Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Research Objectives and Scope
2.1. Literature Search Strategy
2.2. Inclusion Criteria
2.3. Exclusion Criteria
3. Overview of Emodin’s Anti-Metastatic Properties
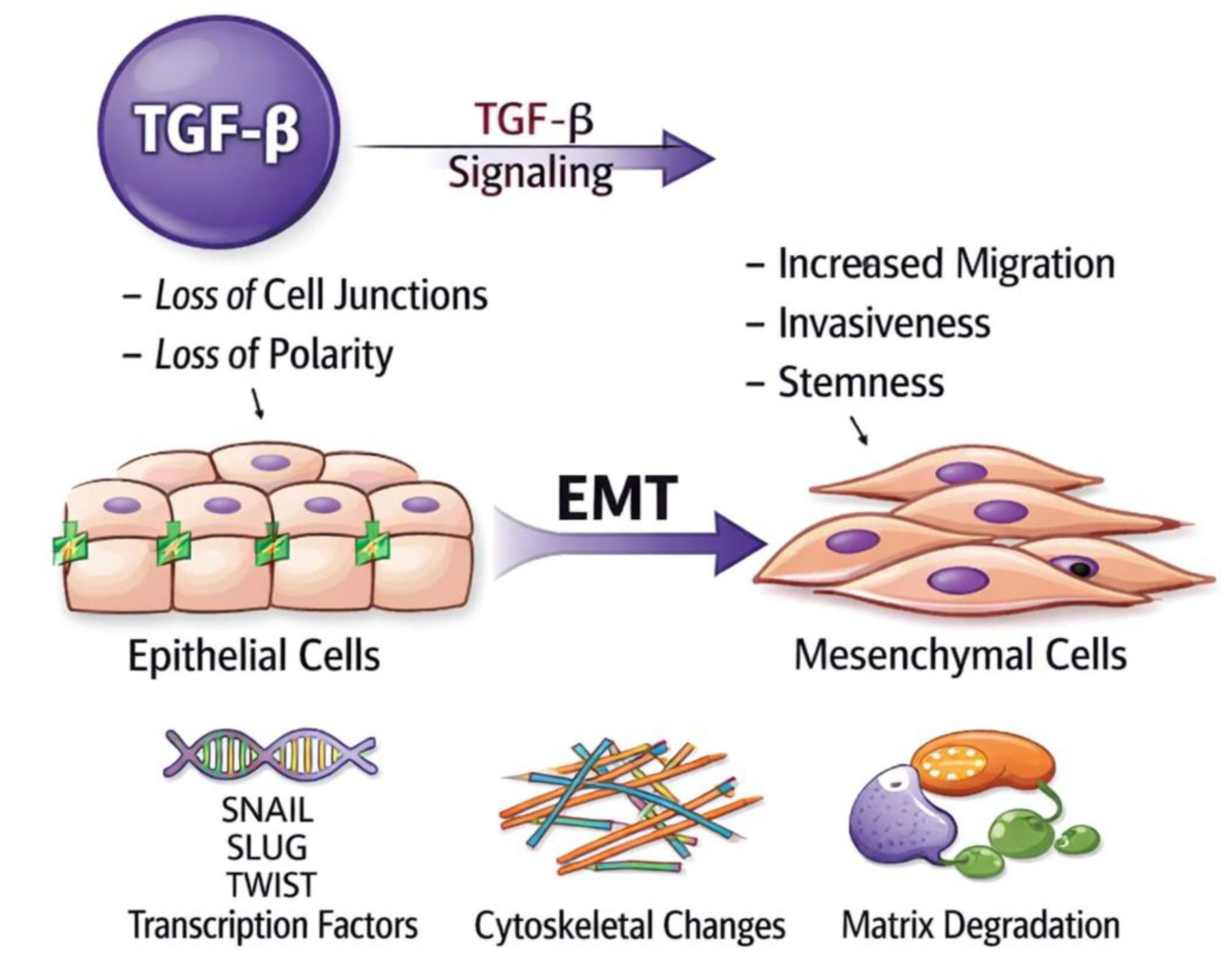
3.1. Mechanisms of Metastasis and EMT in Cancer
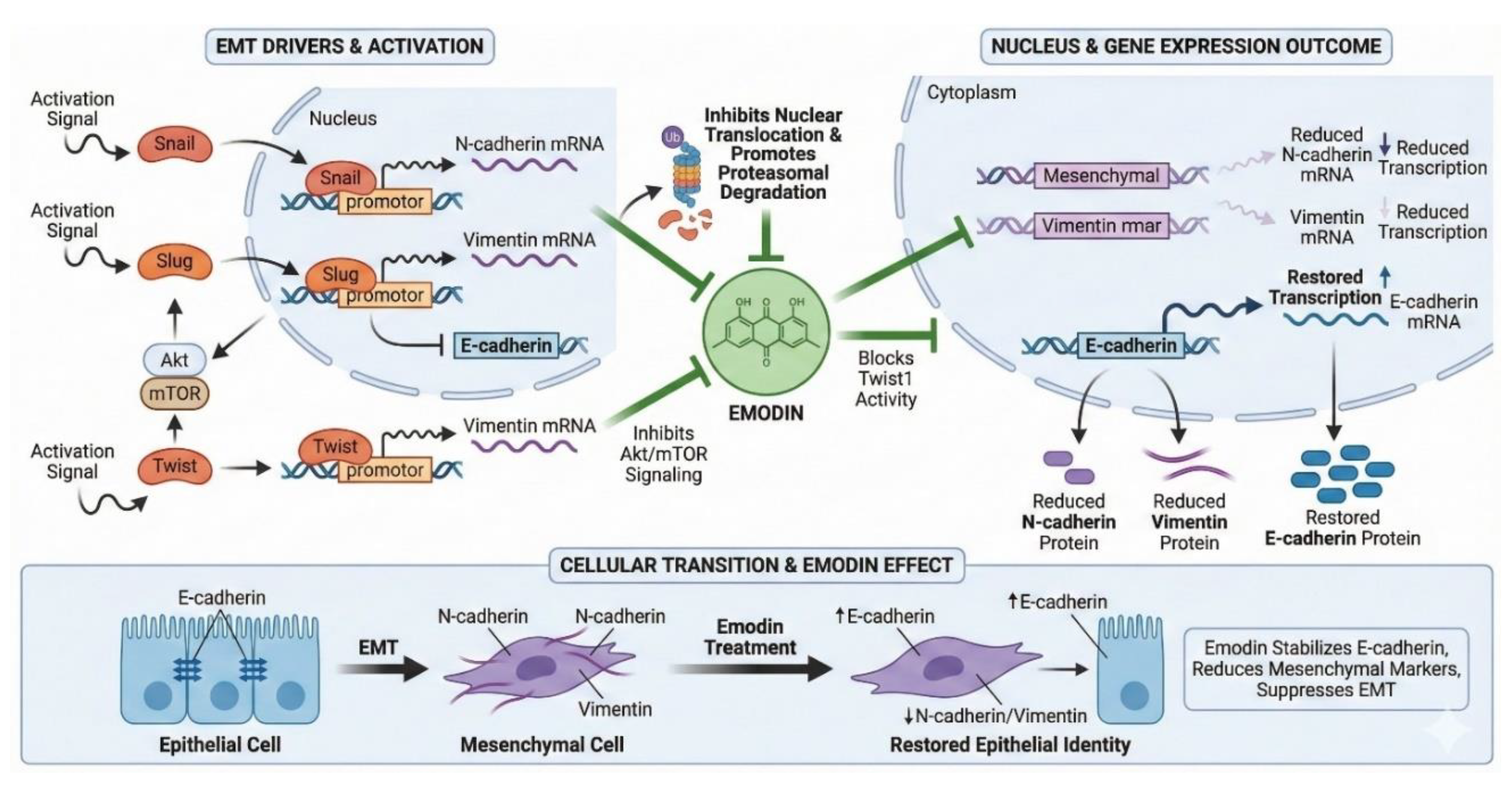
- E-cadherin to N-cadherin Switch: During EMT, E-cadherin levels decrease while N-cadherin levels increase. This change facilitates cell detachment and migration.
- Cytoskeletal Reorganization: EMT causes major changes in the cytoskeleton. Cells lose epithelial cytokeratin networks and gain mesenchymal vimentin filaments.
- Extracellular Matrix Remodeling: MMPs, especially MMP-2 and MMP-9, degrade basement membrane components, creating pathways for tumor cell invasion.
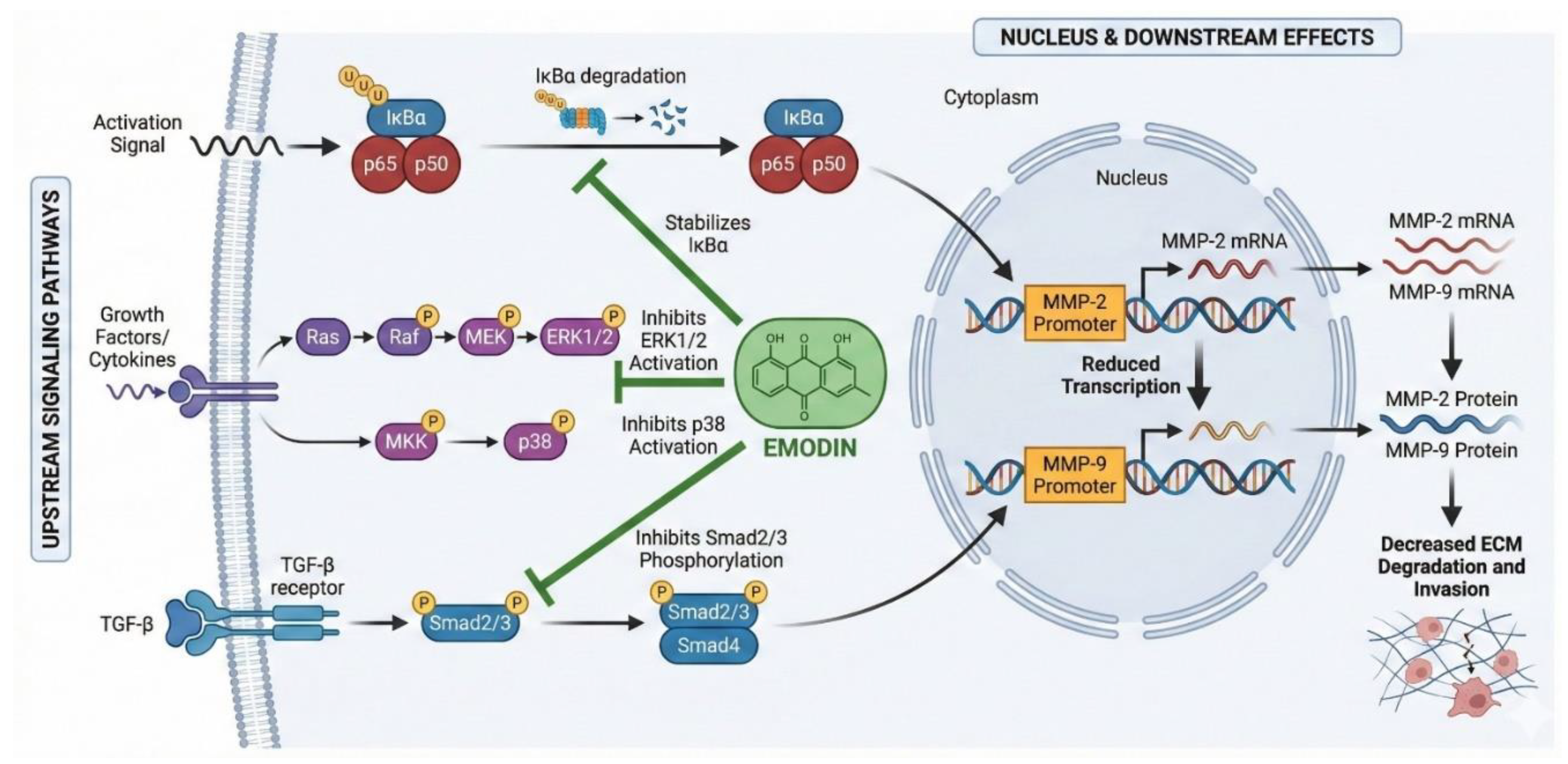
3.2. Emodin-Directed downregulation of MMP-2 and MMP-9
3.3. Cancer Type-Specific Effects
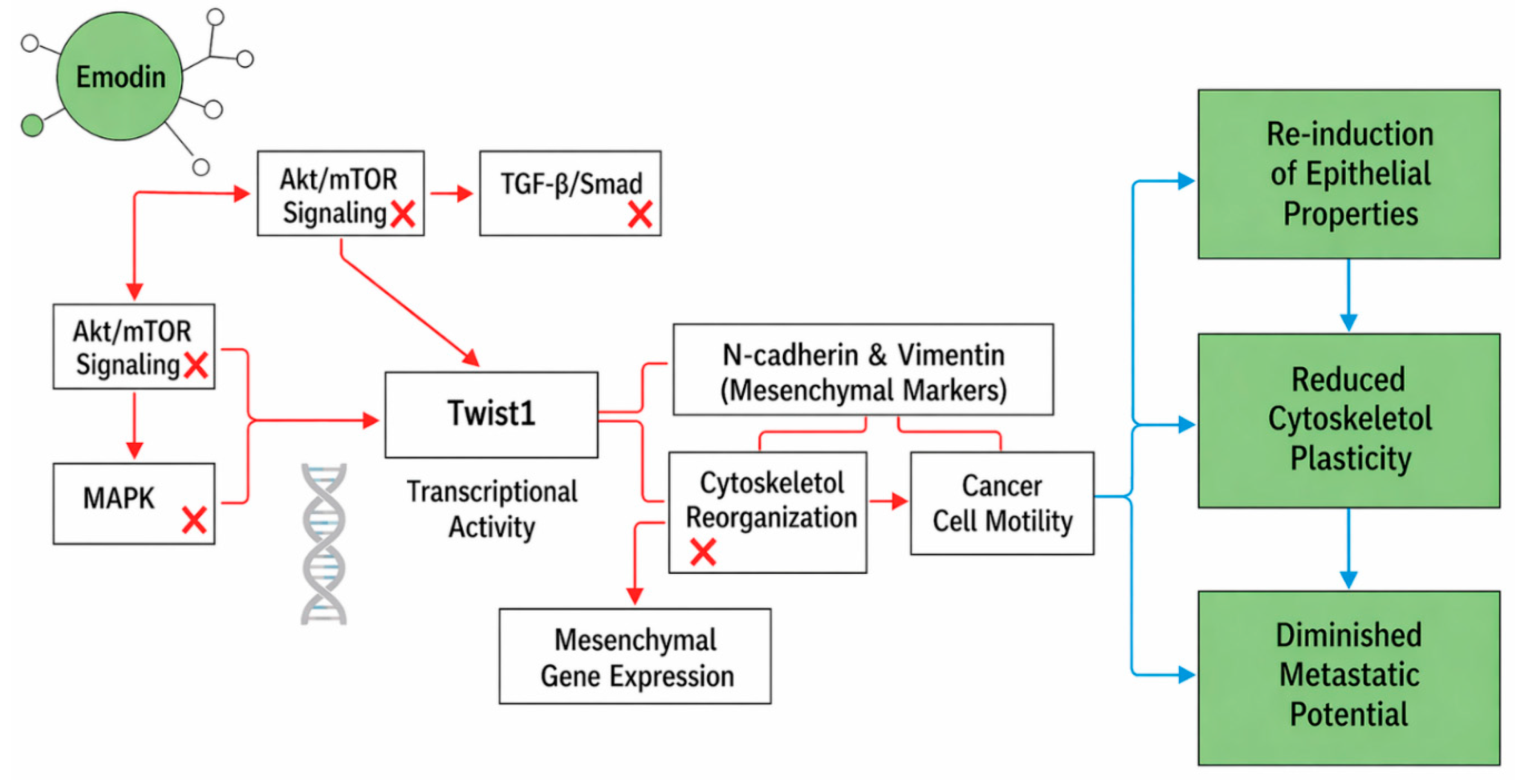
4. Emodin-mediated suppression of EMT by blocking Snail, Slug, and Twist
5. Transforming Growth Factor-Beta
