Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Laboratory Set-Up and Experimental Procedure
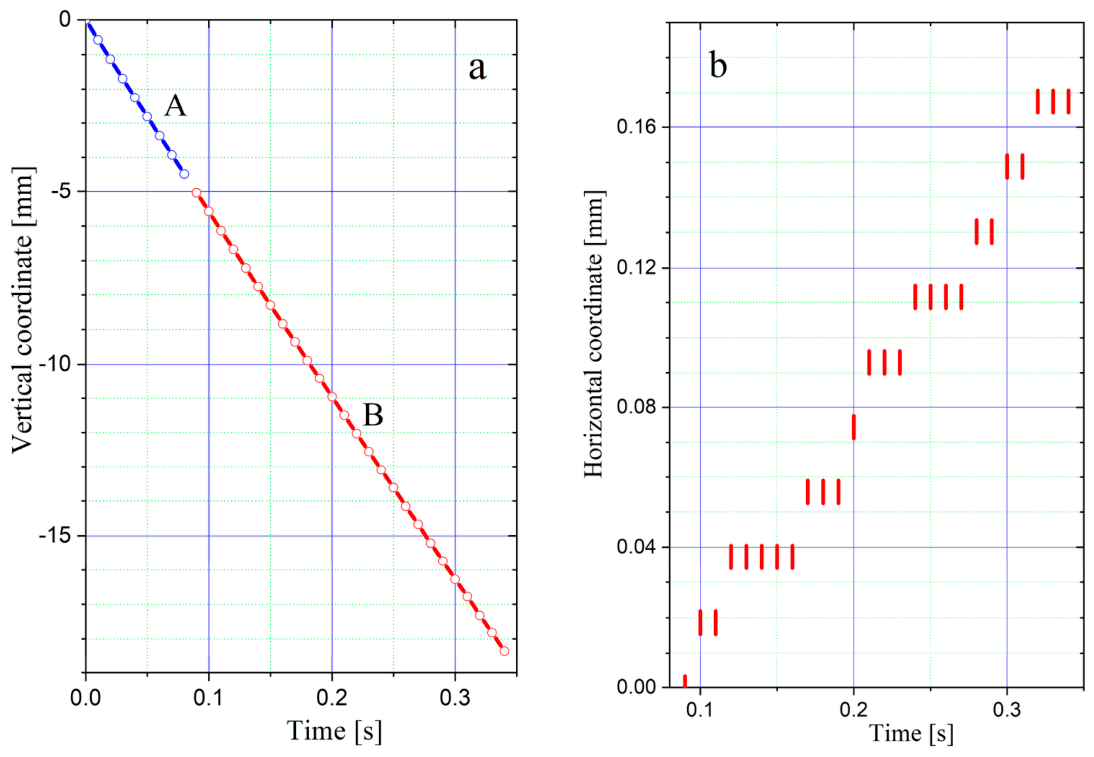
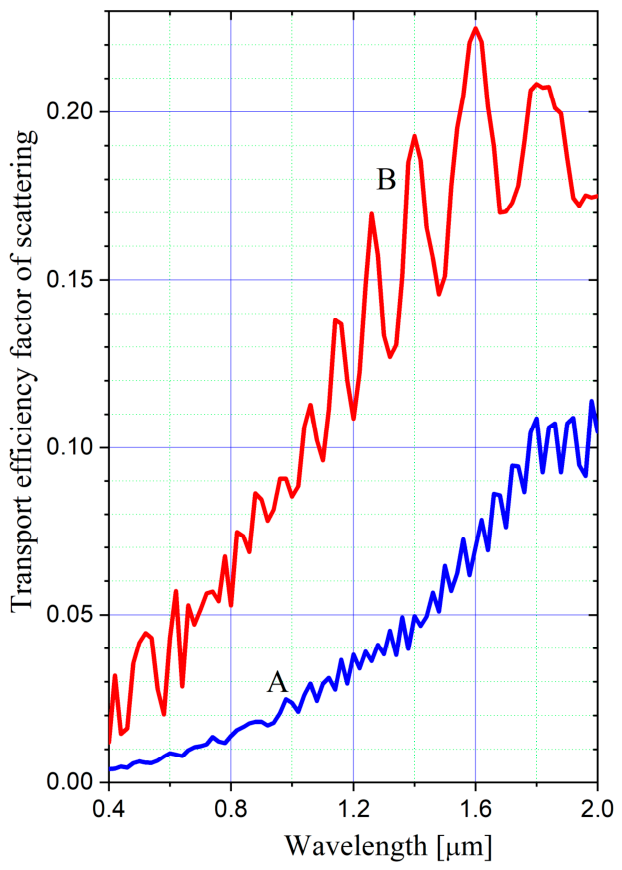
3. Experimental Results
4. Some Applications
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vehring, R. Pharmaceutical particle engineering via spray drying. Pharm. Res. 2008, 25(5), 999–1022. [Google Scholar] [CrossRef]
- Katon, H.; Wandrey, A.J.; Gander, B. Kinetics of solvent extraction/evaporation process for PLGA microparticle fabrication. Int. J. Pharmaceut. 2008, 364, 45–53. [Google Scholar]
- Van Eedenbrugh, B.; Baird, J.A.; Taylor, L.S. Cystallization tendency of active pharmaceutical ingredients following rapid solvent evaporation – classification and comparison with crystallization tendency from undercooled melts. J. Pharmaceut. Sci. 2010, 99(9), 3826–3838. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Zafar, N.; Fessi, H.; Elaissari, A. Double emulsion solvent evaporation techniques used for drug encapsulation. Int. J. Pharmaceut. 2015, 462(2), 179–190. [Google Scholar] [CrossRef] [PubMed]
- Zang, D.; Tarafdar, S.; Tarasevich, Yu.Yu.; Choudhurry, M.D.; Dutta, T. Evaporation of a droplet: From physics to applications. Phys. Rep. 2019, 804, 1–56. [Google Scholar] [CrossRef]
- Ziaee, A.; Albadarin, A.B.; Padrela, L.; Femmer, T.; O’Reilly, E.; Walker, G. Spray drying of pharmaceuticals and biopharmaceuticals: Critical parameters and experimental process optimization approaches. Eur. J. Pharm. Sci. 2019, 127, 300–318. [Google Scholar] [CrossRef]
- Korornaczyk, M.O.; Würtenberger, S.; Baumgartner, S. Impact of succussion on pharmaceutical preparations analysed by means of patterns from evaporated droplets. Sci. Rep. 2020, 570. [Google Scholar] [CrossRef]
- Boel, E.; Koerjekx, R.; Dedroog, S.; Babkin, I.; Vetrano, M.R.; et al. Unravelling particle formation: From single droplet drying to spray drying and electrospraying. Pharmaceutics 2020, 12, 625. [Google Scholar] [CrossRef]
- Baumann, J.M.; Adam, M.S.; Wood, J.D. Engineering advances in spray drying for pharmaceuticals. Annu. Rev. Chem. Biomol. Eng. 2021, 12, 217–240. [Google Scholar] [CrossRef]
- Chen, Q.; Thu, K.; Bui, T.D.; Li, Y.; Ng, K.C.; Chua, K.J. Development of a model for spray evaporation based on droplet analysis. Desalination 2016, 399, 69–77. [Google Scholar] [CrossRef]
- Chen, Q.; Kum, Ja.M.; Li, Y.; Chua, K.J. Experimental and mathematical study of the spray flash evaporation phenomena. Appl. Therm. Eng. 2018, 130, 598–610. [Google Scholar] [CrossRef]
- Dong, C.; Huang, Y.; Zhang, L. Slug flow-enhanced vacuum membrane distillation for seawater desalination: Flux improvement and anti-fouling effect. Sep. Purif. Tech. 2023, 320, 124178. [Google Scholar] [CrossRef]
