Submitted:
16 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mathematical Model
2.1. Model Description
2.2. Model Implementation and Validation
2.3. Analysis Parameters
2.3.1. Particle Size and Aspect Ratio
2.3.2. Initial Moisture Content
2.3.3. Lignocellulosic Composition
2.3.4. Reactor Temperature
2.3.5. Heating Rate
3. Results and Discussion
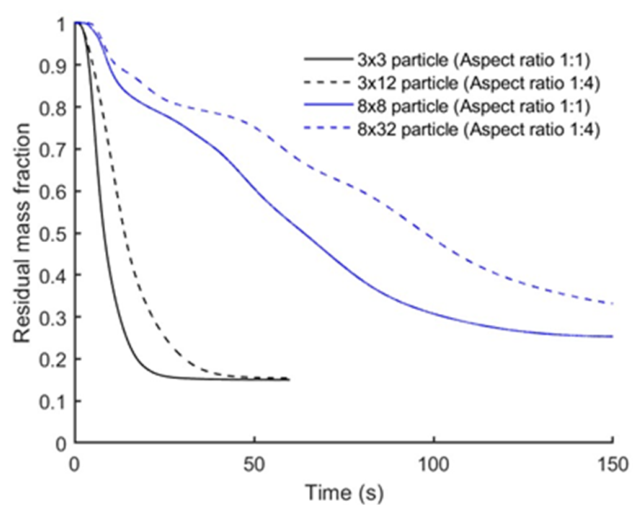
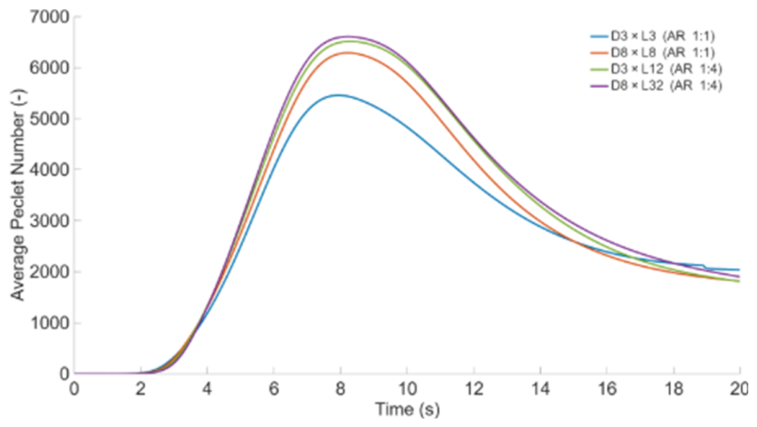
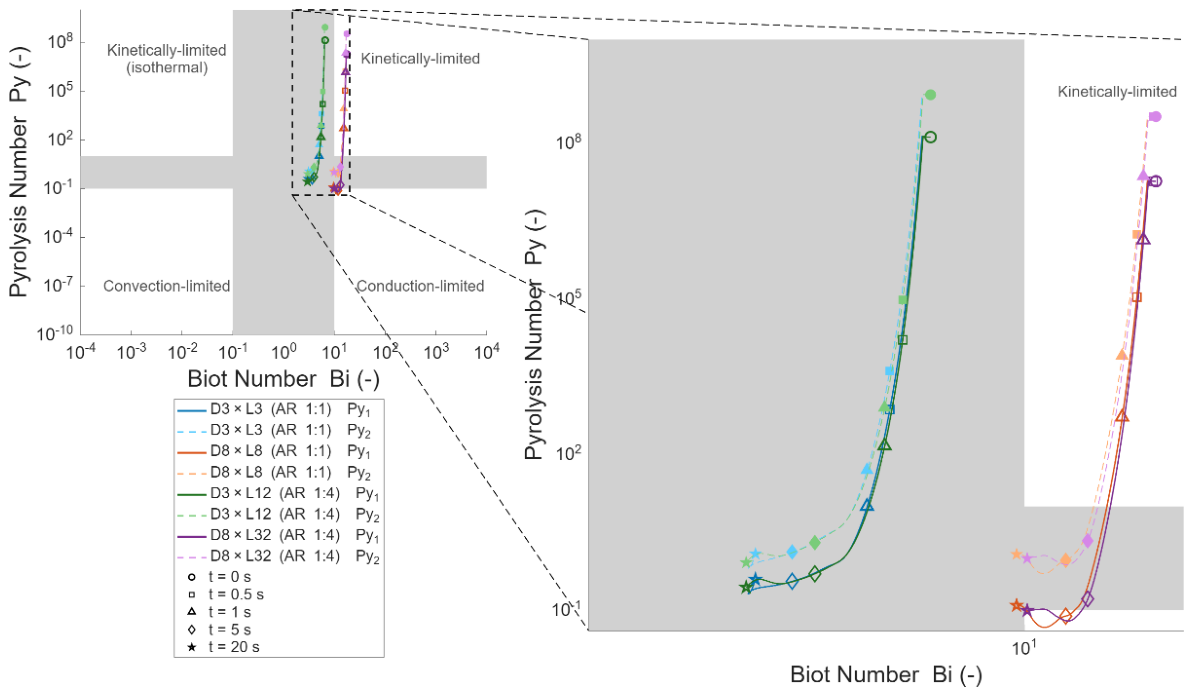
3.1. Effect of Particle Size and Aspect Ratio
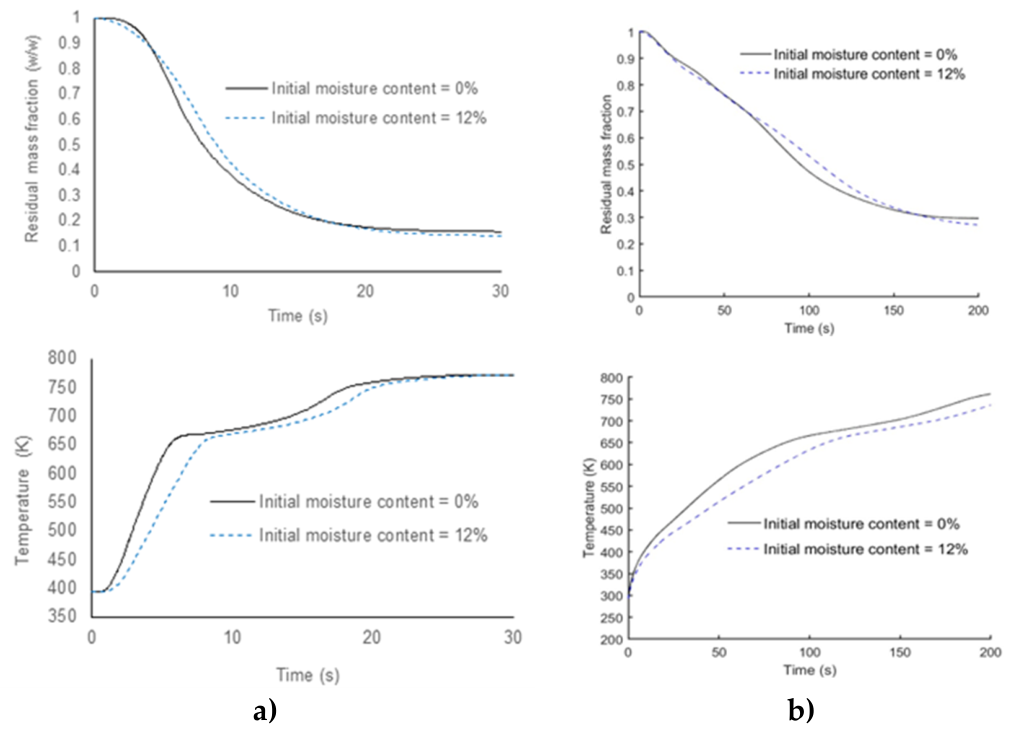
3.2. Effect of the Initial Moisture Content
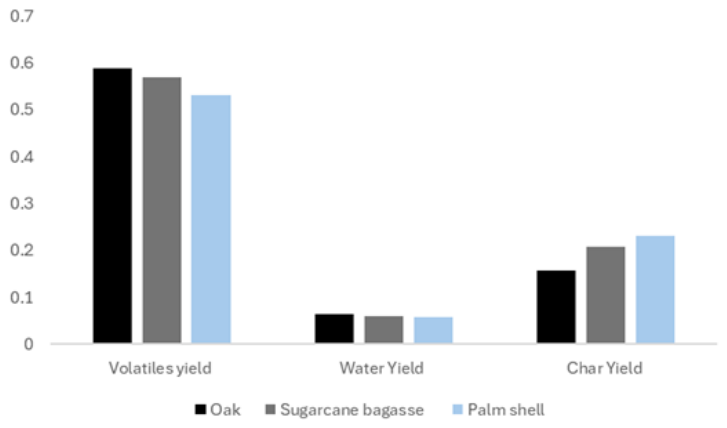
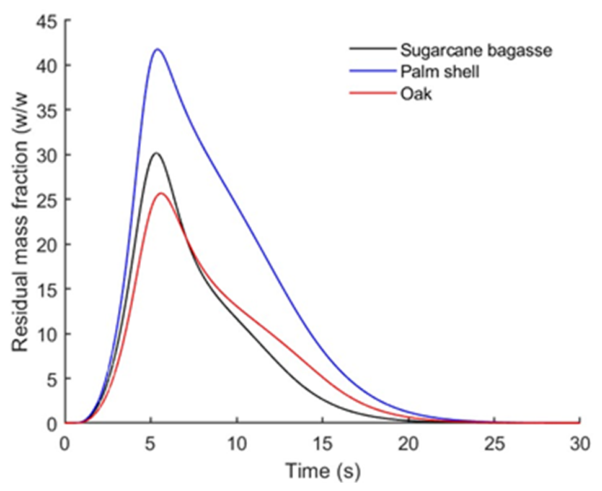
3.3. Effect of Lignocellulosic Composition on Product Yields and Liquid Intermediates Evolution
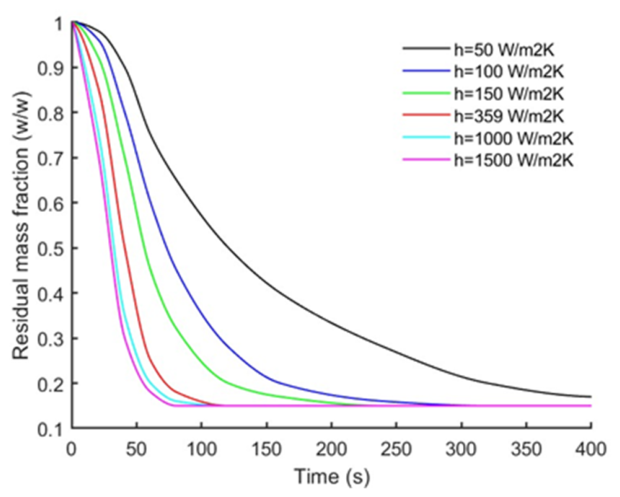
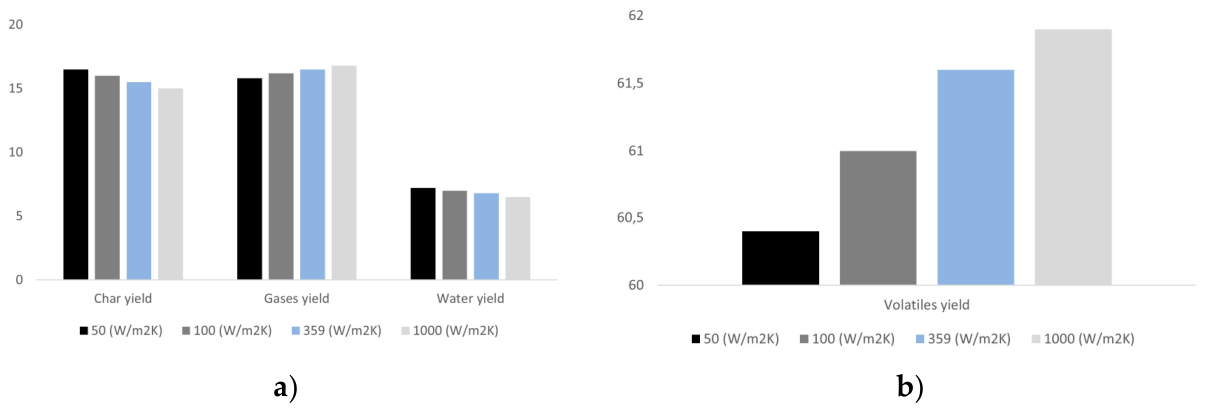
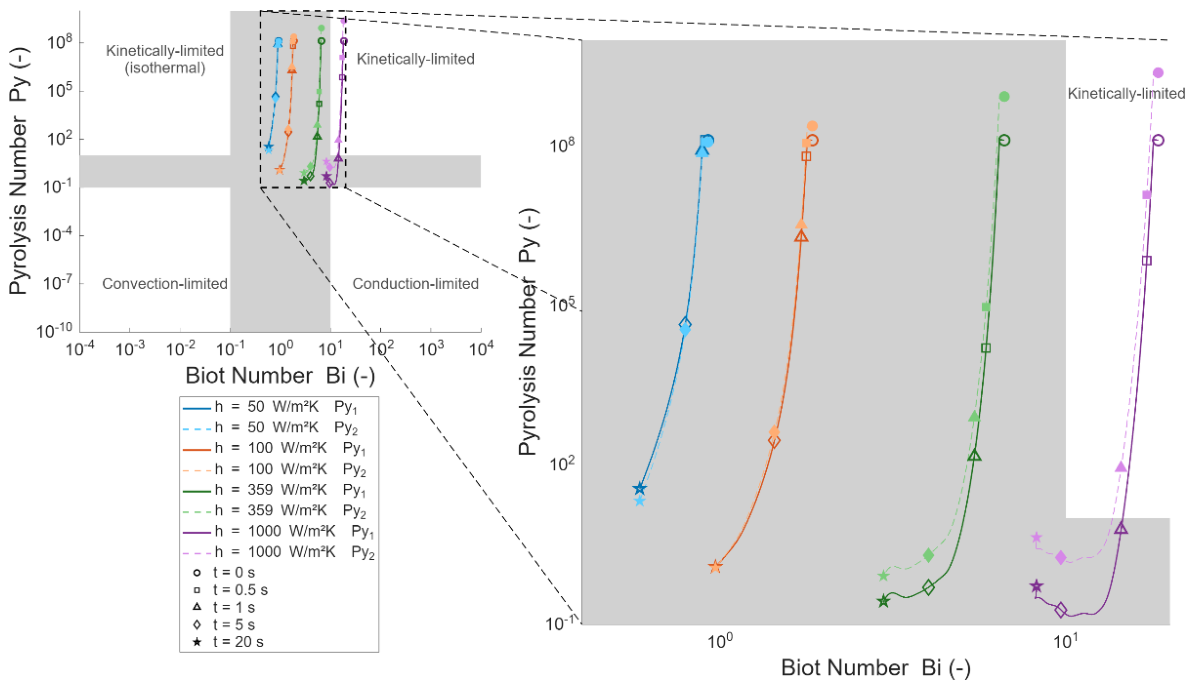
3.4. Effect of Heating Rate on Particle Mass Loss and Products Yields
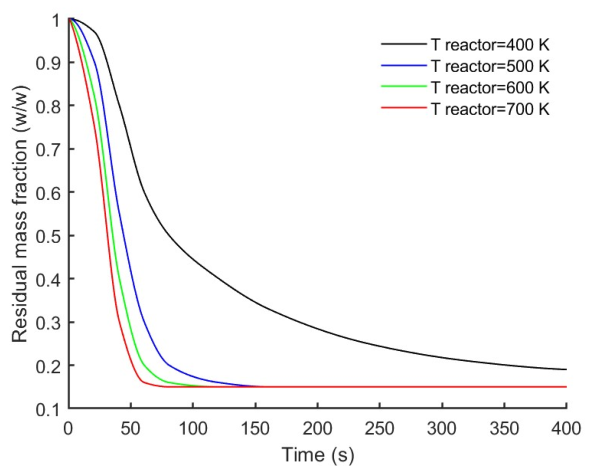
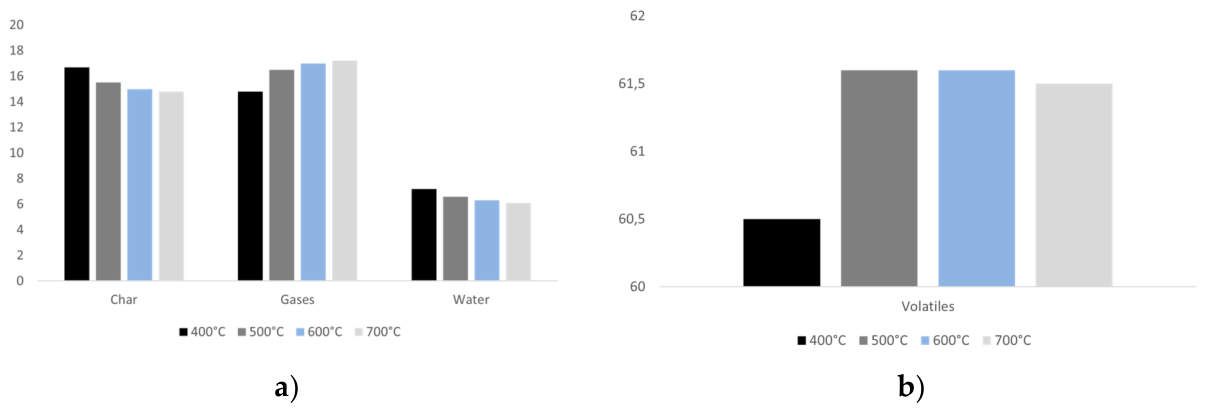
3.5. Effect of Reactor Temperature on Particle Mass Loss and Products Yields
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| Symbol | Description | Units |
| b | biomass | - |
| char | char | - |
| CELL | Cellulose | - |
| CELLA | Active cellulose | - |
| Particle local heat capacity | J·kg-1K-1 | |
| heat capacity of the gaseous components | J·kg-1K-1 | |
| D | Particle diameter | m |
| Effective diffusivity of species in the gas phase | m2·s-1 | |
| G | Gas phase (permanent gases, volatiles, water vapor) | - |
| GAS | Permanent gases | - |
| h | External convective heat transfer coefficient | W·m-2·K-1 |
| HCE | Hemicellulose | - |
| HCE1 | Active hemicellulose type 1 | - |
| HCE2 | Active hemicellulose type 2 | - |
| Water vapor | - | |
| Heat of reaction for reaction j | kJ·kg-1 | |
| Species or the reaction mechanism | - | |