| Mechanism | Molecular Target | Effect |
Cancer Type |
Key Findings | Clinical Relevance |
| Smad2/3 Phosphorylation Inhibition | TβRI kinase activity | Blocks nuclear translocation and EMT gene transcription | Breast Cancer (MDA-MB-231) | 65% reduction in Smad2/3 phosphorylation; restored E-cadherin expression | Potential biomarker for treatment response |
| Non-Smad PI3K/Akt Suppression | PI3K/Akt signaling | Decreases cell survival and motility | Hepatocellular Carcinoma (HepG2) | 70% reduction in Akt phosphorylation; enhanced apoptosis | Combination potential with PI3K inhibitors |
| Non-Smad MAPK/ERK Inhibition | ERK1/2 phosphorylation | Limits migratory and invasive capacity | Colorectal Cancer (SW480) | 60% decrease in ERK activation; reduced MMP-9 expression | Predictive biomarker for MAPK pathway dependency |
| MMP-2/MMP-9 Downregulation | MMP transcription | Reduces ECM degradation and invasion | Lung Cancer (A549) | 80% reduction in MMP activity; decreased invasion by 75% | Monitoring biomarker for therapeutic efficacy |
| E-cadherin Stabilization | Adherens junction integrity | Maintains cell-cell adhesion | Pancreatic Cancer (PANC-1) | 3-fold increase in E-cadherin; reduced cytoskeletal remodeling | Prognostic indicator for treatment response |
| CSC Property Suppression | CD44+/CD24- population | Reduces self-renewal and chemoresistance | Breast Cancer (MCF-7) | 50% decrease in CSC population; 2-fold enhanced doxorubicin sensitivity | Combination therapy with chemotherapy |
| Resistance Mechanism | Notch/Wnt pathway activation | Compensatory EMT induction | Glioblastoma (U87) | Partial EMT restoration via Notch signaling upregulation | Need for combination with pathway inhibitors |
6. Translational Evidence for Emodin in Tumor Progression and Metastasis
7. Clinical Applications and Future Prospects
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Castaneda, M., Den Hollander, P., Kuburich, N. A., Rosen, J. M., & Mani, S. A. (2022). Mechanisms of cancer metastasis. Seminars in Cancer Biology, 87, 17–31. [CrossRef]
- Ribatti, D., Tamma, R., & Annese, T. (2020). Epithelial-Mesenchymal Transition in Cancer: A Historical Overview. Translational Oncology, 13(6), 100773. [CrossRef]