- Li, A.; He, M.; Zheng, M.; Liu, H.; Liu, Y.; et al. Bioinspired multi-scale core-spun yarn-based solar evaporator for ultra-efficient and durable high-salinity brine desalination. Fundam. Res. 2025, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Dombrovsky, L.A.; Levashov, V.Yu.; Kryukov, A.P.; Dembele, S.; Wen, J.X. A comparative analysis of shielding of thermal radiation of fires using mist curtains containing droplets of pure water or sea water. Int. J. Therm. Sci. 2020, 152, 106299. [Google Scholar] [CrossRef]
- Dombrovsky, L.A.; Dembele, S. An improved solution for shielding of thermal radiation of fires using mist curtains of pure water or seawater. Comput. Therm. Sci. 2022, 14(4), 1–18. [Google Scholar] [CrossRef]
- Zhou, Z.; Cao, C.; Cao, L.; Zheng, L.; Xu, J.; et al. Evaporation kinetics of surfactant solution droplets on rice (Oryza sativa) leaves. PLOS One 2017, 12, e0176870. [Google Scholar] [CrossRef]
- Zhou, Z.; Cao, C.; Cao, L.; Zheng, L.; Xu, J.; et al. Effect of surfactant concentration on the evaporation of droplets on cotton (Gossypium hirsutum L.) leaves. Colloids Surf. B 2018, 167, 206–212. [Google Scholar] [CrossRef]
- Li, H.; Cryer, S.; Raymond, J.; Acharya, L. Interpreting atomization of agricultural spray image patterns using latent Dirichlet allocation techniques. Art. Intel. Agric. 2020, 4, 253–261. [Google Scholar] [CrossRef]
- Gao, K.; Haddad, S.; Paolini, R.; Feng, J.; Altheeb, M.; et al. The use of green infrastructure and irrigation in the mitigation of urban heat in a desert city. Build Simul. 2024, 17, 679–694. [Google Scholar] [CrossRef]
- Lankinen, Å; Witzell, J.; Aleklett, K.; Furenhed, S.; ·Green, K.K.; et al. Challenges and opportunities for increasing the use of low-risk plant protection products in sustainable production. A review. Agron. Sustain. Dev. 2024, 44, 21. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, Y.; Yang, Y.; Wu, S.; Zhang, X.; Ren, X. Movement and control of evaporating droplets released from an open surface tank in the push‒pull ventilation system. Build Simul. 2016, 9, 443–457. [Google Scholar] [CrossRef]
- Duverge, J.J.; Rajagopalan, P. Assessment of factors influencing the energy and water performance of aquatic centres. Build Simul. 2020, 13, 771–786. [Google Scholar] [CrossRef]
- Shahidzadeh, N.; Schut, M.F.L.; Desarnaud, J.; Prat, M.; Bonn, D. Salt stains from evaporating droplets. Sci. Rep. 2015, 5, 10355. [Google Scholar] [CrossRef]
- Chi, J.W.; Li, W.J.; Zhang, D.Z.; Zhang, J.C.; Lin, Y.T.; et al. Sea salt aerosols as a reactive surface for inorganic and organic acidic gases in the Arctic troposphere. Atmos. Chem. Phys. 2015, 15, 11341–11353. [Google Scholar] [CrossRef]