| Reactions of the reaction mechanism | - | |
| External convective mass transfer coefficient | kg·s-1·m-2 | |
| Solid permeability to the gas mixture flow (axial direction) | m2 | |
| Particle length | ||
| Initial particle length | ||
| Final particle length | ||
| LIG-C | Carbon riched lignin | - |
| LIG-CC | Intermediate component from lignin | - |
| LIG-H | Hydrogen riched lignin | - |
| LIG-O | Oxygen riched lignin | - |
| LIG-OH | Intermediate component from lignin | - |
| LIG | Intermediate component from lignin | - |
| Nr | Total number of reactions | - |
| Mass flux in the axial direction | kg·m-2·s-1 | |
| Pressure of the gas mixture | ||
| Heat flux in the axial direction | W·m-2 | |
| Heat flux in the radial direction | W·m-2 | |
| Radial axis | m | |
| Reaction rate for the specie i in the reaction j | mol·m-3·s-1 | |
| Initial particle radius | ||
| Final particle radius | ||
| time | s-1 | |
| Temperature | K | |
| Temperature in the bulk phase | K | |
| VOL. | Volatiles (light condensables) | - |
| Gas velocity | m·s-1 | |
| Axial axis | m | |
| Greek symbols | ||
| Component/phase fraction | - | |
| Average conversion of the particle | - | |
| Effective conductivity in the axial direction | W·m-1·K-1 | |
| Effective conductivity in the radial direction | W·m-1·K-1 | |
| Viscosity of the gas mixture | Pa·s | |
| Relation between axial and radial effective thermal conductivity | - | |
| Local particle density (estimated for each node) | kg·m-3 | |
| Mass concentration of the component i per particle volume | kg·m-3 | |
| Mass concentration of the component i in the gas phase per particle volume | kg·m-3 | |
| Concentration of the specie i in the bulk phase | kg·m-3 | |
References
- Mettler, M. S.; Vlachos, D. G.; Dauenhauer, P. J. Top ten fundamental challenges of biomass pyrolysis for biofuels. Energy Environ. Sci. 2012, vol. 5(no. 7), 7797–7809. [Google Scholar] [CrossRef]