- Serrano-Gomez, S. J., Maziveyi, M., & Alahari, S. K. (2016). Regulation of epithelial-mesenchymal transition through epigenetic and post-translational modifications. Molecular Cancer, 15(1), 18. [CrossRef]
- Tuli, H. S., Aggarwal, V., Tuorkey, M., Aggarwal, D., Parashar, N. C., Varol, M., Savla, R., Kaur, G., Mittal, S., & Sak, K. (2021). Emodin: A metabolite that exhibits anti-neoplastic activities by modulating multiple oncogenic targets. Toxicology in Vitro, 73, 105142. [CrossRef]
- Zhang, A., Miao, K., Sun, H., & Deng, C. X. (2022). Tumor heterogeneity reshapes the tumor microenvironment to influence drug resistance. International Journal of Biological Sciences, 18(7), 3019–3033. [CrossRef]
- Zhang, R., Jiao, Y., Wang, S., Wang, L., Leng, Y., Xiong, Z., & Yang, M. (2026). Mechanistic study of the role of emodin in targeting and inhibiting the Rap1 signaling pathway to regulate epithelial-mesenchymal transition and reverse cisplatin resistance in hepatocellular carcinoma. Transl Cancer Res, 15(2), 93. https://tcr.amegroups.org/article/view/112975.