- Schiffer, J.M.; Mael, L.E.; Prather, K.A.; Amaro, R.E.; Grassian, V.H. Sea spray aerosols: Where marine biology meets atmospheric chemistry. ACS Cent. Sci. 2018, 4(12), 1617–1623. [Google Scholar] [CrossRef] [PubMed]
- Brian, H.; Froyd, K.; Murphy, D.M.; Dibb, J.; Darmenov, A.; et al. Observationally constrained analysis of sea salt aerosols in the marine atmosphere. Atmos. Chem. Phys. 2019, 19(16), 10773–10785. [Google Scholar]
- Ackerman, K.L.; Nugent, A.D.; Taing, C. Mechanisms controlling giant sea salt aerosols size distributions along a tropical orographic coastline. Atmos. Chem. Phys. 2023, 23(21), 13735–13753. [Google Scholar] [CrossRef]
- Walton, D.E.; Mumford, C.J. The morphology of spray-dried particles: The effect of process variables upon the morphology of spray-dried particles. Chem. Eng. Res. Des. 1999, 77(5), 442–460. [Google Scholar] [CrossRef]
- Vehring, R.; Foss, W.R.; Lechuga-Ballesteros, D. Particle formation in spray drying. Aerosol Sci. 2007, 38(7), 728–746. [Google Scholar] [CrossRef]
- Nandiyanto, A.B.D.; Okuyama, K. Progress in developing spray-drying methods for the production of controlled morphology particles: From the nanometer to submicrometer size ranges. Adv. Powder Tech. 2011, 22(1), 1–19. [Google Scholar] [CrossRef]
- Sadek, C.; Schuck, P.; Fallourd, Y.; Pradeau, N.; Le Froch-Fouéré, C.; Jeantet, R. Drying of a single droplet to investigate process–structure–function relationships: a review. Dairy Sci. Tech. 2015, 95, 771–794. [Google Scholar] [CrossRef]
- De Souza Lima, R.; Ré, M.-I.; Arlabosse, P. Drying droplet as a template for solid formation: A review. Powder Tech. 2020, 359, 161–171. [Google Scholar] [CrossRef]
- Dombrovsky, L.A.; Levashov, V.Yu.; Shoval, S.; Bormashenko, Ed. Progress in understanding of evaporation of droplets: Fundamentals and applications. Adv. Coll. Interface Sci. 2025, 344, 103605. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.G.; Blanchard, D.C.; Cipriano, R.J. The formation of hollow sea-salt particles from the evaporation of drops of seawater. Atmos. Res. 1988, 22(1), 15–25. [Google Scholar] [CrossRef]
- Handscomb, C.S.; Kraft, M.; Bayly, A.E. A new model for the drying of droplets containing suspended solids after shell formation. Chem. Eng. Sci. 2009, 64(2), 228–246. [Google Scholar] [CrossRef]
- Handscomb, C.S.; Kraft, M.; Bayly, A.E. A new model for the drying of droplets containing suspended solids. Chem. Eng. Sci. 2009, 64(4), 628–637. [Google Scholar] [CrossRef]
- Fairhurst, D. Droplets of ionic solutions, Chap. 20 in “Droplet Wetting and Evaporation. From Pure to Complex Fluids”; Brutin, D., Ed.; Acad. Press: New York, 2015; pp. 295–313. [Google Scholar]
