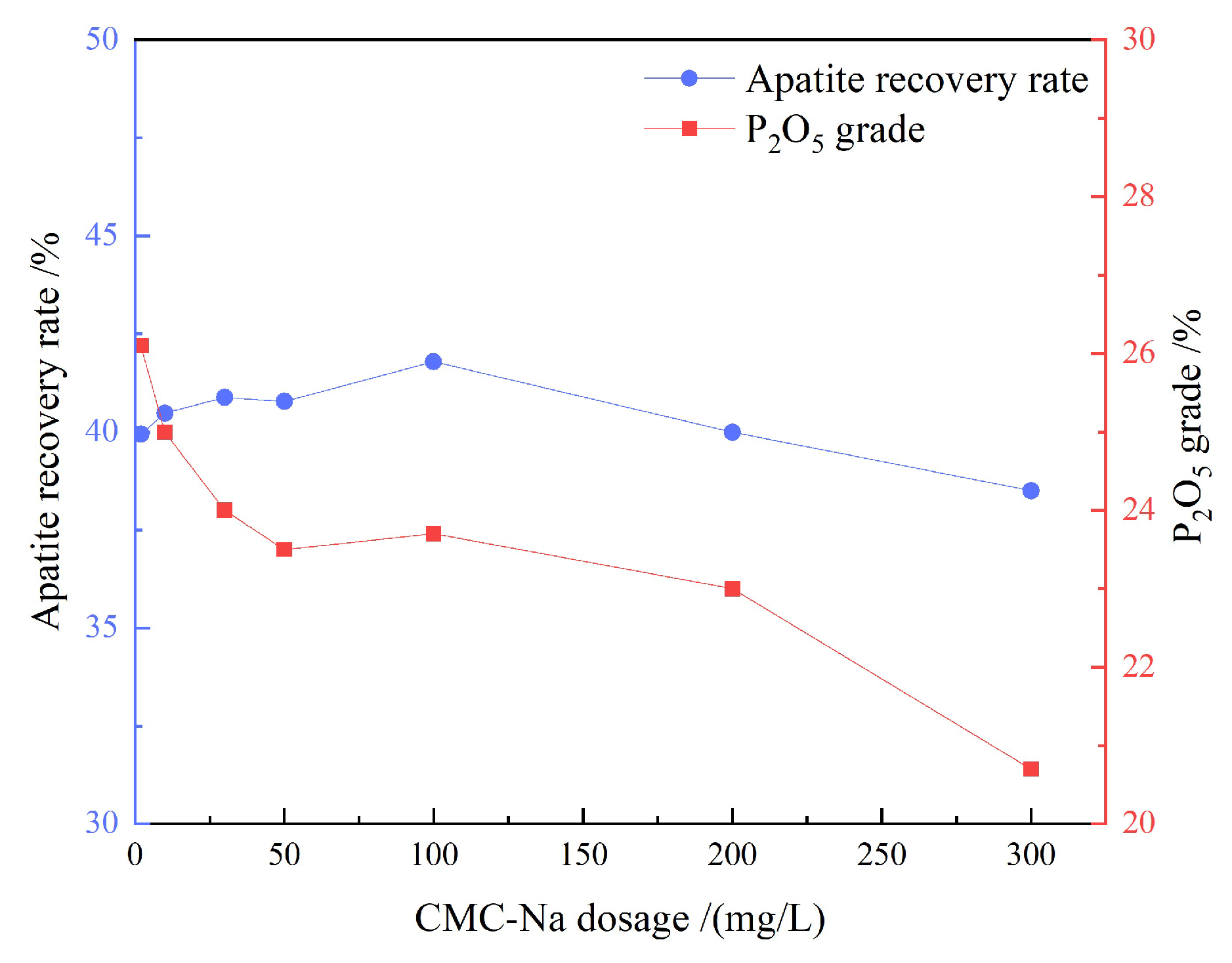
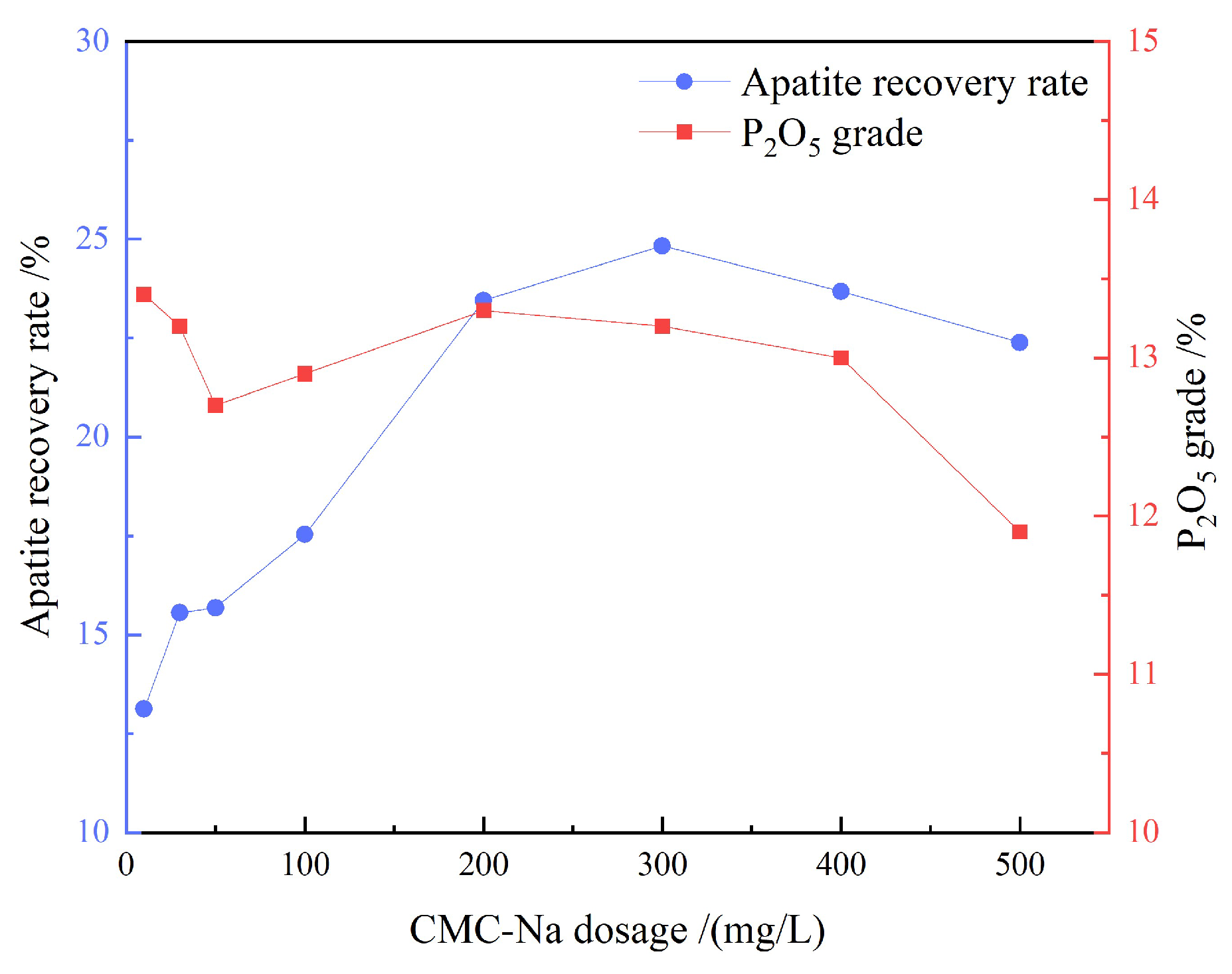
3.1.4. Artificial Mixed Mineral Tests
3.2. Mechanism Analysis
3.2.1. Contact Angle Measurements