- Ren, S., Song, L., Tian, Y., Zhu, L., Guo, K., Zhang, H., & Wang, Z. (2021). Emodin-Conjugated PEGylation of Fe3O4 Nanoparticles for FI/MRI Dual-Modal Imaging and Therapy in Pancreatic Cancer. Int J Nanomedicine, 16, 7463-7478. [CrossRef]
- Sherekar, P., Suke, S. G., Dani, R., Upadhyay, A., & Ahmad, S. (2024). Nanoformulation, Characterization, and In Vivo Pharmacokinetic Studies of Diosgenin- and Emodin-Loaded Polymeric Nanoparticles. BioNanoSci. 14, 164–174. [CrossRef]
- Huang, Y., Hong, W., & Wei, X. (2022). The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J Hematol Oncol, 15, 129. [CrossRef]
- Zhang, Y., & Weinberg R. A. (2018). Epithelial-to-mesenchymal transition in cancer: complexity and opportunities. Front Med, 12(4), 361–373. [CrossRef]
- Kuburich, N. A., Sabapathy, T., Demestichas, B. R., Maddela, J. J., Den Hollander, P., & Mani, S. A. (2023). Proactive and reactive roles of TGF-β in cancer. Seminars in Cancer Biology, 95, 120–139. [CrossRef]
- Gonzalez-Avila, G., Sommer, B., Mendoza-Posada, D. A., Ramos, C., Garcia-Hernandez, A. A., & Falfan-Valencia, R. (2019). Matrix metalloproteinases participation in the metastatic process and their diagnostic and therapeutic applications in cancer. Critical Reviews in Oncology/Hematology, 137, 57–83. [CrossRef]
- Liu, S., Li, L., & Ren, D. (2023). Anti-Cancer Potential of Phytochemicals: The Regulation of the Epithelial-Mesenchymal Transition. Molecules, 28(13), 5069. [CrossRef]
- Ghorbian, S. (2025). Cancer cell plasticity and therapeutic resistance: mechanisms, crosstalk, and translational perspectives. Hereditas, 162(1), 188. [CrossRef]
- Zheng, Q., Li, S., Li, X., & Liu, R. (2021). Advances in the study of emodin: an update on pharmacological properties and mechanistic basis. Chin Med, 16(1), 102. [CrossRef]
- Kessenbrock, K., Wang, C.-Y., & Werb, Z. (2015). Matrix metalloproteinases in stem cell regulation and cancer. Matrix Biology, 44(46), 184–190. [CrossRef]
- Taherkhani, S., Sheibani, M., Mohammadkhanizadeh, A., Virag, JAI., de Castro Braz L., & Azizi, Y. (2025). Metalloproteinases (MMPs) in hypertensive disorders: role, function, pharmacology, and potential strategies to mitigate pathophysiological changes. Front Pharmacol, 16, 1559288. [CrossRef]
- Shoari, A., Ashja Ardalan, A., Dimesa, A. M., & Coban, M. A. (2024). Targeting Invasion: The Role of MMP-2 and MMP-9 Inhibition in Colorectal Cancer Therapy. Biomolecules, 15(1), 35. [CrossRef]
- Baojiang Lv., Kenan Zheng., Yifan Sun., Lulu Wu., Lijun Qiao., Zhibing Wu., Yuanqi Zhao., & Zequan Zheng. (2022). Network Pharmacology Experiments Show That Emodin Can Exert a Protective Effect on MCAO Rats by Regulating Hif-1α/VEGF-A Signaling. ACS Omega, 7(26), 22577–22593. [CrossRef]
- Guo, Y., Pan, W., Liu, S., Shen, Z., Xu, Y., & Hu, L. (2020). ERK/MAPK signalling pathway and tumorigenesis (Review). Experimental and Therapeutic Medicine. [CrossRef]
- Liao, J., Chen, R., Lin, B., Deng, R., Liang, Y., Zeng, J., Ma, S., & Qiu, X. (2024). Cross-Talk between the TGF-β and Cell Adhesion Signaling Pathways in Cancer. International Journal of Medical Sciences, 21(7), 1307–1320. [CrossRef]
- Thacker, P. C., & Karunagaran, D. (2015). Curcumin and Emodin Down-Regulate TGF-β Signaling Pathway in Human Cervical Cancer Cells. PLOS ONE, 10(3), e0120045. [CrossRef]
- Luo, N., Fang, J., Wei, L., Sahebkar, A., Little, P. J., Xu, S., Luo, C., & Li, G. (2020). Emodin in atherosclerosis prevention: Pharmacological actions and therapeutic potential. European Journal of Pharmacology, 890, 173617. [CrossRef]
- Bharadwaj, A. G., Holloway, R. W., Miller, V. A., & Waisman, D. M. (2021). Plasmin and Plasminogen System in the Tumor Microenvironment: Implications for Cancer Diagnosis, Prognosis, and Therapy. Cancers, 13(8), 1838. [CrossRef]
- Manimaran, A., Manoharan, S., & Neelakandan, M. (2016). EMODIN EFFICACY ON THE AKT, MAPK, ERK AND DNMT EXPRESSION PATTERN DURING DMBA-INDUCED ORAL CARCINOMA IN GOLDEN SYRIAN HAMSTERS. Afr J Tradit Complement Altern Med, 13(6), 186–193. [CrossRef]
- Jia, X., Yu, F., Wang, J., Iwanowycz, S., Saaoud, F., Wang, Y., Hu, J., Wang, Q., & Fan, D. (2014). Emodin suppresses pulmonary metastasis of breast cancer accompanied with decreased macrophage recruitment and M2 polarization in the lungs. Breast Cancer Res Treat, 148(2), 291–302. [CrossRef]
- Sun, Y., Wang, X., Zhou, Q., Lu, Y., Zhang, H., Chen, Q., Zhao, M., & Su, S. (2015). Inhibitory effect of emodin on migration, invasion and metastasis of human breast cancer MDA-MB-231 cells in vitro and in vivo. Oncol Rep, 33(1), 338–46. [CrossRef]
- Yang, M., Xiong, Z., Deng, H., Chen, X., Lai, Q., Wang, H., & Leng, Y. (2023). Effect of emodin combined with cisplatin on the invasion and migration of HepG2 hepatoma cells. J Physiol Pharmacol, 74(4). https://pubmed.ncbi.nlm.nih.gov/37865957/.
