Submitted:
22 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Isolation and Purification of Endophytic Bacteria
2.3. Screening of Antagonistic Bacteria
2.3.1. Primary Screening
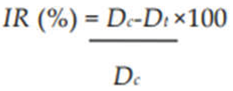
2.3.2. Secondary Screening
2.4. Morphological, Physiological, and Biochemical Identification
2.5. Molecular Identification
2.6. Antifungal Activity of Bacterial Volatile Organic Compounds (VOCs)
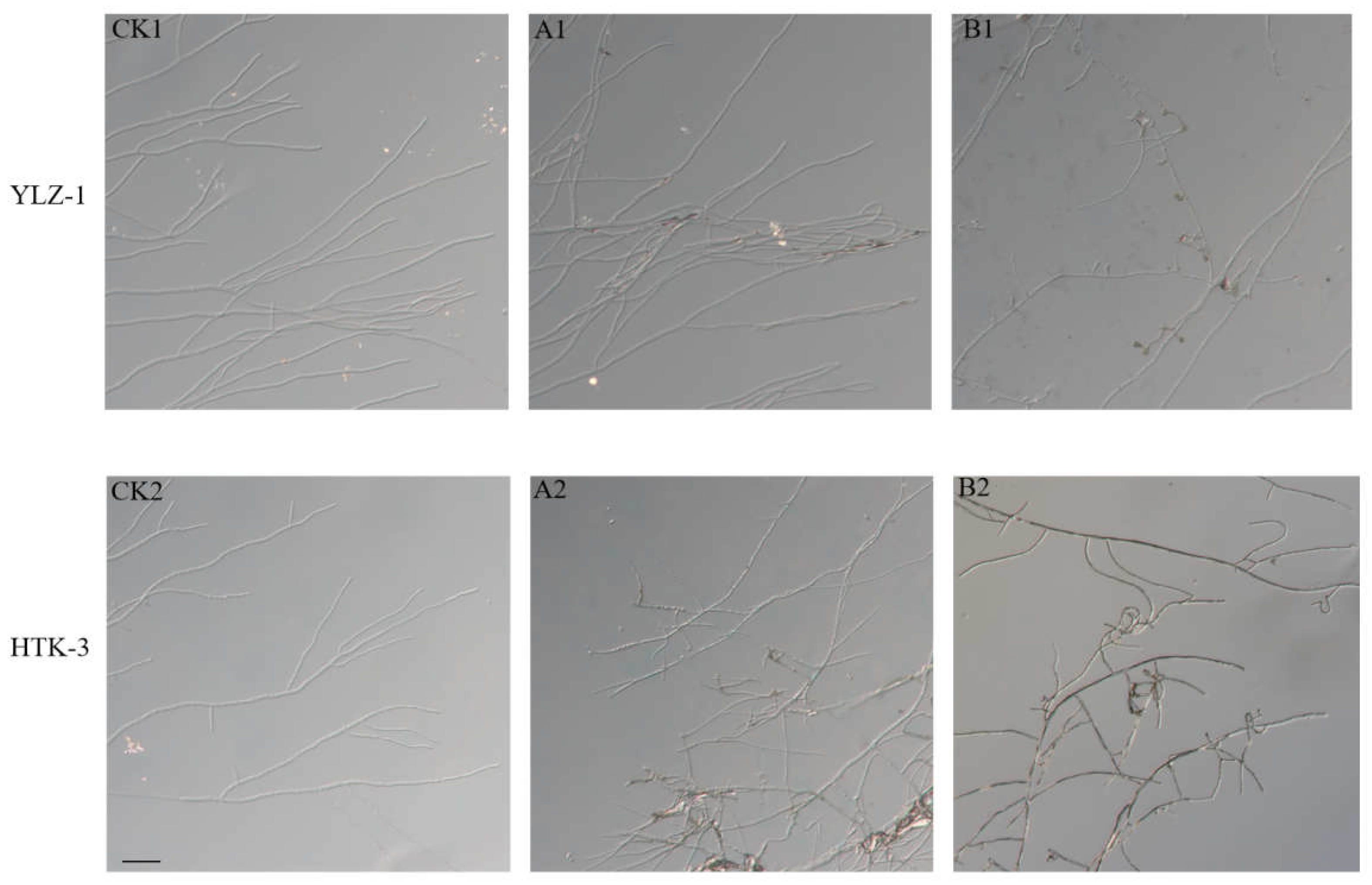
2.7. Effect of the Antagonistic Bacterium on Pathogen Mycelial Growth
2.8. Determination of Extracellular Enzyme Activities and IAA Production
2.9. Determination of the Antifungal Spectrum
2.10. Extraction and Activity Assay of Secondary Metabolites
2.11. GC-MS Identification of Major Components
2.12. In Vitro Toxicity Assay of Core Single Compounds
2.13. In Vivo Biocontrol Efficacy on Potted R. chinensis Seedlings
2.14. Statistical Analysis
3. Results
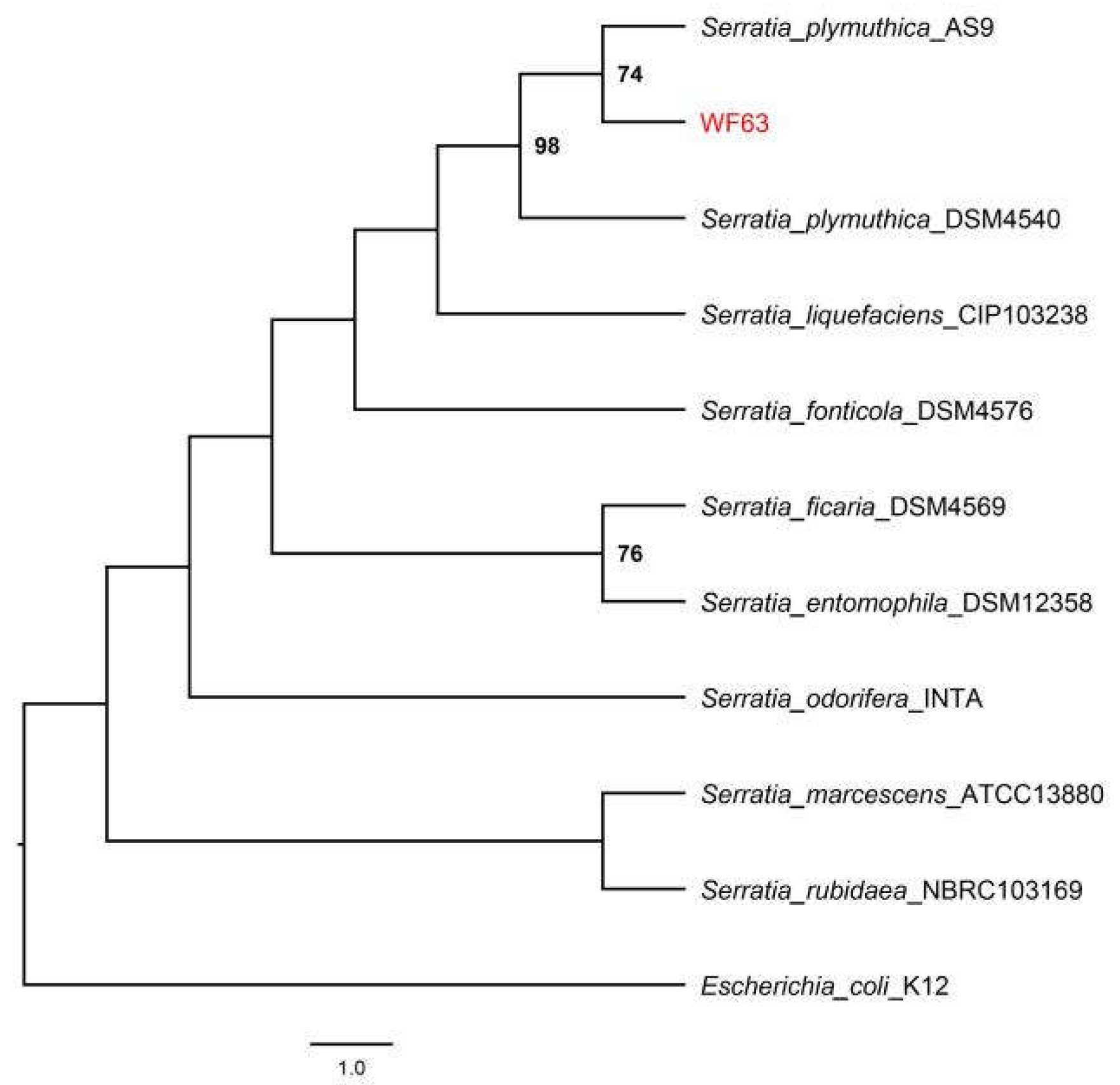
3.1. Isolation and Identification of Endophytic Antagonistic Bacteria
3.2. Inhibitory Activity of Volatile Organic Compounds (VOCs) from Strain WF63
3.3. Inhibitory Activity of the Cell-Free Fermentation Broth Against Target Pathogens
3.4. Determination of Extracellular Enzyme Activities and IAA Production
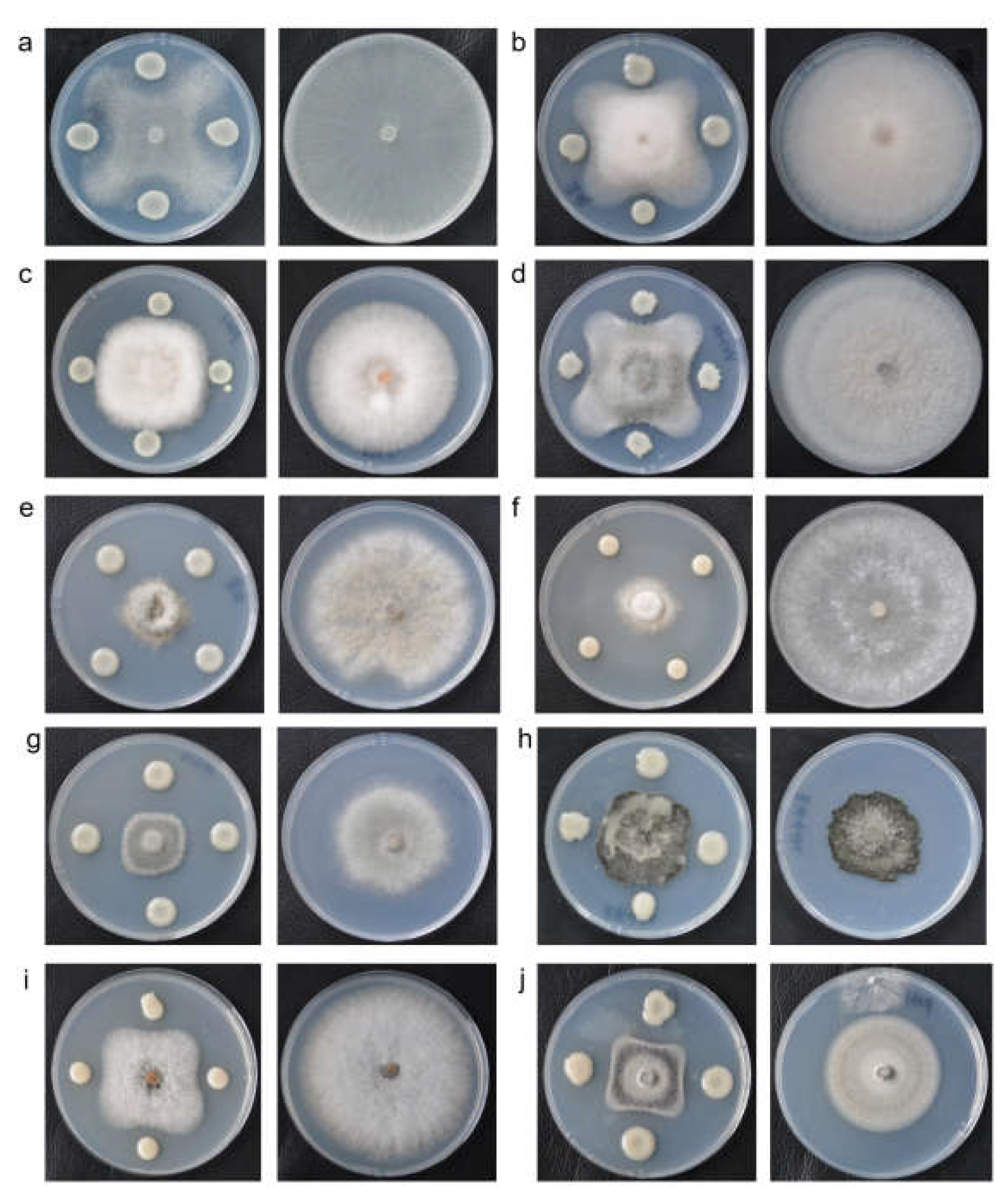
3.5. Inhibitory Effects Against Ten Phytopathogenic Fungi
3.6. Identification of Antifungal Active Substances from Strain WF63
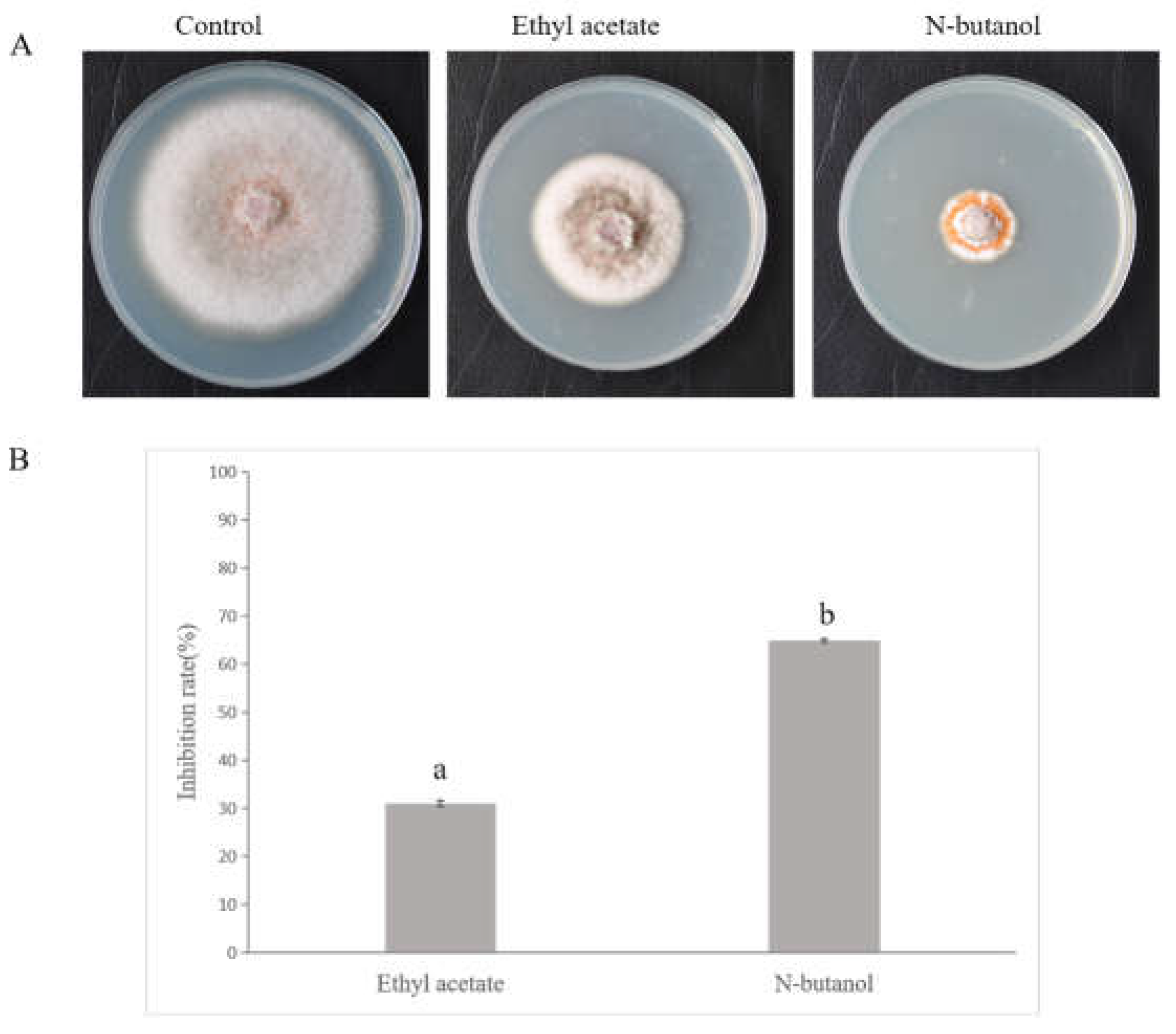
3.7. In Vitro Toxicity Assay of Major Fermentation Components
3.8. In Vivo Biocontrol Efficacy Against Gallnut Anthracnose
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Djakpo, O.; Yao, W. Rhus chinensis and Galla Chinensis—folklore to modern evidence: review. Phytother. Res. 2010, 24, 1739–1747. [Google Scholar] [CrossRef]