- Crowley, M. F.; et al. Quantifying Impacts of Biomass Pelletization on Fast Pyrolysis Using a Single-Particle Reactor, X-ray Computed Tomography, and Computational Modeling. Energy & Fuels 2026, vol. 40(no. 6), 3136–3158. [Google Scholar] [CrossRef]
- Pecha, M. B.; Arbelaez, J. I. M.; Garcia-Perez, M.; Chejne, F.; Ciesielski, P. N. Progress in understanding the four dominant intra-particle phenomena of lignocellulose pyrolysis: chemical reactions, heat transfer, mass transfer, and phase change. Green Chem. 2019, vol. 21(no. 11), 2868–2898. [Google Scholar] [CrossRef]
- Pyle, D. L.; Zaror, C. A. Heat transfer and kinetics in the low temperature pyrolysis of solids. Chem. Eng. Sci. 1984, vol. 39(no. 1), 147–158. [Google Scholar] [CrossRef]
- Anca-Couce, A.; Zobel, N. Numerical analysis of a biomass pyrolysis particle model: Solution method optimized for the coupling to reactor models. Fuel 2012, vol. 97, 80–88. [Google Scholar] [CrossRef]
- Lu, H.; Ip, E.; Scott, J.; Foster, P.; Vickers, M.; Baxter, L. L. Effects of particle shape and size on devolatilization of biomass particle. Fuel 2010, vol. 89(no. 5), 1156–1168. [Google Scholar] [CrossRef]
- Shi, X.; Ronsse, F.; Pieters, J. G. Finite element modeling of intraparticle heterogeneous tar conversion during pyrolysis of woody biomass particles. Fuel Processing Technology 2016, vol. 148, 302–316. [Google Scholar] [CrossRef]
- Gentile, G.; Debiagi, P. E. A.; Cuoci, A.; Frassoldati, A.; Ranzi, E.; Faravelli, T. A computational framework for the pyrolysis of anisotropic biomass particles. Chemical Engineering Journal 2017, vol. 321, 458–473. [Google Scholar] [CrossRef]
- Blondeau, J.; Jeanmart, H. Biomass pyrolysis at high temperatures: Prediction of gaseous species yields from an anisotropic particle. Biomass Bioenergy 2012, vol. 41, 107–121. [Google Scholar] [CrossRef]