- Wahi, D., Soni, D., & Grover, A. (2021). A Double-Edged Sword: The Anti-Cancer Effects of Emodin by Inhibiting the Redox-Protective Protein MTH1 and Augmenting ROS in NSCLC. Journal of Cancer, 12(3), 652–681. [CrossRef]
- Gordian, E., Welsh, E. A., Gimbrone, N., Siegel, E. M., Shibata, D., Creelan, B. C., Cress, W. D., Eschrich, S. A., Haura, E. B., & Muñoz-Antonia, T. (2021). Correction: Transforming growth factor β-induced epithelial-to-mesenchymal signature predicts metastasis-free survival in non-small cell lung cancer. Oncotarget, 12, 1325–1325. Retrieved from https://www.oncotarget.com/article/28004/.
- Hsu, H. C., Liu, L. C., Wang, H. Y., Hung, C. M., Lin, Y. C., Ho, C. T., & Way, T. D. (2017). Stromal Fibroblasts from the Interface Zone of Triple Negative Breast Carcinomas Induced Epithelial-Mesenchymal Transition and its Inhibition by Emodin. PLoS One, 12(1), e0164661. [CrossRef]
- Thakral, F., Prasad, B., Sehgal, R., Gupta, S., Sharma, U., Singh, B. J., Sharma, B., Tuli, H. S., Haque, S., & Ahmad, F. (2025). Role of emodin to prevent gastrointestinal cancers: recent trends and future prospective. Discov Oncol, 16(1), 468. [CrossRef]
- Wei, W., Lin, S., Liu, D., & Wang, Z. (2013). The distinct mechanisms of the antitumor activity of emodin in different types of cancer (Review). Oncology Reports, 30, 2555–2562. [CrossRef]
- Rijo, P., Athanassopoulos, C. M., & Carpinella, M. C. (2022). Editorial: Considering plant metabolites and their synthetic derivatives as candidates for the development of drugs against multidrug resistant (MDR) tumors. Front. Pharmacol, 13, 1108252. [CrossRef]
- Seo, J., Ha, J., Kang, E., & Cho, S. (2021). The role of epithelial–mesenchymal transition-regulating transcription factors in anti-cancer drug resistance. Archives of Pharmacal Research, 44(3), 281–292. [CrossRef]
- Lu, J., Xu, Y., Wei, X., Zhao, Z., Xue, J., & Liu, P. (2016). Emodin Inhibits the Epithelial to Mesenchymal Transition of Epithelial Ovarian Cancer Cells ILK/GSK-3 β /Slug Signaling Pathway. BioMed Research International, 2016, 6253280. [CrossRef]
- Loh, C.-Y., Chai, J. Y., Tang, T. F., Wong, W. F., Sethi, G., Shanmugam, M. K., Chong, P. P., & Looi, C. Y. (2019). The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells, 8(10), 1118. [CrossRef]
- Qin, B., Zeng, Z., Xu, J., Shangwen, J., Ye, Z. J., Wang, S., Wu, Y., Peng, G., Wang, Q., Gu, W., & Tang, Y. (2022). Emodin inhibits invasion and migration of hepatocellular carcinoma cells via regulating autophagy-mediated degradation of snail and β-catenin. BMC Cancer, 22(1), 671. [CrossRef]
- Hu, C., Dong, T., Li, R., Lu, J., Wei, X., & Liu, P. (2016). Emodin inhibits epithelial to mesenchymal transition in epithelial ovarian cancer cells by regulation of GSK-3β/β-catenin/ZEB1 signaling pathway. Oncology Reports, 35, 2027–2034. [CrossRef]
- Ponnusamy, L., Kothandan, G., & Manoharan, R. (2020). Berberine and Emodin abrogates breast cancer growth and facilitates apoptosis through inactivation of SIK3-induced mTOR and Akt signaling pathway. Biochim Biophys Acta Mol Basis Dis, 1866(11), 165897. [CrossRef]
- Zhang, J., Hu, Z., Horta, C. A., & Yang, J. (2023). Regulation of epithelial-mesenchymal transition by tumor microenvironmental signals and its implication in cancer therapeutics. Seminars in Cancer Biology, 88, 46–66. [CrossRef]
- Qin, M. Y., Huang, S. Q., Zou, X. Q., Zhong, X. B., Yang, Y. F., Zhang, Y. T., Mi, Z. C., Zhang, Y. S., & Huang, Z. G. (2021). Drug-containing serum of rhubarb-astragalus capsule inhibits the epithelial-mesenchymal transformation of HK-2 by downregulating TGF-β1/p38MAPK/Smad2/3 pathway. J Ethnopharmacol, 280, 114414. [CrossRef]
- Jiang, J., Zhou, N., Ying, P., Zhang, T., Liang, R., & Jiang, X. (2019). Emodin Promotes Apoptosis of Human Endometrial Cancer Through Regulating the MAPK and PI3K/ AKT Pathways. Open Life Sci, 13, 489–496. [CrossRef]
- Peng, S., Wang, J., Lu, C., Xu, Z., Chai, J., Ke, Q., & Deng, X. (2021). Emodin enhances cisplatin sensitivity in non-small cell lung cancer through Pgp downregulation. Oncology Letters, 21, 230. [CrossRef]
- Bhattacharjee, M., Upadhyay, P., Sarker, S., Basu, A., Das, S., Ghosh, A., Ghosh, S., & Adhikary, A. (2020). Combinatorial therapy of Thymoquinone and Emodin synergistically enhances apoptosis, attenuates cell migration and reduces stemness efficiently in breast cancer. Biochim Biophys Acta Gen Subj, 1864(11), 129695. [CrossRef]
