Submitted:
21 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
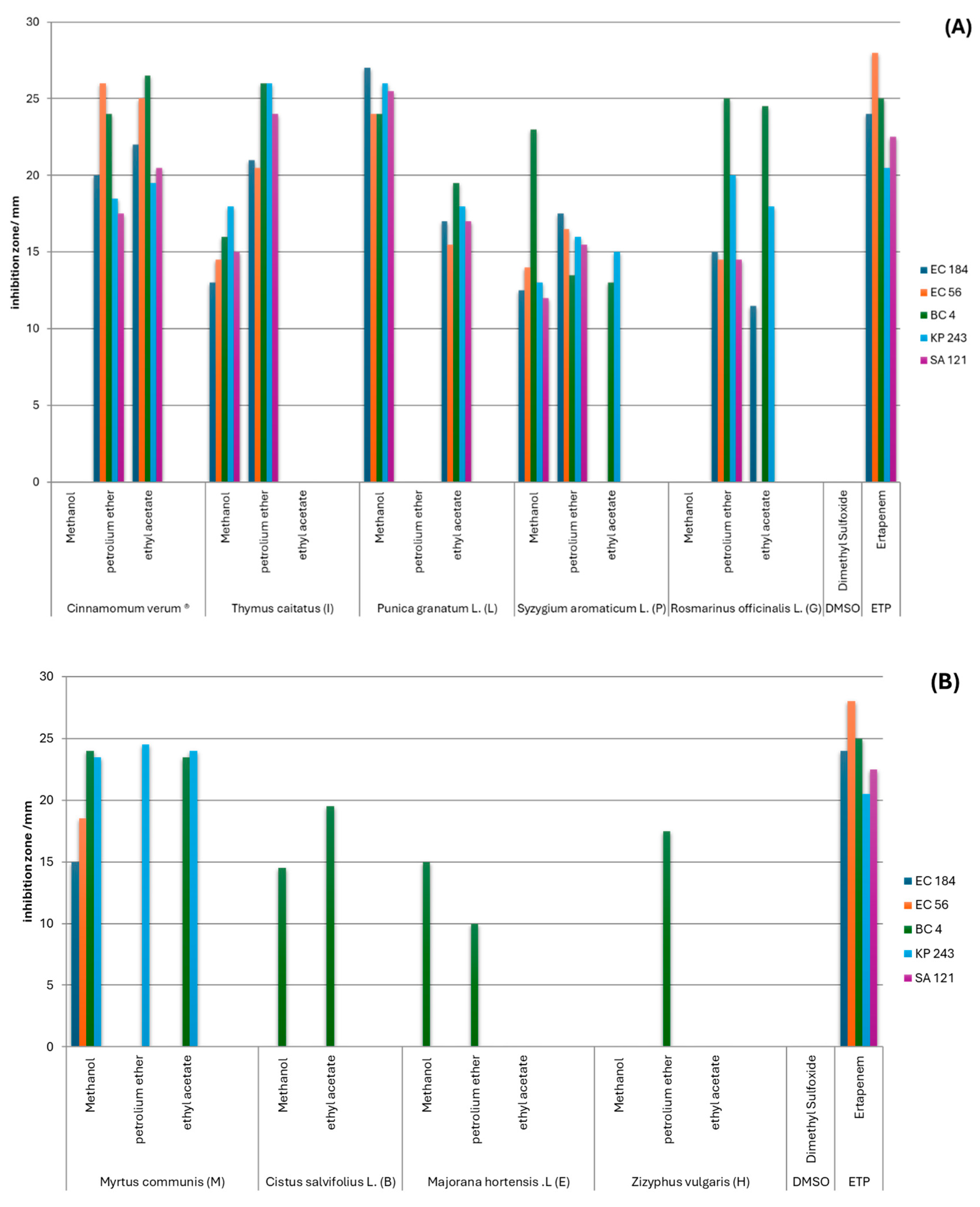
2.1. Screening of Antibacterial Activity by Agar Well Diffusion
2.2. Comparative Activity of Extracts by Solvent Type
2.3. Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
3. Discussion
4. Materials and Methods
4.1. Plant Collection and Identification
4.2. Preparation of Plant Extracts
4.3. Bacterial Strains
4.4. Determination of Antibacterial Activity (Agar Well Diffusion Method)
4.5. Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) Determination
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MIC | Minimum Inhibitory Concentration |
| MBC | Minimum Bactericidal Concentration |
| AMR | Antimicrobial Resistance |
| MRSA | Methicillin-Resistant Staphylococcus aureus |
| ESBL | Extended-Spectrum β-Lactamase |
| FLBC | Foodborne Libyan-Type Bacterial Collection |
References
- Al-Siddiq, M.S.; Omran, M.L.; Youssef, H.H. Efficacy of aqueous and alcoholic ginger extract on some types of pathogenic bacteria. Libyan J. Ecol. Environ. Sci. Technol. 2023, 5, 65–68. [Google Scholar]
- Al-Zereini, W.A.; Al-Trawneh, I.N.; Al-Qudah, M.A.; TumAllah, H.M.; Abudayeh, Z.H.; Hijazin, T. Antibacterial, antioxidant, and cytotoxic activities of Syzygium aromaticum (L.) Merr. & Perry essential oil with identification of its chemical constituents. Z. Naturforsch. C. 2023, 78, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Aouadhi, C.; Jouini, A.; Maaroufi, K. Antibacterial effect of eight essential oils against bacteria implicated in bovine mastitis and characterization of primary action mode of Thymus capitatus essential oil. Antibiotics 2024, 13, 237. [Google Scholar] [CrossRef] [PubMed]
- Asfa, B.; Woldemichael, D.N.; Tesfaw, L.; Asefa, L.; Desta, S.; Girma, S.; Tolera, T.S.; Tufa, T.B. Evaluating antimicrobial activity of selected medicinal plant extracts against pasteurellosis-causing bacteria in small ruminants. Front. Vet. Sci. 2025, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Azwai, S.M.; Lawila, A.F.; Eshamah, H.L.; Sherif, J.A.; Farag, S.A.; Naas, H.T.; Garbaj, A.M.; El Salabi, A.A.; Gammoudi, F.T.; Eldaghayes, I.M. Antimicrobial susceptibility profile of Klebsiella pneumoniae isolated from some dairy products in Libya as a foodborne pathogen. Vet. World 2024, 17, 1168–1176. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, S.; Tiwari, N.; Dutta, S. Study on the antibacterial and antioxidant activities of Punica granatum (pomegranate) peel extracts. J. Integr. Sci. Technol. 2025, 13, 1126. [Google Scholar] [CrossRef]