- Ma, W.; Wu, Y.; Wu, M.; Ren, Z. Cloning characterization and expression of chalcone synthase from medicinal plant Rhus chinensis. J. Plant Biochem. Biotechnol. 2015, 24, 18–24. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, O.; Mi, H.; Yi, J.; Cai, S. Rhus chinensis Mill. fruits prevent necrotizing enterocolitis in rat pups via regulating the expressions of key proteins involved in multiple signaling pathways. J. Ethnopharmacol. 2022, 290, 115103. [Google Scholar] [CrossRef]
- Shin, S.; Park, J.; Choi, H.Y.; Bu, Y.; Lee, K. Blood-pressure-lowering and endothelium-dependent vasorelaxant effects of nutgall tree in rats. Foods 2024, 13, 1041. [Google Scholar] [CrossRef]
- Xu, X.W.; Li, K.; Zhang, Z.Y.; Wang, Y. X.; Huang, F. X.; Li, J. Y.; Zha, Y. P. Identification of pathogen causing Rhus chinensis anthracnose. Acta Phytopathol. Sin. (In Chinese) 2024, 54, 664–668. [Google Scholar]
- Xu, X.W.; Zhang, Z.Y.; Wang, Y.X.; Li, J. Y.; Zha, Y. P. First report of colletotrichum fioriniae causing anthracnose disease of Rhus chinensis in China. Plant Dis. 2023, 107, 4034. [Google Scholar] [CrossRef]
- Ali, M.A.; Ahmed, T.; Ibrahim, E.; Rizwan, M.; Chong, K.P.; Yong, J.W.H. A review on mechanisms and prospects of endophytic bacteria in biocontrol of plant pathogenic fungi and their plant growth-promoting activities. Heliyon 2024, 10, e31573. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, J.; Tong, Y.; Luan, Z.; Hou, J.; Luan, F. Genomic insights into combating anthracnose with an endophytic Bacillus amyloliquefaciens Strain. Plant Dis. 2024, 108, 1976–1981. [Google Scholar] [CrossRef]