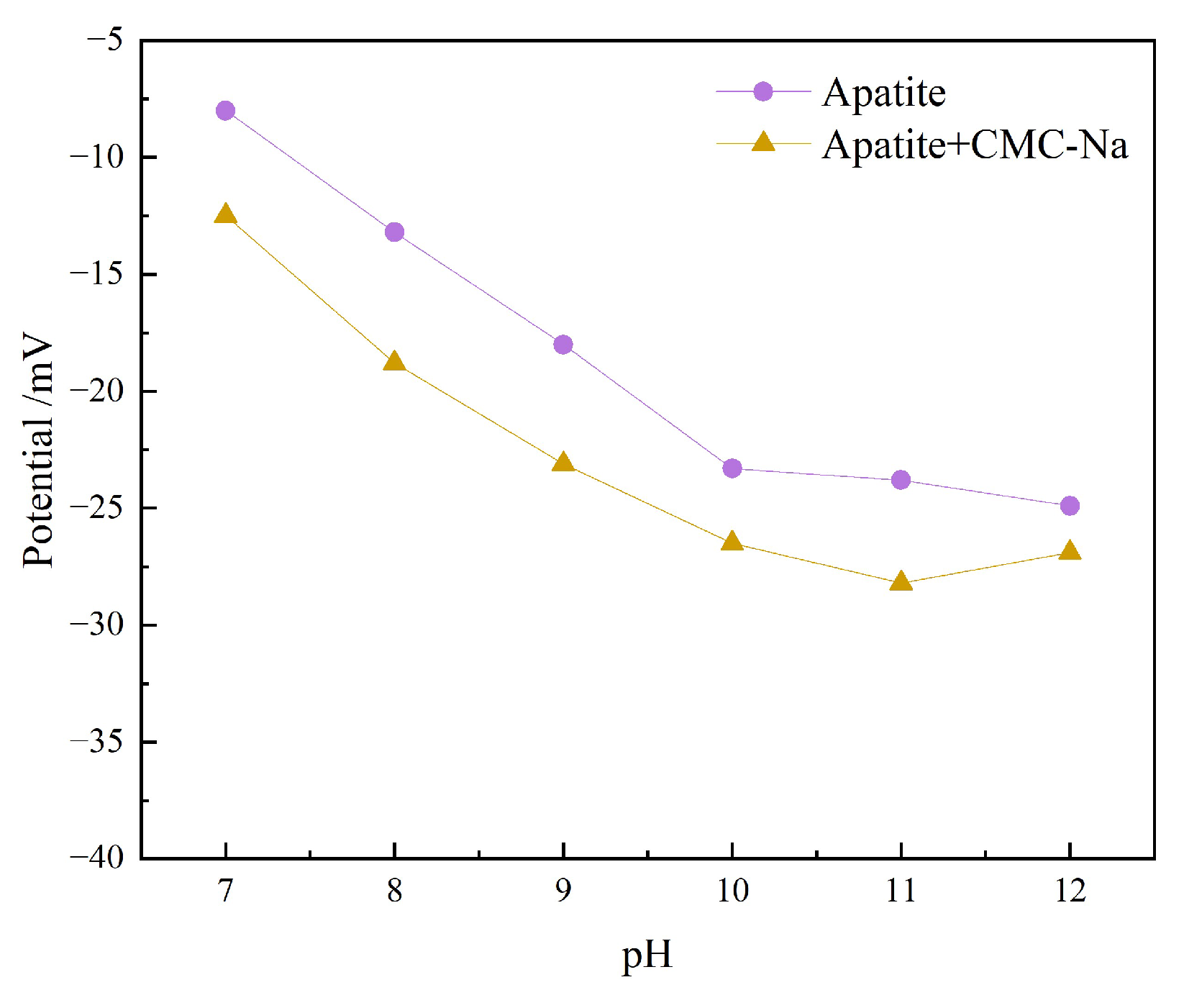
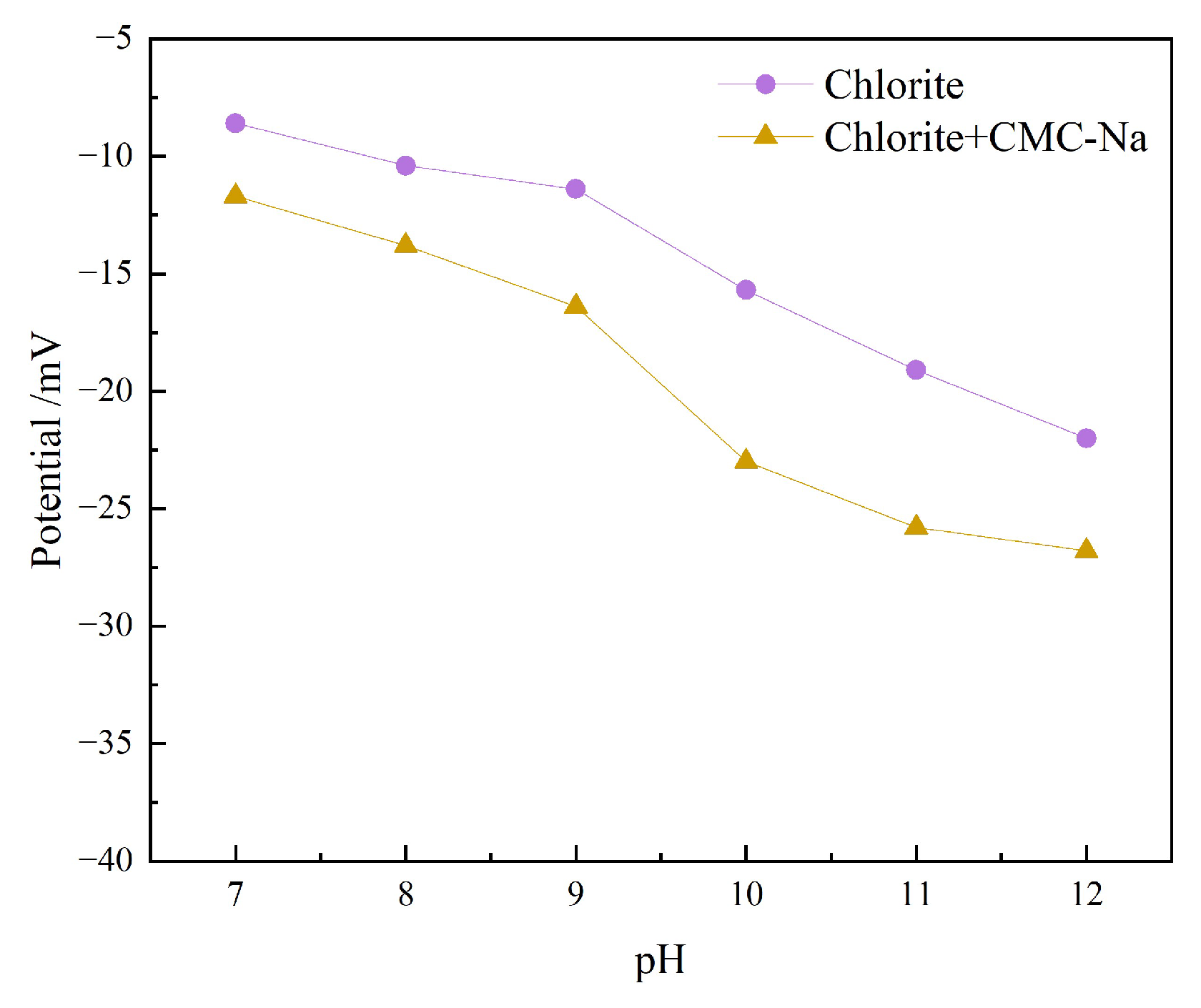
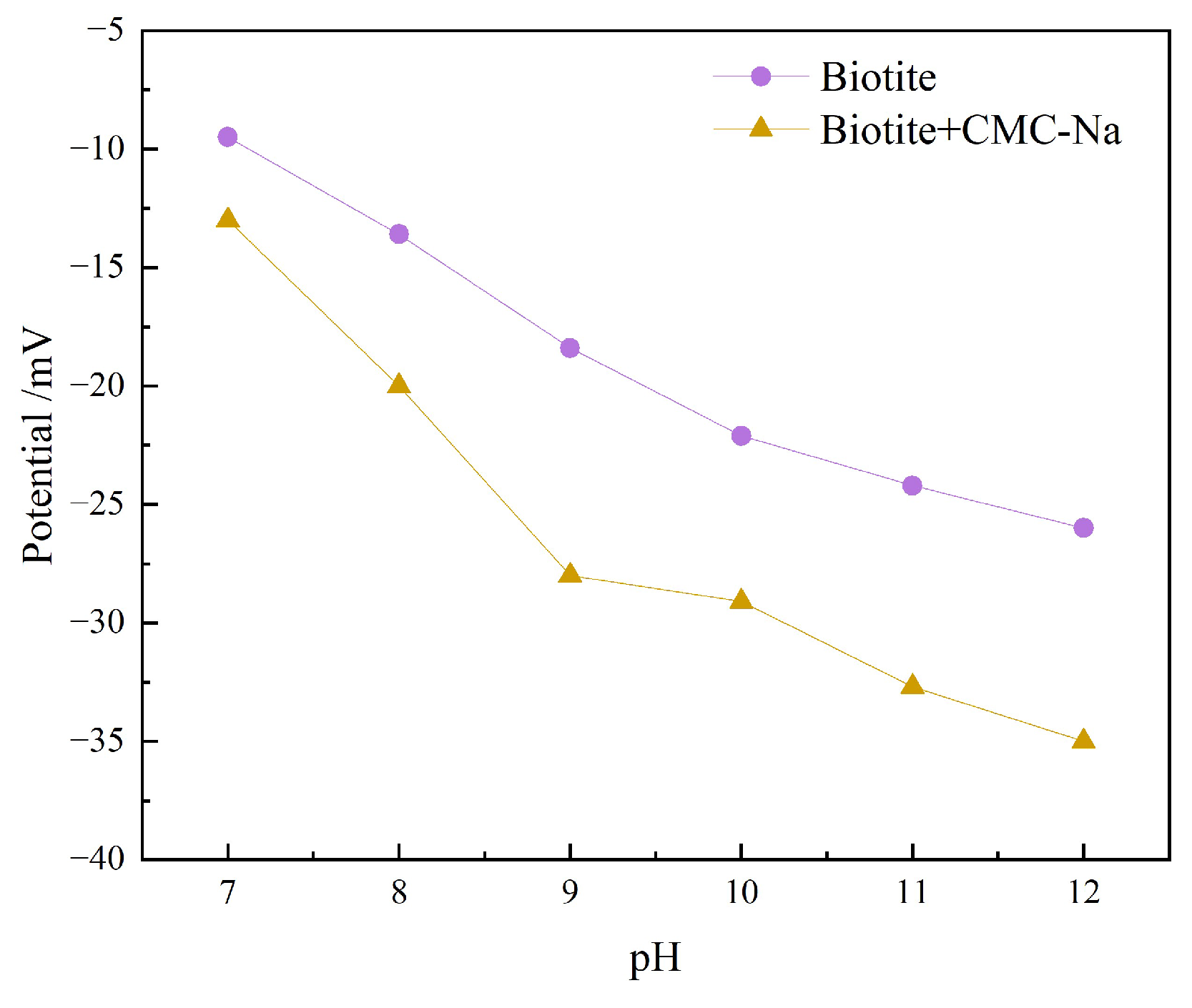
3.2.2. Zeta Potential Analysis
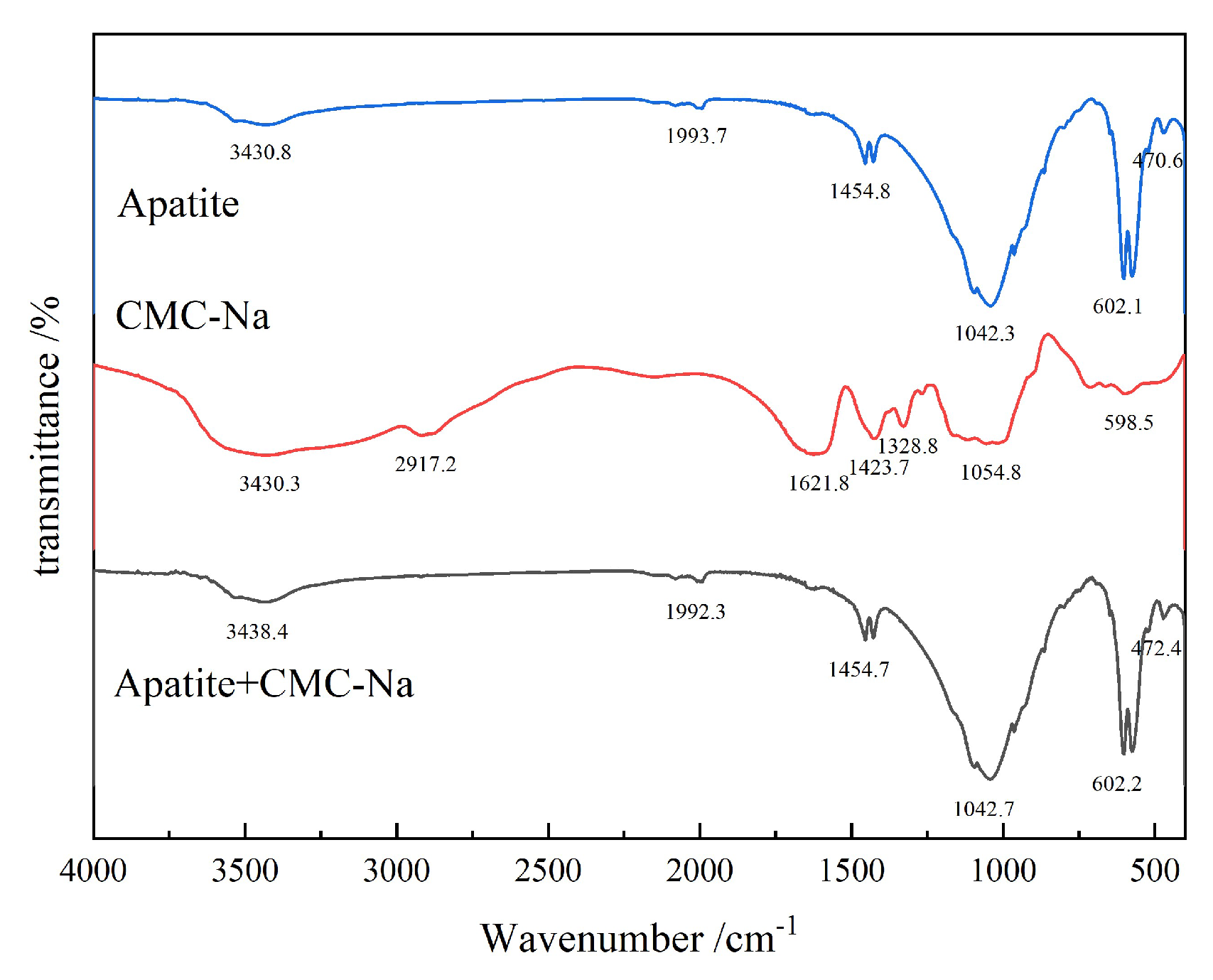
