Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
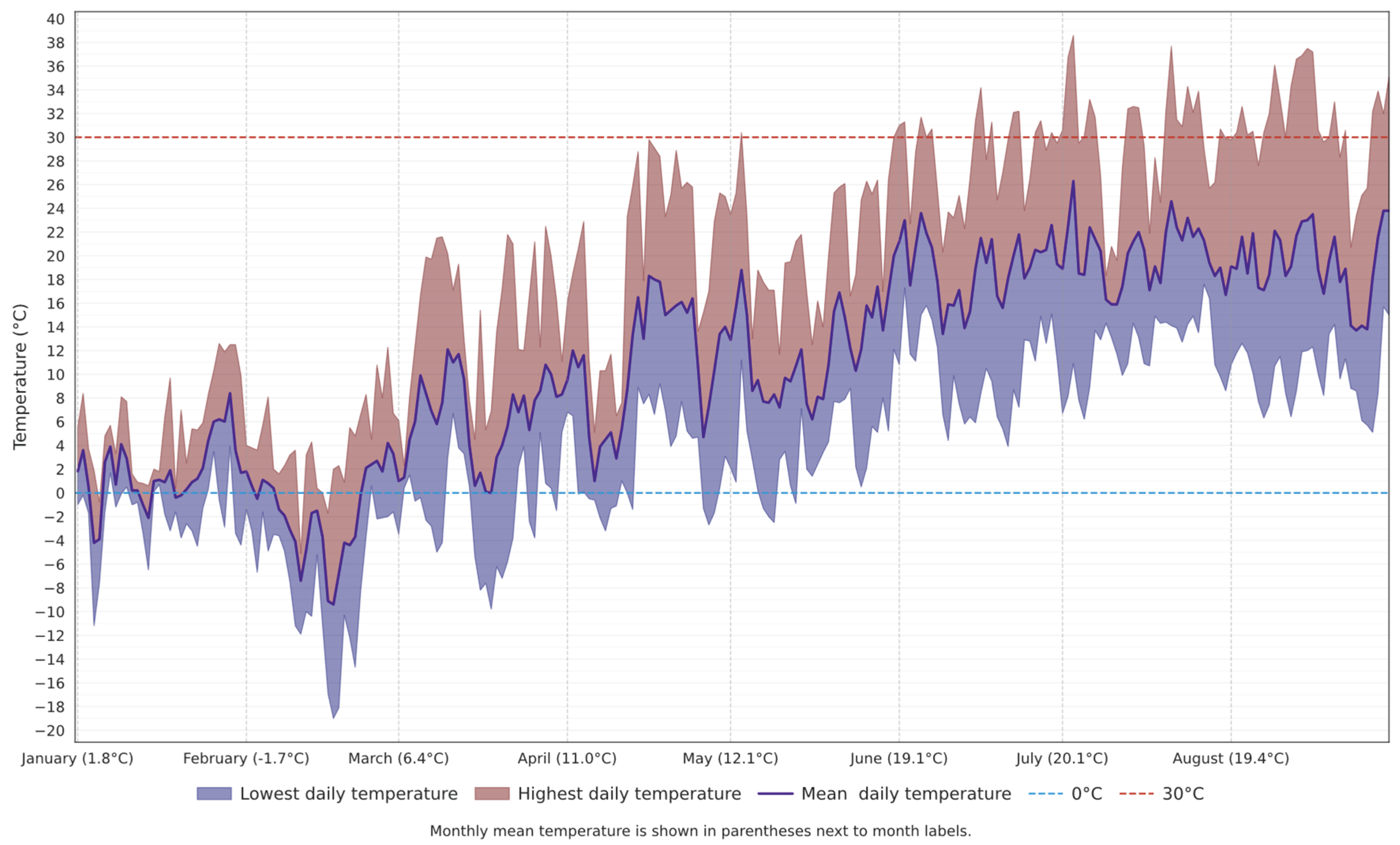
2.1. Overview of the Research Area
2.2. Biostimulation
2.3. Measurements
2.3.1. Yield
2.3.2. Multispectral Imaging
2.4. Analysis of Results
2.4.1. Statistical Analysis
2.4.2. Multispectral Data Analysis
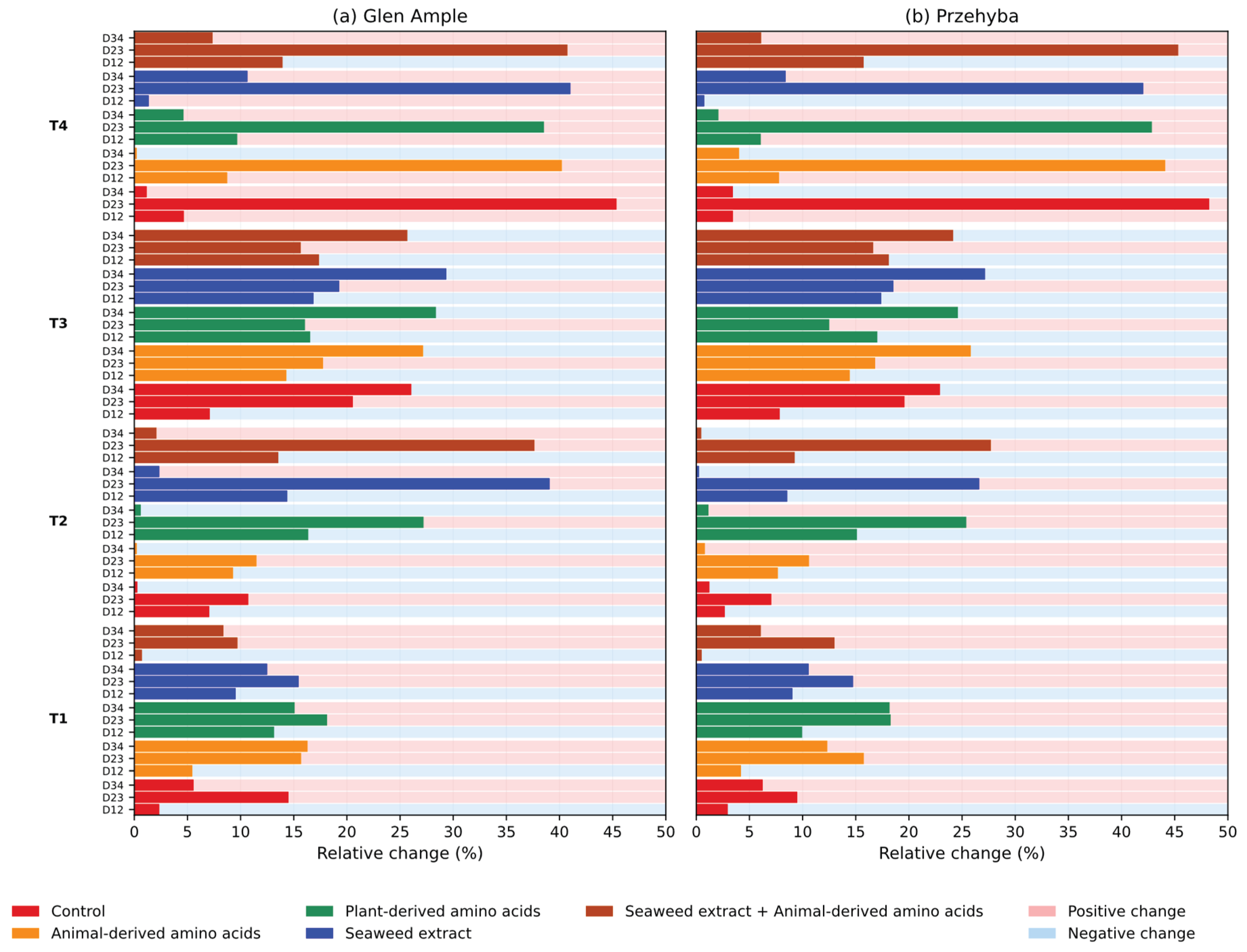
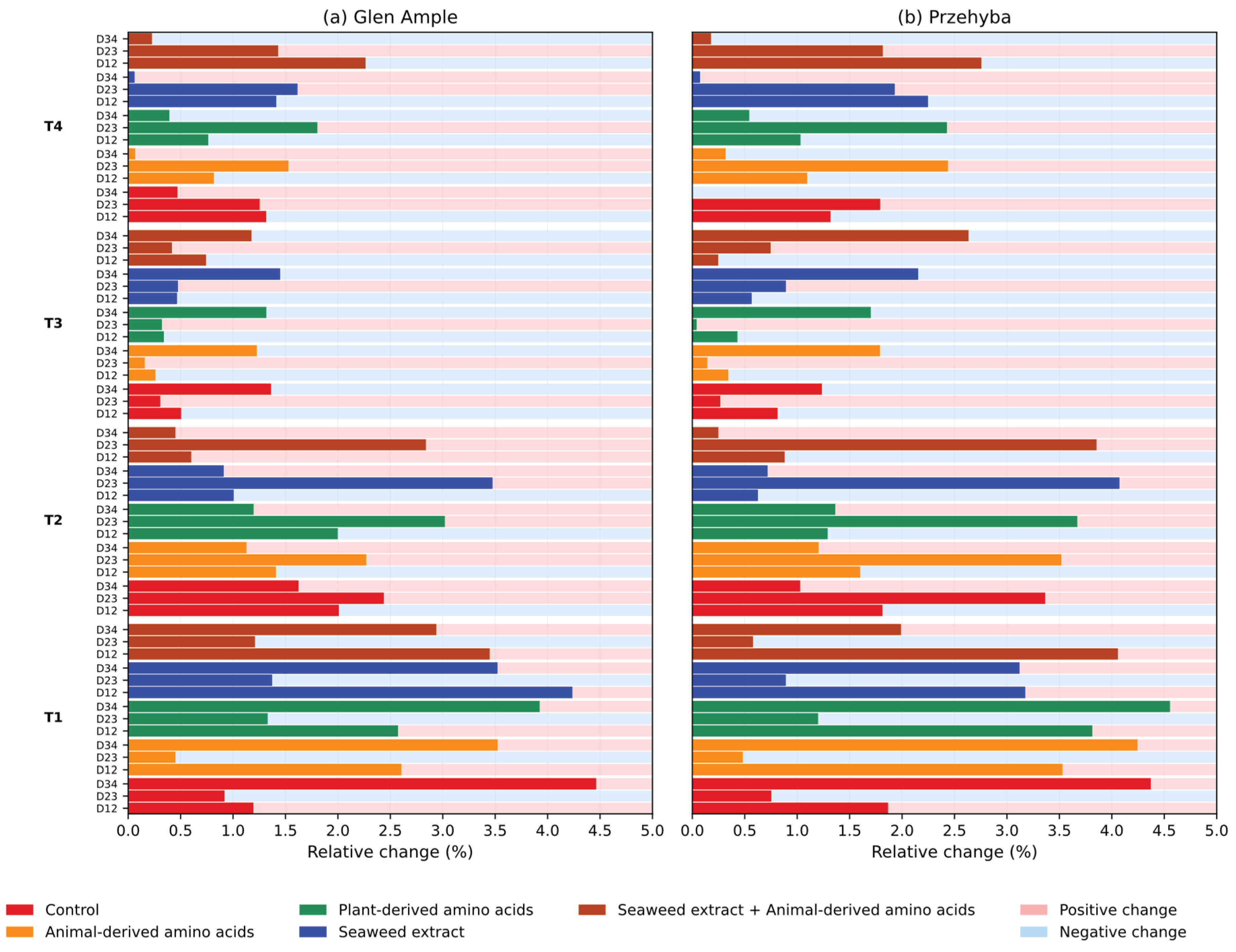
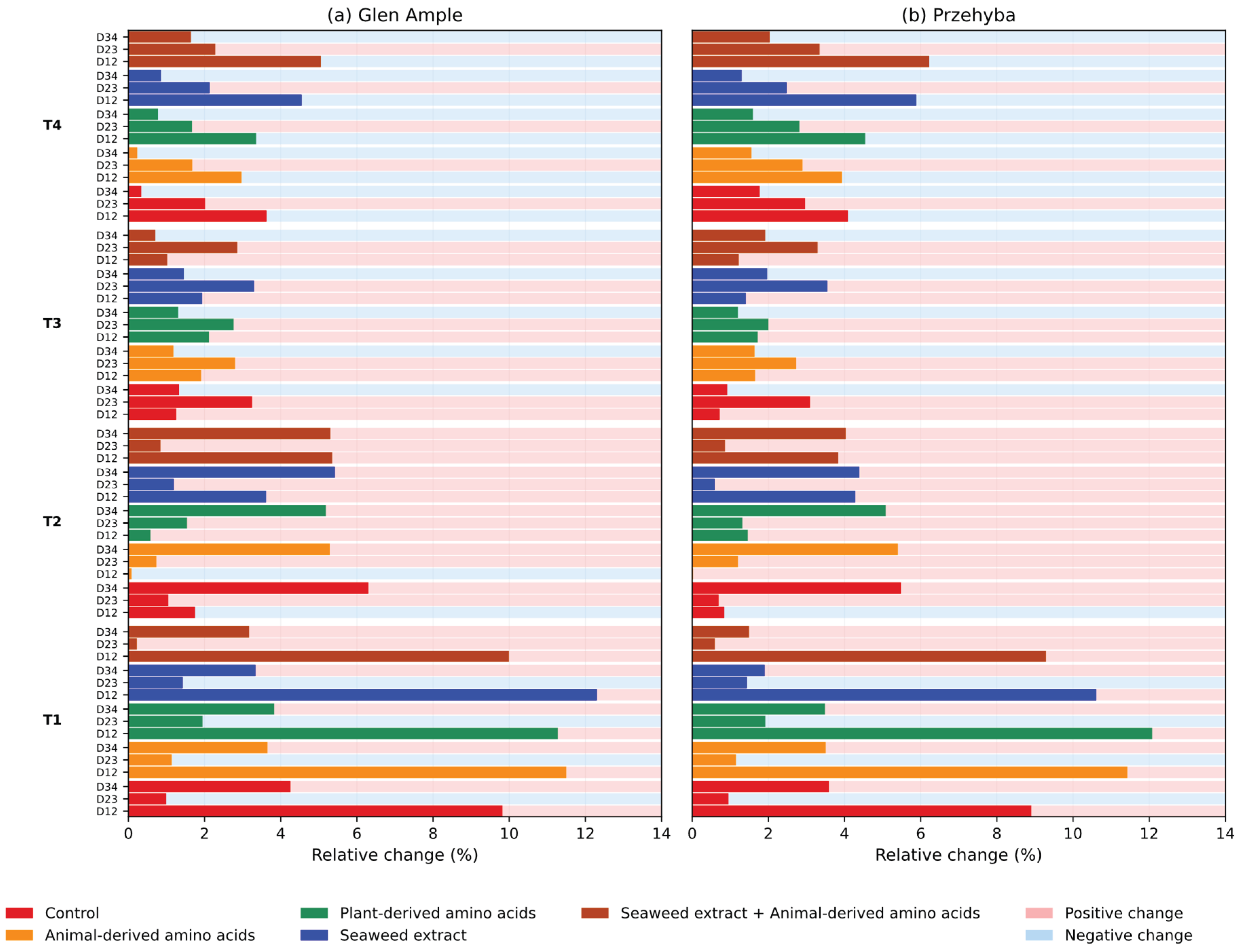
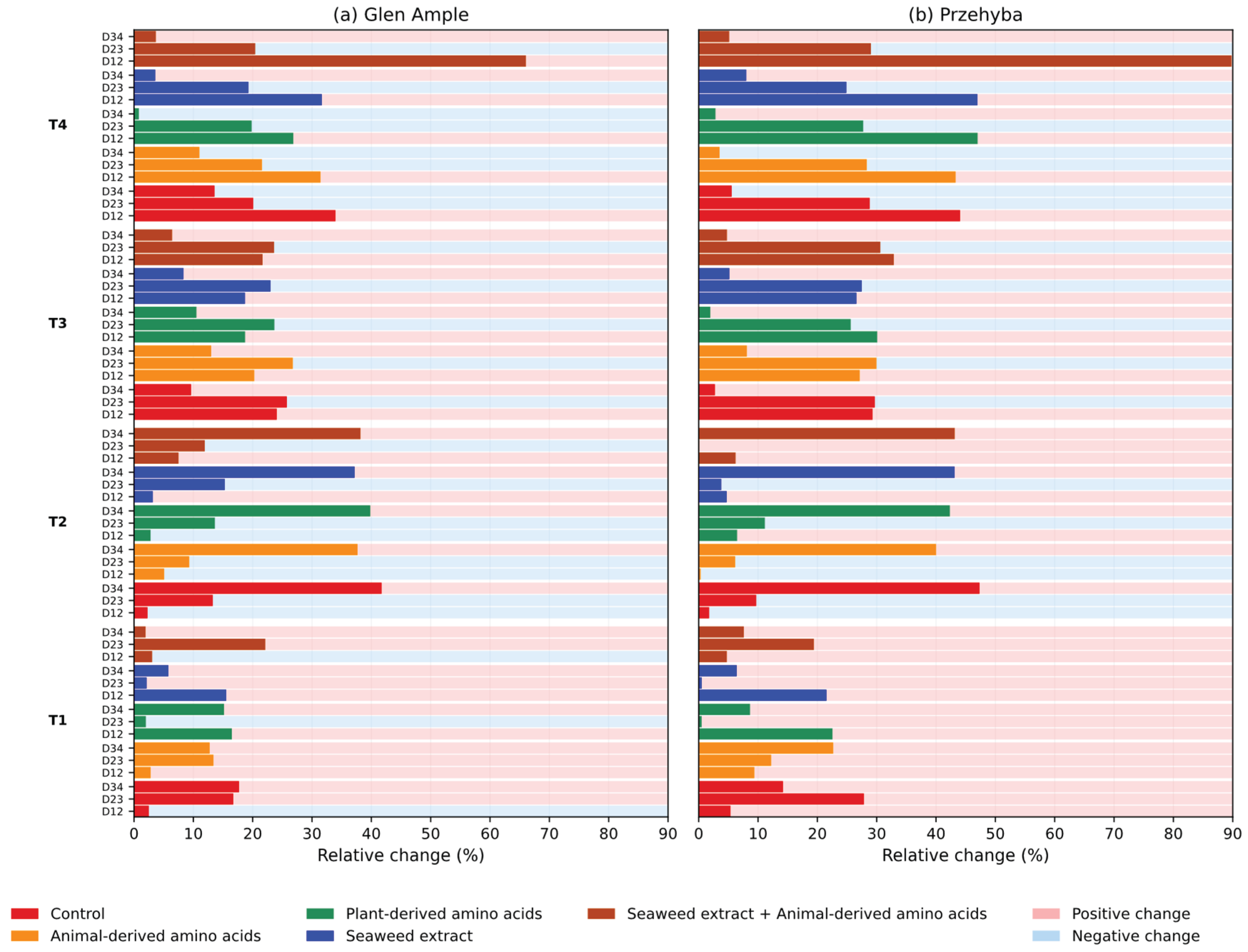
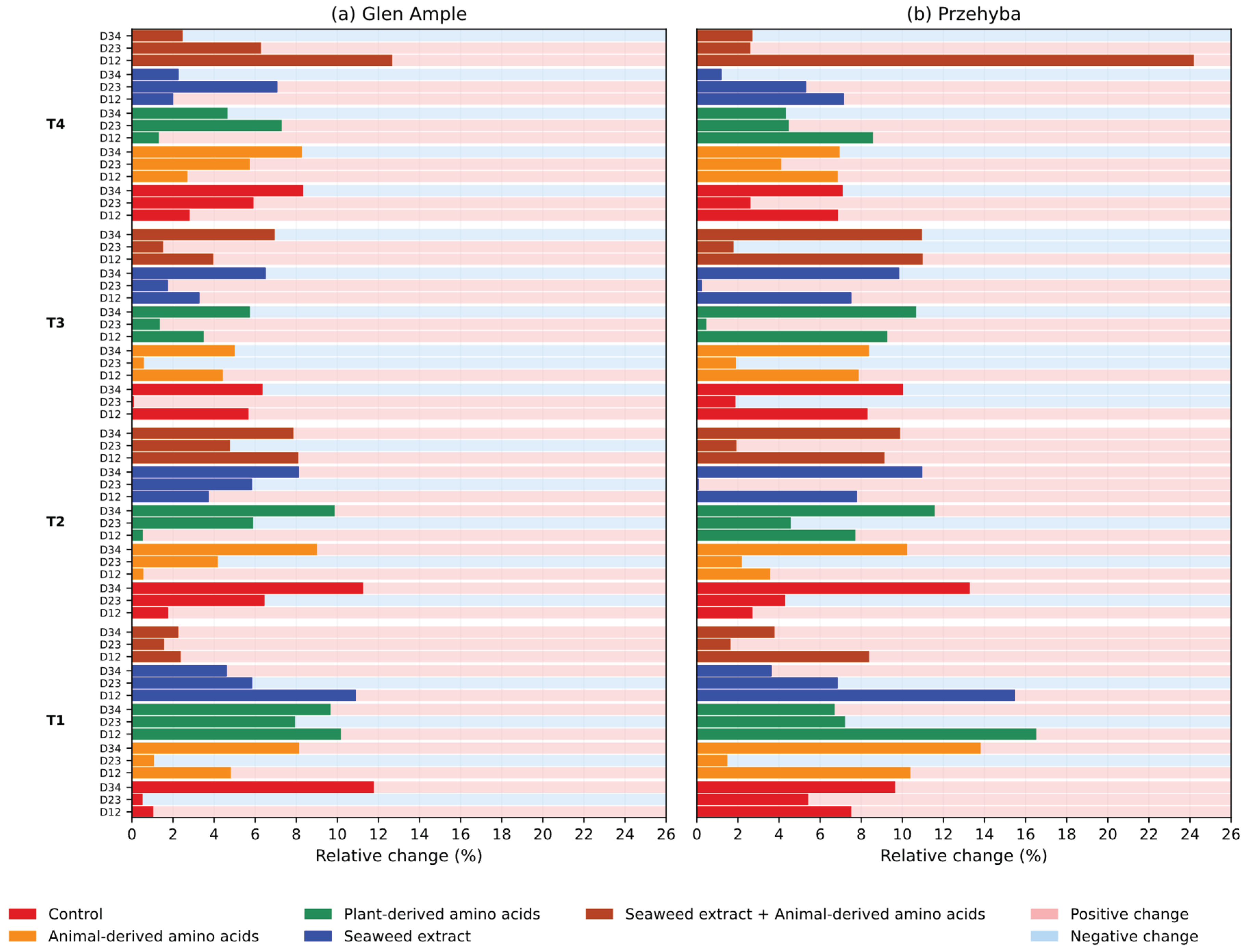
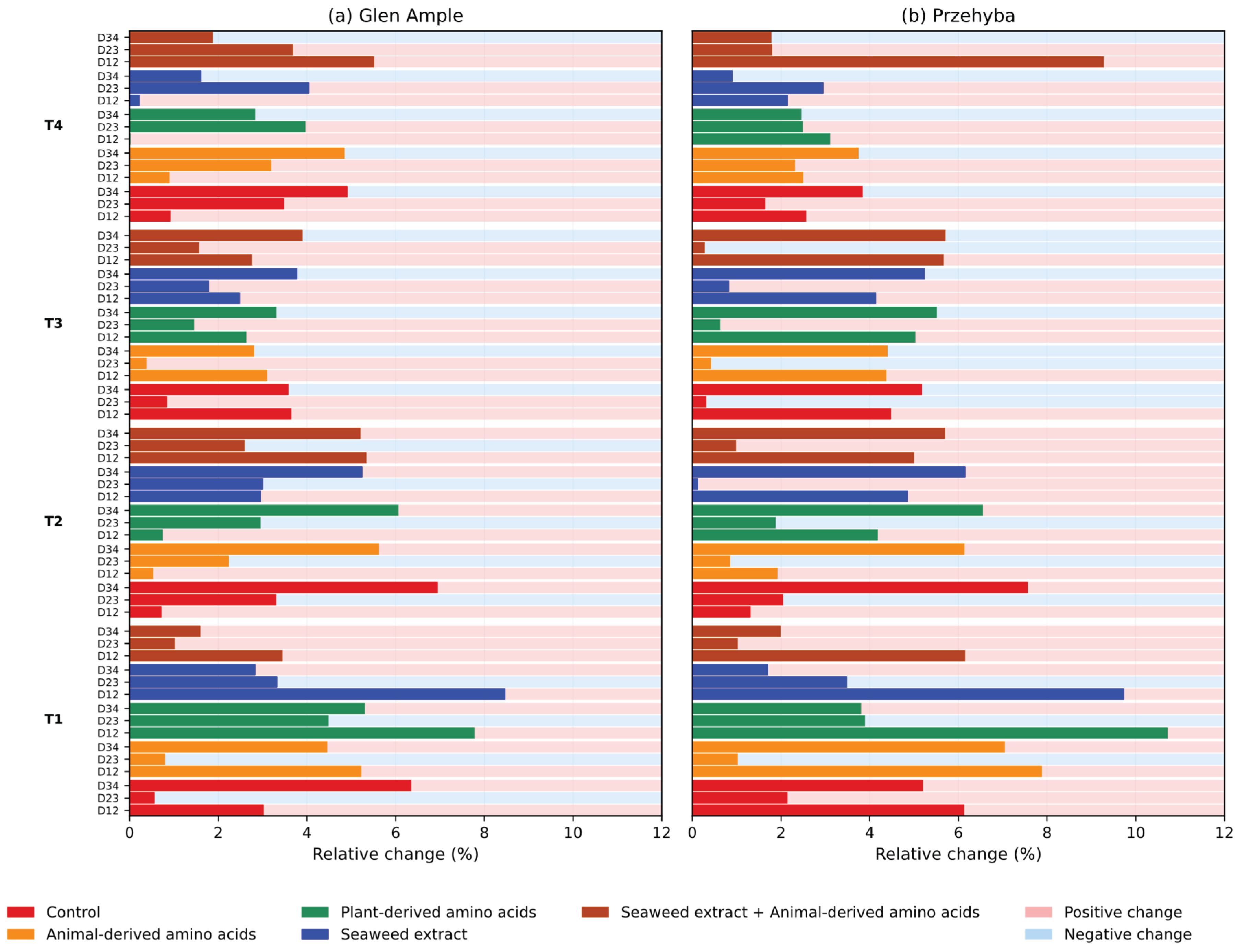
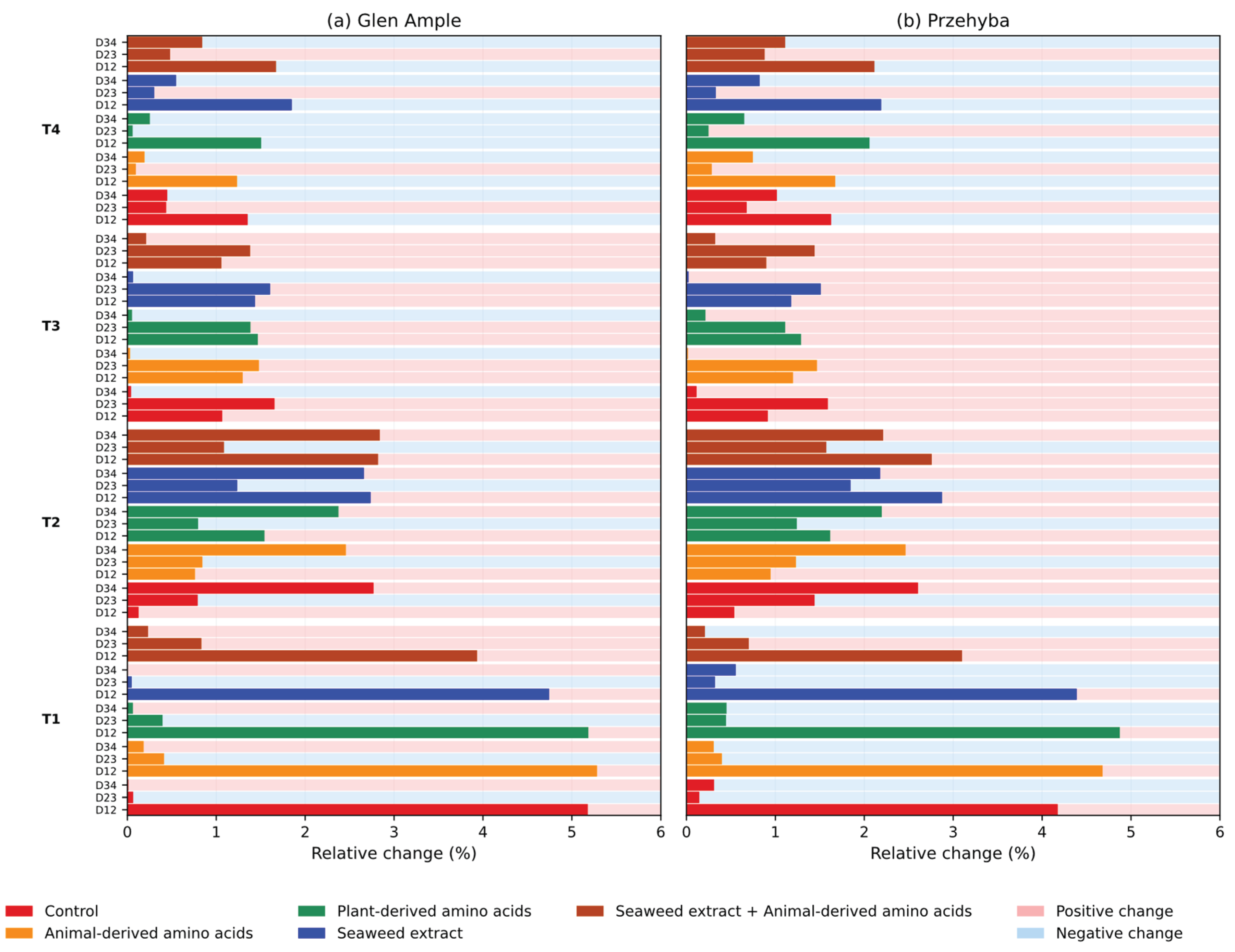
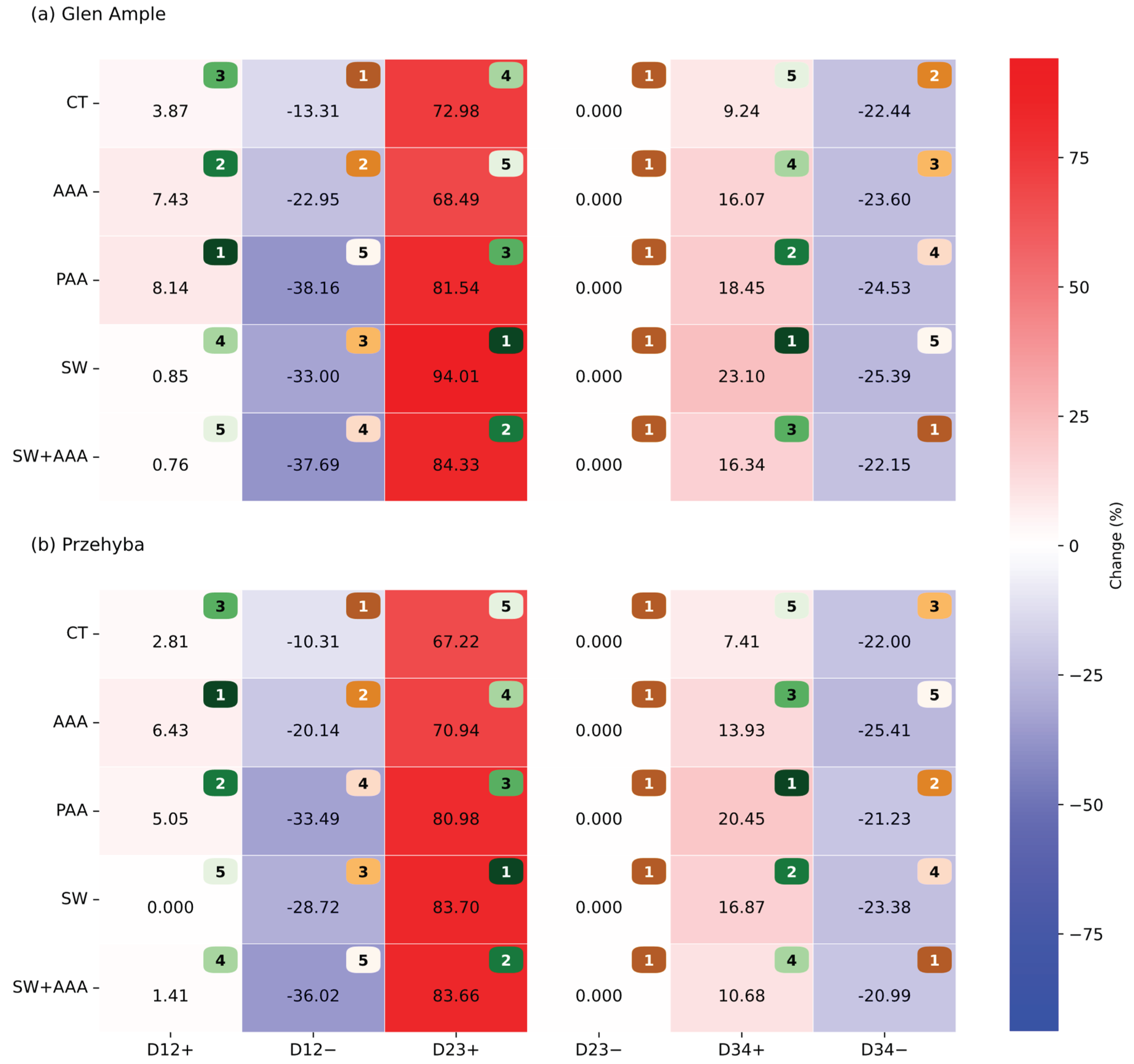
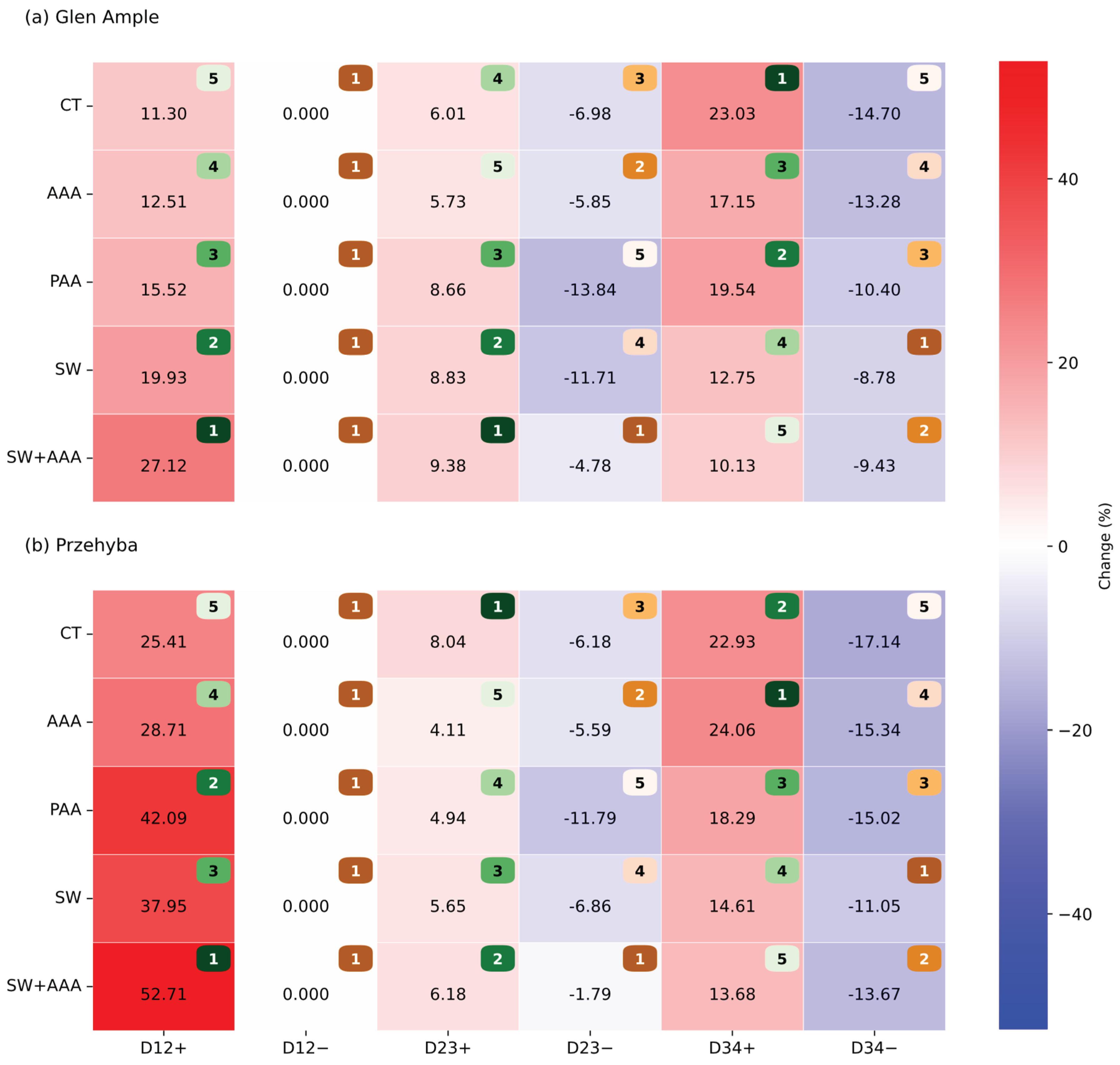
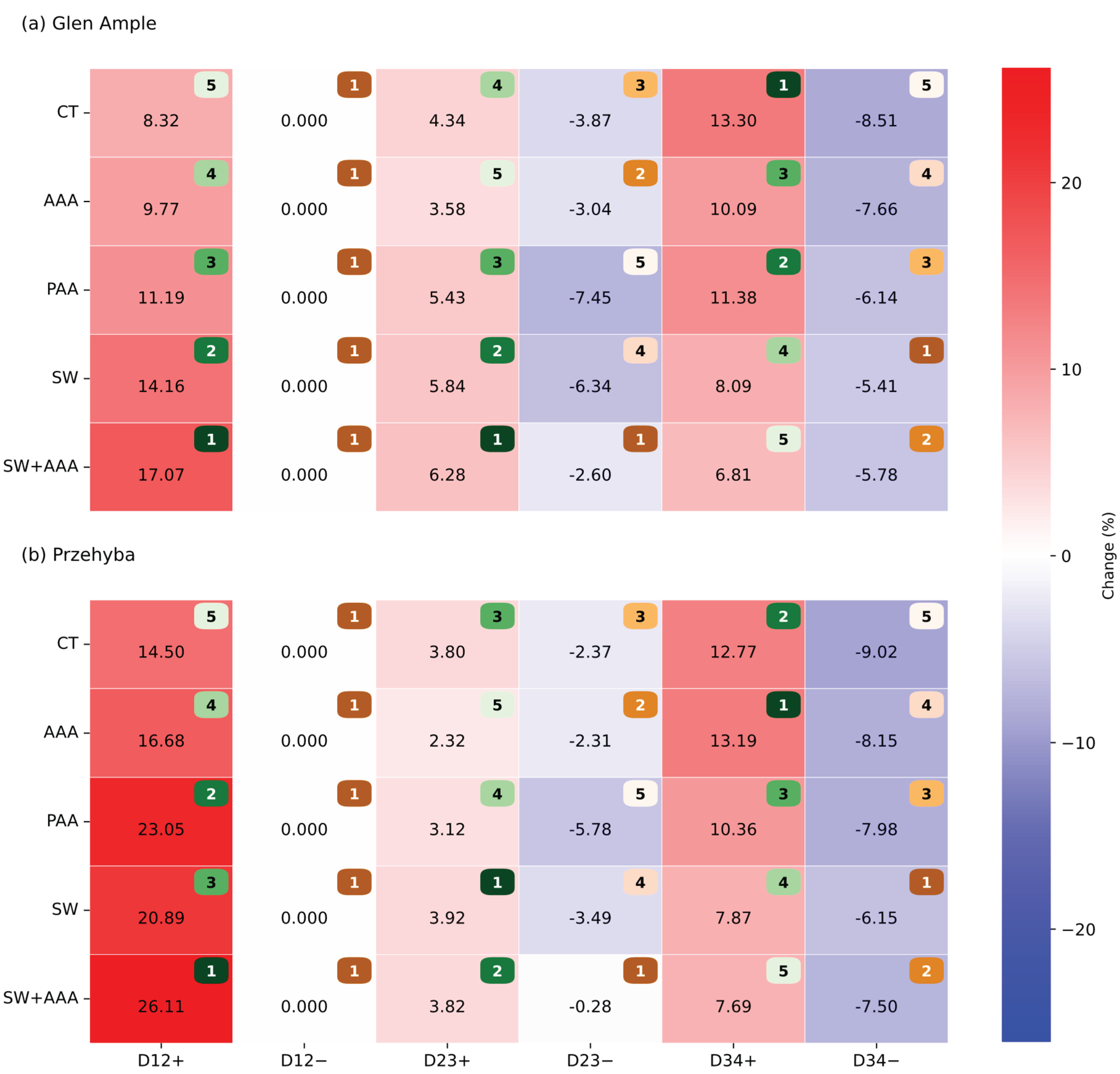
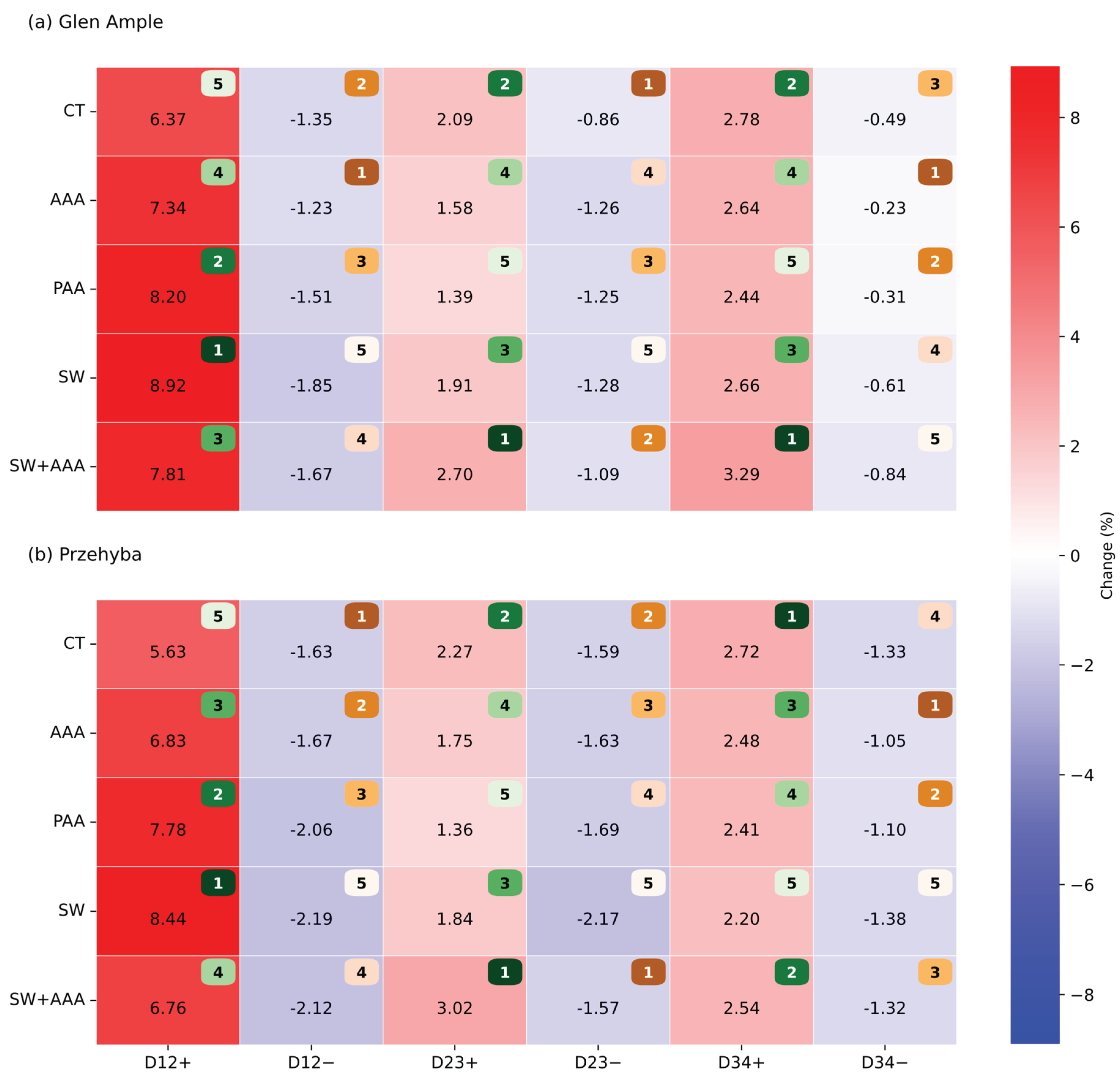
3. Results
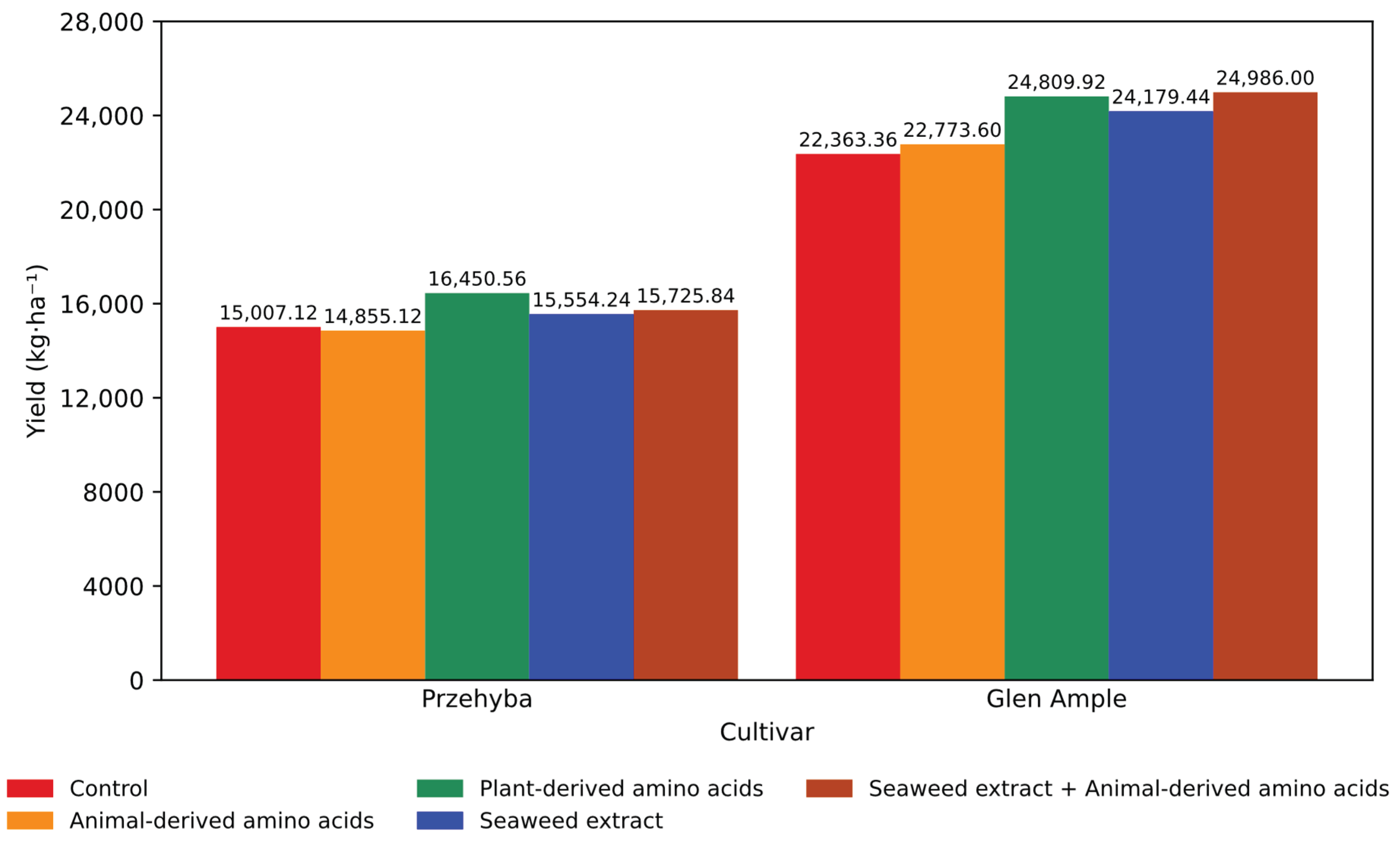
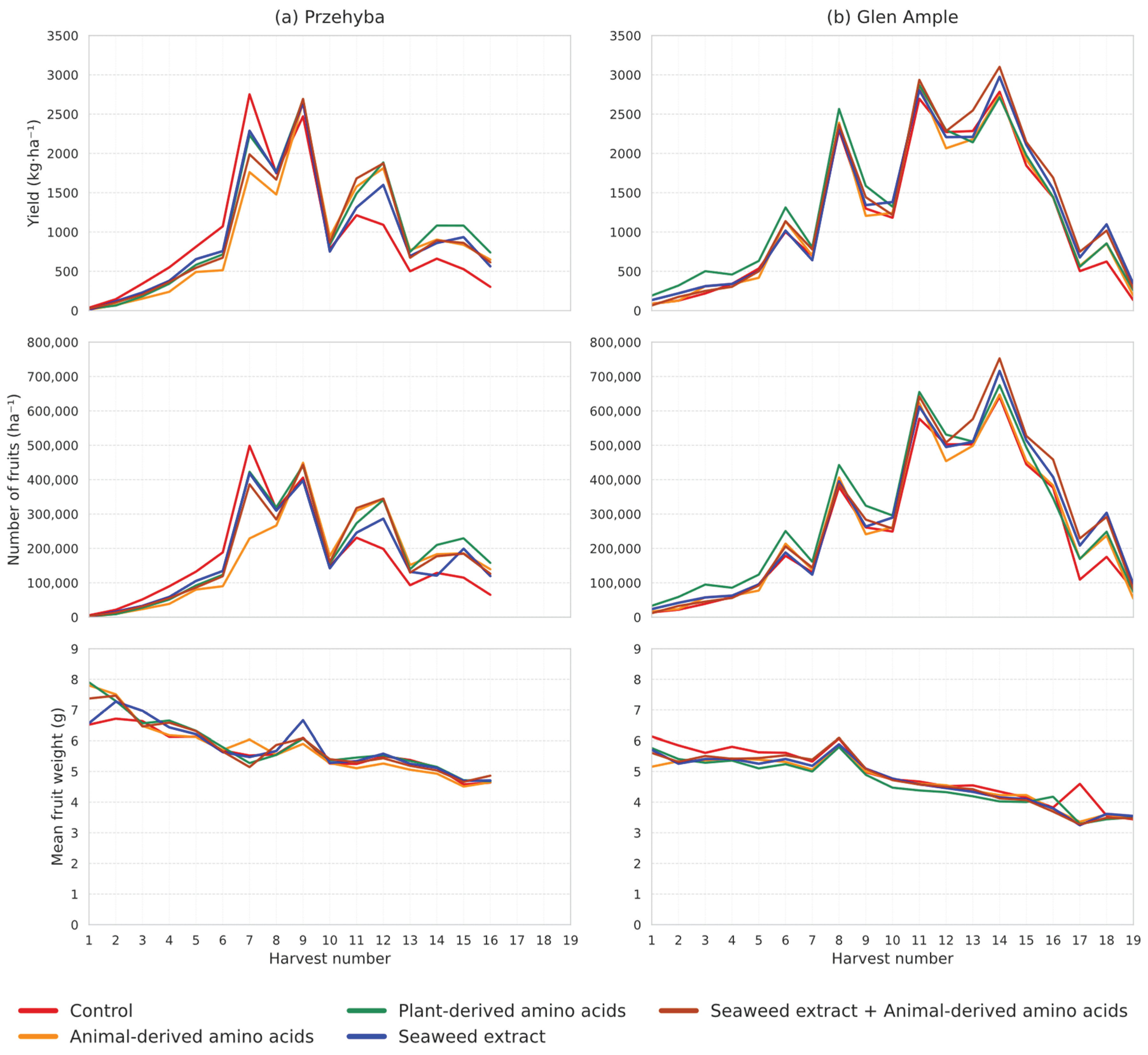
3.1. Yield Parameters
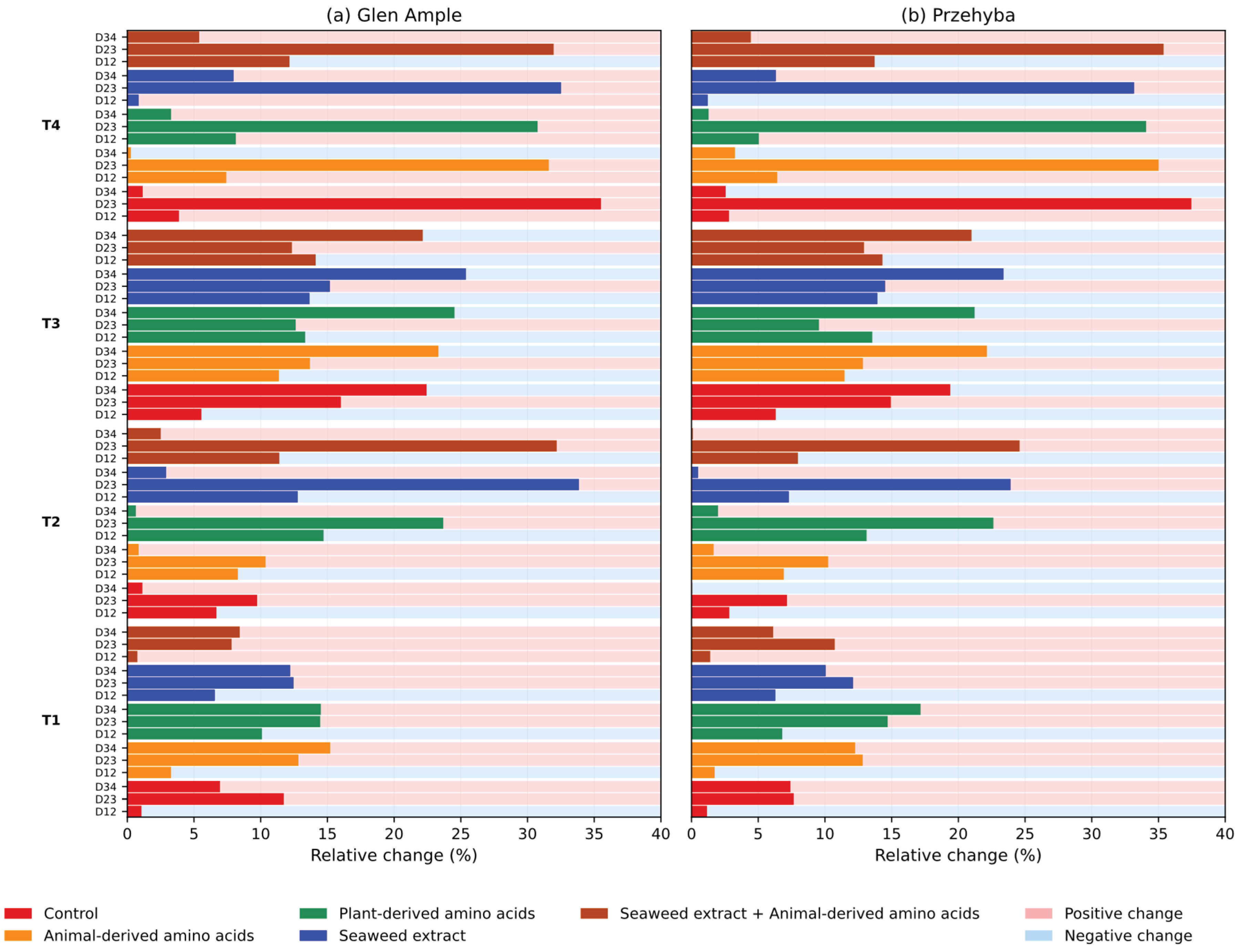
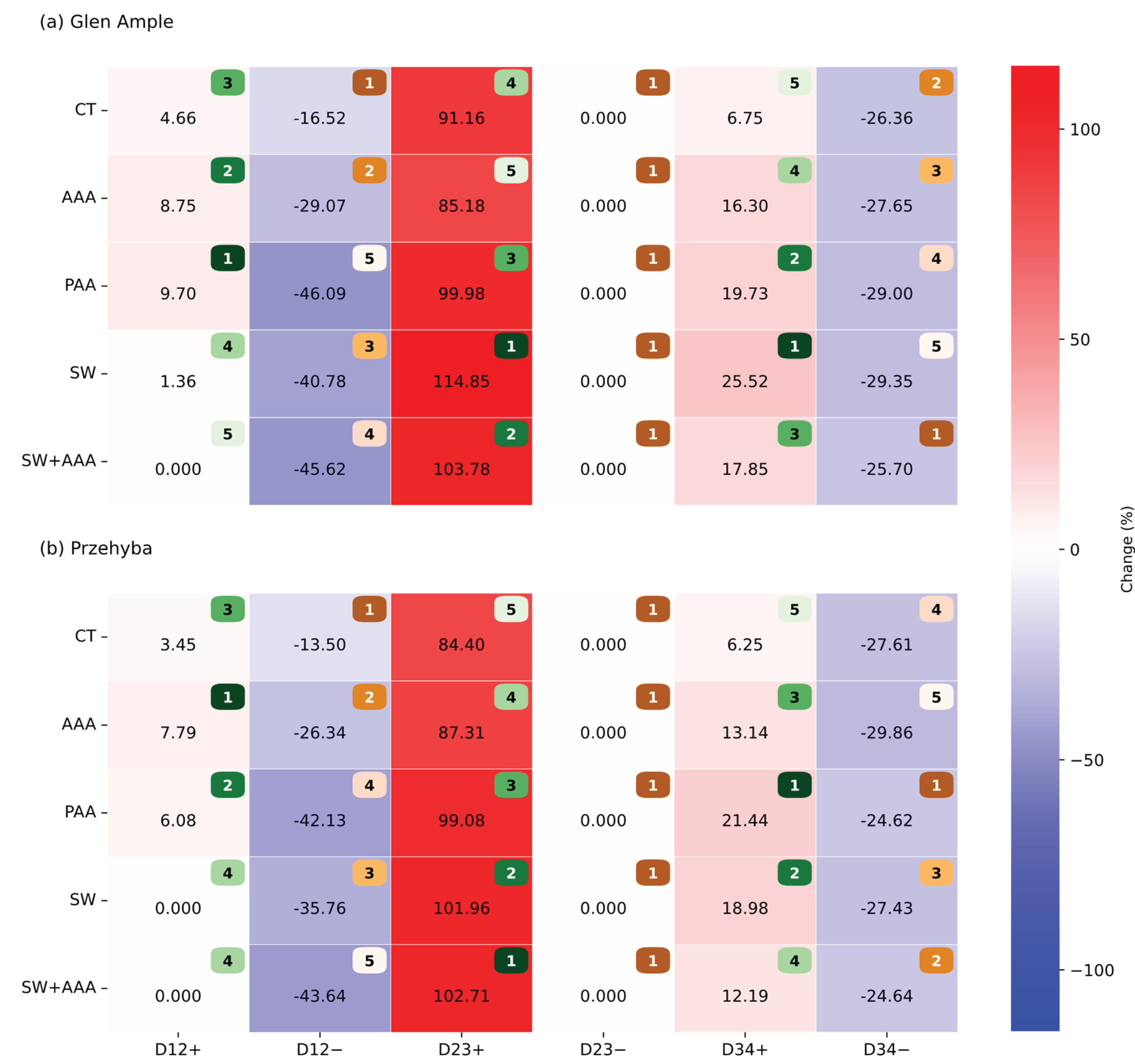