- Wu, Y.; Tan, Y.; Peng, Q.; Xiao, Y.; Xie, J.; Li, Z.; Ding, H.; Pan, H.; Wei, L. Biocontrol potential of endophytic bacterium Bacillus altitudinis GS-16 against tea anthracnose caused by Colletotrichum gloeosporioides. PeerJ 2024, 12, e16761. [Google Scholar] [CrossRef]
- Kim, J.D.; Jeon, B.J.; Han, J.W.; Park, M.Y.; Kang, S.A.; Kim, B.S. Evaluation of the endophytic nature of Bacillus amyloliquefaciens strain GYL4 and its efficacy in the control of anthracnose. Pest Manag. Sci. 2016, 72, 1529–1536. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, M.; Tang, L.; Huang, S.; Guo, T.; Li, Q. Screening and characterization of biocontrol bacteria isolated from Ageratum conyzoides against Collectotrichum fructicola causing Chinese plum (Prunus salicina Lindl.) anthracnose. Front. Microbiol. 2023, 14, 1296755. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.; Cottet, L.; Santos, C.; Castillo, A. Antifungal activity of Serratia plymuthica against the phytopathogenic fungus Alternaria tenuissima. Microb. Pathog. 2024, 193, 106750. [Google Scholar] [CrossRef] [PubMed]
- Kamensky, M.; Ovadis, M.; Chet, I.; Chernin, L. Soil-borne strain IC14 of Serratia plymuthica with multiple mechanisms of antifungal activity provides biocontrol of Botrytis cinerea and Sclerotinia sclerotiorum diseases. Soil Biol. Biochem. 2003, 35, 323–331. [Google Scholar] [CrossRef]
- Levenfors, J.J.; Hedman, R.; Thaning, C.; Gerhardson, B.; Welch, C. Broad-spectrum antifungal metabolites produced by the soil bacterium Serratia plymuthica A 153. Soil Biol. Biochem. 2004, 36, 677–685. [Google Scholar] [CrossRef]
- Neupane, S.; Finlay, R.D.; Alström, S.; Elfstrand, M.; Hogberg, N. Transcriptional responses of the bacterial antagonist Serratia plymuthica to the fungal phytopathogen Rhizoctonia solani. Environ. Microbiol. Rep. 2015, 7, 123–127. [Google Scholar] [CrossRef]