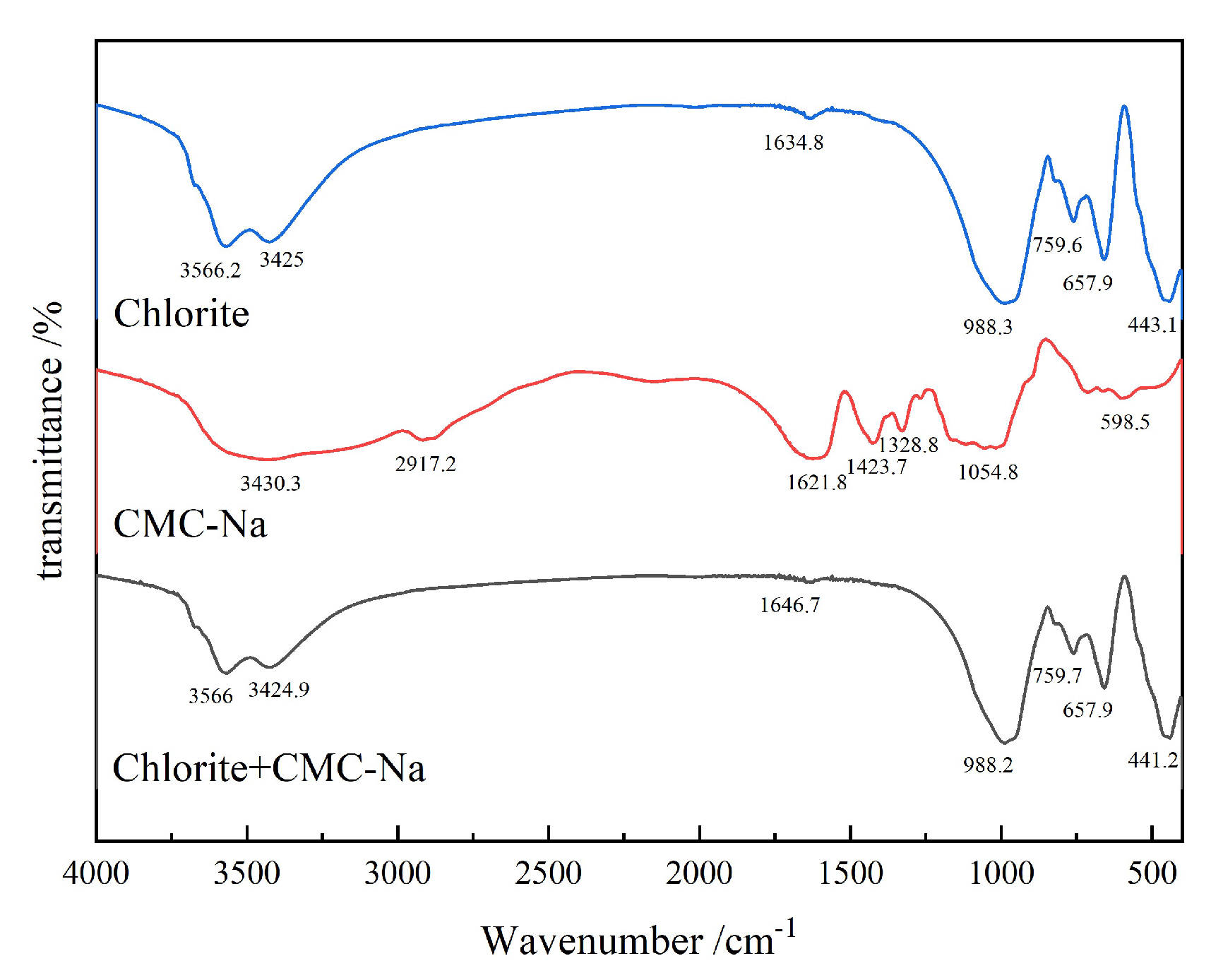
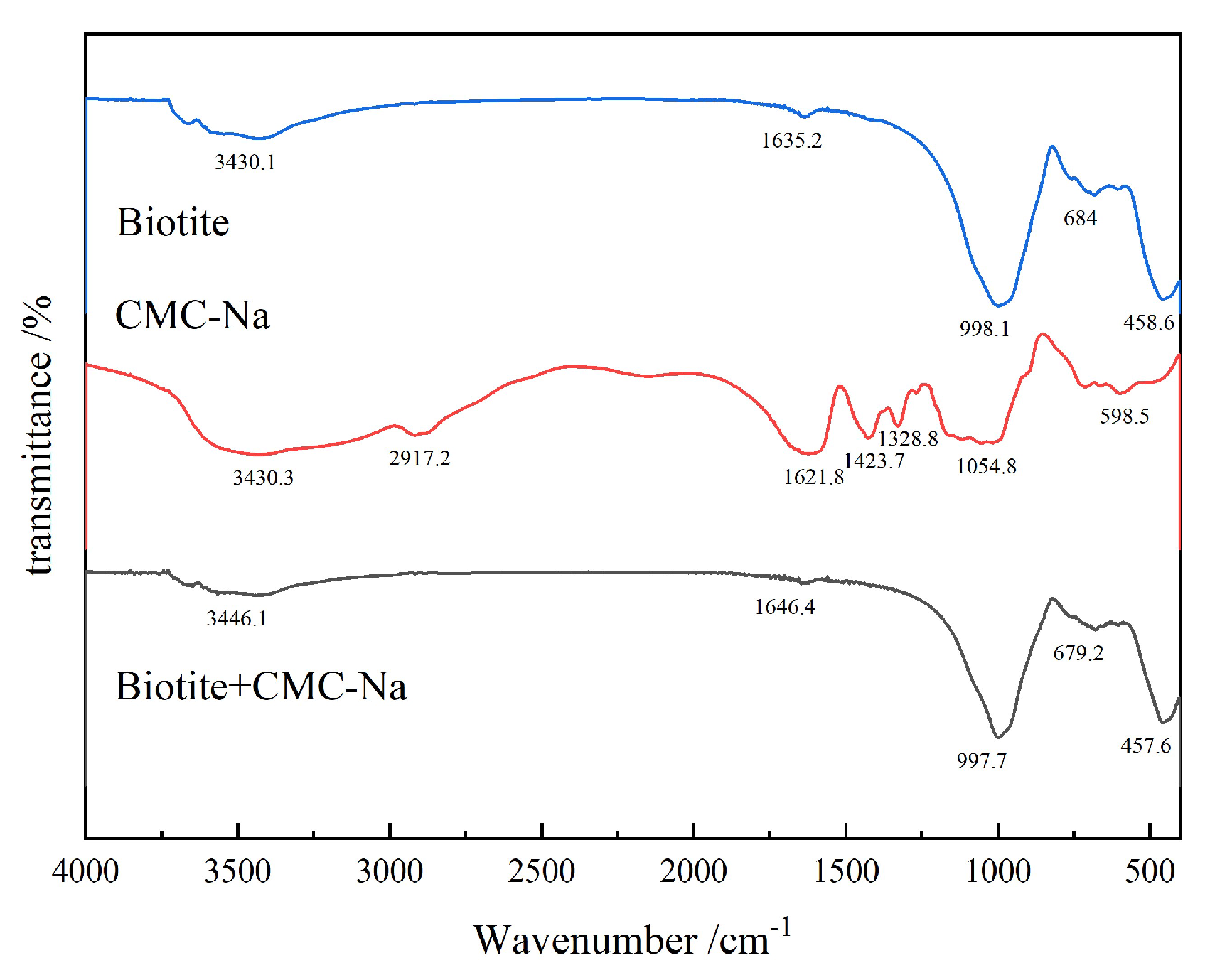
3.2.3. FTIR Analysis
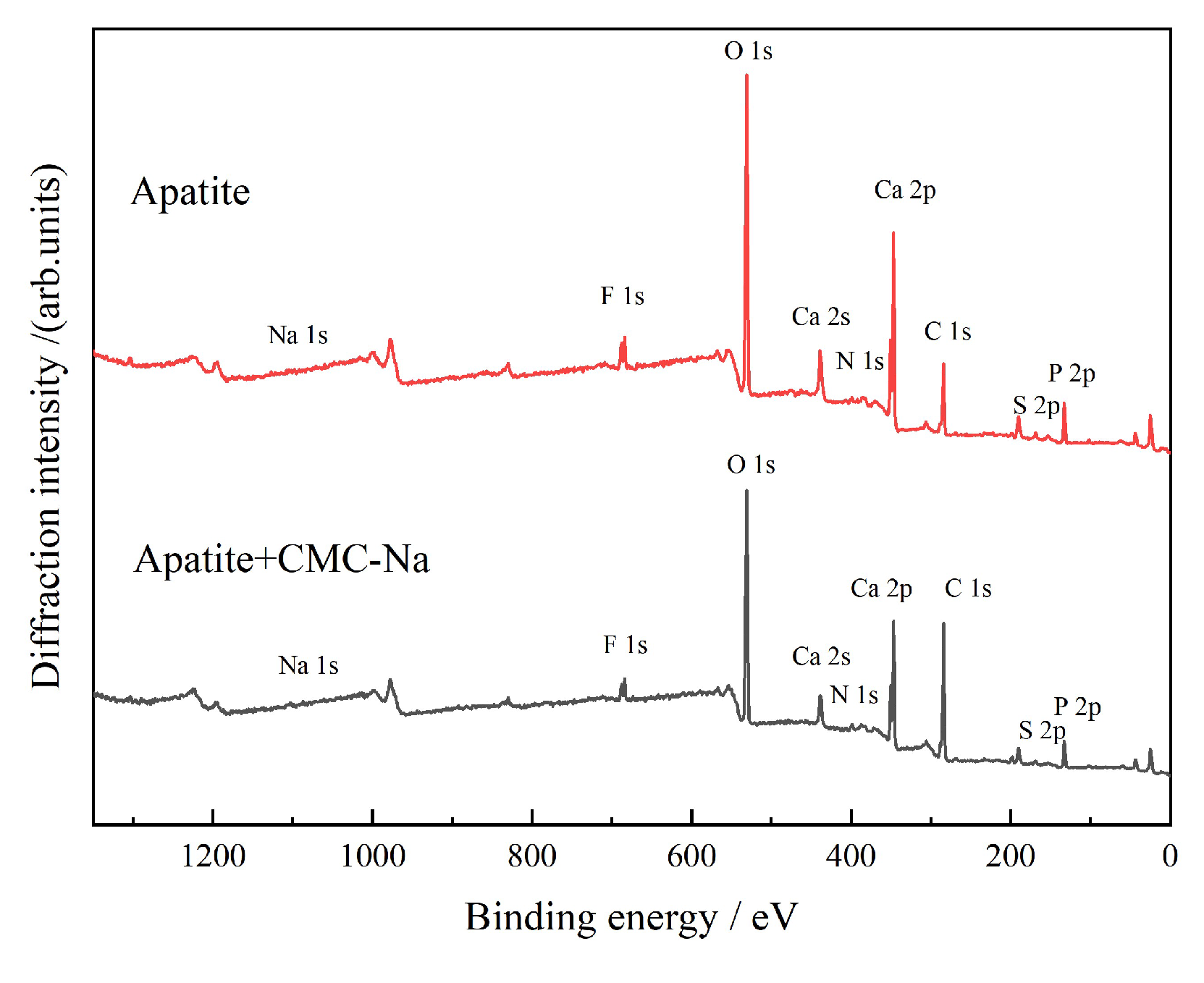
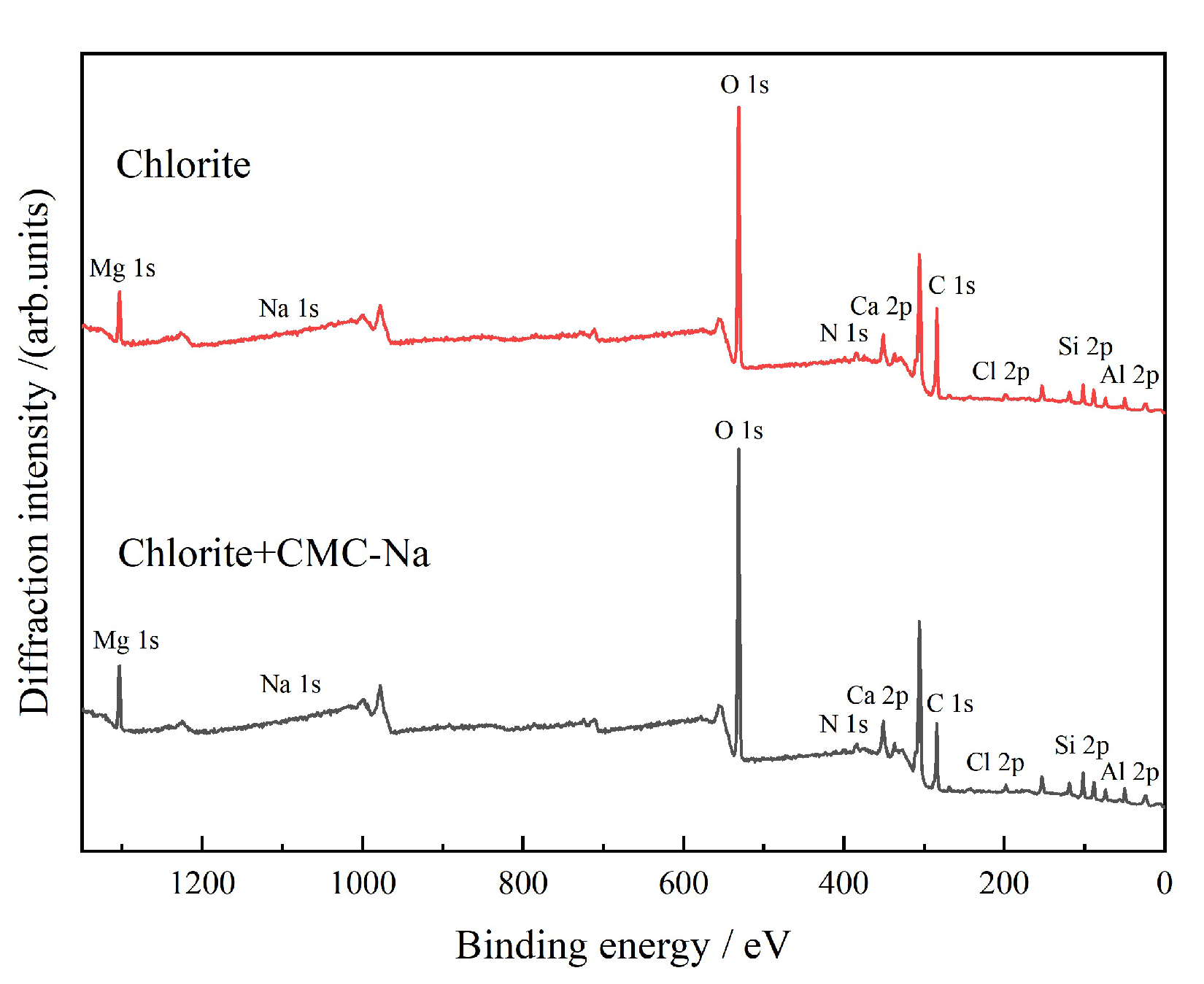