- Mezhericher, M.; Levi, A.; Borde, I. Multi-scale multiphase modeling of transport phenomena in spray-drying processes. Drying Tech. 2015, 33(1), 2–23. [Google Scholar] [CrossRef]
- Quilaquero, M.; Aguilera, J.M. Crystallization of NaCl by fast evaporation of water droplets of NaCl solutions. Food Res. Int. 2016, 84, 143–149. [Google Scholar] [CrossRef]
- Gregson, F.K.A.; Robinson, J.F.; Miles, R.E.H.; Royall, C.P.; Reid, J.P. Drying kinetics of salt solution droplets: Water evaporation rates and crystallization. J. Phys. Chem. B 2019, 123(1), 266–276. [Google Scholar] [CrossRef]
- Nandiyanto, A.B.D.; Ogi, T.; Wang, W.-N.; Gradon, L.; Okuyama, K. Template-assisted spray-drying method for the fabrication of porous particles with tunable structures. Adv. Powder Tech. 2019, 30(12), 2908–2924. [Google Scholar] [CrossRef]
- Rezaei, M.; Netz, R.R. Water evaporation from solute-containing aerosol droplets: Effects of internal concentration and diffusivity profiles and onset of crust formation. Phys. Fluids 2021, 33, 091901. [Google Scholar] [CrossRef]
- Hardy, D.A.; Archer, J.; Lemaitre, P.; Vehring, R.; Reid, J.P.; Walker, J.S. High time resolution measurements of droplet evaporation kinetics and particle crystallization. Phys. Chem. Chem. Phys. 2021, 23(34), 18568−18579. [Google Scholar] [CrossRef]
- Hardy, D.A.; Robinson, J.F.; Hilditch, T.G.; Neal, E.; Lemaitre, P.; Walker, J.S.; Reid, J.P. Accurate measurements and simulations of the evaporation and trajectories of individual solution droplets. J. Phys. Chem. B 2023, 127(15), 3416−3430. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, K.; Olaleye, A.K.; Van den Akker, H.E.A. A porous-crust drying model for a single dairy droplet. Chem. Eng. Res. Des. 2023, 200, 741–752. [Google Scholar] [CrossRef]
- Licsandru, G.; Noiriel, C.; Duru, O.; Geoffroy, S.; Abou-Chakra, A.; Prat, M. Evaporative destabilization of a salt crust with branched pattern formation. Sci. Rep. 2023, 13, 5132. [Google Scholar] [CrossRef] [PubMed]
- Miles, B.E.A.; Winter, E.; Mirembe, S.; Hardy, D.; Mahato, L.K.; Miles, R.E.H.; Reid, J.P. Evaporation kinetics and final particle morphology of multicomponent salt solution droplets. J. Phys. Chem. A 2025, 129(3), 762–773. [Google Scholar] [CrossRef]
- Mignot, B.; Mahmud, T.; Heggs, P.J.; Ghadiri, M.; Roberts, K.J. Modelling and experimental validation of the evaporation and crystallization of a saline droplet. Chem. Eng. Sci. 2026, 322, 123114. [Google Scholar] [CrossRef]
- Wang, J.; Fiebig, M. Absolute measurements of the thermal diffusivity of aqueous solutions of sodium chloride. Int. J. Thermophys. 1998, 19(1), 15–25. [Google Scholar] [CrossRef]