- Feudjio, C.; Yameen, M.A.; Njateng, G.S.S.; Khan, M.A.; Tamekou, S.L.; Mpetga, J.D.S.; Kuiate, J.R. The influence of solvent, host and phenological stage on the yield, chemical composition, and antidiabetic and antioxidant properties of Phragmanthera capitata (Spengel) S. Balle. Evid.-Based Complement. Altern. Med. 2020, 2020, 1–16. [Google Scholar] [CrossRef]
- Garbaj, A.M.; Awad, E.M.; Azwai, S.M.; Abolghait, S.K.; Naas, H.T.; Moawad, A.A.; Gammoudi, F.T.; Barbieri, I.; Eldaghayes, I.M. Enterohemorrhagic Escherichia coli O157 in milk and dairy products from Libya: Isolation and molecular identification by partial sequencing of 16S rDNA. Vet. World 2016, 9, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, A.; Saleem, M.; Riaz, A. Determination of antibacterial and antioxidant potential of organic crude extracts from Malus domestica, Cinnamomum verum and Trachyspermum ammi. Sci. Rep. 2025, 15, 976. [Google Scholar] [CrossRef] [PubMed]
- Justice-Alucho, C.H.; Braide, W. Antimicrobial activity of Syzygium aromaticum (clove) against Staphylococcus aureus and Listeria monocytogenes is enhanced by optimized extraction methods. bioRxiv 2026. [Google Scholar] [CrossRef]
- Maniki, E.; Kostoglou, D.; Paterakis, N.; Nikolaou, A.; Kourkoutas, Y.; Papachristoforou, A.; Giaouris, E. Chemical composition, antioxidant, and antibiofilm properties of essential oil from Thymus capitatus plants organically cultured on the Greek island of Lemnos. Molecules 2023, 28, 1154. [Google Scholar] [CrossRef] [PubMed]
- Muhaisen, H.M.H.; Ab-Mous, M.M.; Ddeeb, F.A.; Rtemi, A.A.; Taba, O.M.; Parveen, M. Antimicrobial agents from selected medicinal plants in Libya. Chin. J. Integr. Med. 2016, 22, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Naas, H.T.; Edarhoby, R.A.; Garbaj, A.M.; Azwai, S.M.; Abolghait, S.K.; Gammoudi, F.T.; Moawad, A.A.; Barbieri, I.; Eldaghayes, I.M. Occurrence, characterization, and antibiogram of Staphylococcus aureus in meat, meat products and some seafood from Libyan retail markets. Vet. World 2019, 12, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Naas, H.T.; Zurghani, M.M.; Garbaj, A.M.; Azwai, S.M.; Eshamah, H.L.; Gammoudi, F.T.; Abolghait, S.K.; Moawad, A.A.; Barbieri, I.; Eldaghayes, I.M. Bacillus cereus as an emerging public health concern in Libya: Isolation and antibiogram from food of animal origin. Libyan J. Med. Sci. 2018, 2, 61–65. [Google Scholar]