- Wang, Y.; Piao, F.; Di, Y.; Xu, J.; Wang, Z.; Wang, T.; Li, P.; Hu, C.; Du, N.; Zhang, T.; Dong, X.; Dong, H.; Shen, S.; Guo, Z. Serratia plymuthica HK9-3 enhances tomato resistance against Phytophthora capsici by modulating antioxidant defense systems and rhizosphere micro-ecological condition. Physiol. Plant. 2024, 176, e14323. [Google Scholar] [CrossRef]
- Aisyah, S.N.; Harnas, H.; Sulastri, S.; Retmi, R.; Fuaddi, H.; Fatchiyah, F.; Bakhtiar, A.; Jamsari, J. Enhancement of a novel isolate of Serratia plymuthica as potential candidate for an antianthracnose. Pak. J. Biol. Sci. 2016, 19, 250–258. [Google Scholar] [CrossRef]
- Dong, X.Z.; Cai, M.Y. Common manual for systematic identification of bacteria; (In Chinese). Science Press: Beijing, China, 2001; pp. 62–65. [Google Scholar]
- Holt, J.G.; Krieg, N.R.; Sneath, P.H.A.; Staley, J.T.; Williams, S.T. Bergey’s manual of determinative bacteriology, 9th ed.; Williams and Wilkins Press: Baltimore, MD, USA, 1994; pp. 195–256. [Google Scholar]
- Li, Z.; Ma, J.; Li, J.; Chen, Y.; Xie, Z.; Tian, Y.; Su, X.; Tian, T.; Shen, T. A biocontrol strain of Serratia plymuthica MM promotes growth and controls fusarium wilt in watermelon. Agronomy 2023, 13, 2437. [Google Scholar] [CrossRef]
- Abdelkhalek, A.; Behiry, S.I.; Al-Askar, A.A. Bacillus velezensis PEA1 inhibits Fusarium oxysporum growth and induces systemic resistance to cucumber mosaic virus. Agronomy 2020, 10, 1312. [Google Scholar] [CrossRef]
- Benhamou, N.; Gagné, S.; Le Quéré, D.; Dehbi, L. Bacterial-mediated induced resistance in cucumber: beneficial effect of the endophytic bacterium Serratia plymuthica on the protection against infection by Pythium ultimum. Phytopathology 2000, 90, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Meziane, H.; Gavriel, S.; Ismailov, Z.; Chet, I.; Chernin, L.; Hofte, M. Control of green and blue mould on orange fruit by Serratia plymuthica strains IC14 and IC1270 and putative modes of action. Postharvest Biol. Technol. 