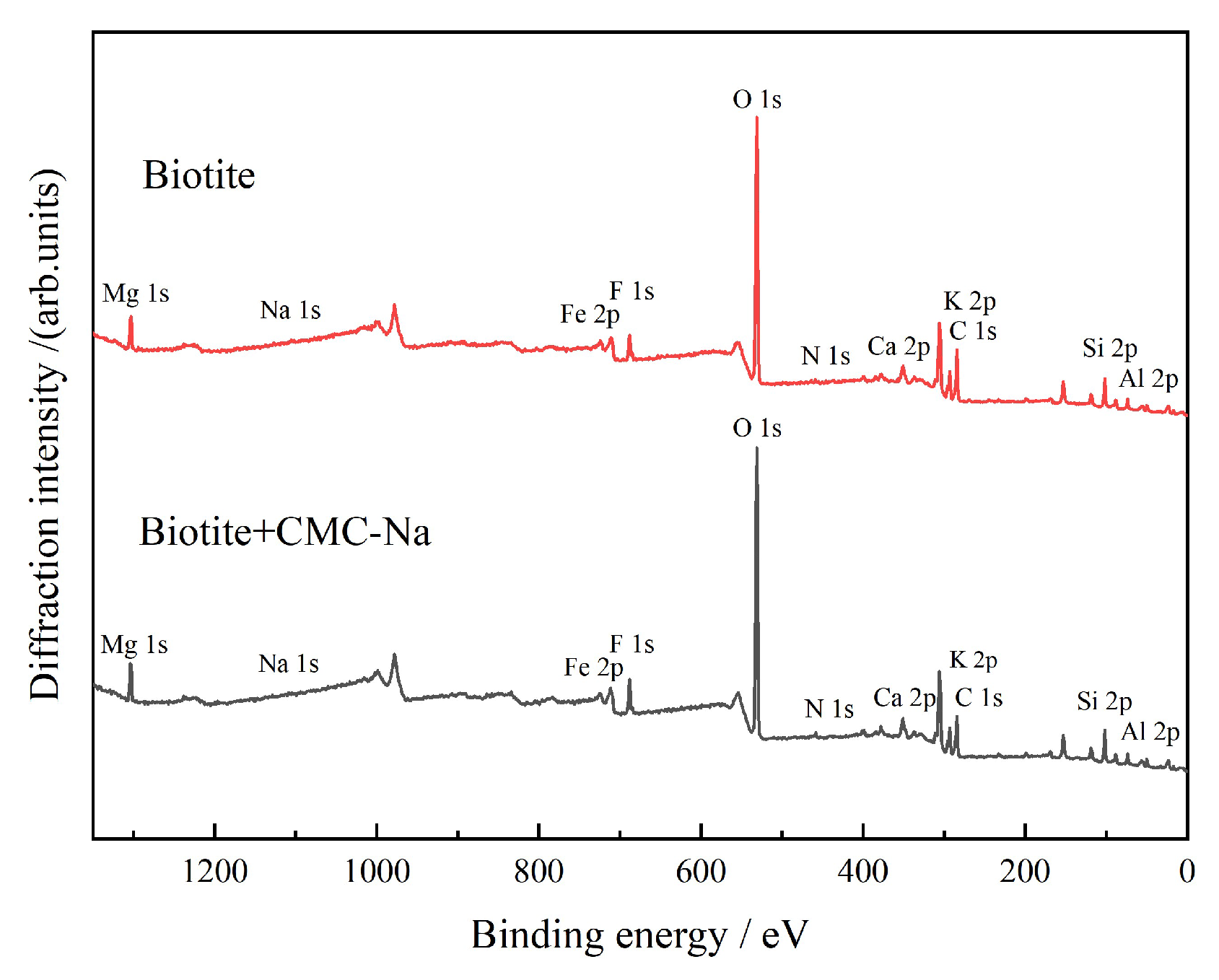
3.2.4. XPS Analysis
- (1)
- (2)
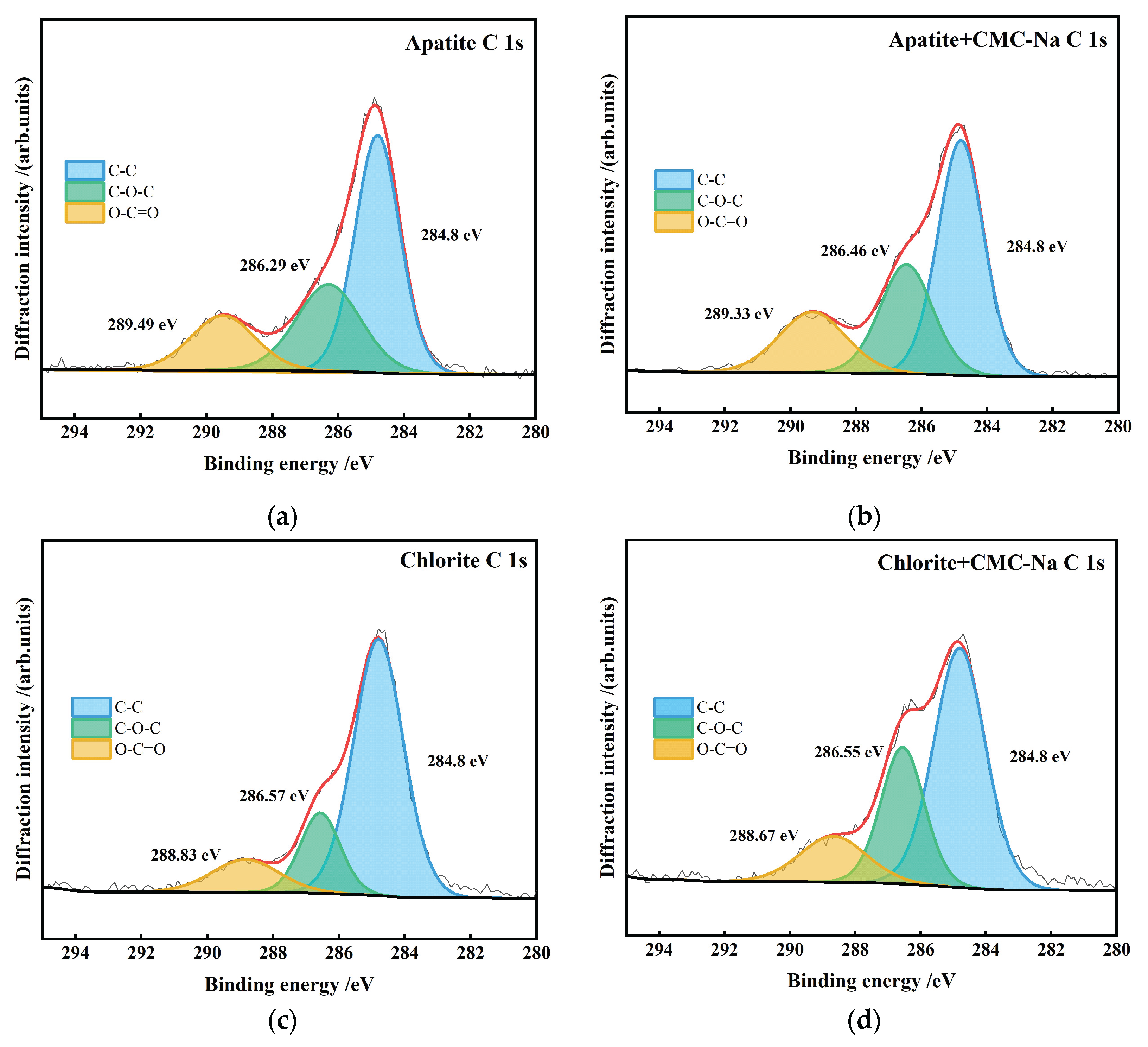
- Figure 24 presents the peak-fitting results