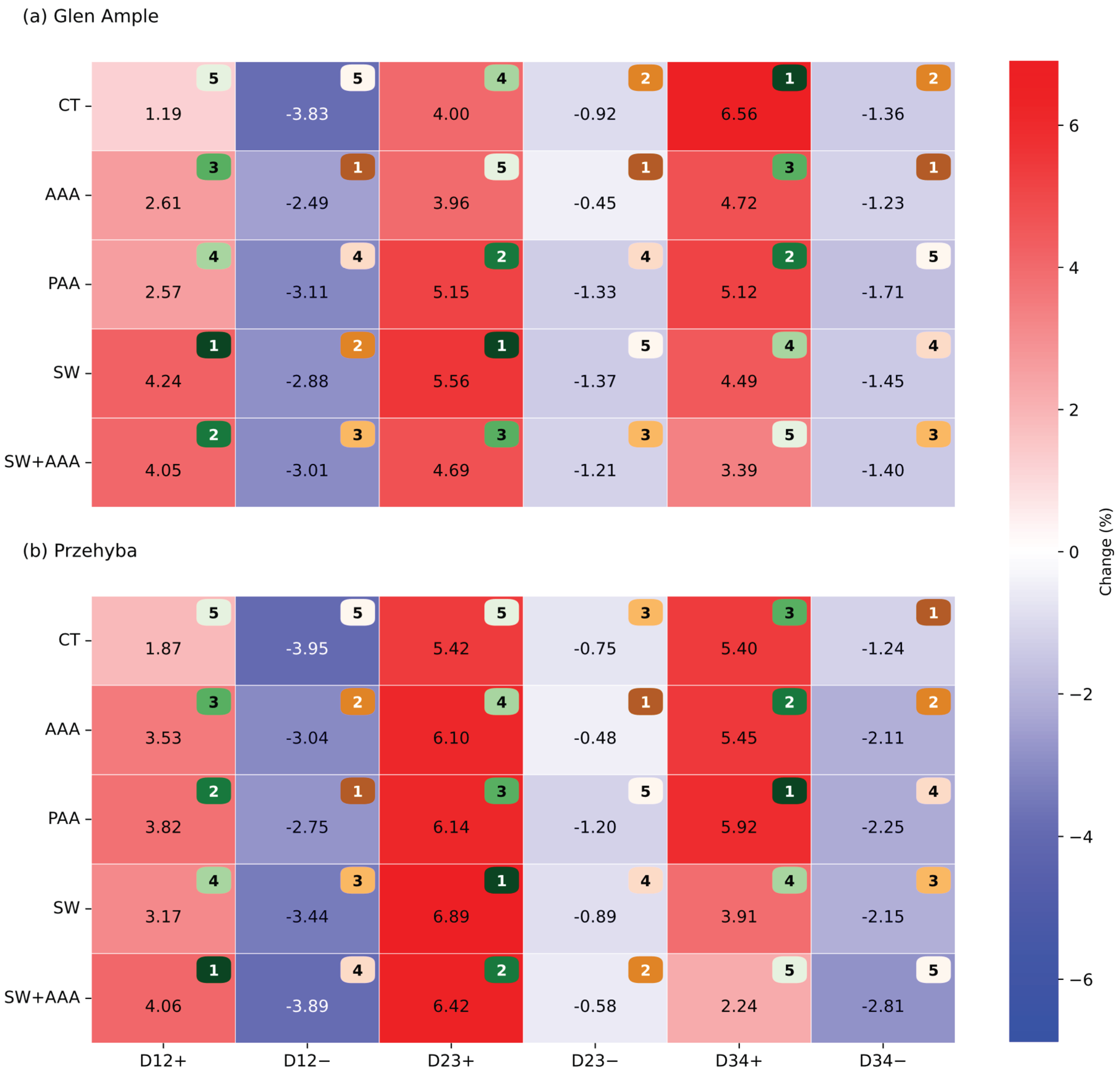
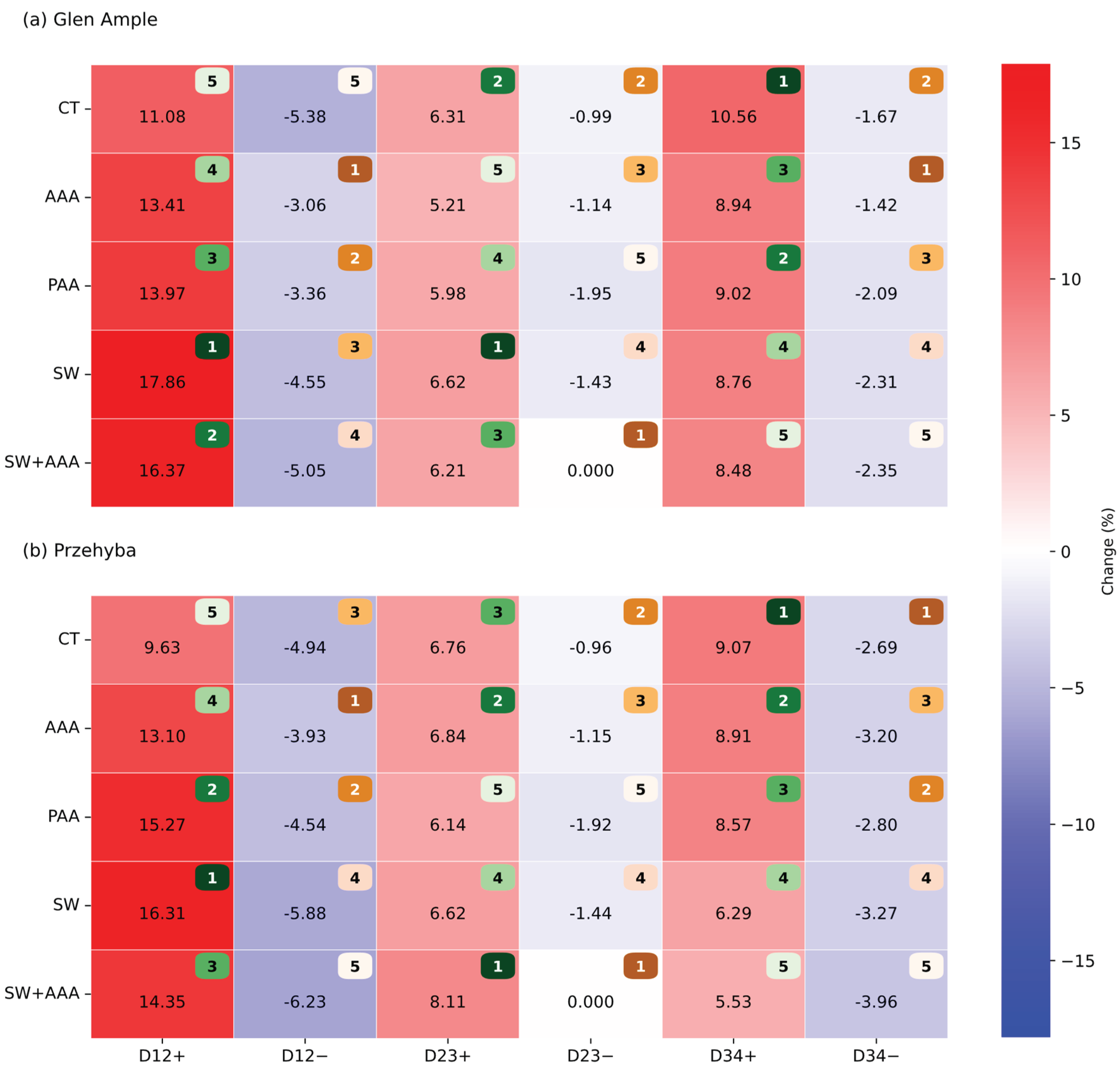
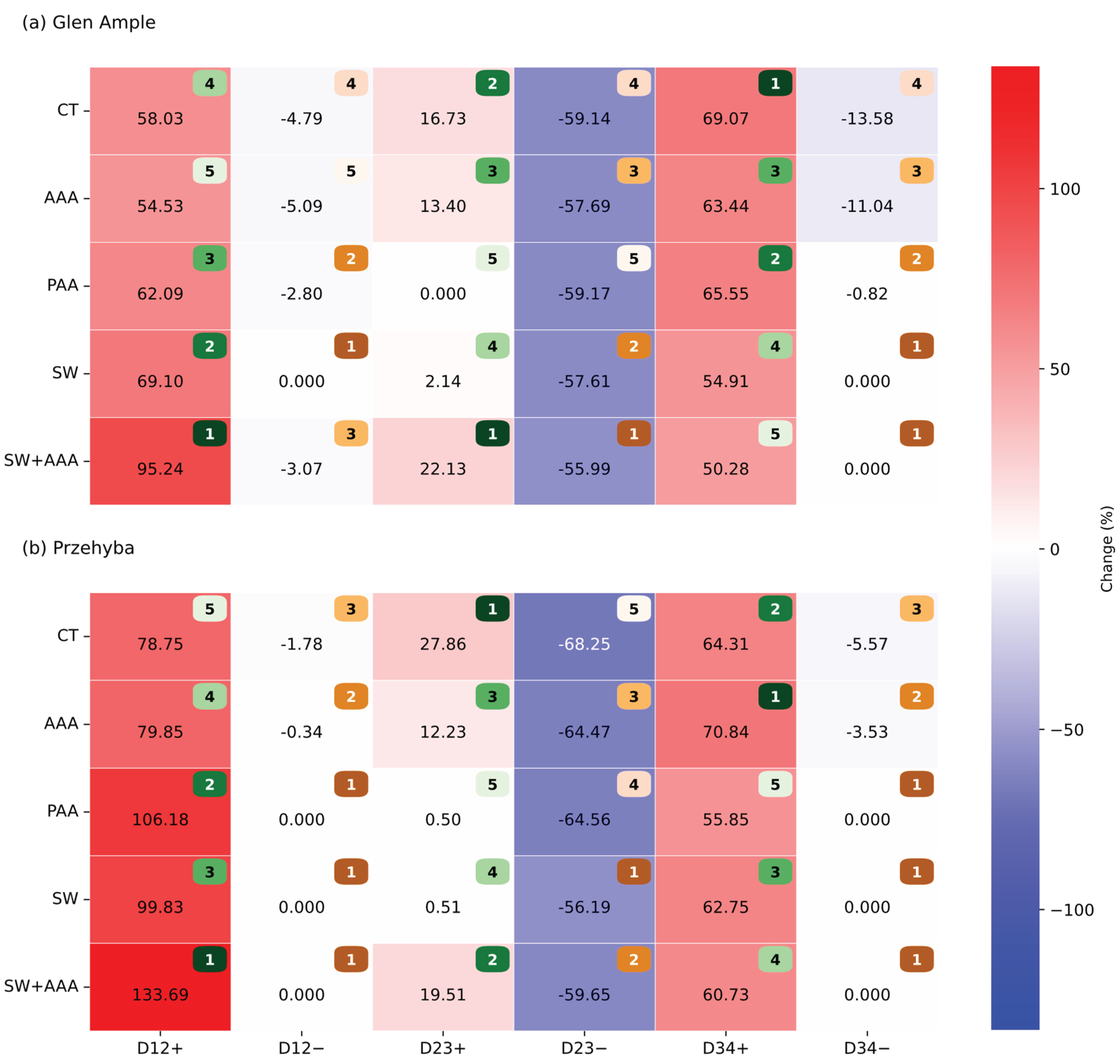
3.1. Vegetation Indices
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chen, B.; Zou, C.; Zhang, Y.; Gou, C.; Li, J. The Current Status, Opportunities, Challenges and Coping Strategies of Sustainable Agriculture. Discov Sustain 2025, 6, 1282. [Google Scholar] [CrossRef]
- Boix-Fayos, C.; De Vente, J. Challenges and Potential Pathways towards Sustainable Agriculture within the European Green Deal. Agricultural Systems 2023, 207, 103634. [Google Scholar] [CrossRef]
- Terán-Samaniego, K.; Robles-Parra, J.M.; Vargas-Arispuro, I.; Martínez-Téllez, M.Á; Garza-Lagler, M.C.; Félix-Gurrlola, D.; Maycotte-de La Peña, M.L.; Tafolla-Arellano, J.C.; García-Figueroa, J.A.; Espinoza-López, P.C. Agroecology and Sustainable Agriculture: Conceptual Challenges and Opportunities—A Systematic Literature Review. Sustainability 2025, 17, 1805. [Google Scholar] [CrossRef]
