Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
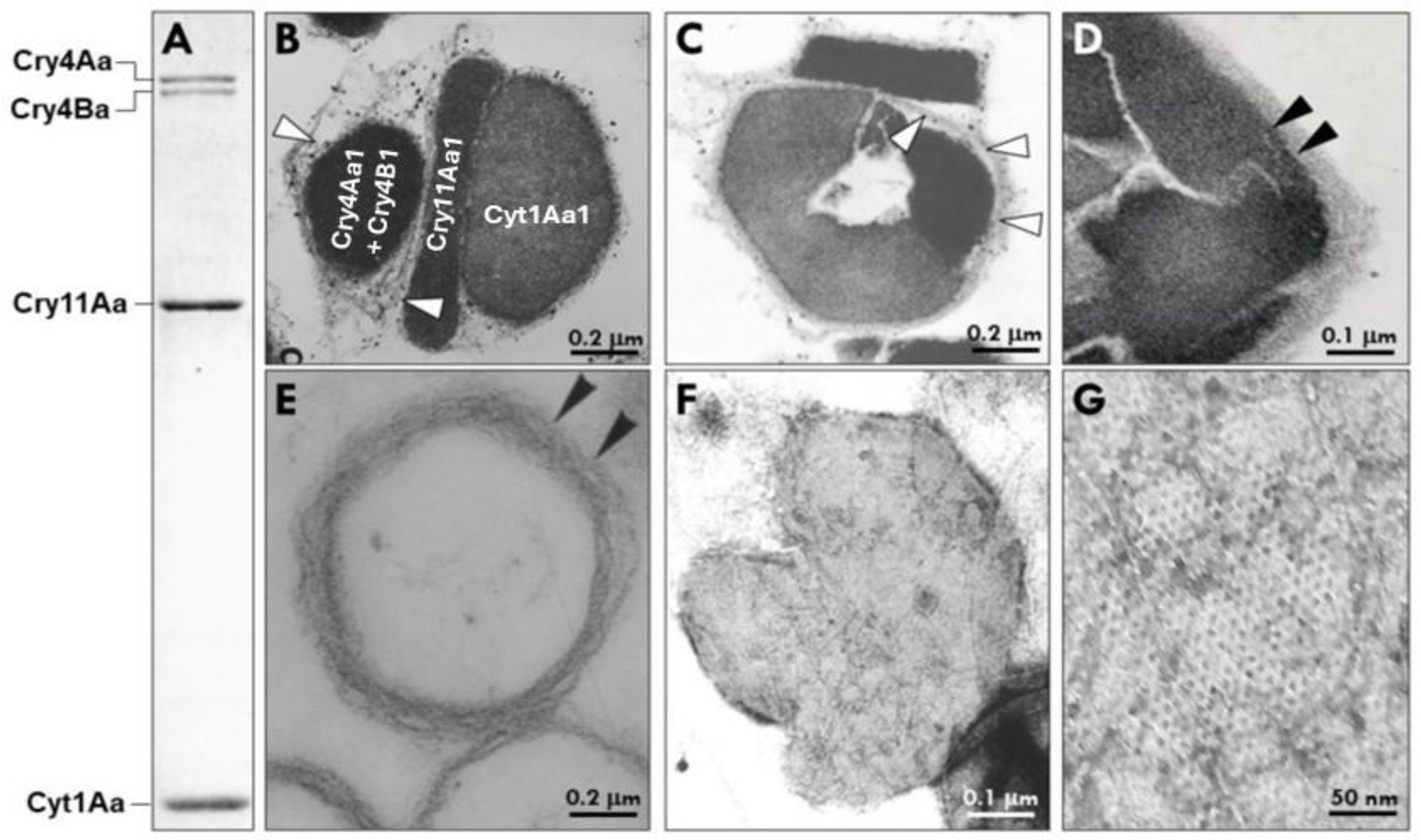
Bacillus thuringiensis subsp. israelensis (Bti) is the most widely used biological larvicide for mosquito control worldwide and a cornerstone of environmentally sustainable vector-management programs. Its long-term global deployment reflects a well-characterized balance between public-health benefit and manageable ecological tradeoffs within integrated vector management (IVM) frameworks. Bti combines high larvicidal efficacy, operational simplicity, and strong target specificity, resulting in an exceptional safety profile for humans and vertebrate wildlife. Decades of laboratory and field studies demonstrate that Bti is biologically selective rather than ecologically inert, with reproducible yet context-dependent effects confined to closely related non-target aquatic dipterans. This review links the molecular and toxicological foundations of Bti to its operational performance, ecological selectivity, resistance-mitigating properties, and sustained utility in mosquito-control programs. Beyond its established larvicidal function, Bti’s prokaryotic insect larvicidal organelle (PILO) represents an underexplored platform for heterologous intracellular protein assembly. Its dense packing, structural stability, and resistance to environmental and biochemical stress indicate an evolutionary specialization for high-capacity protein storage during sporulation. These properties support noncanonical applications in biomolecule storage and stabilization and motivate cautious exploration of environmentally responsive protein release strategies. Although significant mechanistic and translational challenges remain, particularly with respect to cargo trafficking, modularity, and purification, the architectural principles that have enabled effective mosquito control provide a strong foundation for extending PILO-based platforms beyond larvicidal applications.