- Okekunle, P. O.; Watanabe, H.; Pattanotai, T.; Okazaki, K. Effect of biomass size and aspect ratio on intra-particle tar decomposition during wood cylinder pyrolysis. Journal of Thermal Science and Technology 2012, vol. 7(no. 1), 1–15. [Google Scholar] [CrossRef]
- Kwiatkowski, K.; Górecki, B.; Korotko, J.; Gryglas, W.; Dudyński, M.; Bajer, K. Numerical modeling of biomass pyrolysis - Heat and mass transport models. Numeri. Heat Transf. A Appl. 2013, vol. 64(no. 3), 216–234. [Google Scholar] [CrossRef]
- Pecha, B. , Integrated Particle- and Reactor-Scale Simulation of Pine Pyrolysis in a Fluidized Bed. Energy and Fuels 2018, vol. 32(no. 10), 10683–10694. [Google Scholar] [CrossRef]
- Di Blasi, C. Physico-chemical processes occurring inside a degrading two-dimensional anisotropic porous medium. Int. J. Heat Mass Transf. 1998, vol. 41(no. 24), 4139–4150. [Google Scholar] [CrossRef]
- Sánchez, M.; Maya, J. C.; Pecha, B.; Chejne, F.; Quinchía-Figueroa, A. M. Effect of particle characteristics, kinetics and transport phenomena on the prediction of particle mass loss and products yields during biomass fast pyrolysis. J. Anal. Appl. Pyrolysis 2022, vol. 168, 105786. [Google Scholar] [CrossRef]
- Debiagi, P.; Gentile, G.; Cuoci, A.; Frassoldati, A.; Ranzi, E.; Faravelli, T. A predictive model of biochar formation and characterization. J. Anal. Appl. Pyrolysis 2018, vol. 134, no. June, 326–335. [Google Scholar] [CrossRef]
- Maduskar, S.; Facas, G. G.; Papageorgiou, C.; Williams, C. L.; Dauenhauer, P. J. Five Rules for Measuring Biomass Pyrolysis Rates: Pulse-Heated Analysis of Solid Reaction Kinetics of Lignocellulosic Biomass. ACS Sustain. Chem. Eng. 2018, vol. 6(no. 1), 1387–1399. [Google Scholar] [CrossRef]
- Lu, L.; et al. Investigating biomass composition and size effects on fast pyrolysis using global sensitivity analysis and CFD simulations. Chemical Engineering Journal 2021, vol. 421, 127789. [Google Scholar] [CrossRef]