- Hamann, C.H.; Hamnett, A.; Vielstich, W. Electrochemistry. Second Edition; Wiley: Weinheim, Germany, 2007. [Google Scholar]
- Zhang, J.; Clennell, M.B.; Dewhurst, D.N. Transport properties of NaCl in aqueous solution and hydrogen solubility in brine. J. Phys. Chem. B 2023, 127, 8900–8915. [Google Scholar] [CrossRef]
- Fedorets, A.A.; Shcherbakov, D.V.; Levashov, V.Yu.; Dombrovsky, L.A. Self-stabilization of droplet clusters levitating over heated salt water. Int. J. Therm. Sci. 2022, 182, 107822. [Google Scholar] [CrossRef]
- Fedorets, A.A.; Kolmakov, E.E.; Dombrovsky, L.A. Experimental study of the effect of water salinity on the parameters of an equilibrium droplet cluster levitating over a water layer. Front. Heat Mass Transfer 2024, 22(1), 1–14. [Google Scholar] [CrossRef]
- Fedorets, A.A.; Kolmakov, E.E.; Nasyrova, A.V.; Medvedev, D.N.; Mayorov, V.O.; Levashov, V.Yu.; Dombrovsky, L.A. Experimental method for studying the effect of dissolved substances on the evaporation rate of water droplets suspended in air. Front. Heat Mass Transfer 2025, 24(4), 1091–1102. [Google Scholar] [CrossRef]
- Goossens, W.R.A. Review of the empirical correlations for the drag coefficient of rigid spheres. Powder Tech. 2019, 352, 350–359. [Google Scholar] [CrossRef]
- Singh, N.; Kroells, M.; Li, C.; Ching, E.; Ihme, M.; Hogan, C.J.; Schwartzentruber, T.E. General drag coefficient for flow over spherical particles. AIAA J. 2022, 60(2), 587–597. [Google Scholar] [CrossRef]
- Van de Hulst, H.C. Light Scattering by Small Particles; Dover: New York, USA, 1981. [Google Scholar]
- Bohren, C.F.; Huffman, D.R. Absorption and Scattering of Light by Small Particles; Wiley: New York, USA, 1998. [Google Scholar]
- Mishchenko, M.I.; Travis, L.D.; Lacis, A.A. Multiple Scattering of Light by Particles: Radiative Transfer and Coherent Backscattering; Cambridge University Press: Cambridge (UK), 2006. [Google Scholar]
- Dombrovsky, L.A.; Baillis, D. Thermal Radiation in Disperse Systems: An Engineering Approach; Begell House: New York, USA, 2010. [Google Scholar]
- Mishchenko, M.I. Electromagnetic Scattering by Particles and Particle Groups: An Introduction; Cambridge University Press: New York, USA, 2014. [Google Scholar]
- Mishchenko, M.I. “Independent” and “dependent” scattering by particles in a multi-particle group. OSA Continuum 2018, 1(1), 243–260. [Google Scholar] [CrossRef]
- Galy, T.; Huang, D.; Pilon, L. Revisiting independent versus dependent scattering regimes in suspensions or aggregates of spherical particles. J. Quant. Spectr. Radiat. Transfer 2020, 246, 106924. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