of high-resolution C 1s spectra of the three minerals before and after CMC-Na treatment.
- (3)
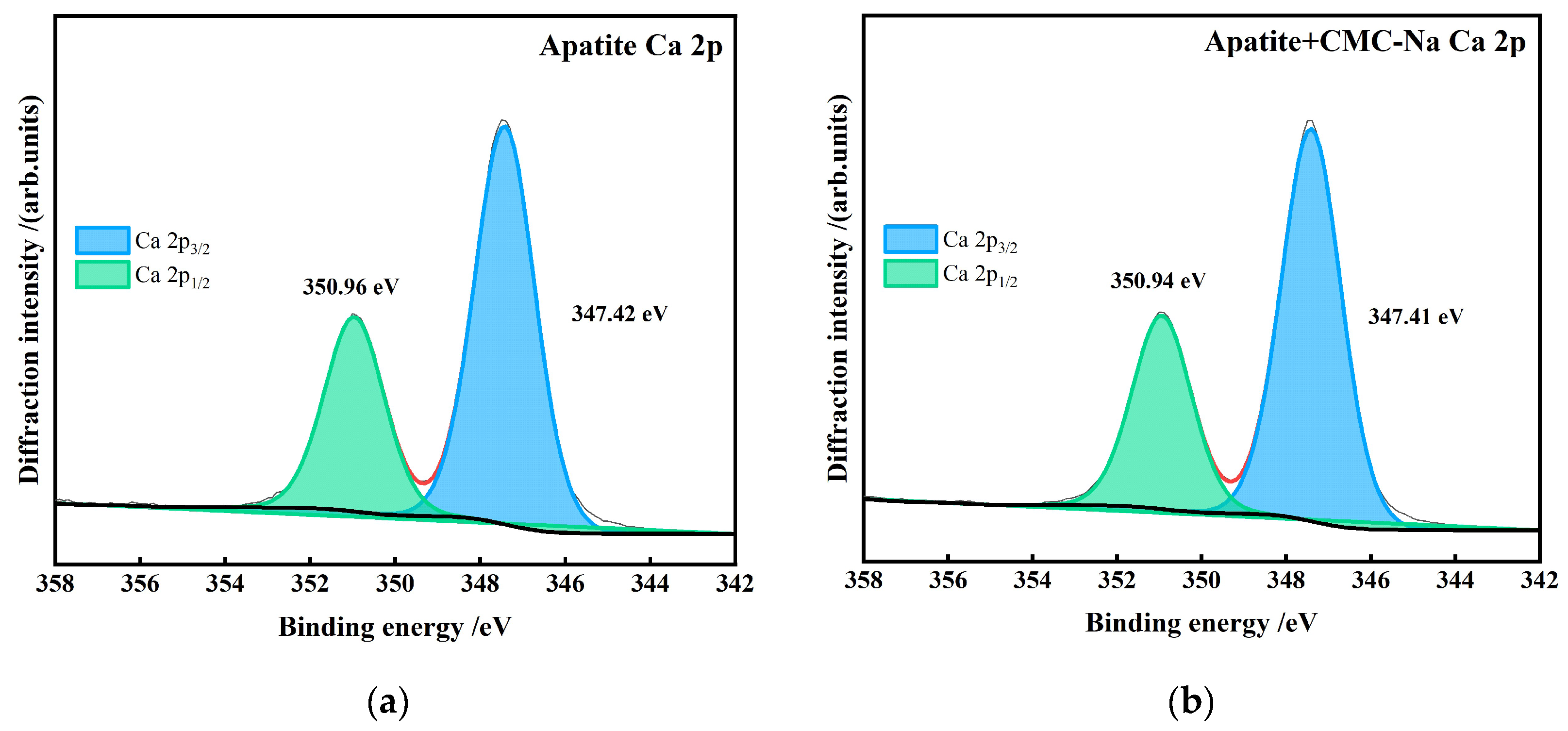
- The apatite surface is rich in Ca2+, which exhibits strong chemisorption interaction with CMC-Na. Figure 25 presents the peak-fitting results of high-resolution Ca 2p spectra of apatite before and after CMC-Na treatment.