- Lihra, T.; Cloutier, A.; Zhang, S.-Y. Longitudinal and transverse permeability of balsam fir wetwood and normal heartwood. Wood and Fiber Science 2000, vol. 32(no. 2), 164–178. Available online: https://www.scopus.com/inward/record.uri?eid=2-s2.0-0041036536&partnerID=40&md5=19c177f01699b9eb39cb8c3f6c9c375b.
- Kollmann, F. F. P.; Kuenzi, E. W.; Stamm, A. J. Principles of Wood Science and Technology. In Principles of Wood Science and Technology; 1968. [Google Scholar] [CrossRef]
- Ciesielski, P. N.; et al. Biomass Particle Models with Realistic Morphology and Resolved Microstructure for Simulations of Intraparticle Transport Phenomena. Energy & Fuels 2015, vol. 29(no. 1), 242–254. [Google Scholar] [CrossRef]
- Bellais, M.; Davidsson, K. O.; Liliedahl, T.; Sjöström, K.; Pettersson, J. B. C. Pyrolysis of large wood particles: A study of shrinkage importance in simulations. Fuel 2003, vol. 82(no. 12), 1541–1548. [Google Scholar] [CrossRef]
- Di Blasi, C. Modeling chemical and physical processes of wood and biomass pyrolysis. Prog. Energy Combust. Sci. 2008, vol. 34(no. 1), 47–90. [Google Scholar] [CrossRef]
- Grønli, M. G.; Várhegyi, G.; Di Blasi, C. Thermogravimetric analysis and devolatilization kinetics of wood. Ind. Eng. Chem. Res. 2002, vol. 41(no. 17), 4201–4208. [Google Scholar] [CrossRef]
- Dufour, A.; Ouartassi, B.; Bounaceur, R.; Zoulalian, A. Modelling intra-particle phenomena of biomass pyrolysis. Chemical Engineering Research and Design 2011, vol. 89(no. 10), 2136–2146. [Google Scholar] [CrossRef]
- Montoya, J.; Pecha, B.; Janna, F. C.; Garcia-Perez, M. Micro-explosion of liquid intermediates during the fast pyrolysis of sucrose and organosolv lignin. J. Anal. Appl. Pyrolysis 2016, vol. 122, 106–121. [Google Scholar] [CrossRef]