Keywords:
1. Introduction
2. General Biology of Bti
2.1. General Structural Features of Bti’s Cry and Cyt Toxins
2.2. Mosquito-Larvicidal Activity of Bti’s Cry and Cyt Proteins
3. Ecological Selectivity, Persistence, and Integrated Use of Bti
3.1. Nontarget Sensitivity Boundaries: Aquatic Invertebrates and the Chironomid Effect
3.2. Human and Vertebrate Safety: Mechanistic and Empirical Evidence
3.3. Evaluating Concerns of Broadened Toxicity Due to Lipophilic Cyt1Aa1
3.4. Governance, Long-Term Ecological Tradeoffs, and Sustainable Deployment
4. Translational Robustness of Bti: From Commercial Larvicides to Programmable Protein Platforms
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Vector-borne diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases.
- Ramalho-Ortigao, M.; Gubler, D.J. Human diseases associated with vectors (arthropods in disease transmission). In Hunter’s Tropical Medicine and Emerging Infectious Diseases, 10th ed.; Ryan, E.T., Hill, D.R., Solomon, T., Aronson, N.E., Endy, T.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1063–1069. [Google Scholar] [CrossRef]
- Hiscox, A.; Jones, R.T.; Dennehy, J.; Dyall, W.; Paris, L.; Spencer, F.I.; Keating, F.; Seelig, F.; Narendran, A.; Das, A.; Logan, J.G. An exploration of current and future vector-borne disease threats and opportunities for change. Front. Public Health 2025, 13, 1585412. [Google Scholar] [CrossRef]
- Ryan, S.J.; Carlson, C.J.; Mordecai, E.A.; Johnson, L.R. Global expansion and redistribution of Aedes-borne virus transmission risk with climate change. PLoS Negl. Trop. Dis. 2019, 13, e0007213. [Google Scholar] [CrossRef] [PubMed]
- Mordecai, E.A.; Cohen, J.M.; Evans, M.V.; Gudapati, P.; Johnson, L.R.; Lippi, C.A.; Miazgowicz, K.; Murdock, C.C.; Rohr, J.R.; Ryan, S.J.; et al. Thermal biology of mosquito-borne disease. Ecol. Lett. 2019, 22, 1690–1708. [Google Scholar] [CrossRef]
- Mordecai, E.A.; Ryan, S.J.; Caldwell, J.M.; Shah, M.M.; LaBeaud, A.D. Climate change could shift disease burden from malaria to arboviruses in Africa. The Lancet Planetary Health 2020, 4, e416–e424. [Google Scholar] [CrossRef]
- Thomson, M.C.; Stanberry, L.R. Climate change and vectorborne diseases. N. Engl. J. Med. 2022, 387, 1969–1978. [Google Scholar] [CrossRef]
- de Souza, W.M.; Weaver, S.C. Effects of climate change and human activities on vector-borne diseases. Nat. Rev. Microbiol. 2024, 22, 476–491. [Google Scholar] [CrossRef]
- Taylor, R.; Messenger, L.A.; Abeku, T.A.; Clarke, S.E.; Yadav, R.S.; Lines, J. Invasive Anopheles stephensi in Africa: Insights from Asia. Trends Parasitol. 2024, 40, 731–743. [Google Scholar] [CrossRef]
- World Health Organization. Elimination of human onchocerciasis: Progress report, 2023-2024. Wkly. Epidemiol. Rec. 2024, 99, 577–590. Available online: https://www.who.int/publications/i/item/who-wer-9941-577-590.
- World Health Organization. Onchocerciasis. Available online: https://www.who.int/news-room/fact-sheets/detail/onchocerciasis.