- Izquierdo, J.; Arriagada, O.; García-Pintos, G.; Ortiz, R.; García-Pintos, M.; García-Pintos, M. Humic Field Biostimulation as a Sustainable Agricultural Practice to Increase Yield of Main Grains: Evidence from on-Farm Trials. Front. Plant Sci. 2025, 16, 1709876. [Google Scholar] [CrossRef]
- Meena, D.C.; Birthal, P.S.; Kumara, T.M.K. Biostimulants for Sustainable Development of Agriculture: A Bibliometric Content Analysis. Discov Agric 2025, 3, 2. [Google Scholar] [CrossRef]
- Ruzzi, M.; Colla, G.; Rouphael, Y. Editorial: Biostimulants in Agriculture II: Towards a Sustainable Future. Front. Plant Sci. 2024, 15, 1427283. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in Plant Science: A Global Perspective. Front. Plant Sci. 2017, 7. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Synergistic Biostimulatory Action: Designing the Next Generation of Plant Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 1655. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Carillo, P.; Garcia-Perez, P.; Cardarelli, M.; Senizza, B.; Miras-Moreno, B.; Colla, G.; Lucini, L. Plant Biostimulants from Seaweeds or Vegetal Proteins Enhance the Salinity Tolerance in Greenhouse Lettuce by Modulating Plant Metabolism in a Distinctive Manner. Scientia Horticulturae 2022, 305, 111368. [Google Scholar] [CrossRef]
- Mironenko, G.A.; Zagorskii, I.A.; Bystrova, N.A.; Kochetkov, K.A. The Effect of a Biostimulant Based on a Protein Hydrolysate of Rainbow Trout (Oncorhynchus Mykiss) on the Growth and Yield of Wheat (Triticum Aestivum L.). Molecules 2022, 27, 6663. [Google Scholar] [CrossRef]
- Delgado, J.A.; Short, N.M.; Roberts, D.P.; Vandenberg, B. Big Data Analysis for Sustainable Agriculture on a Geospatial Cloud Framework. Front. Sustain. Food Syst. 2019, 3, 54. [Google Scholar] [CrossRef]