- Zhou, S.; Pecha, B.; van Kuppevelt, M.; McDonald, A. G.; Garcia-Perez, M. Slow and fast pyrolysis of Douglas-fir lignin: Importance of liquid-intermediate formation on the distribution of products. Biomass Bioenergy 2014, vol. 66, 398–409. [Google Scholar] [CrossRef]
- Teixeira, A. R. , Aerosol generation by reactive boiling ejection of molten cellulose. Energy Environ. Sci. 2011, vol. 4(no. 10), 4306–4321. [Google Scholar] [CrossRef]
- Jendoubi, N.; Broust, F.; Commandre, J. M.; Mauviel, G.; Sardin, M.; Lédé, J. Inorganics distribution in bio oils and char produced by biomass fast pyrolysis: The key role of aerosols. J. Anal. Appl. Pyrolysis 2011, vol. 92(no. 1), 59–67. [Google Scholar] [CrossRef]
- Okekunle, P. O.; Pattanotai, T.; Watanabe, H.; Okazaki, K. Numerical and experimental investigation of intra-particle heat transfer and tar decomposition during pyrolysis of wood biomass. Journal of Thermal Science and Technology 2011, vol. 6(no. 3), 360–375. [Google Scholar] [CrossRef]
- Pecha, M. B. , Impacts of Anisotropic Porosity on Heat Transfer and Off-Gassing during Biomass Pyrolysis. Energy & Fuels 2021, vol. 35(no. 24), 20131–20141. [Google Scholar] [CrossRef]
- Montoya, J.; Pecha, B.; Chejne, F.; Garcia-Perez, M. Single particle model for biomass pyrolysis with bubble formation dynamics inside the liquid intermediate and its contribution to aerosol formation by thermal ejection. J. Anal. Appl. Pyrolysis 2017, vol. 124, 204–218. [Google Scholar] [CrossRef]
- Kamila, B.; Sadhukhan, A. K.; Gupta, P. 2D CFD modeling for pyrolysis of a large biomass particle: Effect of L/D ratio, internal convection and shrinkage. Chemical Engineering Research and Design 2024, vol. 208, 921–933. [Google Scholar] [CrossRef]
- Fatehi, H.; Weng, W.; Li, Z.; Bai, X. S.; Aldén, M. Recent development in numerical simulations and experimental studies of biomass thermochemical conversion. In American Chemical Society; 06 May 2021. [Google Scholar] [CrossRef]