- Carver, W., Fix, E., Fix, C., Fan, D., Chakrabarti, M., & Azhar, M. (2021). Effects of emodin, a plant-derived anthraquinone, on TGF-β1-induced cardiac fibroblast activation and function. J Cell Physiol, 236(11), 7440–7449. [CrossRef]
- Lin, W., Zhong, M., Liang, S., Chen, Y., Liu, D., Yin, Z., Cao, Q., Wang, C., & Ling, C. (2016). Emodin inhibits migration and invasion of MHCC-97H human hepatocellular carcinoma cells. Exp Ther Med, 12(5), 3369–3374. [CrossRef]
- Stompor-Gorący, M. (2021). The Health Benefits of Emodin, a Natural Anthraquinone Derived from Rhubarb-A Summary Update. Int J Mol Sci, 22(17), 9522. [CrossRef]
- Iwanowycz, S., Wang, J., Hodge, J., Wang, Y., Yu, F., Fan, D. (2016). Emodin Inhibits Breast Cancer Growth by Blocking the Tumor-Promoting Feedforward Loop between Cancer Cells and Macrophages. Mol Cancer Ther, 15(8), 1931–1942. [CrossRef]
- McDonald, S. J., VanderVeen, B. N., Velazquez, K. T., Enos, R. T., Fairman, C. M., Cardaci, T. D., Fan, D., & Murphy, E. A. (2022). Therapeutic Potential of Emodin for Gastrointestinal Cancers. Integrative Cancer Therapies, 21, 15347354211067469. [CrossRef]
- Trybus, W., Trybus, E., & Król, T. (2022). Emodin Sensitizes Cervical Cancer Cells to Vinblastine by Inducing Apoptosis and Mitotic Death. International Journal of Molecular Sciences, 23(15), 8510. [CrossRef]
- Dong, X., Fu, J., Yin, X., Cao, S., Li, X., Lin, L., Huyiligeqi, & Ni, J. (2016). Emodin: A Review of its Pharmacology, Toxicity and Pharmacokinetics. Phytotherapy Research, 30(8), 1207–1218. [CrossRef]
- Brown, N. F., & Marshall, J. F. (2019). Integrin-Mediated TGFβ Activation Modulates the Tumour Microenvironment. Cancers, 11(9), 1221. [CrossRef]
- Liu, J., Vaziri, D. N., Miao, H., & Zhao, Y. (2025). The Renoprotective Effect, Nephrotoxicity, and Molecular Mechanisms of Emodin. Integrative Medicine in Nephrology and Andrology, 13, e25-00036. [CrossRef]
- Li, F., Song, X., Zhou, X., Chen, L., & Zheng, J. (2023). Emodin attenuates high lipid-induced liver metastasis through the AKT and ERK pathways in vitro in breast cancer cells and in a mouse xenograft model. Heliyon; 9(6), e17052. [CrossRef]
- Liu, Q., Hodge, J., Wang, J., Wang, Y., Wang, L., Singh, U., Li, Y., Yao, Y., Wang, D., Ai, W., Nagarkatti, P., Chen, H., Xu, P., Murphy, E. A., & Fan, D. (2020). Emodin reduces Breast Cancer Lung Metastasis by suppressing Macrophage-induced Breast Cancer Cell Epithelial-mesenchymal transition and Cancer Stem Cell formation. Theranostics, 10(18), 8365–8381. [CrossRef]
- de Streel, G., & Lucas, S. (2021). Targeting immunosuppression by TGF-β1 for cancer immunotherapy. Biochem Pharmacol, 192, 114697. [CrossRef]
- Lin, S. Z., Xu, J. B., Ji, X., Chen, H., Xu, H. T., Hu, P., Chen, L., Guo, J. Q., Chen, M. Y., Lu, D., Wang, Z. H., & Tong, H. F. (2015). Emodin inhibits angiogenesis in pancreatic cancer by regulating the transforming growth factor-β/drosophila mothers against decapentaplegic pathway and angiogenesis-associated microRNAs. Mol Med Rep, 12(4), 5865–5871. [CrossRef]
- Liu, C., Li, Z., Wu, D., Li, C., & Zhang, Y. (2016). Smad3 and phospho-Smad3 are potential markers of invasive nonfunctioning pituitary adenomas. Onco Targets Ther, 9, 2265–2271. [CrossRef]
- Long, H., Chen, H., Yan, J., & Cheng, H. (2022). Emodin exerts antitumor effects in ovarian cancer cell lines by preventing the development of cancer stem cells via epithelial mesenchymal transition. Oncol Lett, 23(3), 95. [CrossRef]
- Chen, R., Zhang, H., Zhao, X., Zhu, L., Zhang, X., Ma, Y., & Xia, L. (2024). Progress on the mechanism of action of emodin against breast cancer cells. Heliyon; 10(21), e38628. [CrossRef]
- Ma, L., Li, H., Zhang, S., Xiong, X., Chen, K., Jiang, P., Jiang, K., & Deng, G. (2018). Emodin ameliorates renal fibrosis in rats via TGF-β1/Smad signaling pathway and function study of Smurf 2. Int Urol Nephrol, 50, 373–382. [CrossRef]
- Zhang, Q., Chen, W. W., Sun, X., Qian, D., Tang, D. D., Zhang, L. L., Li, M. Y., Wang, L.Y., Wu, C. J., & Peng, W. (2022). The versatile emodin: A natural easily acquired anthraquinone possesses promising anticancer properties against a variety of cancers. Int J Biol Sci, 18(8), 3498–3527. https://www.ijbs.com/v18p3498.htm.