- Chergui, N.; Kechadi, M.T. Data Analytics for Crop Management: A Big Data View. J Big Data 2022, 9, 123. [Google Scholar] [CrossRef]
- Ahmad, W.; Jamil, M.; Jabbar, B.; Ahmad, F.; Bukhari, S.L.; Jabeen, S. The Potential of Remote Sensing in Modern Agriculture. In Artificial Intelligence and Data Sciences for Precision Agriculture; Fiaz, S., Nadeem, M.A., Baloch, F.S., Chung, Y.S., Eds.; Springer Nature Switzerland: Cham, 2026; pp. 275–297. ISBN 978-3-032-12769-3. [Google Scholar]
- Victor, N.; Maddikunta, P.K.R.; Mary, D.R.K.; Murugan, R.; Chengoden, R.; Gadekallu, T.R.; Rakesh, N.; Zhu, Y.; Paek, J. Remote Sensing for Agriculture in the Era of Industry 5.0—A Survey. IEEE J. Sel. Top. Appl. Earth Observations Remote Sensing 2024, 17, 5920–5945. [Google Scholar] [CrossRef]
- Tsoulias, N.; Zhao, M.; Paraforos, D.S.; Argyropoulos, D. Hyper- and Multi-Spectral Imaging Technologies. In Encyclopedia of Smart Agriculture Technologies; Zhang, Q., Ed.; Springer International Publishing: Cham, 2023; pp. 1–11. ISBN 978-3-030-89123-7. [Google Scholar]