2006, 39, 125–133. [Google Scholar] [CrossRef]
- Hadizadeh, I.; Peivastegan, B.; Hannukkala, A.; Wolf, J.; Nissinen, R.; Pirhonen, M. Biological control of potato soft rot caused by Dickeya solani and the survival of bacterial antagonists under cold storage conditions. Plant Pathol. 2018, 68, 297–311. [Google Scholar] [CrossRef]
- Yan, H.; Li, H.L.; Lang, B. Isolation and diversity analysis of endophytic bacteria in Ji Yan 9. Chin. Tob. Sci. (In Chinese) 2019, 40, 7. [Google Scholar]
- Saberi Riseh, R.; Vatankhah, M.; Hassanisaadi, M.; Barka, E.A. Unveiling the role of hydrolytic enzymes from soil biocontrol bacteria in sustainable phytopathogen management. Front. Biosci. (Landmark Ed.) 2024, 29, 105. [Google Scholar] [CrossRef]
- Ajuna, H.B.; Lim, H.I.; Moon, J.H.; Won, S.J.; Choub, V.; Choi, S.I.; Yun, J.Y.; Ahn, Y.S. The Prospect of hydrolytic enzymes from Bacillus species in the biological control of pests and diseases in forest and fruit tree production. Int. J. Mol. Sci. 2023, 24, 16889. [Google Scholar] [CrossRef]
- Ben Abdallah, D.; Frika-Gargouri, O.; Tounsi, S. Rizhospheric competence, plant growth promotion and biocontrol efficacy of Bacillus amyloliquefaciens subsp. plantarum strain 32a. Biol. Control 2018, 124, 61–67. [Google Scholar] [CrossRef]
- Wu, R.; Yuan, M.J.; Kang, X.L. Screening, identification and antifungal activity of endophytic antagonistic bacteria against rose anthracnose. J. South. Agric. (In Chinese) 2024, 55, 1046–1059. [Google Scholar]