- Al Naggar, Y.; Fahmy, N.M.; Alkhaibari, A.M.; Al-Akeel, R.K.; Alharbi, H.; Mohamed, A.; Eleftherianos, I.; El-Seedi, H.R.; Giesy, J.P.; Alharbi, H.A. Mechanisms and genetic drivers of resistance of insect pests to insecticides and approaches to its control. Toxics 2025, 13, 681. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.; Gallichotte, E.N.; Randall, J.; Glass, A.; Foy, B.D.; Ebel, G.D.; Kading, R.C. Intrinsic factors driving mosquito vector competence and viral evolution: A review. Front. Cell. Infect. Microbiol. 2023, 13, 1330600. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Scott, T.W. Mode of transmission and the evolution of arbovirus virulence in mosquito vectors. Proc. R. Soc. B 2009, 276, 1369–1378. [Google Scholar] [CrossRef]
- Kim, K.-S. Current challenges in the development of vaccines and drugs against emerging vector-borne diseases. Curr. Med. Chem. 2019, 26, 2974–2986. [Google Scholar] [CrossRef]
- Hassan, A.O.; Akinwale, O.P.; Bello, A.A.; Yusuf, M.S. Malaria vaccine: Prospects and challenges. Madonna Univ. J. Med. Health Sci. 2022, 2, 22–40. [Google Scholar]
- Pintado Silva, J.; Fernandez-Sesma, A. Challenges on the development of a dengue vaccine: A comprehensive review of the state of the art. J. Gen. Virol. 2023, 104, 001831. [Google Scholar] [CrossRef]
- Miranda, L.S.; Rudd, S.R.; Mena, O.; Hudspeth, P.E.; Barboza-Corona, J.E.; Park, H.-W.; Bideshi, D.K. The perpetual vector mosquito threat and its eco-friendly nemeses. Biology 2024, 13, 182. [Google Scholar] [CrossRef]
- Guillet, P.; Kurtak, D.C.; Philippon, B.; Meyer, R. Bacillus thuringiensis israelensis H-14: A biocontrol agent for onchocerciasis control in West Africa. In Bacterial Control of Mosquitoes & Black Flies; de Barjac, H., Sutherland, D.J., Eds.; Rutgers University Press: New Brunswick, NJ, USA, 1990; pp. 187–201. [Google Scholar] [CrossRef]
- Mbewe, R.; Pemba, D.F.; Kazembe, L.; Mhango, C.; Chiotha, S. The impact of Bacillus thuringiensis israelensis (Bti) on adult and larval black fly populations. Malawi J. Sci. Technol. 2014, 10, 1–7. [Google Scholar]
- Molloy, D.P. Progress in the biological control of black flies with Bacillus thuringiensis israelensis, with emphasis on temperate climates. In Bacterial Control of Mosquitoes & Black Flies; de Barjac, H., Sutherland, D.J., Eds.; Rutgers University Press: New Brunswick, NJ, USA, 1990; pp. 161–186. [Google Scholar]
- World Health Organization. Bacillus thuringiensis. In Environmental Health Criteria 217; WHO: Geneva, Switzerland, 1999; Available online: https://inchem.org/documents/ehc/ehc/ehc217.htm.
- U.S. Environmental Protection Agency. Reregistration Eligibility Decision (RED) for Bacillus thuringiensis israelensis; EPA 738-R-98-004; Office of Pesticide Programs: Washington, DC, USA, 1998. [Google Scholar]
- Boisvert, M.; Boisvert, J. Effects of Bacillus thuringiensis var. israelensis on target and non-target organisms: A review of laboratory and field experiments. Biocontrol Sci. Technol. 2000, 10, 517–561. [Google Scholar] [CrossRef]
- Lacey, L.A. Bacillus thuringiensis serovariety israelensis and Bacillus sphaericus for mosquito control. J. Am. Mosq. Control Assoc. 2007, 23, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Suliman, N.S.; Talaei-Hassanloui, R.; Abachi, H.; Zarei, S.; Osdaghi, E. Taxonomic refinement of Bacillus thuringiensis. Front. Microbiol. 2025, 16, 1518307. [Google Scholar] [CrossRef] [PubMed]