- Pinto, L.; Ayala-Zavala, J.F. Application of plant antimicrobials in the food sector: Where do we stand? Foods 2024, 13, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Naveed, M.; Bibi, J.; Kamboh, A.A.; Arain, M.A.; Shah, Q.A.; Alagawany, M.; El-Hack, M.E.A.; Abdel-Latif, M.A.; Yatoo, M.I.; Tiwari, R.; Chakraborty, S.; Dhama, K. The promising pharmacological effects and therapeutic applications of Punica granatum L. (pomegranate). Recent Pat. Food Nutr. Agric. 2023, 12, 1–12. [Google Scholar] [CrossRef]
- Sherif, J.A.; Farag, S.A.A.; Abureema, S.F.; Azwai, S.M.; Garbaj, A.M.; Gammoudi, F.T.; El Salabi, A.A.; Eldaghayes, I.M. Emergence of extended-spectrum beta-lactamase producer and colistin-resistant E. coli in animal-origin foods in Libya. World Vet. J. 2025, 15, 597–611. [Google Scholar] [CrossRef]
- Tayeb, Y.A.; Al Sharif, H.A.; Mansour, S.; Ashour, A.S.; Tayeb, A.Y. Effect of aqueous and alcoholic extract of Capparis spinosa L. leaves on pathogenic bacteria and comparison with antibiotics. Glob. Libyan J. 2022, 57, 1–15. [Google Scholar]
| Plant name | Solvent | MIC (mg/ml) | ||||
|---|---|---|---|---|---|---|
| EC184 | EC56 | BC4 | KP243 | SA121 | ||
| Rosmariums officinalis L. (G) | P | - | - | - | - | - |
| E | 50 | ND | 50 | - | ND | |
| Cistus salvifolius L. (B) | M | ND | ND | 12.5 | ND | ND |
| E | ND | ND | 6.25 | ND | ND | |
| Majorana hortensis L. (E) | M | ND | ND | 25 | ND | ND |
| P | ND | ND | 25 | ND | ND | |
| Zizyphus vulgaris (H) | P | ND | ND | 50 | ND | ND |
| Thymus capitatus (I) | M | 3.125 | 1.5625 | 1.5625 | 1.5625 | 3.125 |
| P | 6.25 | 1.5625 | 1.5625 | 3.125 | 6.25 | |
| Punica granatum L. (L) | M | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 |
| E | 50 | 50 | 25 | 50 | 25 | |
| Myrtus communis (M) | M | 25 | 25 | 12.5 | 12.5 | ND |
| P | ND | ND | ND | 12.5 | ND | |
| E | ND | ND | 6.25 | 12.5 | ND | |
| Syzygium aromaticum L. (P) | M | 1.5625 | 1.5625 | 0.78125 | 3.125 | 3.125 |
| P | 1.5625 | 1.5625 | 0.78125 | 3.125 | 0.78125 | |
| E | ND | ND | 3.125 | 3.125 | ND | |
| Cinnamomum verum (R) | P | 0.78125 | 1.5625 | 0.78125 | 0.78125 | 0.78125 |
| E | 0.78125 | 0.78125 | 0.78125 | 0.78125 | 0.78125 | |
| Plant name | Solvent | MBC (mg/ml) | ||||