- Yuan, S.; Wu, Y.; Jin, J.; Tong, S.; Zhang, L.; Cai, Y. Biocontrol Capabilities of Bacillus subtilis E11 against Aspergillus flavus In Vitro and for Dried Red Chili (Capsicum annuum L.). Toxins 2023, 15, 308. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Liang, X.; Li, H.; Mo, L.; Mo, R.; Chen, W.; Wei, Y.; Wang, T.; Jiang, W. Biocontrol ability of Bacillus velezensis T9 against Apiospora arundinis causing Apiospora mold on sugarcane. Front. Microbiol. 2023, 14, 1314887. [Google Scholar] [CrossRef] [PubMed]
- Lobo, L.; Silva, M.; Castellane, T.; Carvalho, R.; Rigobelo, E. Effect of indole-3-acetic acid on tomato plant growth. Microorganisms 2022, 10, 2212. [Google Scholar] [CrossRef] [PubMed]
- Ek-Ramos, M.J.; Fraire-Velázquez, S. Editorial: Antimicrobial secondary metabolites: sources and opportunities to devise biocontrol strategies to counteract phytopathogens. Front. Plant Sci. 2024, 15, 1382604. [Google Scholar] [CrossRef]
- John, J.; Mallikarjunaswamy, G.E.; Noshad, N. Probiotic rhizospheric Bacillus sp. from Zingiber officinale Rosc. displays antifungal activity against soft rot pathogen Pythium sp. Curr. Plant Biol. 2021, 27, 100217. [Google Scholar] [CrossRef]
- Martins, M.B.; Carvalho, I.D. Diketopiperazines: biological activity and synthesis. Tetrahedron 2007, 63, 9923–9932. [Google Scholar] [CrossRef]
- Kiran, G.S.; Priyadharshini, S.; Sajayan, A.; et al. An antibiotic agent pyrrolo[1,2-a]pyrazine-1,4-dione,hexahydro isolated from a marine bacteria Bacillus tequilensis MSI45 effectively controls multi-drug resistant Staphylococcus aureus. RSC Adv. 2018, 8, 17837–17846. [Google Scholar] [CrossRef] [PubMed]
- Manimaran, M.; Gopal, J.V.; Kannabiran, K. Antibacterial activity of Streptomyces sp. VITMK1 isolated from mangrove soil of pichavaram, Tamil nadu, India. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2017, 87, 499–506. [Google Scholar] [CrossRef]