- Rudd, S.R.; Miranda, L.S.; Curtis, H.R.; Bigot, Y.; Diaz-Mendoza, M.; Hice, R.; Nizet, V.; Park, H.-W.; Blaha, G.; Federici, B.A.; Bideshi, D.K. The parasporal body of Bacillus thuringiensis subsp. israelensis: A unique phage capsid-associated prokaryotic insecticidal organelle. Biology 2023, 12, 1421. [Google Scholar] [CrossRef]
- Berry, C.; O’Neil, S.; Ben-Dov, E.; Jones, A.F.; Murphy, L.; Quail, M.A.; Holden, M.T.; Harris, D.; Zaritsky, A.; Parkhill, J. Complete sequence and organization of pBtoxis, the toxin-coding plasmid of Bacillus thuringiensis subsp. israelensis. Appl. Environ. Microbiol. 2002, 68, 5082–5095. [Google Scholar] [CrossRef]
- Xu, C.; Wang, B.-C.; Yu, Z.; Sun, M. Structural insights into Bacillus thuringiensis Cry, Cyt and Parasporin toxins. Toxins 2014, 6, 2732–2770. [Google Scholar] [CrossRef] [PubMed]
- Tetreau, G.; Banneville, A.S.; Andreeva, E.A.; Brewster, A.S.; Hunter, M.S.; Sierra, R.G.; Teulon, J.M.; Young, I.D.; Burke, N.; Grunewald, T.A.; et al. Serial femtosecond crystallography on in vivo-grown crystals drives elucidation of mosquitocidal Cyt1Aa1 bioactivation cascade. Nat. Commun. 2020, 11, 1153. [Google Scholar] [CrossRef] [PubMed]
- Crickmore, N.; Berry, C.; Panneerselvam, S.; Mishra, R.; Connor, T.R.; Bonning, B.C. A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins. J. Invertebr. Pathol. 2021, 186, 107438. [Google Scholar] [CrossRef] [PubMed]
- Tetreau, G.; Sawaya, M.R.; De Zitter, E.; Andreeva, E.A.; Banneville, A.-S.; Schibrowsky, N.A.; et al. De novo determination of mosquitocidal Cry11Aa and Cry11Ba structures from naturally occurring nanocrystals. Nat. Commun. 2022, 13, 4376. [Google Scholar] [CrossRef]
- Pacheco, S.; Gomez, I.; Pelaez-Aguilar, A.E.; Verduzco-Rosas, L.A.; Garcia-Suarez, R.; do Nascimento, N.A.; Rivera-Najera, L.Y.; Canton, P.E.; Soberon, M.; Bravo, A. Structural changes upon membrane insertion of the insecticidal pore-forming toxins produced by Bacillus thuringiensis. Front. Insect Sci. 2023, 3, 1188891. [Google Scholar] [CrossRef]
- Pigott, C.R.; Ellar, D.J. Role of receptors in Bacillus thuringiensis crystal toxin activity. Microbiol. Mol. Biol. Rev. 2007, 71, 255–281. [Google Scholar] [CrossRef]
- Ben-Dov, E. Bacillus thuringiensis subsp. israelensis and its dipteran-specific toxins. Toxins 2014, 6, 1222–1243. [Google Scholar] [CrossRef]
- Manasherob, R.; Zaritsky, A.; Ben-Dov, E.; Saxena, D.; Barak, Z.; Einav, M. Effect of accessory proteins P19 and P20 on cytolytic activity of Cyt1Aa1 from Bacillus thuringiensis subsp. israelensis in Escherichia coli. Curr. Microbiol. 2001, 43, 355–364. [Google Scholar] [CrossRef]
- Silva-Filha, M.H.; Romao, T.P.; Rezende, T.M.T.; Carvalho, K.S.; Menezes, H.S.G.; do Nascimento, N.A.; Soberon, M.; Bravo, A. Bacterial toxins active against mosquitoes: Mode of action and resistance. Toxins 2021, 13, 523. [Google Scholar] [CrossRef]
- Sharma, M.; Kumar, V. Mosquito-larvicidal binary (BinA/B) proteins for mosquito control programs-Advancements, challenges, and possibilities. Curr. Res. Insect Sci. 2022, 2, 100028. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-W.; Bideshi, D.K.; Federici, B.A. Recombinant strain of Bacillus thuringiensis producing Cyt1A, Cry11B, and the Bacillus sphaericus binary toxin. Appl. Environ. Microbiol. 2003, 69, 1331–1334. [Google Scholar] [CrossRef] [PubMed]