|---|---|---|---|---|---|---|
| EC184 | EC56 | BC4 | KP243 | SA121 | ||
| Rosmariums officinalis L. (G) | P | - | - | - | - | - |
| E | 100 | ND | 100 | - | ND | |
| Cistus salvifolius L. (B) | M | ND | ND | 25 | ND | ND |
| E | ND | ND | 12.5 | ND | ND | |
| Majorana hortensis L. (E) | M | ND | ND | 50 | ND | ND |
| P | ND | ND | 50 | ND | ND | |
| Zizyphus vulgaris (H) | P | ND | ND | 100 | ND | ND |
| Thymus capitatus (I) | M | 6.25 | 3.125 | 3.125 | 3.125 | 6.25 |
| P | 12.5 | 3.125 | 3.125 | 6.25 | 12.5 | |
| Punica granatum L. (L) | M | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 |
| E | 100 | 100 | 50 | 100 | 50 | |
| Myrtus communis (M) | M | 50 | 50 | 25 | 25 | ND |
| P | ND | ND | ND | 25 | ND | |
| E | ND | ND | 12.5 | 25 | ND | |
| Syzygium aromaticum L. (P) | M | 3.125 | 3.125 | 1.5625 | 6.25 | 6.25 |
| P | 3.125 | 3.125 | 1.5625 | 6.25 | 1.5625 | |
| E | ND | ND | 6.25 | 6.25 | ND | |
| Cinnamomum verum (R) | P | 1.5625 | 3.125 | 1.5625 | 1.5625 | 1.5625 |
| E | 1.5625 | 1.5625 | 1.5625 | 1.5625 | 1.5625 | |
| Code | Family | Plant Name | Common English Name |
|---|---|---|---|
| B | Cistaceae | Cistus salvifolius L. | Sage-leaved rockrose |
| E | Labiatae | Majorana hortensis L. | Sweet marjoram |
| F | Cupressineae | Juniperus communis | Common juniper |
| G | Lamiaceae | Rosmarinus officinalis L. | Rosemary |
| H | Rhamneae | Zizyphus vulgaris | Christ's thorn jujube |
| I | Lamiaceae | Thymus capitatus | Cone head thyme |
| J | Ranunculaceae | Nigella sativa L. | Black seed |
| K | Urticaceae | Urtica sp. | Italian Nettle |
| L | Punicaceae | Punica granatum L. | Pomegranate |
| M | Myrtaceae | Myrtus communis | Common myrtle |
| N | Apiaceae | Pituranthos tortuosus | Desert thyme |
| O | Zingiberaceae | Curcurma sp | Turmeric |
| P | Myrtaceae | Syzygium aromaticum L. | Clove |
| Q | Zingiberaceae | Zingiber officinale | Ginger |
| R | Lauraceae | Cinnamomum verum | Ceylon cinnamon |
| S | Malvaceae | Malva parviflora | Cheeseweed mallow |
| T | Urticaceae | Urtica dioica | Stinging nettle |
| U | Rutaceae | Citrus aurantium | Bitter orange |
| V | Caesalpinioideae | Ceratonia siliqua | Carob |
| X | Fabaceae | Retama raetam | White broom |
| Y | Asteraceae | Phagnalon rupestre L. | Rock phagnalon |
| Z | Rutaceae | Haplophyllum tuberculatum | Rue-leaved haplophyllum |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