- (4)
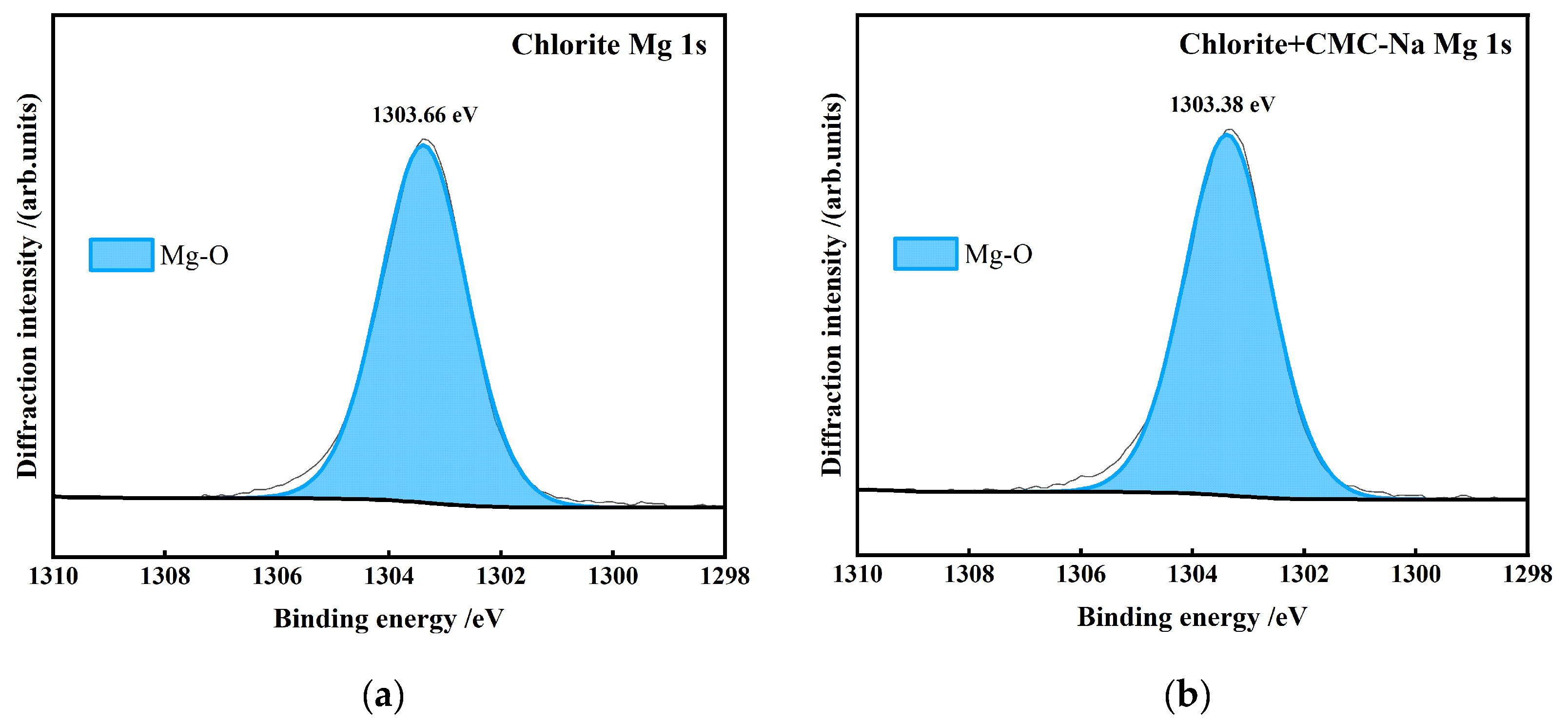
- Chlorite is a magnesium-bearing layered silicate mineral. CMC-Na can adsorb through interaction with Mg2+ in the interlayer or at edges, resulting in negative shift of the Mg 1s peak. Figure 26 presents the peak-fitting results of high-resolution Mg 1s spectra of chlorite before and after CMC-Na treatment.
- (5)
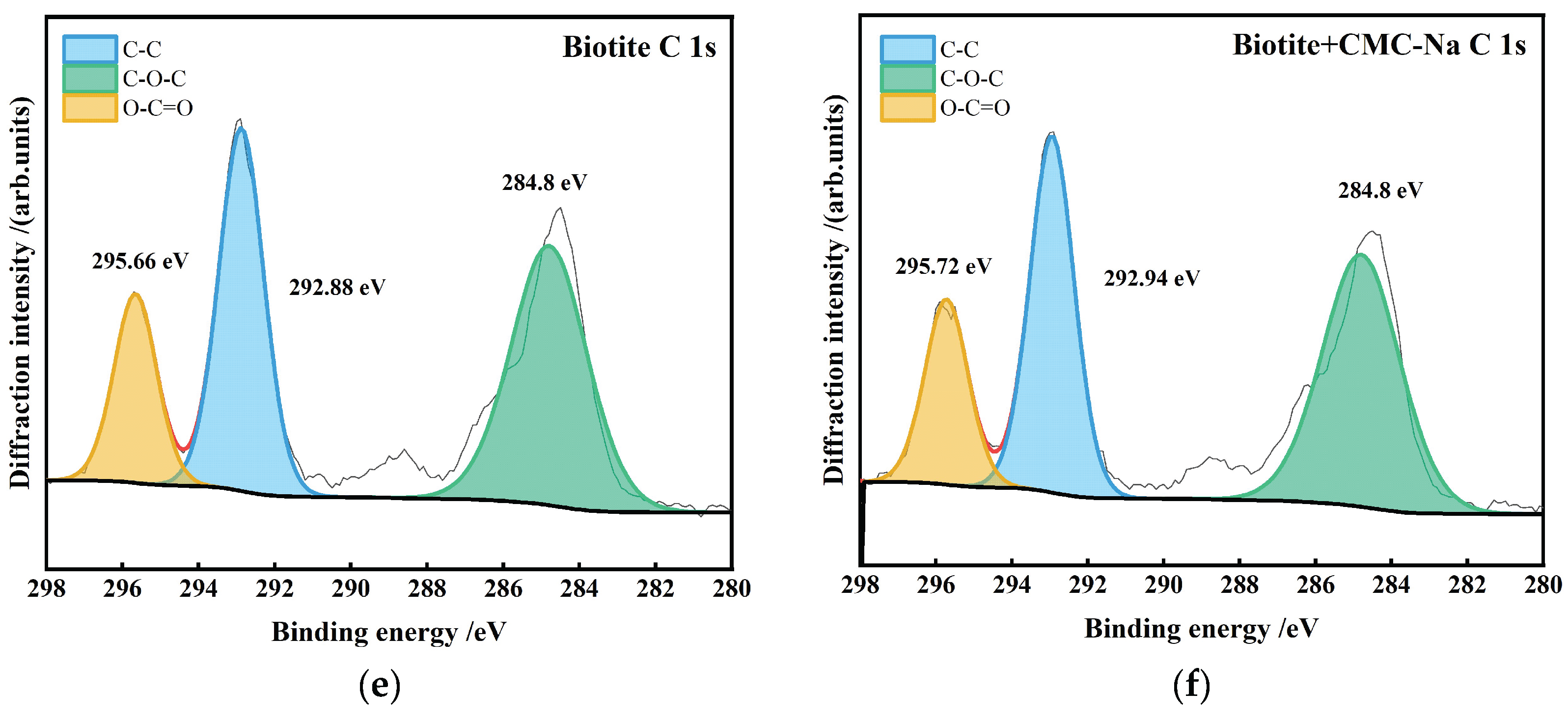
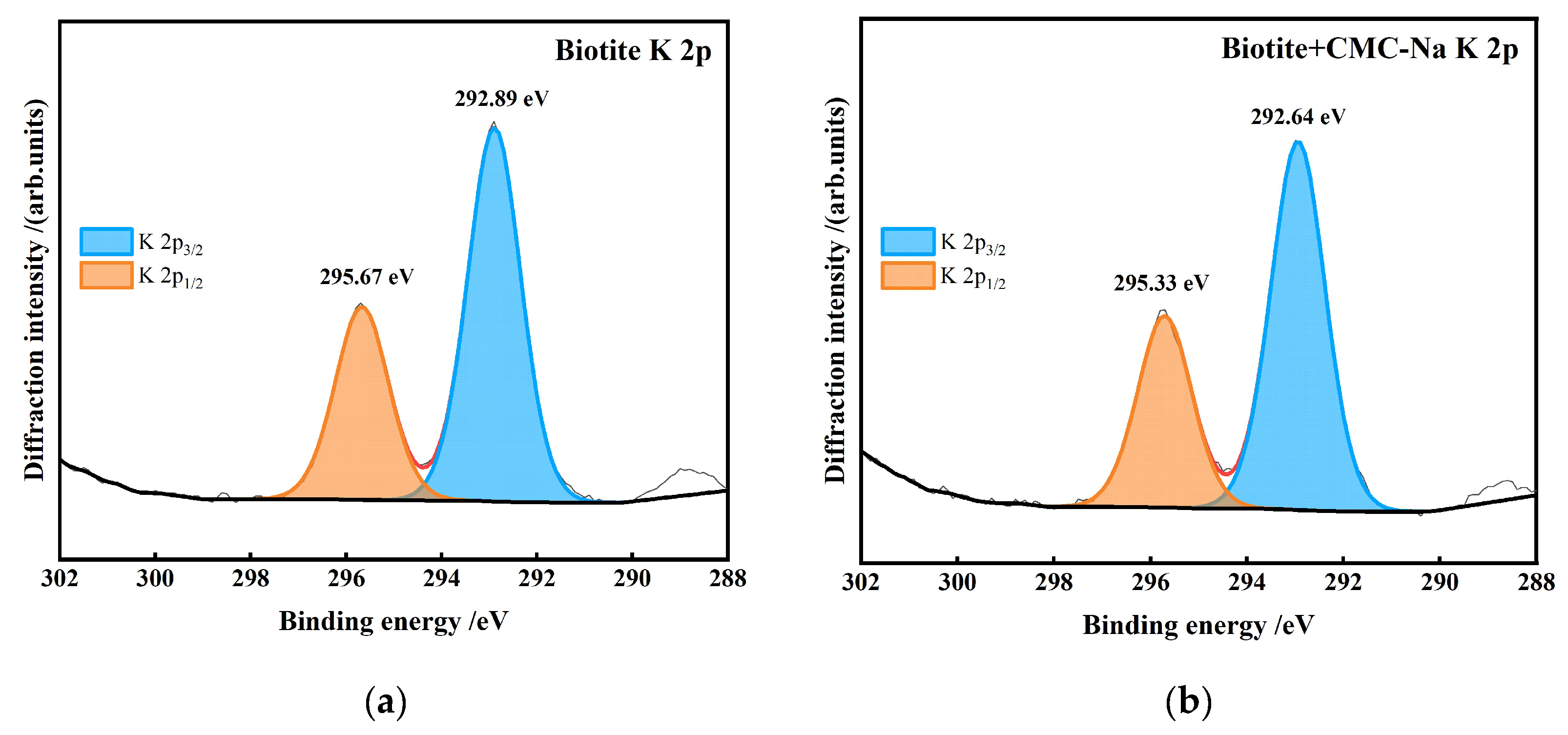
- Biotite is a potassium-bearing layered silicate mineral. The interlayer K⁺ ions are highly active. Figure 27 presents the peak-fitting results of high-resolution K 2p spectra of biotite before and after CMC-Na treatment.