- Tangjitjaroenkun, J. Evaluation of antioxidant, antibacterial, and gas chromatography-mass spectrometry analysis of ethyl acetate extract of Streptomyces omiyaensis SCH2. Asian J. Pharm. Clin. Res. 2018, 11, 271. [Google Scholar] [CrossRef]
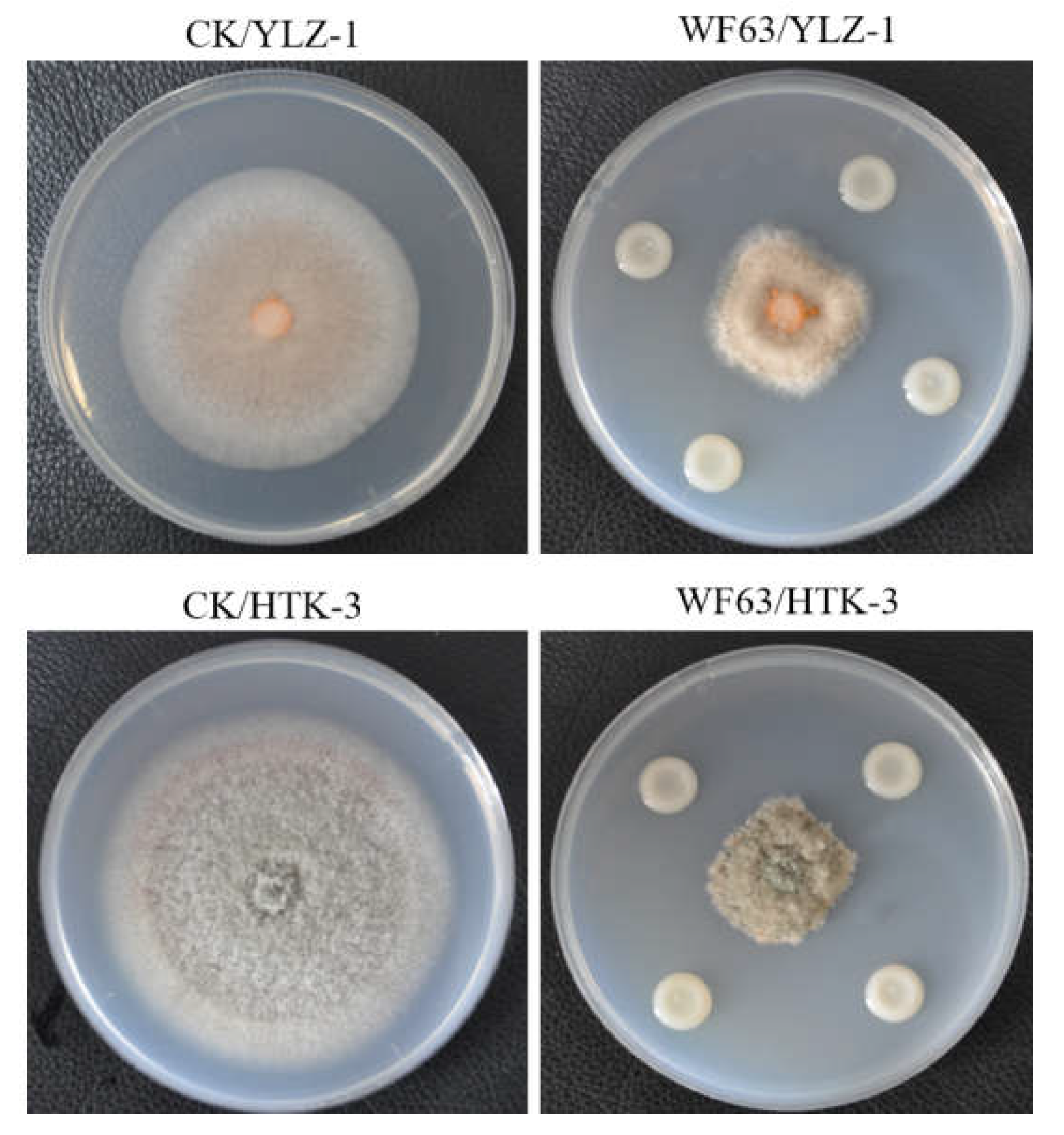
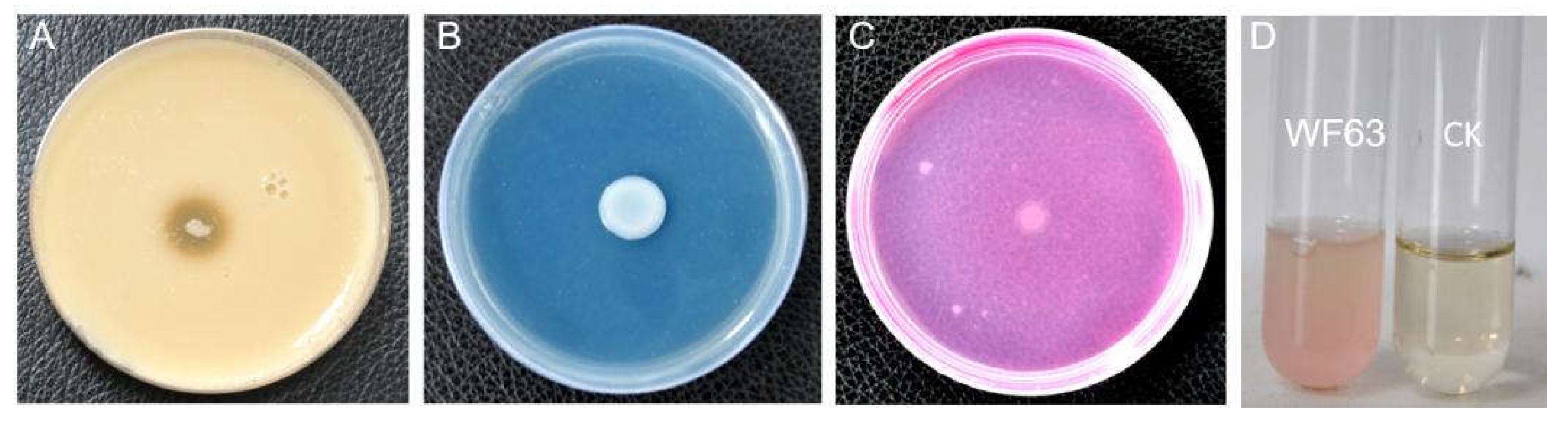

| Treatment | Colony diameter / mm | Inhibition rate / % |
| WF63/YLZ-1 | 47.93±0.15 | 0.44±0.31 |
| WF63/HTK-3 | 47.50±0.51 | 21.80±0.75 |
| CK/YLZ-1 | 48.13±0.15 | — |
| CK/HTK-3 | 60.87±0.12 | — |
| Treatment | Colony diameter / mm | Inhibition rate / % |
| WF63/YLZ-1 | 31.20±0.13 | 50.68±0.43 |
| WF63/HTK-3 | 30.05±0.47 | 61.39±0.27 |
| CK/YLZ-1 | 63.27±0.17 | — |
| CK/HTK-3 | 77.83±0.51 | — |
| Phytopathogen | Plant Disease |
Treatment colony Diameter (mm) |
Control colony diameter(mm) |
Inhibition rate (%) |
| Rhizoctonia solani | Rice sheath blight | 45.13±0.06 | 85.23±0.25 | 47.05±0.18 c |
| Colletotrichum graminicola | Corn anthracnose | 44.30±0.20 | 85.07±0.12 | 47.92±0.18 c |
| Fusarium graminearum | Wheat scab | 48.27±0.25 | 65.8±0.44 | 26.64±0.84 e |
| Colletotrichum siamense | Walnut anthracnose | 39.0±0.20 | 85.17±0.15 | 54.21±0.16 b |
| Botrytis cinera | Strawberry gray mold | 22.17±0.70 | 76.37±0.51 | 70.98±0.81 a |
| Sclerotinia sclerotiorum | Rapeseed sclerotinia stem rot | 25.33±0.99 | 85±0.00 | 70.20±1.16 a |
| Colletotrichum camelliae | Camellia oleifera anthracnose | 27.87±0.12 | 59.27±0.21 | 52.98±0.05 b |
| Bipolaris maydis | Southern corn leaf blight | 35.4±0.53 | 41.3±0.80 | 14.26±2.25 f |
| Colletotrichum gloeosporioides | Mango anthracnose | 40.33±0.15 | 78.73±0.25 | 48.77±0.30 c |