- Wang, J.; Ku, X.; Liu, Z. Three-Dimensional Simulation of the Pyrolysis of a Thermally Thick Biomass Particle. Energy & Fuels 2023, vol. 37(no. 6), 4413–4428. [Google Scholar] [CrossRef]
- Wu, Y.; et al. Effect of biomass components’ interaction on the pyrolysis reaction kinetics and small-molecule product release characteristics. J. Anal. Appl. Pyrolysis 2023, vol. 173, 106039. [Google Scholar] [CrossRef]
- Müller-Hagedorn, M.; Bockhorn, H. Pyrolytic behaviour of different biomasses (angiosperms) (maize plants, straws, and wood) in low temperature pyrolysis. J. Anal. Appl. Pyrolysis 2007, vol. 79(no. 1–2), 136–146. [Google Scholar] [CrossRef]
- Ranzi, E.; Debiagi, P. E. A.; Frassoldati, A. Mathematical Modeling of Fast Biomass Pyrolysis and Bio-Oil Formation. Note II: Secondary Gas-Phase Reactions and Bio-Oil Formation. ACS Sustain. Chem. Eng. 2017, vol. 5(no. 4), 2882–2896. [Google Scholar] [CrossRef]
- Bridgwater, A. V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, vol. 38, 68–94. [Google Scholar] [CrossRef]
- Várhegyi, G.; Bobály, B.; Jakab, E.; Chen, H. Thermogravimetric Study of Biomass Pyrolysis Kinetics. A Distributed Activation Energy Model with Prediction Tests. Energy & Fuels 2011, vol. 25(no. 1), 24–32. [Google Scholar] [CrossRef]
- Antal, M. J.; Grønli, M. The Art, Science, and Technology of Charcoal Production. Ind. Eng. Chem. Res. 2003, vol. 42(no. 8), 1619–1640. [Google Scholar] [CrossRef]
- Luo, H.; et al. Multi-fluid modeling of heat transfer in bubbling fluidized bed with thermally-thick particles featuring intra-particle temperature inhomogeneity. Chemical Engineering Journal 2023, vol. 460. [Google Scholar] [CrossRef]
- Gao, X.; Yu, J.; Lu, L.; Rogers, W. A. Coupling particle scale model and SuperDEM-CFD for multiscale simulation of biomass pyrolysis in a packed bed pyrolyzer. AIChE Journal 2021, vol. 67(no. 4), e17139. [Google Scholar] [CrossRef]
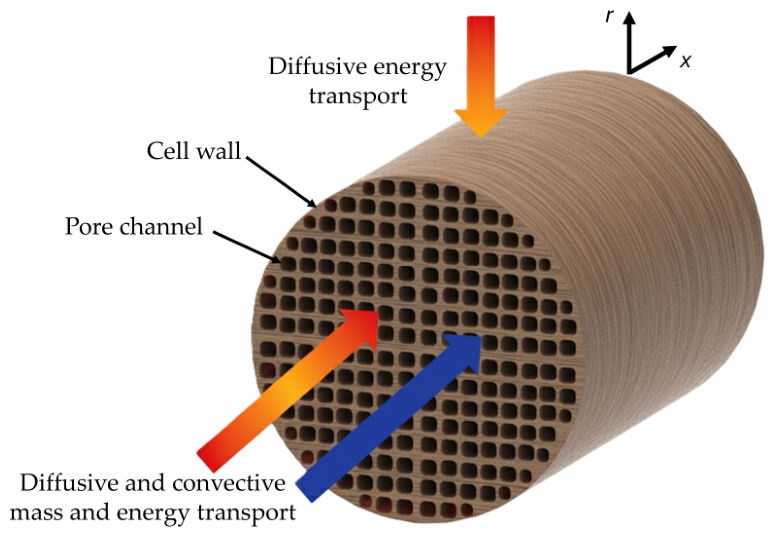