- Nicolis, O.; Gonzalez, C. Wavelet-Based Fractal and Multifractal Analysis for Detecting Mineral Deposits Using Multispectral Images Taken by Drones. In Methods and Applications in Petroleum and Mineral Exploration and Engineering Geology; Elsevier, 2021; pp. 295–307. ISBN 978-0-323-85617-1. [Google Scholar]
- The Future of Imaging Technology; Kheiralipour, K., Ed.; Mechanical engineering theory and applications; Nova Science Publishers: New York, 2024; ISBN 979-8-89113-987-9. [Google Scholar]
- Handoko, R.N.S.; Lin, S.-Y. Integrating Plant Growth Regulators and Biostimulants to Enhance Resilient and Sustainable Raspberry and Blackberry Production. Scientia Horticulturae 2025, 350, 114296. [Google Scholar] [CrossRef]
- Adamczewski, K.; Matysiak, K. Klucz Do Określania Faz Rozwojowych Roślin Jedno- i Dwuliściennych w Skali BBCH; Instytut Ochrony Roślin, 2011; ISBN 978-83-89867-66-7. [Google Scholar]
- DJI Agriculture. Available online: https://ag.dji.com/mavic-3-m (accessed on 20 October 2025).
- Processing Options Pix4Dfields. Available online: https://support.pix4d.com/hc/en-us/articles/360028421272#label7 (accessed on 20 November 2025).
- Kamilczynski. GitHub Repository 2026. Available online: https://github.com/kamilczynski/Foliar-Biostimulation-Primocane-Raspberry-Assessed-Using-UAV-Based-Multispectral-Imaging (accessed on 10 April 2026).
- Buczyński, K.; Kapłan, M.; Jarosz, Z. Impact of Foliar Biostimulant Applications on Primocane Raspberry Assessed by UAV-Based Multispectral Imaging. Agriculture 2026, 16, 835. [Google Scholar] [CrossRef]
- Kamilczynski. GitHub Repository 2026. Available online: https://github.com/kamilczynski/Impact-of-Biostimulation-on-Floricane-Raspberries-Assessed-Using-Drone-Based-Remote-Sensing (accessed on 19 April 2026).