- Zheng, Q., Xu, Y., Lu, J., Zhao, J., Wei, X., & Liu, P. (2016). Emodin inhibits migration and invasion of human endometrial stromal cells by facilitating the mesenchymal-epithelial transition through targeting ILK. Reprod Sci, 23(11), 1526–35. [CrossRef]
- Wei, W., Tang, J., Hu, L., Feng, Y., Li, H., Yin, C., & Tang, F. (2021). Experimental anti-tumor effect of emodin in suspension – in situ hydrogels formed with self-assembling peptide. Drug Delivery, 28(1), 1810–1821. [CrossRef]
- Zou, G., Zhang, X., Wang, L., Li, X., Xie, T., Zhao, J., Yan, J., Wang, L., Ye, H., Jiao, S., Xiang, R., & Shi, Y. (2020). Herb-sourced emodin inhibits angiogenesis of breast cancer by targeting VEGFA transcription. Theranostics, 10(15), 6839–6853. https://www.thno.org/v10p6839.htm.
- Akkol, E. K., Tatlı, I. I., Karatoprak, G. Ş., Ağar, O. T., Yücel, Ç., Sobarzo-Sánchez, E., & Capasso, R. (2021). Is Emodin with Anticancer Effects Completely Innocent? Two Sides of the Coin. Cancers, 13(11), 2733. [CrossRef]
- Zhao, X., Ma, Y., Luo, J., Xu, K., Tian, P., Lu, C., & Song, J. (2024). Blocking the WNT/β-catenin pathway in cancer treatment:pharmacological targets and drug therapeutic potential. Heliyon, 10(16), e35989. [CrossRef]
- Liu, J., Xiao, S., Huang, C., Zhang, X., Cai, R., Qu, S., Peng, Y., & Xie, L. (2025). Emodin nanoparticles-loaded injectable hydrogel with deep tumor penetration for triple-negative breast cancer therapy. Biomater Adv, 177, 214380. [CrossRef]
- Lv, Y., Li, W., Liao, W., Jiang, H., Liu, Y., Cao, J., Lu, W., & Feng, Y. (2024). Nano-Drug Delivery Systems Based on Natural Products. International Journal of Nanomedicine, 19, 541–569. [CrossRef]
- Ai, Z., Liu, B., Chen, J., Zeng, X., Wang, K., Tao, C., Chen, J., Yang, L., Ding, Q., & Zhou, M. (2024). Advances in nano drug delivery systems for enhanced efficacy of emodin in cancer therapy. International Journal of Pharmaceutics: X, 9, 100314. [CrossRef]
- Tang, C., Zou, Y., Meng, H., Xu, J., Long, F., Chen, X., Xiao, Q., Su, K., Nan, Z., Guo, Y., & Li, C. (2025). Synergistic anticancer effects of carrier-free self-assembled doxorubicin and emodin nanoparticles in non-small cell lung cancer. Sci Rep; 15(1), 40205. [CrossRef]
- Lande, P. S., Adhao, V. S., Ambhore, J. P., Gaikwad, K. P., Chandak, C. S., & Joge, L. P. (2023). Anticancer action of naturally occurring emodin for the controlling of cervical cancer. Explor Target Antitumor Ther, 4, 690–8. [CrossRef]
- Wu, C.-C., Chen, M.-S., & Chen, J.-Y. (2024). The Application of Emodin Treatment on Nasopharyngeal Carcinoma Therapy. Biomedicines, 12(3), 486. [CrossRef]
- Hwang, S. Y., Heo, K., Kim, J. S., Im, J. W., Lee, S. M., Cho, M., Kang, D. H., Heo, J., Lee, J. W., Choi, C. W., & Yang, K. (2015). Emodin attenuates radioresistance induced by hypoxia in HepG2 cells via the enhancement of PARP1 cleavage and inhibition of JMJD2B. Oncology Reports, 33, 1691-1698. [CrossRef]
- Sougiannis, A. T., Enos, R. T., VanderVeen, B. N., Velazquez, K. T, Kelly, B., McDonald, S., Cotham, W., Chatzistamou, I., Nagarkatti, M., Fan, D., & Murphy, E. A. (2021). Safety of natural anthraquinone emodin: an assessment in mice. BMC Pharmacol Toxicol, 22(1), 9. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