- Wirth, M.C.; Federici, B.A.; Walton, W.E. Cyt1A from Bacillus thuringiensis synergizes activity of Bacillus sphaericus against Aedes aegypti (Diptera: Culicidae). Appl. Environ. Microbiol. 2000, 66, 1093–1097. [Google Scholar] [CrossRef]
- Wirth, M.C.; Walton, W.E.; Federici, B.A. Cyt1A from Bacillus thuringiensis restores toxicity of Bacillus sphaericus against resistant Culex quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 401–407. [Google Scholar] [CrossRef]
- Nascimento, N.A.; Torres-Quintero, M.C.; Molina, S.L.; Pacheco, S.; Romao, T.P.; Pereira-Neves, A.; Soberon, M.; Bravo, A.; Silva-Filha, M.H. Functional Bacillus thuringiensis Cyt1Aa is necessary to synergize Lysinibacillus sphaericus binary toxin (Bin) against Bin-resistant and refractory mosquito species. Appl. Environ. Microbiol. 2020, 86, e02770-19. [Google Scholar] [CrossRef]
- Drobniewski, F.A.; Ellar, D.J. Investigation of the membrane lesion induced in vitro by two mosquitocidal delta-endotoxins of Bacillus thuringiensis. Curr. Microbiol. 1988, 16, 195–199. [Google Scholar] [CrossRef]
- Knowles, B.H.; Blatt, M.R.; Tester, M.; Horsnell, J.M.; Carroll, J.; Menestrina, G.; Ellar, D.J. A cytolytic delta-endotoxin from Bacillus thuringiensis var. israelensis forms cation-selective channels in planar lipid bilayers. FEBS Lett. 1989, 244, 259–262. [Google Scholar] [CrossRef]
- Butko, P. Cytolytic toxin Cyt1A and its mechanism of membrane damage: Data and hypotheses. Appl. Environ. Microbiol. 2003, 69, 2415–2422. [Google Scholar] [CrossRef]
- Onofre, J.; Pacheco, S.; Torres-Quintero, M.C.; Gill, S.S.; Soberon, M.; Bravo, A. The Cyt1Aa toxin from Bacillus thuringiensis inserts into target membranes via different mechanisms in insects, red blood cells, and lipid liposomes. J. Biol. Chem. 2020, 295, 9606–9617. [Google Scholar] [CrossRef] [PubMed]
- Soberon, M.; Lopez-Diaz, J.A.; Bravo, A. Cyt toxins produced by Bacillus thuringiensis: A protein fold conserved in several pathogenic microorganisms. Peptides 2013, 41, 87–93. [Google Scholar] [CrossRef]
- McClintock, J.T.; Schaffer, C.R.; Sjoblad, R.D. A comparative review of the mammalian toxicity of Bacillus thuringiensis-based pesticides. Pestic. Sci. 1995, 45, 95–105. [Google Scholar] [CrossRef]
- European Food Safety Authority. Conclusion on the peer review of the pesticide risk assessment of the active substance Bacillus thuringiensis subsp. israelensis. EFSA J. 2012, 10, 2958. [Google Scholar] [CrossRef]
- Poulin, B.; Lefebvre, G.; Paz, L. Red flag for green spray: Adverse trophic effects of Bacillus thuringiensis israelensis on breeding birds. J. Appl. Ecol. 2010, 47, 884–889. [Google Scholar] [CrossRef]
- Land, M.; Bundschuh, M.; Hopkins, R.J.; Poulin, B.; McKie, B.G. Effects of mosquito control using Bacillus thuringiensis israelensis on aquatic and terrestrial ecosystems: A systematic review. Environ. Evid. 2023, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Baggs, R.D.; Stewart, J.P. Susceptibility of some Florida chironomids and mosquitoes to various formulations of Bacillus thuringiensis serovar israelensis. J. Econ. Entomol. 1981, 74, 672–677. [Google Scholar] [CrossRef]
- Lagadic, L.; Roucaute, M.; Caquet, T. Bti sprays do not adversely affect npn-target aquatic intervetebrates in French Atlantic costal wetlands. J. Appl. Ecol. 2014, 51, 102–113. [Google Scholar] [CrossRef]