- Kazakov, P.; Alseekh, S.; Ivanova, V.; Gechev, T. Biostimulant-Based Molecular Priming Improves Crop Quality and Enhances Yield of Raspberry and Strawberry Fruits. Metabolites 2024, 14, 594. [Google Scholar] [CrossRef]
- Kapłan, M.; Klimek, K.; Buczyński, K.; Stój, A.; Krupa, T.; Borkowska, A. Evaluation of the Effect of Biostimulation on the Yielding of Golden Delicious Apple Trees. Applied Sciences 2023, 13, 9389. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Jafari, A.; Shirmardi, M. The Effect of Seaweed Foliar Application on Yield and Quality of Apple Cv. ‘Golden Delicious. Scientia Horticulturae 2024, 323, 112529. [Google Scholar] [CrossRef]
- Di-Vaio, C.; Cirillo, A.; Cice, D.; El-Nakhel, C.; Rouphael, Y. Biostimulant Application Improves Yield Parameters and Accentuates Fruit Color of Annurca Apples. Agronomy 2021, 11, 715. [Google Scholar] [CrossRef]
- Rana, V.S.; Lingwal, K.; Sharma, S.; Rana, N.; Pawar, R.; Kumar, V.; Sharma, U. Biostimulatory Effect of Seaweed Extract on the Fruiting and Runner Production of Strawberry. ELSR 2022, 08, 132–141. [Google Scholar] [CrossRef]
- Mattner, S.W.; Villalta, O.N.; McFarlane, D.J.; Islam, M.T.; Arioli, T.; Cahill, D.M. The Biostimulant Effect of an Extract from Durvillaea Potatorum and Ascophyllum Nodosum Is Associated with the Priming of Reactive Oxygen Species in Strawberry in South-Eastern Australia. J Appl Phycol 2023, 35, 1789–1800. [Google Scholar] [CrossRef]
- Zydlik, Piotr; Zydlik, Zofia; Wieczorek, Robert. The Effectiveness of Using a Preparation Containing Amino Acids in the Cultivation of Strawberries under Thermal Stress Conditions. 2021. [Google Scholar] [CrossRef]
- Lenart, A.; Wrona, D.; Klimek, K.; Kapłan, M.; Krupa, T. Assessment of the Impact of Innovative Fertilization Methods Compared to Traditional Fertilization in the Cultivation of Highbush Blueberry. PLoS ONE 2022, 17, e0271383. [Google Scholar] [CrossRef]
- Lopes, T.; Silva, A.P.; Ribeiro, C.; Carvalho, R.; Aires, A.; Vicente, A.A.; Gonçalves, B. Ecklonia Maxima and Glycine–Betaine-Based Biostimulants Improve Blueberry Yield and Quality. Horticulturae 2024, 10, 920. [Google Scholar] [CrossRef]
- Pérez-León, M.I.; González-Fuentes, J.A.; Valdez-Aguilar, L.A.; Benavides-Mendoza, A.; Alvarado-Camarillo, D.; Castillo-Chacón, C.E. Effect of Glutamic Acid and 6-Benzylaminopurine on Flower Bud Biostimulation, Fruit Quality and Antioxidant Activity in Blueberry. Plants 2023, 12, 2363. [Google Scholar] [CrossRef]
- Saleh, Y.M.; Agha, B.S.; Alalam, A.T.S.; Alalaf, A.H.; Adil, A.M.; Al-Ma’athedi, A.F.; Mohamed, M.M.; Abobatta, W.F.; El-hanafy Fekry, W.M.; Mohammed, A.; et al. The Effects of Seaweed Extract and Amino Acid Fertilizers on Growth and Productivity of Two Grape (Vitis Vinifera L.) Cultivars. Sci Rep 2026. [Google Scholar] [CrossRef]
- Arioli, T.; Mattner, S.W.; Hepworth, G.; McClintock, D.; McClinock, R. Effect of Seaweed Extract Application on Wine Grape Yield in Australia. J Appl Phycol 2021, 33, 1883–1891. [Google Scholar] [CrossRef]
- Taskos, D.; Stamatiadis, S.; Yvin, J.-C.; Jamois, F. Effects of an Ascophyllum Nodosum (L.) Le Jol. Extract on Grapevine Yield and Berry Composition of a Merlot Vineyard. Scientia Horticulturae 2019, 250, 27–32. [Google Scholar] [CrossRef]
- Santos, M.; Maia, C.; Meireles, I.; Pereira, S.; Egea-Cortines, M.; Sousa, J.R.; Raimundo, F.; Matos, M.; Gonçalves, B. Effects of Calcium- and Seaweed-Based Biostimulants on Sweet Cherry Profitability and Quality. In Proceedings of the The 3rd International Electronic Conference on Agronomy; MDPI, January 9 2024; p. 45.
- García-Cano, C.; Ferrández-Gómez, B.; Sánchez-Sánchez, A.; Jordá, J.D.; Cerdán, M. Enhancing of Quality, Yield and Aromatic Profile of Sweet Cherries: Comparison between Organic and Conventional Biostimulant Systems. BMC Plant Biol 2025, 25, 869. [Google Scholar] [CrossRef] [PubMed]
- Zhi, H.; Dong, Y. Seaweed-Based Biostimulants Improves Quality Traits, Postharvest Disorders, and Antioxidant Properties of Sweet Cherry Fruit and in Response to Gibberellic Acid Treatment. Scientia Horticulturae 2024, 336, 113454. [Google Scholar] [CrossRef]
- Yan, K.; Gao, S.; Yan, G.; Ma, X.; Chen, X.; Zhu, P.; Li, J.; Gao, S.; Gastellu-Etchegorry, J.-P.; Myneni, R.B.; et al. A Global Systematic Review of the Remote Sensing Vegetation Indices. International Journal of Applied Earth Observation and Geoinformation 2025, 139, 104560. [Google Scholar] [CrossRef]
- Arinaitwe, U.; Yabwalo, D.N.; Hangamaisho, A. Unlocking the Potential of Biostimulants: A Review of Classification, Mode of Action, Formulations, Efficacy, Mechanisms, and Recommendations for Sustainable Intensification. IJPB 2025, 16, 122. [Google Scholar] [CrossRef]
- Khoulati, A.; Ouahhoud, S.; Taibi, M.; Ezrari, S.; Mamri, S.; Merah, O.; Hakkou, A.; Addi, M.; Maleb, A.; Saalaoui, E. Harnessing Biostimulants for Sustainable Agriculture: Innovations, Challenges, and Future Prospects. Discov Agric 2025, 3, 56. [Google Scholar] [CrossRef]





| Cultivar | Country of origin | Breeding program |
| Przehyba | Poland | Sadowniczy Zakład Doświadczalny Brzezna |
| Glen Ample | Scotland | James Hutton Institute |
| Biostimulant Type | Trade Name | Application Dose (l/ha) |
| Animal-derived amino acids | NaturalCrop SL | 1.5 |
| Plant-derived amino acids | Kaishi | 2.0 |
| Seaweed extract | Valkiria Power Alg | 2.0 |
| Seaweed extract + Animal-derived amino acids | Phylgreen Kuma | 3.0 |
| Ortho GSD (cm/pixel) | 1.40 | 2.30 |
| Route altitude (m) | 30.4 | 49.9 |
| Course angle (°) | 111 | 111 |
| Speed (m/s) | 1.3 | 2 |
| Frontal overlap ratio (%) | 90 | 90 |
| Side overlap ratio (%) | 70 | 70 |
| RTK | On | On |
| White balance | Auto | Auto |
| Dewarping | Off | Off |
| Programming language R (4.4.0) | |
| Integrated development environment | Rstudio (2025.9.2.418) |
| Packages | Readxl (1.4.5), Dplyr (1.1.4), Emmeans (2.0.1), Multcomp (1.4.29), MultcompView (0.1.10), ComplexHeatmap (2.22.0), Circlize (0.4.17) |
| Programming language Python (3.12.0) | |
| Integrated development environment | PyCharm (2025.3.2.1) |
| Libraries | Pandas (2.2.3), Seaborn (0.13.2), Matplotlib (3.9.2), NumPy (2.2.0) |
| Parameter | Setting |
| Processing pipeline | Accurate |
| reflectanceTargetUsed | True |
| enableRadiometry | True |
| enableGPU | True |
| orhtoMinGsd | 0 |
| orthoMaxSizeMPixels | 0 |
| enablePanSharpening | False |
| enableOrthoMinGsd | False |
| enableOrthoMaxSizeMPixels | False |
| Treatment | Measurement | Processing settings |
| 1 | 1 | clear sky |
| 2 | overcast | |
| 3 | overcast | |
| 4 | overcast | |
| 2 | 1 | clear sky |
| 2 | clear sky | |
| 3 | clear sky | |
| 4 | overcast | |
| 3 | 1 | clearsky |
| 2 | overcast | |
| 3 | clearsky | |
| 4 | clearsky | |
| 4 | 1 | clear sky |
| 2 | overcast | |
| 3 | clear sky | |
| 4 | clear sky |
| Vegetation index | Formula | |
| Leaf Chlorophyll Index | LCI | |
| Normalized Difference Red Edge | NDRE | |
| Normalized Difference Vegetation Index | NDVI | |
| Green Normalized Difference Vegetation Index | GNDVI | |
| Modified Chlorophyll Absorption in Reflective Index | MCARI | |
| Modified Chlorophyll Absorption in Reflective Index 2 | MCARI2 | |
| Optimized Soil Adjusted Vegetation Index | OSAVI | |
| Structure Intensive Pigment Index 2 |
SIPI2 | |
| Cultivar | Combination | Yield per Plant (g) |
Number of Fruits per Plant (pcs.) |
Fruit Weight (g) |
| Glen Ample | CT | 2,795.42 b | 604.54 b | 4.62 a |
| AAA | 2,846.70 b | 627.53 b | 4.54 a | |
| PAA | 3,101.24 a | 696.82 a | 4.45 a | |
| SW | 3,022.43 ab | 675.64 a | 4.47 a | |
| SW+AAA | 3,123.25 a | 698.00 a | 4.48 a | |
| p-value | 0.004 | < 0.001 | 0.353 | |
| Przehyba | CT | 1,875.89 b | 336.22 b | 5.58 a |
| AAA | 1,856.89 b | 335.05 b | 5.54 a | |
| PAA | 2,056.32 a | 374.79 a | 5.51 a | |
| SW | 1,944.28 ab | 340.18 b | 5.72 a | |
| SW+AAA | 1,965.73 ab | 358.10 ab | 5.49 a | |
| p-value | 0.018 | 0.002 | 0.289 |
| Yield per Plant (g) |
Number of Fruits per Plant (pcs.) |
Fruit Weight (g) | ||
| Cultivar (A) | Przehyba | 1939.82 b | 348.87 b | 5.56 a |
| Glen Ample | 2977.81 a | 660.51 a | 4.51 b | |
| p-value | < 0.001 | < 0.001 | < 0.001 | |
| Combination (B) | CT | 2335.66 b | 470.38 c | 5.10 a |
| AAA | 2351.80 bc | 481.29 c | 5.04 a | |
| PAA | 2578.78 a | 535.80 a | 4.97 a | |
| SW | 2483.36 ac | 507.91 b | 5.09 a | |
| SW+AAA | 2544.49 a | 528.05 ab | 4.98 a | |
| p-value | < 0.001 | < 0.001 | 0.265 | |
| A*B | p-value | 0.162 | < 0.001 | 0.362 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