| Magnaporthe grisea | Rice blast | 31.37±0.29 | 50.27±0.21 | 37.60±0.69 d |
| Retention time/min | Identified compound | Relative area /% | Chemical Formula | Moleculaweight /g·mol-1 | CAS No. |
| 8.216 | CYCLO(-PRO-VAL) | 9.067 | C10H16N2O2 | 196.25 | 2854-40-2 |
| 8.916 | 2H-Pyrido[1,2-a]pyrazin-3(4H)-one,hexahydro-(8CI) | 8.530 | C8H14N2O | 154.21 | 15932-74-8 |
| 9.031 | 3-Isobutyl-2,3,6,7,8,8a-hexahydropyrrolo[1,2-a]pyrazine-1,4-dione | 11.032 | C11H18N2O2 | 210.27 | 5654-86-4 |
| 11.460 | Pyrrolo(2,1-F)pyrazine-1,4-dione, 2,3,6,7,8,8A-hexahydro-3-(phenylmethyl)- | 22.371 | C14H16N2O2 | 244.29 | 14705-60-3 |
| 11.553 | 2,2’-Methylenebis(6-tert-butyl-4-methylphenol) | 14.644 | C23H32O2 | 340.5 | 119-47-1 |
| 12.727 | 4-(Methylthio)benzenemethanol | 10.428 | C8H10OS | 154.23 | 3446-90-0 |
| 12.901 | N-[1-[(4-Hydroxyphenyl)methyl]cyclohexyl]propanamide | 23.645 | C16H23NO2 | 261.37 | 696618-84-5 |
| Compound | CAS No. |
Toxicity regression equation (y = ax + b) |
Coefficient of determination (R2) |
EC50 value (mg·L-1) |
| CYCLO(-PRO-VAL) | 2854-40-2 | y=0.8472x+1.6768 | 0.8808 | >8000 |
| 2H-Pyrido[1,2-a]pyrazin-3(4H)-one,hexahydro-(8CI) | 15932-74-8 | y=1.2036x+2.4402 | 0.9084 | 133.88 |
| 3-Isobutyl-2,3,6,7,8,8a-hexahydropyrrolo[1,2-a]pyrazine-1,4-dione | 5654-86-4 | The inhibition rate was negligible or negative | N/A | >200 |
| Pyrrolo(2,1-F)pyrazine-1,4-dione, 2,3,6,7,8,8A-hexahydro-3-(phenylmethyl)- | 14705-60-3 | The data did not exhibit a significant linear increase | N/A | >200 |
| Treatment | Disease index | Relative control efficacy/ % |
| WF63/YLZ-1 | 25.79±1.83 | 52.12±2.96 |
| WF63/HTK-3 | 27.40±4.25 | 56.04±6.49 |
| CK/YLZ-1 | 53.86±0.79 | — |
| CK/HTK-3 | 62.33±4.57 | — |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