- Schöndorfer, A.; Kolbenschlag, S.; Bauspiess, P.; Schirmel, J.; Bollinger, E.; Manfrin, A.; Bundschuh, M. Long-term consequences of Bacillus thuringiensis israelensis applications on aquatic insect emergence: Insights from a 4-year mesocosm study. Sci. Total Environ. 2025, 296, 118208. [Google Scholar] [CrossRef]
- Payne, J.; Bellmer, D.; Jadeja, R.; Muriana, P. The Potential of Bacillus Species as Probiotics in the Food Industry: A Review. Foods 2024, 13, 2444. [Google Scholar] [CrossRef]
- Williams, N.; Weir, T.L. Spore-Based Probiotic Bacillus subtilis: Current Applications in Humans and Future Perspectives. Fermentation 2024, 10, 78. [Google Scholar] [CrossRef]
- Nicolas, G.M. Secondary Metabolites from Bacillus spp. Probiotics as Potential Treatments for Multidrug-Resistant Pathogens: A Comprehensive Review. Curr. Res. Microb. Sci. 2025, 8, 100392. [Google Scholar] [CrossRef]
- Feltrin, P.F.; Roll, A.A.P.; Von Mühlen, C.; Gatti, N.L.S.; Leite, F.P.L.L.; Corcini, C.D.; Varela Júnior, A.S.; Nunes, A.P.; Xavier, E.G.; Roll, V.F.B. An Assessment of Bacillus thuringiensis var. israelensis Effects on Hematology, Intestinal Morphology, and Testicular Growth in Chickens. Rev. Bras. Saúde Prod. Anim. 2025, 26, e022. Available online: https://www.scielo.br/j/rbspa/a/bpSNNCrq4my6svwhZvrgqnP/?format=pdf&lang=en. [CrossRef]
- Covich, A.P.; Palmer, M.A.; Crowl, T.A. The role of benthic invertebrate species in freshwater ecosystems. BioScience 1999, 49, 119–127. [Google Scholar] [CrossRef]
- Hanowski, J.; Niemi, G.; Lima, A.; Regal, R. Response of breeding bird populations to mosquito control treatments of wetlands. Wetlands 1997, 17, 485–492. [Google Scholar] [CrossRef]
- Kalniņš, G.; Bertins, M.; Vīksna, A.; Tars, K. Functionalization of bacterial microcompartment shell interior with cysteine-containing peptides enhances iron and cobalt loading capacity. BioMeta.
- van Nieuwpoort, J.C.; Schrama, M.; Spitzen, J.; Boerlijst, S.P. Beyond the target insects: Impacts of Bacillus thuringiensis israelensis on aquatic macrofauna communities. Parasites Vectors 2025, 18, 907. [Google Scholar] [CrossRef]
- Levin, D.B. Human Health Effects Resulting from Exposure to Bacillus thuringiensis Applied during Insect Control Programmes. In Use of Microbes for Control and Eradication of Invasive Arthropods; Progress in Biological Control; Springer: Dordrecht, The Netherlands, 2009; pp. 291–303. [Google Scholar] [CrossRef]
- Minnesota Department of Health. Health risk information about Bacillus thuringiensis (Bt). 2025. Available online: https://www.health.state.mn.us/communities/environment/pesticide/bt.html.
- Tayabali, A.F.; Seligy, V.L. Human cell exposure assays of Bacillus thuringiensis commercial insecticides: Production of Bacillus cereuslike cytolytic effects from outgrowth of spores. Environ. Health Perspect. 2000, 108, 919–930. [Google Scholar] [CrossRef]
- Hou, Y.; Li, J.; Wu, Y. Modulation of oral vaccine efficacy by the gut microbiota. npj Vaccines 2025, 10, 179. [Google Scholar] [CrossRef] [PubMed]
- Toussaint, S.; Pompermaier, A.; Chagas, F.B.; Tamagno, W.A.; Freeman, J.L.; Hartmann, P.A.; Hartmann, M. Environmentally relevant concentrations of a Bacillus thuringiensis-based biopesticide impair zebrafish behavior. Aquat. Ecol. 2025, 59, 1549–1556. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (EPA). Bti for mosquito control: Human health assessment. 2025. Available online: https://www.epa.gov/mosquitocontrol/bti-mosquito-control (accessed on 13 April 2026).