4. Results
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CMC-Na | Carboxymethyl Cellulose Sodium |
References
- Zhu, P.C.; LI, X.; LI, F.; Wei, J.X. Process mineralogy research and flotation experiment in a phosphate mine of Yichang. Mining Research and Development 2018, 38, 85–88. [Google Scholar]
- Dong, L.Y.; Cui, Y.R.; Qiao, L.D. A critical review on the flotation of calcium-containing minerals. Separation and Purification Technology 2025, 360, 131082. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, Z.X.; Tao, D.P. Experimental Study on Reverse Flotation Desilication of Phosphate Using a Novel Cationic Collector. Mining Research and Development 2026, 46, 291–300. [Google Scholar]
- Xue, K.; Zhang, R.Y. Advances of researches on the distribution and metallogenic characteristics of phosphorous deposits in China. Acta Mineralogica Sinica 2019, 39, 7–14. [Google Scholar]
- Yin, W.Z. Progress in research on mineral flotation interaction effects. Nonferrous Metals (Mineral Processing Section) 2024, 1–12, 56. [Google Scholar]
- Jia, Y.K.; Yao, J.; Yin, W.Z.; Yang, P. Ultrasonic Regulation of the Interaction Mechanism Between Hematite and Chlorite During Flotation. Metal Mine 2025, 09, 107–113. [Google Scholar]
- Peng, L.Q.; Liu, R.Z.; Li, R.L.; Zhang, C.W.; Luo, K.Y.; Zhang, C. The scrubbing-flotation research on collophanite with higher silica and sesquioxide content. Yunnan Chemical Technology 2024, 51, 69–71. [Google Scholar]
- Guo, Y.J.; Li, j.l.; Fan, P.Q.; Li, H.B.; Chen, C.Y.; Du, L.P.; Xu, S. Experimental Study on a New Process of Classification Flotation Desilication of Phosphogypsum in Yunnan. Non-Metallic Mines 2022, 45, 53–56. [Google Scholar]
- Huang, G.; Xu, H.X.; Ma, L.Q.; Lun, W. Improving coal flotation by classified conditioning. International Journal of Coal Preparation and Utilization 2018, 38, 361–373. [Google Scholar] [CrossRef]
- Ruan, Y.Y.; Zhou, J.H.; Zhou, F.; Luo, H.H.; Wang, X.L.; Chi, R.A.; Deng, N.B. Study on process mineralogy characteristics and mixed reverse flotation test of Houping phosphate rock in Hubei. Industrial Minerals & Processing 2026, 55, 1–7. [Google Scholar]
- Che, Y.H.; Chen, W.; Zhu, Y.Y.; Qi, J.; Zhao, G.; Yang, L. Review on the Research Progress of Regulators Towards Clay Mineral Flotation. Metal Mine 2023, 36–49. [Google Scholar]
- Du, L.P.; Chen, C.Y.; Zhong, J.; Guo, Y.J. Brief analysis of phosphate beneficiation technology progress and problem countermeasures. Chemical Minerals & Processing 2016, 45, 57–61. [Google Scholar]
- Zhang, Q.; Mao, S.; Huang, X.F.; Chen, A.A.; Zhang, W.B. Research progress in surface chemistry of flotation for apatite and gangue minerals in phosphate ore. Mineral Protection and Utilization 2024, 44, 1–15. [Google Scholar]
- Dankwah, J.B.; Asamoah, R.K.; Zanin, M.; Skinner, W. Dense liquid flotation:Can coarse particle flotation performance be enhanced by controlling fluid density. Minerals Engineering 2022, 180, 107513. [Google Scholar] [CrossRef]
- Fatemeh, K.; Ataallah, B.; Yousef, G.; Abolfazl, D.; Morteza, A.; Hadi, F.; Mohsen, H. The interaction and synergic effect of particle size on flotation efficiency: A comparison study of recovery by size,and byliberation between lab and industrial scale dana. Rudarsko-geološko-naftni zbornik 2023, 38, 1–12. [Google Scholar]
- Meftuni, Y.; Sonmez, I. Effect of the hydrophobic fraction and particle size in the collectorless column flotation kinetics. Colloids and Surfaces A:Physicochemical andEngineering Aspects 1997, 121, 9–13. [Google Scholar]
- Cheng, H.Z.; Zhang, B. Course of development of fine coal flotation process in China. Coal Preparation Technology 2024, 52, 56–61. [Google Scholar]
- Meng, C.; Li, L.X.; Shen, S.P.; Qi, S.L.; Yu, J.; Yuan, Z.T. Experiments on classified magnetic separation-classified flotation for a ilmenite. Mineral Protection and Utilization 2017, 59–63. [Google Scholar]
- He, H.T.; Zhang, H.; Yang, W.Q.; Cai, Z.J. Effect of particle size on the desilication of Kunyang Yunnan collophonite by reverse flotation. Yunnan Chemical Industry 2021, 48, 32–34. [Google Scholar]
- Li, Y.H.; Liu, H.; Yang, B.Q.; Zeng, M.Y.; Zhang, H.Q.; He, G.S.; Luo, H.H. Application and its mechanism of sodium lignosulfonatein in magnesium removal from phosphate ore by direct flotation. Nonferrous Metals (Mineral Processing Section) 2023, 3, 152–157. [Google Scholar]
- Li, Z.H.; Xiong, K.; Zuo, K.S.; Gao, P. Effect of Carboxymethyl Cellulose on Flotation of Ascharite/Serpentine and Its Action Mechanism. Metal Mine 2024, 10, 107–111. [Google Scholar]
- Hu, Y.W.; Wang, L.; Wang, J.L.; Cao, Z.; Liu, L.; Li, G. Influences and Mechanisms of Carboxymethyl Cellulose on the Flotation Behavior of Columbite and Ankerite. Nonferrous Metals (Mineral Processing Section) 2025, 11, 109–119. [Google Scholar]
- Wu, H.B. Adsorption behavior and mechanism of cellulose inhibitors in the system of scheelite flotation. Master's Thesis, Jiangxi University of Science and Technology, Ganzhou, China, 2018. [Google Scholar]
- Zhang, W.P. Effects of macromolecular organic depressants on flotation behavior of apatite and calcium carbonate minerals and the mechanism. Master's Thesis, Jiangxi University of Science and Technology, Ganzhou, China, 2020. [Google Scholar]
- Ma, Q.; Li, Y.B.; Li, W.Q.; Yang, X. Study on the reaction mechanism of fluorite and calcite flotation separation by carboxymethyl cellulose. Metal Mine 2022, 7, 187–192. [Google Scholar]
- Guo, F.; Li, J. Separation strategies for Jordanian phosphate rock with siliceous and calcareous gangues. Mineral Processing 2010, 97, 74–78. [Google Scholar] [CrossRef]
- Lópze-valdivieso, A.; Lozano-ledesma, L.A.; Robledo-cabrera; A. Orozco-Navarro, O.A. Carboxymethylcellulose (CMC) as PbS depressant in the processing of Pb-Cu bulk concentrates:Adsorption and floatability studies. Minerals Engineering 2017, 112, 77–83. [Google Scholar] [CrossRef]
- Zhang, H.; Li, H.B.; Zhao, F.T.; Yang, W.Q.; Liu, R.Z. Study and application of determination method of contact angle of phosphate powder. Chemical Minerals & Processing Matters 2020, 49, 22–24. [Google Scholar]
- Huang, X.F.; Gong, F.Z. Measurement of contact angles of solid powder by Washburn osmotic pressure method. Laboratory Research and Exploration 2003, 5, 48–50. [Google Scholar]
- Wang, J.L.; Wang, J.L.; Shi, J.Y.; Wu, X.; Cao, Z. Research progress on surface property testing methods for mineral flotation. Minerals Protection and Utilization 2025, 45, 101–113. [Google Scholar]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