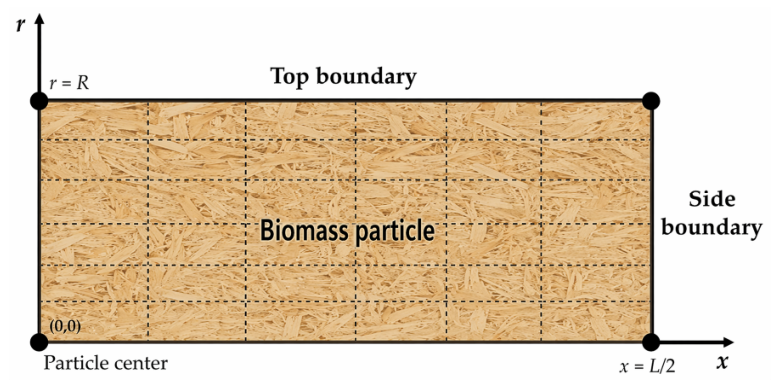
| Aspect | Consideration | Equation | |
|---|---|---|---|
| Particle geometry | Porous anisotropic cylinder, radial and longitudinal shrinkage modeled as functions of average conversion. |
(1) (2) |
|
| Reaction scheme | CRECK mechanism [15]: first-order Arrhenius kinetics |
|
|
| Solid/liquid phase mass balance | Biomass pseudo-components, metaplast and char | (3) | |
| Gas phase mass balance | Diffusive + convective transport in axial direction; diffusive only in radial direction: | (4) | |
| Gas velocity | Darcy's Law | (5) | |
| Energy balance | Diffusive transport (axial + radial) + convective transport (axial) + heat of reactions | (6) | |
| Boundary conditions | Side boundary: convective heat and mass transfer |
(7) (8) |
|
| Top boundary: convective heat and mass transfer |
(9) (10) |
||
| Symmetry boundaries: central axes | (11) | (12) | |
| (13) | (14) | ||
| Parameter | Values studied | Fixed parameters |
|---|---|---|
| Particle size and aspect ratio | D×L: 3×3 mm (A.R. 1:1), 3×12 mm (A.R. 1:4), 8×8 mm (A.R. 1:1), 8×16 mm (A.R. 1:2), 8×24 mm (A.R. 1:3), and 8×32 mm (A.R. 1:4) | T = 500 °C, h = 359 W·m⁻²·K⁻¹, oak, 0% moisture |
| Initial moisture content | 0% and 12% (w/w) | D = 3 mm, L = 12 mm, T = 500 °C, h = 359 W·m⁻²·K⁻¹, oak D = 8 mm, L = 32 mm, T = 500 °C, h = 359 W·m⁻²·K⁻¹, oak |
| Lignocellulosic composition | Oak (CELL 44%, HCE 34%, LIG 22%), Sugarcane bagasse (CELL 28%, HCE 44%, LIG 23%, ashes 5%), Palm shell (CELL 27%, HCE 23%, LIG 50%) | D = 3 mm, L = 12 mm, T = 500 °C, h = 359 W·m⁻²·K⁻¹, 0% moisture |
| Heating rate (convective coefficient) | h = 50, 100, 150, 359, 1000, 1500 W·m⁻²·K⁻¹ | D = 3 mm, L = 12 mm, T = 500 °C, oak; 0% moisture |
| Reactor temperature | 400, 500, 600, 700 °C | D = 3 mm, L = 12 mm, h = 359 W·m⁻²·K⁻¹, oak, 0% moisture |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