- Perez, C.; Fernandez, L.E.; Sun, J.; Folch, J.L.; Gill, S.S.; Soberon, M.; Bravo, A. Bacillus thuringiensis subsp. israelensis Cyt1Aa synergizes Cry11Aa toxin by functioning as a membrane-bound receptor. Proc. Natl. Acad. Sci. USA 2005, 102, 18303–18308. [Google Scholar] [CrossRef] [PubMed]
- Wirth, M.C.; Park, H.-W.; Walton, W.E.; Federici, B.A. Cyt1A of Bacillus thuringiensis delays evolution of resistance to Cry11A in the mosquito Culex quinquefasciatus. Appl. Environ. Microbiol. 2005, 71, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Crickmore, N.; Bone, E.J.; Williams, J.A.; Ellar, D.J. Contribution of the individual components of the delta-endotoxin crystal to the mosquitocidal activity of Bacillus thuringiensis subsp. israelensis. FEMS Microbiol. Lett. 1995, 131, 249–254. [Google Scholar] [CrossRef]
- Wirth, M.C.; Georghiou, G.P.; Federici, B.A. CytA enables CryIV endotoxins of Bacillus thuringiensis to overcome high levels of CryIV resistance in the mosquito Culex quinquefasciatus. Proc. Natl. Acad. Sci. USA 1997, 94, 10536–10540. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Johnson, J.J.; Federici, B.A. Synergism of mosquitocidal toxicity between CytA and CryIVD proteins using inclusions produced from cloned genes of Bacillus thuringiensis. Mol. Microbiol. 1994, 13, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, J.; Ehlers, R.U.; Martinez-Ramirez, A.C.; Real, M.D. Binding of Cyt1Aa and Cry11Aa toxins of Bacillus thuringiensis serovar israelensis to brush border membrane vesicles of Tipula paludosa (Diptera: Nematocera) and subsequent pore formation. Appl. Environ. Microbiol. 2007, 73, 3623–3629. [Google Scholar] [CrossRef]
- Perez, C.; Munoz-Garay, C.; Portugal, L.C.; Sanchez, J.; Gill, S.S.; Soberon, M.; Bravo, A. Bacillus thuringiensis subsp. israelensis Cyt1Aa enhances activity of Cry11Aa toxin by facilitating the formation of a pre-pore oligomeric structure. Cell. Microbiol. 2007, 9, 2931–2937. [Google Scholar] [CrossRef]
- Fernandez-Luna, M.T.; Tabashnik, B.E.; Lanz-Mendoza, H.; Bravo, A.; Soberon, M.; Miranda-Rios, J. Single-concentration tests show synergism among Bacillus thuringiensis subsp. israelensis toxins against the malaria vector mosquito Anopheles albimanus. J. Invertebr. Pathol. 2010, 104, 231–233. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Vector Control Response 2017-2030: Progress and Challenges; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- Liu, Y.; Xu, Q. Balancing public health benefits and ecological risks of Bt-based mosquito control. Bt Res. 2025, 16, 20–28. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for malaria vector control and integrated vector management; WHO: Geneva, Switzerland, 2023. [Google Scholar]
- European Parliament and Council of the European Union. Regulation (EU) No 528/2012 concerning the making available on the market and use of biocidal products. Official Journal of the European Union. 2012. Available online: https://www.legislation.gov.uk/eur/2012/528.
- European Union. Official Journal of the European Union, L 2025/2299. 2025. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=OJ:L_202502299 (accessed on April 2026).
- Tilquin, M.; Paris, M.; Reynaud, S.; Després, L.; Ravanel, P.; Geremia, R.A.; Gury, J. Long-lasting persistence of Bacillus thuringiensis subsp. israelensis in mosquito natural habitats. PLoS ONE 2008, 3, e3432. [Google Scholar] [CrossRef]
- Young, J.O.; Bowden, J.J.; Moise, E.R.D.; Scott, R.; Schmidt, B.C.; Warren, J. Effects of Bacillus thuringiensis subsp. kurstaki on NonTarget Macromoth Communities Following Repeated Applications. Forests 2025, 16, 1666. [Google Scholar] [CrossRef]
- Belousova, M.E.; Malovichko, Y.V.; Shikov, A.E.; Nizhnikov, A.A.; Antonets, K.S. Dissecting the Environmental Consequences of Bacillus thuringiensis Application for Natural Ecosystems. Toxins 2021, 13, 355. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for malaria vector control and integrated vector management; WHO: Geneva, Switzerland, 2023. [Google Scholar]
- Groupe de Démoustication du Golfe du Morbihan (GDG). Scientific, regulatory and technical note on the uses of Bacillus thuringiensis israelensis (Bti). 2025. Available online: https://www.gdg.ca/wpcontent/uploads/2025/07/BTI_MISEAJOUR_EN_27JUIN2025.pdf (accessed on April 2026).
- Wu, Z.Q. Using Bacillus thuringiensis var. israelensis to control mosquito larvae in aquaculture: An ecological control strategy. J. Mosq. Res. 2024, 14, 67–75. [Google Scholar] [CrossRef]
- Schnepf, E.; Crickmore, N.; Van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Peng, Q.; Song, F.; Lereclus, D. Regulation of cry gene expression in Bacillus thuringiensis. Toxins 2014, 6, 2194–2209. [Google Scholar] [CrossRef] [PubMed]
- Park, H.W.; Bideshi, D.K.; Wirth, M.C.; Johnson, J.J.; Walton, W.E.; Federici, B.A. Recombinant larvicidal bacteria with markedly improved efficacy against culex vectors of west nile virus. Am. J. Trop. Med. Hyg. 2005, 72, 732–738. [Google Scholar] [CrossRef]
- Juárez-Hernández, E.O.; Casados-Vázquez, L.E.; del Rincón-Castro, M.C.; Salcedo-Hernández, R.; Bideshi, D.K.; Barboza-Corona, J.E. Bacillus thuringiensis subsp. israelensis producing endochitinase ChiA74Δsp inclusions and its improved activity against Aedes aegypti. J. Appl. Microbiol. 2016, 120, 389–398. [Google Scholar] [CrossRef]
- Diaz-Mendoza, M.; Bideshi, D.K.; Federici, B.A. A 54-kilodalton protein encoded by pBtoxis is required for parasporal body structural integrity in Bacillus thuringiensis subsp. israelensis. J. Bacteriol. 2012, 194, 1562–1571. [Google Scholar] [CrossRef]
- Doron, L.; Kerfeld, C.A. Bacterial microcompartments as a next-generation metabolic engineering tool: Utilizing nature’s solution for confining challenging catabolic pathways. Biochem. Soc. Trans. 2024, 52, 997–1010. [Google Scholar] [CrossRef]
- Rose, S.M.; Radhakrishnan, A.; Sinha, S. Innate and engineered attributes of bacterial microcompartments for applications in biomaterials science. J. Mater. Chem. B 2023, 11, 4842–4854. [Google Scholar] [CrossRef]
- McDowell, H.B.; Hoiczyk, E. Bacterial nanocompartments: Structures, functions, and applications. J. Bacteriol. 2022, 204, e0034621. [Google Scholar] [CrossRef]
- Quinton, A.R.; McDowell, H.B.; Hoiczyk, E. Encapsulins: Nanotechnology’s future in a shell. Adv. Appl. Microbiol. 2023, 125, 1–48. [Google Scholar] [CrossRef]
- Kerfeld, C.A.; Aussignargues, C.; Zarzycki, J.; Cai, F.; Sutter, M. Bacterial microcompartments. Nat. Rev. Microbiol. 2018, 16, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.A.; Giessen, T.W. Advances in encapsulin nanocompartment biology and engineering. Biotechnol. Bioeng. 2020, 118, 491–505. [Google Scholar] [CrossRef] [PubMed]
- Chmelyuk, N.S.; Oda, V.V.; Gabashvili, A.N.; et al. Encapsulins: Structure, properties, and biotechnological applications. Biochemistry (Moscow) 2023, 88, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.; Giessen, T.W. Engineering encapsulin nanocages for drug delivery. Mater. Adv. 2025, 6, 10412–10430. [Google Scholar] [CrossRef]
- WHO Cold chain and logistic management. Available online: https://cdn.who.int/media/docs/default-source/searo/india/publications/immunization-handbook-107-198-part2.pdf.
- Zhang, Z.; Hong, W.; Zhang, Y.; Li, X.; Que, H.; Wei, X. Mucosal immunity and vaccination strategies: Current insights and future perspectives. Mol. Biomed. 2025, 6, 57. [Google Scholar] [CrossRef]
- Sinha, D.; Gopalakrishna, P.K.; Paul, S.; Longet, S. A user’s guide to designing efficient and safe mucosal vaccines: Challenges and potentials. Oxford Open Immunol. 2025, 6, iqaf007. [Google Scholar] [CrossRef]
- Seo, H.; Duan, Q.; Zhang, W. Vaccines against gastroenteritis: Current progress and challenges. Gut Microbes 2020, 11, 1486–1517. [Google Scholar] [CrossRef]
- Eshaghi, B.; Schudel, A.; Sadeghi, I.; Chen, Z.; Lee, A.H.; Kanelli, M.; Tierney, F.; Han, J.; Ingalls, B.; Francis, D.M.; Li, G.; von Andrian, U.; Langer, R.; Jaklenec, A. The role of engineered materials in mucosal vaccination strategies. Nat. Rev. Mater. 2024, 9, 29–45. [Google Scholar] [CrossRef]
- Senthilkumar, M.; Amaresan, N.; Sankaranarayanan, A. Purification and Characterization of Crystal Proteins. In Plant–Microbe Interactions; Springer Protocols Handbooks; Humana: New York, NY, USA, 2021; pp. 239–241. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



